Submitted:
31 July 2024
Posted:
02 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
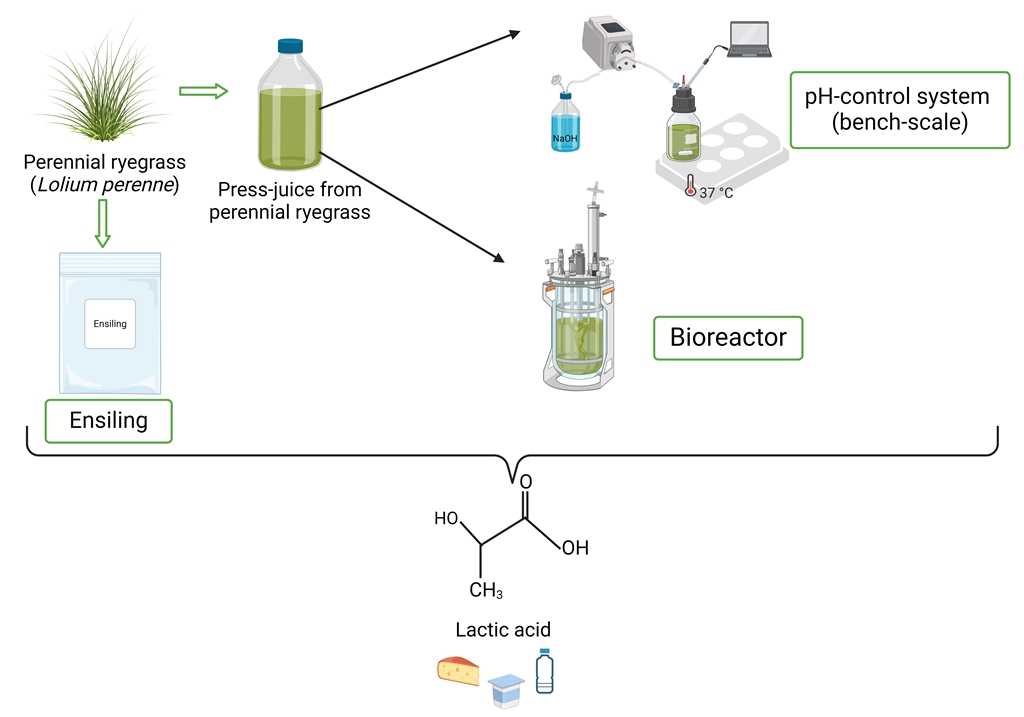
2. Materials and Methods
2.1. Raw Material
| Variety | Ploidy | Breeder |
| Agaska | 2x | DLF |
| Honroso | 2x | DSV |
| Arvicola | 4x | Freudenberger |
| Barmigo | 4x | Barenbrug |
| Exlosion | 4x | DSV |
2.3 Pretreatment and Press- Juice Preparation
2.2. Microorganism, Preculture and Complex Medium
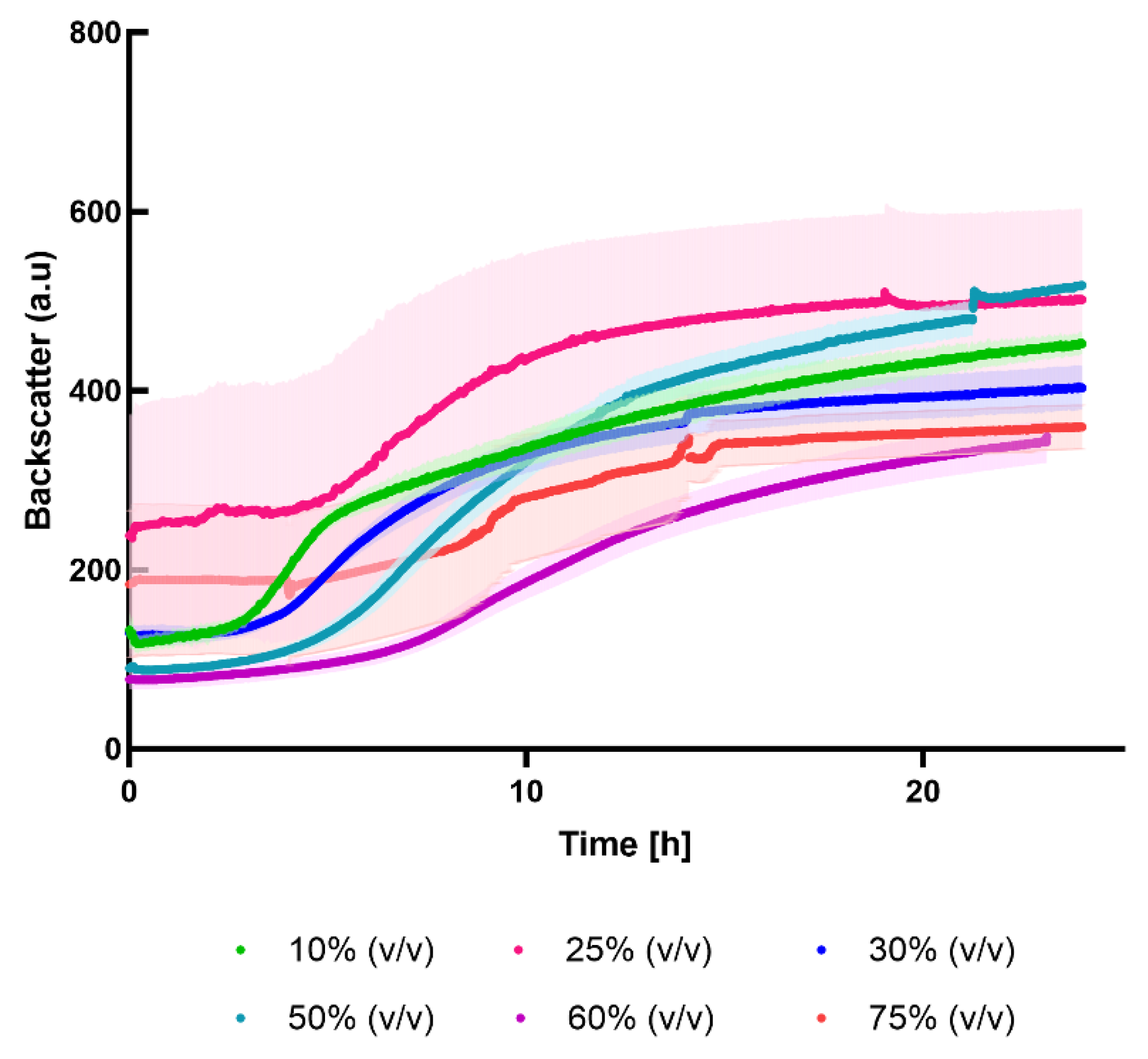
2.3. Cultivation in Different Percentage of Press-Juice
2.4. Bench-Scale Fermentation in Schott Bottle
2.5. Scaled-Up Fermentation in Bioreactor
2.6. Ensiling of Different Varieties of Lolium perenne Whole Grass Fractions
2.7. Analytical Methods
2.8. Data Processing and Evaluation
3. Results and Discussion
3.1. Influence of Raw Material
| Variety | Cut of the Year | Harvest Date | DM (dt/ha) |
CF (%) |
CP (%) |
WSC (%) |
| Agaska | 2 | 07.07.2021 | 26.76 | 25.73 | 10.81 | 16.15 |
| Arvicola | 2 | 07.07.2021 | 20.79 | 21.52 | 11.34 | 21.41 |
| Barmigo | 2 | 07.07.2021 | 23.55 | 24.76 | 10.04 | 18.37 |
| Explosion | 1 | 03.06.2022 | 51.95 | 25.95 | 5.82 | 22.62 |
| Honroso | 1 | 03.06.2022 | 43.33 | 25.29 | 7.95 | 20.37 |
3.2. Press-Juices Analysis
| Arvicola | Agaska | Barmigo | |
| Total sugar [g/L] | 23.8 | 39.5 | 28.3 |
| Protein [mg/L] | 559 | 683 | 642 |
| Amino acid [mg/L] | n. a | 3130.5 | 2560.9 |
| Cations [g/L] | 9.3 | 8.1 | 8.2 |
| Anions [g/L] | 6.0 | 5.9 | 4.8 |
| Sample | Glucose content [% (m/m)] |
Deviation | Fructose content [% (m/m)] |
Deviation |
| Honroso | 2,93 % | 0,41 % | 5,32 % | 1,20 % |
| Explosion | 5,09 % | 0,79 % | 7,56 % | 3,00 % |
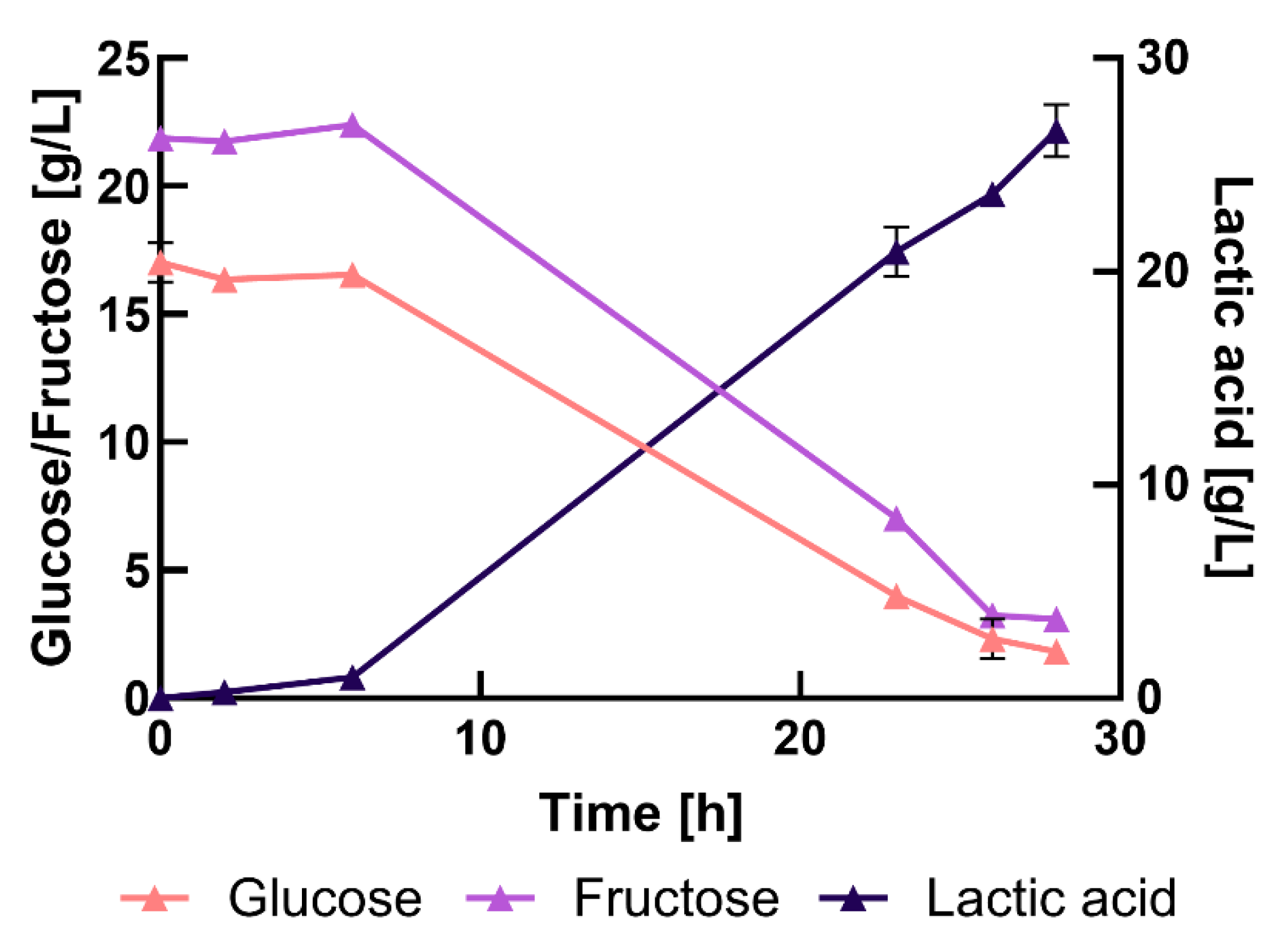
3.3. Bench-Scale Fermentation in Complex Medium
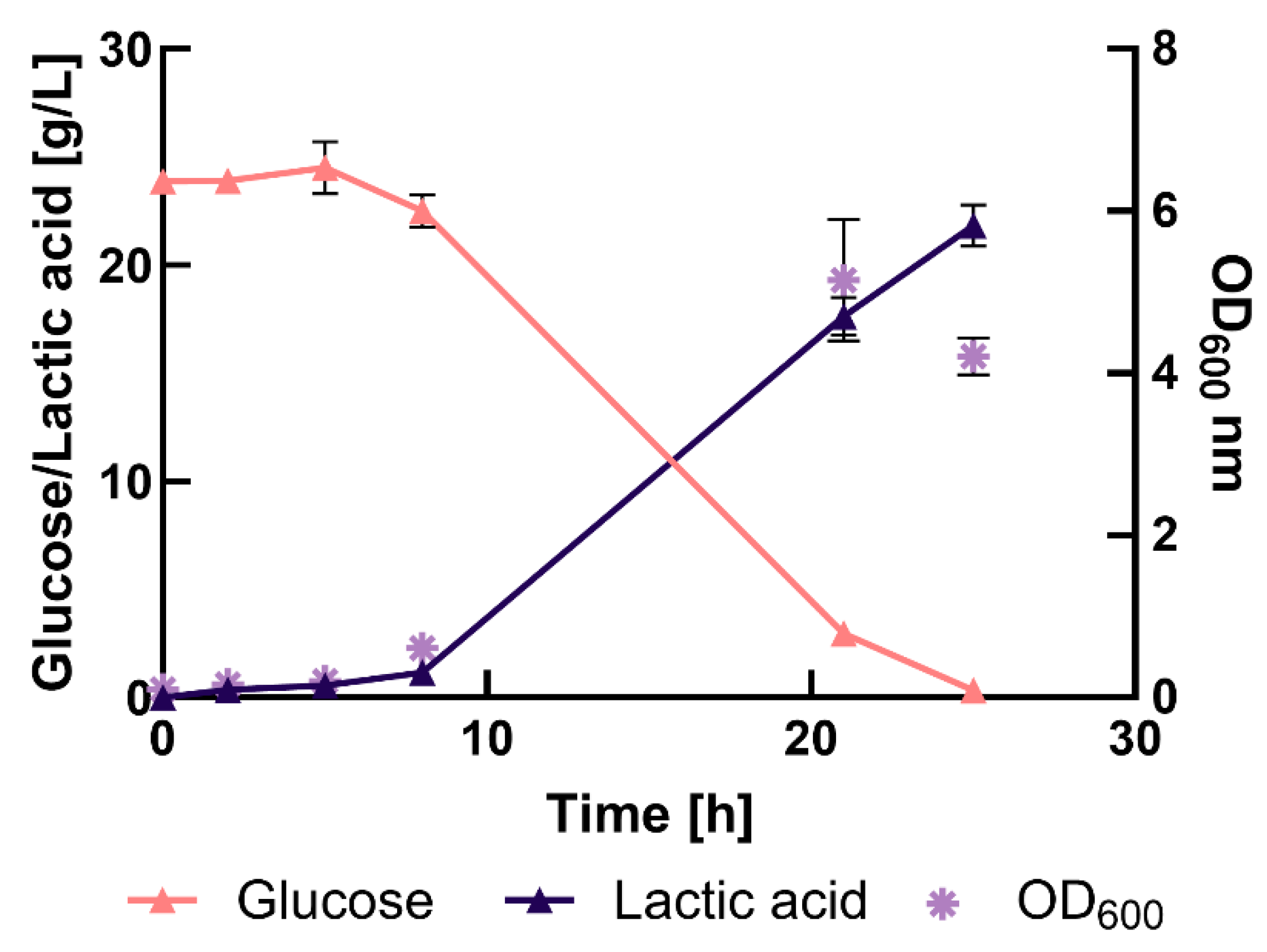
3.4. Cultivation in Different Percentage of Press-Juice
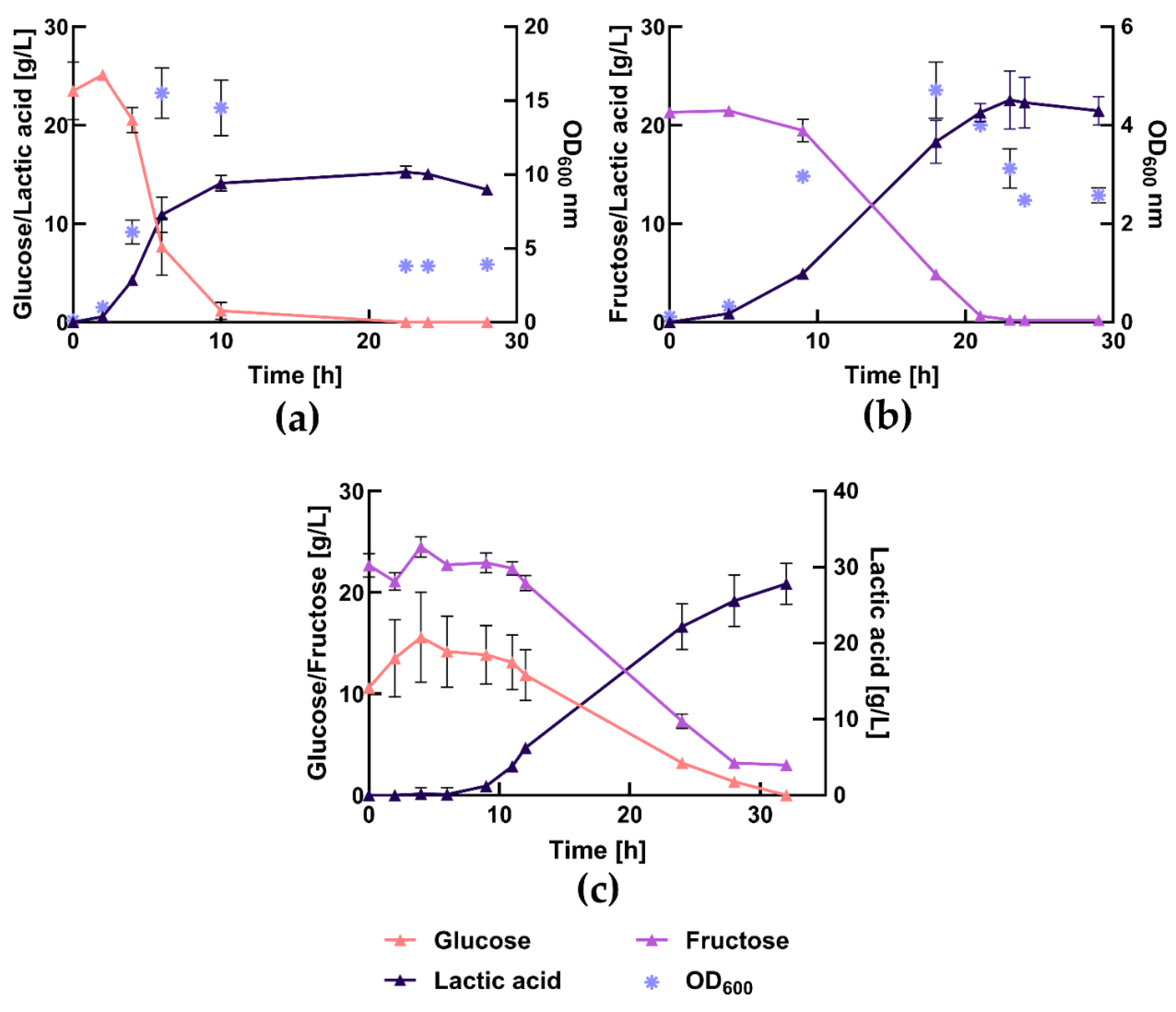
3.5. Bench-Scale Fermentation Using the Press-Juice
3.6. Comparison between Complex-Medium and Press-Juice in Bioreactor
| MRS medium with glucose1 | MRS medium with fructose1 | 75% (v/v) press-juice2 | |
|---|---|---|---|
| Si [g/L] | 23.5 ± 2.9 | 21.3 ± 0.3 | 33.3 ± 0.84 |
| Sugar(s) remained [g/L] | 0 ± 0.00 | 0.21 ± 00 | 2.9 ± 0.00 |
| Su [%] | 94.6 ± 4.5 | 77.2 ± 1.43 | 102.7 ± 3.5 |
| rS [g/Lh] | -2.23 ± 0.36 | -0.91 ± 0.03 | -1.03 ±0.04 |
| CLA [g/L] | 13.47 ± 0.57 | 21.45 ± 1.45 | 27.81 ± 2.69 |
| S [g/g] | 0.59 ± 0.11 | 1.02 ± 0.08 | 0.84 ± 0.01 |
| YP/S [g/g] | 0.63 ± 0.02 | 1.02 ± 0.08 | 0.91 ± 0.07 |
| QLA [g/L h] | 0.48 ± 0.02 | 0.69 ± 0.00 | 0.87 ± 0.10 |
| Fermentation efficiency [%] | 51.3 ± 1.9 | 101.7 ± 8.4 | 91.4 ± 6.7 |
| YX/S [g/g] | 0.17 ± 0.02 | 0.12 ± 0.01 | n.a |
| QCELLS [g/L h] | 0.16 ± 0.00 | 0.09 ± 0.01 | n.a |
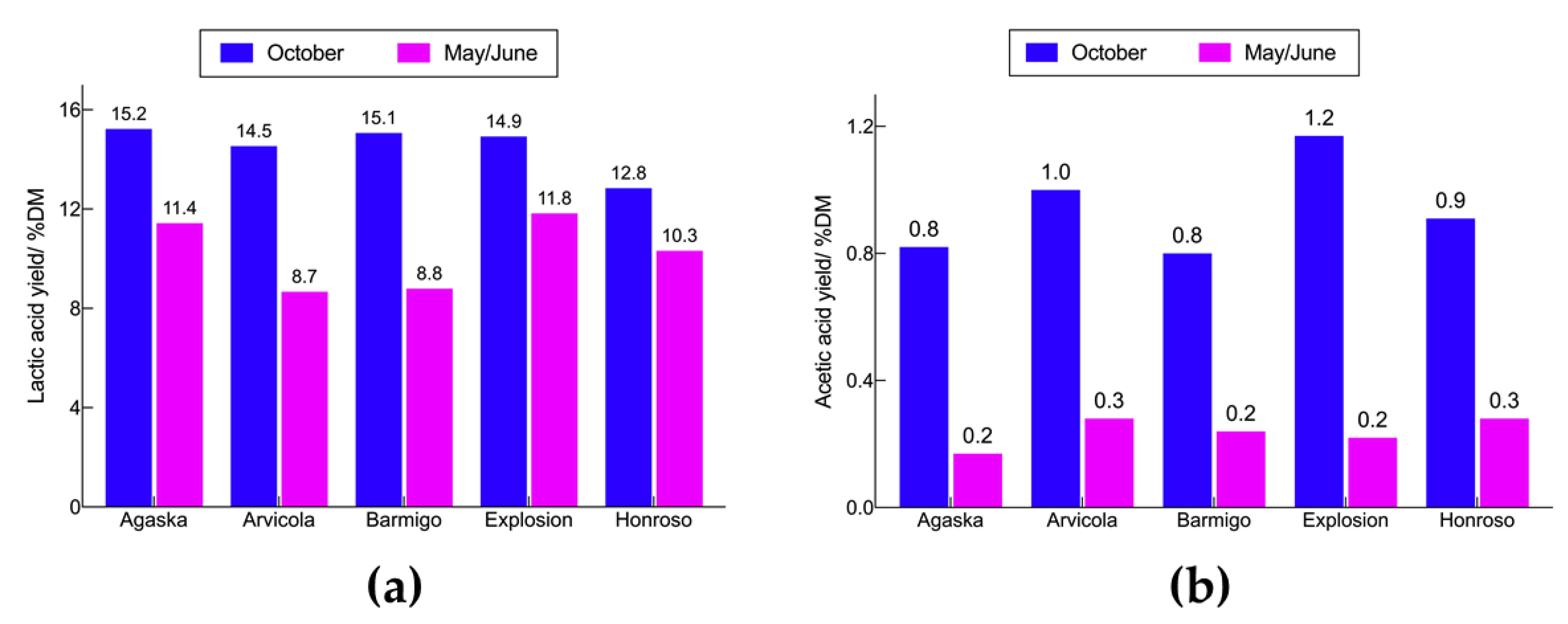
3.6. Lactic Acid Yields from Ensiling Whole Grass Fractions of Different Varieties of Lolium perenne
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Antar, M.; Lyu, D.; Nazari, M.; Shah, A.; Zhou, X.; Smith, D.L. Biomass for a Sustainable Bioeconomy: An Overview of World Biomass Production and Utilization. Renewable and Sustainable Energy Reviews 2021, 139, 110691. [Google Scholar] [CrossRef]
- Yadav, V.G.; Yadav, G.D.; Patankar, S.C. The Production of Fuels and Chemicals in the New World: Critical Analysis of the Choice between Crude Oil and Biomass Vis-à-Vis Sustainability and the Environment. Clean Technol Environ Policy 2020, 22, 1757–1774. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Feng, Y.; Fu, J.; Guo, H.; Guo, Y.; Han, B.; Jiang, Z.; Kong, L.; Li, C.; Liu, H.; et al. Catalytic Conversion of Lignocellulosic Biomass into Chemicals and Fuels. Green Energy and Environment 2023, 8, 10–114. [Google Scholar] [CrossRef]
- McEniry, J.; O’Kiely, P. Developments in Grass-/Forage-Based Biorefineries. In Advances in Biorefineries: Biomass and Waste Supply Chain Exploitation; Waldron, K., Ed.; Woodhead Publishing, 2014; pp. 335–363 ISBN 9780857095213.
- Abdel-Rahman, M.A.; Tashiro, Y.; Sonomoto, K. Recent Advances in Lactic Acid Production by Microbial Fermentation Processes. Biotechnol Adv 2013, 31, 877–902. [Google Scholar] [CrossRef] [PubMed]
- Balla, E.; Daniilidis, V.; Karlioti, G.; Kalamas, T.; Stefanidou, M.; Bikiaris, N.D.; Vlachopoulos, A.; Koumentakou, I.; Bikiaris, D.N. Poly(Lactic Acid): A Versatile Biobased Polymer for the Future with Multifunctional Properties.Applications. Polymers (Basel) 2021, 13, 1822. [Google Scholar] [CrossRef] [PubMed]
- López-Gómez, J.P.; Alexandri, M.; Schneider, R.; Venus, J. A Review on the Current Developments in Continuous Lactic Acid Fermentations and Case Studies Utilising Inexpensive Raw Materials. Process Biochemistry 2019, 79, 1–10. [Google Scholar] [CrossRef]
- Alves de Oliveira, R.; Komesu, A.; Vaz Rossell, C.E.; Maciel Filho, R. Challenges and Opportunities in Lactic Acid Bioprocess Design—From Economic to Production Aspects. Biochem Eng J 2018, 133, 219–239. [Google Scholar] [CrossRef]
- Varriale, L.; Ulber, R. Fungal-Based Biorefinery: From Renewable Resources to Organic Acids. ChemBioEng Reviews 2023, 10, 272–292. [Google Scholar] [CrossRef]
- Ajala, E.O.; Olonade, Y.O.; Ajala, M.A.; Akinpelu, G.S. Lactic Acid Production from Lignocellulose – A Review of Major Challenges and Selected Solutions. ChemBioEng Reviews 2020, 38–49. [Google Scholar] [CrossRef]
- Li, L.; Cai, D.; Wang, C.; Han, J.; Ren, W.; Zheng, J.; Wang, Z.; Tan, T. Continuous L-Lactic Acid Production from Defatted Rice Bran Hydrolysate Using Corn Stover Bagasse Immobilized Carrier. RSC Adv 2015, 5, 18511–18517. [Google Scholar] [CrossRef]
- Kuo, Y.C.; Yuan, S.F.; Wang, C.A.; Huang, Y.J.; Guo, G.L.; Hwang, W.S. Production of Optically Pure L-Lactic Acid from Lignocellulosic Hydrolysate by Using a Newly Isolated and d-Lactate Dehydrogenase Gene-Deficient Lactobacillus Paracasei Strain. Bioresour Technol 2015, 198, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.Y.; Qian, H.; Zhang, W.G. Enhancement of L-Lactic Acid Production in Lactobacillus Casei from Jerusalem Artichoke Tubers by Kinetic Optimization and Citrate Metabolism. J Microbiol Biotechnol 2010, 20, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Karp, S.G.; Igashiyama, A.H.; Siqueira, P.F.; Carvalho, J.C.; Vandenberghe, L.P.S.; Thomaz-Soccol, V.; Coral, J.; Tholozan, J.L.; Pandey, A.; Soccol, C.R. Application of the Biorefinery Concept to Produce L-Lactic Acid from the Soybean Vinasse at Laboratory and Pilot Scale. Bioresour Technol 2011, 102, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Hama, S.; Mizuno, S.; Kihara, M.; Tanaka, T.; Ogino, C.; Noda, H.; Kondo, A. Production of D-Lactic Acid from Hardwood Pulp by Mechanical Milling Followed by Simultaneous Saccharification and Fermentation Using Metabolically Engineered Lactobacillus Plantarum. Bioresour Technol 2015, 187, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.M.; Kim, J.S.; Nguyen, T.N.; Kim, S.K.; Choi, G.J.; Choi, Y.H.; Jang, K.S.; Kim, J.C. Production of L- and d-Lactic Acid from Waste Curcuma Longa Biomass through Simultaneous Saccharification and Cofermentation. Bioresour Technol 2013, 146, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Lin, Y.; Zhang, Z.; Xiang, T.; Mei, Y.; Zhao, S.; Liang, Y.; Peng, N. High-Titer Lactic Acid Production by Lactobacillus Pentosus FL0421 from Corn Stover Using Fed-Batch Simultaneous Saccharification and Fermentation. Bioresour Technol 2016, 214, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Tirpanalan, Ö.; Reisinger, M.; Smerilli, M.; Huber, F.; Neureiter, M.; Kneifel, W.; Novalin, S. Wheat Bran Biorefinery - An Insight into the Process Chain for the Production of Lactic Acid. Bioresour Technol 2015, 180, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Cingadi, S.; Srikanth, K.; E.V.R, A.; Sivaprakasam, S. Statistical Optimization of Cassava Fibrous Waste Hydrolysis by Response Surface Methodology and Use of Hydrolysate Based Media for the Production of Optically Pure D-Lactic Acid. Biochem Eng J 2015, 102, 82–90. [CrossRef]
- Pontes, R.; Romaní, A.; Michelin, M.; Domingues, L.; Teixeira, J.; Nunes, J. L-Lactic Acid Production from Multi-Supply Autohydrolyzed Economically Unexploited Lignocellulosic Biomass. Ind Crops Prod 2021, 170. [Google Scholar] [CrossRef]
- Karnaouri, A.; Asimakopoulou, G.; Kalogiannis, K.G.; Lappas, A.; Topakas, E. Efficient D-Lactic Acid Production by Lactobacillus Delbrueckii Subsp. Bulgaricus through Conversion of Organosolv Pretreated Lignocellulosic Biomass. Biomass Bioenergy 2020, 140, 105672. [Google Scholar] [CrossRef]
- Tsapekos, P.; Alvarado-Morales, M.; Baladi, S.; Bosma, E.F.; Angelidaki, I. Fermentative Production of Lactic Acid as a Sustainable Approach to Valorize Household Bio-Waste. Frontiers in Sustainability 2020, 1, 1–12. [Google Scholar] [CrossRef]
- Costa, S.; Summa, D.; Semeraro, B.; Zappaterra, F.; Rugiero, I.; Tamburini, E. Fermentation as a Strategy for Bio-Transforming Waste into Resources: Lactic Acid Production from Agri-Food Residues. Fermentation 2020, 7, 1–12. [Google Scholar] [CrossRef]
- Akermann, A.; Weiermüller, J.; Christmann, J.; Guirande, L.; Glaser, G.; Knaus, A.; Ulber, R. Brewers’ Spent Grain Liquor as a Feedstock for Lactate Production with Lactobacillus Delbrueckii Subsp. Lactis. Eng Life Sci 2020, 20, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Grewal, J.; Khare, S.K. One-Pot Bioprocess for Lactic Acid Production from Lignocellulosic Agro-Wastes by Using Ionic Liquid Stable Lactobacillus Brevis. Bioresour Technol 2018, 251, 268–273. [Google Scholar] [CrossRef]
- Montipó, S.; Ballesteros, I.; Fontana, R.C.; Liu, S.; Martins, A.F.; Ballesteros, M.; Camassola, M. Integrated Production of Second Generation Ethanol and Lactic Acid from Steam-Exploded Elephant Grass. Bioresour Technol 2018, 249, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Ahring, B.K.; Traverso, J.J.; Murali, N.; Srinivas, K. Continuous Fermentation of Clarified Corn Stover Hydrolysate for the Production of Lactic Acid at High Yield and Productivity. Biochem Eng J 2016, 109, 162–169. [Google Scholar] [CrossRef]
- Zhang, Z.; Xie, Y.; He, X.; Li, X.; Hu, J.; Ruan, Z.; Zhao, S.; Peng, N.; Liang, Y. Comparison of High-Titer Lactic Acid Fermentation from NaOH-and NH3-H2O2-Pretreated Corncob by Bacillus Coagulans Using Simultaneous Saccharification and Fermentation. Sci Rep 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wee, Y.J.; Kim, J.N.; Yun, J.S.; Ryu, H.W. Utilization of Sugar Molasses for Economical L(+)-Lactic Acid Production by Batch Fermentation of Enterococcus Faecalis. Enzyme Microb Technol 2004, 35, 568–573. [Google Scholar] [CrossRef]
- Selim, M.T.; Salem, S.S.; Fouda, A.; El-Gamal, M.S.; Abdel-Rahman, M.A. Use of Corn-Steep Water Effluent as a Promising Substrate for Lactic Acid Production by Enterococcus Faecium Strain Wh51-1. Fermentation 2021, 7, 1–18. [Google Scholar] [CrossRef]
- Di Lorenzo, R.D.; Serra, I.; Porro, D.; Branduardi, P. State of the Art on the Microbial Production of Industrially Relevant Organic Acids. Catalysts 2022, 12, 234. [Google Scholar] [CrossRef]
- Abedi, E.; Hashemi, S.M.B. Lactic Acid Production – Producing Microorganisms and Substrates Sources-State of Art. Heliyon 2020, 6, e04974. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, M.; Gobbetti, M. Lactobacillus SPP.: General Characteristics ☆. In Reference Module in Food Science; Elsevier, 2016.
- Talukder, M.M.R.; Das, P.; Wu, J.C. Microalgae (Nannochloropsis Salina) Biomass to Lactic Acid and Lipid. Biochem Eng J 2012, 68, 109–113. [Google Scholar] [CrossRef]
- Kim, J.; Kim, Y.-M.; Lebaka, V.R.; Wee, Y.-J. Lactic Acid for Green Chemical Industry: Recent Advances in and Future Prospects for Production Technology, Recovery, and Applications. Fermentation 2022, 8, 609. [Google Scholar] [CrossRef]
- Büyükkileci, A.O.; Harsa, S. Batch Production of L(+) Lactic Acid from Whey by Lactobacillus Casei (NRRL B-441). Journal of Chemical Technology and Biotechnology 2004, 79, 1036–1040. [Google Scholar] [CrossRef]
- Hayek, S.A.; Ibrahim, S.A. Current Limitations and Challenges with Lactic Acid Bacteria: A Review. Food Nutr Sci 2013, 2013, 73–87. [Google Scholar] [CrossRef]
- Pessione, A.; Lamberti, C.; Pessione, E. Proteomics as a Tool for Studying Energy Metabolism in Lactic Acid Bacteria. Mol Biosyst 2010, 6, 1419–1430. [Google Scholar] [CrossRef] [PubMed]
- Varriale, L.; Volkmar, M.; Weiermüller, J.; Ulber, R. Effects of Pretreatment on the Biocatalysis of Renewable Resources. Chem Ing Tech 2022, 94, 1818–1826. [Google Scholar] [CrossRef]
- Baruah, J.; Nath, B.K.; Sharma, R.; Kumar, S.; Deka, R.C.; Baruah, D.C.; Kalita, E. Recent Trends in the Pretreatment of Lignocellulosic Biomass for Value-Added Products. Front Energy Res 2018, 6. [Google Scholar] [CrossRef]
- Gonçalves, M.C.P.; Romanelli, J.P.; Cansian, A.B.M.; Pucci, E.F.Q.; Guimarães, J.R.; Tardioli, P.W.; Saville, B.A. A Review on the Production and Recovery of Sugars from Lignocellulosics for Use in the Synthesis of Bioproducts. Ind Crops Prod 2022, 186. [Google Scholar] [CrossRef]
- Singhvi, M.; Zendo, T.; Sonomoto, K. Free Lactic Acid Production under Acidic Conditions by Lactic Acid Bacteria Strains: Challenges and Future Prospects. Appl Microbiol Biotechnol 2018, 102, 5911–5924. [Google Scholar] [CrossRef] [PubMed]
- Mussatto, S.I.; Fernandes, M.; Mancilha, I.M.; Roberto, I.C. Effects of Medium Supplementation and PH Control on Lactic Acid Production from Brewer’s Spent Grain. Biochem Eng J 2008, 40, 437–444. [Google Scholar] [CrossRef]
- Si, H.; Liang, X.; Liu, S.; Xu, M.; Wang, J.; Hua, D. Efficient Production of L-Lactic Acid from Corn Straw Hydrolysate. IOP Conf Ser Earth Environ Sci 2020, 514. [Google Scholar] [CrossRef]
- Hébert, E.M.; Raya, R.R.; De Giori, G.S. Nutritional Requirements of Lactobacillus Delbrueckii Subsp. Lactis in a Chemically Defined Medium. Curr Microbiol 2004, 49, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Savijoki, K.; Ingmer, H.; Varmanen, P. Proteolytic Systems of Lactic Acid Bacteria. Appl Microbiol Biotechnol 2006, 71, 394–406. [Google Scholar] [CrossRef] [PubMed]
- Richter, K.; Träger, A. L(+)-Lactic Acid from Sweet Sorghum by Submerged and Solid-state Fermentations. Acta Biotechnol 1994, 14, 367–378. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, M.; Cai, D.; Wang, B.; Wang, Z.; Qin, P.; Tan, T. Efficient L-Lactic Acid Production from Sweet Sorghum Bagasse by Open Simultaneous Saccharification and Fermentation. RSC Adv 2016, 6, 35771–35777. [Google Scholar] [CrossRef]
- Iino, T.; Uchimura, T.; Komagata, K. The Effect of Sodium Acetate on the Growth Yield, the Production of L- and D-Lactic Acid, and the Activity of Some Enzymes of the Glycolytic Pathway of Lactobacillus Sakei NRIC 1071 and Lactobacillus Plantarum NRIC 1067. Journal of General and Applied Microbiology 2002, 48, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Coelho, L.F.; De Lima, C.J.B.; Rodovalho, C.M.; Bernardo, M.P.; Contiero, J. Lactic Acid Production by New Lactobacillus Plantarum LMISM6 Grown in Molasses: Optimization of Medium Composition. Brazilian Journal of Chemical Engineering 2011, 28, 27–36. [Google Scholar] [CrossRef]
- Hébert, E.M.; Raya, R.R.; De Giori, G.S. Nutritional Requirements of Lactobacillus Delbrueckii Subsp. Lactis in a Chemically Defined Medium. Curr Microbiol 2004, 49, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, Z.E.; Mousavi, S.M.; Razavi, S.H.; Emam-Djomeh, Z.; Kiani, H. Fermentation of Pomegranate Juice by Probiotic Lactic Acid Bacteria. World J Microbiol Biotechnol 2011, 27, 123–128. [Google Scholar] [CrossRef]
- Volkmar, M.; Maus, A.L.; Weisbrodt, M.; Bohlender, J.; Langsdorf, A.; Holtmann, D.; Ulber, R. Municipal Green Waste as Substrate for the Microbial Production of Platform Chemicals. Bioresour Bioprocess 2023, 10. [Google Scholar] [CrossRef] [PubMed]
- Boakye-Boaten, N.A.; Xiu, S.; Shahbazi, A.; Wang, L.; Li, R.; Schimmel, K. Uses of Miscanthus Press Juice within a Green Biorefinery Platform. Bioresour Technol 2016, 207, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Santamaria-Fernandez, M.; Ambye-Jensen, M.; Damborg, V.K.; Lübeck, M. Demonstration-Scale Protein Recovery by Lactic Acid Fermentation from Grass Clover – a Single Case of the Production of Protein Concentrate and Press Cake Silage for Animal Feeding Trials. Biofuels, Bioproducts and Biorefining 2019, 13, 502–513. [Google Scholar] [CrossRef]
- Rolfe, M.D.; Rice, C.J.; Lucchini, S.; Pin, C.; Thompson, A.; Cameron, A.D.S.; Alston, M.; Stringer, M.F.; Betts, R.P.; Baranyi, J.; et al. Lag Phase Is a Distinct Growth Phase That Prepares Bacteria for Exponential Growth and Involves Transient Metal Accumulation. J Bacteriol 2012, 194, 686–701. [Google Scholar] [CrossRef] [PubMed]
- Hamill, P.G.; Stevenson, A.; McMullan, P.E.; Williams, J.P.; Lewis, A.D.R.; Sudharsan, S.; Stevenson, K.E.; Farnsworth, K.D.; Khroustalyova, G.; Takemoto, J.Y.; et al. Microbial Lag Phase Can Be Indicative of, or Independent from, Cellular Stress. Sci Rep 2020, 10, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Petrut, S.; Rusu, E.; Tudorache, I.S.; Pelinescu, D.; Sarbu, I.; Stoica, I.; Vassu, T. Influence of Various Carbon Sources on Growth and Biomass Accumulation of Some Lactic Acid Bacteria Strains. Revista de Chimie 2019, 70, 2434–2438. [Google Scholar] [CrossRef]
- Chen, H.; Niu, J.; Qin, T.; Ma, Q.; Wang, L.; Shu, G. Optimization of the Medium for Lactobacillus Acidophilus by Plackett-Burman and Steepest Ascent Experiment. Acta Sci Pol Technol Aliment 2015, 14, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.T.; Hong, Z.S.; Cheng, C.L.; Ng, I.S.; Lo, Y.C.; Nagarajan, D.; Chang, J.S. Exploring Fermentation Strategies for Enhanced Lactic Acid Production with Polyvinyl Alcohol-Immobilized Lactobacillus Plantarum 23 Using Microalgae as Feedstock. Bioresour Technol 2020, 308, 123266. [Google Scholar] [CrossRef] [PubMed]
- de la Torre, I.; Acedos, M.G.; Ladero, M.; Santos, V.E. On the Use of Resting L. Delbrueckii Spp. Delbrueckii Cells for D-Lactic Acid Production from Orange Peel Wastes Hydrolysates. Biochem Eng J 2019, 145, 162–169. [Google Scholar] [CrossRef]
- Pontes, R.; Romaní, A.; Michelin, M.; Domingues, L.; Teixeira, J.; Nunes, J. L-Lactic Acid Production from Multi-Supply Autohydrolyzed Economically Unexploited Lignocellulosic Biomass. Ind Crops Prod 2021, 170. [Google Scholar] [CrossRef]
- Erliana, W.H.; Widjaja, T.; Altway, A.; Pudjiastuti, L. Synthesis of Lactic Acid from Sugar Palm Trunk Waste (Arenga Pinnata): Preliminary Hydrolysis and Fermentation Studies. Biodiversitas 2020, 21, 2281–2288. [Google Scholar] [CrossRef]
- Kung, L.; Shaver, R. Interpretation and Use of Silage Fermentation Analysis Reports; Vol. 3;
- Johnson, H.E.; Merry, R.J.; Davies, D.R.; Kell, D.B.; Theodorou, M.K.; Griffith, G.W. Vacuum Packing: A Model System for Laboratory-Scale Silage Fermentations. J Appl Microbiol 2005, 98, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Haag, N.L.; Nägele, H.J.; Fritz, T.; Oechsner, H. Effects of Ensiling Treatments on Lactic Acid Production and Supplementary Methane Formation of Maize and Amaranth - An Advanced Green Biorefining Approach. Bioresour Technol 2015, 178, 217–225. [Google Scholar] [CrossRef] [PubMed]
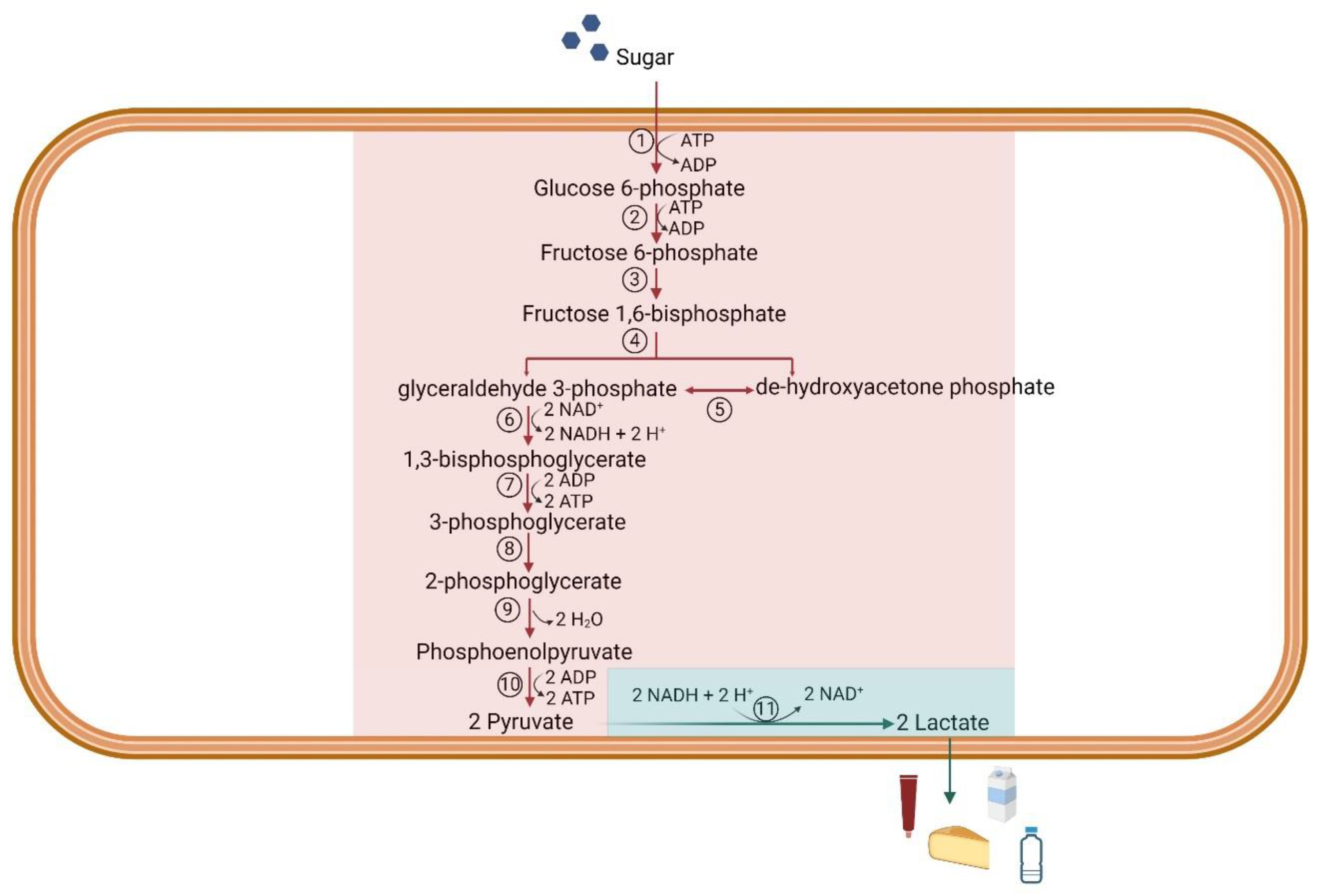
| Microorganism | Feedstock | Cultivation | Yield [g/g] | Ref. |
|---|---|---|---|---|
| L. rhamnosus LA-04-01a | Defatted rice brain hydrolysate | Batch | 0.95 | [11] |
| Continuous | 0.98 | |||
| L. paracasei 7BLa | Wood chips | Fed-Batch | 0.96 | [12] |
| Rice straw | 0.97 | |||
| L. casei G-02a | Jerusalem artichoke | Fed-Batch (SSF) | 0.96 | [13] |
| L. agilis LPB 56b | Soybean vinasse | Batch | 0.85 | [14] |
| L. plantarum NCIMB 8826a | Delignified hardwood pulp | Batch (SSF) | 0.88 | [15] |
| L. paracasei LA104a | Curcuma longa waste | Batch (SSF) | 0.69 | [16] |
| L. coryniformis ATCC 25600b | 0.65 | |||
| L. pentosus FL0421a | Corn stover | Fed-batch (SSF) | 0.66 | [17] |
| L. pentosus DSM20314a | Wheat bran | Batch | 0.73 | [18] |
| L. delbrueckii NBRC 3202b | Cassava fibrous waste | Batch | 0.50 | [19] |
| L. rhamosus ATCC 7469a | Lignocellulosic mixture | Batch (SSF) | 0.97 | [20] |
| L. delbrueckii subsp. bulgarius ATCC 11842b | Beechwood hydrolysaete | Batch (SSF) | 0.69 | [21] |
| Pine hydrolysate | 0.40 | |||
| L. delbrueckii DSM 20074b | Household bio-waste | Batch | 0.65 | [22] |
| L. casei DSM 20011a | Agro-industrial waste | Batch | 0.78 | [23] |
| L. delbrueckii subsp. lactis DSMZ 20072b | Brewers‘ spent grain | Batch | 0.89 | [24] |
| L. brevis MTCC 4460c | Cottonseed cake | Batch (SSF) | 0.22 | [25] |
| Wheat straw | 0.49 | |||
| Sugarcane bagasse | 0.52 | |||
| L. buchneri NRRL B-30929c | Elephant grass liquor | Batch | 0.50 | [26] |
| B. coagulans A107b | Tapioca starch hydrolysate | Continuous | 0.80 | [7] |
| B. coagulans ADb | Corn stover hydrolysate | Continuous | 0.95 | [27] |
| B. coagulans LA204b | Corncob | Fed-Batch (SSF) | 0.77 | [28] |
| B. coagulans LA1507b | Sweet sorghum bagasse | Open-Fed-batch (SSF) | 0.44 | [31] |
| E. faecalis RKY1b | Molasses | Batch | 0.95 | [29] |
| E. faecium WH51-1b | Corn steep water effluent | Batch | 0.89 | [30] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




