Submitted:
13 August 2024
Posted:
13 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Animals and Treatments
2.3. Western Blot Detection of TREM2
2.4. Immunohistochemistry of Brain Sections
2.5. Study Population
2.6. Separation and Treatment of Monocyte-Derived Macrophages (MDM)
2.7. Real-Time Quantitative Polymerase Chain Reaction (qPCR) and RNA-Sequencing
2.8. Enzyme-Linked Immunosorbent Assay (ELISA)
2.9. Immunocytochemistry for TREM2
2.10. Statistical Analysis
3. Results
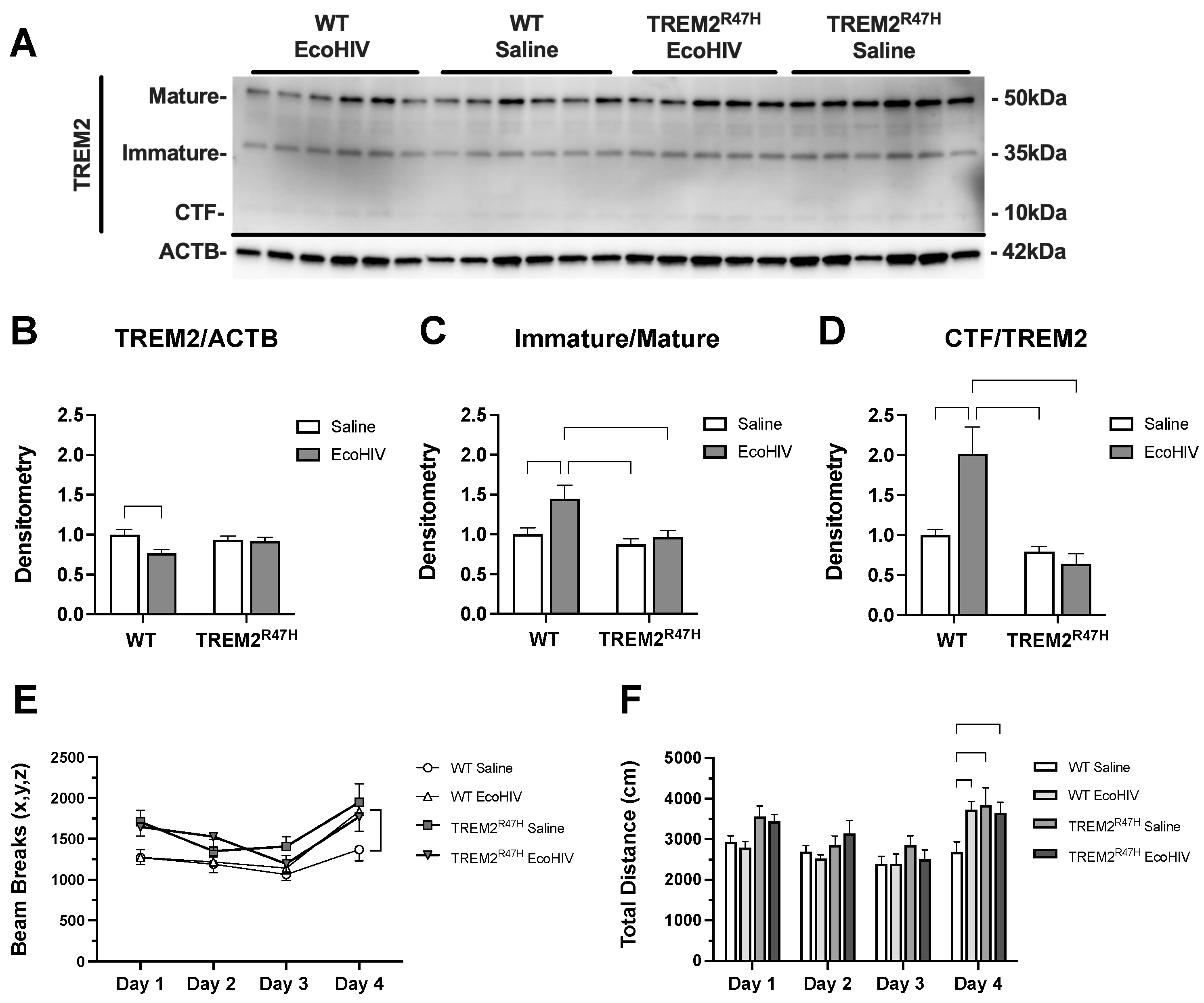
3.1. EcoHIV Reduces Levels of TREM2 and Alters Memory in Wildtype and TREM2R47H Mice
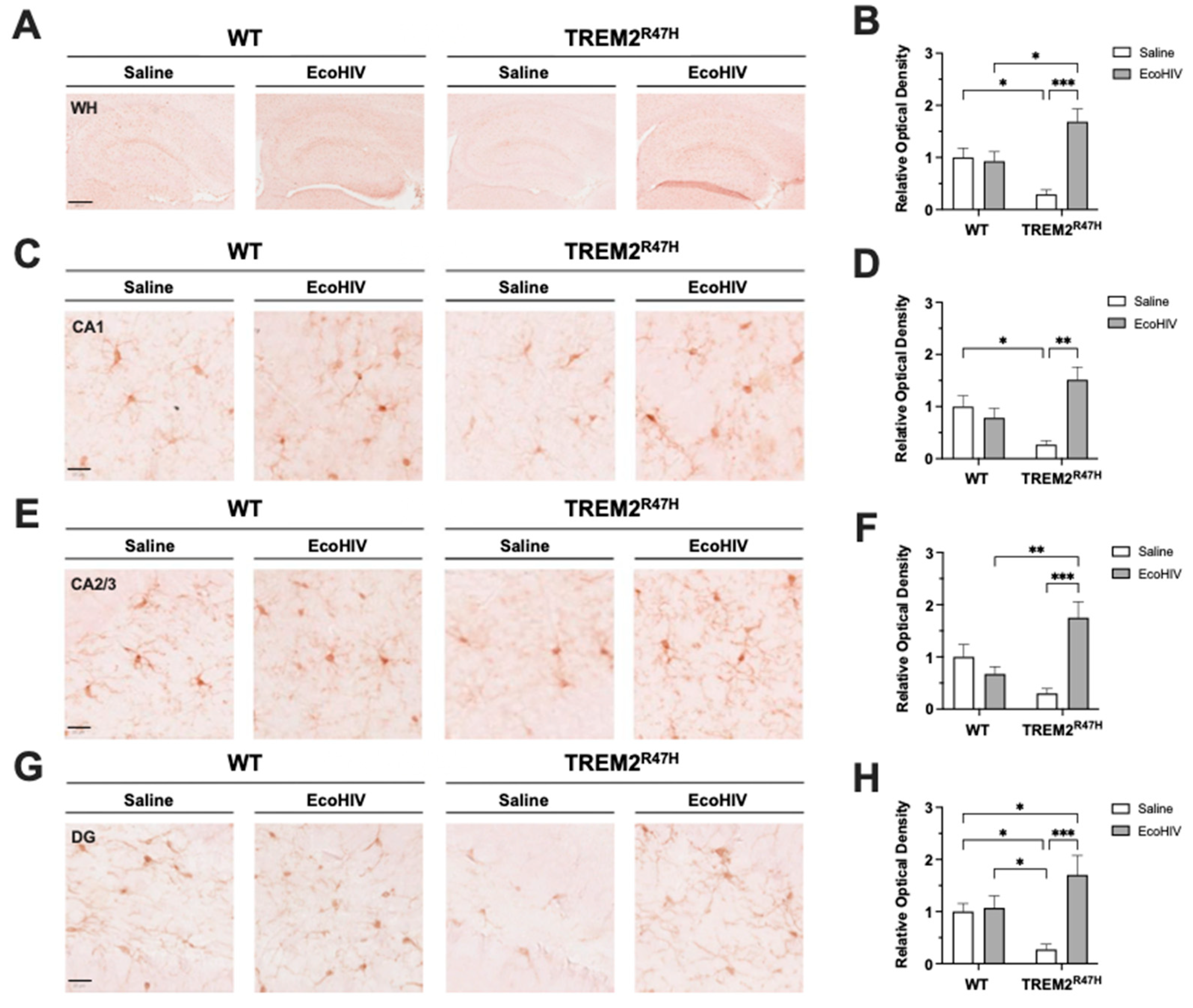
3.2. EcoHIV Increases Hippocampal IBA1 Expression in TREM2R47H Mice
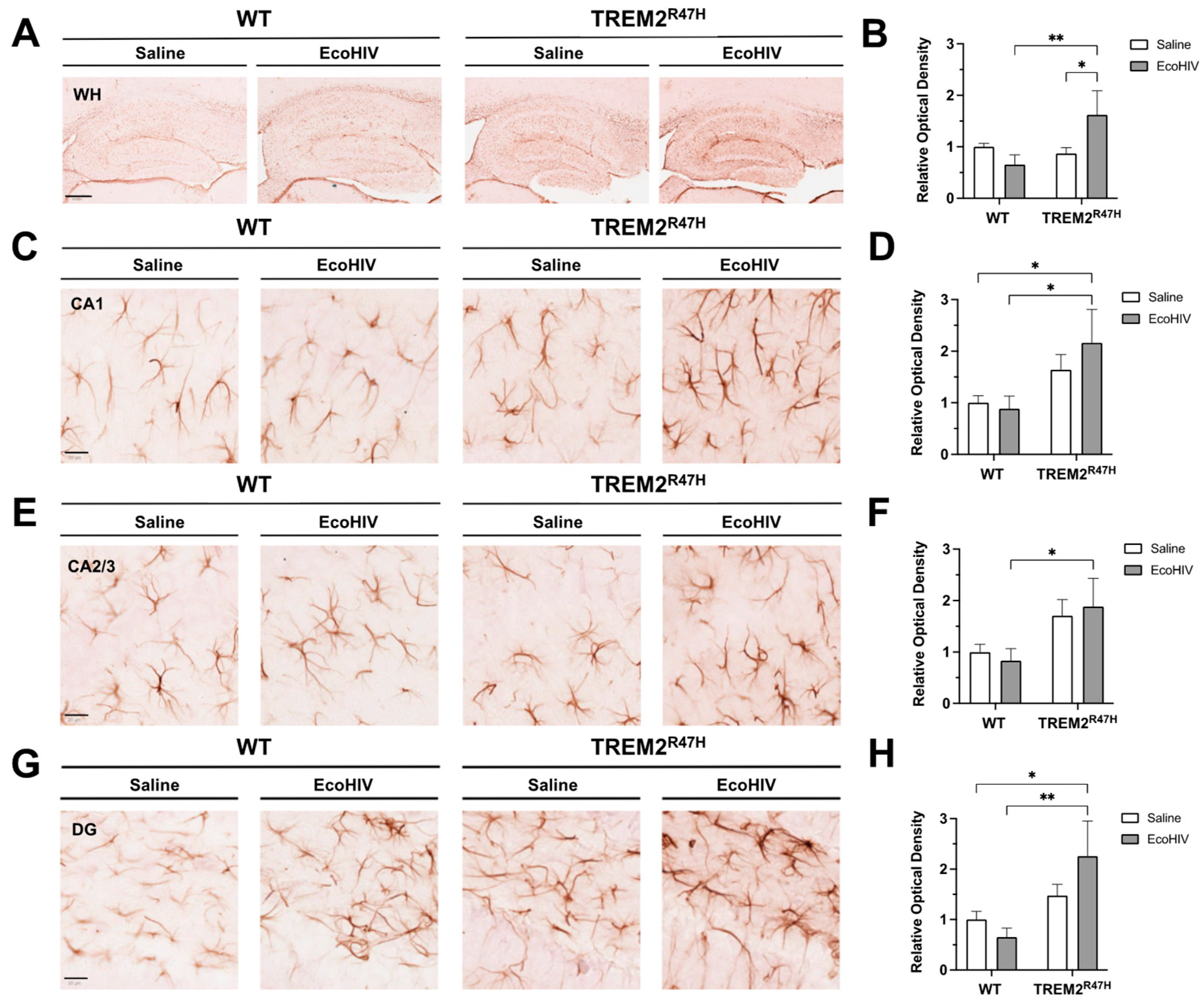
3.3. Hippocampal GFAP Expression Is Increased in TREM2R47H Mice Infected with EcoHIV
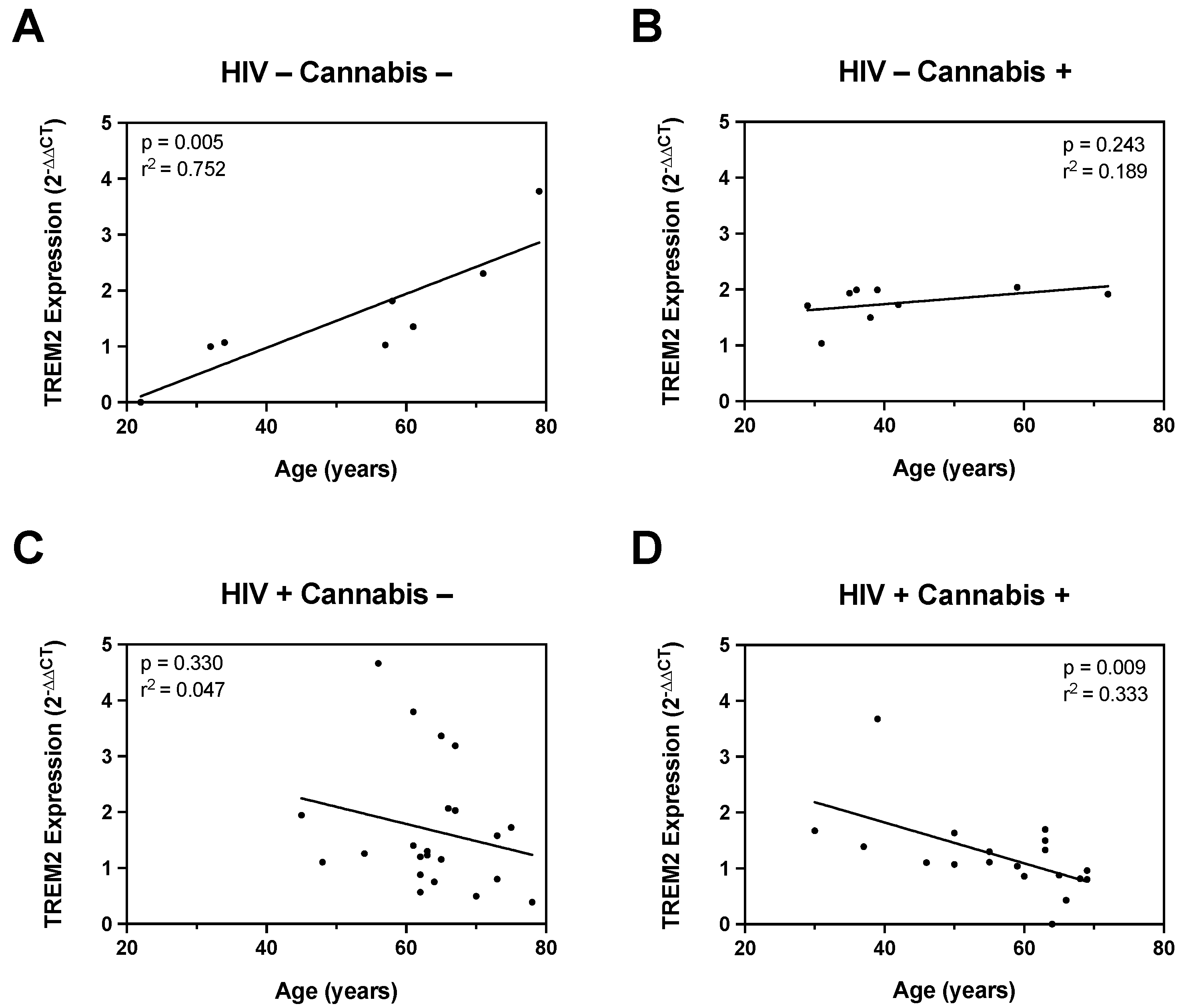
3.4. The Relationship between TREM2 and Age Is Differentially Affected by Cannabis and HIV
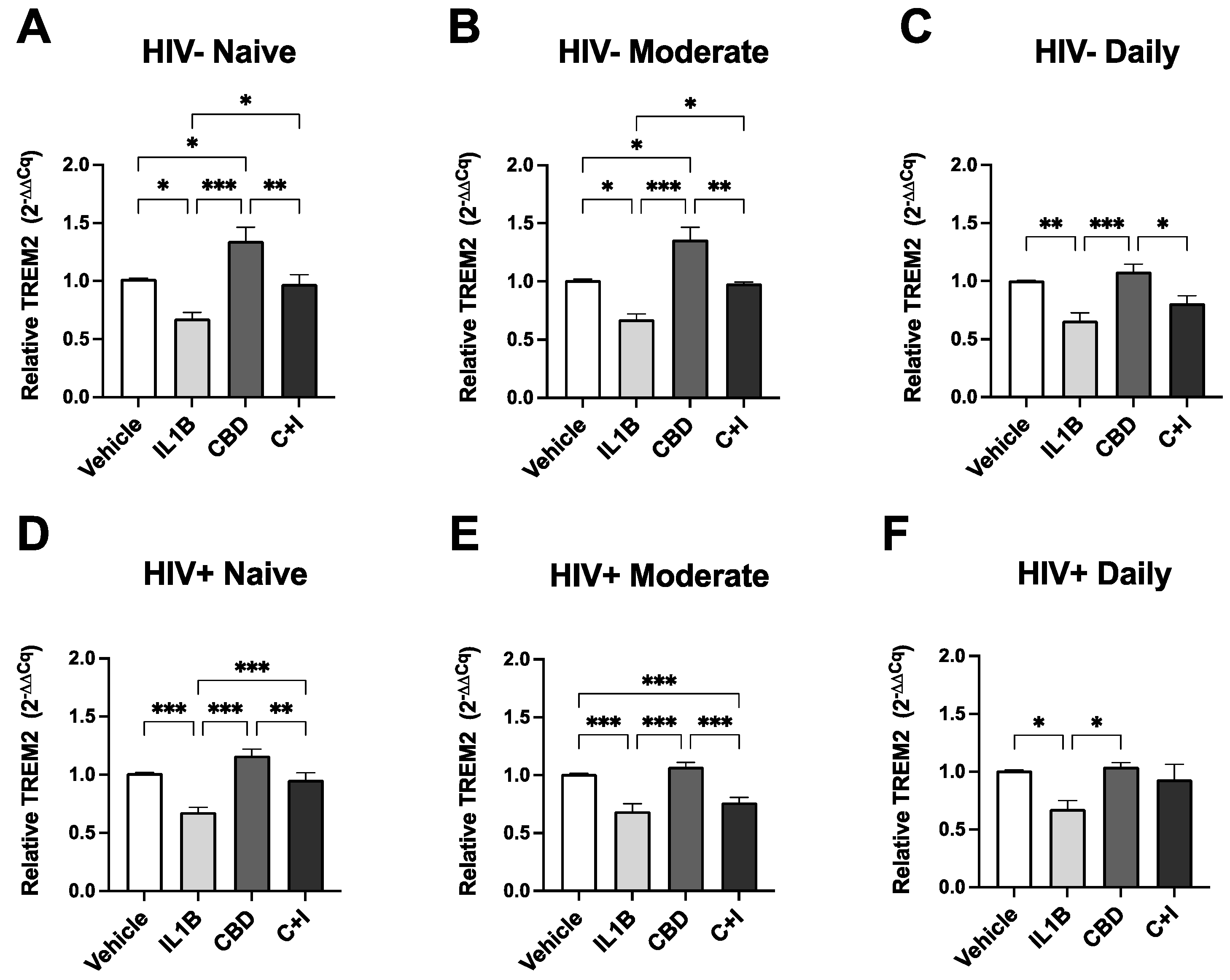
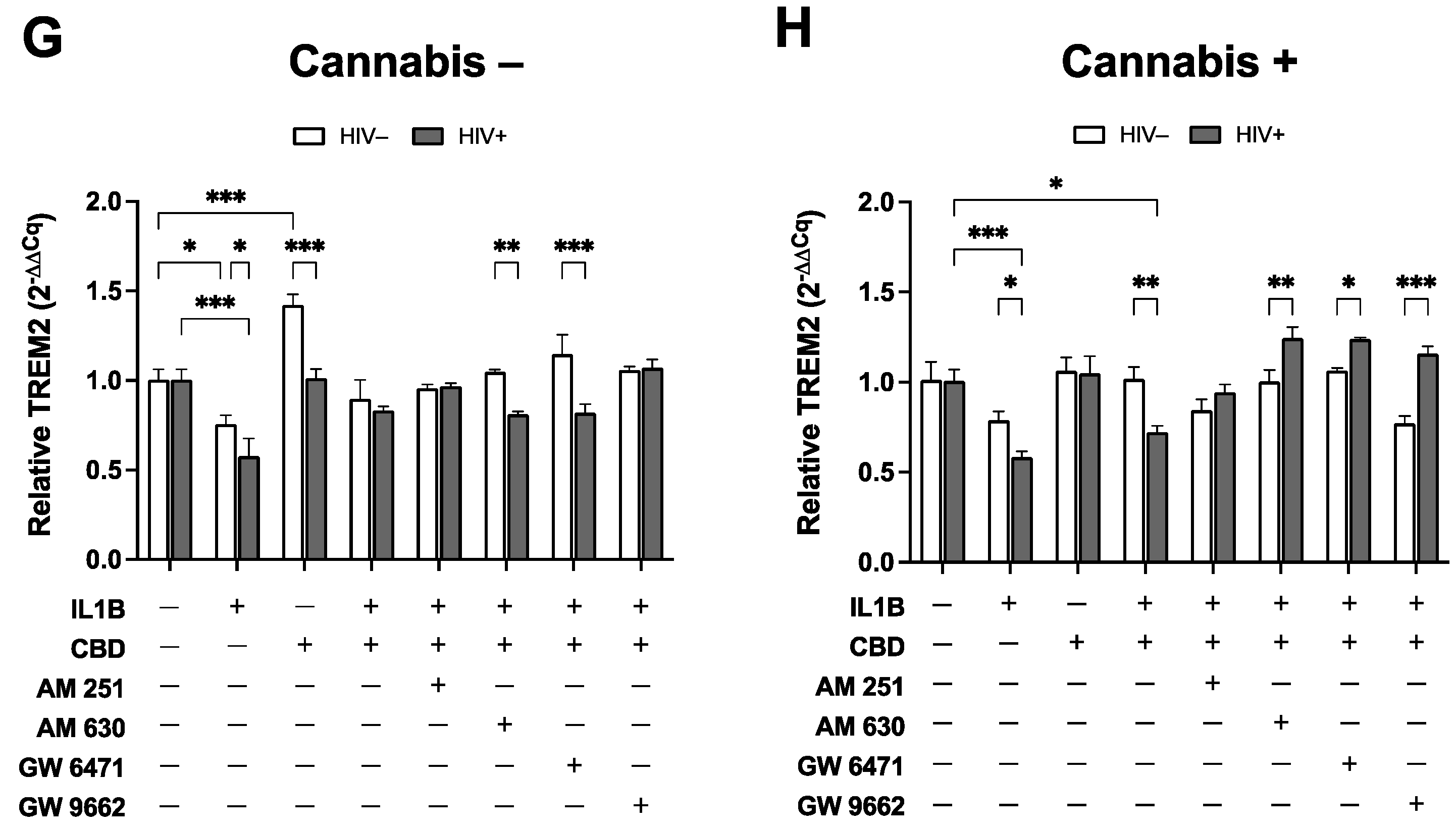
3.5. TREM2 Expression in MDMs Treated with CBD is Differentially Modulated by Cannabis Use, HIV Status, and Cannabinoid/PPAR Antagonists
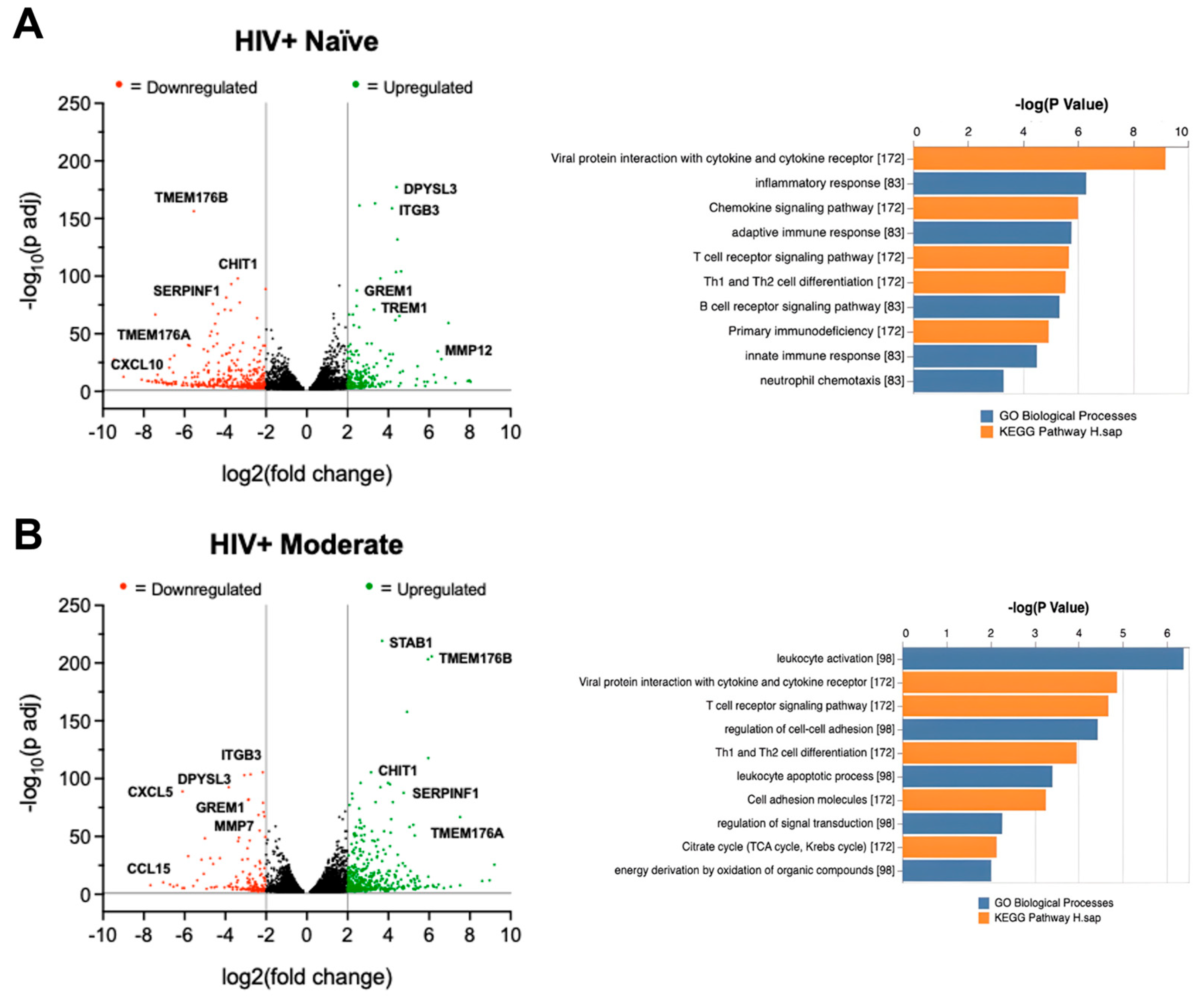
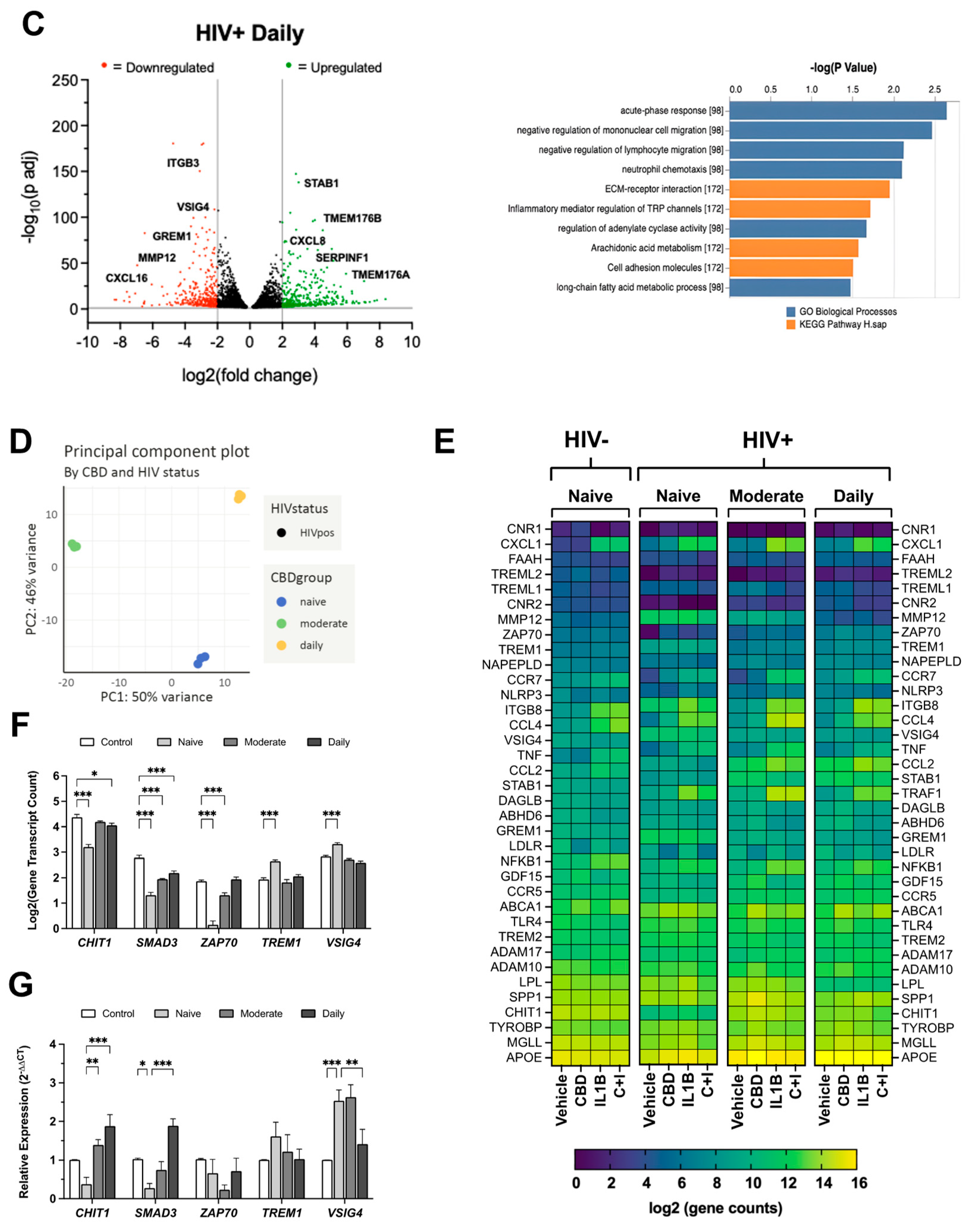
3.6. Cannabis Use Is Associated with Altered TREM2 and TREM2-Related mRNA Expression in MDMs Isolated from Blood Samples of HIV Patients
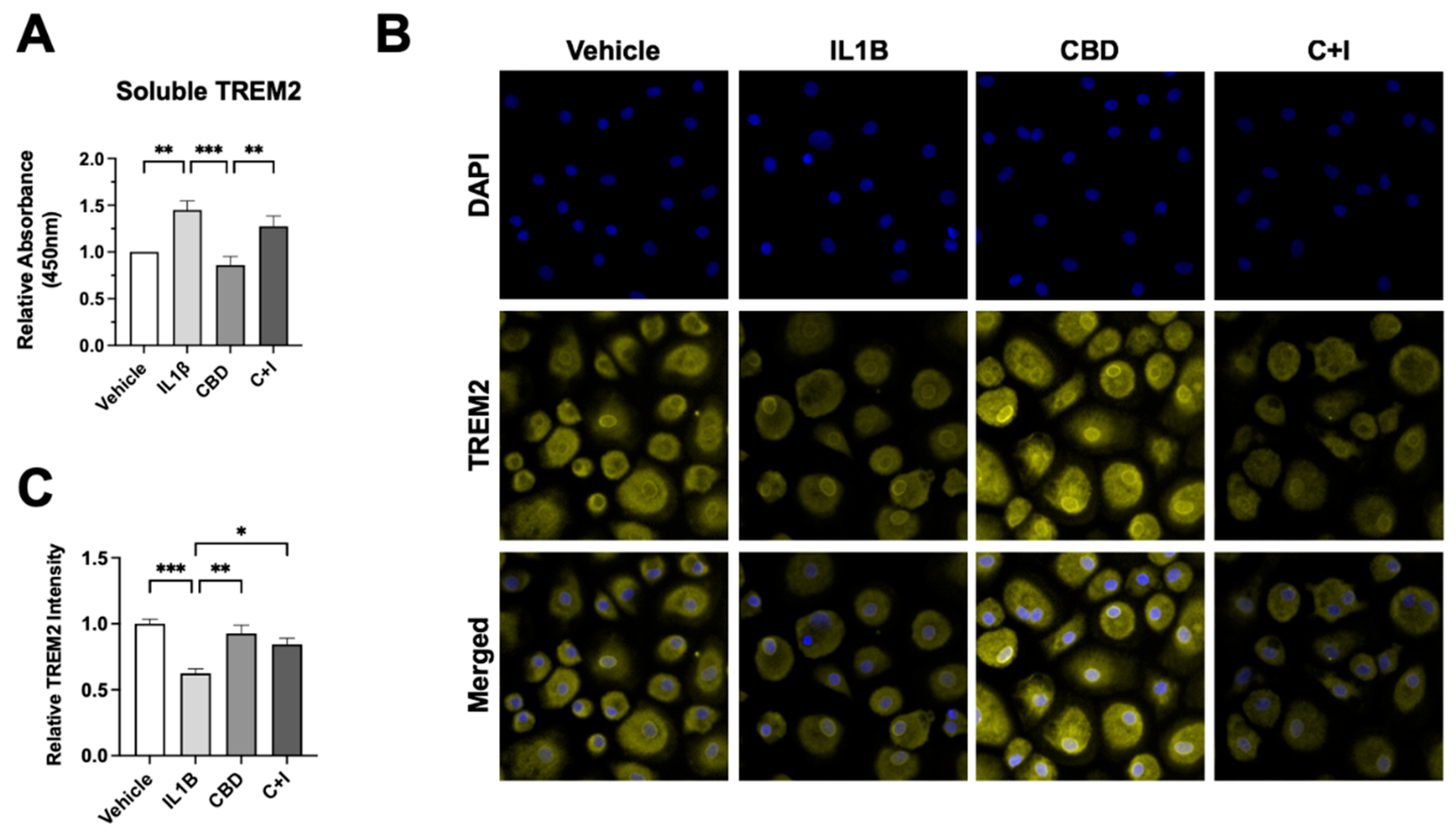
3.7. CBD Reduces sTREM2 and Increases Membrane-Bound TREM2 in Cultured MDMs
4. Discussion
5. Conclusions
References
- Zenebe, Y., et al., Worldwide Occurrence of HIV-Associated Neurocognitive Disorders and Its Associated Factors: A Systematic Review and Meta-Analysis. Front Psychiatry, 2022. 13: p. 814362.
- Mekuriaw, B., et al., Prevalence and variability of HIV/AIDS-associated neurocognitive impairments in Africa: a systematic review and meta-analysis. BMC Public Health, 2023. 23(1): p. 997.
- Nightingale, S., et al., Cognitive impairment in people living with HIV: consensus recommendations for a new approach. Nature Reviews Neurology, 2023. 19(7): p. 424-433.
- Diagnoses of HIV Infection in the United States and Dependent Areas 2019: National Profile, in HIV Surveillance Report, C.f.D.C.a. Prevention, Editor. 2021.
- Alakkas, A., et al., White matter damage, neuroinflammation, and neuronal integrity in HAND. J Neurovirol, 2019. 25(1): p. 32-41.
- Keledjian, K., et al., Correlation of HIV-Induced Neuroinflammation and Synaptopathy with Impairment of Learning and Memory in Mice with HAND. J Clin Med, 2023. 12(16).
- Solomon, I.H., et al., White Matter Abnormalities Linked to Interferon, Stress Response, and Energy Metabolism Gene Expression Changes in Older HIV-Positive Patients on Antiretroviral Therapy. Mol Neurobiol, 2020. 57(2): p. 1115-1130.
- Mackiewicz, M.M., et al., Pathogenesis of age-related HIV neurodegeneration. J Neurovirol, 2019. 25(5): p. 622-633.
- Lopez Angel, C.J., et al., Signatures of immune dysfunction in HIV and HCV infection share features with chronic inflammation in aging and persist after viral reduction or elimination. Proc Natl Acad Sci U S A, 2021. 118(14).
- Silvin, A., J. Qian, and F. Ginhoux, Brain macrophage development, diversity and dysregulation in health and disease. Cell Mol Immunol, 2023. 20(11): p. 1277-1289.
- Koenig, S., et al., Detection of AIDS virus in macrophages in brain tissue from AIDS patients with encephalopathy. Science, 1986. 233(4768): p. 1089-93.
- Gu, C.J., et al., EcoHIV infection of mice establishes latent viral reservoirs in T cells and active viral reservoirs in macrophages that are sufficient for induction of neurocognitive impairment. PLoS Pathog, 2018. 14(6): p. e1007061.
- Potash, M.J., et al., A mouse model for study of systemic HIV-1 infection, antiviral immune responses, and neuroinvasiveness. Proc Natl Acad Sci U S A, 2005. 102(10): p. 3760-5.
- Kim, B.-H., et al., EcoHIV Infection of Primary Murine Brain Cell Cultures to Model HIV Replication and Neuropathogenesis. Viruses, 2024. 16(5): p. 693.
- Burdo, T.H., A. Lackner, and K.C. Williams, Monocyte/macrophages and their role in HIV neuropathogenesis. Immunol Rev, 2013. 254(1): p. 102-13.
- Fields, J.A., et al., Alterations in brain TREM2 and Amyloid-beta levels are associated with neurocognitive impairment in HIV-infected persons on antiretroviral therapy. J Neurochem, 2018.
- Fields, J., et al., Age-dependent molecular alterations in the autophagy pathway in HIVE patients and in a gp120 tg mouse model: reversal with beclin-1 gene transfer. J Neurovirol, 2013. 19(1): p. 89-101.
- Guha, D., et al., CSF Extracellular Vesicle Abeta42 and Tau/Abeta42 Ratio Are Associated with Cognitive Impairment in Older People with HIV. Viruses, 2023. 16(1).
- Porcheray, F., et al., Macrophage activation and human immunodeficiency virus infection: HIV replication directs macrophages towards a pro-inflammatory phenotype while previous activation modulates macrophage susceptibility to infection and viral production. Virology, 2006. 349(1): p. 112-20.
- Ye, L., et al., IL-1beta and TNF-alpha induce neurotoxicity through glutamate production: a potential role for neuronal glutaminase. J Neurochem, 2013. 125(6): p. 897-908.
- Jay, T.R., V.E. von Saucken, and G.E. Landreth, TREM2 in Neurodegenerative Diseases. Mol Neurodegener, 2017. 12(1): p. 56.
- Li, Y., et al., TREM2 Regulates High Glucose-Induced Microglial Inflammation via the NLRP3 Signaling Pathway. Brain Sci, 2021. 11(7).
- Zhu, Z. Zhu, Z., et al., TREM2 suppresses the proinflammatory response to facilitate PRRSV infection via PI3K/NF-kappaB signaling. PLoS Pathog, 2020. 16(5): p. e1008543.
- Li, C., et al., TREM2 inhibits inflammatory responses in mouse microglia by suppressing the PI3K/NF-κB signaling. Cell Biol Int, 2019. 43(4): p. 360-372.
- Rivest, S., TREM2 enables amyloid beta clearance by microglia. Cell Res, 2015. 25(5): p. 535-6.
- Jin, S.C., et al., TREM2 is associated with increased risk for Alzheimer’s disease in African Americans. Mol Neurodegener, 2015. 10: p. 19.
- Lue, L.F., et al., TREM2 Protein Expression Changes Correlate with Alzheimer’s Disease Neurodegenerative Pathologies in Post-Mortem Temporal Cortices. Brain Pathol, 2015. 25(4): p. 469-80.
- Yeh, F.L., et al., TREM2 Binds to Apolipoproteins, Including APOE and CLU/APOJ, and Thereby Facilitates Uptake of Amyloid-Beta by Microglia. Neuron, 2016. 91(2): p. 328-40.
- Colonna, M. and Y. Wang, TREM2 variants: new keys to decipher Alzheimer disease pathogenesis. Nat Rev Neurosci, 2016. 17(4): p. 201-7.
- Kobayashi, M., et al., TREM2/DAP12 Signal Elicits Proinflammatory Response in Microglia and Exacerbates Neuropathic Pain. J Neurosci, 2016. 36(43): p. 11138-11150.
- Henjum, K., et al., Cerebrospinal fluid soluble TREM2 in aging and Alzheimer’s disease. Alzheimers Res Ther, 2016. 8(1): p. 17.
- Zheng, H., et al., TREM2 Promotes Microglial Survival by Activating Wnt/beta-Catenin Pathway. J Neurosci, 2017. 37(7): p. 1772-1784.
- Zhong, L., et al., Soluble TREM2 induces inflammatory responses and enhances microglial survival. J Exp Med, 2017. 214(3): p. 597-607.
- Raha, A.A., et al., Neuroprotective Effect of TREM-2 in Aging and Alzheimer’s Disease Model. J Alzheimers Dis, 2017. 55(1): p. 199-217.
- Foley, J.F., sTREM2 and neuroinflammation. Sci Signal, 2017. 10(470).
- Le Page, A., et al., Role of the peripheral innate immune system in the development of Alzheimer’s disease. Exp Gerontol, 2018. 107: p. 59-66.
- Morris, J.C. and A.L. Price, Pathologic correlates of nondemented aging, mild cognitive impairment, and early-stage Alzheimer’s disease. J Mol Neurosci, 2001. 17(2): p. 101-18.
- Nixon, R.A., Autophagy, amyloidogenesis and Alzheimer disease. J Cell Sci, 2007. 120(Pt 23): p. 4081-91.
- Dinkins, C., J. Arko-Mensah, and V. Deretic, Autophagy and HIV. Semin Cell Dev Biol, 2010. 21(7): p. 712-8.
- Hult, B., et al., Neurobiology of HIV. Int Rev Psychiatry, 2008. 20(1): p. 3-13.
- Gisslen, M., et al., CSF concentrations of soluble TREM2 as a marker of microglial activation in HIV-1 infection. Neurol Neuroimmunol Neuroinflamm, 2019. 6(1): p. e512.
- Walter, J., The Triggering Receptor Expressed on Myeloid Cells 2: A Molecular Link of Neuroinflammation and Neurodegenerative Diseases. J Biol Chem, 2016. 291(9): p. 4334-41.
- Knight, A.C., et al., Differential regulation of TREM2 and CSF1R in CNS macrophages in an SIV/macaque model of HIV CNS disease. J Neurovirol, 2020. 26(4): p. 511-519.
- Azzolini, F., et al., Neuroinflammation Is Associated with GFAP and sTREM2 Levels in Multiple Sclerosis. Biomolecules, 2022. 12(2).
- La Rosa, F., et al., TREM2 Expression and Amyloid-Beta Phagocytosis in Alzheimer’s Disease. Int J Mol Sci, 2023. 24(10).
- Jonsson, T., et al., Variant of TREM2 associated with the risk of Alzheimer’s disease. N Engl J Med, 2013. 368(2): p. 107-16.
- Tran, K.M., et al., A Trem2(R47H) mouse model without cryptic splicing drives age- and disease-dependent tissue damage and synaptic loss in response to plaques. Mol Neurodegener, 2023. 18(1): p. 12.
- Fields, J.A., et al., Alterations in brain TREM2 and Amyloid-beta levels are associated with neurocognitive impairment in HIV-infected persons on antiretroviral therapy. J Neurochem, 2018. 147(6): p. 784-802.
- Abrams, D.I., et al., Cannabis in painful HIV-associated sensory neuropathy: a randomized placebo-controlled trial. Neurology, 2007. 68(7): p. 515-21.
- Rizzo, M.D., et al., HIV-infected cannabis users have lower circulating CD16+ monocytes and IFN-gamma-inducible protein 10 levels compared with nonusing HIV patients. AIDS, 2018. 32(4): p. 419-429.
- Manuzak, J.A., et al., Heavy Cannabis Use Associated With Reduction in Activated and Inflammatory Immune Cell Frequencies in Antiretroviral Therapy-Treated Human Immunodeficiency Virus-Infected Individuals. Clin Infect Dis, 2018. 66(12): p. 1872-1882.
- Ellis, R.J., et al., Recent cannabis use in HIV is associated with reduced inflammatory markers in CSF and blood. Neurol Neuroimmunol Neuroinflamm, 2020. 7(5).
- Liu, Z., et al., Cannabis Use Associates With Reduced Proviral Burden and Inflammatory Cytokine in Tissues From Men With Clade C HIV-1 on Suppressive Antiretroviral Therapy. J Infect Dis, 2024.
- Watson, C.W., et al., Cannabis Exposure is Associated With a Lower Likelihood of Neurocognitive Impairment in People Living With HIV. J Acquir Immune Defic Syndr, 2020. 83(1): p. 56-64.
- Ellis, R.J., et al., Beneficial Effects of Cannabis on Blood-Brain Barrier Function in Human Immunodeficiency Virus. Clin Infect Dis, 2021. 73(1): p. 124-129.
- Esposito, G., et al., Cannabidiol in vivo blunts beta-amyloid induced neuroinflammation by suppressing IL-1beta and iNOS expression. Br J Pharmacol, 2007. 151(8): p. 1272-9.
- Esposito, G., et al., Cannabidiol reduces Aβ-induced neuroinflammation and promotes hippocampal neurogenesis through PPARγ involvement. PLoS One, 2011. 6(12): p. e28668.
- Borgonetti, V., et al., Non-psychotropic Cannabis sativa L. phytocomplex modulates microglial inflammatory response through CB2 receptors-, endocannabinoids-, and NF-κB-mediated signaling. Phytother Res, 2022. 36(5): p. 2246-2263.
- Mammana, S., et al., Could the Combination of Two Non-Psychotropic Cannabinoids Counteract Neuroinflammation? Effectiveness of Cannabidiol Associated with Cannabigerol. Medicina (Kaunas), 2019. 55(11).
- Turcotte, C., et al., The CB 2 receptor and its role as a regulator of inflammation. Cell Mol Life Sci, 2016. 73(23): p. 4449-4470.
- Rizzo, M.D., et al., Targeting Cannabinoid Receptor 2 on Peripheral Leukocytes to Attenuate Inflammatory Mechanisms Implicated in HIV-Associated Neurocognitive Disorder. J Neuroimmune Pharmacol, 2020. 15(4): p. 780-793.
- Schmittgen, T.D. and K.J. Livak, Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc, 2008. 3(6): p. 1101-8.
- Wang, Y., et al., TREM2 lipid sensing sustains the microglial response in an Alzheimer’s disease model. Cell, 2015. 160(6): p. 1061-71.
- Jay, T.R., et al., Disease Progression-Dependent Effects of TREM2 Deficiency in a Mouse Model of Alzheimer’s Disease. J Neurosci, 2017. 37(3): p. 637-647.
- Giraldo, M., et al., Variants in triggering receptor expressed on myeloid cells 2 are associated with both behavioral variant frontotemporal lobar degeneration and Alzheimer’s disease. Neurobiol Aging, 2013. 34(8): p. 2077.e11-8.
- Guerreiro, R., et al., TREM2 variants in Alzheimer’s disease. N Engl J Med, 2013. 368(2): p. 117-27.
- Do, T.H., et al., TREM2 macrophages induced by human lipids drive inflammation in acne lesions. Sci Immunol, 2022. 7(73): p. eabo2787.
- Olufunmilayo, E.O. and R.M.D. Holsinger, Variant TREM2 Signaling in Alzheimer’s Disease. J Mol Biol, 2022. 434(7): p. 167470.
- Swinton, M.K., et al., Alterations in Brain Cannabinoid Receptor Levels Are Associated with HIV-Associated Neurocognitive Disorders in the ART Era: Implications for Therapeutic Strategies Targeting the Endocannabinoid System. Viruses, 2021. 13(9).
- Vallée, A., et al., Effects of cannabidiol interactions with Wnt/β-catenin pathway and PPARγ on oxidative stress and neuroinflammation in Alzheimer’s disease. Acta Biochim Biophys Sin (Shanghai), 2017. 49(10): p. 853-866.
- Marini, S., et al., Oral Cannabidiol Treatment Is Associated with an Anti-Inflammatory Gene Expression Signature in Myeloid Cells of People Living with HIV. Cannabis Cannabinoid Res, 2024.
- Tito, P.A.L., et al., Cannabidiol prevents lipopolysaccharide-induced sickness behavior and alters cytokine and neurotrophic factor levels in the brain. Pharmacol Rep, 2021. 73(6): p. 1680-1693.
- Yndart Arias, A., et al., Anti-inflammatory effects of CBD in human microglial cell line infected with HIV-1. Sci Rep, 2023. 13(1): p. 7376.
- Yang, S., et al., Cannabidiol Enhances Microglial Beta-Amyloid Peptide Phagocytosis and Clearance via Vanilloid Family Type 2 Channel Activation. Int J Mol Sci, 2022. 23(10).
- Choi, S.M., et al., Classical monocyte-derived macrophages as therapeutic targets of umbilical cord mesenchymal stem cells: comparison of intratracheal and intravenous administration in a mouse model of pulmonary fibrosis. Respiratory Research, 2023. 24(1): p. 68.
- Feng, D., et al., Monocyte-derived macrophages orchestrate multiple cell-type interactions to repair necrotic liver lesions in disease models. The Journal of Clinical Investigation, 2023. 133(15).
- Leri, M., et al., EVOO Polyphenols Exert Anti-Inflammatory Effects on the Microglia Cell through TREM2 Signaling Pathway. Pharmaceuticals (Basel), 2023. 16(7).
- Liu, W., et al., Trem2 promotes anti-inflammatory responses in microglia and is suppressed under pro-inflammatory conditions. Hum Mol Genet, 2020. 29(19): p. 3224-3248.
- Yu, X., et al., Chitotriosidase attenuates brain inflammation via HDAC3/NF-κB pathway in D-galactose and aluminum-induced rat model with cognitive impairments. Neuroscience Research, 2021. 172: p. 73-79.
- Yim, L.Y., et al., Transforming Growth Factor β Signaling Promotes HIV-1 Infection in Activated and Resting Memory CD4(+) T Cells. J Virol, 2023. 97(5): p. e0027023.
- Hung, C.-H., et al., HIV-1 Nef Assembles a Src Family Kinase-ZAP-70/Syk-PI3K Cascade to Downregulate Cell-Surface MHC-I. Cell Host & Microbe, 2007. 1(2): p. 121-133.
- Campbell, G.R., R.K. To, and S.A. Spector, TREM-1 Protects HIV-1-Infected Macrophages from Apoptosis through Maintenance of Mitochondrial Function. mBio, 2019. 10(6).
- Campbell, G.R., et al., HIV-1 Tat Upregulates TREM1 Expression in Human Microglia. J Immunol, 2023. 211(3): p. 429-442.
- Li, J., et al., VSIG4 inhibits proinflammatory macrophage activation by reprogramming mitochondrial pyruvate metabolism. Nat Commun, 2017. 8(1): p. 1322.
- Watson, C.W., et al., Daily Cannabis Use is Associated With Lower CNS Inflammation in People With HIV. J Int Neuropsychol Soc, 2021. 27(6): p. 661-672.
| HIV Status | Sex (M/F/TBD) |
Ethnicity (% Hispanic) |
Race (% White) |
Education (years) |
|---|---|---|---|---|
| HIV- | 11 / 3 / 3 | 14.3 | 14.3 | 15.5 |
| HIV+ | 32 / 4 / 6 | 13.3 | 76.7 | 15.6 |
| Group |
Age (years ± SEM) |
GDS (score ± SEM) |
POMS (score ± SEM) |
Infection Duration (years ± SEM) |
| HIV+ Naive (n=22) | 63.68 ± 1.69 | 0.37 ± 0.08 | 1.78 ± 0.29 | 27.46 ± 2.25 |
| HIV+ Moderate (n=12) | 55.83 ± 3.51 | 0.46 ± 0.13 | 2.05 ± 0.52 | 22.42 ± 2.76 |
| HIV+ Daily (n=8) | 53.62 ± 5.19 | 0.82 ± 0.3 | 2.55 ± 0.26 | 28.49 ± 1.91 |
| HIV- Naïve (n=8) | 51.75 ± 7.14 | 0.28 ± 0.05 | 1.20 ± 0.30 | N/A ± N/A |
| HIV- Moderate (n=1) | 29.0 ± N/A | 0.06 ± N/A | 0.19 ± N/A | N/A ± N/A |
| HIV- Daily (n=8) | 44.0 ± 4.98 | 0.25 ± N/A | 2.94 ± N/A | N/A ± N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




