Submitted:
19 July 2024
Posted:
19 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Animals
2.3. Preparation of Semen Extender
2.4. Semen Collection
2.5. Sperm Freezing Process and Post-Thaw Sperm Evaluation
2.6. Motility Assessment
2.7. Assessment of Sperm Viability
2.8. Assessment of Acrosome Integrity
2.9. Gene Expression Level
2.9.1. RNA Extraction
2.9.2. Quantitative RT-PCR
2.10. Experimental Design
2.11. Statistical Analyses
3. Results
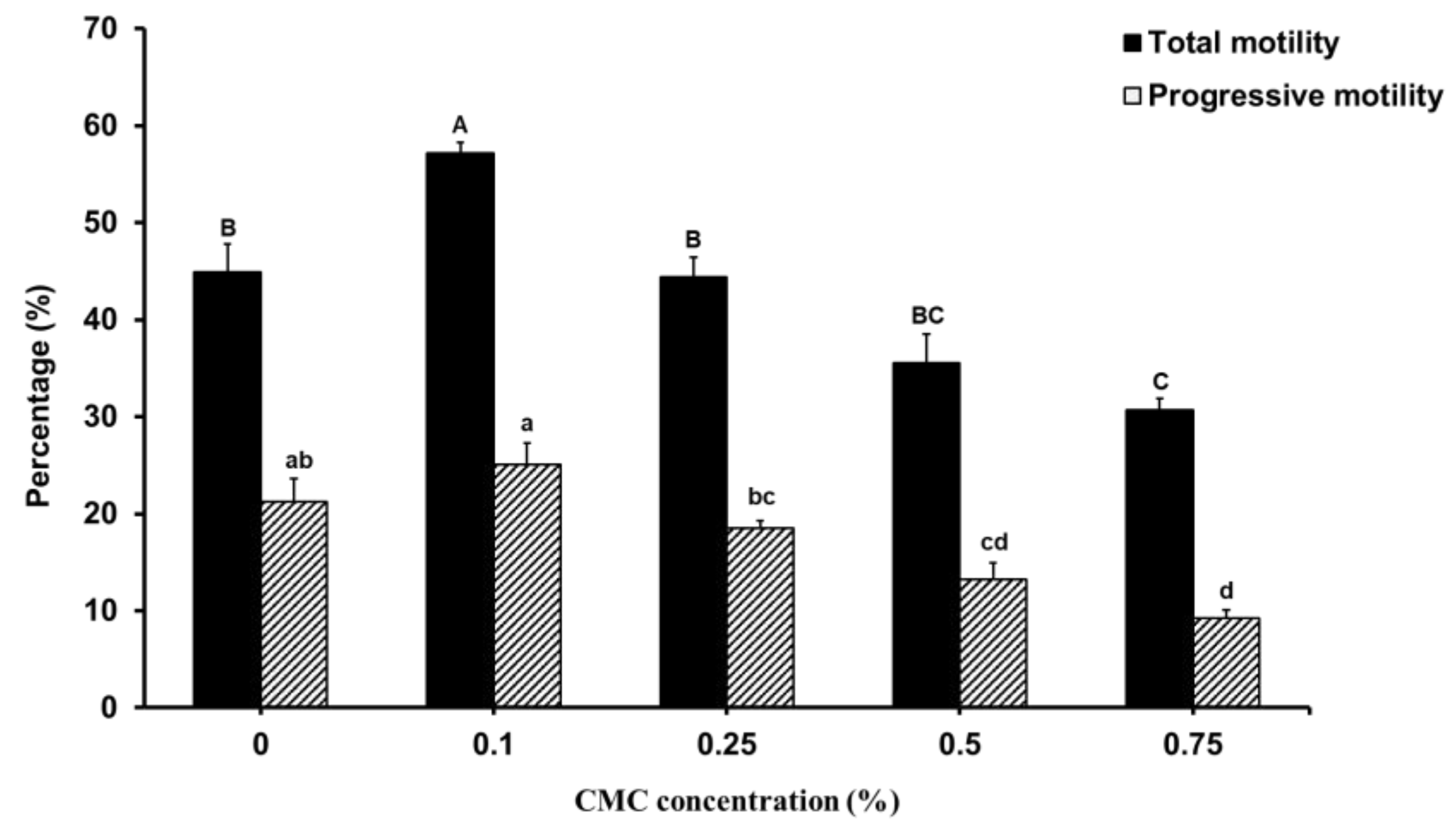
3.1. Effect of CMC on Sperm Motility, Viability and Acrosome Integrity
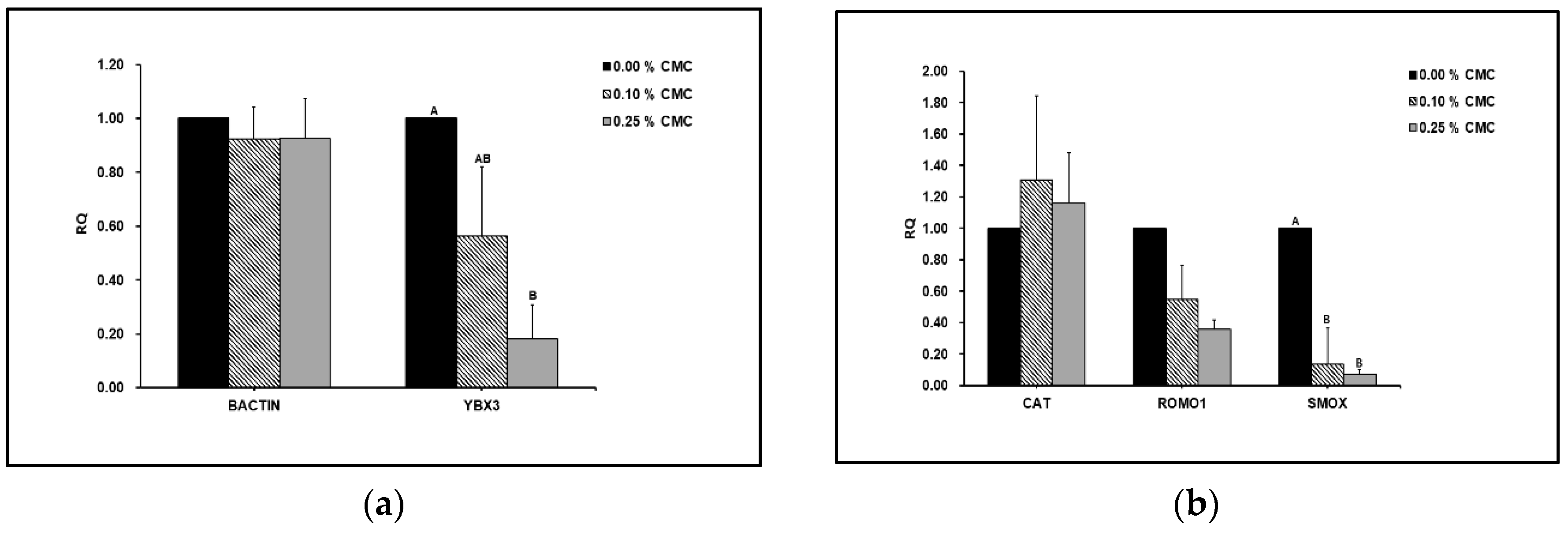
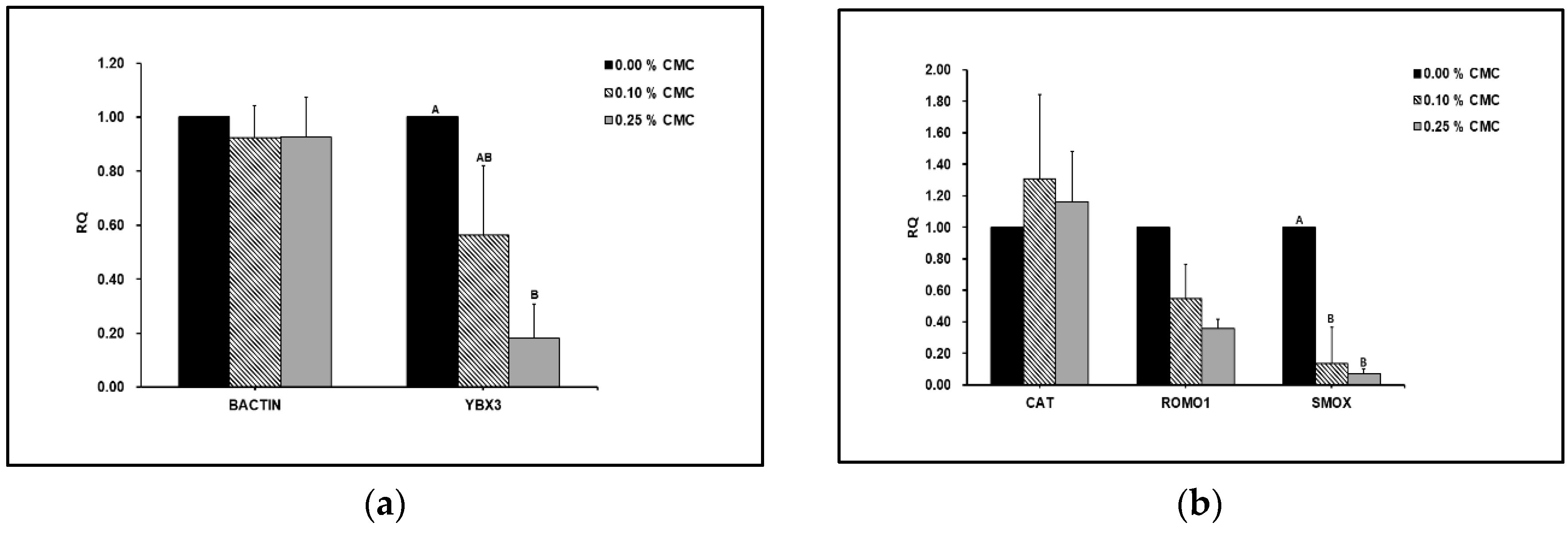
3.2. Effect of CMC on Gene Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- England, G. C.; Millar, K. M. The ethics and role of AI with fresh and frozen semen in dogs. Reprod Domest Anim 2008, 43 Suppl 2, 165-71. [CrossRef]
- Thomassen, R.; Farstad, W. Artificial insemination in canids: a useful tool in breeding and conservation. Theriogenology 2009, 71, 190-9. [CrossRef]
- Puja, I. K.; Sawitri, N. M.; Maharani, N.; Heryani, L.; Dharmayudha, A.; Gunawan, I. Preservation of semen from Kintamani Bali dogs by freezing method. J Adv Vet Anim Res 2019, 6, 158-162. [CrossRef]
- Hezavehei, M.; Sharafi, M.; Kouchesfahani, H. M.; Henkel, R.; Agarwal, A.; Esmaeili, V.; Shahverdi, A. Sperm cryopreservation: A review on current molecular cryobiology and advanced approaches. Reproductive BioMedicine Online 2018, 37, 327-339. [CrossRef]
- Oldenhof, H.; Gojowsky, M.; Wang, S.; Henke, S.; Yu, C.; Rohn, K.; Wolkers, W. F.; Sieme, H. Osmotic stress and membrane phase changes during freezing of stallion sperm: mode of action of cryoprotective agents. Biol Reprod 2013, 88, 68. [CrossRef]
- Paul, R. K.; Kumar, D.; Singh, R. Carboxymethyl cellulose and glycerol act synergistically as cryoprotectant during cryopreservation of ram semen. Cryobiology 2021, 101, 61-66. [CrossRef]
- Aisen, E. G.; Medina, V. H.; Venturino, A. Cryopreservation and post-thawed fertility of ram semen frozen in different trehalose concentrations. Theriogenology 2002, 57, 1801-8. [CrossRef]
- Tariq, A.; Ahmad, M.; Iqbal, S.; Riaz, M. I.; Tahir, M. Z.; Ghafoor, A.; Riaz, A. Effect of carboxylated poly l-Lysine as a cryoprotectant on post-thaw quality and in vivo fertility of Nili Ravi buffalo (Bubalus bubalis) bull semen. Theriogenology 2020, 144, 8-15. [CrossRef]
- Ezzati, M.; Shanehbandi, D.; Hamdi, K.; Rahbar, S.; Pashaiasl, M. Influence of cryopreservation on structure and function of mammalian spermatozoa: an overview. Cell Tissue Bank 2020, 21, 1-15. [CrossRef]
- Ďuračka, M.; Benko, F.; Tvrdá, E. Molecular Markers: A New Paradigm in the Prediction of Sperm Freezability. In International Journal of Molecular Sciences2023; Vol. 24. [CrossRef]
- Guerra, S. M.; Valcarce, D. G.; Cabrita, E.; Robles, V. Analysis of transcripts in gilthead seabream sperm and zebrafish testicular cells: mRNA profile as a predictor of gamete quality. Aquaculture 2013, 406, 28-33.
- Valcarce, D. G.; Cartón-García, F.; Herráez, M. P.; Robles, V. Effect of cryopreservation on human sperm messenger RNAs crucial for fertilization and early embryo development. Cryobiology 2013, 67, 84-90.
- Linde-Forsberg, C. Regulations and recommendations for international shipment of chilled and frozen canine semen. In Recent advances in small animal reproduction, International Veterinary Information Service (www. ivis. org), Document 2001; Vol. 1209.
- Rahman, M. A.; Park, S. H.; Yu, I. J. Dog sperm cryopreservation using one step dilution with glycerol-free tris extender. Cryo Letters 2016, 37, 137-41. Retrieved from https://www.ingentaconnect.com/content/cryo/cryo/2016/00000037/00000002/art00009.
- Dorado, J.; Rijsselaere, T.; Munoz-Serrano, A.; Hidalgo, M. Influence of sampling factors on canine sperm motility parameters measured by the Sperm Class Analyzer. Syst Biol Reprod Med 2011, 57, 318-25.
- Yu, I. J. Canine sperm cryopreservation using glucose in glycerol-free Tris. Cryo Letters 2014, 35, 101-7.
- Livak, K. J.; Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402-8. [CrossRef]
- Bahr, M. M.; Amer, M. S.; Abo-El-Sooud, K.; Abdallah, A. N.; Shehab, G. G.; El-Tookhy, O. S. Proficiency of Carboxymethylcellulose as a Cryoprotectant. Clinical and Histological Evaluation of Cryopreserved Heterogenous Mesenchymal Stem Cell-Exosomal Hydrogel on Critical Size Skin Wounds in Dogs. Int J Hematol Oncol Stem Cell Res 2021, 15, 178-191. [CrossRef]
- Nagy, S.; Sinkovics, G.; Kovács, A. Viability and acrosome integrity of rabbit spermatozoa processed in a gelatin-supplemented extender. Animal Reproduction Science 2002, 70, 283-286. [CrossRef]
- Yániz, J.; Martí, J. I.; Silvestre, M. A.; Folch, J.; Santolaria, P.; Alabart, J. L.; López-Gatius, F. Effects of solid storage of sheep spermatozoa at 15 degrees C on their survival and penetrating capacity. Theriogenology 2005, 64, 1844-51. [CrossRef]
- Anaya, M. C. G.; Barón, F. J.; Guerrero, J. M.; García-Marín, L. J.; Gil, J. Increasing extender viscosity improves the quality of cooled boar semen. Journal of Agricultural Science 2014, 6, 12.
- Asa, E.; Ahmadi, R.; Mahmoodi, M.; Mohammadniya, A. Supplementation of freezing media with alpha lipoic acid preserves the structural and functional characteristics of sperm against cryodamage in infertile men with asthenoteratozoospermia. Cryobiology 2020, 96, 166-174. [CrossRef]
- Salamon, S.; Maxwell, W. M. C. Frozen storage of ram semen I. Processing, freezing, thawing and fertility after cervical insemination. Animal Reproduction Science 1995, 37, 185-249. [CrossRef]
- Oltvai, Z. N.; Milliman, C. L.; Korsmeyer, S. J. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 1993, 74, 609-19. [CrossRef]
- Vaskivuo, T. E.; Stenbäck, F.; Tapanainen, J. S. Apoptosis and apoptosis-related factors Bcl-2, Bax, tumor necrosis factor-alpha, and NF-kappaB in human endometrial hyperplasia and carcinoma. Cancer 2002, 95, 1463-71. [CrossRef]
- Wang, X. L.; Zhang, Y. X.; Yang, C. G.; Zhang, B.; Chen, S. L. Cloning, characterization and expression analysis of a cold shock domain family member YB-1 in turbot Scophthalmus maximus. Fish Shellfish Immunol 2012, 33, 1215-21. [CrossRef]
- Snyder, E.; Soundararajan, R.; Sharma, M.; Dearth, A.; Smith, B.; Braun, R. E. Compound Heterozygosity for Y Box Proteins Causes Sterility Due to Loss of Translational Repression. PLoS Genet 2015, 11, e1005690. [CrossRef]
- Cervelli, M.; Amendola, R.; Polticelli, F.; Mariottini, P. Spermine oxidase: ten years after. Amino Acids 2012, 42, 441-50. [CrossRef]
- Amendola, R.; Cervelli, M.; Tempera, G.; Fratini, E.; Varesio, L.; Mariottini, P.; Agostinelli, E. Spermine metabolism and radiation-derived reactive oxygen species for future therapeutic implications in cancer: an additive or adaptive response. Amino Acids 2014, 46, 487-98. [CrossRef]
- Pegg, A. E. Functions of Polyamines in Mammals. J Biol Chem 2016, 291, 14904-12. [CrossRef]
- Amini, M. A.; Karimi, M.; Talebi, S. S.; Piri, H.; Karimi, J. The Association of Oxidative Stress and Reactive Oxygen Species Modulator 1 (ROMO1) with Infertility: A Mini Review. Chonnam Med J 2022, 58, 91-95. [CrossRef]
- Fallahi, S.; Rajaei, M.; Hesam, M. J.; Koolivand, M.; Malekzadeh, K. The effect of Phoenix dactylifera pollen on the expression of NRF2, SOD2, CAT, and GPX4 genes, and sperm parameters of fertile and infertile men: A controlled clinical trial. Int J Reprod Biomed 2021, 19, 545-558. [CrossRef]
| Gene | Primer sequence | Fragment length (bp) | Annealing temperature (°C) | Accession number. |
|---|---|---|---|---|
| GAPDH | F: GGAGAAAGCTGCCAAATATG R: ACCAGGAAATGAGCTTGACA |
152 | 53.4 | NM_001003142.2 |
| BCL2 | F: CTCCTGGCTGTCTCTGAAGG R: GTGGCAGGCCTACTGACTTC |
172 | 59.5 | NM_001002949.1 |
| BAX | F: GACGGCCTCTCTCCTACTT R: GGTGAGTGACGCAGTAAGCA |
120 | 57.4 | NM_001003011.1 |
| ANAXA2 | F: CTGGGGACTGATGAGGACTC R: CGGGCATCTTGGTCAATCAG |
227 | 57.4 | NM_001002961.1 |
| BACTIN | F: ACTGGGACGACATGGAGAAG R: CATGGTTGGGGTGTTGAAGG |
164 | 57.4 | Z70044.1 |
| YBX3 | F: TGAAGGAGAGAAGGGTGCAG R: CCAATCTCACCGGCATTACG |
150 | 57.4 | NM_001003127.1 |
| CAT | F: CCAGCCCTGACAAAATGCTT R: CACCCTGATTGTCGAGCATG |
165 | 57.4 | NM_001002984.1 |
| ROMO1 | F: TGTCTCAGGATGGGAATGCG R: TCCCGATGGCCATGAATGTG |
219 | 57.4 | XM_038433666.1 |
| SMOX | F: AGAAGTGTGATGACGAGGCG R: TCGGAAGTATGGGTTGCTGC |
128 | 57.4 | XM_855324.3 |
| CMC (%, w/v) |
VCL (µm/s) |
VAP (µm/s) |
VSL (µm/s) |
STR (%) |
LIN (%) |
WOB (%) |
ALH (µm/s) |
BCF (Hz) |
|---|---|---|---|---|---|---|---|---|
| 0 | 43.1 ± 2.8 | 29.8 ± 3.3 | 23.5 ± 3.2 | 59.0 ± 1.2 | 39.6 ± 2.4 | 61.4 ± 2.8 | 1.7 ± 0.1 | 5.7 ± 0.3 |
| 0.1 | 40.9 ± 3.0 | 30.4 ± 3.1 | 24.3 ± 2.8 | 61.8 ± 0.5 | 45.0 ± 1.2 | 66.8 ± 1.1 | 1.4 ± 0.0 | 6.0 ± 0.1 |
| 0.25 | 35.4 ± 3.3 | 26.0 ± 4.2 | 20.4 ± 4.5 | 60.6 ± 2.6 | 43.9 ± 4.2 | 66.2 ± 3.4 | 1.3 ± 0.0 | 5.5 ± 0.4 |
| 0.5 | 32.1 ± 2.6 | 22.5 ± 2.7 | 16.9 ± 3.0 | 58.7 ± 1.3 | 40.9 ± 2.0 | 63.5 ± 1.3 | 1.3 ± 0.0 | 5.0 ± 0.3 |
| 0.75 | 26.7 ± 1.8 | 17.9 ± 1.6 | 12.2 ± 1.7 | 56.8 ± 0.3 | 39.1 ± 0.8 | 63.5 ± 0.8 | 1.3 ± 0.0 | 4.1 ± 0.4 |
| CMC (%, w /v) | Viability | Acrosome integrity |
|---|---|---|
| 0 | 46.5 ± 1.0 | 48.2 ± 2.1 |
| 0.1 | 50.2 ± 0.9 | 50.6 ± 2.2 |
| 0.25 | 49.2 ± 1.4 | 40.6 ± 4.7 |
| 0.5 | 49.8 ± 1.6 | 46.3 ± 4.0 |
| 0.75 | 49.6 ± 1.0 | 49.0 ± 1.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




