Submitted:
02 August 2024
Posted:
03 August 2024
You are already at the latest version
Abstract
Keywords:
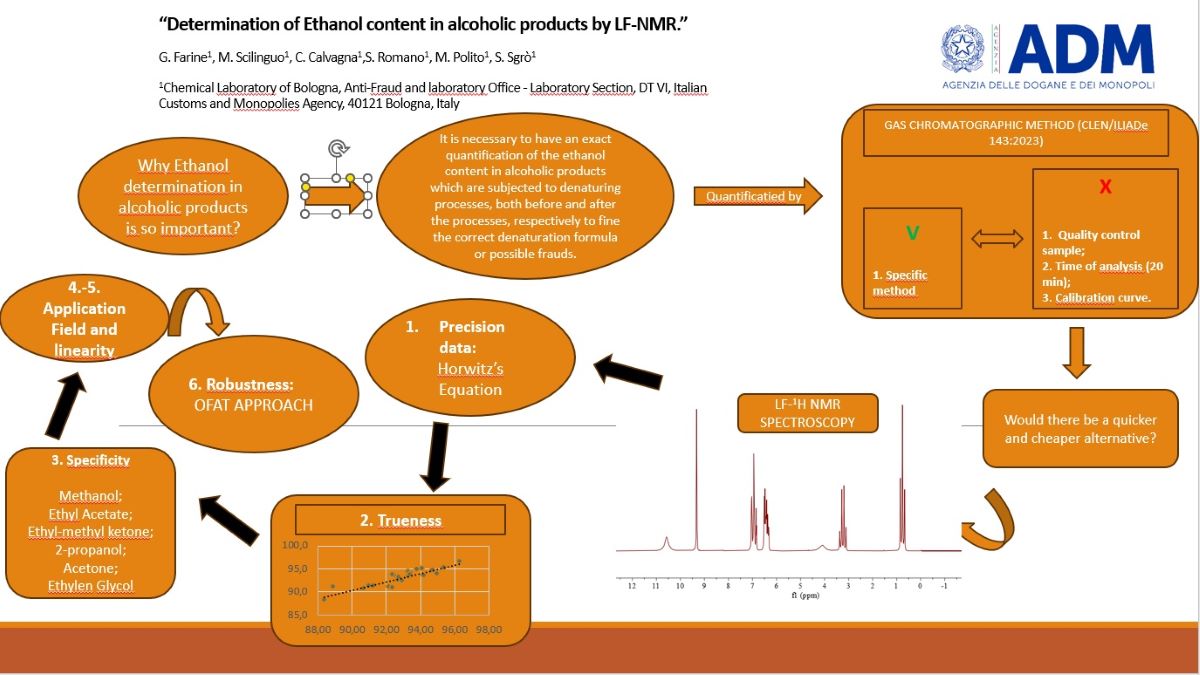
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Chemical Analysis
2.2.1. Cromatographic Method
2.2.2. Densimetric Method
2.2.3. Spectroscopic (LF-NMR) Method
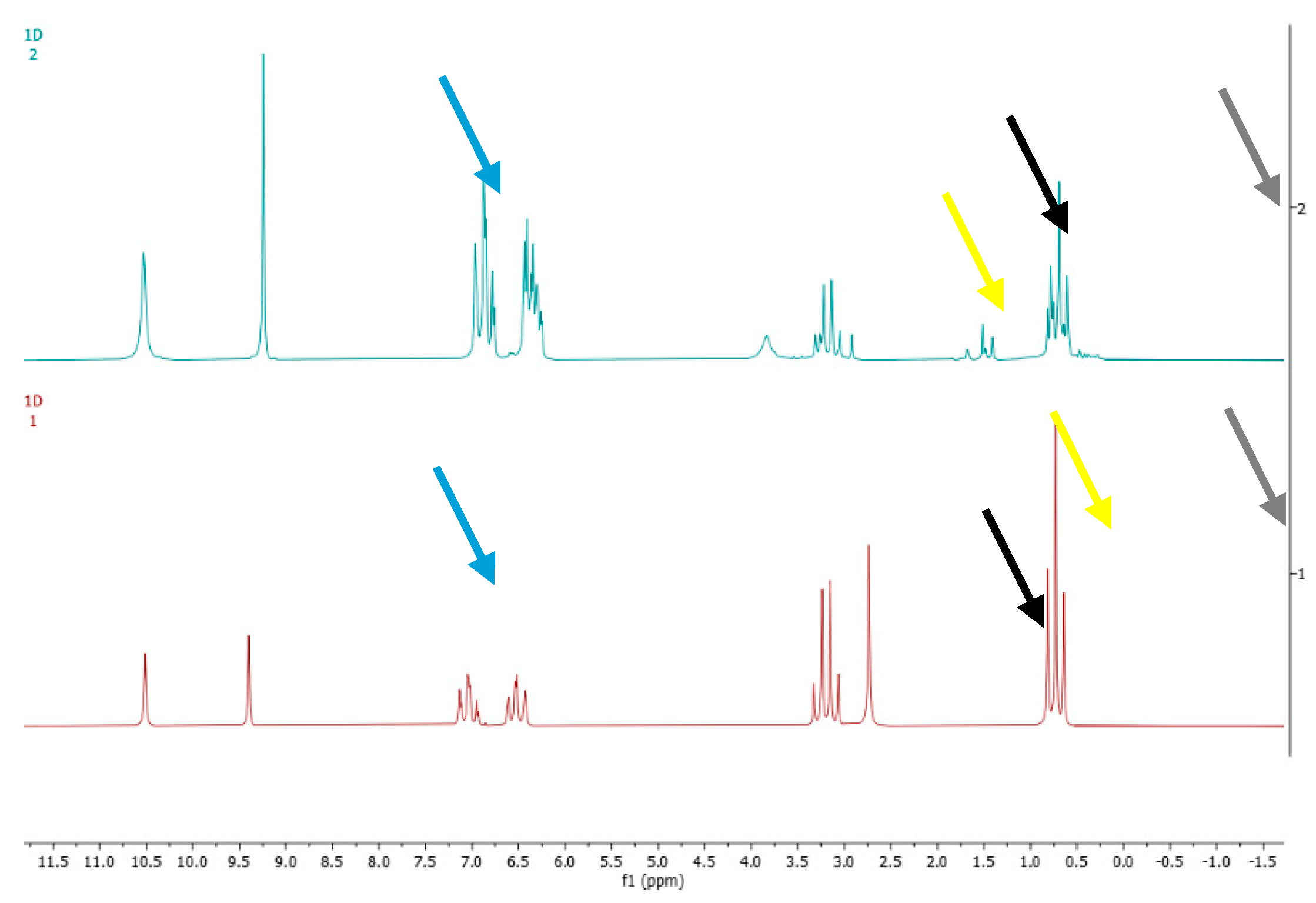
3. Results and Discussion
- Precision data;
- Trueness;
- Specificity;
- Application field;
- Linearity;
- Robustness.
3.1. Precision Data (Repeatabilty and Uncertainty of Measurement)
3.2. Trueness
- Comparison with a PT sample (interlaboratory circuit residual);
3.2.1. Comparison with a PT Sample
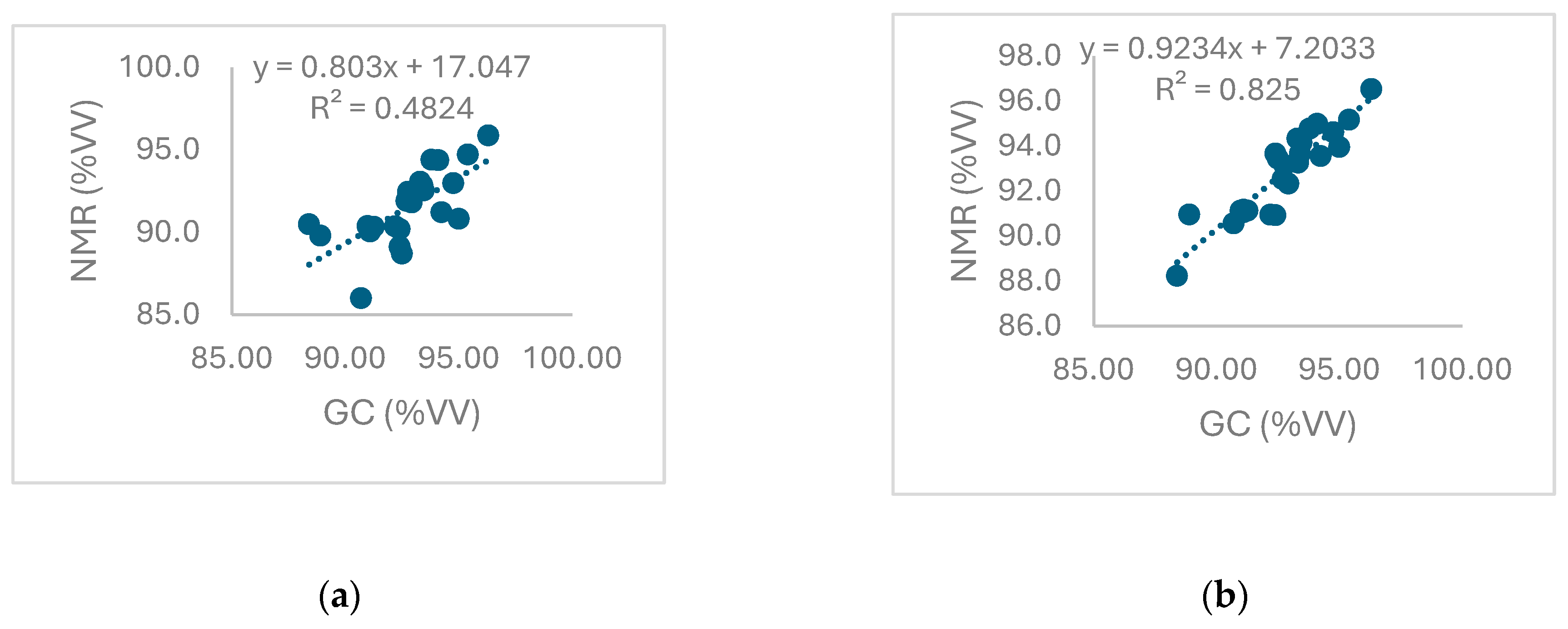
3.2.2. Comparison between Results Obtained by NMR Spectroscopic and Reference Cromatographic Methods
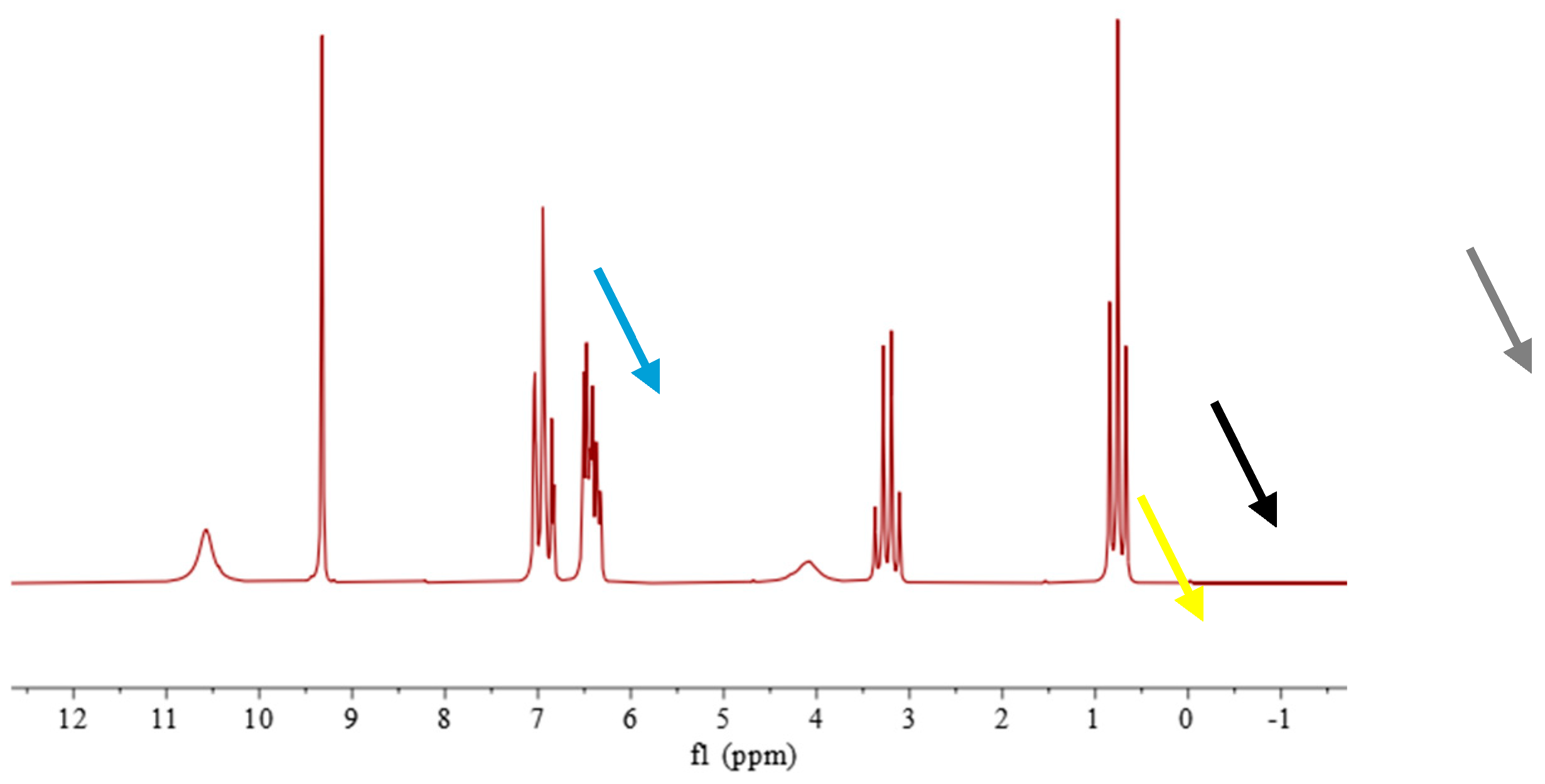
3.3. Specificity
- ethanol standard solution containing all the 14 denaturants mentioned above;
- ethanol standard solution containing the two most common volatile substances generally found in crude alcohol: methanol and ethyl acetate;
- ethanol standard solutions containing, singularly, common or critical denaturants as received by our laboratory.
3.3.1. Analysis of Ethanol Standard Solution Containing 14 Denaturants
3.3.2. Analysis of Ethanol Standard Solution Containing Methanol and Ethyl Acetate
3.3.3. Analysis of Ethanol Standard Solutions Containing, Singularly, Common or Critical Denaturants
3.4. Application Field
3.5. Linearity
3.6. Robustness
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Council Directive 92/83/EEC of 19 October 1992 on the harmonization of the structures of excise duties on alcohol and alcoholic beverages, OJ L 316, 31.10.1992, p. 21–27 (ES, DA, DE, EL, EN, FR, IT, NL, PT).
- COMMISSION REGULATION (EC) No 3199/93 of 22 November 1993 on the mutual recognition of procedures for the complete denaturing of alcohol for the purposes of exemption from excise duty.
- I.W. Burton, M. A. Quilliam, and J. A. Walter, “Quantitative 1H NMRwith external standards: use in preparation of calibrationsolutions for algal toxins and other natural products,” Analytical Chemistry, vol. 77, no. 10, pp. 3123–3131, 2005. [CrossRef]
- S. K. Bharti and R. Roy, “Quantitative 1H NMR spectroscopy,” TrAC Trends in Analytical Chemistry, vol. 35, pp. 5–26, 2012.
- H.-S. Son, M. K. Kim, F. Van den Berg et al., “ 1H nuclearmagnetic resonance-based metabolomic characterization of wines by grape varieties and production areas,” Journal of Agricultural and Food Chemistry, vol. 56, no. 17, pp. 8007–8016, 2008. [CrossRef]
- G. F. Pauli, T. G¨odecke, B. U. Jaki, and D. C. Lankin, “Quantitative 1H NMR. Development and potential of an analytical method: an update,” Journal of Natural Products, vol. 75, no. 4, pp. 834–851, 2012. [CrossRef]
- J. C. Edwards, J. M. Hunter, and B. V. Nemzer, “Multinuclear NMR of calcium fructoborate complex—structure, stability, and quantitation in the presence of other ingredients, excipients or adulterants,” Journal of Food Research, vol. 3, no. 3, pp. 115–131, 2014. [CrossRef]
- G.Ramtahal, I. C. Yen, I. Bekele et al., “Cost-effective method of analysis for the determination of cadmium, copper, nickel and zinc in cocoa beans and chocolates,” Journal of Food Research, vol. 4, no. 1, pp. 193–199, 2014. [CrossRef]
- P. K. Bowyer, “The measurement of alcohol levels in wine,” in The AustralianGrapegrower andWinemaker, pp. 90–96,AnnualTechnical Issue, 2006.
- P. Maes, Y. B. Monakhova, T. Kuballa, H. Reusch, and D. W. Lachenmeier, “Qualitative and quantitative control of carbonated cola beverages using 1H NMR spectroscopy,” Journal of Agricultural and Food Chemistry, vol. 60, no. 11, pp. 2778–2784,2012. [CrossRef]
- B. Lorrain, I. Ky, L. Pechamat, and P.-L. Teissedre, “Evolution of analysis of polyphenols from grapes, wines, and extracts,”Molecules, vol. 18, no. 1, pp. 1076–1100, 2013. [CrossRef]
- ILIADe 143:2023| CLEN Method “Determination of Ethanol in Alcoholic Products by GC-FID” Version 25 July 2023.
- EA guidelines on the expression of uncertainty in quantitative testing, European co-operation for Accreditation, EA-4/16.
- UNICHIM Manuals n. 179/0, 179/1, 179/2 2011 Edition “Guidelines for the validation of analytical methods in chemical laboratories”.
- Decreto legge del 30/08/1993 n.331 “Armonizzazione delle disposizioni in materia di imposte sugli oli minerali, sull’alcole, sulle bevande alcoliche, sui tabacchi lavorati e in materia di IVA con quelle recate da direttive CEE e modificazioni conseguenti a detta armonizzazione, nonche’ disposizioni concernenti la disciplina dei centri autorizzati di assistenza fiscale, le procedure dei rimborsi di imposta, l’esclusione dall’ILOR dei redditi di impresa fino all’ammontare corrispondente al contributo diretto lavorativo, l’istituzione per il 1993 di un’imposta erariale straordinaria su taluni beni ed altre disposizioni tributarie”; Gazzetta Ufficiale n. 203 del 30/08/1993 null n. 427 del 29/10/1993.
- Decreto ministeriale 9/07/1996, n. 524 , “Regolamento recante norme per disciplinare l’impiego dell’alcole etilico e delle bevande alcoliche in usi esenti da accisa”, (GU Serie Generale n.237 del 09-10-1996).
- COMMISSION IMPLEMENTING REGULATION (EU) 2017/2236 of 5 December 2017 amending Regulation (EC) No 3199/93 on the mutual recognition of procedures for the complete denaturing of alcohol for the purposes of exemption from excise duty.

| Matrix | EtOH content determined by GC-FID |
|---|---|
| Anhydrous ethyl alcohol | EtOH: 99.9 % v/v |
| Denatured ethyl alcohol (euro DG) | EtOH: 88.5 % v/v |
| Crude alcohol | EtOH: 92.7 % v/v |
| Hydroalcoholic mixture ethyl alcohol/water 80/20 %v/v | EtOH: 82.7 % v/v |
| Matrix | rLAB (%v/v) -CH3 signal |
rLAB (%v/v) -CH2 signal |
UH (%v/v) |
|---|---|---|---|
| Anhydrous ethyl alcohol | 1.64 | 0.74 | 2.00 |
| Denatured ethyl alcohol (euro DG) | 1.84 | ||
| Crude alcohol | 1.90 | ||
| Hydroalcoholic mixture ethyl alcohol/water 80/20 %v/v | 1.76 |
| Matrix | X (%v/v) |
Xpt (%v/v) |
spt | Z-score | Acceptability (-2 ≤ z ≤ +2) |
|---|---|---|---|---|---|
| PT CLEN 2019-Burning Alcohol (-CH3) – Repetition 1 | 86.76 | 89.62 |
0.65 | -4.40 | NO |
| PT CLEN 2019-Burning Alcohol (-CH3) – Repetition 2 | 87.61 | -3.09 | NO | ||
| PT CLEN 2019-Burning Alcohol (-CH3) – Repetition 3 | 86.31 | -5.09 | NO | ||
| PT CLEN 2019-Burning Alcohol (-CH2) – Repetition 1 | 89.25 | -0.57 | YES | ||
| PT CLEN 2019-Burning Alcohol (-CH2) – Repetition 2 | 90.04 | 0.65 | YES | ||
| PT CLEN 2019-Burning Alcohol (-CH2) – Repetition 3 | 88.78 | -1.30 | YES |
|
-CH3 signal |
-CH3 signal |
-CH3 signal |
Acceptability |
-CH2 signal |
-CH2 signal |
-CH2 signal |
Acceptability | ||
|---|---|---|---|---|---|---|---|---|---|
| Anhydrous ethyl alcohol | 100.88 | 99.49 | 1.39 | 0.16 | NO | 100.03 | 0.85 | 0.14 | NO |
| Denatured ethyl alcohol (euro DG) | 88.08 | 89.07 | 1.00 | 0.26 | NO | 88.28 | 0.20 | 0.40 | YES |
| Crude alcohol | 92.73 | 92.76 | 0.03 | 0.56 | YES | 92.92 | 0.19 | 0.65 | YES |
| Hydroalcoholic mixture ethyl alcohol/water 80/20 %v/v | 82.72 | 81.32 | 1.40 | 0.25 | NO | 82.99 | 0.26 | 0.33 | YES |
| CH3 |
Acceptability | CH2 |
Acceptability | |
|---|---|---|---|---|
| Anhydrous ethyl alcohol - CRM | 0.47 | YES | 0.11 | YES |
| Equation -CH3 signal |
a (intercept) ± s(a) -CH3 signal |
b (slope) ± s(b) -CH3 signal |
Equation -CH2 signal |
a (intercept) ± s(a) -CH2 signal |
b (slope) ± s(b) -CH2 signal |
|---|---|---|---|---|---|
| y = 0.803x + 17.047 | 17.0±16.1 | 0.80±0.17 | y = 0.9234x + 7.2033 | 7.20±8.22 | 0.92±0.09 |
| Cexperimental -CH3 signal |
Cexperimental -CH2 signal |
Ctheoretical | Recovery -CH3 signal |
Recovery -CH2 signal |
|
|---|---|---|---|---|---|
| Ethanol standard solution containing 14 denaturants | 98.65w/w % | 103.41w/w % | 54.99 w/w % | 179.39 % | 188.06% |
| Cexperimental -CH3 signal |
Cexperimental -CH2 signal |
Ctheoretical | Recovery -CH3 signal |
Recovery -CH2 signal |
|
|---|---|---|---|---|---|
|
Ethanol std + methanol & ethyl acetate |
83.13 %w/w | 81.31%w/w | 80.26 %w/w | 103.58% | 101.31% |
| Cexperimental -CH3 signal |
Cexperimental -CH2 signal |
Ctheoretical | Recovery -CH3 signal |
Recovery -CH2 signal |
|
|---|---|---|---|---|---|
| Ethanol std + acetone | 88.52 | 91.43 | 90.10 | 98.25 | 101.47 |
| Ethanol std + MEK | 89.99 | 90.53 | 89.99 | 100.60 | 101.75 |
| Ethanol std + 2-propanol | 103.69 | 92.69 | 90.25 | 114.89 | 102.70 |
| Ethanol std + ethylen glycol | 85.67 | 83.41 | 85.67 | 97.37 | 124.80 |
| Ethanol std + n-propanol | 89.54 | 90.95 | 89.54 | 101.58 | 109.98 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




