Submitted:
07 August 2024
Posted:
12 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Neutronic
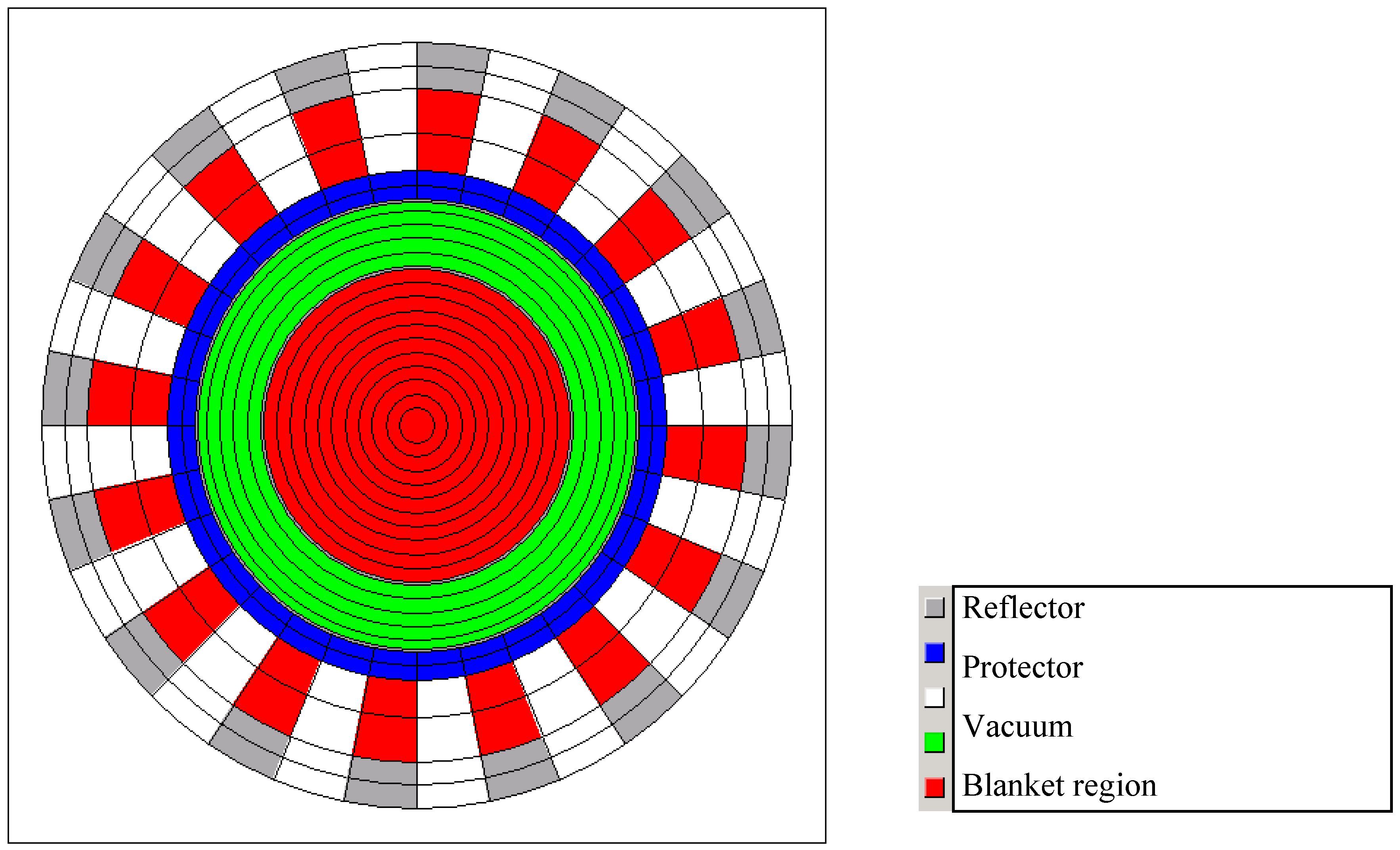
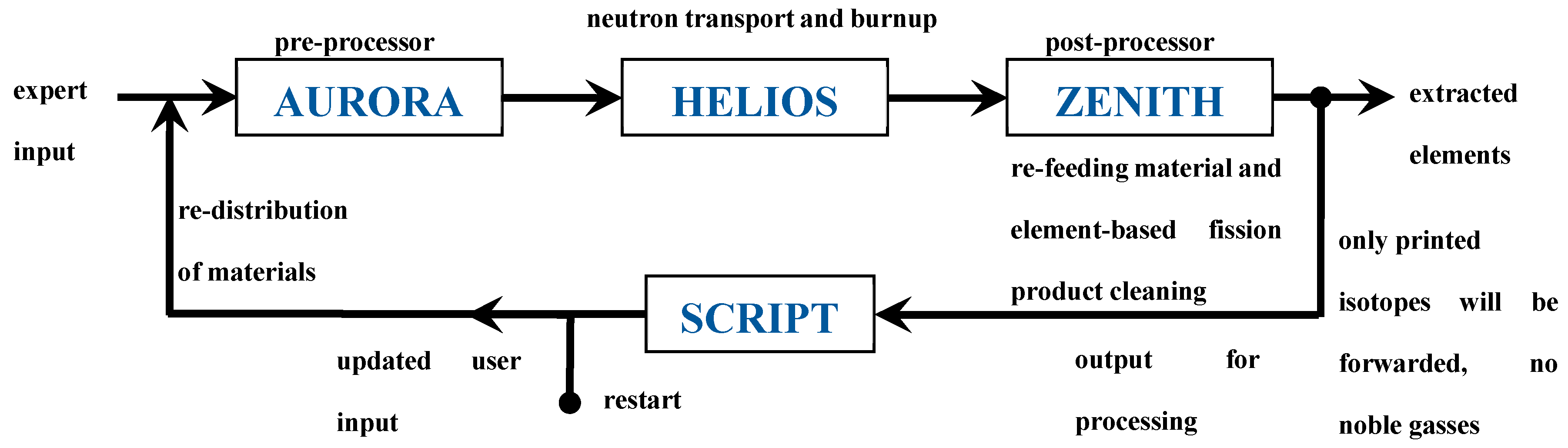
2.1. Code, Model and Methods
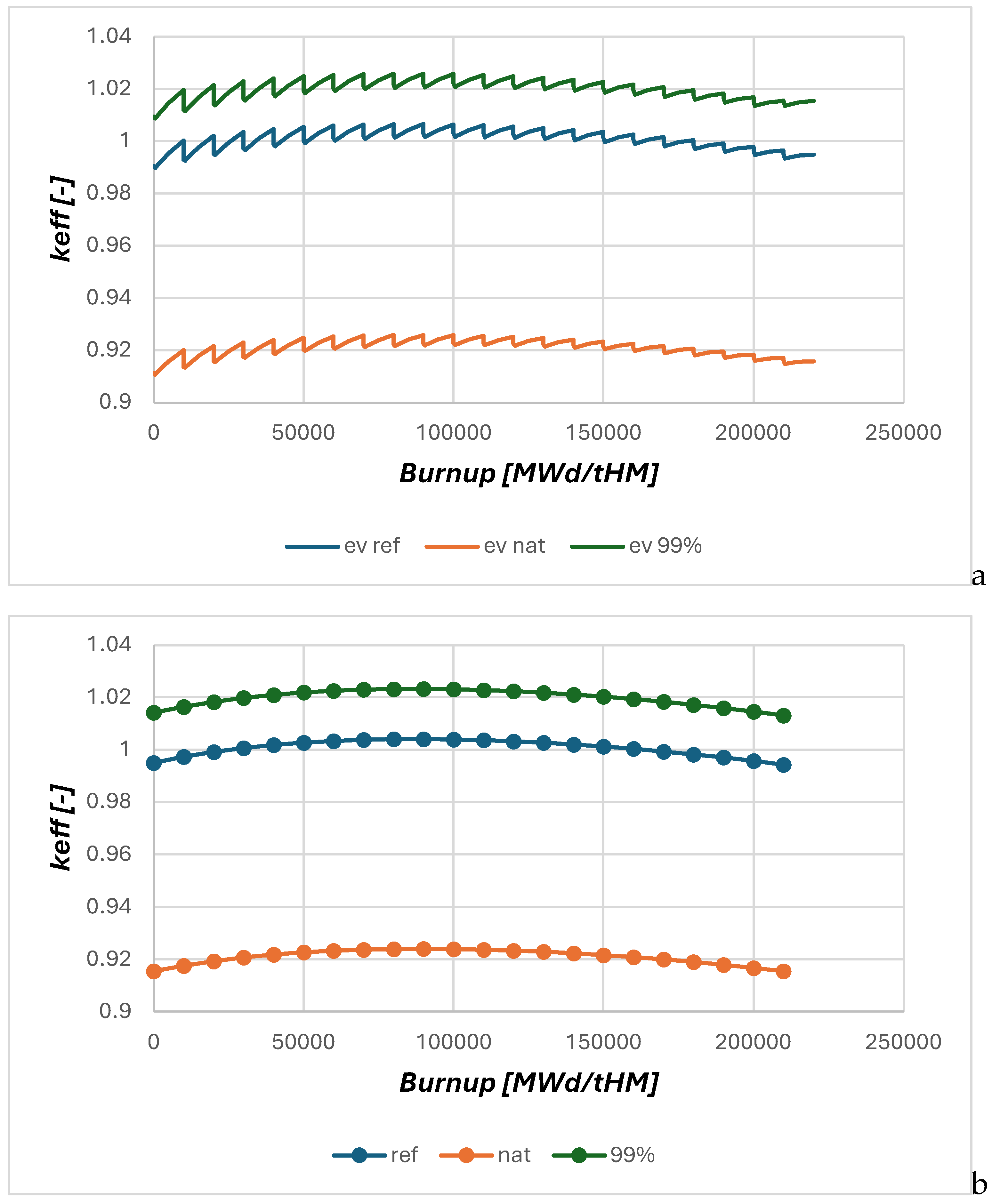
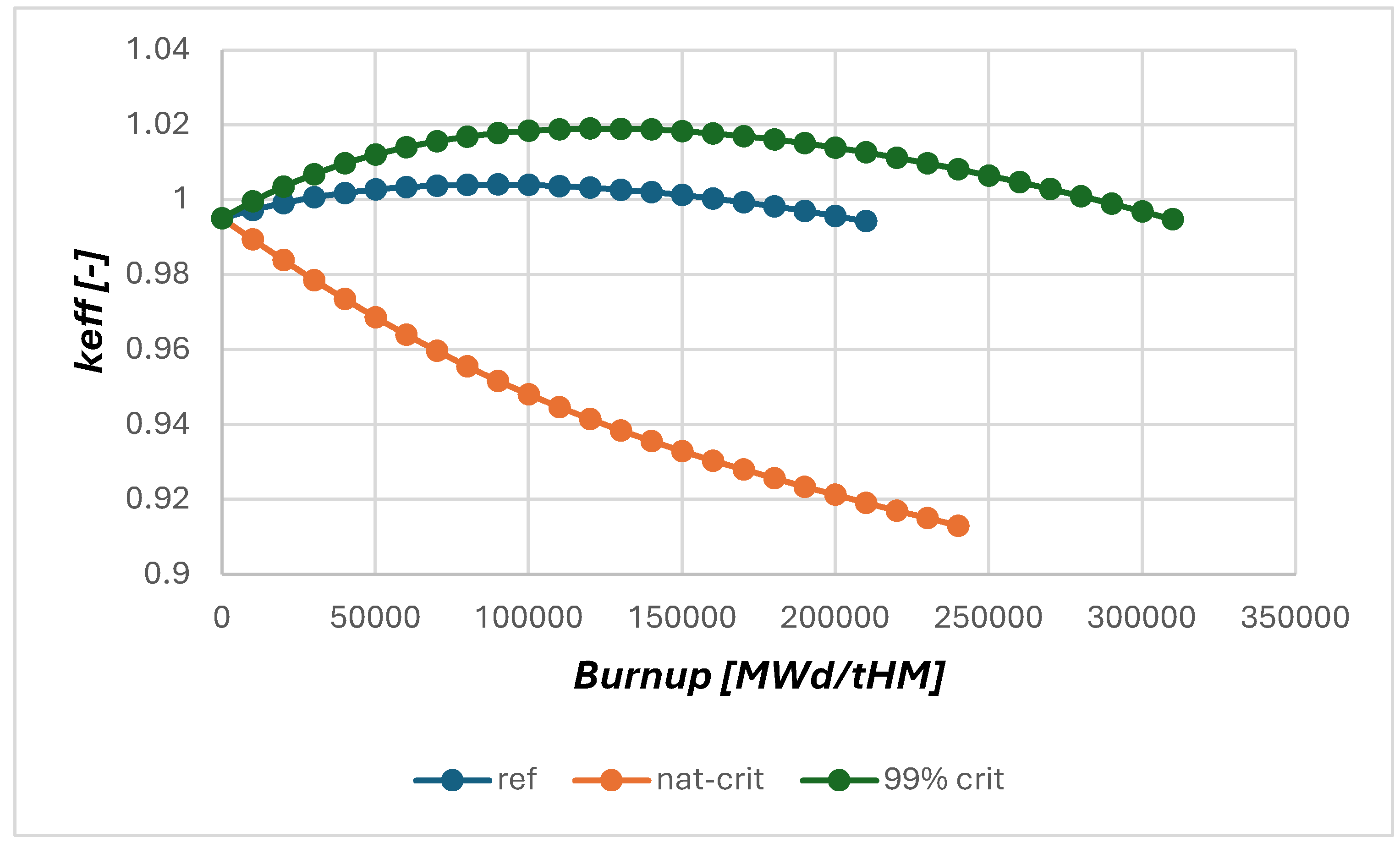
2.2. Neutronic Results
3. Isotope Separations
- based directly on the atomic weight of the isotope,
- based on the small differences in chemical reaction rate, and,
- based on properties such as spectroscopic not directly connected to atomic weight.
3.1. Physical Separations Methods
Separation by Fractionated Distillation…
Separation by Electromagnetic Separation
Separation by Diffusion
Separation by Gas Centrifugation
- -
- length of the gas centrifuge
- -
- radius of the centrifuge
- -
- axial location of the feed point
- -
- parameters of the scoops
- -
- feed flow rate,
- -
- pressure at the wall and on physical properties
- -
- peripheral rotation speed
- -
- feed rate of the working gas
- -
- temperature T0 at injection
- -
- temperature distribution on the wall
- -
- pressure of the gas at the rotor wall
- -
- molar weight of the isotope reagent
- -
- viscosity of the gas
- -
- molar weight of the carrier gas (if any)
- -
- product density media
- -
- product diffusion coefficient.
3.2. Chemical Separation Methods
Separation Using Solid Ion Exchangers
Separation Using Liquid - Liquid Extraction
From the Solid - Liquid to the Liquid - Liquid Extraction
Separation by Photochemistry
Electrolytic Separation
Separation by Ionic Migration
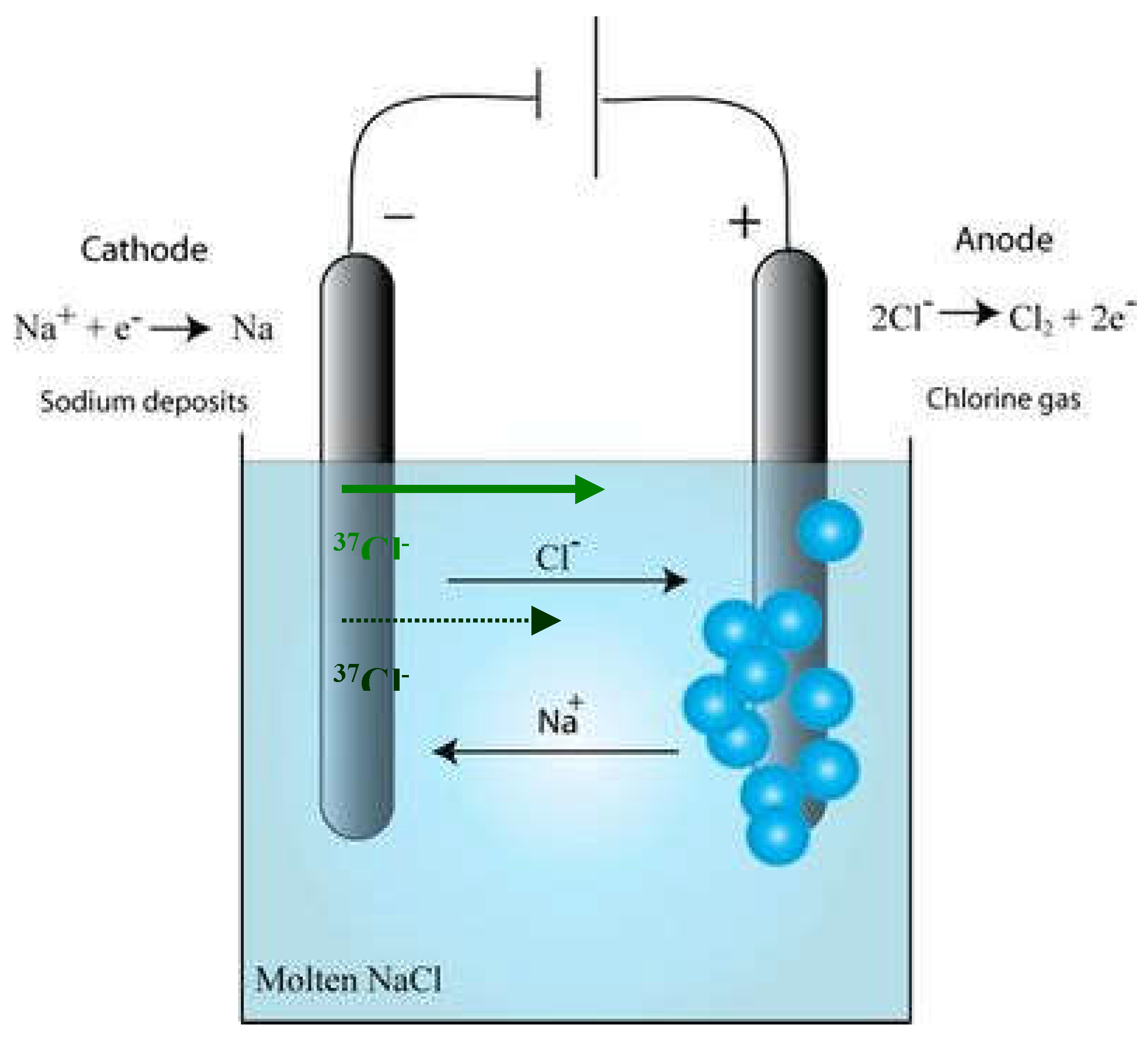
Separation by Chloride Electrolysis
4. Discussion
Isotopic Exchanges
Kinetic Effects…
Convection and Mixing
From Single Stage to Multi-Stage Separation
Combined Processes
Evaporation - Precipitation
Sorption - Volatilisation
Combining Neutronic and Separation
5. Conclusion
Acknowledgement
References
- B. Merk, D. Litskevich, A. Peakman, M. Bankhead, IMAGINE, A disruptive change to nuclear or how can we make more out of the existing spent nuclear fuel and what has to be done to make it possible in the UK? ATW- internat. Journal for Nuclear Power, 64 (2019) 353-359.
- C. Degueldre, R Dawson, V Najdanovic, 'Nuclear fuel cycle, with a liquid ore and fuel: toward renewable energy', Sustainable Energy and Fuels, 3 (2019) 1693-1700. [CrossRef]
- R. Roper, M. Harkema, P. Sabharwall, C. Riddle, B. Chisholm, B. Day, P. Marotta, Molten salt for advanced energy applications: A review. Annals of Nuclear Energy 169 (2022) 108924. [CrossRef]
- B. Merk, A. Detkina, S. Atkinson, D. Litskevich, G. Cartland-Glover, Evaluation of the breeding performance of a NaCl-UCl-based reactor system. Energy, 12 (2019). [CrossRef]
- Z. Mausolff, M. DeHart, S. Goluoglu, Design and assessment of a molten chloride fast reactor , Nuclear Engineering and Design, 379 (2021) 111181. [CrossRef]
- J. Krepel, K. J. Kramer, Molten Chloride Fast Reactors (MCFRs), Editor(s): Ehud Greenspan, Encyclopedia of Nuclear Energy, Elsevier, (2021) 625-642. [CrossRef]
- J. Serp, M. Allibert, O. Beneš, S. Delpech, O. Feynberg, V. Ghetta, D. Heuer, D. Holcomb, V Ignatiev, J L Kloosterman, Lelio Luzzi, E Merle-Lucotte, J Uhlíř, R Yoshioka, D Zhimin, The molten salt reactor (MSR) in generation IV: Overview and perspectives, Progress in Nuclear Energy, 77(2014) 308-319. [CrossRef]
- L. Martinot, Thermodynamic properties of infinitely dilute solutions of UCl3 and UCl4 in the KCl-NaCl-MgCl2 (30:20:50) ternary eutectic, Journal of the Less Common Metals, 86 (1982) 203-210. [CrossRef]
- L.Y. He, G.C. Li, S.P. Xia, Jin-Gen Chen, Yang Zou, Gui-Min Liu, Effect of 37Cl enrichment on neutrons in a molten chloride salt fast reactor. Nucl. Sci. Tech., 31 (2020) 27. [CrossRef]
- F. G. Gosling, The Manhattan Project: Making the Atomic Bomb (DOE/MA-0001; Washington: History Division, Department of Energy, January 1999), 5-6.
- M. E. Gooden, T. A. Bredeweg, J. B. Wilhelmy, M. M. Fowler, R. S. Rundberg, A. J. Silano, A. . P Tonchev, 35Cl(n,p)35S reaction cross-section using monoenergetic neutrons in the intermediate and fast energy regions, 2018-04-1 Los Alamos Technical ReportLA-UR-18-23117. [CrossRef]
- HELIOS2 Methods Manual (version 2.03.01), Studsvik, SSP-11/452 Rev 6, January 12, 20214.
- E.A. Villarino, R.J.J. Stammler, A. Ferri, J.J. Casal, HELIOS: angularly dependent collision probabilities, Nuclear Science and Engineering 112 (1992) 16-31. [CrossRef]
- C.A. Wemple, H-N.M. Gheorghiu , R.J.J. Stamm’ler , E.A. Villarino (2008) Recent Advances in the HELIOS-2 Lattice Physics Code, International Conference on the Physics of Reactors “Nuclear Power: A Sustainable Resource”, Interlaken, Switzerland, September 14-19, 2008.
- AURORA USER MANUAL, Studsvik, SSP-11/451 Rev 8, January 12, 2021.
- ZENITH USER MANUAL, Studsvik, SSP-11/460 Rev. 5, December 8, 2020.
- Evaluation and Viability of Liquid Fuel Fast Reactor System EVOL, DELIVERABLE D2.1, Design parameters definition for most stable salt flux, rev 3 30/04/2012.
- M. Brovchenko, J-L Kloosterman, L Luzzi 3, E Merle, D Heuer, A Laureau, O Feynberg, V. Ignatiev, M. Aufiero, A. Cammi, C. Fiorina, F. Alcaro, S. Dulla, P Ravetto, L. Frima, D Lathouwers, B. Merk., Neutronic benchmark of the molten salt fast reactor in the frame of the EVOL and MARS collaborative projects, EPJ Nuclear Sci. Technol. 5, (2019). [CrossRef]
- B. Merk, A. Detkina, D, Litskevich, S. Atkinson, G. Cartland-Glover, The Interplay between Breeding and Thermal Feedback in a Molten Chlorine Fast Reactor. Energies, 13 (2020) 1609. [CrossRef]
- M. A. Jessee, J. J. Jarrell, W. A. Wieselquist, M. L. Williams, K. S. Kim, T. M. Evans, S. P. Hamilton, C. A. Gentry (2017),“POLARIS - 2D LIGHT WATER REACTOR LATTICE PHYSICS MODULE, in SCALE Code System”, edited by B.T. Rearden, M.A. Jessee, February 2014.
- B. Merk, U. Rohde, V. Glivici-Cotruta, D. Litskevich, S. Scholl, On the use of a Molten Salt Fast Reactor to apply an idealized, Transmutation Scenario for the Nuclear Phase Out. PLOS ONE, 9 (2014). [CrossRef]
- Merk B, Litskevich D. Transmutation of All German Transuranium under Nuclear Phase Out Conditions - Is This Feasible from Neutronic Point of View? PLoS One., 10 (2015) :e0145652. [CrossRef]
- B. Merk, D. Litskevich, A disruptive approach to eliminating weapon-grade plutonium – Pu burning in a molten salt fast reactor. PLoS ONE 13 (2018) e0201757. [CrossRef]
- R. Rachamin, C. Wemple, E. Fridman, Neutronic analysis of SFR core with HELIOS-2, Serpent, and DYN3D codes, Annals of Nuclear Energy, 55 (2013). [CrossRef]
- Batchelder et al., Phys. Rev. C, vol. 99, no. 4, 2019.
- Kuvin et al., Physical Review C, 102, issue 2, 2020, (https://www.oecdnea.org/janisweb/book/neutrons/Cl35/MT103which).
- Palmiotti, G., Gehin, J. C., & Cisneros, T. (2021). Assessment of nuclear data needs for advanced reactor demonstrations: application to the molten chloride reactor experiment (MCRE) (No. INL/CON-21-64838-Rev000). Idaho National Lab.(INL), Idaho Falls, ID (United States).
- Ortwin Renn (Hrsg.), Partitionierung und Transmutation Forschung – Entwicklung – Gesellschaftliche Implikationen, acatech SUDIE Dezember 2013.
- J. Bigeleise, Isotope Separation Practice, in Spindel Ed., Isotope Effects in Chemical Processes Advances in Chemistry; American Chemical Society (1969). [CrossRef]
- J. W. Beams, C. Skarstrom, The Concentration of Isotopes by the Evaporative Centrifuge Method, Phys. Rev. 56 (1939) 266 - 272. [CrossRef]
- P. Baertschi, W. Kuhn. Vapor pressure differences of isotopic compounds. Infrared as the cause of higher volatility of the heavier species. Helv. Chim. Acta 40 (1957) 1084-1103. [CrossRef]
- A Liebscher, J Barnes Z. Sharp, Chlorine isotope vapor–liquid fractionation during experimental fluid-phase separation at 400 °C/23 MPa to 450 °C/42 MPa, Chemical Geology, 2342 (2006) 340-345. [CrossRef]
- Sun, Q. Xu, S. Xu, H. Shen, J. Sun, Y. Zhang, Separation and analysis of chlorine isotopes in higher plants, Chemical Geology, 381 (2014) 21-25. [CrossRef]
- Degueldre, The analysis of nuclear materials and their environments, Springer – Nature (2017). [CrossRef]
- Derrick Quarles Jr. Andrew D.Toms RonaldSmithJr. PatrickSullivan DavidBass JohnLeone. Automated ICP-MS method to measure bromine, chlorine, and iodine species and total metals content in drinking water, Talanta Open, 1 (2020) 100002. [CrossRef]
- A. Z. Kranz, W. W. Watson, Chlorine Isotope Separation by Thermal Diffusion, Phys. Rev. 91, (1953) 1469. [CrossRef]
- R. Scott Kemp, Gas Centrifuge Theory and Development: A Review of U.S. Programs, The Technical Basis for Arms Control, Disarmament, and Nonproliferation Initiatives, 17 (2009) 1-19. [CrossRef]
- E.M Aisen, V.D Borisevich, E.V Levin, G.E Popov, A.V Tikhomirov, S.V Yupatov, Study of isotope separation of some chemical elements in a gas centrifuge. Separation Science and Technology, 374 (1996) 127-131. [CrossRef]
- S. V. Bogovalov, Isotope Separation in Concurrent Gas Centrifuges. Physics Procedia, 72 (2015) 297-304. [CrossRef]
- Ch. Ying, Sh. Zeng., Y. Nie, X. .Shang, H.G. Wood, Over All Separation Factors for Stable isotopes by Gas Centrifuge, Separation Science and Technology 36 ( 2006) 2006159-175. [CrossRef]
- M. Musashi, T. Oi, H. G.M. Eggenkamp, Y. Yato, M. Matsuo, Anion-exchange chromatographic study of the chlorine isotope effect accompanying hydration, Journal of Chromatography A, 1140 (2007) 121-125. [CrossRef]
- Kl. G. Heumann, Kl. Baier, Dependence of Chlorine Isotope Separation in Ion Exchange Chromatography on the Nature and Concentration of the Eluent. Z. Naturforsch. 35b (1980) 1538-1540. [CrossRef]
- Kl. G. Heumann, Kl. Baier, G. Wibmer, Chlor-Isotopenseparation an einem wasserhaltigen Zirkondioxidaustauscher / Chlorine Isotope Separation Using an Hydrous Zirconium Dioxide Exchanger, Zeitschrift fur Naturforschung B 35 (1980). [CrossRef]
- M. Musashi, T. Oi, H. G.M.Eggenkamp, Experimental determination of chlorine isotope separation factor by anion-exchange chromatography, Analytica Chimica Acta, 508 (2004) 37-40. [CrossRef]
- Zezheng Zhang, Yongzhong Jia,, Bing Liu, Huaxin Sun, Yan Jing, Quanyou Zhang, Fei Shao, Mixiang Qi, Ying Yao, Extraction and separation of lithium isotopes by using organic liquid film extraction system of crown ether-ionic liquid, Fusion Engineering and Design, 161 (2020) 112015. [CrossRef]
- A V Khoroshilov, P I Ivanov, Separation of boron isotopes by chemical exchange in liquid-liquid systems, J. Phys.: Conf. Ser. 1099 (2018) 012006. [CrossRef]
- G.D. Rabinovich, V.P. Ivakhnik, Chlorine isotope separation by liquid-phase thermal diffusion. At Energy, 45 (1978) 879–882. [CrossRef]
- P.N. Nesterenko, Ion Exchange. Overview., M. Miró, P. Worsfold, C. Poole, A. Townsend (Eds.), Encyclopedia of Analytical Science (third ed.), Academic Press, Oxford (2019), pp. 204-210. [CrossRef]
- [ ] Bio-Rad, AG@ 1, AG MP-l and AG 2 Strtng, Anion Exchange Resin Instruction Manual BioRad Laboratories, LIT212 Rev C (2023).
- J.J.van Deemter, F.J.Zuiderweg, A.Klinkenberg, Longitudinal diffusion and resistance to mass transfer as causes of nonideality in chromatography Chemical Engineering Science, 5 (1956) 271-289. [CrossRef]
- W. Kuhn, H. Martin. Photochemical separation of isotopes, Naturwissenschaften 20, 772(1932). 3. - 27.
- W. Kuhn, H. Martin, K. H. Eldau. Enrichment of the chlorine isotopes by irradiation of chlorine dioxide at low pressure (the photochemistry of chlorine dioxide). Z. physik. Chem. 5O (1941) 213-254, Chem. Zentr. I (1942) 579.
- D. D. S. Liu, S. Datta, R. N. Zare, Laser separation of chlorine isotopes. Photochemical reaction of electronically excited iodine monochloride with halogenated olefins, J. Am. Chem. Soc., 97, (1975) 2557–2558. [CrossRef]
- R. N. Zare, Laser isotope separation by selective excited state photochemistry. Annual progress report, March 31, 1976--February 28, 1977. United States: N. p., 1977. [CrossRef]
- CA Lucy, TL McDonald. Separation of chloride isotopes by capillary electrophoresis based on the isotope effect on ion mobility. Anal Chem., 67 (1995) 1074-1078. [CrossRef]
- M. Petek, D.W. Ramey, R. D. Taylor, E.H. Kobisk, Tritium isotope separation from light and heavy water by bipolar electrolysis. United States: N. p., 1980. CONF-800427-15.
- J. Wild, P. Wang, T. Jin, Y. Yang, Modeling Isotope Separation in Electrochemical Lithium Deposition, Journal of The Electrochemical Society, 169 (2022) 032504. [CrossRef]
- Klemm. Separation of isotopes by electrolytic migration of ions, Colloques intern. centr. natl. recherche sci. 39, Electrolyse C18 -C24 (1952).
- Klemm. A. Lunden, Isotopenanreicherung beim Chlor durch elektrolytische Überführung in geschmolzenem Bleichlorid, Z. Naturforsch., 10a (1955) 282-284.
- C. Wang, H. Ju, X. Zhou, P. Zhang, Z. Xue, L. Mao, F. Shao, Y. Jing, Y. Jia, J. Sun, Separation of lithium isotopes: Electromigration coupling with crystallization (1999), Journal of Molecular Liquids, Volume 355 (2022) 118911. [CrossRef]
- H. L. Johnston, D. A. Hutchison, Efficiency of the Electrolytic Separation of Chlorine Isotopes, J. Chem. Phys. 10 (1942) 469. [CrossRef]
- Yongtao Li , Ed J George, Analysis of perchlorate in water by reversed-phase LC/ESI-MS/MS using an internal standard technique, Anal Chem. 77 (2005) 4453-8. [CrossRef]
- J. R. Christie, W. D. Johnson, A. G. Loudon, Allan MacColl, M. N. Mruzek, Heavy atom kinetic isotope effects. Part 3.—The chlorine kinetic isotope effect in the gas-phase unimolecular decomposition of ethyl chloride. J. Chem. Soc., Faraday Trans. 71, (1975) 1937-1942. [CrossRef]
- Lewandowicz, J. Rudziński, L. Tronstad, M. Widersten, P. Ryberg, O. Matsson, P. Paneth. Chlorine Kinetic Isotope Effects on the Haloalkane Dehalogenase Reaction. J. Am. Chem. Soc., 123 (2001) 4550–4555. [CrossRef]
- M. Musashi, Takao Oi, Hans G.M.Eggenkamp, YumioYato, Motoyuki Matsuo, Anion-exchange chromatographic study of the chlorine isotope effect accompanying hydration, Journal of Chromatography A, 1140 (2007) 121-125. [CrossRef]
- V. Ghazanfari, M. M. Shadman, Numerical solution of a comprehensive form of convection–diffusion equation for binary isotope in the gas centrifuge, Annals of Nuclear Energy, 175, 2022, 109220. [CrossRef]
- Klemm. Infinitively long separation tube with end volumes, Z. Naturforsch 3a (1948) 211-216.
- T.F. Johns, Isotope separation by multistage methods, in Progress in Nuclear Physics, 6 (2013) 1-25. [CrossRef]
- Chong Guang Luo, Ying Kai Xiao, Hai Zhou Ma, Yun Qi Ma, Yan Ling Zhang, Mao Yong He Stable isotope fractionation of chlorine during evaporation of brine from a saline lake, Chinese Science Bulletin , 57 (2012) 1833–1843. [CrossRef]
- H. Holmstrand , Manolis Mandalakis , Zdenek Zencak , Örjan Gustafsson , Per Andersson , Chlorine isotope fractionation of a semi-volatile organochlorine compound during preparative megabore-column capillary gas chromatography, Journal of Chromatography A 1103 (2006) 133-138. [CrossRef]
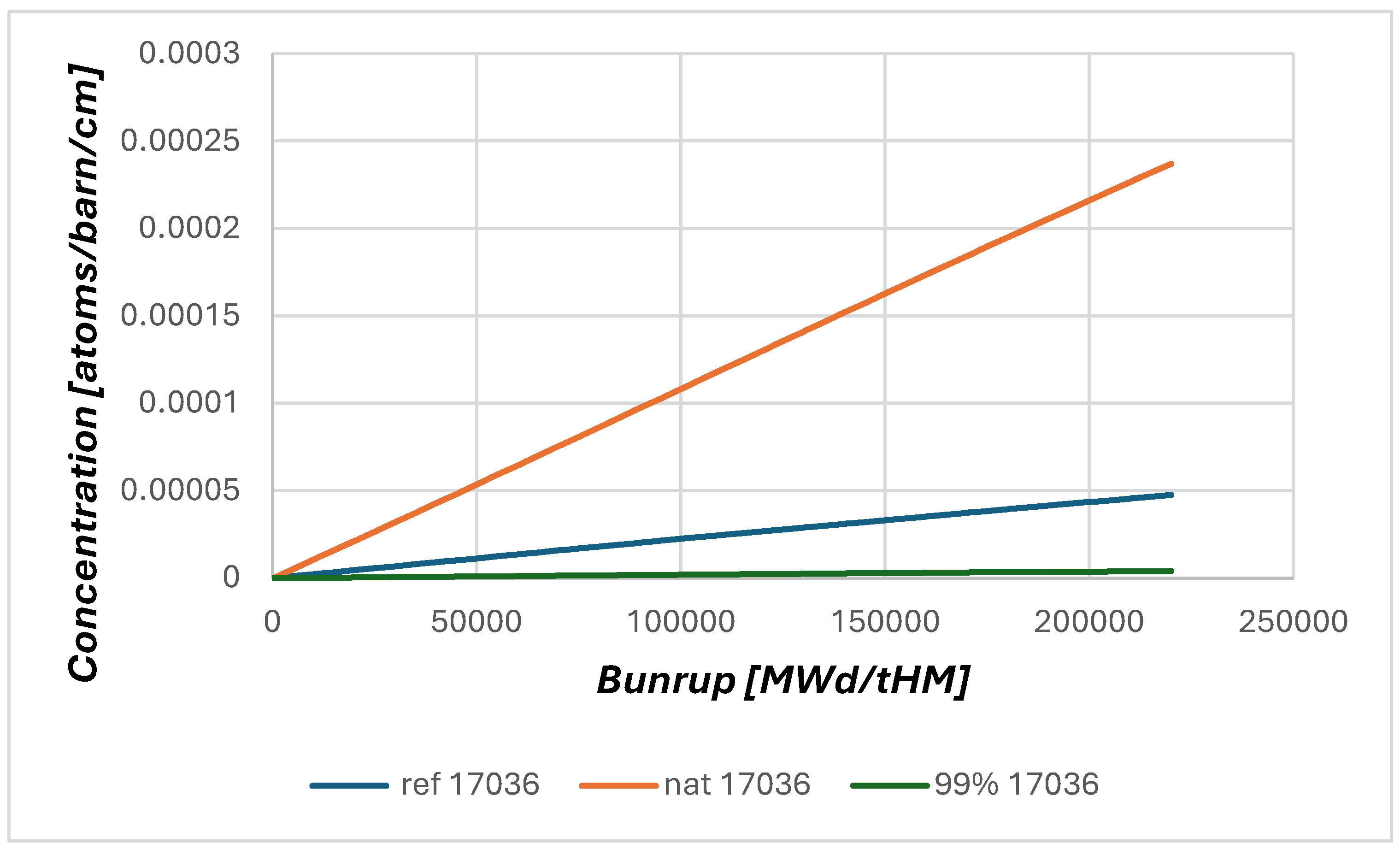
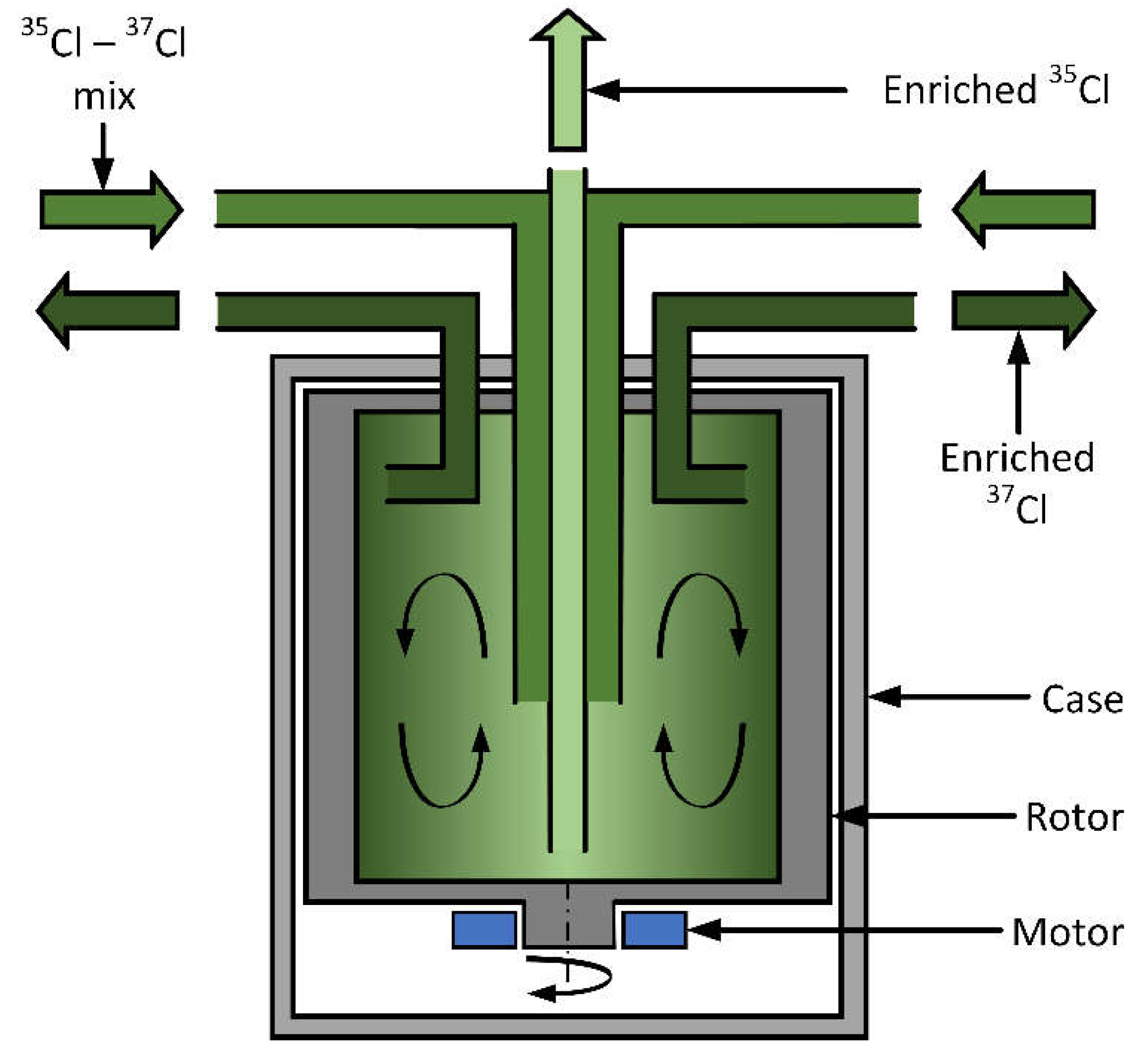
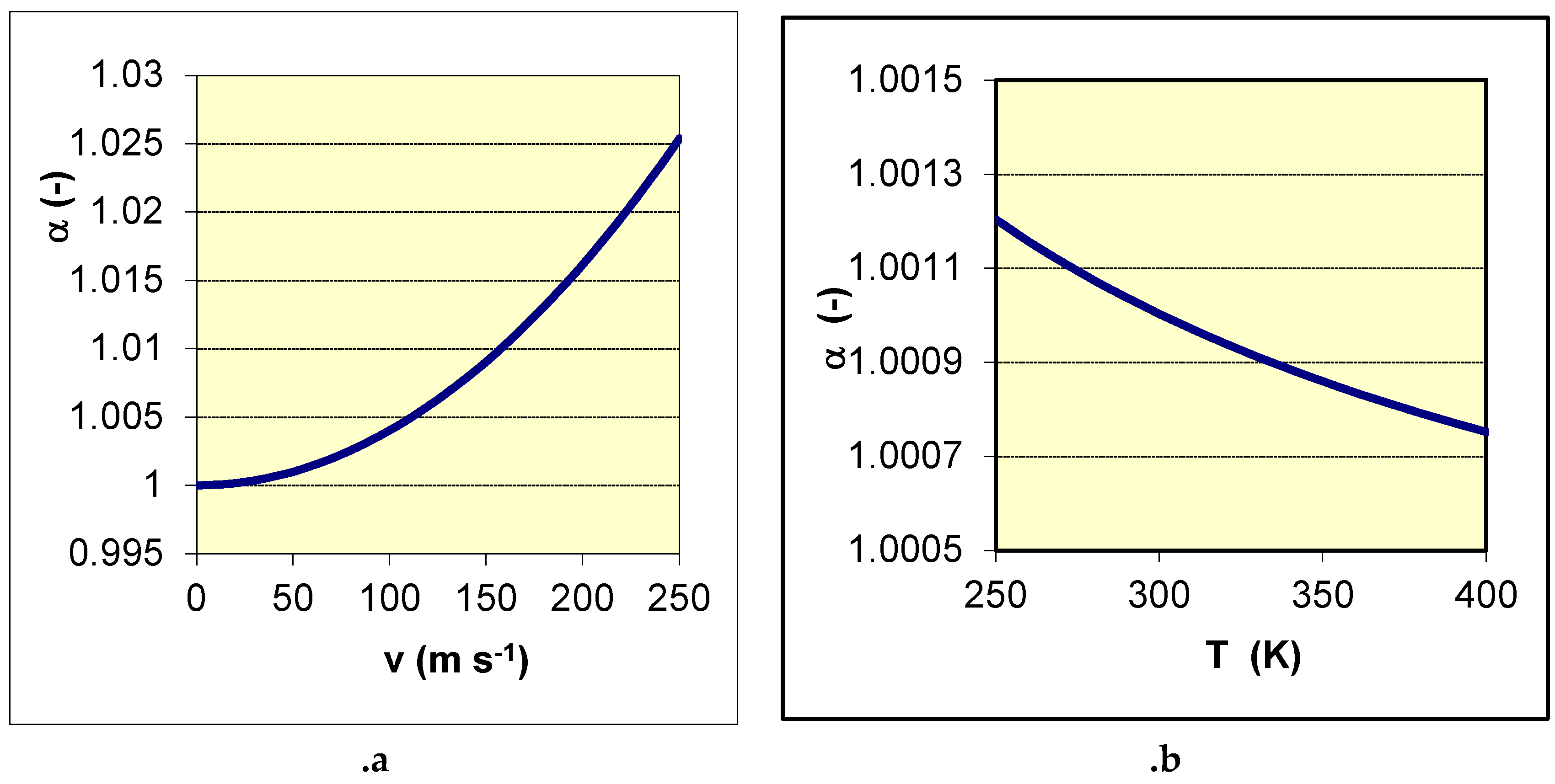
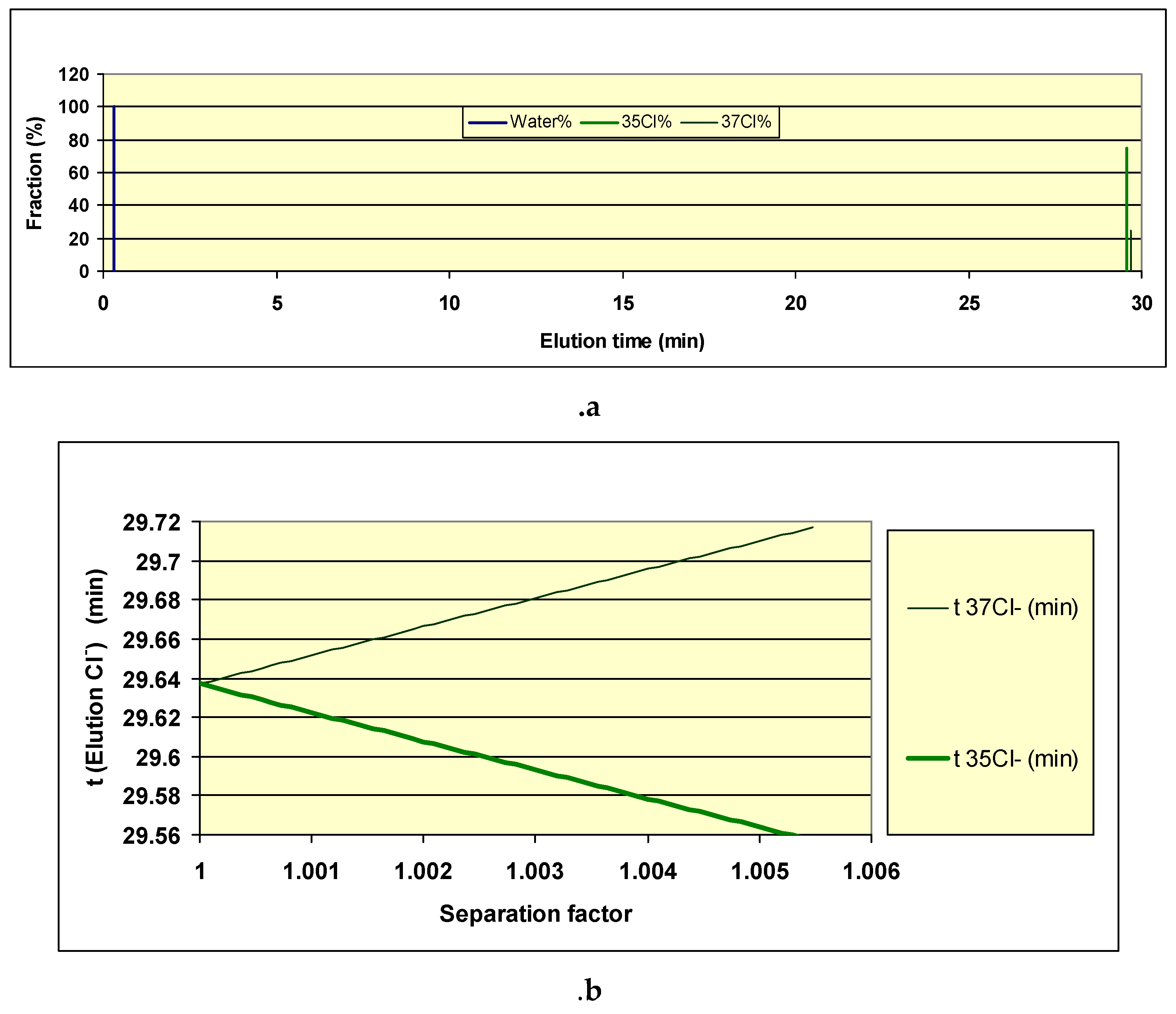
| 35Cl (%) | 37Cl (%) | RG (pcm) |
|---|---|---|
| 75.8 | 24.2 | 0000 |
| 05.0 | 95.0 | 4118 |
| 01.0 | 99.0 | 4365 |
| 00.1 | 99.9 | 4421 |
| Separation | Conditions | α | Ref |
|---|---|---|---|
| Centrifugation | T: 300 K | 1.0020 | This work |
| Distillation | T: 300 K | 1.0060 | Liebscher et al. (2006) |
| Chromatography | T: 300 K | 1.0003 | Musashi et al. (2007) |
| Electrolysis | T: 300 K Pt | 1.0060 | Johnston & Hutchison (1942) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




