Submitted:
09 January 2025
Posted:
10 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Heat Shock Proteins, Chaperones and Chaperonins
3. Structure of the Cardiac Muscle Cell and Role of Mitochondria
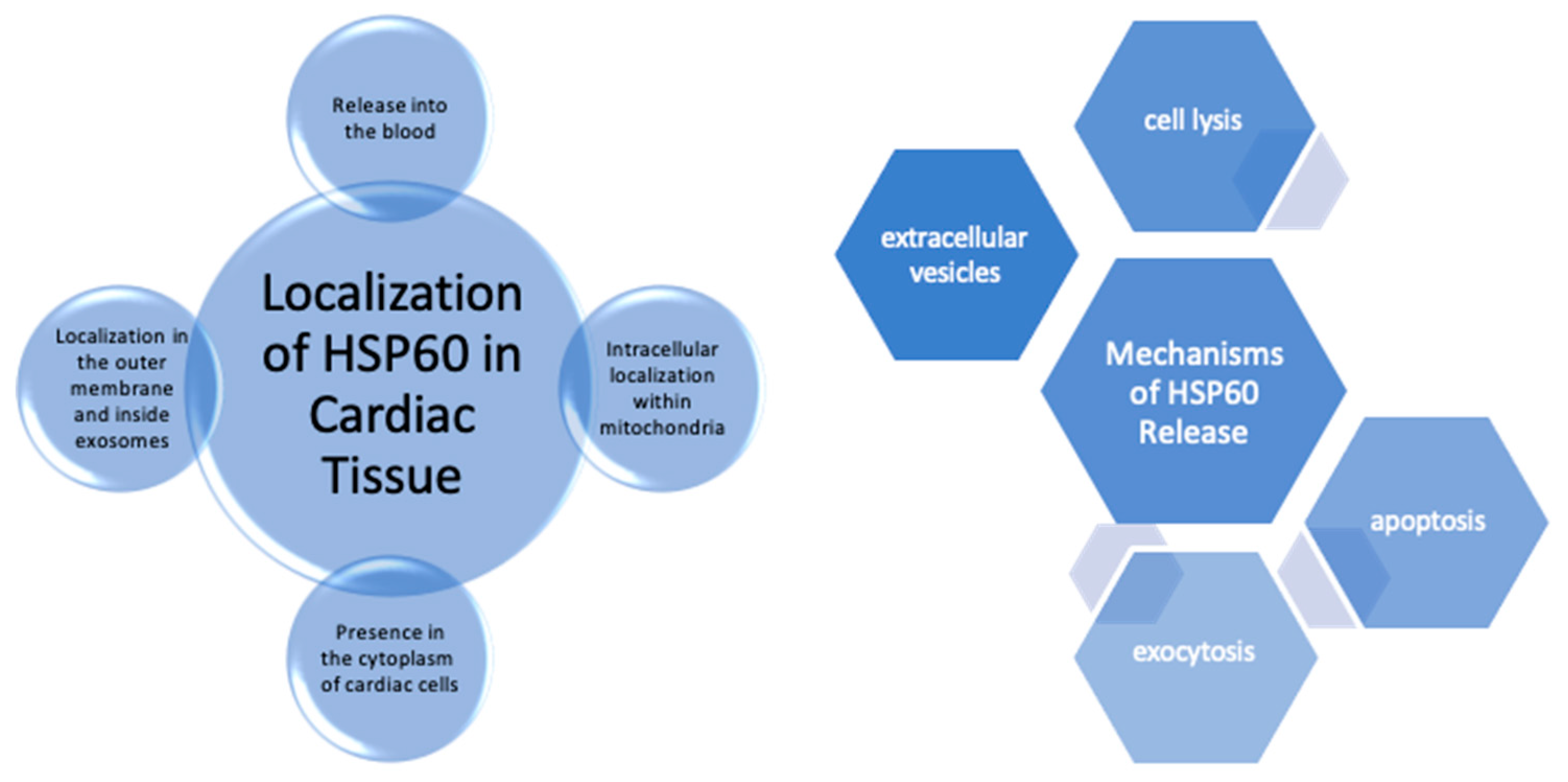
4. Hsp60 Expression and Localization in the Healthy Heart Tissue
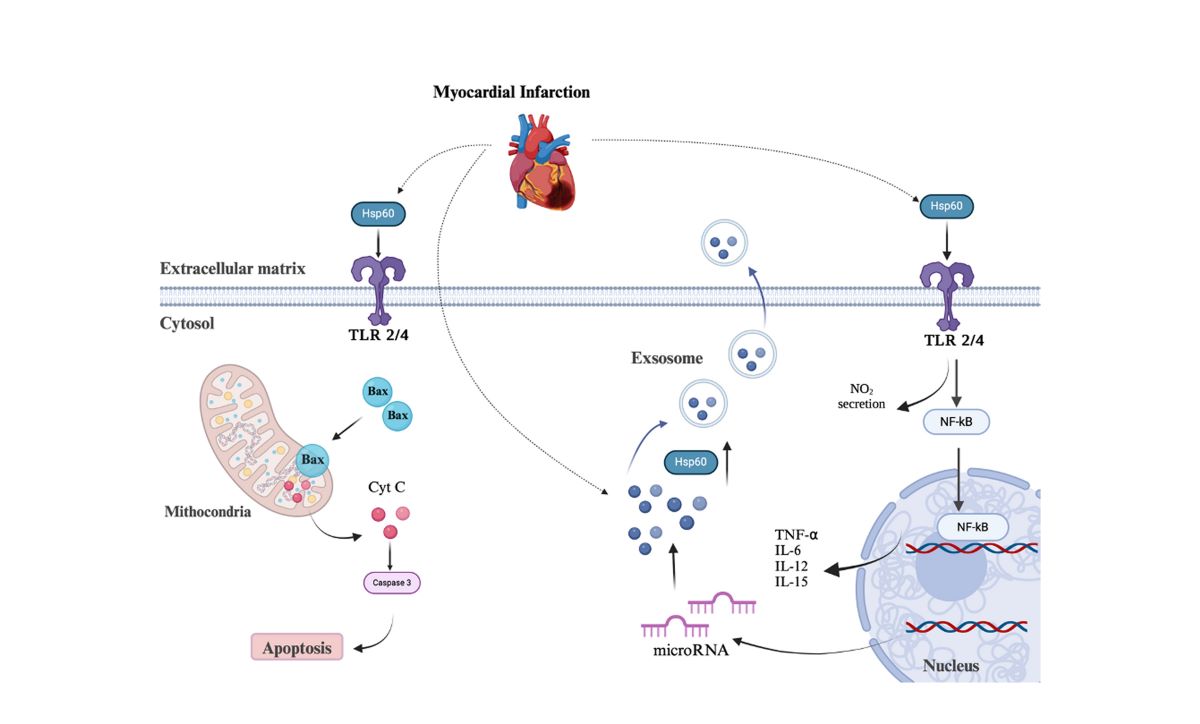
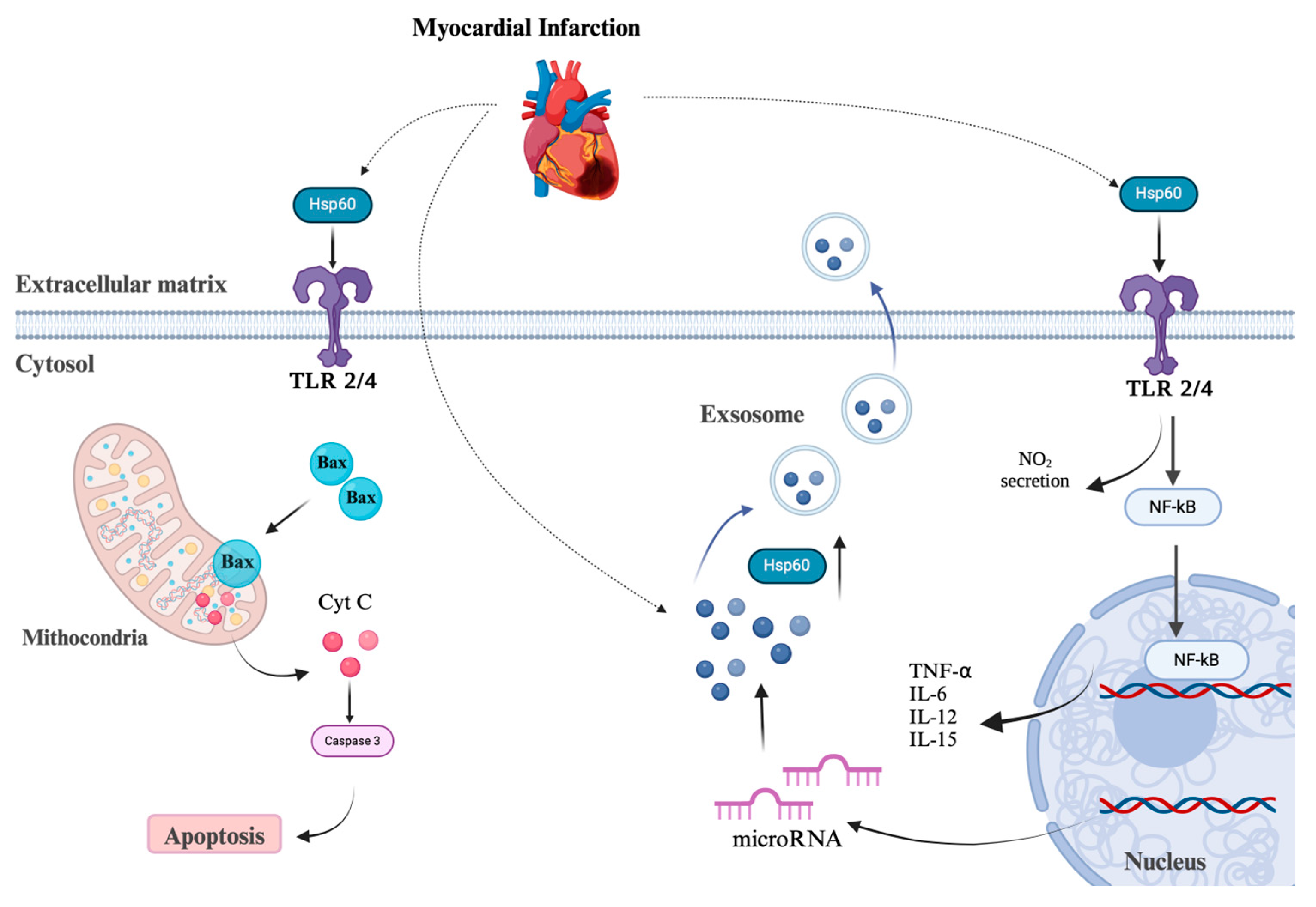
5. Mitochondrial Dysfunction in Cardiac Diseases: The Role of HSP60 Release and Its Protective Function
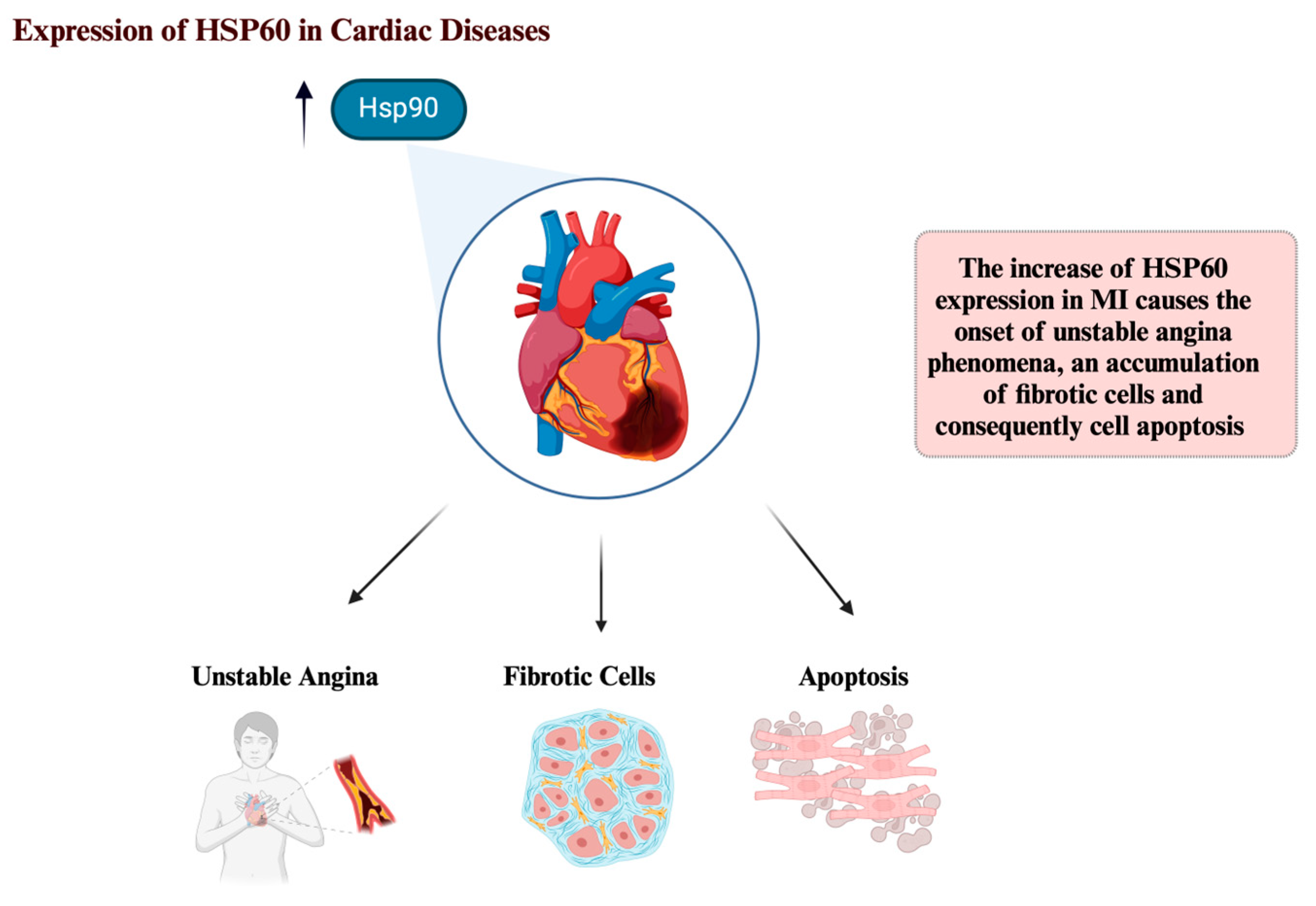
6. Hsp60 and Cardiac Diseases
7. Conclusions
Author Contributions
Funding
Abbreviations
| Adenosine triphosphate | ATP |
| Oxidative phosphorylation | OXPHOS |
| Electron transport chain | ETC |
| Mitochondrial antioxidant manganese superoxide dismutase | MnSOD, SOD2 |
| Endoplasmic reticulum | ER |
| Inner mitochondrial membrane | IMM |
| Toll-like receptors | TLRs |
| Damage Associated Molecular Pattern | DAMP |
| Nitric oxide | NO |
| Interleukin | IL |
| Major histocompatibility complex | MHC |
| Antigen-presenting cells | APCs |
| Myocardial infarction | MI |
| Atrial fibrillation | AF |
| Left ventricle | LV |
| Acute myocardial infarction | AMI |
| Cardiovascular diseases | CVDs |
| Dilated cardiomyopathy | DCM |
| Chronic Heart Failure | CHF |
| Embryonic days | ED |
| unfolded mitochondrial protein response | UPRmt |
| ischaemia/reperfusion | I/R |
References
- Tzameli, I. The evolving role of mitochondria in metabolism. Trends in Endocrinology & Metabolism. 2012, 23, 417–419. [Google Scholar]
- Spinelli JB, Haigis MC. The multifaceted contributions of mitochondria to cellular metabolism. Nature cell biology. 2018, 20, 745–54. [Google Scholar] [CrossRef]
- Bock FJ, Tait SW. Mitochondria as multifaceted regulators of cell death. Nature reviews Molecular cell biology. 2020, 21, 85–100. [Google Scholar] [CrossRef] [PubMed]
- de Souza Breda CN, Davanzo GG, Basso PJ, Câmara NOS, Moraes-Vieira PMM. Mitochondria as central hub of the immune system. Redox biology. 2019;26:101255.
- Papa L, Djedaini M, Hoffman R. Mitochondrial role in stemness and differentiation of hematopoietic stem cells. Stem cells international. 2019;2019(1):4067162.
- Murphy E, Ardehali H, Balaban RS, DiLisa F, Dorn GW, Kitsis RN, et al. Mitochondrial function, biology, and role in disease: a scientific statement from the American Heart Association. Circulation research. 2016, 118, 1960–91. [Google Scholar] [CrossRef]
- Wang X, Zhang X, Wu D, Huang Z, Hou T, Jian C, et al. Mitochondrial flashes regulate ATP homeostasis in the heart. Elife. 2017;6:e23908.
- Duan Y, Tang H, Mitchell-Silbaugh K, Fang X, Han Z, Ouyang K. Heat Shock Protein 60 in Cardiovascular Physiology and Diseases. Front Mol Biosci. 2020;7:73.
- Yan J, Bao E, Yu J. Heat shock protein 60 expression in heart, liver and kidney of broilers exposed to high temperature. Research in Veterinary Science. 2009;86(3):533-8.
- Hartl FU, Bracher A, Hayer-Hartl M. Molecular chaperones in protein folding and proteostasis. Nature. 2011;475(7356):324-32.
- Kampinga HH, Hageman J, Vos MJ, Kubota H, Tanguay RM, Bruford EA, et al. Guidelines for the nomenclature of the human heat shock proteins. Cell Stress and Chaperones. 2009;14(1):105-11.
- Han D, Huang SS, Wang W-F, Deng D-F, Hung SS. Starvation reduces the heat shock protein responses in white sturgeon larvae. Environmental biology of fishes. 2012;93:333-42.
- Michaud MR, Teets NM, Peyton JT, Blobner BM, Denlinger DL. Heat shock response to hypoxia and its attenuation during recovery in the flesh fly, Sarcophaga crassipalpis. Journal of insect physiology. 2011;57(1):203-10.
- Bukau B, Horwich AL. The Hsp70 and Hsp60 chaperone machines. Cell. 1998;92(3):351-66.
- Hartl FU, Hayer-Hartl M. Converging concepts of protein folding in vitro and in vivo. Nature structural & molecular biology. 2009;16(6):574-81.
- Feldman DE, Frydman J. Protein folding in vivo: the importance of molecular chaperones. Current opinion in structural biology. 2000;10(1):26-33.
- Braig, K. Chaperonins. Current opinion in structural biology. 1998;8(2):159-65.
- Fink, AL. Chaperone-mediated protein folding. Physiological reviews. 1999;79(2):425-49.
- Agashe VR, Hartl F-U, editors. Roles of molecular chaperones in cytoplasmic protein folding. Seminars in cell & developmental biology; 2000: Elsevier.
- Hartl FU, Hayer-Hartl M. Molecular chaperones in the cytosol: from nascent chain to folded protein. Science. 2002;295(5561):1852-8.
- Duan Y, Tang H, Mitchell-Silbaugh K, Fang X, Han Z, Ouyang K. Heat shock protein 60 in cardiovascular physiology and diseases. Frontiers in molecular biosciences. 2020;7:73.
- Cheng MY, Hartl F-U, Martin J, Pollock RA, Kalousek F, Neuper W, et al. Mitochondrial heat-shock protein hsp60 is essential for assembly of proteins imported into yeast mitochondria. Nature. 1989;337(6208):620-5.
- Ostermann J, Horwich AL, Neupert W, Hartl F-U. Protein folding in mitochondria requires complex formation with hsp60 and ATP hydrolysis. Nature. 1989;341(6238):125-30.
- Ishida R, Okamoto T, Motojima F, Kubota H, Takahashi H, Tanabe M, et al. Physicochemical properties of the mammalian molecular chaperone HSP60. International Journal of Molecular Sciences. 2018;19(2):489.
- Gomez-Llorente Y, Jebara F, Patra M, Malik R, Nisemblat S, Chomsky-Hecht O, et al. Structural basis for active single and double ring complexes in human mitochondrial Hsp60-Hsp10 chaperonin. Nature Communications. 2020;11(1):1916.
- Bie AS, Cömert C, Körner R, Corydon TJ, Palmfeldt J, Hipp MS, et al. An inventory of interactors of the human HSP60/HSP10 chaperonin in the mitochondrial matrix space. Cell Stress Chaperones. 2020;25(3):407-16.
- Kao TY, Chiu YC, Fang WC, Cheng CW, Kuo CY, Juan HF, et al. Mitochondrial Lon regulates apoptosis through the association with Hsp60-mtHsp70 complex. Cell Death Dis. 2015;6(2):e1642.
- Meng Q, Li BX, Xiao X. Toward developing chemical modulators of Hsp60 as potential therapeutics. Frontiers in molecular biosciences. 2018;5:35.
- Henderson B, Fares MA, Lund PA. Chaperonin 60: a paradoxical, evolutionarily conserved protein family with multiple moonlighting functions. Biological Reviews. 2013;88(4):955-87.
- Gupta S, Knowlton AA. HSP60 trafficking in adult cardiac myocytes: role of the exosomal pathway. American Journal of Physiology-Heart and Circulatory Physiology. 2007;292(6):H3052-H6.
- Lin L, Kim S-C, Wang Y, Gupta S, Davis B, Simon SI, et al. HSP60 in heart failure: abnormal distribution and role in cardiac myocyte apoptosis. American Journal of Physiology-Heart and Circulatory Physiology. 2007;293(4):H2238-H47.
- Pockley AG, Wu R, Lemne C, Kiessling R, de Faire U, Frostegård J. Circulating heat shock protein 60 is associated with early cardiovascular disease. Hypertension. 2000;36(2):303-7.
- Lewthwaite J, Owen N, Coates A, Henderson B, Steptoe A. Circulating human heat shock protein 60 in the plasma of British civil servants: relationship to physiological and psychosocial stress. Circulation. 2002;106(2):196-201.
- Giannessi D, Colotti C, Maltinti M, Del Ry S, Prontera C, Turchi S, et al. Circulating heat shock proteins and inflammatory markers in patients with idiopathic left ventricular dysfunction: their relationships with myocardial and microvascular impairment. Cell stress & chaperones. 2007;12(3):265.
- Blasi C, Kim E, Knowlton AA. Improved metabolic control in diabetes, HSP60, and proinflammatory mediators. Autoimmune Diseases. 2012;2012(1):346501.
- Wang X, Zhang X, Wu D, Huang Z, Hou T, Jian C, et al. Mitochondrial flashes regulate ATP homeostasis in the heart. Elife. 2017;6.
- Nguyen BY, Ruiz-Velasco A, Bui T, Collins L, Wang X, Liu W. Mitochondrial function in the heart: the insight into mechanisms and therapeutic potentials. Br J Pharmacol. 2019;176(22):4302-18.
- Bhattacharyya A, Chattopadhyay R, Mitra S, Crowe SE. Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol Rev. 2014;94(2):329-54.
- Singh, H. Mitochondrial ion channels in cardiac function. Am J Physiol Cell Physiol. 2021;321(5):C812-c25.
- Paradies G, Paradies V, Ruggiero FM, Petrosillo G. Role of Cardiolipin in Mitochondrial Function and Dynamics in Health and Disease: Molecular and Pharmacological Aspects. Cells. 2019;8(7).
- Murphy E, Ardehali H, Balaban RS, DiLisa F, Dorn GW, 2nd, Kitsis RN, et al. Mitochondrial Function, Biology, and Role in Disease: A Scientific Statement From the American Heart Association. Circ Res. 2016;118(12):1960-91.
- Giannessi D, Colotti C, Maltinti M, Del Ry S, Prontera C, Turchi S, et al. Circulating heat shock proteins and inflammatory markers in patients with idiopathic left ventricular dysfunction: their relationships with myocardial and microvascular impairment. Cell Stress Chaperones. 2007;12(3):265-74.
- Gupta S, Knowlton AA. HSP60 trafficking in adult cardiac myocytes: role of the exosomal pathway. Am J Physiol Heart Circ Physiol. 2007;292(6):H3052-6.
- Pockley AG, Wu R, Lemne C, Kiessling R, de Faire U, Frostegård J. Circulating heat shock protein 60 is associated with early cardiovascular disease. Hypertension. 2000;36(2):303-7.
- Cheng Y, Sun J, Chen H, Adam A, Tang S, Kemper N, et al. Expression and location of HSP60 and HSP10 in the heart tissue of heat-stressed rats. Exp Ther Med. 2016;12(4):2759-65.
- Di Felice V, Barone R, Trovato E, D'Amico D, Macaluso F, Campanella C, et al. Physiactisome: A New Nanovesicle Drug Containing Heat Shock Protein 60 for Treating Muscle Wasting and Cachexia. Cells. 2022;11(9).
- Osterloh A, Meier-Stiegen F, Veit A, Fleischer B, von Bonin A, Breloer M. Lipopolysaccharide-free heat shock protein 60 activates T cells. J Biol Chem. 2004;279(46):47906-11.
- Pockley AG, Muthana M, Calderwood SK. The dual immunoregulatory roles of stress proteins. Trends Biochem Sci. 2008;33(2):71-9.
- Swaroop S, Sengupta N, Suryawanshi AR, Adlakha YK, Basu A. HSP60 plays a regulatory role in IL-1β-induced microglial inflammation via TLR4-p38 MAPK axis. J Neuroinflammation. 2016;13:27.
- Boyd JH, Mathur S, Wang Y, Bateman RM, Walley KR. Toll-like receptor stimulation in cardiomyoctes decreases contractility and initiates an NF-kappaB dependent inflammatory response. Cardiovasc Res. 2006;72(3):384-93.
- Duan Y, Tang H, Mitchell-silbaugh K, Fang X, Han Z, Ouyang K. Heat Shock Protein 60 in Cardiovascular Physiology and Diseases. Frontiers in Molecular Biosciences. 2020;7.
- Krishnan-Sivadoss I, Mijares-Rojas IA, Villarreal-Leal RA, Torre-Amione G, Knowlton AA, Guerrero-Beltrán CE. Heat shock protein 60 and cardiovascular diseases: An intricate love-hate story. Med Res Rev. 2021;41(1):29-71.
- Rocca C, Soda T, De Francesco EM, Fiorillo M, Moccia F, Viglietto G, et al. Mitochondrial dysfunction at the crossroad of cardiovascular diseases and cancer. Journal of Translational Medicine. 2023;21(1):635.
- Zhang X, He M, Cheng L, Chen Y, Zhou L, Zeng H, et al. Elevated heat shock protein 60 levels are associated with higher risk of coronary heart disease in Chinese. Circulation. 2008;118(25):2687-93.
- Yang L, Wang B, Zhou Q, Wang Y, Liu X, Liu Z, et al. MicroRNA-21 prevents excessive inflammation and cardiac dysfunction after myocardial infarction through targeting KBTBD7. Cell Death Dis. 2018;9(7):769.
- Hu Y, Chen X, Li X, Li Z, Diao H, Liu L, et al. MicroRNA-1 downregulation induced by carvedilol protects cardiomyocytes against apoptosis by targeting heat shock protein 60. Molecular medicine reports. 2019;19(5):3527-36.
- Cohen-Sfady M, Nussbaum G, Pevsner-Fischer M, Mor F, Carmi P, Zanin-Zhorov A, et al. Heat shock protein 60 activates B cells via the TLR4-MyD88 pathway. The Journal of Immunology. 2005;175(6):3594-602.
- Zanin-Zhorov A, Cahalon L, Tal G, Margalit R, Lider O, Cohen IR. Heat shock protein 60 enhances CD4+ CD25+ regulatory T cell function via innate TLR2 signaling. The Journal of clinical investigation. 2006;116(7):2022-32.
- Kol A, Lichtman AH, Finberg RW, Libby P, Kurt-Jones EA. Cutting edge: heat shock protein (HSP) 60 activates the innate immune response: CD14 is an essential receptor for HSP60 activation of mononuclear cells. The Journal of Immunology. 2000;164(1):13-7.
- Ohashi K, Burkart V, Flohé S, Kolb H. Cutting edge: heat shock protein 60 is a putative endogenous ligand of the toll-like receptor-4 complex. The Journal of Immunology. 2000;164(2):558-61.
- Hu Y, Chen X, Li X, Li Z, Diao H, Liu L, et al. MicroRNA-1 downregulation induced by carvedilol protects cardiomyocytes against apoptosis by targeting heat shock protein 60. Mol Med Rep. 2019;19(5):3527-36.
- Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation. 2019;139(10):e56-e528.
- Farmakis D, Parissis J, Filippatos G. Acute heart failure: epidemiology, classification, and pathophysiology. The ESC textbook of intensive and acute cardiovascular care, 2nd ed Oxford. 2015:459-69.
- Komajda M, Anker SD, Cowie MR, Filippatos GS, Mengelle B, Ponikowski P, et al. Physicians' adherence to guideline-recommended medications in heart failure with reduced ejection fraction: data from the QUALIFY global survey. European journal of heart failure. 2016;18(5):514-22.
- Santhanakrishnan R, Wang N, Larson MG, Magnani JW, McManus DD, Lubitz SA, et al. Atrial fibrillation begets heart failure and vice versa: temporal associations and differences in preserved versus reduced ejection fraction. Circulation. 2016;133(5):484-92.
- Henning RH, Brundel BJ. Proteostasis in cardiac health and disease. Nature Reviews Cardiology. 2017;14(11):637-53.
- Brundel BJ, Shiroshita-Takeshita A, Qi X, Yeh Y-H, Chartier D, Van Gelder IC, et al. Induction of heat shock response protects the heart against atrial fibrillation. Circulation research. 2006;99(12):1394-402.
- Wiersma M, Meijering RA, Qi XY, Zhang D, Liu T, Hoogstra-Berends F, et al. Endoplasmic reticulum stress is associated with autophagy and cardiomyocyte remodeling in experimental and human atrial fibrillation. Journal of the American Heart Association. 2017;6(10):e006458.
- Kirmanoglou K, Hannekum A, Schäfler AE. Expression of mortalin in patients with chronic atrial fibrillation. Basic research in cardiology. 2004;99:404-8.
- Yang M, Tan H, Cheng L, He M, Wei Q, Tanguay RM, et al. Expression of heat shock proteins in myocardium of patients with atrial fibrillation. Cell stress & chaperones. 2007;12(2):142.
- Kim SC, Stice JP, Chen L, Jung JS, Gupta S, Wang Y, et al. Extracellular heat shock protein 60, cardiac myocytes, and apoptosis. Circ Res. 2009;105(12):1186-95.
- Lin L, Kim SC, Wang Y, Gupta S, Davis B, Simon SI, et al. HSP60 in heart failure: abnormal distribution and role in cardiac myocyte apoptosis. Am J Physiol Heart Circ Physiol. 2007;293(4):H2238-47.
- Kim S-C, Stice JP, Chen L, Jung JS, Gupta S, Wang Y, et al. Extracellular heat shock protein 60, cardiac myocytes, and apoptosis. Circulation research. 2009;105(12):1186-95.
- Tian J, Guo X, Liu X-M, Liu L, Weng Q-F, Dong S-J, et al. Extracellular HSP60 induces inflammation through activating and up-regulating TLRs in cardiomyocytes. Cardiovascular Research. 2013;98(3):391-401.
- Kawai T, Akira S, editors. TLR signaling. Seminars in immunology; 2007: Elsevier.
- Kirchhoff S, Gupta S, Knowlton A. Cytosolic heat shock protein 60, apoptosis, and myocardial injury. Circulation. 2002;105(24):2899-904.
- Gupta S, Knowlton A. Cytosolic heat shock protein 60, hypoxia, and apoptosis. Circulation. 2002;106(21):2727-33.
- Xanthoudakis S, Roy S, Rasper D, Hennessey T, Aubin Y, Cassady R, et al. Hsp60 accelerates the maturation of pro-caspase-3 by upstream activator proteases during apoptosis. The EMBO journal. 1999.
- Chandra D, Choy G, Tang DG. Cytosolic accumulation of HSP60 during apoptosis with or without apparent mitochondrial release: evidence that its pro-apoptotic or pro-survival functions involve differential interactions with caspase-3. Journal of Biological Chemistry. 2007;282(43):31289-301.
- Samali A, Cai J, Zhivotovsky B, Jones DP, Orrenius S. Presence of a pre-apoptotic complex of pro-caspase-3, Hsp60 and Hsp10 in the mitochondrial fraction of Jurkat cells. The EMBO journal. 1999.
- Ghosh JC, Dohi T, Kang BH, Altieri DC. Hsp60 regulation of tumor cell apoptosis. Journal of Biological Chemistry. 2008;283(8):5188-94.
- Ghosh JC, Siegelin MD, Dohi T, Altieri DC. Heat shock protein 60 regulation of the mitochondrial permeability transition pore in tumor cells. Cancer research. 2010;70(22):8988-93.
- Gupta S, Knowlton A. HSP60, Bax, apoptosis and the heart. Journal of cellular and molecular medicine. 2005;9(1):51-8.
- Jürgensmeier JM, Xie Z, Deveraux Q, Ellerby L, Bredesen D, Reed JC. Bax directly induces release of cytochrome c from isolated mitochondria. Proceedings of the National Academy of Sciences. 1998;95(9):4997-5002.
- Cappello F, Logozzi M, Campanella C, Bavisotto CC, Marcilla A, Properzi F, et al. Exosome levels in human body fluids: A tumor marker by themselves? Eur J Pharm Sci. 2017;96:93-8.
- Eckhardt A, Kulhava L, Miksik I, Pataridis S, Hlavackova M, Vasinova J, et al. Proteomic analysis of cardiac ventricles: baso-apical differences. Mol Cell Biochem. 2018;445(1-2):211-9.
- Oyama J, Maeda T, Sasaki M, Higuchi Y, Node K, Makino N. Repetitive hyperthermia attenuates progression of left ventricular hypertrophy and increases telomerase activity in hypertensive rats. Am J Physiol Heart Circ Physiol. 2012;302(10):H2092-101.
- Yang M, Tan H, Cheng L, He M, Wei Q, Tanguay RM, et al. Expression of heat shock proteins in myocardium of patients with atrial fibrillation. Cell Stress Chaperones. 2007;12(2):142-50.
- Toga W, Tanonaka K, Takeo S. Changes in Hsp60 level of the failing heart following acute myocardial infarction and the effect of long-term treatment with trandolapril. Biol Pharm Bull. 2007;30(1):105-10.
- Williams JW, Huang LH, Randolph GJ. Cytokine Circuits in Cardiovascular Disease. Immunity. 2019;50(4):941-54.
- Fan F, Duan Y, Yang F, Trexler C, Wang H, Huang L, et al. Deletion of heat shock protein 60 in adult mouse cardiomyocytes perturbs mitochondrial protein homeostasis and causes heart failure. Cell Death Differ. 2020, 27, 587–600. [Google Scholar] [CrossRef]
- Knowlton AA, Kapadia S, Torre-Amione G, Durand JB, Bies R, Young J, et al. Differential expression of heat shock proteins in normal and failing human hearts. J Mol Cell Cardiol. 1998;30(4):811-8.
- Niizeki T, Takeishi Y, Watanabe T, Nitobe J, Miyashita T, Miyamoto T, et al. Relation of serum heat shock protein 60 level to severity and prognosis in chronic heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol. 2008;102(5):606-10.
- Al-Zghoul MB, Ismail ZB, Dalab AE, Al-Ramadan A, Althnaian TA, Al-Ramadan SY, et al. Hsp90, Hsp60 and HSF-1 genes expression in muscle, heart and brain of thermally manipulated broiler chicken. Res Vet Sci. 2015;99:105-11.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




