Submitted:
18 July 2024
Posted:
19 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1 Cell lines and Plasmids
2.2. Peptides
2.3. Production of Hybridomas
2.4. Purification of Antibodies
2.5. ELISA
2.6. Flow Cytometric Analysis
2.7. Western Blotting
3. Results
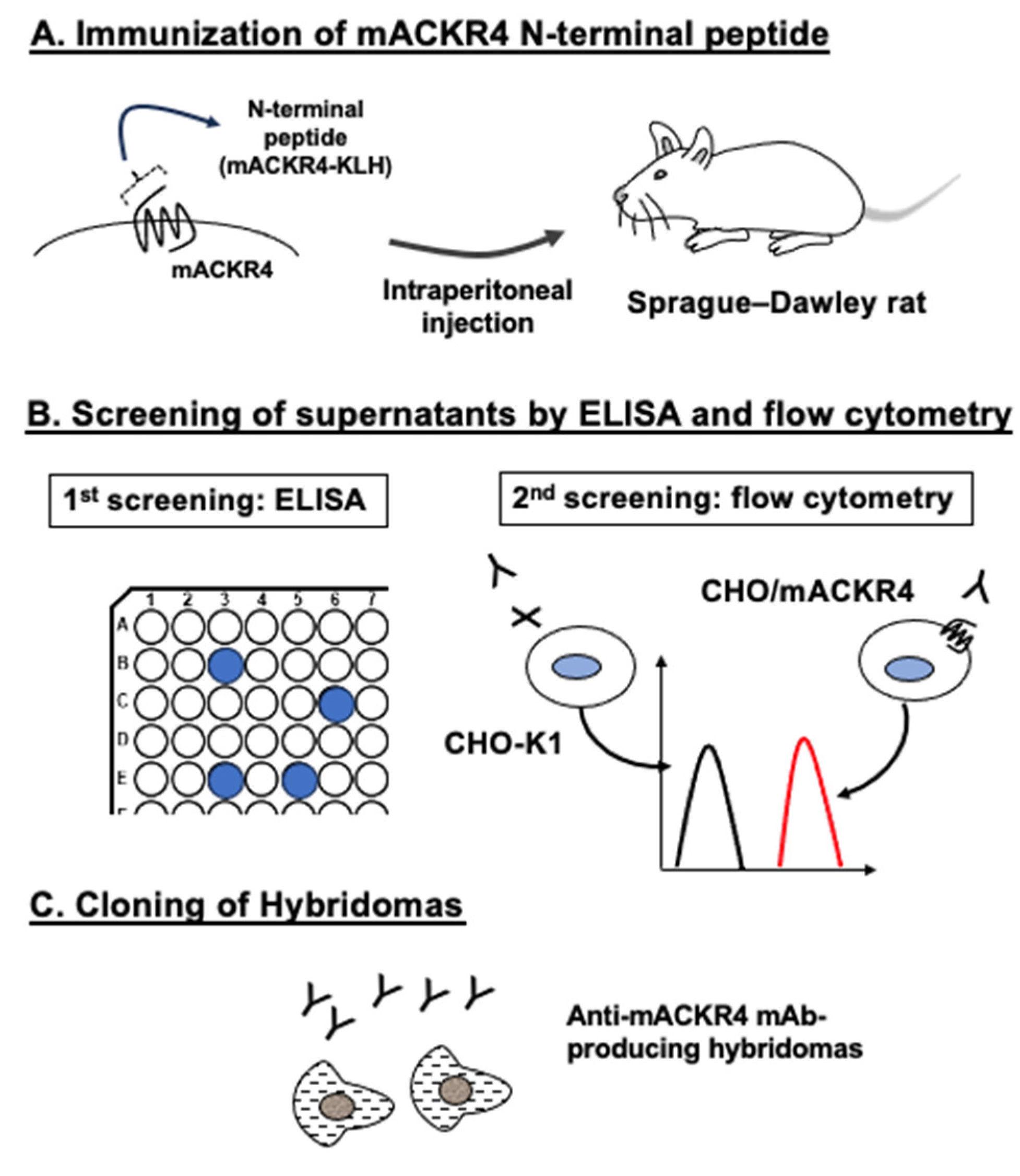
3.1. Development of Anti-mACKR4 mAbs Using N-Terminal Peptide Immunization
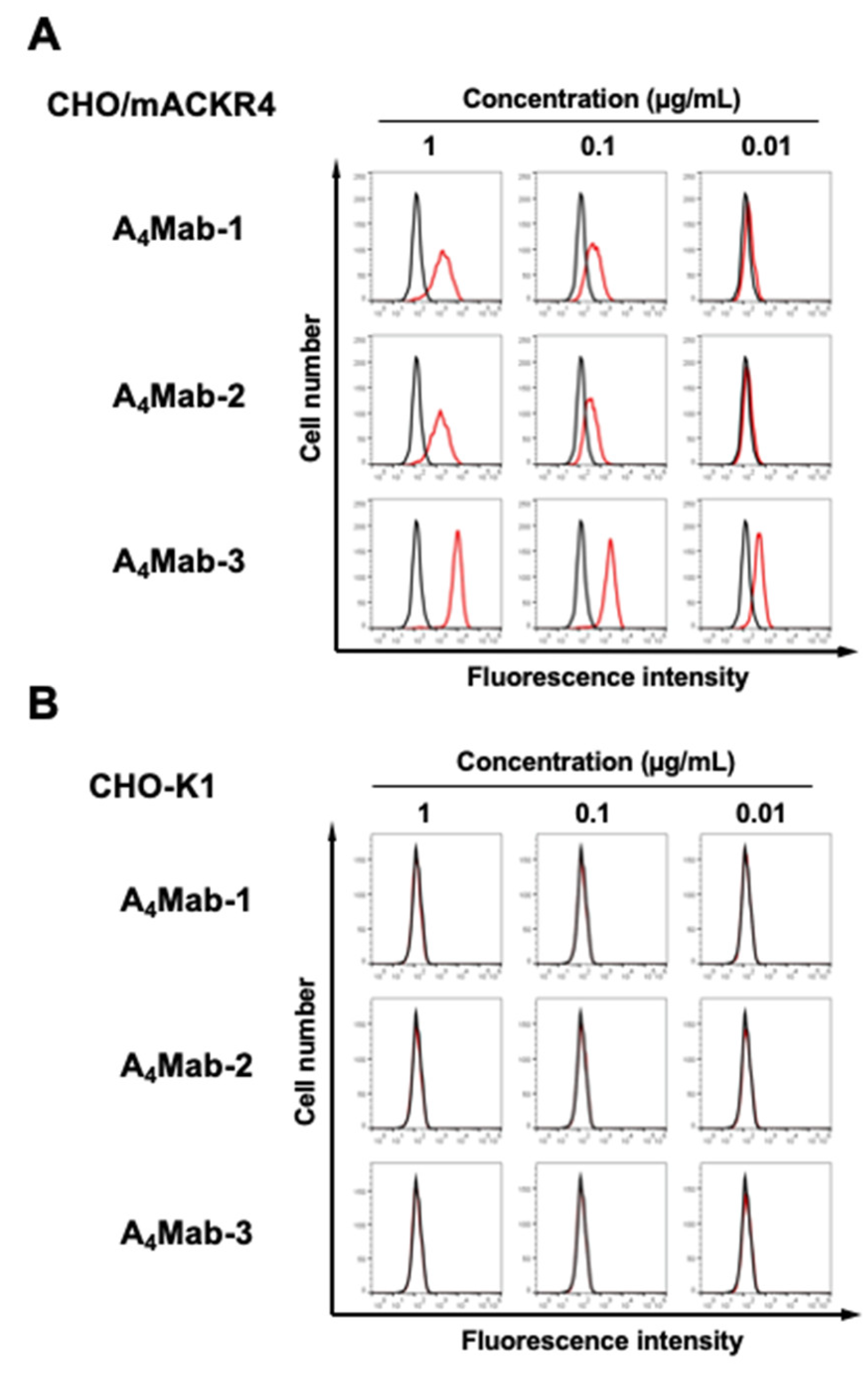
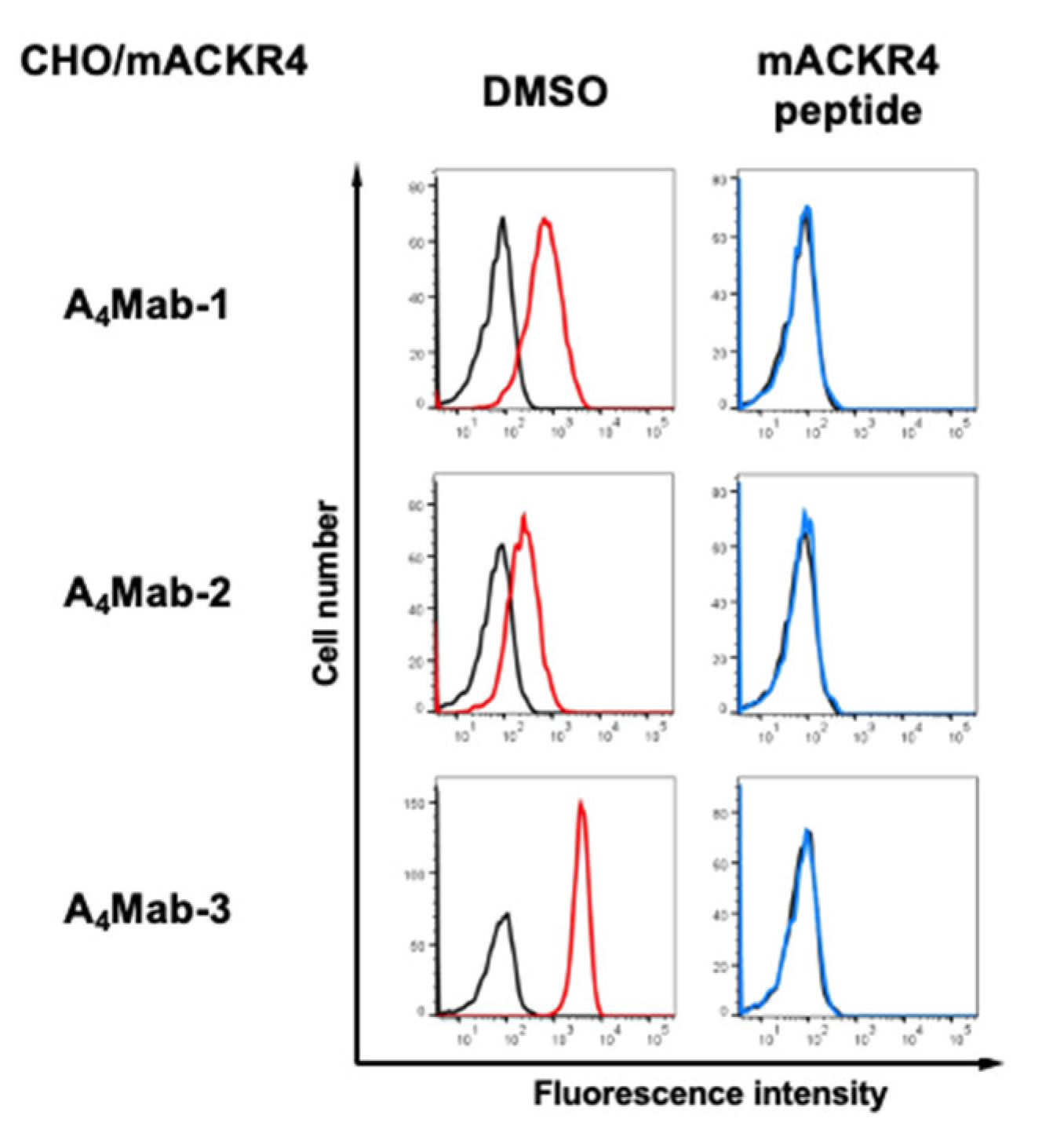
3.2. Flow Cytometric Analysis Using A4Mab-1, A4Mab-2, and A4Mab-3
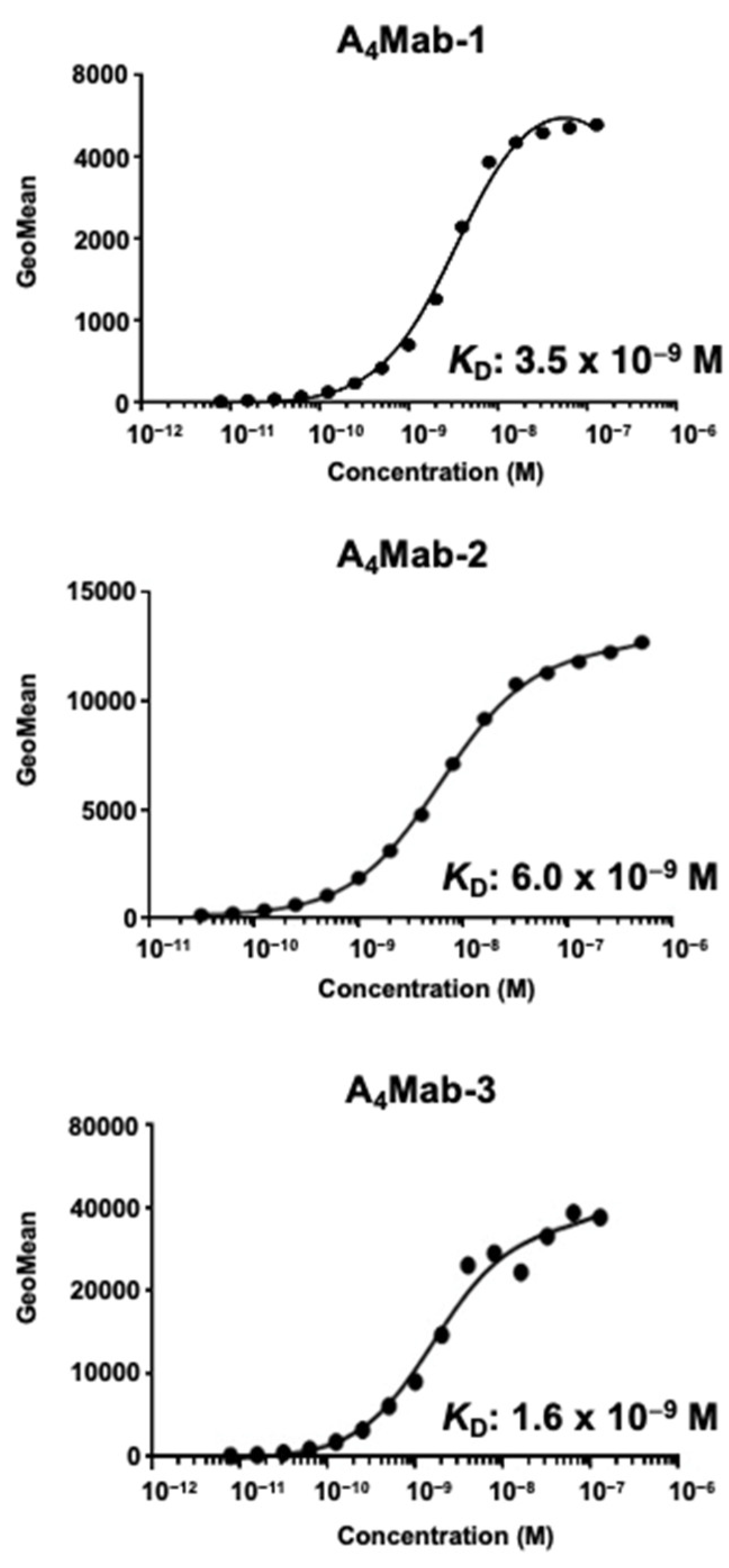
3.3. Determination of the Binding Affinity of A4Mab-1, A4Mab-2, and A4Mab-3 Using Flow Cytometry
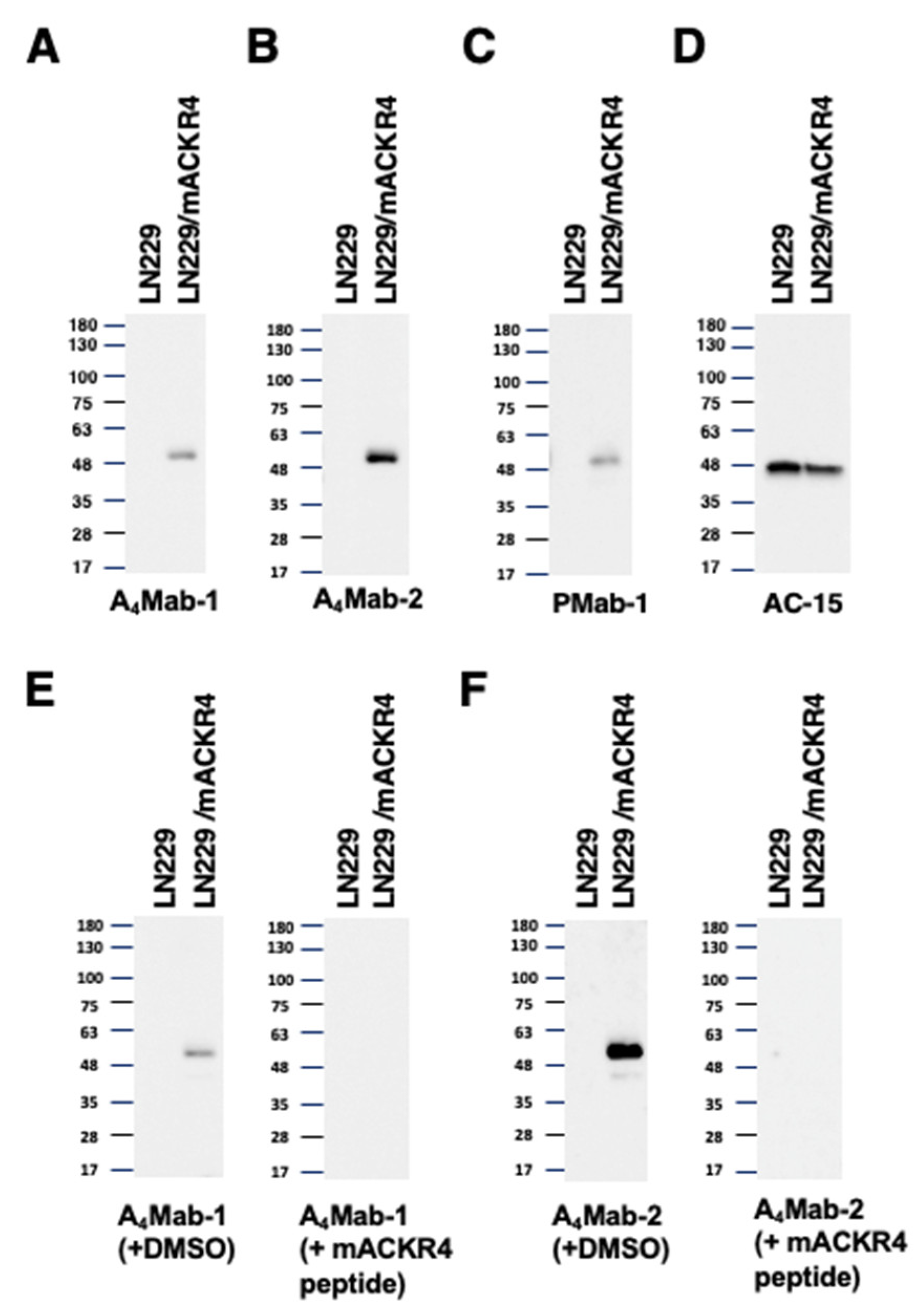
3.4. Detection of mACKR4 Using A4Mab-1, A4Mab-2, and A4Mab-3 by Western Blotting
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hughes, C.E.; Nibbs, R.J.B. A guide to chemokines and their receptors. Febs j 2018, 285, 2944–2971. [Google Scholar] [CrossRef]
- Comerford, I.; McColl, S.R. Atypical chemokine receptors in the immune system. Nat Rev Immunol 2024. [Google Scholar] [CrossRef]
- Bachelerie, F.; Ben-Baruch, A.; Burkhardt, A.M.; Combadiere, C.; Farber, J.M.; Graham, G.J.; Horuk, R.; Sparre-Ulrich, A.H.; Locati, M.; Luster, A.D.; et al. International Union of Basic and Clinical Pharmacology. [corrected]. LXXXIX. Update on the extended family of chemokine receptors and introducing a new nomenclature for atypical chemokine receptors. Pharmacol Rev 2014, 66, 1–79. [Google Scholar] [CrossRef] [PubMed]
- Proudfoot, A.E. Chemokine receptors: multifaceted therapeutic targets. Nat Rev Immunol 2002, 2, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Bonecchi, R.; Graham, G.J. Atypical Chemokine Receptors and Their Roles in the Resolution of the Inflammatory Response. Front Immunol 2016, 7, 224. [Google Scholar] [CrossRef]
- Bryce, S.A.; Wilson, R.A.; Tiplady, E.M.; Asquith, D.L.; Bromley, S.K.; Luster, A.D.; Graham, G.J.; Nibbs, R.J. ACKR4 on Stromal Cells Scavenges CCL19 To Enable CCR7-Dependent Trafficking of APCs from Inflamed Skin to Lymph Nodes. J Immunol 2016, 196, 3341–3353. [Google Scholar] [CrossRef]
- Kara, E.E.; Bastow, C.R.; McKenzie, D.R.; Gregor, C.E.; Fenix, K.A.; Babb, R.; Norton, T.S.; Zotos, D.; Rodda, L.B.; Hermes, J.R.; et al. Atypical chemokine receptor 4 shapes activated B cell fate. J Exp Med 2018, 215, 801–813. [Google Scholar] [CrossRef]
- Matti, C.; Salnikov, A.; Artinger, M.; D’Agostino, G.; Kindinger, I.; Uguccioni, M.; Thelen, M.; Legler, D.F. ACKR4 Recruits GRK3 Prior to β-Arrestins but Can Scavenge Chemokines in the Absence of β-Arrestins. Front Immunol 2020, 11, 720. [Google Scholar] [CrossRef]
- Nibbs, R.J.; Graham, G.J. Immune regulation by atypical chemokine receptors. Nat Rev Immunol 2013, 13, 815–829. [Google Scholar] [CrossRef]
- Wangmo, D.; Premsrirut, P.K.; Yuan, C.; Morris, W.S.; Zhao, X.; Subramanian, S. ACKR4 in Tumor Cells Regulates Dendritic Cell Migration to Tumor-Draining Lymph Nodes and T-Cell Priming. Cancers (Basel) 2021, 13. [Google Scholar] [CrossRef]
- Whyte, C.E.; Osman, M.; Kara, E.E.; Abbott, C.; Foeng, J.; McKenzie, D.R.; Fenix, K.A.; Harata-Lee, Y.; Foyle, K.L.; Boyle, S.T.; et al. ACKR4 restrains antitumor immunity by regulating CCL21. J Exp Med 2020, 217. [Google Scholar] [CrossRef]
- Ouchida, T.; Isoda, Y.; Nakamura, T.; Yanaka, M.; Tanaka, T.; Handa, S.; Kaneko, M.K.; Suzuki, H.; Kato, Y. Establishment of a Novel Anti-Mouse CCR1 Monoclonal Antibody C(1)Mab-6. Monoclon Antib Immunodiagn Immunother 2024. [Google Scholar] [CrossRef]
- Tateyama, N.; Asano, T.; Suzuki, H.; Li, G.; Yoshikawa, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Epitope Mapping of Anti-Mouse CCR3 Monoclonal Antibodies Using Flow Cytometry. Antibodies (Basel) 2022, 11. [Google Scholar] [CrossRef]
- Saito, M.; Harigae, Y.; Li, G.; Asano, T.; Tanaka, T.; Suzuki, H.; Kaneko, M.K.; Kato, Y. C(3)Mab-2: An Anti-Mouse CCR3 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022, 41, 45–49. [Google Scholar] [CrossRef]
- Asano, T.; Suzuki, H.; Tanaka, T.; Saito, M.; Li, G.; Goto, N.; Nanamiya, R.; Kaneko, M.K.; Kato, Y. C(3)Mab-3: A Monoclonal Antibody for Mouse CC Chemokine Receptor 3 for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2022, 41, 74–79. [Google Scholar] [CrossRef]
- Suzuki, H.; Tanaka, T.; Li, G.; Ouchida, T.; Kaneko, M.K.; Kato, Y. Development of a Sensitive Anti-Mouse CCR5 Monoclonal Antibody for Flow Cytometry Monoclon Antib Immunodiagn. Immunother 2024, in press. [Google Scholar]
- Suzuki, H.; Saito, M.; Asano, T.; Tanaka, T.; Kitamura, K.; Kudo, Y.; Kaneko, M.K.; Kato, Y. C(8)Mab-3: An Anti-Mouse CCR8 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022, 41, 110–114. [Google Scholar] [CrossRef]
- Saito, M.; Tanaka, T.; Asano, T.; Nakamura, T.; Yanaka, M.; Handa, S.; Komatsu, Y.; Harigae, Y.; Tateyama, N.; Nanamiya, R.; et al. C(8)Mab-2: An Anti-Mouse C-C Motif Chemokine Receptor 8 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022, 41, 115–119. [Google Scholar] [CrossRef]
- Saito, M.; Suzuki, H.; Tanaka, T.; Asano, T.; Kaneko, M.K.; Kato, Y. Development of an Anti-Mouse CCR8 Monoclonal Antibody (C(8)Mab-1) for Flow Cytometry and Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022, 41, 333–338. [Google Scholar] [CrossRef]
- Li, G.; Tanaka, T.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Cx1Mab-1: A Novel Anti-mouse CXCR1 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024, in press. [Google Scholar] [CrossRef]
- Ouchida, T.; Isoda, Y.; Tanaka, T.; Kaneko, M.K.; Suzuki, H.; Kato, Y. Cx(3)Mab-4: A Novel Anti-Mouse CXCR3 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024. [Google Scholar] [CrossRef]
- Ouchida, T.; Suzuki, H.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Cx(4)Mab-1: A Novel Anti-Mouse CXCR4 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2023. [Google Scholar] [CrossRef]
- Tanaka, T.; Li, G.; Asano, T.; Saito, M.; Kaneko, M.K.; Suzuki, H.; Kato, Y. Development of a Novel Anti-Mouse CCR2 Monoclonal Antibody (C(2)Mab-6) by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022, 41, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Asano, T.; Suzuki, H.; Goto, N.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Establishment of Novel Anti-Mouse CCR3 Monoclonal Antibodies (C(3)Mab-6 and C(3)Mab-7) by N-terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022, 41, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Takei, J.; Suzuki, H.; Asano, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Development of a Novel Anti-Mouse CCR4 Monoclonal Antibody (C(4)Mab-1) by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022, 41, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Ubukata, R.; Suzuki, H.; Tanaka, T.; Li, G.; Kaneko, M.K.; Kato, Y. Development of Sensitive Anti-Mouse CCR5 Monoclonal Antibodies Using the N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2024. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Asano, T.; Suzuki, H.; Tanaka, T.; Yoshikawa, T.; Kaneko, M.K.; Kato, Y. Establishment of a Sensitive Monoclonal Antibody Against Mouse CCR9 (C(9)Mab-24) for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2023, 42, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, K.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Cx(6)Mab-1: A Novel Anti-Mouse CXCR6 Monoclonal Antibody Established by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022, 41, 133–141. [Google Scholar] [CrossRef]
- Fujii, Y.; Kaneko, M.K.; Kato, Y. MAP Tag: A Novel Tagging System for Protein Purification and Detection. Monoclon Antib Immunodiagn Immunother 2016, 35, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Bastow, C.R.; Bunting, M.D.; Kara, E.E.; McKenzie, D.R.; Caon, A.; Devi, S.; Tolley, L.; Mueller, S.N.; Frazer, I.H.; Harvey, N.; et al. Scavenging of soluble and immobilized CCL21 by ACKR4 regulates peripheral dendritic cell emigration. Proc Natl Acad Sci U S A 2021, 118. [Google Scholar] [CrossRef]
- Comerford, I.; Nibbs, R.J.; Litchfield, W.; Bunting, M.; Harata-Lee, Y.; Haylock-Jacobs, S.; Forrow, S.; Korner, H.; McColl, S.R. The atypical chemokine receptor CCX-CKR scavenges homeostatic chemokines in circulation and tissues and suppresses Th17 responses. Blood 2010, 116, 4130–4140. [Google Scholar] [CrossRef]
- Comerford, I.; Milasta, S.; Morrow, V.; Milligan, G.; Nibbs, R. The chemokine receptor CCX-CKR mediates effective scavenging of CCL19 in vitro. Eur J Immunol 2006, 36, 1904–1916. [Google Scholar] [CrossRef] [PubMed]
- Samus, M.; Rot, A. Atypical chemokine receptors in cancer. Cytokine 2024, 176, 156504. [Google Scholar] [CrossRef] [PubMed]
- Isoda, Y.; Tanaka, T.; Suzuki, H.; Asano, T.; Nakamura, T.; Yanaka, M.; Handa, S.; Komatsu, Y.; Okuno, S.; Takahashi, N.; et al. Epitope Mapping of an Anti-Mouse CXCR6 Monoclonal Antibody (Cx(6)Mab-1) Using the 2 × Alanine Scanning Method. Monoclon Antib Immunodiagn Immunother 2022, 41, 275–278. [Google Scholar] [CrossRef]
- He, X.; Xu, C. Immune checkpoint signaling and cancer immunotherapy. Cell Res 2020, 30, 660–669. [Google Scholar] [CrossRef]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef]
- Galon, J.; Costes, A.; Sanchez-Cabo, F.; Kirilovsky, A.; Mlecnik, B.; Lagorce-Pagès, C.; Tosolini, M.; Camus, M.; Berger, A.; Wind, P.; et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 2006, 313, 1960–1964. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



