Submitted:
24 July 2024
Posted:
25 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Methods of Membrane Modification
Modification of membranes with magnesium chloride (MgCl2)
Modification of membranes with reduced graphene oxide
Modification of membranes with chitosan
2.2.2. Scanning Electron Microscopy
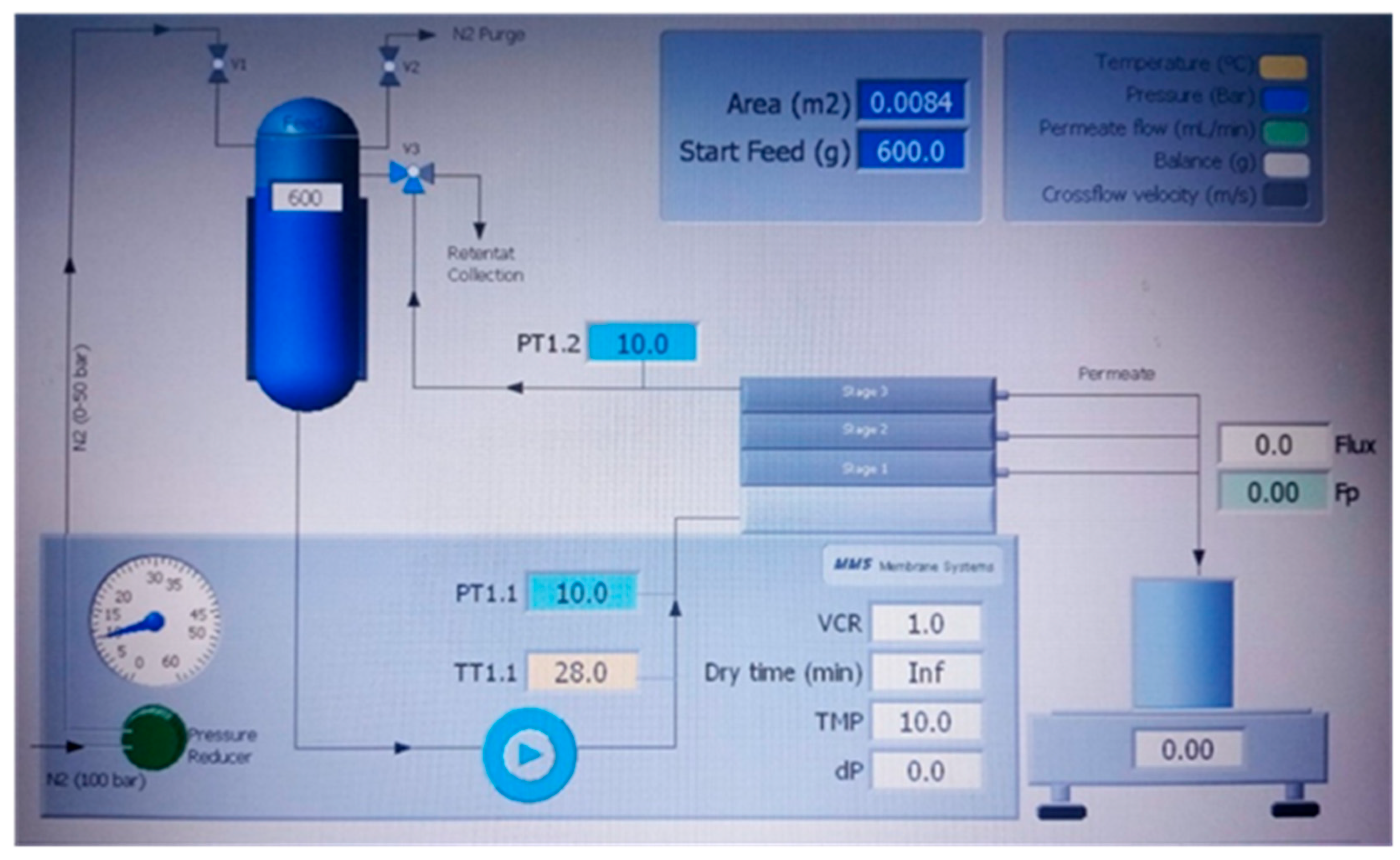
2.2.3. Filtration Experiments
Solvent permeability test
Tests with sulfamethoxazole pollutant
2.2.4. Data Processing
3. Results
3.1. Membrane Modification
3.2. Macroscopic Analysis through Operational Parameters
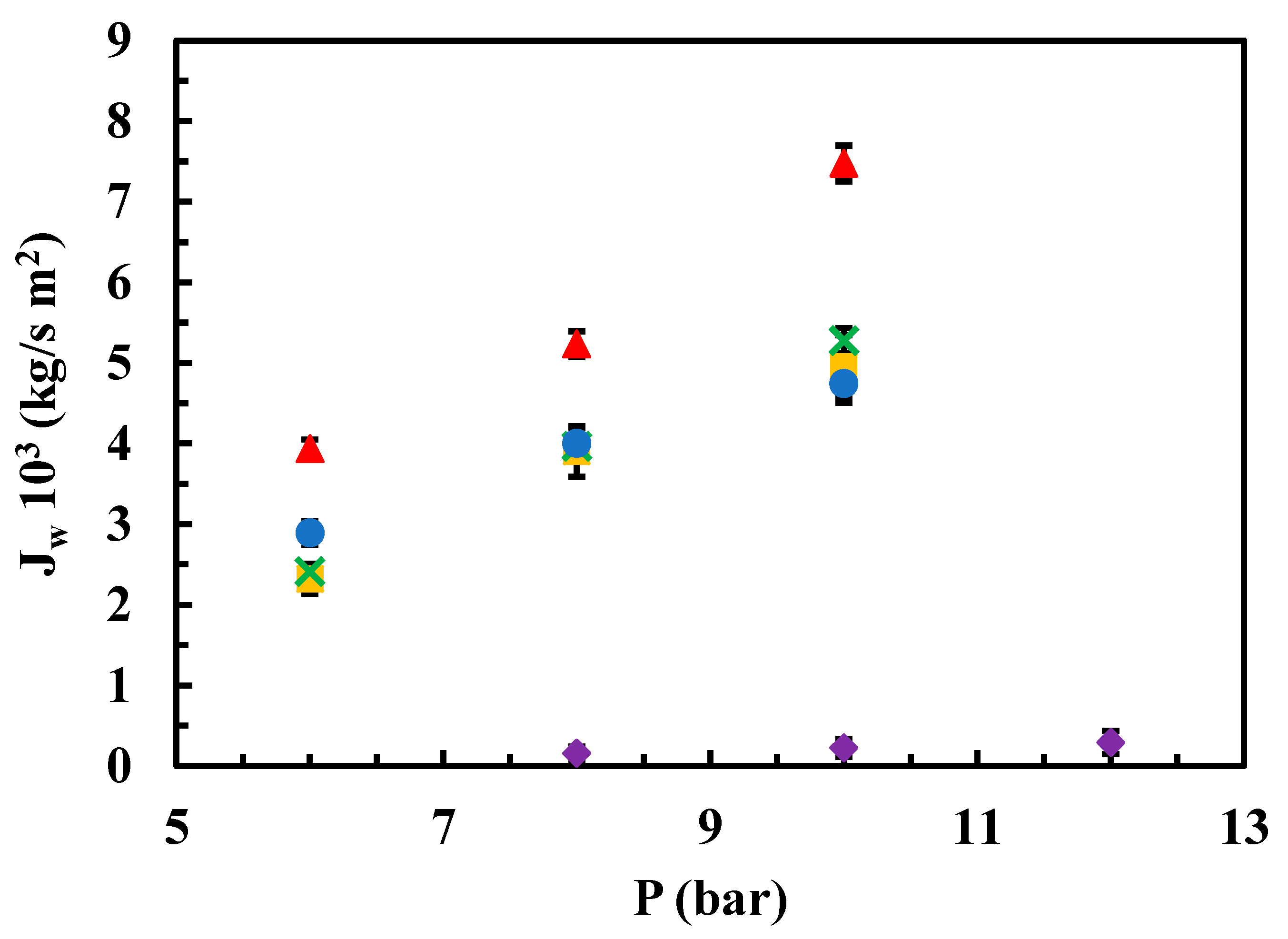
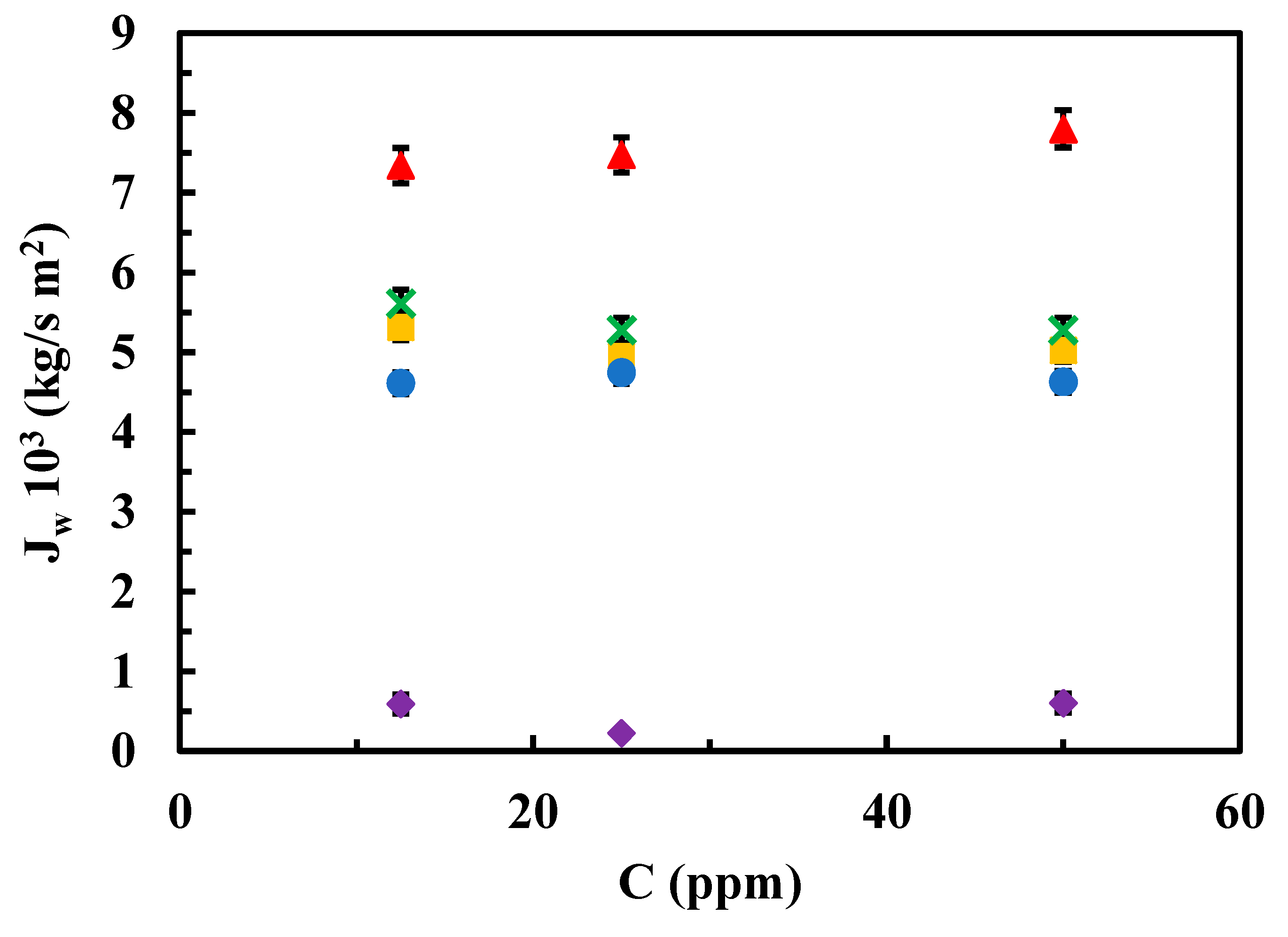
3.2.1. Permeability
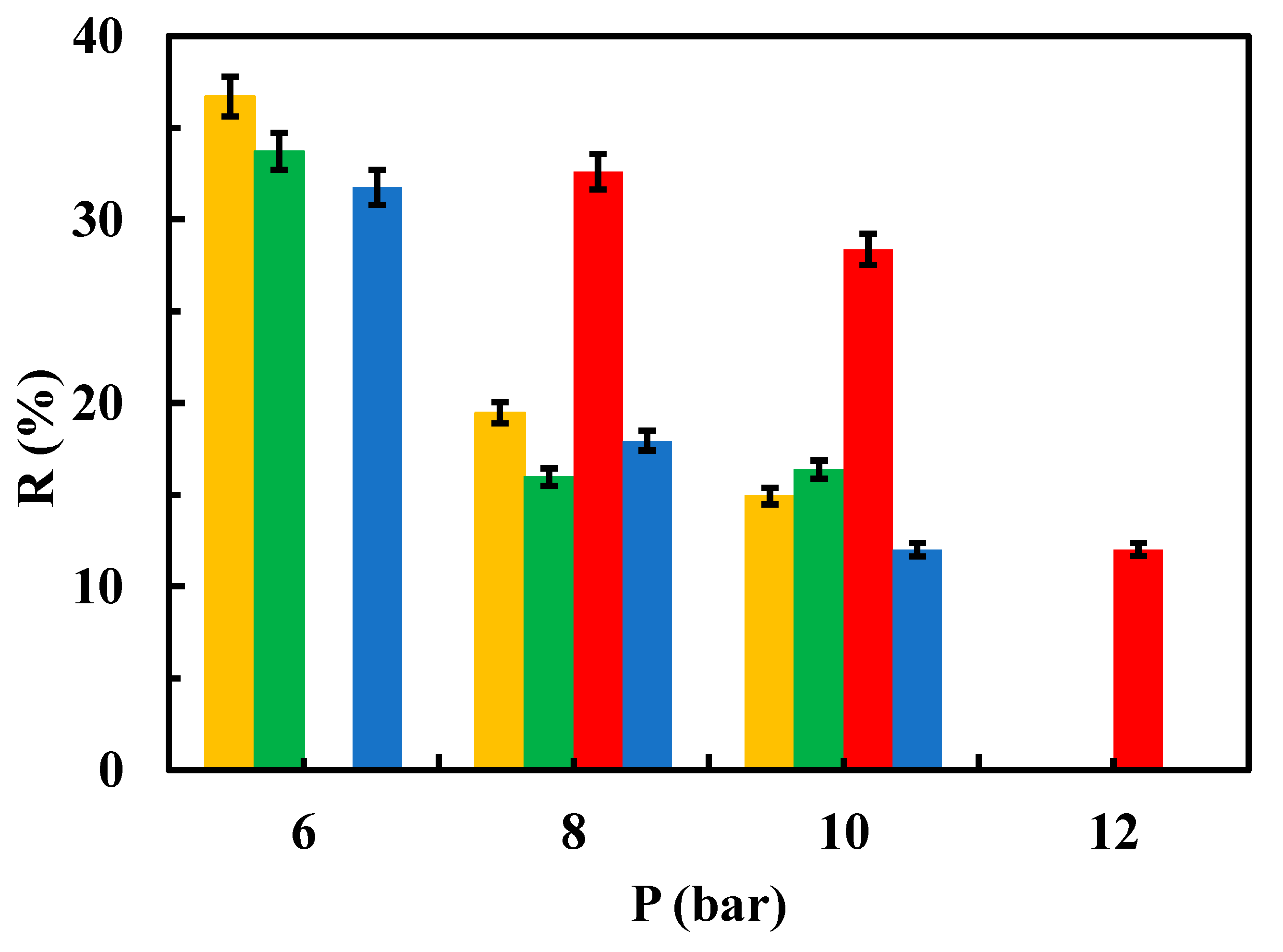
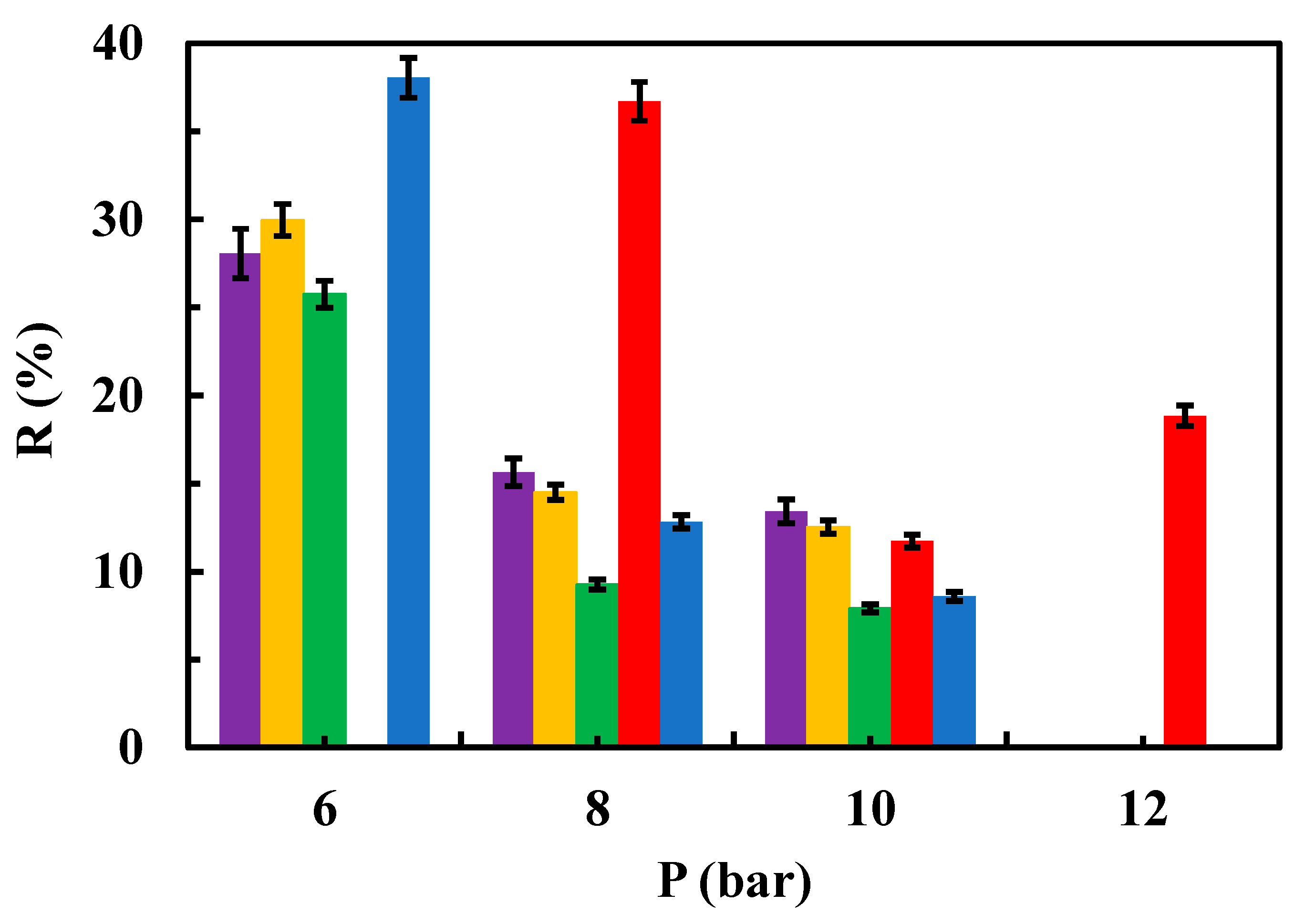
3.2.2. Sulfamethoxazole Removal
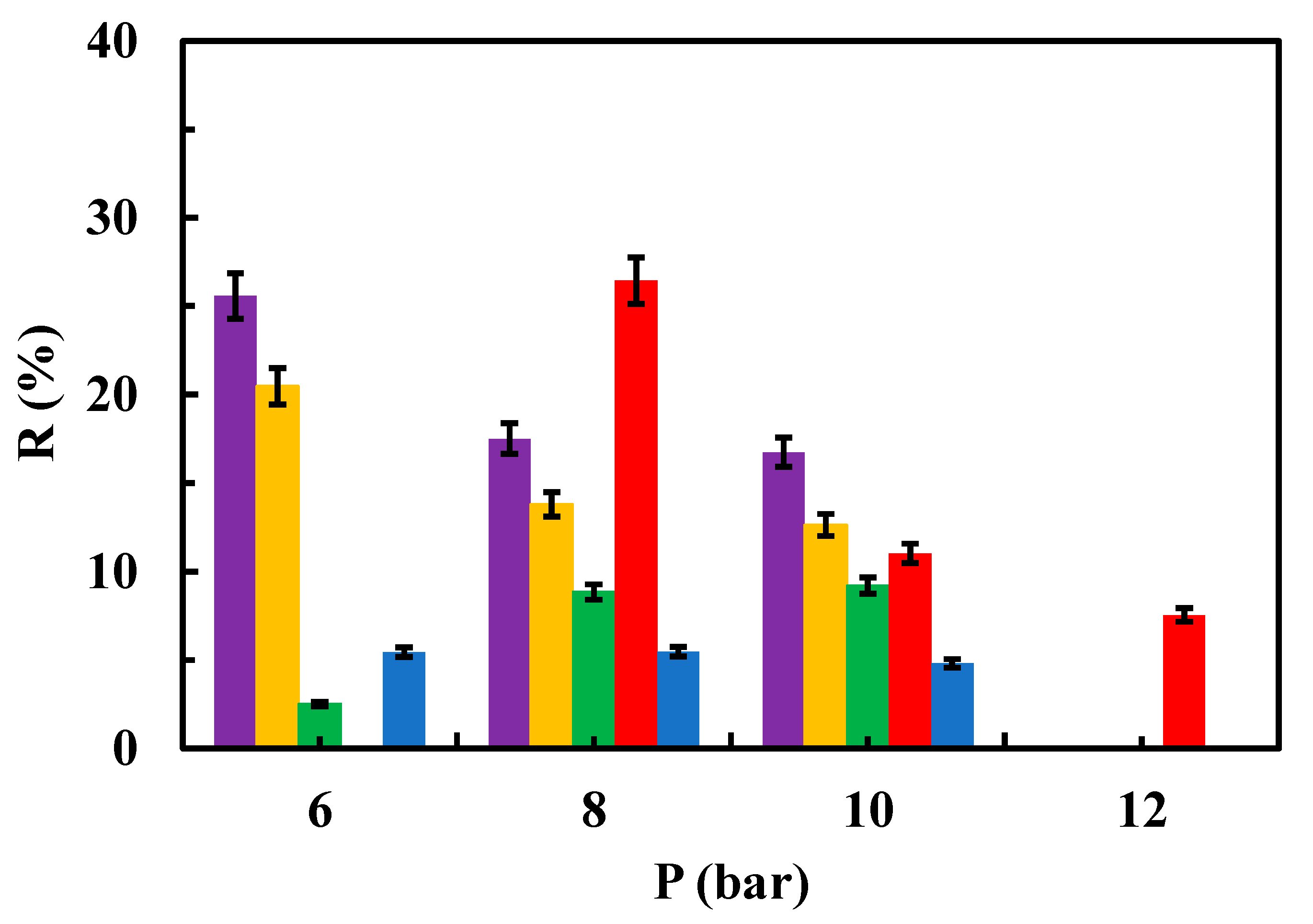
3.3. Fouling Parameters
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ng, L.Y.; Mohammad, A.W.; Leo, C.P.; Hilal, N. Polymeric membranes incorporated with metal/metal oxide nanoparticles: a comprehensive review. Desalination 2013, 308, 15–33. [Google Scholar] [CrossRef]
- Upreti, D.; Rajendran, A.; Lenka, N.; Srivastava, R.; Sen Gupta, R.; Maiti, B.; Bose, S.; Patro, T.U. Designing a robust biocompatible porous polymeric membrane using Laponite and graphene oxide for versatile and selective adsorption of water contaminants. Chem. Eng. J. 2023, 464, 142738. [Google Scholar] [CrossRef]
- Jiang, S.; Ladewig, B.P. Green synthesis of polymeric membranes: Recent advances and future prospects. Curr. Opin. Green Sustain. Chem. 2020, 21, 1–8. [Google Scholar] [CrossRef]
- Yahya, L.A.; Tobiszewski, M.; Kubica, P.; Koronkiewicz, S.; Vakh, C. Polymeric porous membranes as solid support and protective material in microextraction processes: A review. TrAC, Trends Anal. Chem. 2024, 173, 117651. [Google Scholar] [CrossRef]
- Al-Maliki, R.M.; Alsalhy, Q.F.; Al-Jubouri, S.; Salih, I.K.; AbdulRazak, A.A.; Shehab, M.A.; Németh, Z.; Hernadi, K. Classification of nanomaterials and the effect of graphene oxide (GO) and recently developed nanoparticles on the ultrafiltration membrane and their applications: a review. Membranes. 2022, 12, 1043. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Ye, W.; Zhong, K.; Shen, J.; Jullok, N.; Sotto, A.; Van der Bruggen, B. Enhancement of polyethersulfone (PES) membrane doped by monodisperse Stöber silica for water treatment. Chem. Eng. Process. 2016, 107, 194–205. [Google Scholar] [CrossRef]
- Chauhan, D.; Nagar, P.K.; Pandey, K.; Pandey, H. Simulations of novel mixed-patterned membrane surfaces: Enhanced hydrodynamics and concentration polarization to mitigate fouling in water treatment. J. Water Process Eng. 2024, 62, 105371. [Google Scholar] [CrossRef]
- Alkhouzaam, A.; Qiblawey, H. Novel polysulfone ultrafiltration membranes incorporating polydopamine functionalized graphene oxide with enhanced flux and fouling resistance. J. Memb. Sci. 2021, 620, 118900. [Google Scholar] [CrossRef]
- Saeki, D.; Minami, R.; Matsuyama, H. Effects of operating conditions on biofouling in crossflow ultrafiltration membrane processes. Sep. Purif. Technol. 2017, 189, 138–144. [Google Scholar] [CrossRef]
- Ng, C.Y.; Mohammad, A.W.; Ng. L.Y.; Jahim, J.M. Membrane fouling mechanisms during ultrafiltration of skimmed coconut milk. J. Food Eng. 2014, 142, 190–200. [Google Scholar] [CrossRef]
- Kammakakam, I.; Lai, Z. Next-generation ultrafiltration membranes: A review of material design, properties, recent progress, and challenges. Chemosphere. 2023, 316, 137669. [Google Scholar] [CrossRef] [PubMed]
- Ocakoglu, K.; Dizge, N.; Colak, S.G.; Ozay, Y.; Bilici, Z.; Yalcin, M.S.; Ozdemir, S.; Yatmaz, H.C. Polyethersulfone membranes modified with CZTS nanoparticles for protein and dye separation: Improvement of antifouling and self-cleaning performance. Coll. Surf. A Physicochem. Eng. Asp. 2021, 616, 126230. [Google Scholar] [CrossRef]
- Al-Araji, D.; Al-Ani, F.; Alsalhy, Q. The Permeation and separation characteristics of polymeric membranes incorporated with nanoparticles for dye removal and interaction mechanisms between polymer and nanoparticles: a mini review. Eng. Technol. J. 2022, 40, 1399–1411. [Google Scholar] [CrossRef]
- Palanisamy, G.; Muhammed, A.P.; Thangarasu, S.; Oh, T.H. Investigating the sulfonated chitosan/polyvinylidene fluoride-based proton exchange membrane with fSiO2 as filler in microbial fuel cells. Membranes, 2023, 13, 758. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.B.; Sun, X.F.; Wang, L.; Wang, S.Y.; Liu, R.D.; Wang, S.G. Polyethersulfone membranes modified with D-tyrosine for biofouling mitigation: synergistic effect of surface hydrophility and anti-microbial properties. Chem. Eng. J. 2017, 311, 135–142. [Google Scholar] [CrossRef]
- Lalia, B.S.; Kochokodan, V.; Hashaike, R.; Hilal, N. A review on membrane fabrication: Structure, properties and performance relationship. Desalination. 2013, 326, 77–95. [Google Scholar] [CrossRef]
- Cano, M.; Khan, U.; Sainsbury, T.; O’Neill, A.; Wang, Z.; McGovern, I.T.; Maser, W.K.; Benito, A.M.; Coleman, J.N. Improving the mechanical properties of graphene oxide-based materials by covalent attachment of polymer chains. Carbon. 2013, 52, 363–371. [Google Scholar] [CrossRef]
- Chang, X.; Wang, Z.; Quan, S.; Xu, Y.; Jiang, Z.; Shao, L. Exploring the synergetic effects of graphene oxide (GO) and polyvinylpyrrodione (PVP) on poly(vinylylidenefluoride) (PVDF) ultrafiltration membrane performance. Appl. Surf. Sci. 2014, 316, 537–548. [Google Scholar] [CrossRef]
- Zinadini, S.; Zinatizadeh, A.A.; Rahimi, M.; Vatanpour, V.; Zangeneh, H. Preparation of a novel antifouling mixed matrix PES membrane by embedding graphene oxide nanoplates. J. Membr. Sci. 2014, 453, 292–301. [Google Scholar] [CrossRef]
- Xu, B.; Wang, J. Radiation-induced modification of chitosan and applications for water and wastewater treatment. J. Clean. Prod. 2024, 467, 142924. [Google Scholar] [CrossRef]
- Spoiala, A.; Ilie, C.I.; Ficai, D.; Ficai, A.; Andronescu, E. Chitosan based nanocomposite polymeric membranes for water purification-a review. Materials. 2021, 14, 2091. [Google Scholar] [CrossRef]
- Samoila, P.; Humelnicu, A.C.; Cojocaru, M.I.C.; Harabagiu, V. Chitin and chitosan for water purification. In Chitin and chitosan properties and applications, 1st ed.; Lambertus, A.M.; van den Broken, Boeriu, C.G., Eds.; John Wiley & Sons Ltd, 2020, pp. 429–460.
- Mu, T.; Tan, G.; Du, G.; He, L.; Li, Z.; Li, X. Novel charged chitosan composite nanofiltration membranes containing mesogenic group. Polym. Eng. Sci. 2017, 57, 22–30. [Google Scholar] [CrossRef]
- Jiménez-Gómez, C.P.; Cecilia, J.A. Chitosan: a natural biopolymer with a wide and varied range of applications. Molecules. 2020, 25, 3981. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Li, Y.; Zhao, X.; Wang, J.; Zhu, X.; Lai, C.; Wu, D.; Cheng, X.; Xu, J.; Liang, H. (2023) pH-Responsive chitosan sacrificial layer for simultaneous enhancement of ultrafiltration performance and sustainable membrane fouling control. ACS Appl. Polym. Mater. 2023, 5, 6875–6885. [Google Scholar] [CrossRef]
- Hamzah, S.; Ali, N.; Mohammad, A.W.; Ariffin, M.M.; Ali, A. Desing of chitosan/Psf self-assembly membrane to mitigate fouling and enhance performance in trypsin separation. J. Chem. Technol. Biotechnol. 2012, 87, 1157–1166. [Google Scholar] [CrossRef]
- Ghiggi, F.F.; Pollo, L.D.; Cardozo, N.D.M.; Tessaro, I.C. Preparation and characterization of polyethersulfone/N-phthalogy-chitosan ultrafiltration membrane with antifouling property. Eur. Polym. J. 2017, 92, 61–70. [Google Scholar] [CrossRef]
- Darwish, N.B.; Abdulgader, H.A.; AlRomaih, H.; Alalawi, A. Effect of ultrafiltration membranes modifications by chitosan on humic acid fouling. J. Water Process. Eng. 2019, 27, 32–36. [Google Scholar] [CrossRef]
- ICM. Institut de Ciènces del Mar. Microscopía Electronica. Available online: https://micro.icm.csic.es/es/site-page/hitachi-s-3500n (accessed on 15 November 2022).
- Murcia, M.D.; Hidalgo, A.M.; Gómez, M.; León, G.; Gómez, E.; Martínez, M. Ultrafiltration membranes modified with reduced graphene oxide: Effect on methyl green removal from aqueous solution. Materials, 2023, 16, 1369. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, M.; Ahmed, R.; Ali, A.W.; Lee, S.J. SEM and ESEM techniques used for analysis of asphalt binder and mixture: A state of the art review. Constr. Build. Mater. 2018, 186, 313–329. [Google Scholar] [CrossRef]
- Hidalgo, A.M.; Gómez, M.; Murcia, M.D.; León, G.; Miguel, B.; Gago, I.; Martínez, P.M. Ibuprofen removal by graphene oxide and reduced graphene oxide coated polysulfone nanofiltration membranes. Membranes 2022, 12, 562. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, M.; Chen, M.; Ma, L.; Yang, B.; Li, L.; Tu, W. A polydopamine-modified reduced graphene oxide (RGO)/MOFs nanocomposite with fast rejection capacity for organic dye. Chem. Eng. J. 2019, 359, 47–57. [Google Scholar] [CrossRef]
- Mahlangu, O.T.; Motsa, M.M.; Nkambule, T.I.; Mamba, B.B. Rejection of trace organic compounds by membrane processes: mechanisms, challenges, and opportunities. Rev. Chem. Eng. 2022, 39, 875–910. [Google Scholar] [CrossRef]
- Bellona, C.; Drewes, E. The role of membrane surface charge and solute physico-chemical properties in the rejection of organic acids by NF membranes. J. Membr. Sci. 2005, 249, 227–234. [Google Scholar] [CrossRef]
- Verliefde, A.; Van der Meeren, P.; Van der Bruggen, B. Solution-diffusion processes. In Encyclopedia of Membrane Science and Technology; Hoek, E.M.V., Tarabara, V.V., Eds.; Wiley and Sons: Hoboken, NY, USA, 2013; p. 4013. [Google Scholar]
- Zhao, C.; Xue, J.; Ran, F.; Sun, S. Modification of polyethersulfone membranes – A review of methods. Prog. Mater. Sci. 2013, 58, 76–150. [Google Scholar] [CrossRef]
- Rahimpour, A.; Madaeni, S.S.; Taheri, A.H.; Mansourpanah, Y. Coupling TiO2 nanoparticles with UV irradiation for modification of polyethersulfone ultrafiltration membranes. J. Membr. Sci. 2008, 313, 158–169. [Google Scholar] [CrossRef]
- Deng, B.; Li, J.; Hou, Z.; Yao, S.; Shi, L.; Liang, G.; Sheng, K. Microfiltration membranes prepared from polyethersulfone powder grafted with acrylic acid by simultaneous irradiation and their pH dependence. Radiat. Phys. Chem. 2007, 77, 898–906. [Google Scholar] [CrossRef]
- Celik, E.; Park, H.; Choi, H.; Choi, H. Carbon nanotube blended polyethersulfone membranes for fouling control in water treatment. Water Res. 2011, 45, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Macedo, A.; Duarte, E.; Pinho, M. The role of concentration polarization in ultrafiltration of ovine cheese whey. J. Membr. Sci. 2011, 381, 34–40. [Google Scholar] [CrossRef]
- Galanakis, C.M.; Chasiotis, S.; Botsaris, G.; Gekas, V. Separation and recovery proteins and sugars from Halloumi cheese whey. Food Res. Int. 2014, 65, 477–483. [Google Scholar] [CrossRef]
- Corbatón-Báguena, M.J.; Álvarez-Blanco, S.; Vicent-Vela, M.C. Cleaning of ultrafiltration membranes fouled with BSA by means of saline solutions. Sep. Purif. Technol. 2014, 125, 1–10. [Google Scholar] [CrossRef]
- Stanley, C.; Rau, D.C. Evidence for water structuring forces between surfaces. Curr. Opin. Colloid Interface Sci. 2011, 16, 551–556. [Google Scholar] [CrossRef]
- Botton, S.; Verliefde, A.R.D.; Quach, N.T.; Cornelissen, E.R. Surface characterisation of biofouled NF membranes: role of surface energy for improved rejection predictions. Water Sci. Technol. 2012, 66, 2122–2130. [Google Scholar] [CrossRef] [PubMed]



| Characteristics | Technical specifications |
|---|---|
| Supplier’s signature | Alfa Laval |
| Designation | GR95PP |
| Filtration type | Ultrafiltration |
| MWCO value (kDa | 2 |
| Active layer | Polyethersulfone |
| Support material | polypropylene |
| Operating pressure (bar) | 1 – 12 |
| Maximum tolerable pressure (bar) | 10 |
| Tolerated pH range | 1 – 13 |
| Temperature range (ºC) | 5 – 75 |
| Membrane | C (%) | O (%) | S (%) | Ca (%) | Si (%) | Al (%) | |
|---|---|---|---|---|---|---|---|
| Native | 67.1 | 21.9 | 10.9 | 0.2 | - | - | |
| Before | MgCl2-modified | ||||||
| filtering | rGO-modified | 68.8 | 19.7 | 11.3 | 0.2 | 0.1 | - |
| contaminant | Chitosan-modified | 67.5 | 18.3 | 13.7 | - | 0.1 | 0.5 |
| After | MgCl2-modified | 68.5 | 21 | 10.4 | - | 0.1 | - |
| filtering | rGO-modified | 71.1 | 17.5 | 11.3 | - | 0.1 | - |
| contaminant | Chitosan-modified | 67.9 | 20.5 | 11.4 | - | 0.1 | 0.2 |
| Membrane | Aw ∙ 108 (s/m) Before pollutant |
Aw ∙ 108 (s/m) After pollutant |
|---|---|---|
| Native | 2.1019 | - |
| MgCl2(0.5 g/L) modified | - | 0.4671 |
| MgCl2(1.0 g/L) modified | 0.4671 | 0.7358 |
| rGO modified | 0.1257 | 0.0794 |
| Chitosan modified | 1.0885 | 0.4506 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




