Submitted:
18 July 2024
Posted:
19 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Theoretical Backgrounds: Inertial Focusing
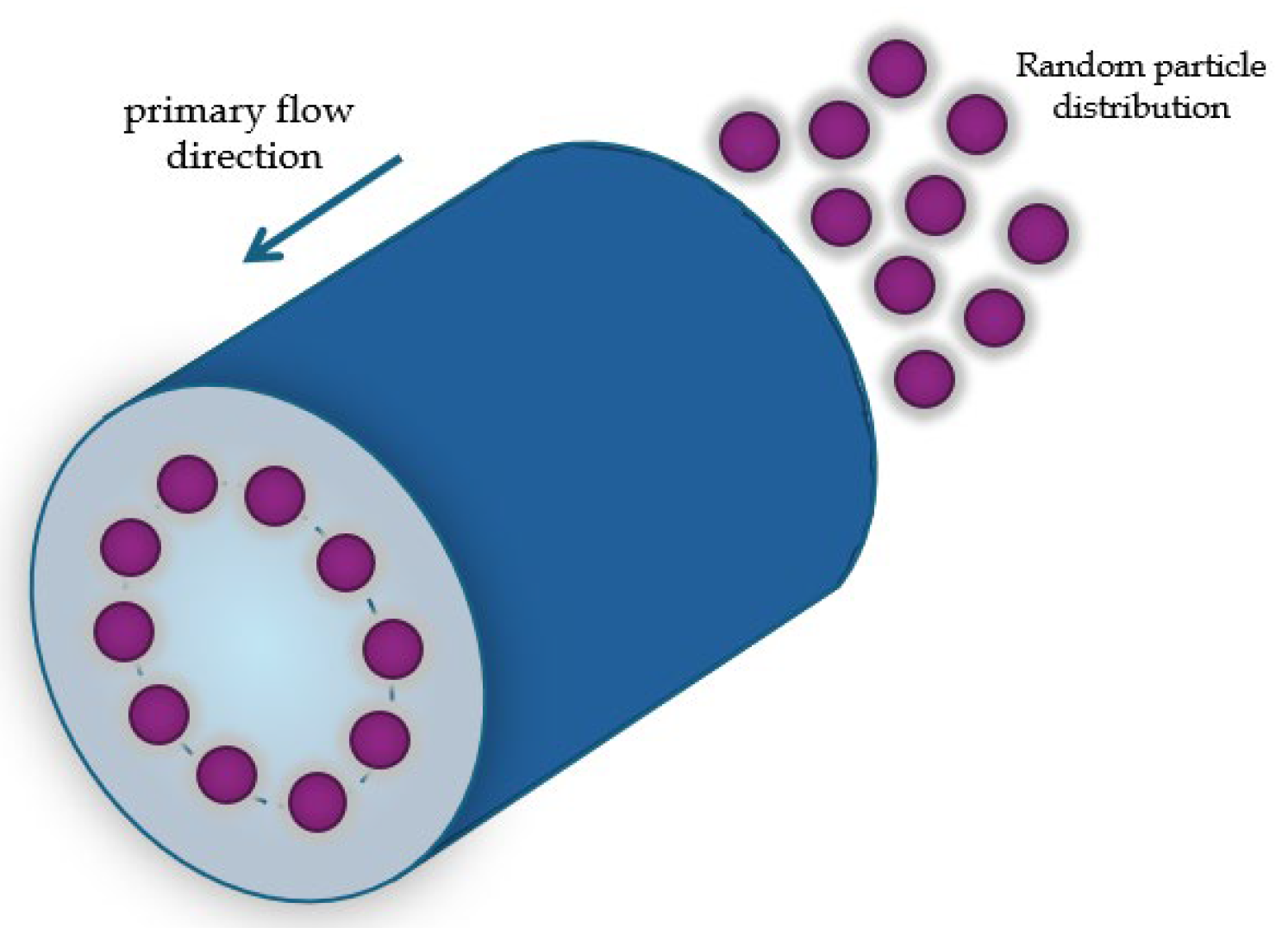
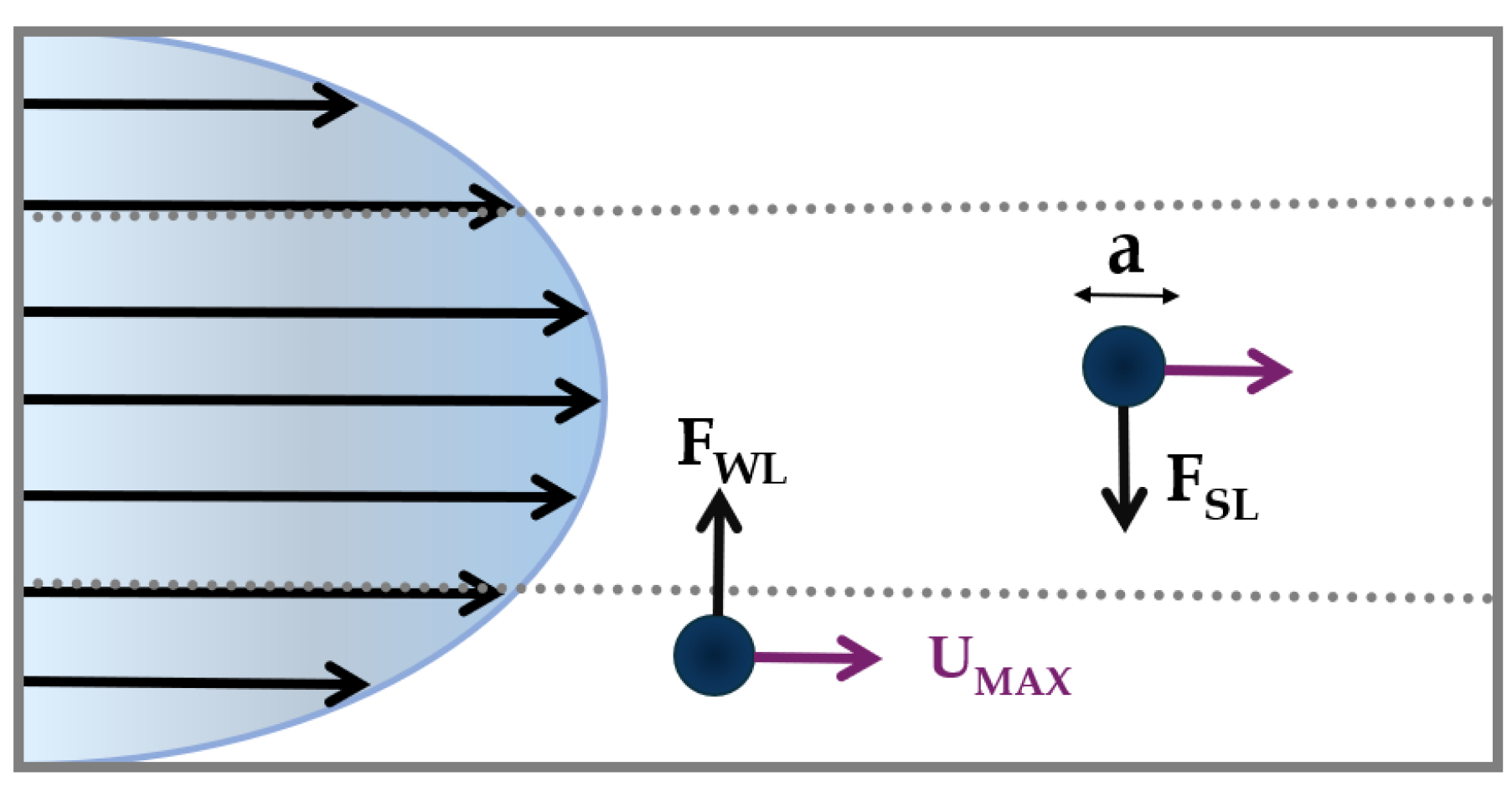
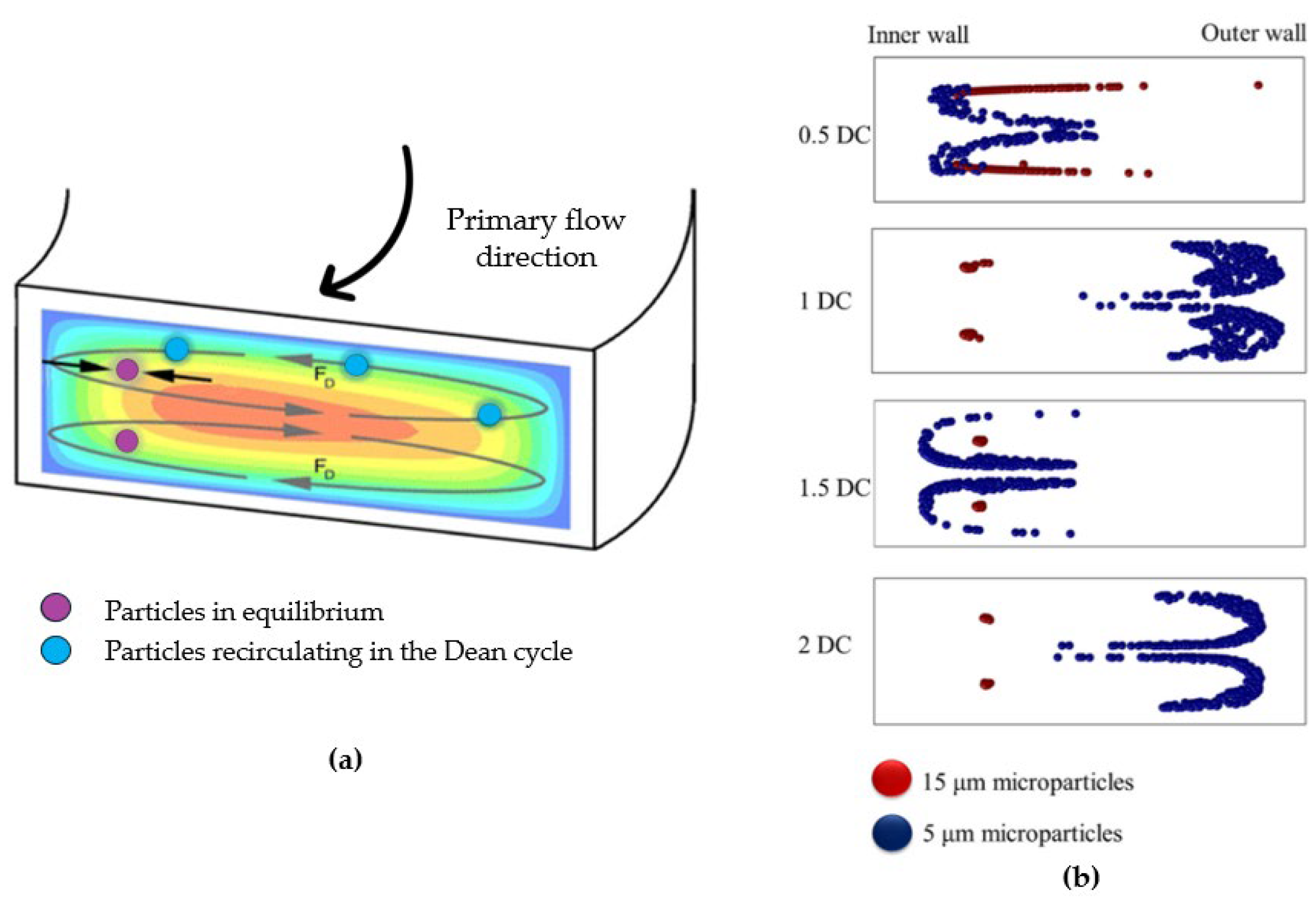
2.1. Curvilinear Microchannel and Dean Vortices
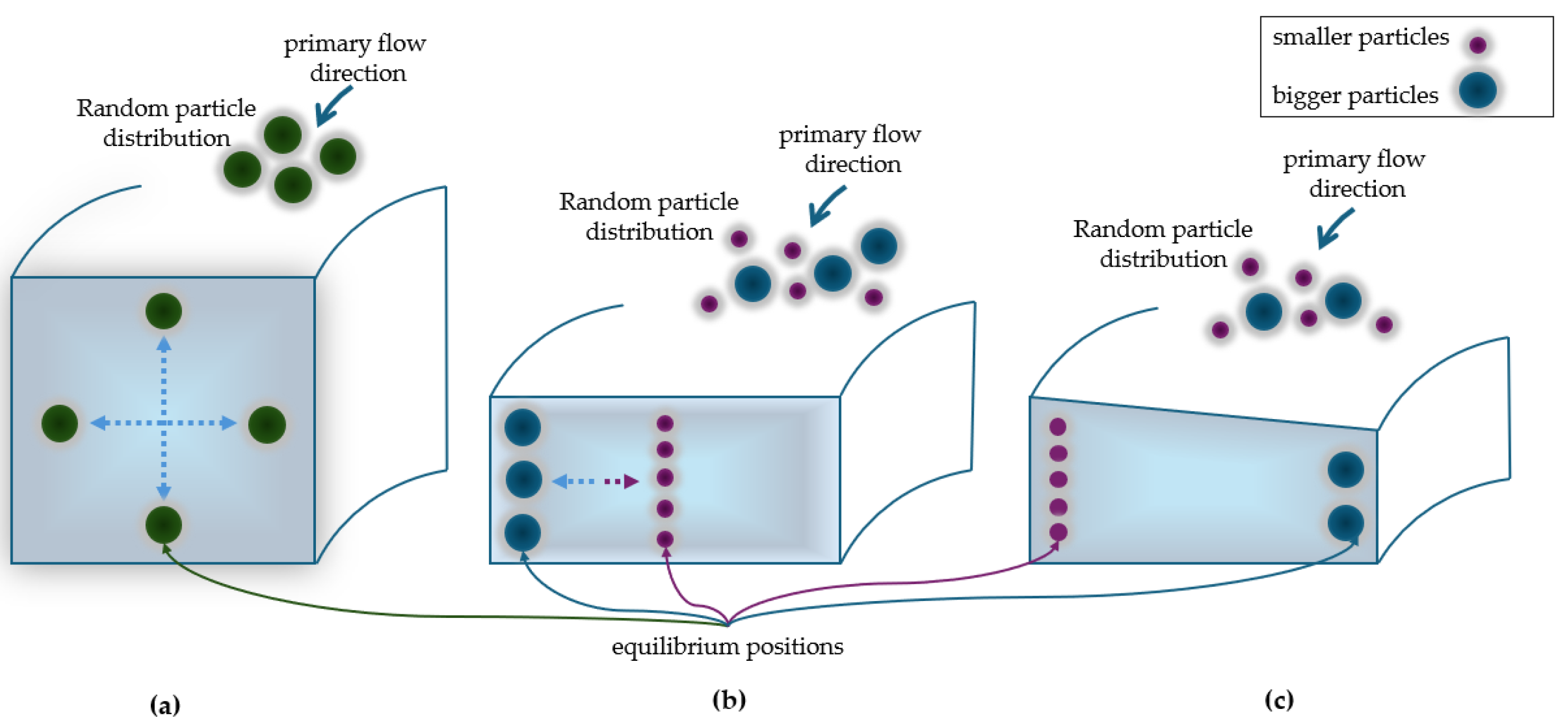
2.2. Rectangular and Trapezoidal Spiral Microchannels
3. Materials and Prototyping Techniques for Spiral Microchannels
3.1. Materials
3.2. Fabrication Techniques
3.2.1. Techniques by Deposition
3.2.2. Techniques by Removal
| Material | Fabrication technique | Ref. |
|---|---|---|
| PMMA | CO2 laser ablation | [2,66] |
| PDMS | Soft lithography | [1,34,42,47,50,51,52,54,55,56,72,73,74,75,76,77,79,85] |
| xurography | [80] | |
| Glass | Yb:KGW solid state laser ablation | [46,84] |
| Silicon | etching | [63] |
| Resin | 3-D printing | [78] |
| Hydrogel | 3D-printing | [68] |
| Wax | 3D-printing + soft lihography | [69] |
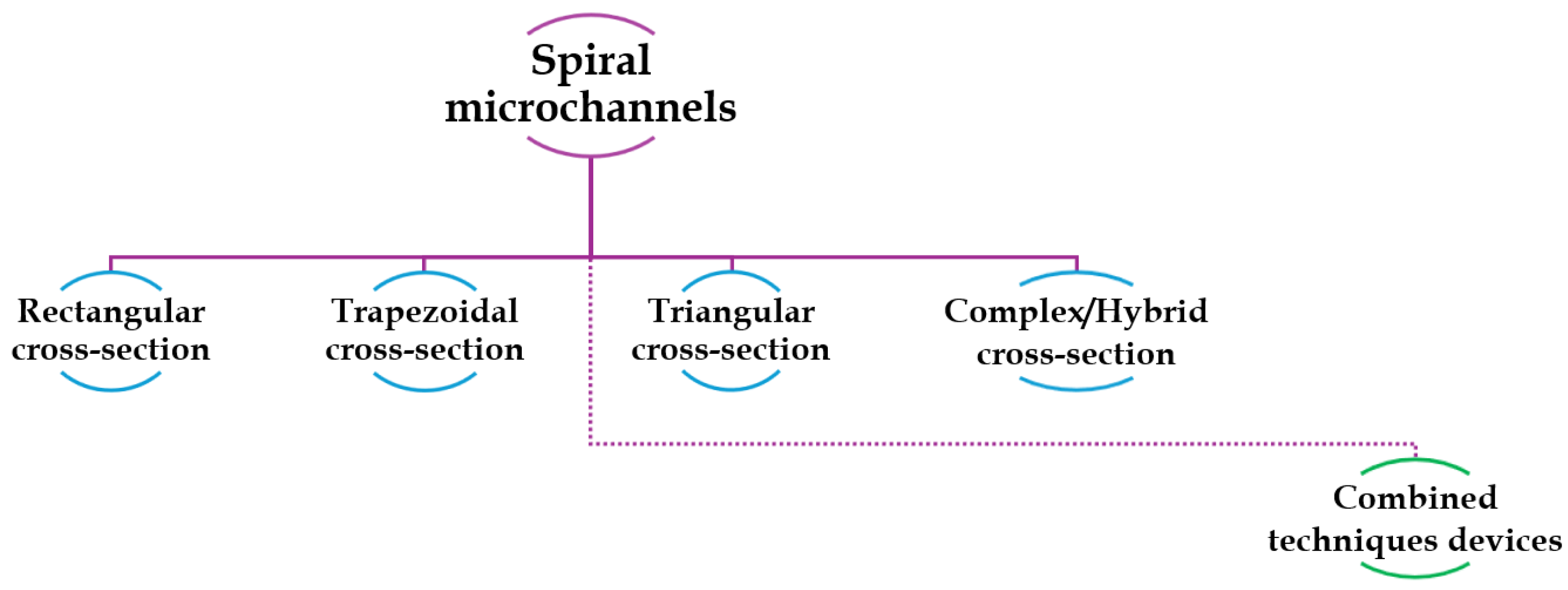
4. Overview on Different Cross-Sectional Shapes
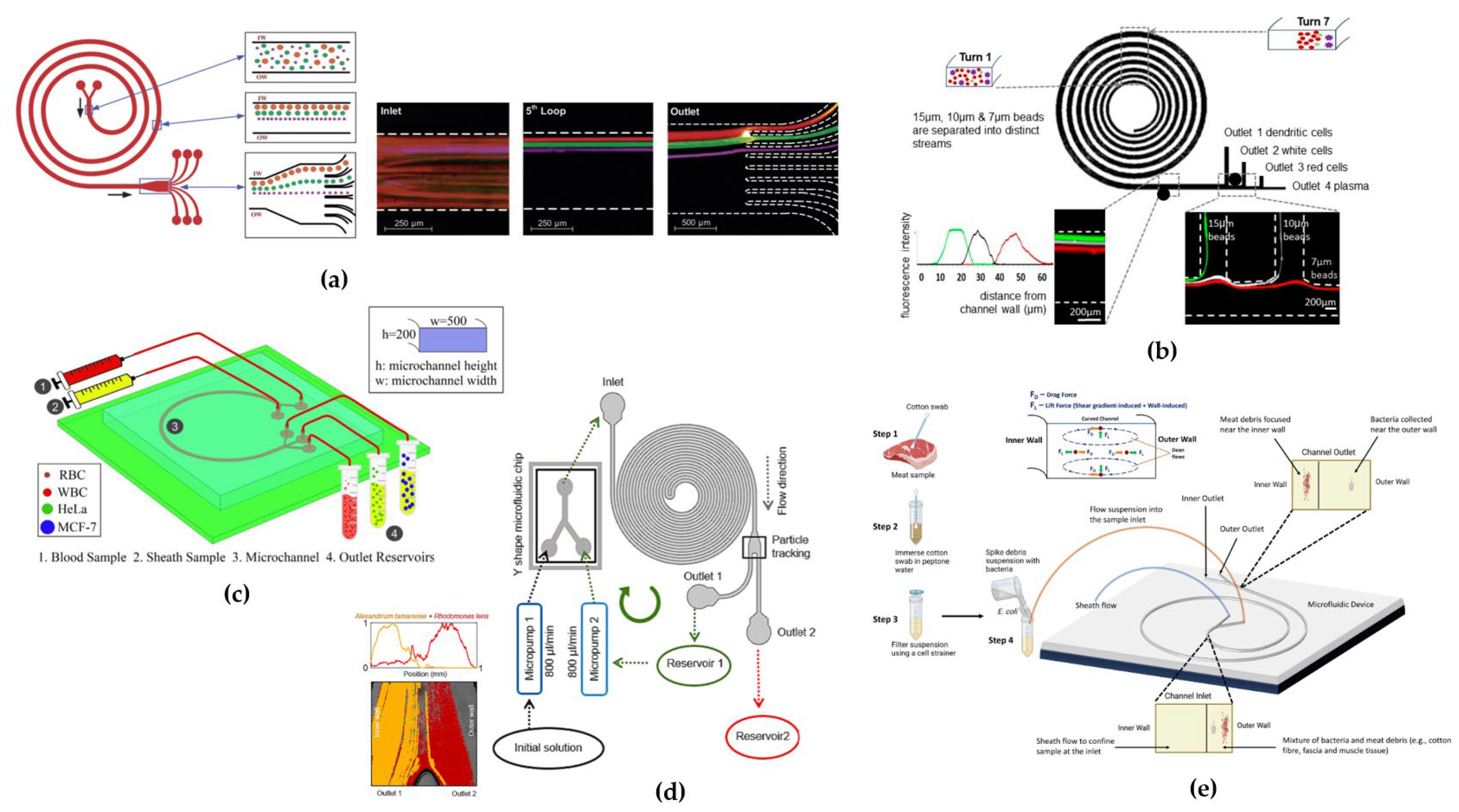
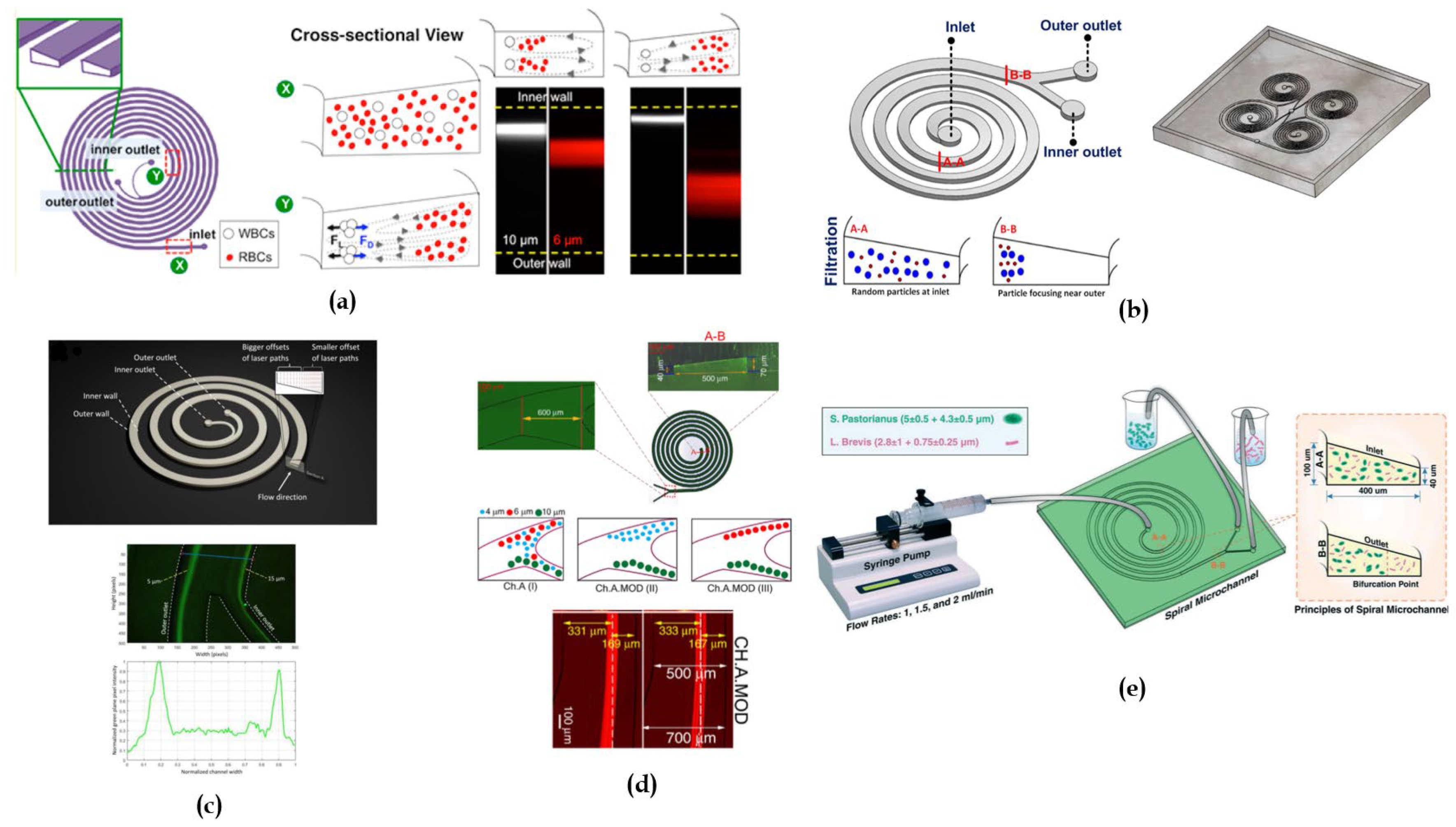
4.1. Rectangular Cross-Sectional Channels
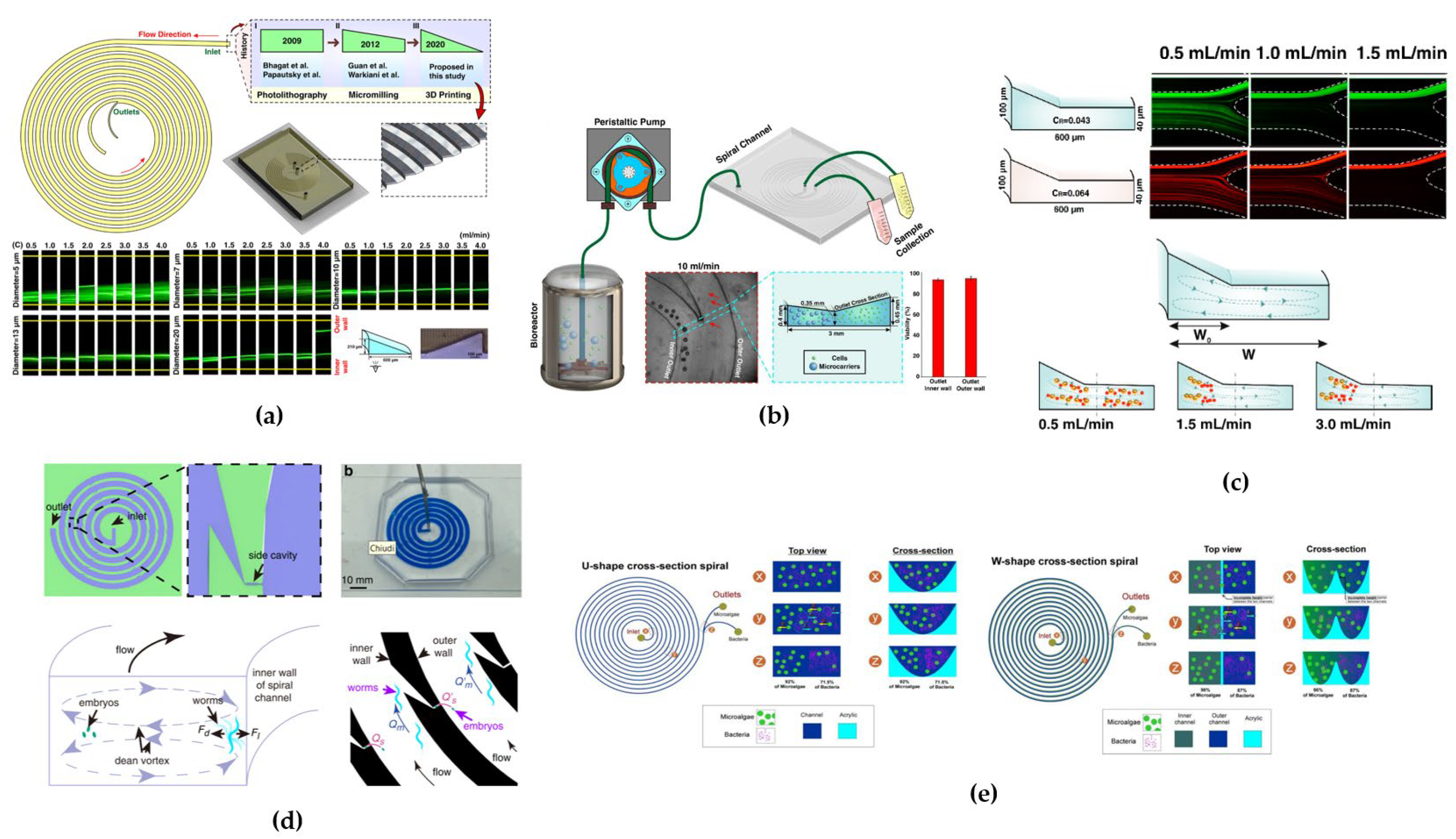
4.2. Trapezoidal Cross-Sectional Devices
4.3. Triangular Cross-Sectional Devices
4.4. Hybrid/Hybrid Cross-Sectional Devices
4.5. Combined Techniques Devices
| loop | Channel dimensions |
Particles’ size | Flowrate | Separation Efficiency (%) |
Ref. | ||
|---|---|---|---|---|---|---|---|
| Rectangular cross section | |||||||
| >10 | W=100 µm H=1250 µm |
polystyrene beads: 1, 8, 10 µm |
2 mL/min | Not specified | [63] | ||
| >5 | |||||||
| 5 | W=100 µm H=50 µm |
polystyrene beads=7.32 and 1.9 µm | 10 μL/min | 100% | [72] | ||
| 5 | W=500 µm H=130 µm |
polystyrene beads: 10, 15, 20 µm |
3 mL/min | 90% | [42] | ||
| W=500 µm H=120 µm |
SH-SY5Y neuroblastoma cells and C6 glioma cells | 80% | |||||
| 4 | W=500 µm H=150 µm |
RBCs~7 µm WBCs~ 10-20 µm |
1.8 mL/min | 95% |
[34] |
||
| W =250 µm H =75 µm |
polystyrene beads: 7.32, 10, 15 and 20 µm |
1-3 mL/min | All in the first outlets/no separation |
||||
| 4 | W=150 µm H=50 µm |
Sperm cells ~9 µm RBCs ~9 µm |
0.52 µL/min | 81-99% | [88] | ||
| 3x2 | W=500 µm H=170 µm |
polystyrene beads: 6, 10 and 15 µm |
100 µL/min | 90% | [47] | ||
| CTCs>15µm WBCs~7–15 µm |
3 mL/h (0.1mL/min) |
||||||
| 7 | W=from 200 to 600 H=100 |
polystyrene beads: 7, 10 and 15 µm |
1.6 mL/min | 72 % | [80] | ||
| Human dendritic cells ~ 10-15 µm RBCs~7 µm WBC~7–15 µm | |||||||
| 1 | W=500 H=200 |
RBCs~7 µm CTCs (HeLa and MCF-7) ~ 16-24 µm |
~113-139 mL/h | 100% | [86] | ||
| 2x5 | W=300 µm H=100µm |
polystyrene beads: 6, 10, 20 and 40 μm algal dinoflagellate species ~ 20-60 µm |
2000 µL/min | >94% (loss <6%) |
[79] | ||
| 2 | W=200 µm H=70µm |
polystyrene beads: 1.84, 6.04 and 10.6 μm |
400 µL/min | 50% | [76] | ||
| bacteria (Escherichia coli / Staphylococcus aureus) ~ 1 µm ground meat debris | |||||||
| Trapezoidal cross section | |||||||
| 8 | W=500 µm Hin=70µm Hout=100µm |
Polystyrene beads: 6,10, 15.5 µm in water |
0.8 mL/min | >80% | [54] | ||
| polymorphonuclear leukocytes (PMNs), mononuclear leukocytes (MNLs) and haematocrits | |||||||
| 8 | W=600 µm Hin=80µm Hou=130µm |
Polystyrene beads: 5.8, 9.8, 15.5, 26.25 µm |
0.5-7.5 mL/min | 92% | [50] | ||
| 8 | W=600 µm Hin=80µm Hou=130µm |
RBCs ~ 7 µm different CTCs ~ 15-20 µm |
1700 µL/min | Not specified | [8] | ||
| 8 | W=600 µm Hin=80µm Hou=130µm |
Polystyrene beads: 10 µm and 15 µm | 6 mL/min | 92% | [51] | ||
| Mammalian cells: Chinese Hamster Ovary cells ~ 10-20 µm | |||||||
| W=450 µm Hin=30µm Hou=70µm |
Polystyrene beads: 4 µm | 2 mL/min | 90% | ||||
| Yeast cells: Saccharomyces cerevisiae ~ 3-5 µm | |||||||
| 8 | W=600 µm Hin=80µm Hou=130µm |
Mesenchymal stem cells (hMSCs) ~ 11-25 µm | 3 mL/min | Not specified | [4] | ||
| 8 | W=600 µm Hin=80µm Hout=130µm |
Polystyrene beads: 6 µm and 10 µm | 1 mL/min | 80-91% | [55] | ||
| Microalgae: Tetraselmis suecica ~ 10.7 µm; Phaeodactylum tricornutum ~ 25.7 and 3.5 µm | |||||||
| 8 | W=580 µm Hin=85µm Hout=133µm |
Mesenchymal stem cells (hMSCs) ~ 11-25 µm | 1.5 mL/min | Not specified | [90] | ||
| 8 | W=600 µm Hin=50µm Hout=90µm |
Polystyrene beads: 5, 10 and 15 µm | 1-5 mL/min | Not specified | [46] | ||
| 8 | W=200 µm Hin=40µm Hout=90µm |
Polystyrene beads: 2,5 and 10 µm | 0.6-1 mL/min | Not specified | [84] | ||
| 4 | W=400 µm Hin=40µm Hout=100µm |
Beer Spoilage Bacteria ~ 2-5 µm |
1.5 mL/min | 90%/>50% | [1] | ||
| 6 | W=500 µm Hin=40µm Hout=70µm |
Polystyrene beads: 4, 6 and 10 µm | 1.5 mL/min | 98% | [77] | ||
| 6 | W=600 µm Hin=80µm Hout=130µm |
Polystyrene beads: 15.45 µm | 1.5 mL/min | Not specified | [91] | ||
| Chinese hamster ovary cells ~ 17.7 µm | |||||||
| 8 | W=600 µm Hout=70µm Hin=110µm |
RBCs ~7 µm WBCs ~ 10-15µm |
800 µL/min | 90% | [66] | ||
| Triangular cross-section | |||||||
| 5 | W= 600 µm Hmax=210µm Hmin= 0 µm |
polystyrene beads: 5, 7, 10, 13 and 20 µm | 4 mL/min | Not specified | [78] | ||
| Hybrid/complex cross-section | |||||||
| Stair-like | |||||||
|
4 |
W1=500 µm H1=110µm W2=100 µm H2=70µm |
7.32 and 20 μm human umbilical vein endothelial cells (HUVEC) and fibroblast cells |
2300 μL/min | Not specified | [56] |
||
| (rectangular + double trapezoidal) | |||||||
| 5 | Dh = 250 µm | Microcarriers ~ 180 µm Mesenchymal Stem Cells (hMSCs)~ 15-30 µm |
10 mL/min | 98% | [69] | ||
| rectangular + trapezoidal | |||||||
| 4.5 | W=400 µm Hin=40µm Hout=100µm |
Polystyrene beads: 4, 6 and 10 µm | 1.5 mL/min | Not specified | [52] | ||
| W=500 µm Hin=40µm Hout=100µm |
Not specified | ||||||
| W=600 µm Hin=40µm Hout=100µm |
97-98% | ||||||
| rectangular + cavities | |||||||
| 5 | W=1600 µm H=50µm |
C. elegans embryos ~ 24 µm adult worms ~ 26, 32, 40, 61 and 78 μm |
1 mL/min | 85% | [75] | ||
| U-shape and W-shape | |||||||
| 10 | W=227 µm Hmax=210µm Hmin=175µm |
microalgaes (Desmodesmus sp) ~ 15 µm bacteria (Escherichia coli) ~ 1 µm |
0.7 mL/min | 92%-72% | [2] | ||
| W=220 µm Hmax=210µm Hmin=162µm |
96%- 66% | ||||||
| Combined techniques devices | |||||||
| Rectangular spiral + cavities + magnetic actuator | |||||||
| 3.5 | W=250 µm H=130µm |
breast cancer cell lines (MCF-7 ~ 15-17 μm and MDA-MB-231 ~12 µm) BCs |
150 μL/min | ~ 97 - 85% |
[6] | ||
| Rectangular spiral + DLD | |||||||
| 2 x 5 | W=200 µm H=80µm |
Polystyrene beads: 5, 8, 15, and 24µm | 1.2 mL/min – 2.2 mL/min |
≤97% | [73] | ||
| RBC: 7.34 µm – WBC: ~12µm lung cancer cells (A549): ~ 10-15µm breast cancer cells (MCF-7): ~15-25µm | |||||||
| Rectangular spiral + magnetic actuator | |||||||
| 4 | W=500 µm H=130µm |
Monodisperse beads: 5 and 15.6 μm Polydisperse beads: from 2 to 20 μm |
1200 μL/min | 86-80 % | [85] | ||
| Rectangular spiral + U turn | |||||||
| 4 | W=500 µm H=180µm |
polydisperse beads: 10µm; monodispersed beads: 5µm and 15µm | 1.7 mL/min | 93% | [74] | ||
5. Conclusions and Perspectives
- Using inexpensive, easy-to-process, recyclable, durable, non-toxic, biocompatible materials suitable for real-time imaging (like PDMS and PMMA) makes these devices potentially Affordable and Delivered.
- Rapid manufacturing techniques that allow mass production and minimize waste materials make these devices affordable. Although soft lithography is widespread, it has issues such as time-consuming and material-intensive pre-fabrication steps and lower precision achievable for complex shapes. In contrast, additive manufacturing (3D printing) allows for precise fabrication of various geometries, though it is limited by the materials available. Ultra-short laser ablation, while less common, offers high precision, speed, and the ability to work with various materials, including PMMA.
- Designing these microfluidic structures on few square centimetres, single or multi-layer chips without the need for external mechanical or electrical forces makes them Equipment-free and User-friendly; the capacity to operate under high flow pressure conditions also makes them Robust.
- By better matching the channel and cross-section dimensions with cell sizes, inertial spiral microchannels could become a Sensitive and Specific sorting technique with high throughput and efficiency.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Condina, M.R.; Dilmetz, B.A.; Razavi Bazaz, S.; Meneses, J.; Ebrahimi Warkiani, M.; Hoffmann, P. Rapid Separation and Identification of Beer Spoilage Bacteria by Inertial Microfluidics and MALDI-TOF Mass Spectrometry. Lab Chip 2019, 19, 1961–1970. [Google Scholar] [CrossRef]
- Abdel-Mawgood, A.L.; Ngum, L.F.; Matsushita, Y.; El-Mashtoly, S.F.; Fath El-Bab, A.M.R. Separation of Microalgae from Bacterial Contaminants Using Spiral Microchannel in the Presence of a Chemoattractant. 2024. [CrossRef]
- Xavier, M.; Oreffo, R.O.C.; Morgan, H. Skeletal Stem Cell Isolation: A Review on the State-of-the-Art Microfluidic Label-Free Sorting Techniques. Biotechnol Adv 2016, 34, 908–923. [Google Scholar] [CrossRef]
- Poon, Z.; Lee, W.C.; Guan, G.; Nyan, L.M.; Lim, C.T.; Han, J.; Van Vliet, K.J. Bone Marrow Regeneration Promoted by Biophysically Sorted Osteoprogenitors From Mesenchymal Stromal Cells. Stem Cells Transl Med 2015, 4, 56–65. [Google Scholar] [CrossRef]
- Vaidyanathan, R.; Yeo, T.; Lim, C.T. Microfluidics for Cell Sorting and Single Cell Analysis from Whole Blood. In Methods in Cell Biology; Academic Press Inc., 2018; Vol. 147, pp. 151–173 ISBN 9780128142820.
- Kwak, B.; Lee, J.; Lee, J.; Kim, H.S.; Kang, S.; Lee, Y. Spiral Shape Microfluidic Channel for Selective Isolating of Heterogenic Circulating Tumor Cells. Biosens Bioelectron 2018, 101, 311–316. [Google Scholar] [CrossRef]
- Mohammadi, A.; Rahmanian, M. Label-Free Isolation of Circulating Tumor Cells Using Lateral Dielectrophoresis- Assisted Inertial. Microfluid Nanofluidics 2023. [Google Scholar] [CrossRef]
- Warkiani, M.E.; Guan, G.; Luan, K.B.; Lee, W.C.; Bhagat, A.A.S.; Kant Chaudhuri, P.; Tan, D.S.W.; Lim, W.T.; Lee, S.C.; Chen, P.C.Y.; et al. Slanted Spiral Microfluidics for the Ultra-Fast, Label-Free Isolation of Circulating Tumor Cells. Lab Chip 2014, 14, 128–137. [Google Scholar] [CrossRef]
- Stephenson, J. Lab-on-a-Chip Shows Promise in Defining and Diagnosing Cancers, 1999.
- Zhang, J.; Yan, S.; Yuan, D.; Alici, G.; Nguyen, N.T.; Ebrahimi Warkiani, M.; Li, W. Fundamentals and Applications of Inertial Microfluidics: A Review. Lab Chip 2016, 16, 10–34. [Google Scholar] [CrossRef] [PubMed]
- Wyatt Shields Iv, C.; Reyes, C.D.; López, G.P. Microfluidic Cell Sorting: A Review of the Advances in the Separation of Cells from Debulking to Rare Cell Isolation. Lab Chip 2015, 15, 1230–1249. [Google Scholar] [CrossRef] [PubMed]
- Antoniadi, I.; Skalický, V.; Sun, G.; Ma, W.; Galbraith, D.W.; Novák, O.; Ljung, K. Fluorescence Activated Cell Sorting—A Selective Tool for Plant Cell Isolation and Analysis. Cytometry Part A 2022, 101, 725–736. [Google Scholar] [CrossRef]
- Yaman, S.; Anil-Inevi, M.; Ozcivici, E.; Tekin, H.C. Magnetic Force-Based Microfluidic Techniques for Cellular and Tissue Bioengineering. Front Bioeng Biotechnol 2018, 6. [Google Scholar] [CrossRef]
- Bonaccorso, F.; Zerbetto, M.; Ferrari, A.C.; Amendola, V. Sorting Nanoparticles by Centrifugal Fields in Clean Media. Journal of Physical Chemistry C 2013, 117, 13217–13229. [Google Scholar] [CrossRef]
- Orfao, A.; Ruiz-Arguelles, A. General Concepts About Cell Sorting Techniques; 1996; Vol. 29;
- Rahman, N.A.; Ibrahim, F.; Yafouz, B. Dielectrophoresis for Biomedical Sciences Applications: A Review. Sensors (Switzerland) 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Alnaimat, F.; Dagher, S.; Mathew, B.; Hilal-Alnqbi, A.; Khashan, S. Microfluidics Based Magnetophoresis: A Review. Chemical Record 2018, 18, 1596–1612. [Google Scholar] [CrossRef] [PubMed]
- Dao, M.; Suresh, S.; Huang, T.J.; Li, P.; Mao, Z.; Peng, Z.; Zhou, L.; Chen, Y.; Huang, P.H.; Truica, C.I.; et al. Acoustic Separation of Circulating Tumor Cells. Proc Natl Acad Sci U S A 2015, 112, 4970–4975. [Google Scholar] [CrossRef]
- Kim, G.Y.; Han, J.I.; Park, J.K. Inertial Microfluidics-Based Cell Sorting. Biochip J 2018, 12, 257–267. [Google Scholar] [CrossRef]
- Volpe, A.; Paiè, P.; Ancona, A.; Osellame, R. Polymeric Fully Inertial Lab-on-a-Chip with Enhanced-Throughput Sorting Capabilities. Microfluid Nanofluidics 2019, 23. [Google Scholar] [CrossRef]
- Shen, Y.; Yalikun, Y.; Tanaka, Y. Recent Advances in Microfluidic Cell Sorting Systems. Sens Actuators B Chem 2019, 282, 268–281. [Google Scholar] [CrossRef]
- Whitesides, G.M. The Origins and the Future of Microfluidics. Nature 2006, 442, 368–373. [Google Scholar] [CrossRef]
- Mickey Urdea; Laura A. Penny; Stuart S. Olmsted; Maria Y. Giovanni; Peter Kaspar; Andrew Shepherd; Penny Wilson; Carol A. Dahl; Steven Buchsbaum; Gerry Moeller; et al. Requirements for High Impact Diagnostics in Developing World. Nature.
- De Timàry, G.; Cappello, J.; Scheid, B. Enhanced Pinch Flow Fractionation Using Inertial Streamline Crossing. 2022. [CrossRef]
- McGrath, J.; Jimenez, M.; Bridle, H. Deterministic Lateral Displacement for Particle Separation: A Review. Lab Chip 2014, 14, 4139–4158. [Google Scholar] [CrossRef]
- Choi, S.; Song, S.; Choi, C.; Park, J.K. Continuous Blood Cell Separation by Hydrophoretic Filtration. Lab Chip 2007, 7, 1532–1538. [Google Scholar] [CrossRef] [PubMed]
- Van Holten, R.W.; Autenrieth, S.; Boose, J.A.; Hsieh, W.-T.; Dolan, S. Removal of Prion Challenge from an Immune Globulin Preparation by Use of a Size-Exclusion Filter, 2002.
- Bhattacharjee, C.; Saxena, V.K.; Dutta, S. Static Turbulence Promoters in Cross-Flow Membrane Filtration: A Review. Chem Eng Commun 2020, 207, 413–433. [Google Scholar] [CrossRef]
- Paiè, P.; Bragheri, F.; Di Carlo, D.; Osellame, R. Particle Focusing by 3D Inertial Microfluidics. Microsyst Nanoeng 2017, 3. [Google Scholar] [CrossRef]
- Volpe, A.; Gaudiuso, C.; Ancona, A. Sorting of Particles Using Inertial Focusing and Laminar Vortex Technology: A Review. Micromachines (Basel) 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- D. J. Tritton Physical Fluid Dynamics; 1977; ISBN 978-0-442-30132-3 Published:. 01 January 1997.
- Matas, J.P.; Morris, J.F.; Guazzelli, E. Lateral Forces on a Sphere Solid/Liquid Dispersions in Drilling and Production Fluides Chargés En Forage et Production Pétrolière; 2004; Vol. 59;
- Di Carlo, D.; Irimia, D.; Tompkins, R.G.; Toner, M. Continuous Inertial Focusing, Ordering, and Separation of Particles in Microchannels. Proc Natl Acad Sci U S A 2007, 104, 18892–18897. [Google Scholar] [CrossRef] [PubMed]
- Nivedita, N.; Papautsky, I. Continuous Separation of Blood Cells in Spiral Microfluidic Devices. Biomicrofluidics 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- G. Segrè; A. Silberberg Radial Particle Displacements in Poiseuille Flow. Nature 1960. [Google Scholar]
- Smits, A.J. A Physical Introduction to Fluid Mechanics, 2024.
- Tabeling Patrick Introduction to Microfluidics, Oxford University Press, Ed.; Second edition. 2005.
- 2005.
- J. Hansson1; J.M. Karlsson2; T. Haraldsson2; W. van der Wijngaart2; A. Russom INERTIAL PARTICLE FOCUSING IN PARALLEL MICROFLUIDIC CHANNELS FOR HIGH-THROUGHPUT FILTRATION; IEEE, 2011; ISBN 9781457701566.
- Asmolov, E.S. The Inertial Lift on a Spherical Particle in a Plane Poiseuille Flow at Large Channel Reynolds Number; 1999; Vol. 381;
- Zhou, J.; Papautsky, I. Fundamentals of Inertial Focusing in Microchannels. Lab Chip 2013, 13, 1121–1132. [Google Scholar] [CrossRef]
- Kuntaegowdanahalli, S.S.; Bhagat, A.A.S.; Kumar, G.; Papautsky, I. Inertial Microfluidics for Continuous Particle Separation in Spiral Microchannels. Lab Chip 2009, 9, 2973–2980. [Google Scholar] [CrossRef]
- Walz D., G. F. The Radial Velocity of Spherical Particles in Tubular Pinch Effect Experiments, 1973. [Google Scholar]
- Bhagat, A.A.S.; Kuntaegowdanahalli, S.S.; Papautsky, I. Inertial Microfluidics for Continuous Particle Filtration and Extraction. Microfluid Nanofluidics 2009, 7, 217–226. [Google Scholar] [CrossRef]
- Dean, W.R.; Hurst, J.M.; The London, E. and D.P.M. and J. XVI Note on the Motion of Fluid in a Curved Pipe, 1959. [Google Scholar]
- Al-Halhouli, A.; Al-Faqheri, W.; Alhamarneh, B.; Hecht, L.; Dietzel, A. Spiral Microchannels with Trapezoidal Cross Section Fabricated by Femtosecond Laser Ablation in Glass for the Inertial Separation of Microparticles. Micromachines (Basel) 2018, 9. [Google Scholar] [CrossRef]
- Warkiani, M.E.; Khoo, B.L.; Wu, L.; Tay, A.K.P.; Bhagat, A.A.S.; Han, J.; Lim, C.T. Ultra-Fast, Label-Free Isolation of Circulating Tumor Cells from Blood Using Spiral Microfluidics. Nat Protoc 2016, 11, 134–148. [Google Scholar] [CrossRef] [PubMed]
- Ramya, S.; Kumar, S.P.; Ram, G.D.; Lingaraja, D. A Short Review of Spiral Microfluidic Devices with Distinct Cross-Sectional Geometries. Microfluid Nanofluidics 2022, 26. [Google Scholar] [CrossRef]
- Chun, B.; Ladd, A.J.C. Inertial Migration of Neutrally Buoyant Particles in a Square Duct: An Investigation of Multiple Equilibrium Positions. Physics of Fluids 2006, 18. [Google Scholar] [CrossRef]
- Guan, G.; Wu, L.; Bhagat, A.A.; Li, Z.; Chen, P.C.Y.; Chao, S.; Ong, C.J.; Han, J. Spiral Microchannel with Rectangular and Trapezoidal Cross-Sections for Size Based Particle Separation. Sci Rep 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Warkiani, M.E.; Tay, A.K.P.; Guan, G.; Han, J. Membrane-Less Microfiltration Using Inertial Microfluidics. Sci Rep 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Mihandoust, A.; Bazaz, S.R.; Maleki-Jirsaraei, N.; Alizadeh, M.; Taylor, R.A.; Warkiani, M.E. High-Throughput Particle Concentration Using Complex Cross-Section Microchannels. Micromachines (Basel) 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Zhu, S.; Jiang, D.; Zhu, L.; Yang, J.; Xiang, N. Channel Innovations for Inertial Microfluidics. Lab Chip 2020, 20, 3485–3502. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Guan, G.; Hou, H.W.; Bhagat, A.A.S.; Han, J. Separation of Leukocytes from Blood Using Spiral Channel with Trapezoid Cross-Section. Anal Chem 2012, 84, 9324–9331. [Google Scholar] [CrossRef] [PubMed]
- Syed, M.S.; Rafeie, M.; Vandamme, D.; Asadnia, M.; Henderson, R.; Taylor, R.A.; Warkiani, M.E. Selective Separation of Microalgae Cells Using Inertial Microfluidics. Bioresour Technol 2018, 252, 91–99. [Google Scholar] [CrossRef]
- Ghadami, S.; Kowsari-Esfahan, R.; Saidi, M.S.; Firoozbakhsh, K. Spiral Microchannel with Stair-like Cross Section for Size-Based Particle Separation. Microfluid Nanofluidics 2017, 21. [Google Scholar] [CrossRef]
- Waldbaur, A.; Rapp, H.; Länge, K.; Rapp, B.E. Let There Be Chip - Towards Rapid Prototyping of Microfluidic Devices: One-Step Manufacturing Processes. Analytical Methods 2011, 3, 2681–2716. [Google Scholar] [CrossRef]
- Abgrall, P.; Gué, A.M. Lab-on-Chip Technologies: Making a Microfluidic Network and Coupling It into a Complete Microsystem - A Review. Journal of Micromechanics and Microengineering 2007, 17. [Google Scholar] [CrossRef]
- Ren, K.; Zhou, J.; Wu, H. Materials for Microfluidic Chip Fabrication. Acc Chem Res 2013, 46, 2396–2406. [Google Scholar] [CrossRef] [PubMed]
- Roy, E.; Pallandre, A.; Zribi, B.; Horny, M.-C.; Delapierre, F.D.; Cattoni, A.; Gamby, J.; Haghiri-Gosnet, A.-M. Overview of Materials for Microfluidic Applications. In Advances in Microfluidics - New Applications in Biology, Energy, and Materials Sciences; InTech, 2016.
- Gale, B.K.; Jafek, A.R.; Lambert, C.J.; Goenner, B.L.; Moghimifam, H.; Nze, U.C.; Kamarapu, S.K. A Review of Current Methods in Microfluidic Device Fabrication and Future Commercialization Prospects. Inventions 2018, 3. [Google Scholar] [CrossRef]
- Huang, D.; Man, J.; Jiang, D.; Zhao, J.; Xiang, N. Inertial Microfluidics: Recent Advances. Electrophoresis 2020, 41, 2166–2187. [Google Scholar] [CrossRef] [PubMed]
- Gregoratto, I.; McNeil, C.J.; Reeks, M.W. Micro-Devices for Rapid Continuous Separation of Suspensions for Use in Micro-Total-Analysis-Systems (ΜTAS). In Proceedings of the Microfluidics, BioMEMS, and Medical Microsystems V; SPIE, January 20 2007; Vol. 6465; p. 646503. [Google Scholar]
- Rodrigues, R.O.; Lima, R.; Gomes, H.T.; Silva, A.M.T. Polymer Microfluidic Devices: An Overview of Fabrication Methods. U.Porto Journal of Engineering 2015, 1, 67–79. [Google Scholar] [CrossRef]
- Nielsen, J.B.; Hanson, R.L.; Almughamsi, H.M.; Pang, C.; Fish, T.R.; Woolley, A.T. Microfluidics: Innovations in Materials and Their Fabrication and Functionalization. Anal Chem 2020, 92, 150–168. [Google Scholar] [CrossRef]
- Adel, M.; Allam, A.; Sayour, A.E.; Ragai, H.F.; Umezu, S.; Fath El-Bab, A.M.R. Fabrication of Spiral Low-Cost Microchannel with Trapezoidal Cross Section for Cell Separation Using a Grayscale Approach. Micromachines (Basel) 2023, 14, 1340. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.G.A.; Ambhorkar, P.; Samanipour, R.; Yang, A.; Ghafoor, A.; Kim, K. Microfluidics-Based Fabrication of Cell-Laden Microgels. Biomicrofluidics 2020, 14. [Google Scholar] [CrossRef]
- Shen, C.; Li, Y.; She, W.; Meng, Q. Layer-by-Layer Adhesion of Hydrogels for Constructing Heterogeneous Microfluidic Chips. 2023. [CrossRef]
- Raoufi, M.A.; Razavi Bazaz, S.; Niazmand, H.; Rouhi, O.; Asadnia, M.; Razmjou, A.; Ebrahimi Warkiani, M. Fabrication of Unconventional Inertial Microfluidic Channels Using Wax 3D Printing. Soft Matter 2020, 16, 2448–2459. [Google Scholar] [CrossRef]
- Tumbleston, J.R.; Shirvanyants, D.; Ermoshkin, N.; Janusziewicz, R.; Johnson, A.R.; Kelly, D.; Chen, K.; Pinschmidt, R.; Rolland, J.P.; Ermoshkin, A.; et al. Continuous Liquid Interface Production of 3D Objects. Science (1979) 2015, 347, 1349–1352. [Google Scholar] [CrossRef] [PubMed]
- Bhagat, A.A.S.; Kuntaegowdanahalli, S.S.; Papautsky, I. Continuous Particle Separation in Spiral Microchannels Using Dean Flows and Differential Migration. Lab Chip 2008, 8, 1906–1914. [Google Scholar] [CrossRef] [PubMed]
- Abdulla, A.; Liu, W.; Gholamipour-Shirazi, A.; Sun, J.; Ding, X. High-Throughput Isolation of Circulating Tumor Cells Using Cascaded Inertial Focusing Microfluidic Channel. Anal Chem 2018, 90, 4397–4405. [Google Scholar] [CrossRef] [PubMed]
- Omrani, V.; Targhi, M.Z.; Rahbarizadeh, F.; Nosrati, R. High-Throughput Isolation of Cancer Cells in Spiral Microchannel by Changing the Direction, Magnitude and Location of the Maximum Velocity. Sci Rep 2023, 13. [Google Scholar] [CrossRef] [PubMed]
- Pan, P.; Qin, Z.; Sun, W.; Zhou, Y.; Wang, S.; Song, P.; Wang, Y.; Ru, C.; Wang, X.; Calarco, J.; et al. A Spiral Microfluidic Device for Rapid Sorting, Trapping, and Long-Term Live Imaging of Caenorhabditis Elegans Embryos. Microsyst Nanoeng 2023, 9. [Google Scholar] [CrossRef]
- Esan, A.; Vanholsbeeck, F.; Swift, S.; McGoverin, C.M. Continuous Separation of Bacterial Cells from Large Debris Using a Spiral Microfluidic Device. Biomicrofluidics 2023, 17. [Google Scholar] [CrossRef] [PubMed]
- Mihandoust, A.; Maleki-Jirsaraei, N.; Rouhani, S.; Safi, S.; Alizadeh, M. Improvement of Size-Based Particle Separation Throughput in Slanted Spiral Microchannel by Modifying Outlet Geometry. Electrophoresis 2020, 41, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Razavi Bazaz, S.; Rouhi, O.; Raoufi, M.A.; Ejeian, F.; Asadnia, M.; Jin, D.; Ebrahimi Warkiani, M. 3D Printing of Inertial Microfluidic Devices. Sci Rep 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, V.; Pinto, V.; Sousa, P.; Gonçalves, L.; Fernández, E.; Minas, G. Spiral Inertial Microfluidics for Separation and Concentration of Phytoplankton. Algal Res 2023, 76. [Google Scholar] [CrossRef]
- Caffiyar, M.Y.; Lim, K.P.; Kamal Basha, I.H.; Hamid, N.H.; Cheong, S.C.; Wei Ho, E.T. Label-Free, High-Throughput Assay of Human Dendritic Cells from Whole-Blood Samples with Microfluidic Inertial Separation Suitable for Resource-Limited Manufacturing. Micromachines (Basel) 2020, 11. [Google Scholar] [CrossRef]
- Eaton, S.M.; De Marco, C.; Martinez-Vazquez, R.; Ramponi, R.; Turri, S.; Cerullo, G.; Osellame, R. Femtosecond Laser Microstructuring for Polymeric Lab-on-Chips. J Biophotonics 2012, 5, 687–702. [Google Scholar] [CrossRef] [PubMed]
- Volpe, A.; Trotta, G.; Krishnan, U.; Ancona, A. Prediction Model of the Depth of the Femtosecond Laser Micro-Milling of PMMA. Opt Laser Technol 2019, 120. [Google Scholar] [CrossRef]
- Volpe, A.; Krishnan, U.; Chiriacò, M.S.; Primiceri, E.; Ancona, A.; Ferrara, F. A Smart Procedure for the Femtosecond Laser-Based Fabrication of a Polymeric Lab-on-a-Chip for Capturing Tumor Cell. Engineering 2021, 7, 1434–1440. [Google Scholar] [CrossRef]
- Al-Halhouli, A.; Albagdady, A.; Dietzel, A. Sheath-Less High Throughput Inertial Separation of Small Microparticles in Spiral Microchannels with Trapezoidal Cross-Section. RSC Adv 2019, 9, 41970–41976. [Google Scholar] [CrossRef]
- Tabatabaei, S.A.; Zabetian, M.; Zabetian Targhi, M. Design and Experimental Investigation of a Novel Spiral Microouidic Chip to Separate Wide Size Range of Micro-Particles Design and Experimental Investigation of a Novel Spiral Microfluidic Chip to Separate Wide Size Range of Micro-Particles. 2020. [CrossRef]
- Shiriny, A.; Bayareh, M. Inertial Focusing of CTCs in a Novel Spiral Microchannel. Chem Eng Sci 2021, 229. [Google Scholar] [CrossRef]
- McFaul, S.M.; Lin, B.K.; Ma, H. Cell Separation Based on Size and Deformability Using Microfluidic Funnel Ratchets. Lab Chip 2012, 12, 2369–2376. [Google Scholar] [CrossRef]
- Son, J.; Murphy, K.; Samuel, R.; Gale, B.K.; Carrell, D.T.; Hotaling, J.M. Non-Motile Sperm Cell Separation Using a Spiral Channel. Analytical Methods 2015, 7, 8041–8047. [Google Scholar] [CrossRef]
- Keating, A. Mesenchymal Stromal Cells: New Directions. Cell Stem Cell 2012, 10, 709–716. [Google Scholar] [CrossRef]
- Yin, L.; Wu, Y.; Yang, Z.; Tee, C.A.; Denslin, V.; Lai, Z.; Lim, C.T.; Lee, E.H.; Han, J. Microfluidic Label-Free Selection of Mesenchymal Stem Cell Subpopulation during Culture Expansion Extends the Chondrogenic Potential: In Vitro. Lab Chip 2018, 18, 878–889. [Google Scholar] [CrossRef]
- Yin, L.; Au, W.Y.; Yu, C.C.; Kwon, T.; Lai, Z.; Shang, M.; Warkiani, M.E.; Rosche, R.; Lim, C.T.; Han, J. Miniature Auto-Perfusion Bioreactor System with Spiral Microfluidic Cell Retention Device. Biotechnol Bioeng 2021, 118, 1951–1961. [Google Scholar] [CrossRef]
- Saha, S.C.; Francis, I.; Nassir, T. Computational Inertial Microfluidics: Optimal Design for Particle Separation. Fluids 2022, 7. [Google Scholar] [CrossRef]
- Milo, R.; Jorgensen, P.; Moran, U.; Weber, G.; Springer, M. BioNumbers The Database of Key Numbers in Molecular and Cell Biology. Nucleic Acids Res 2009, 38. [Google Scholar] [CrossRef] [PubMed]
- Truongvo, T.N.; Kennedy, R.M.; Chen, H.; Chen, A.; Berndt, A.; Agarwal, M.; Zhu, L.; Nakshatri, H.; Wallace, J.; Na, S.; et al. Microfluidic Channel for Characterizing Normal and Breast Cancer Cells. Journal of Micromechanics and Microengineering 2017, 27. [Google Scholar] [CrossRef]
- Kwak, B.; Lee, J.; Lee, D.; Lee, K.; Kwon, O.; Kang, S.; Kim, Y. Selective Isolation of Magnetic Nanoparticle-Mediated Heterogeneity Subpopulation of Circulating Tumor Cells Using Magnetic Gradient Based Microfluidic System. Biosens Bioelectron 2017, 88, 153–158. [Google Scholar] [CrossRef]
- Warkiani, M.E.; Guan, G.; Luan, K.B.; Lee, W.C.; Bhagat, A.A.S.; Kant Chaudhuri, P.; Tan, D.S.W.; Lim, W.T.; Lee, S.C.; Chen, P.C.Y.; et al. Slanted Spiral Microfluidics for the Ultra-Fast, Label-Free Isolation of Circulating Tumor Cells. Lab Chip 2014, 14, 128–137. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




