Submitted:
27 July 2024
Posted:
30 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Lines
2.3. Animals and Ethics Statement
2.4. Synthesis
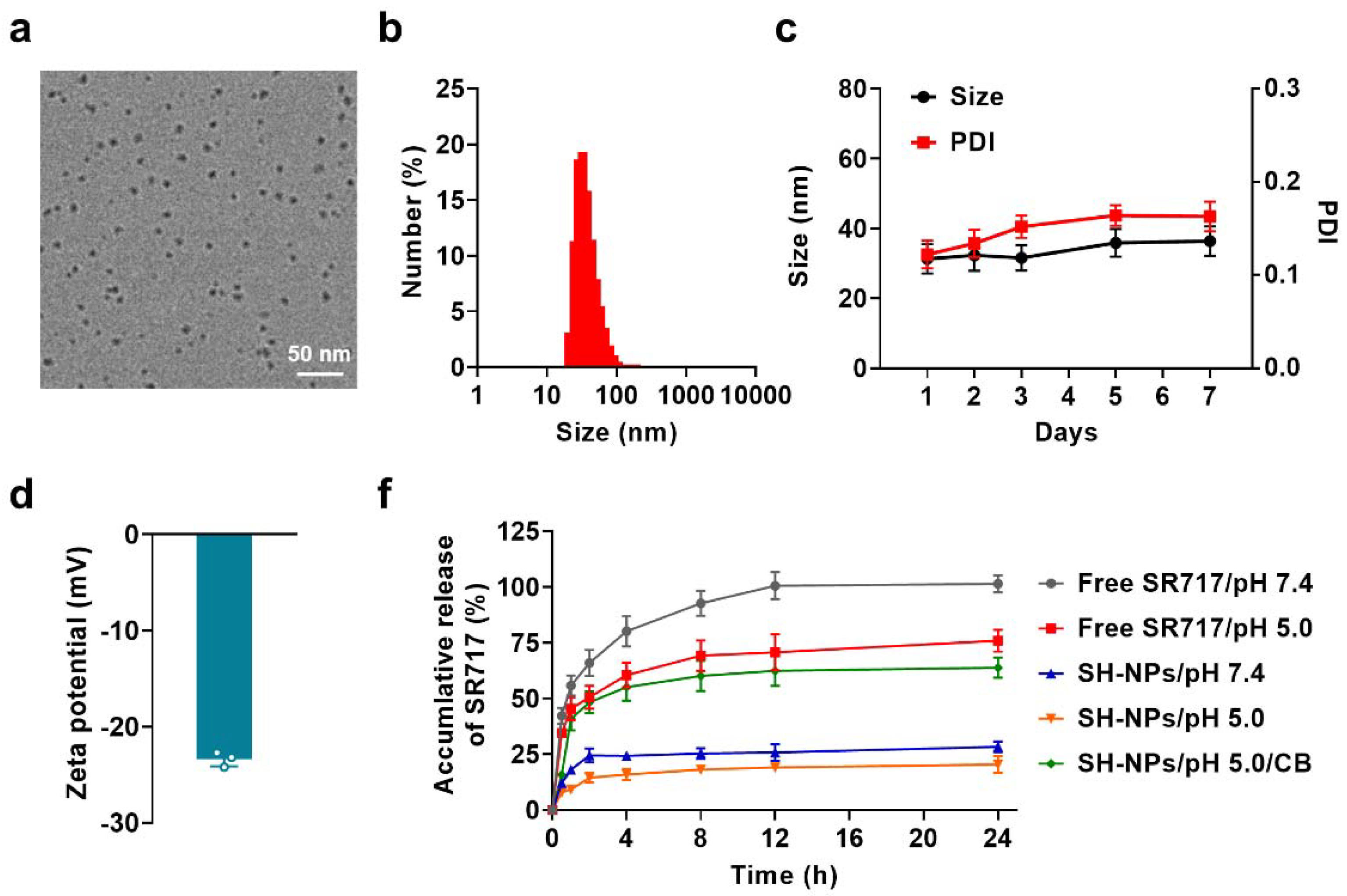
2.5. Characterization
2.6. Drug Release
2.7. Cellular Uptakes and Endocytic Pathway
2.8. In Vitro Cytotoxicity
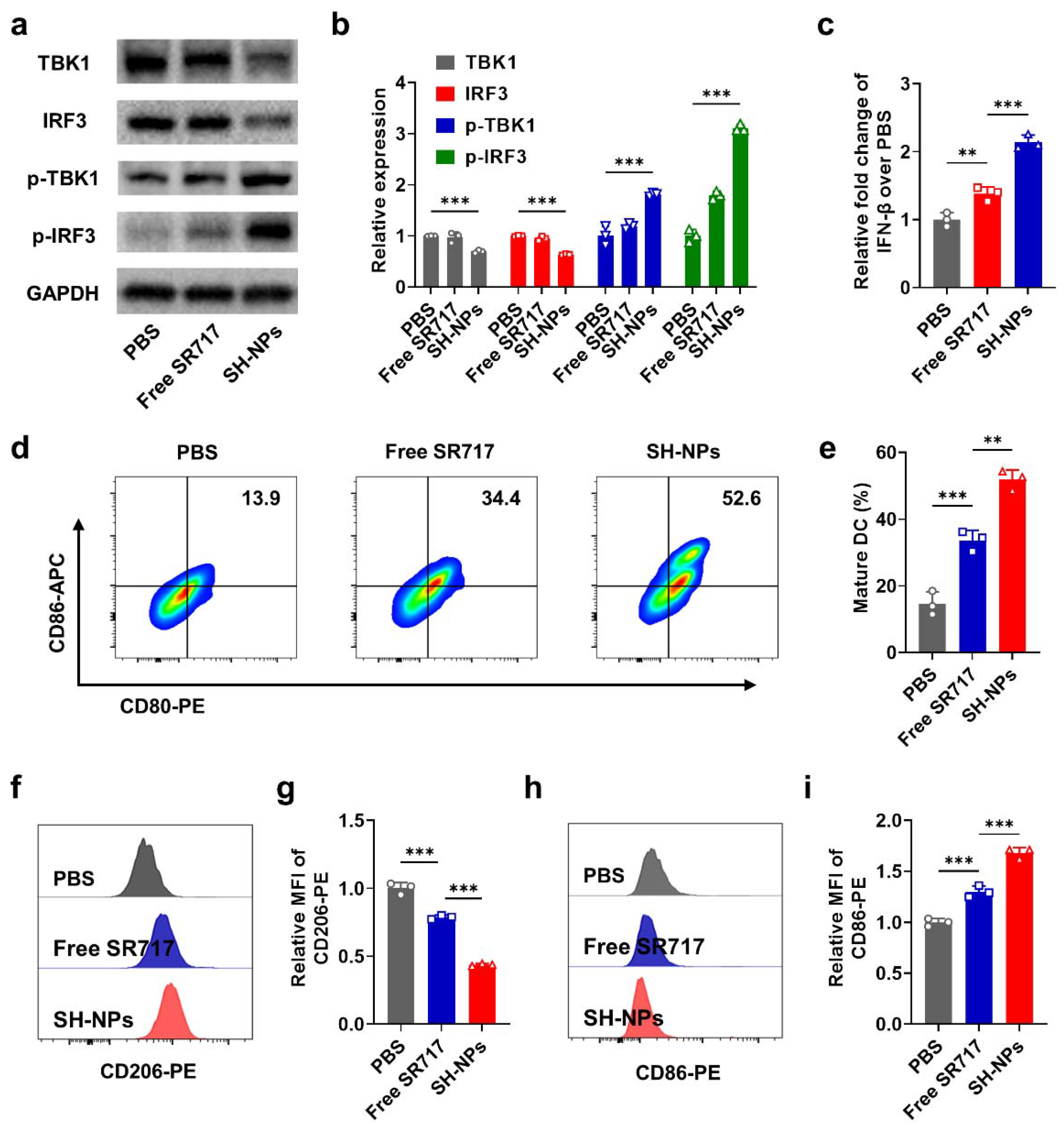
2.9. Western Blot
2.10. DC Maturation
2.11. Macrophage Polarization
2.12. Construction of RENCA Tumor Models
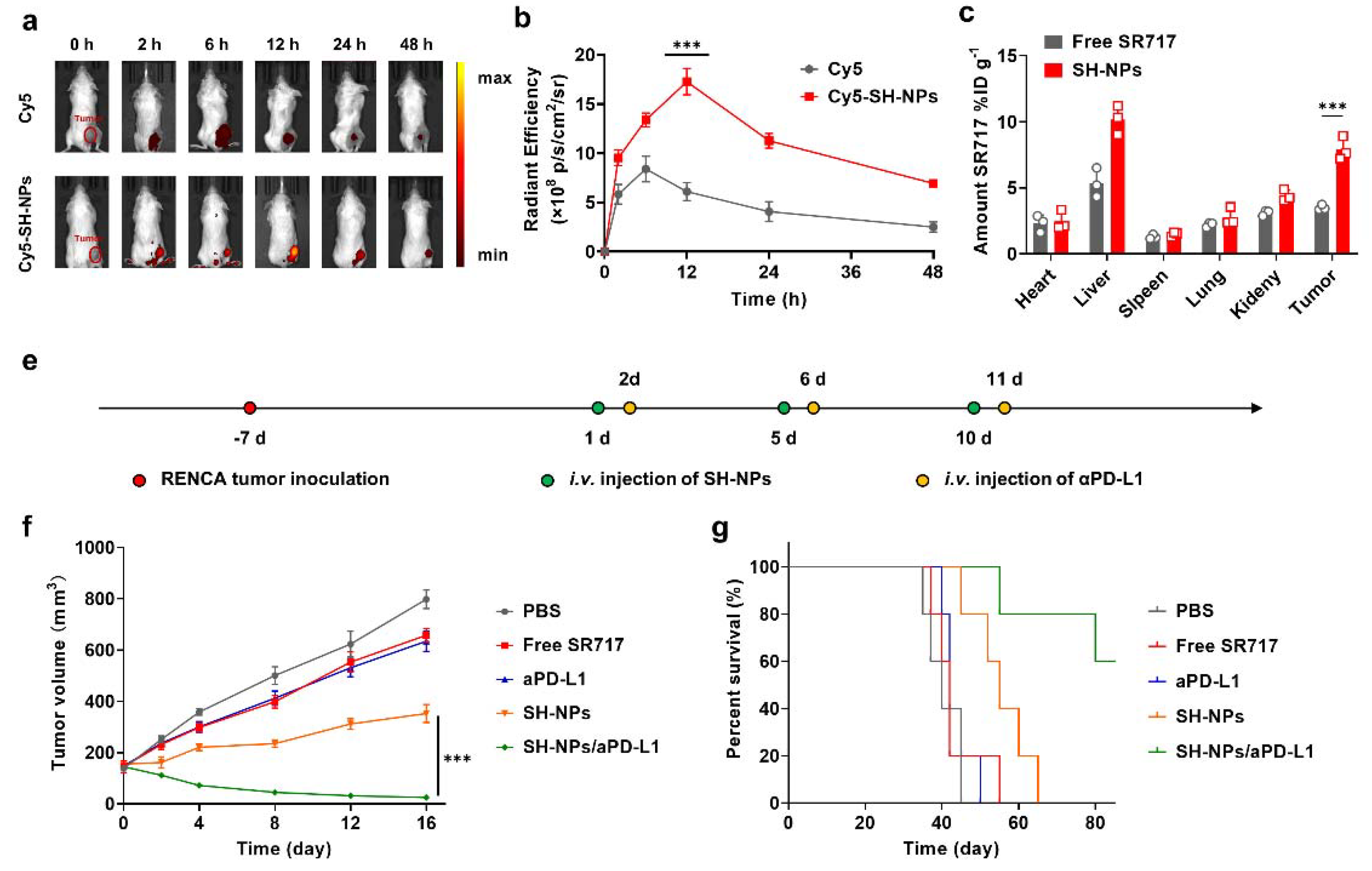
2.13. In Vivo Tumor Targeting
2.14. In Vivo Biodistribution
2.15. In Vivo Antitumor Efficacy
2.16. Cytokine Analysis
2.17. Antitumor Immune Response
2.18. Evaluation of Antitumor Immune Response in Resected Human Tissues
2.19. Statistics and Reproducibility
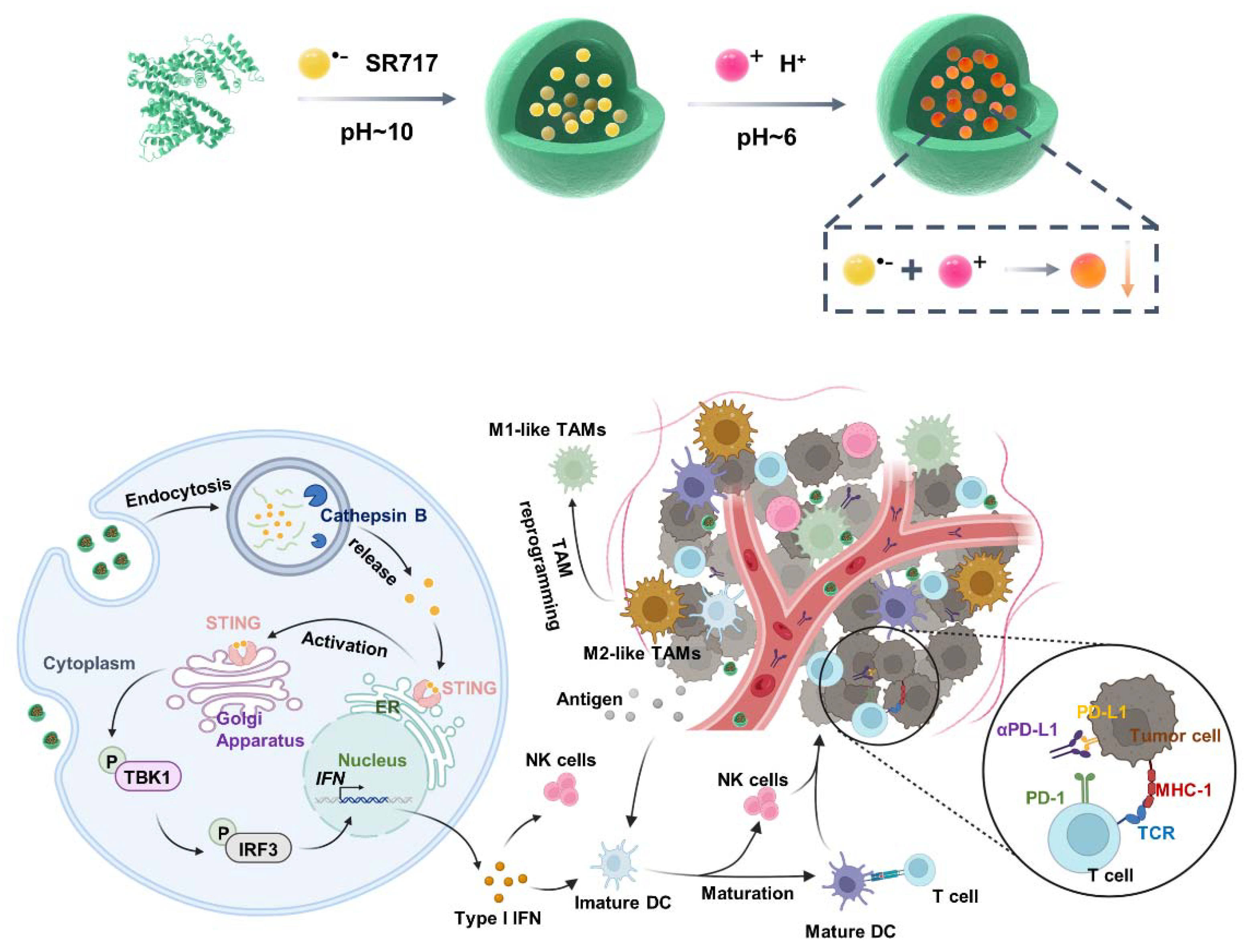
3. Results
3.1. Preparation and Characterization of SH-NPs
3.2. STING Activation and Immune Activation of SH-NPs Inside Immune Cells
3.3. Ttumor Accumulation and Antitumor Efficacy of SH-NPs
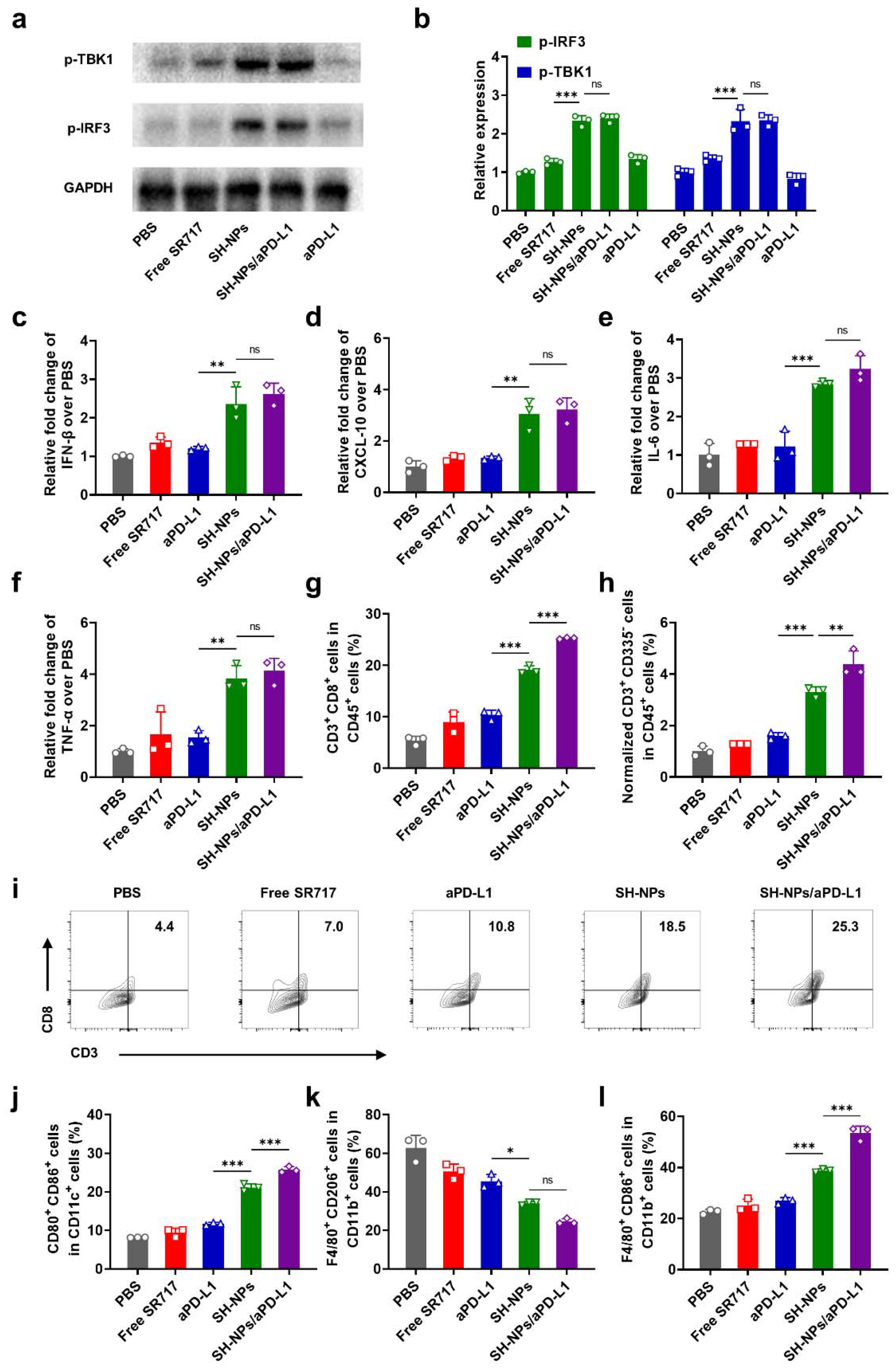
3.4. STING Activation and Immune Responses of SH-NPs in Mice Bearing Renal Tumor
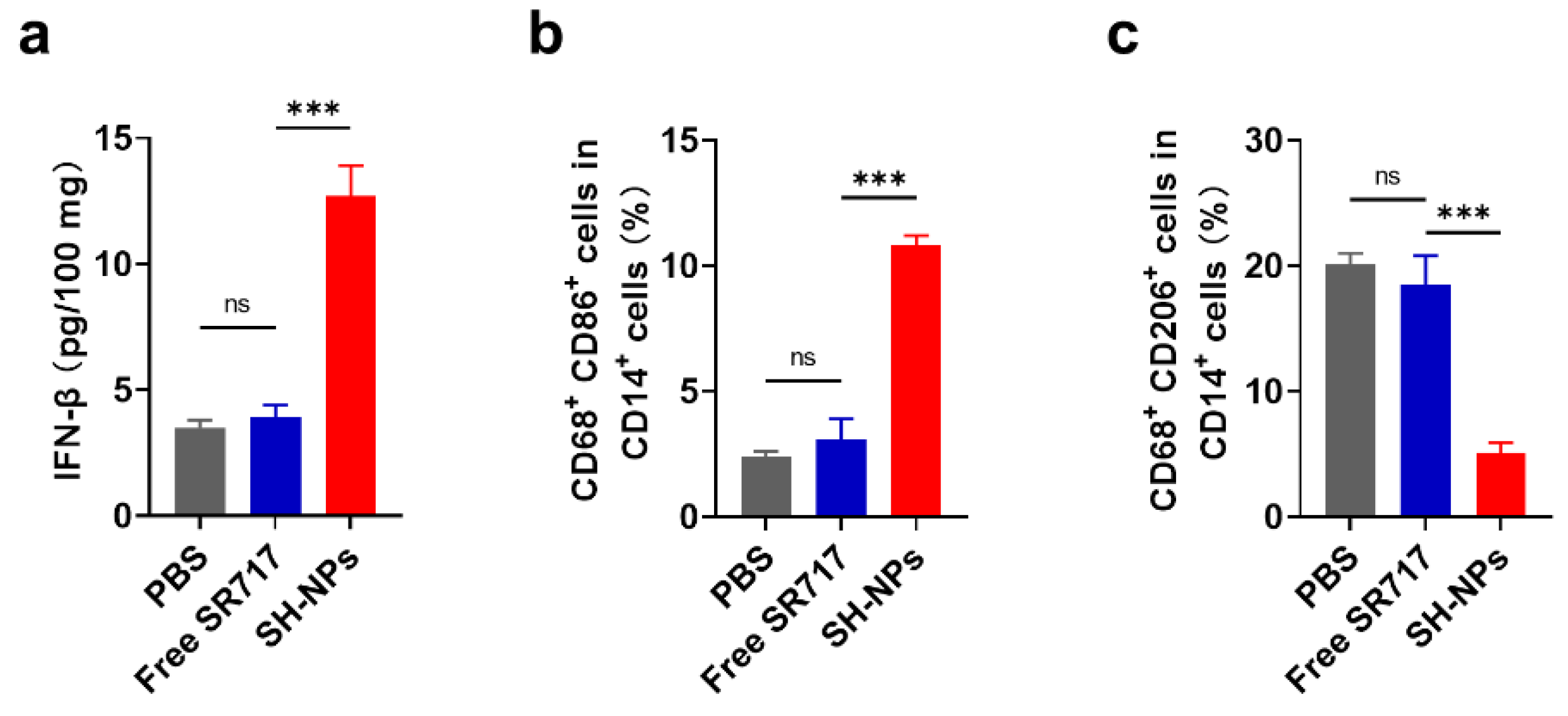
3.5. STING Activation in Human Tissues
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, C.; Zhang, S.J.A.A.N.M. Advantages of nanomedicine in cancer therapy: A review. ACS Appl. Nano Mater. 2023, 6, 22594–22610. [Google Scholar] [CrossRef]
- Kruger, S.; Ilmer, M.; Kobold, S.; Cadilha, B.L.; Endres, S.; Ormanns, S.; Schuebbe, G.; Renz, B.W.; D’Haese, J.G.; Schloesser, H.J.J. o. E.; et al. Advances in cancer immunotherapy 2019–latest trends. J. Exp. Clin. Cancer Res. 2019, 38, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mathew, D.; Marmarelis, M.E.; Foley, C.; Bauml, J.M.; Ye, D.; Ghinnagow, R.; Ngiow, S.F.; Klapholz, M.; Jun, S.; Zhang, Z.J.S. Combined JAK inhibition and PD-1 immunotherapy for non–small cell lung cancer patients. Science 2024, 384, eadf1329. [Google Scholar] [CrossRef]
- Lyu, J.; Bai, L.; Li, Y.; Wang, X.; Xu, Z.; Ji, T.; Yang, H.; Song, Z.; Wang, Z.; Shang, Y.J.N.C. Plasma proteome profiling reveals dynamic of cholesterol marker after dual blocker therapy. Nat. Commun. 2024, 15, 3860. [Google Scholar] [CrossRef]
- Xu, Y.; Yan, J.; Tao, Y.; Qian, X.; Zhang, C.; Yin, L.; Gu, P.; Liu, Y.; Pan, Y.; Tang, R.J.S. Pituitary hormone α-MSH promotes tumor-induced myelopoiesis and immunosuppression. Science. 2022, 377, 1085–1091. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, P.J.T. i. p. s. Preserving the CTLA-4 checkpoint for safer and more effective cancer immunotherapy. Trends Pharmacol. Sci. 2020, 414, 412–418. [Google Scholar] [CrossRef]
- Rowshanravan, B.; Halliday, N.; Sansom, D.M.J.B. , The Journal of the American Society of Hematology. CTLA-4: a moving target in immunotherapy. J. Am. Soc. Hematol. 2018, 131, 58–67. [Google Scholar]
- Sharma, P.; Allison, J.P.J.S. The future of immune checkpoint therapy. J. Am. Soc. Hematol. 2015, 348, 56–61. [Google Scholar] [CrossRef] [PubMed]
- de Miguel, M.; Calvo, E.J.C. c. Clinical challenges of immune checkpoint inhibitors. Cancer Cell. 2020, 38, 326–333. [Google Scholar] [CrossRef]
- Sharma, P.; Allison, J.P.J.N.R.I. Dissecting the mechanisms of immune checkpoint therapy. Nat. Rev. Immunol. 2020, 20, 75–76. [Google Scholar] [CrossRef]
- Topalian, S.L.; Drake, C.G.; Pardoll, D.M.J.C. c. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer cell. 2015, 27, 450–461. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Siddiqui, B.A.; Anandhan, S.; Yadav, S.S.; Subudhi, S.K.; Gao, J.; Goswami, S.; Allison, J.P.J.C. d. The next decade of immune checkpoint therapy. Cancer Disco. 2021, 11, 838–857. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.A.; Minn, A.J.J.I. Combination cancer therapy with immune checkpoint blockade: mechanisms and strategies. Cancer Disco. 2018, 48, 417–433. [Google Scholar] [CrossRef] [PubMed]
- Pardoll, D.M.J.N. r. c. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer. 2012, 12, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Bitton, K.; Michot, J.-M.; Barreau, E.; Lambotte, O.; Haigh, O.; Marabelle, A.; Voisin, A.-L.; Mateus, C.; Rémond, A.-L.; Couret, C.J.A.J. o. O. Prevalence and clinical patterns of ocular complications associated with anti-PD-1/PD-L1 anticancer immunotherapy. Am. J. Ophthalmol. 2019, 202, 109–117. [Google Scholar] [CrossRef]
- Ichihara, E.; Harada, D.; Inoue, K.; Sato, K.; Hosokawa, S.; Kishino, D.; Watanabe, K.; Ochi, N.; Oda, N.; Hara, N.J.L.C. The impact of body mass index on the efficacy of anti-PD-1/PD-L1 antibodies in patients with non-small cell lung cancer. Lung Cancer. 2020, 139, 140–145. [Google Scholar] [CrossRef] [PubMed]
- de Jong, F.C.; Rutten, V.C.; Zuiverloon, T.C.; Theodorescu, D.J.I. j. o. m. s. Improving anti-PD-1/PD-L1 therapy for localized bladder cancer. Int. J. Mol. Sc. 2021, 22, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Mosallanejad, K.; Kagan, J.C.J.I.; biology, c. Control of innate immunity by the cGAS-STING pathway. Immunol. Cell Biol. 2022, 100, 409–423. [Google Scholar] [CrossRef] [PubMed]
- Hopfner, K.-P.; Hornung, V.J.N. r. M. c. b. Molecular mechanisms and cellular functions of cGAS–STING signalling. Nat. Rev. Mol. Cell Biol. 2020, 21, 501–521. [Google Scholar] [CrossRef]
- Decout, A.; Katz, J.D.; Venkatraman, S.; Ablasser, A.J.N.R.I. The cGAS–STING pathway as a therapeutic target in inflammatory diseases. Nat. Rev. Immuno. 2021, 21, 548–569. [Google Scholar] [CrossRef]
- Motwani, M.; Pesiridis, S.; Fitzgerald, K.A.J.N.R.G. DNA sensing by the cGAS–STING pathway in health and disease. Nat. Rev. Genet. 2019, 20, 657–674. [Google Scholar] [CrossRef] [PubMed]
- Celias, D.P.; Hänggi, K.; Ruffell, B.J.C.R. Investigating the mechanisms involved in HMGB1-dependent DNA uptake and STING activation in dendritic cells. Cancer Res. 2023, 83, 678–678. [Google Scholar] [CrossRef]
- Umemura, K.; Kawamoto, Y.; Takahashi, Y.; Takakura, Y.J.M.P. Development of a Cytosolic DNA Sensor Agonist Using GALA Peptide-Conjugated DNA and Long Single-Stranded DNA. Mol. Pharmaceutics. 2024, 21, 1204–1213. [Google Scholar] [CrossRef] [PubMed]
- Morehouse, B.R.; Govande, A.A.; Millman, A.; Keszei, A.F.; Lowey, B.; Ofir, G.; Shao, S.; Sorek, R.; Kranzusch, P.J.J.N. STING cyclic dinucleotide sensing originated in bacteria. Nature. 2020, 586, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hou, X.; Du, S.; Xue, Y.; Yan, J.; Kang, D.D.; Zhong, Y.; Wang, C.; Deng, B.; McComb, D.W.J.N.N. Close the cancer–immunity cycle by integrating lipid nanoparticle–mRNA formulations and dendritic cell therapy. Nat. Nanotechnol. 2023, 18, 1364–1374. [Google Scholar] [CrossRef] [PubMed]
- Crunkhorn, S.J.N.R.I. Strengthening the sting of immunotherapy. Nat. Nanotechnol. 2020, 20, 589. [Google Scholar]
- Nguyen, P.H.D.; Jayasinghe, M.K.; Le, A.H.; Peng, B.; Le, M.T.J.A. n. Advances in drug delivery systems based on red blood cells and their membrane-derived nanoparticles. ACS nano. 2023, 17, 5187–5210. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Su, Y.-Y.; Jiang, X.-C.; Gao, J.-Q. J. D. D.; Research, T. Cell membrane-coated nanoparticles: a novel multifunctional biomimetic drug delivery system. Drug Deliv. Transl. Res. 2023, 13, 716–737. [Google Scholar] [CrossRef] [PubMed]
- Vyas, K.; Rathod, M.; Patel, M.M.J.N.N. , Biology; Medicine. Insight on nano drug delivery systems with targeted therapy in treatment of oral cancer. Nanomedicine: NBM. 2023, 49, 102662. [Google Scholar] [CrossRef]
- Senti, M.E.; Del Valle, L.G.; Schiffelers, R.M.J.A.D.D.R. mRNA delivery systems for cancer immunotherapy: lipid nanoparticles and beyond. Adv. Drug Deliv. 2024, 115190. [Google Scholar] [CrossRef]
- Zhou, Q.; Dutta, D.; Cao, Y.; Ge, Z.J.A. n. Oxidation-responsive PolyMOF nanoparticles for combination photodynamic-immunotherapy with enhanced STING activation. ACS Nano. 2023, 17, 9374–9387. [Google Scholar] [CrossRef]
- Lu, Q.; Chen, R.; Du, S.; Chen, C.; Pan, Y.; Luan, X.; Yang, J.; Zeng, F.; He, B.; Han, X.J.B. Activation of the cGAS-STING pathway combined with CRISPR-Cas9 gene editing triggering long-term immunotherapy. Biomaterials. 2022, 291, 121871. [Google Scholar] [CrossRef]
- Gou, S.; Liu, W.; Wang, S.; Chen, G.; Chen, Z.; Qiu, L.; Zhou, X.; Wu, Y.; Qi, Y.; Gao, Y.J.N.L. Engineered nanovaccine targeting Clec9a+ dendritic cells remarkably enhances the cancer immunotherapy effects of STING agonist. Nano Lett. 2021, 21, 9939–9950. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Y.; Wei, C.; Deng, Y.; Chen, H.; Shen, J.; Ke, H.J.J. o. M. C. B. Biomineralized iron oxide–polydopamine hybrid nanodots for contrast-enhanced T 1-weighted magnetic resonance imaging and photothermal tumor ablation. J. Mater. Chem. 2021, 9, 1781–1786. [Google Scholar]
- Li, M.; Wang, Y.; Li, T.; Zhang, J.; Wang, X.; Luo, J.; You, M.; Yang, T.; Deng, Y.; Yang, H.J.A.B. Albumin-templated platinum (II) sulfide nanodots for size-dependent cancer theranostics. Acta Biomater. 2023, 155, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Zhang, Y.; Zhu, J.; Zhang, F.; Xu, A. a.; Zhou, T.; Li, Y.; Liu, M.; Ke, H.; Yang, T.J.A.M. A pH-Activatable Copper-Biomineralized Proenzyme for Synergistic Chemodynamic/Chemo-Immunotherapy against Aggressive Cancers. Adv. Mater. 2023, 35, 2210201. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Liu, M.; Yang, T.; Luo, J.; Wei, C.; Shen, J.; Song, X.; Ke, H.; Sun, P.; Guo, M.J.J. o. C. R. Self-activated arsenic manganite nanohybrids for visible and synergistic thermo/immuno-arsenotherapy. J. Control. Release. 2022, 350, 761–776. [Google Scholar] [CrossRef]
- Liu, B.; Jiao, J.; Xu, W.; Zhang, M.; Cui, P.; Guo, Z.; Deng, Y.; Chen, H.; Sun, W.J.A.M. Highly Efficient Far-Red/NIR-Absorbing Neutral Ir (III) Complex Micelles for Potent Photodynamic/Photothermal Therapy. Adv. Mater. 2021, 33, 2100795. [Google Scholar] [CrossRef]
- Xue, X.; Qu, H.; Li, Y. Stimuli-responsive crosslinked nanomedicine for cancer treatment. Exploration. 2022, 2, 20210134. [Google Scholar] [CrossRef]
- Lucas, E.D.; Schafer, J.B.; Matsuda, J.; Kraus, M.; Burchill, M.A.; Tamburini, B.A.J. PD-L1 reverse signaling in dermal dendritic cells promotes dendritic cell migration required for skin immunity. Cell reports. 2020 33, 2, 108258. [Google Scholar]
- Lei, H.; Pei, Z.; Jiang, C.; et al. Recent progress of metal-based nanomaterials with anti-tumor biological effects for enhanced cancer therapy. Exploration. 2023, 3, 20220001. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



