Submitted:
08 July 2024
Posted:
19 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Centers Involved
2.2. LEVO-D Data Collection
2.3. REGALAD Data Collection
2.4. Endpoints
2.5. Statistical Analysis
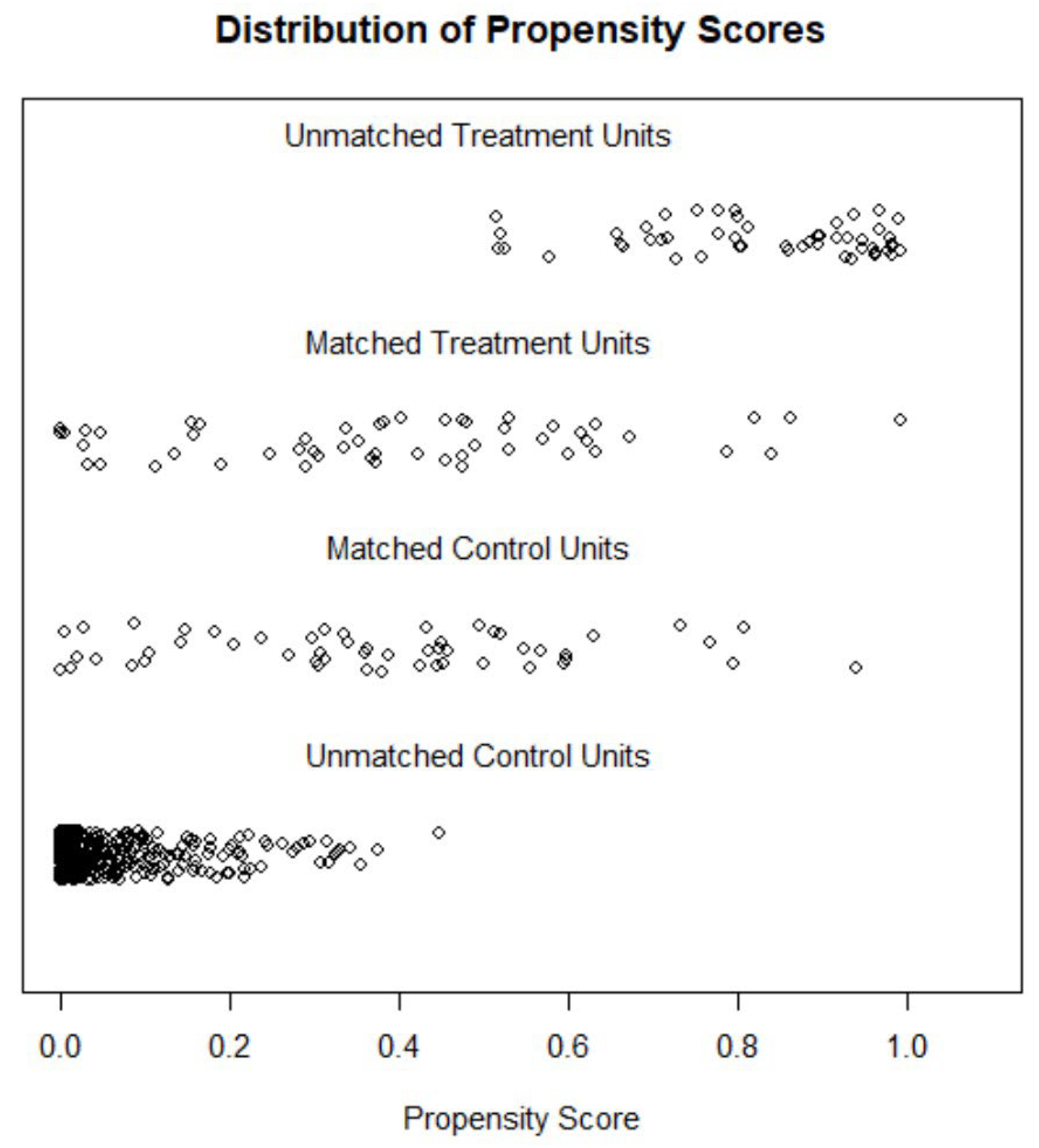
2.6. Propensity Matching
3. Results
3.1. Demographics
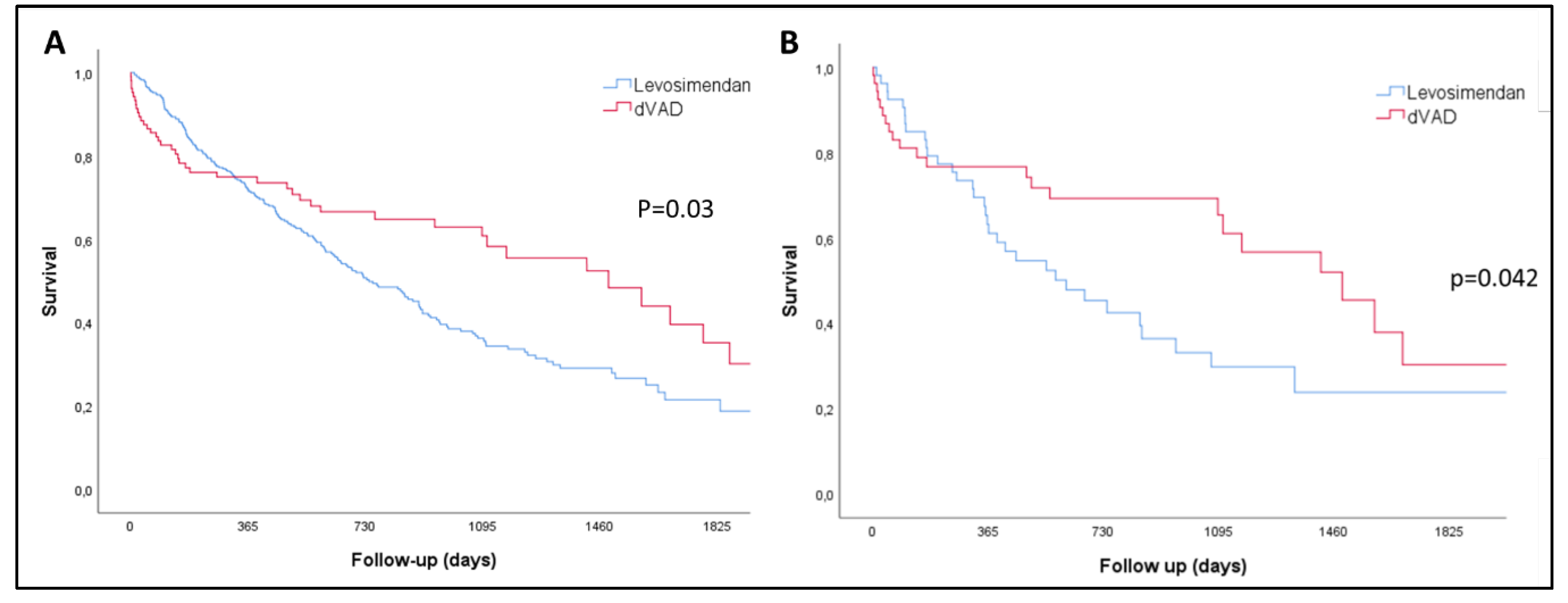
3.2. Survival and HF events
3.3. Propensity Score Matching
4. Discussion
4.1. Patients Characteristics
4.2. Long and Short-Term Outcomes
4.3. Clinical Implications
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McDonagh TA, Metra M, Adamo M; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42:3599-3726 . [CrossRef]
- Mehra MR, Cleveland JC Jr, Uriel N; et al. Primary results of long-term outcomes in the MOMENTUM 3 pivotal trial and continued access protocol study phase: A study of 2200 HeartMate 3 left ventricular assist device implants. Eur J Heart Fail. 2021;23:1392-1400 . [CrossRef]
- Comín-Colet J, Manito N, Segovia-Cubero J; et al. LION-HEART Study Investigators. Efficacy and safety of intermittent intravenous outpatient administration of levosimendan in patients with advanced heart failure: The LION-HEART multicentre randomised trial. Eur J Heart Fail. 2018;20:1128-1136. [CrossRef]
- García-González MJ, Aldea Perona A, Lara Padron A; et al. Efficacy and safety of intermittent repeated levosimendan infusions in advanced heart failure patients: The LAICA study. ESC Heart Fail. 2021;8:4820-4831. [CrossRef]
- Dobarro D, Donoso-Trenado V, Solé-González E; et al. Intermittent inotropic support with levosimendan in advanced heart failure as destination therapy: The XXXX1 registry. ESC Heart Fail. 2023. [CrossRef]
- Laufer-Perl M, Sadon S, Zahler D, et al Repetitive milrinone therapy in ambulatory advanced heart failure patients. Clin Cardiol. 2022;45:488-494 .
- Eaton RE, Kissling KT, Haas GJ; et al. Rehospitalization of Patients with Advanced Heart Failure Receiving Continuous, Palliative Dobutamine or Milrinone. Am J Cardiol. 2022;184:80-89 . [CrossRef]
- Gómez-Bueno M, Pérez de la Sota E, Forteza Gil A; et al. First report of the XXXX2 registry. Rev Esp Cardiol (Engl Ed). 2022 . Epub ahead of print. [CrossRef]
- Stevenson LW, Pagani FD, Young JB; et al. INTERMACS profiles of advanced heart failure: The current picture. J Heart Lung Transplant. 2009;28:535-41 . [CrossRef]
- Emerson D, Chikwe J, Catarino P; et al. Contemporary Left Ventricular Assist Device Outcomes in an Aging Population: An STS INTERMACS Analysis. J Am Coll Cardiol. 2021;78:883-894 .
- Mehra M.R., Nayak A., Morris A.A.; et al. Prediction of survival after implantation of a fully magnetically levitated left ventricular assist device. J Am Coll Cardiol HF 2022;10:948-959 . [CrossRef]
- Oliva F, Perna E, Marini M; et al. Scheduled intermittent inotropes for Ambulatory Advanced Heart Failure. The RELEVANT-HF multicentre collaboration. Int J Cardiol. 2018;272:255-259 .
- Rose EA, Gelijns AC, Moskowitz AJ; et al. Long-term use of a left ventricular assist device for end-stage heart failure. N Engl J Med. 2001;345:1435-43 . [CrossRef]
- Rogers JG, Butler J, Lansman SL; et al. Chronic mechanical circulatory support for inotrope-dependent heart failure patients who are not transplant candidates: Results of the INTrEPID Trial. J Am Coll Cardiol. 2007;50:741-7 . [CrossRef]
- Ambardekar AV, Kittleson MM, Palardy M; et al. Outcomes with ambulatory advanced heart failure from the Medical Arm of Mechanically Assisted Circulatory Support (MedaMACS) Registry. J Heart Lung Transplant. 2019;38:408-417. [CrossRef]
- Nizamic T, Murad MH, Allen LA; et al. Ambulatory Inotrope Infusions in Advanced Heart Failure: A Systematic Review and Meta-Analysis. JACC Heart Fail. 2018;6:757-767 .
- Goldstein DJ, Naka Y, Horstmanshof D; et al. Association of Clinical Outcomes With Left Ventricular Assist Device Use by Bridge to Transplant or Destination Therapy Intent: The Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy With HeartMate 3 (MOMENTUM 3) Randomized Clinical Trial. JAMA Cardiol. 2020;5:411-419 .
- Shah P, Yuzefpolskaya M, Hickey GW; et al. Twelfth Interagency Registry for Mechanically Assisted Circulatory Support Report: Readmissions After Left Ventricular Assist Device. Ann Thorac Surg. 2022;113:722-737 . [CrossRef]
- Aleksova N, Alba AC, Fan CS; et al. The Effect of Age on Outcomes After Destination-Therapy Left Ventricular Assist Device Implantation: An Analysis of the IMACS Registry. Can J Cardiol. 2021;37:467-475 . [CrossRef]
- Karason K, Lund LH, Dalén M; et al. Randomized trial of a left ventricular assist device as destination therapy versus guideline-directed medical therapy in patients with advanced heart failure. Rationale and design of the SWEdish evaluation of left Ventricular Assist Device (SweVAD) trial. Eur J Heart Fail. 2020;22:739-750 .
- DeFilippis EM, Breathett K, Donald EM; et al. Psychosocial Risk and Its Association With Outcomes in Continuous-Flow Left Ventricular Assist Device Patients. Circ Heart Fail. 2020;13:e006910 . [CrossRef]
- Fakoya OA, McCorry NK, Donnelly M. Loneliness and social isolation interventions for older adults: A scoping review of reviews. BMC Public Health. 2020;20:129 . [CrossRef]
| LEVO-D | REGALAD | p Value | |
|---|---|---|---|
| Age | 69.4±11.4 | 67.8±5.7 | 0.046 |
| Gender (male) | 79.4% | 80.8% | 0.76 |
| BSA | 1.84±0.2 | 1.85±0.2 | 0.40 |
| Hypertension | 68.7% | 67.3% | 0.78 |
| Diabetes Mellitus | 49.1% | 46.2% | 0.59 |
| Atrial arrythmias | 60.8% | 30.8% | <0.001 |
| HF diagnosis < 1month | 3.8% | 4.8% | 0.76 |
| HF diagnosis 1-12 months | 12.6% | 11.5% | |
| HF diagnosis 1-2 years | 9.8% | 5.8% | |
| HF diagnosis > 2 years | 73.3% | 77.9% | |
| HF aetiology DCM | 26.8% | 28.8% | 0.002 |
| HF aetiology IHD | 52.6% | 65.4% | |
| HF aetiology Others | 20.6% | 5.8% | |
| LVEF | 27.5±9.4 | 23.2±6.2 | <0.001 |
| LVEDD | 63.2±9.3 | 67.5±10.2 | <0.001 |
| TAPSE | 15.0±4.3 | 16.0±3.7 | 0.036 |
| SPAP | 51.2±15.8 | 47.5±15.5 | 0.044 |
| Severe MR | 10.6% | 22.1% | 0.005 |
| Severe TR | 9.2% | 5.8% | 0.14 |
| ACE-i/ARB/ARNI | 70.0% | 62.5% | 0.14 |
| Betablockers | 78.9% | 65.4% | 0.004 |
| MRA | 69.7% | 68.3% | 0.78 |
| Amiodarone | 23.0% | 36.5% | 0.005 |
| Anticoagulation | 69.7% | 54.4% | 0.003 |
| Digoxin | 19.1% | 16.7% | 0.57 |
| Furosemide or equivalent | 96% | 97.1% | 0.60 |
| ICD | 55.1% | 80.6% | <0.001 |
| CRT | 30.8% | 36.7% | 0.06 |
| NYHA IV | 12.9% | 57.7% | <0.001 |
| HR | 73.3±13.6 | 74.5±12.8 | 0.27 |
| SBP | 106.5±15.5 | 102.5±14.7 | 0.019 |
| DBP | 63.4±9.6 | 63.2±9.9 | 0.83 |
| HF admissions previous year | 1.7±1.6 | 2.0±0.7 | 0.003 |
| Sodium | 138.6±4.2 | 137.4±4.5 | 0.018 |
| Potassium | 4.2±0.6 | 4.1±0.5 | 0.12 |
| Urea | 91.5±51.2 | 78.8±44.2 | 0.018 |
| Creatinin | 1.6±0.7 | 1.4±0.5 | 0.001 |
| Billirubin | 1.2±0.8 | 0.9±0.5 | 0.002 |
| Albumin | 3.9±0.5 | 3.8±0.6 | 0.07 |
| Uric acid | 8.2±2.8 | 7.9±2.2 | 0.32 |
| NT-ProBNP (mean) | 9654±11879 | 6203±5995 | <0.001 |
| Haemoglobin | 12.6±1.9 | 12.0±1.9 | 0.003 |
| Lymphocites (%) | 19.8±9.4 | 19.0±7.8 | 0.44 |
| HF admissions year after | 1.12±1.8 | 0.94±1.64 | 0.35 |
| Alive without admission 1 year after | 49.6% | 33.7% | 0.004 |
| LEVO-D (53) | REGALAD (53) | P Value | |
|---|---|---|---|
| Age | 69.6±8.9 | 68.4±4.8 | 0.39 |
| Sex (male) | 73.6% | 77.4% | 0.87 |
| BMI | 25.8±3.9 | 26.9±3.8 | 0.16 |
| Hypertension | 64.2% | 66% | 0.84 |
| Diabetes | 52.8% | 52.8% | 0.99 |
| Atrial Tachy | 39.6% | 35.8% | 0.84 |
| IHD | 60.4% | 60.4% | 0.99 |
| LVEF | 24.1% | 23.5% | 0.66 |
| LVEDD | 66.5±5.7 | 67.4±10.8 | 0.60 |
| RV dysfunc | 54.7% | 54.7% | 0.99 |
| MR III-IV | 52.8% | 56.6% | 0.85 |
| TR III-IV | 37.7% | 37.7% | 0.99 |
| SPAP | 49.2±10.4 | 47.5±13.3 | 0.47 |
| SBP | 104.3±16.6 | 103.2±16.5 | 0.75 |
| HR | 75.9±13.2 | 75.3±12.7 | 0.81 |
| MDRD4 | 53.7±29.4 | 52±23.6 | 0.67 |
| NT-ProBNP | 7910±7000 | 7157±6317 | 0.56 |
| Haemoglobin | 11.9±1.7 | 11.9±2.1 | 0.87 |
| ACE-i/ARB/ARNI | 62.2% | 68% | 0.68 |
| Betablockers | 67.9% | 67.9% | 0.99 |
| ARM | 69.7% | 69.8% | 0.99 |
| Amiodarone | 28.3% | 30.2% | 0.98 |
| OAC | 50.9% | 56.6% | 0.69 |
| Digoxin | 18.9% | 20.8% | 0.99 |
| Furosemide | 94.3% | 94.3% | 0.99 |
| ICD | 67.9% | 71.7% | 0.83 |
| CRT | 41.5% | 47.2% | 0.70 |
| NYHA IV | 41.5% | 39.6% | 0.99 |
| 3 or more HF admissions | 20.8% | 24.5% | 0.82 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




