Submitted:
11 July 2024
Posted:
12 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Overview on the Early Morphogenesis of Vertebrate Hearts
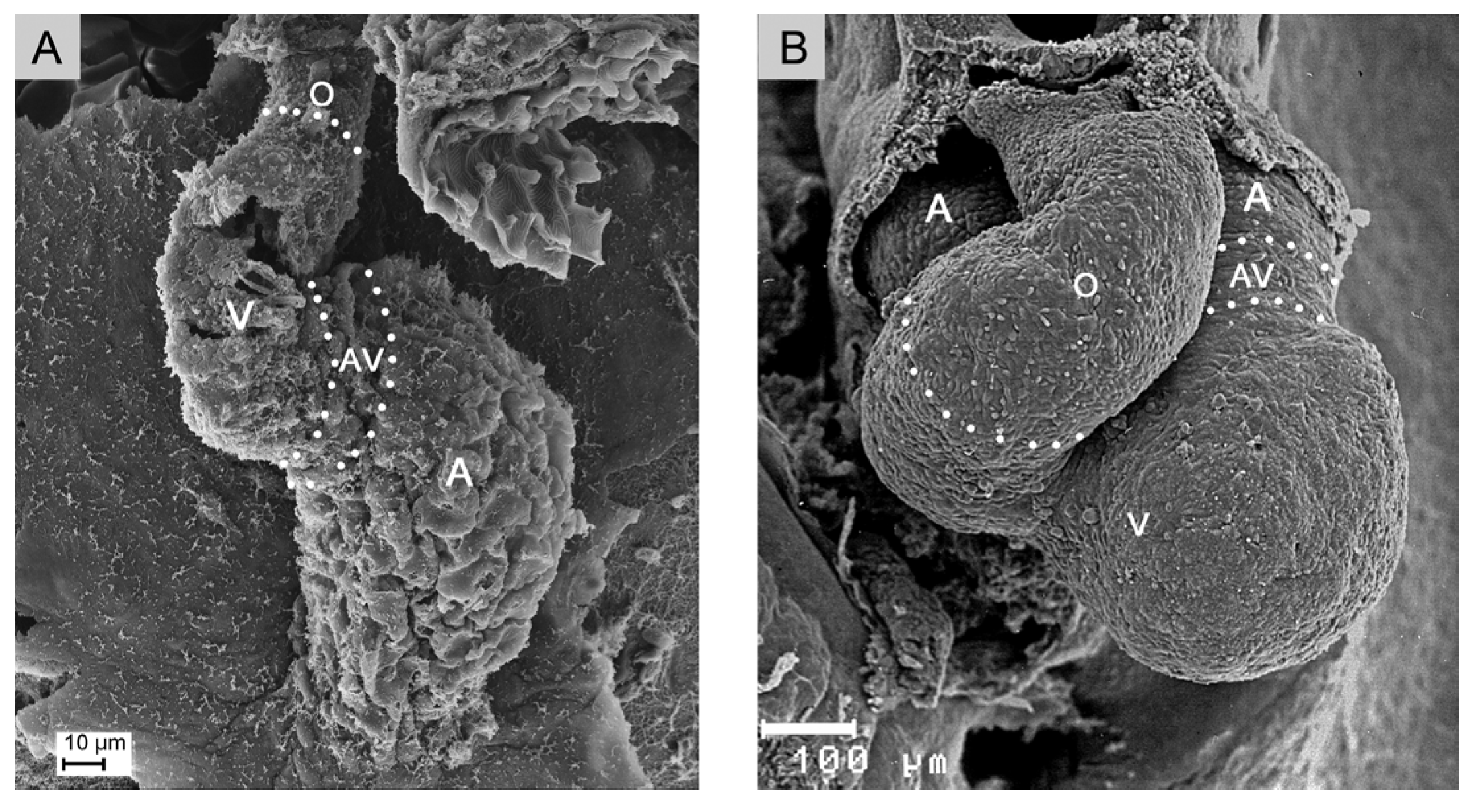
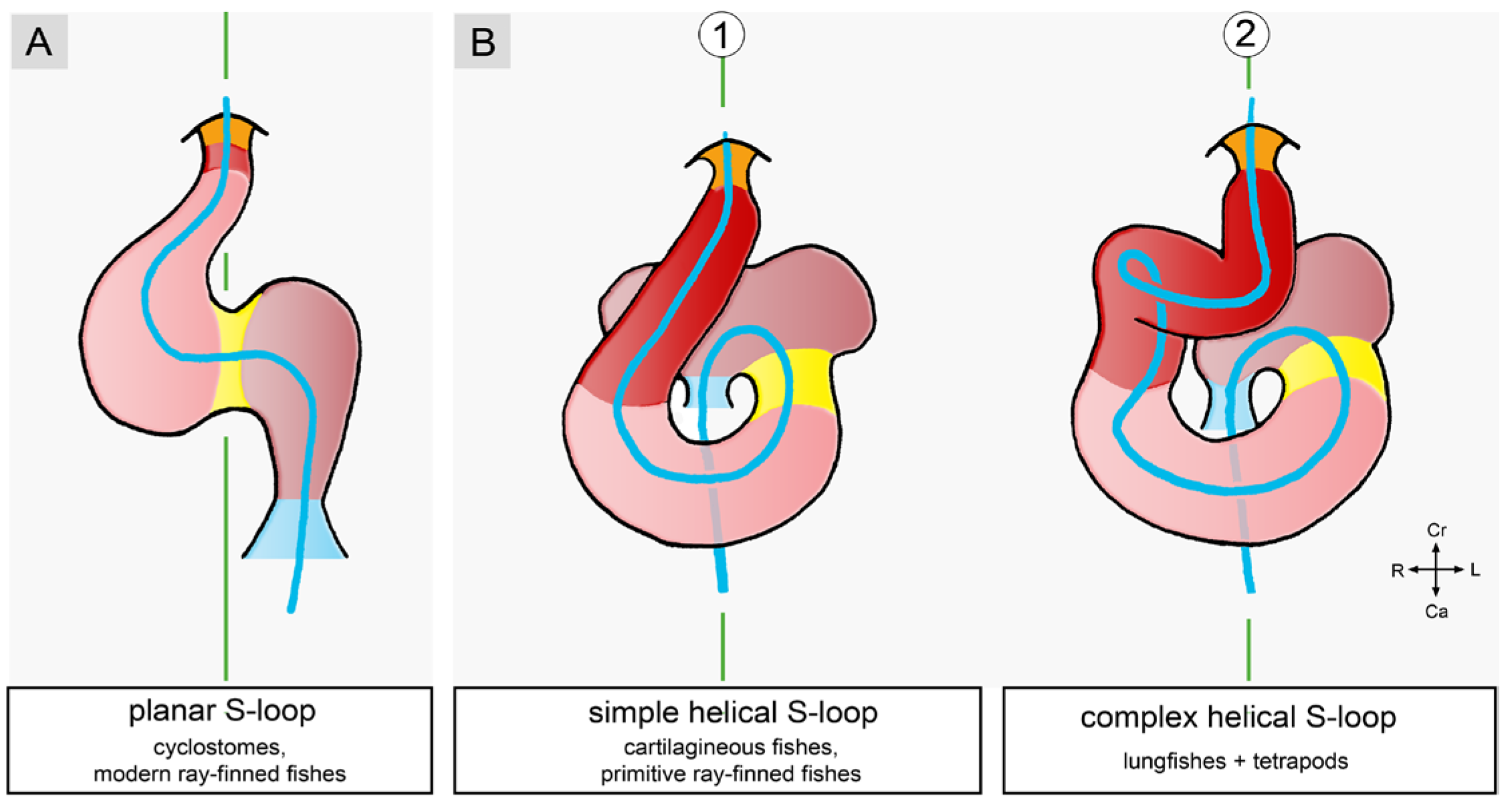
2.1. Comparative Aspects of Cardiac Looping among Vertebrates
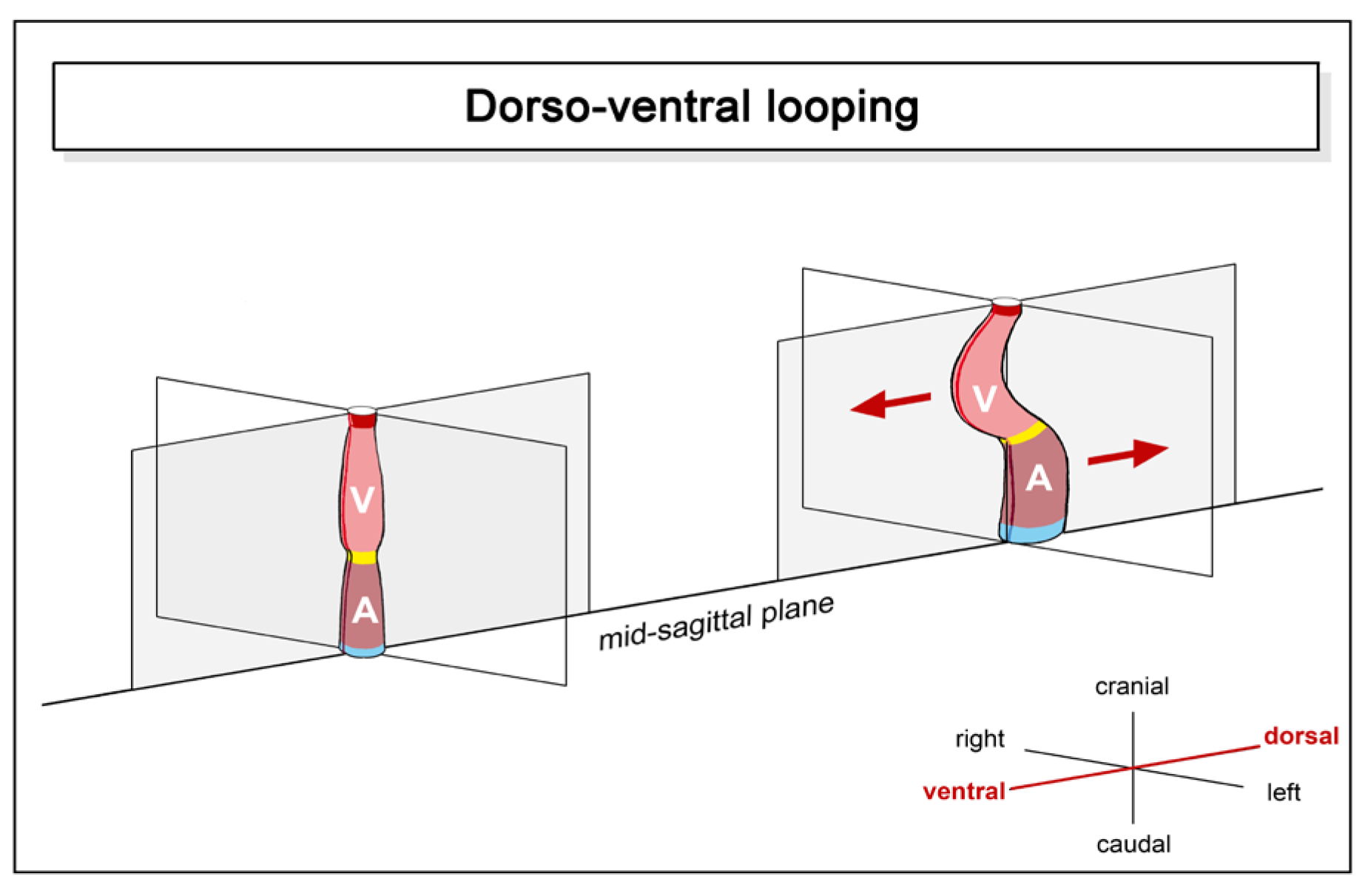
- (1), positional and morphological changes along the original dorso-ventral heart axis.
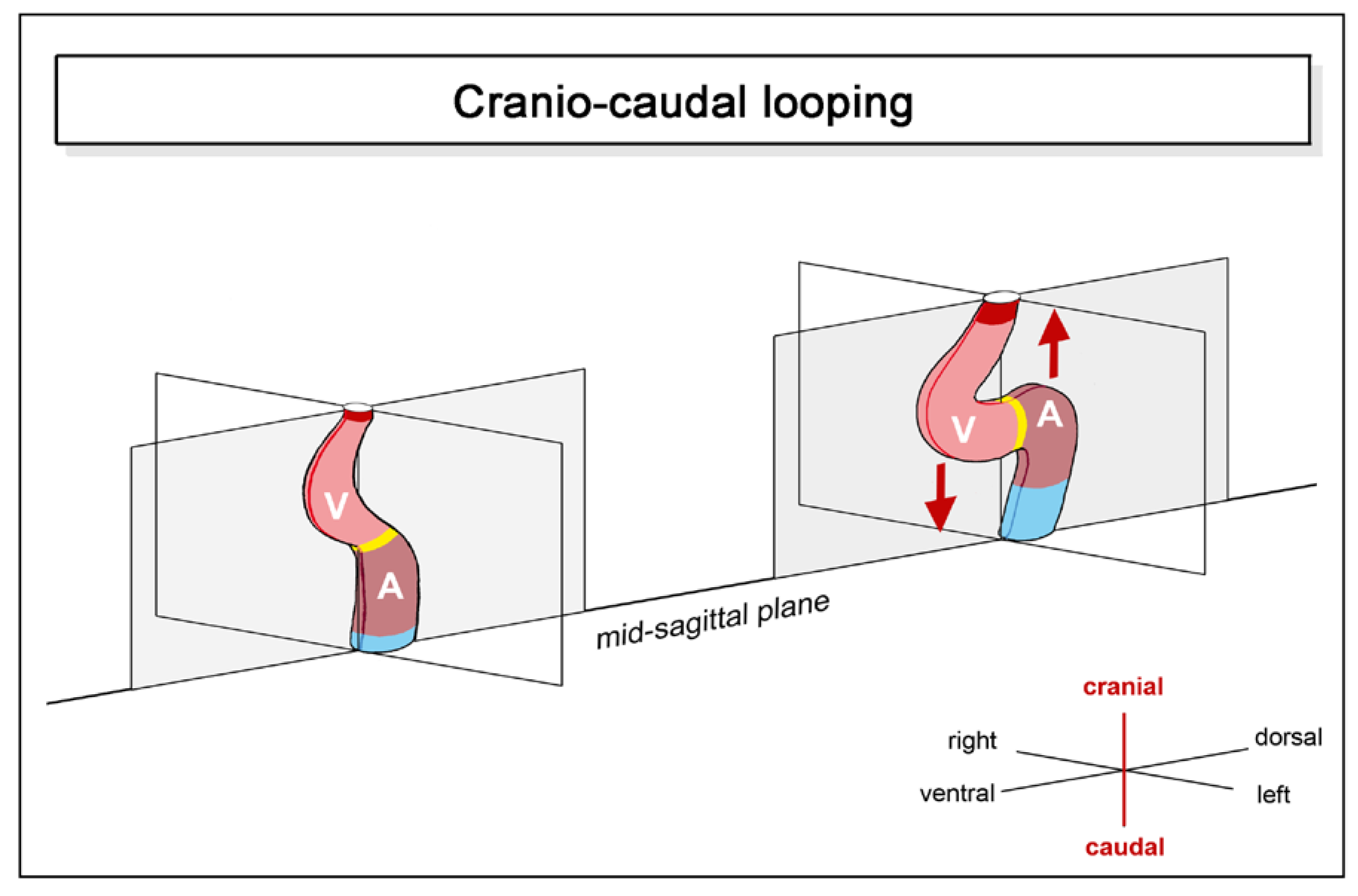
- (2), positional and morphological changes along the cranio-caudal body axis.
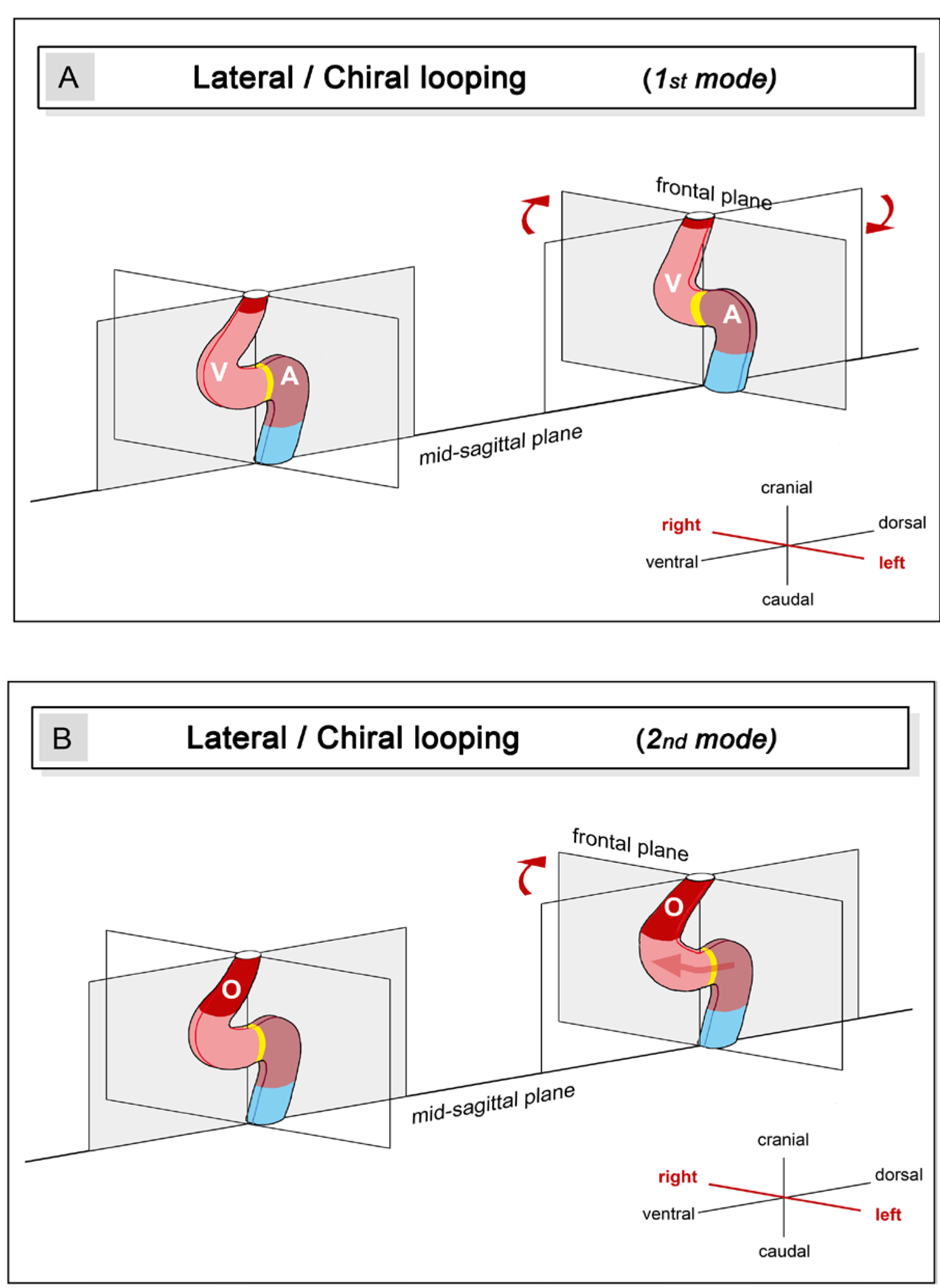
- (3), positional and morphological changes along the so-called left-right body axis, which lead to a bilaterally asymmetric heart shape. This component may be named “lateral looping” or “chiral looping”.
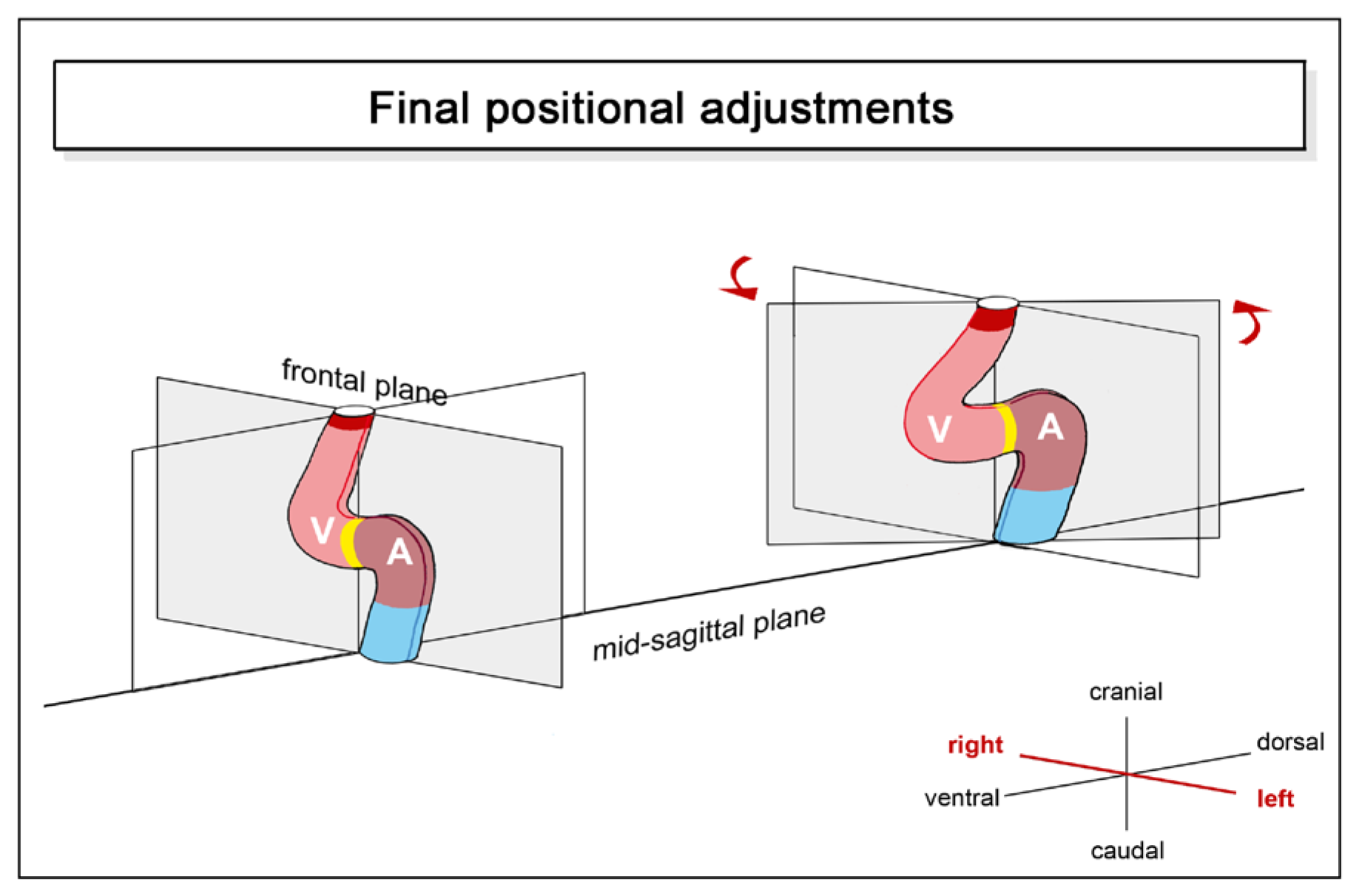
- (4), positional and morphological changes, which reduce the degree of lateral/chiral looping reached at earlier stages of cardiac looping. This component may be named “final positional adjustments.”
3. Comparative Anatomy of the Looped Design of Mature Vertebrate Hearts
3.1. Comparative Anatomy of the Sigmoid Routing of the Flow Path(s) of Mature Vertebrate Hearts
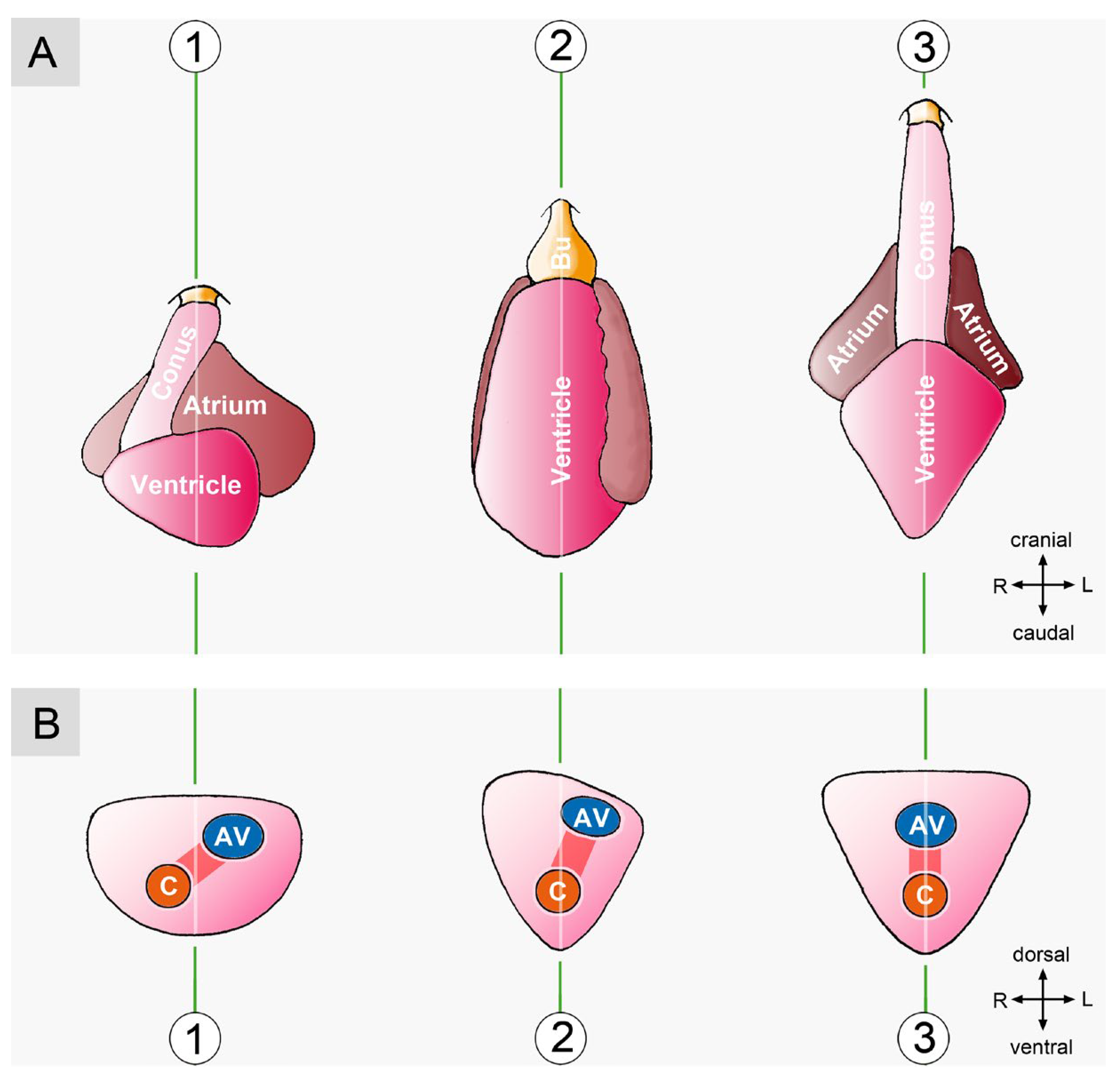
3.2. Comparative Anatomy of the Bilaterally Asymmetric (chiral) Routing of the Flow Path(s) of Mature Vertebrate Hearts
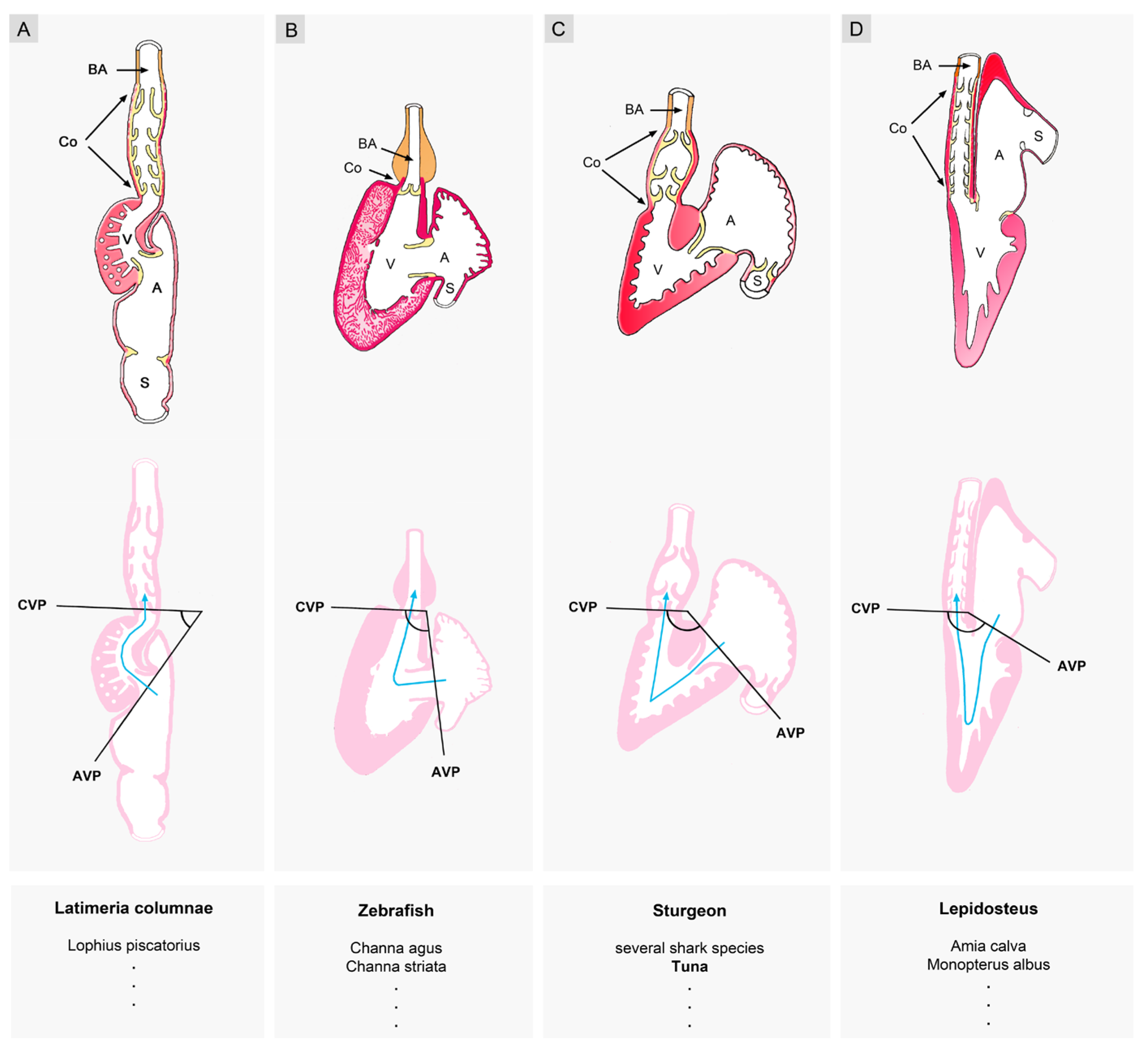
3.2.1. Hearts with a Visually Conspicuous Bilateral Asymmetry
3.2.2. Hearts with a Visually Obscured Bilateral Asymmetry
3.2.3. Hearts with a Nearly Perfect Bilateral Symmetry
4. The Functional Significance of the Looped Design of Embryonic and Mature Vertebrate Hearts
4.1. A Closer View on the Functional Design of Embryonic Vertebrate Hearts
4.2. A Closer View on the Functional Design of Mature Vertebrate Hearts
4.2.1. Looping Dictates the Alignments and Separations of the Systemic and Pulmonary Flow Paths
4.2.2. Looping Might Optimize the Cardiac Pumping Efficiency
4.2.3. A critical Evaluation of the Proposed Functional Advantages of the Sigmoid Routing of the Cardiac Flow Path(s)
4.2.3.1. Does Looping Really Minimize the Loss of Momentum of the Inflowing Blood?
4.2.3.2. Does Looping Really Enhance Ventriculo-Atrial Coupling?
4.2.3.3. Implications for the Usage of Fishes in Cardiovascular Research
5. Summary and Conclusions
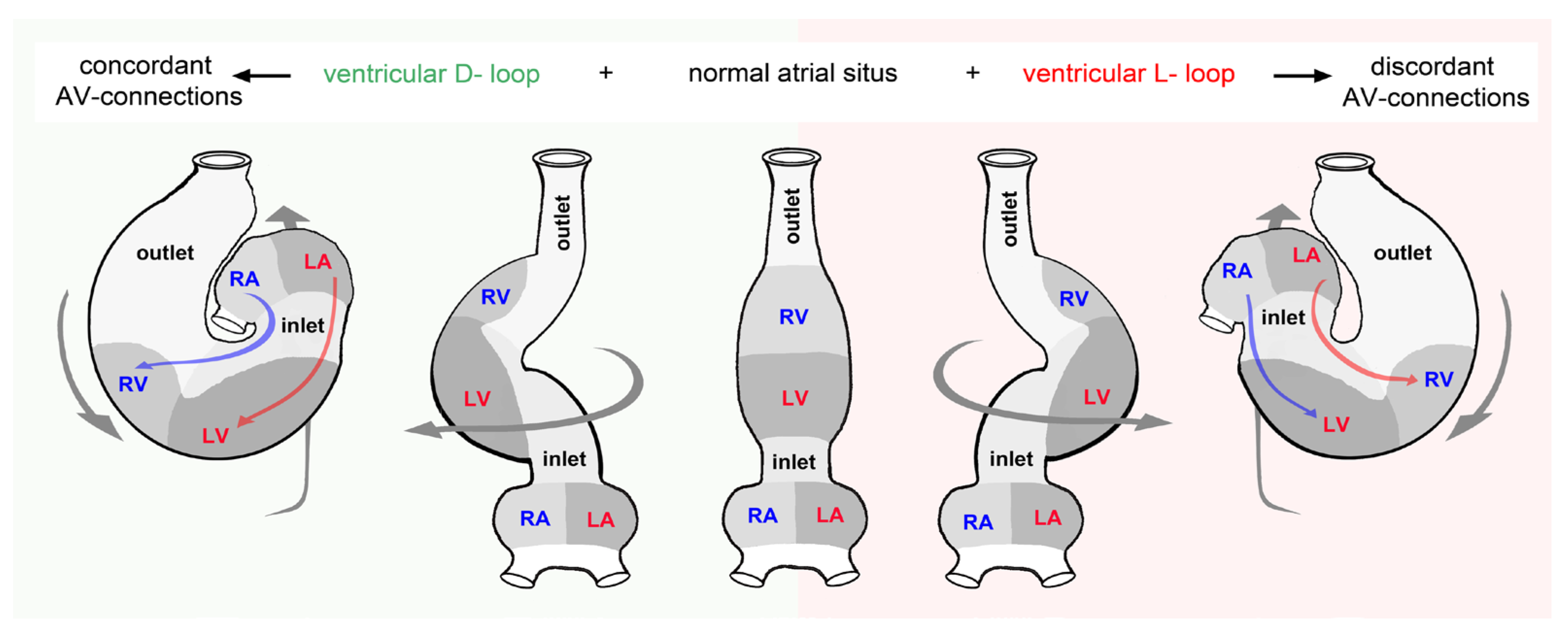
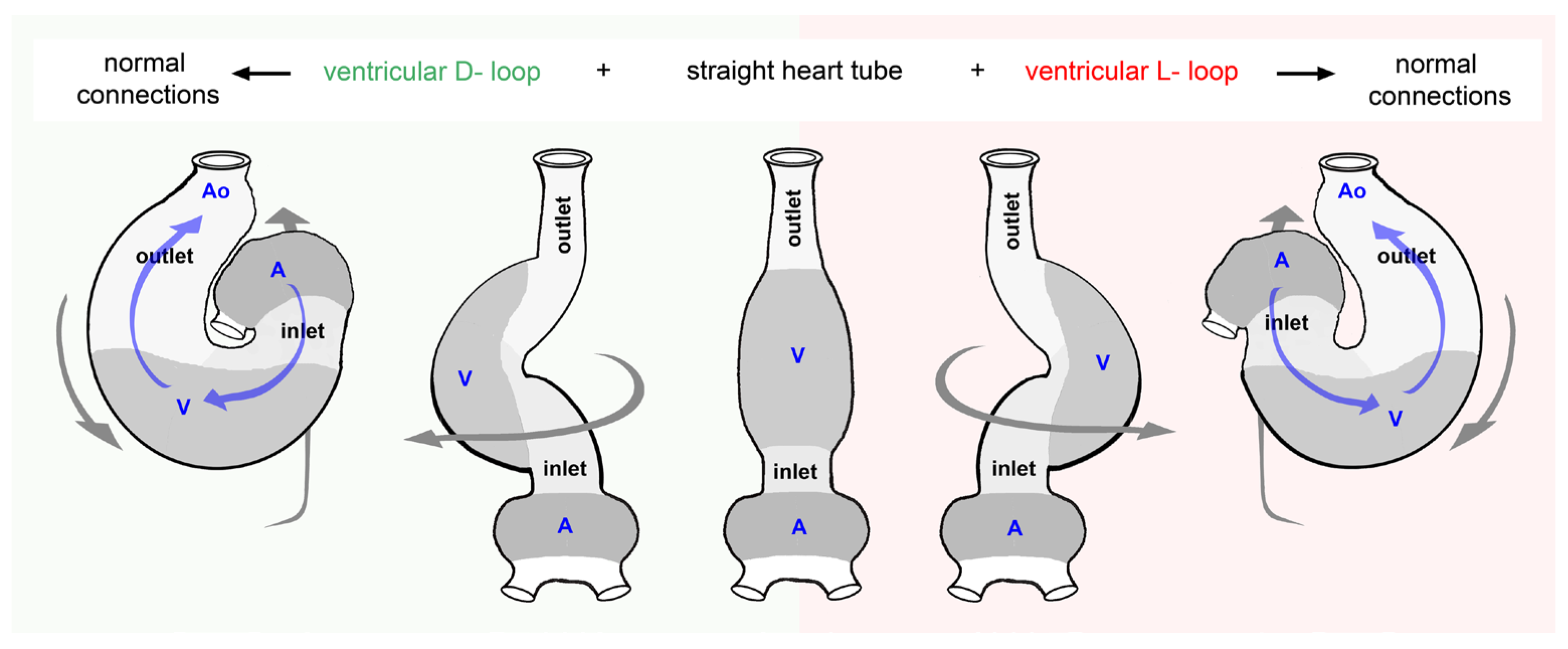
- (1), the looped design of vertebrate hearts is roughly characterized by (a), an s-shaped (sigmoid) routing of the cardiac flow paths, and (b,) a bilaterally asymmetric (chiral) routing of the cardiac flow paths.
- (2), the fully looped heart tube of vertebrate embryos regularly shows sigmoid as well as chiral routing of its flow path.
- (3), data from physical pump models suggest that the looped design of valveless embryonic heart tubes might improve the pumping efficiency.
- (4), among the mature (chambered) hearts of vertebrates, sigmoid as well as chiral routing of the flow path(s) is regularly found only in the multi-chambered hearts of lungfish and tetrapods. Here, the bilaterally asymmetric (chiral) anatomy is regarded as the main determinant of the alignment and functional or structural separation of the systemic and pulmonary flow paths.
- (5), among the two-chambered heart of fishes, only sigmoid routing of the flow path is a regular feature, while a bilaterally asymmetric (chiral) configuration is not a regular feature. Thus, among fishes we can find species with bilaterally asymmetric hearts as well as species with bilaterally symmetric hearts.
- (6), the presence of bilateral symmetry in the mature hearts of some fishes seems to be the consequence of a process of back-rotation of the heart during the post-embryonic (larval) period of development.
- (7), the presence of a nearly perfect bilateral symmetry in the high performance hearts of tuna, and other active fishes, suggests that chiral routing of the cardiac flow path may not significantly improve the pumping function of two-chambered hearts. Thus, if we assume that the pumping function of two-chambered hearts may profit from a looped design, such a benefit should be attributed to the sigmoid routing of the cardiac flow path, which is a feature found in the two-chambered as well as multi-chambered hearts of vertebrates.
- (8), the bilaterally asymmetric (chiral) anatomy of some fish hearts seems to represent no more than the solution of a packing problem.
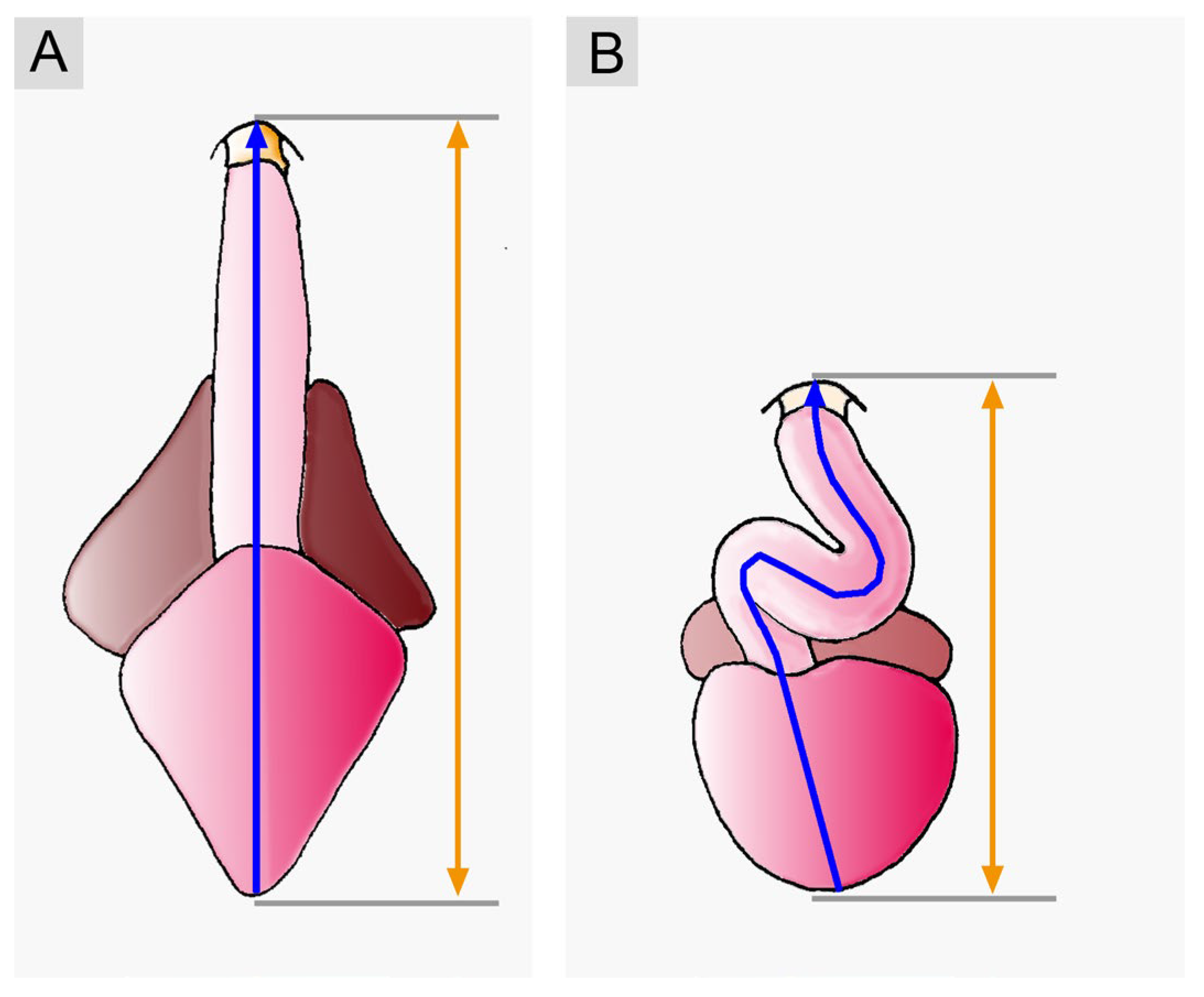
- (9), it was frequently stated that the evolution of the vertebrate heart was characterized by an increase in the degree of sigmoid routing of the cardiac flow path(s). The lowest degree of sigmoid routing was ascribed to the hearts of basal vertebrates (jawless fishes). A moderate degree was ascribed to the hearts of jawed fishes, while high degrees of sigmoid routing of the cardiac flow path(s) were ascribed to the hearts of lungfish and tetrapods. A review of original data from fishes, however, shows that this phylogenetic scenario does not match with the reality. Among jawed fishes, we can find a spectrum of various degrees of sigmoid routing of the cardiac flow path that ranges from low degrees, resulting from an atrial position that is dorso-caudal to the ventricle, to high degrees, resulting from an atrial position that is dorso-cranial to the ventricle. Thus, a high degree of sigmoid routing of the cardiac flow path(s) is not exclusively found in the mature hearts of tetrapods.
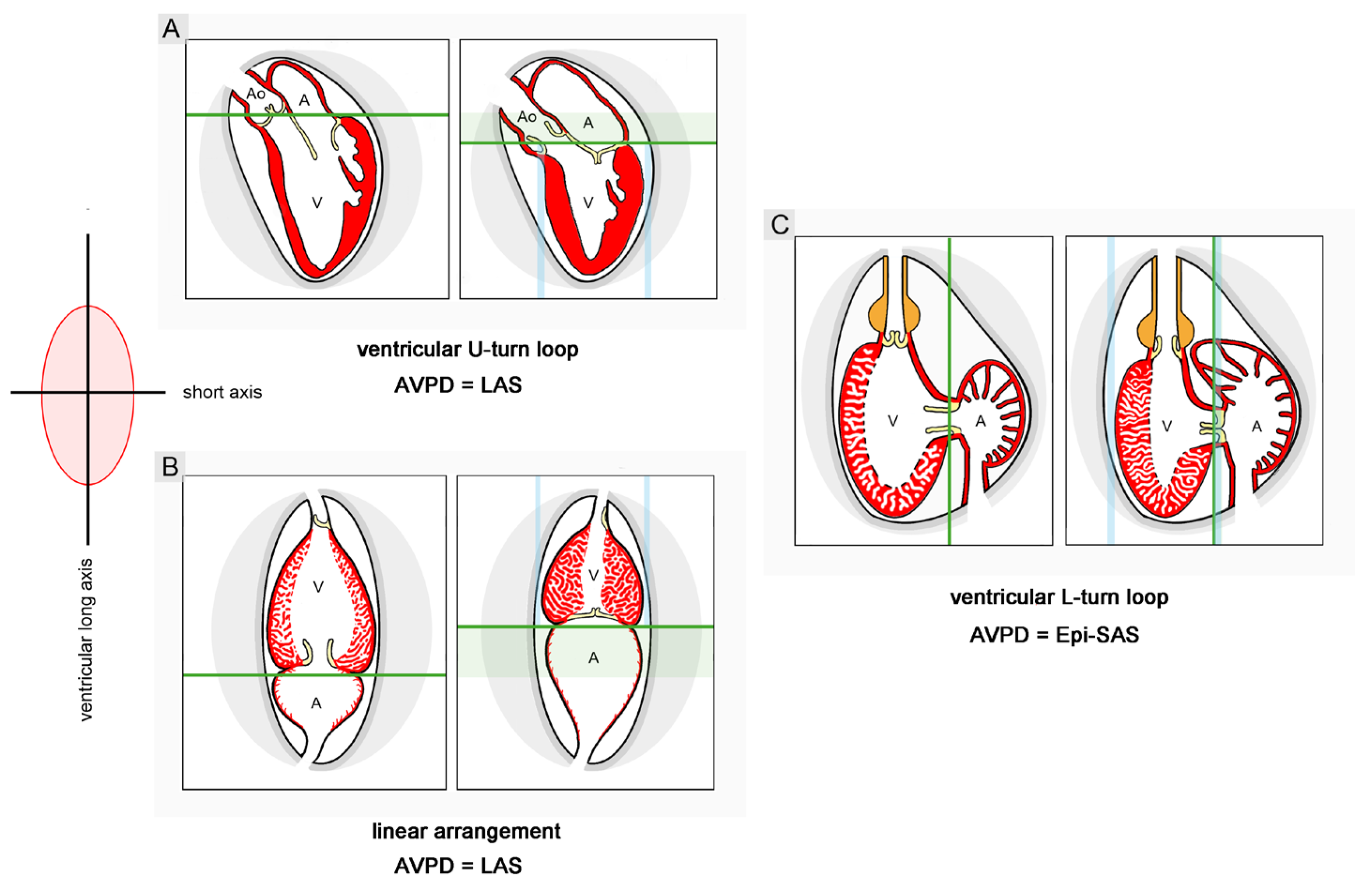
- (10), Kilner an co-authors have postulated that sigmoid routing of the cardiac flow path(s) may produce effects supporting the long-axis displacement of the AVP, while a linear routing of the cardiac flow path(s) may produce effects suppressing the long-axis displacement of the AVP particularly in the exercising state. Data from snail hearts and physical models cast doubt on the validity of this hypothesis. The relation between the looped design and the pumping function of vertebrate hearts remains an enigma.
Funding
Conflicts of Interest
References
- Von Baer, K.E. Untersuchungen über die Entwickelungsgeschichte der Fische nebst einem Anhange über die Schwimmblase. F.C.W. Vogel: Leipzig, Germany, 1835.
- Bischoff, T.L.W. Entwickelungsgeschichte des Säugethier- und Menscheneies. L. Voß: Leipzig, Germany, 1842.
- His, W. Anatomie menschlicher Embryonen III.. F.C.W. Vogel: Leipzig, Germany, 1885.
- Sobotta, J. Über die Entwickelung des Blutes, des Herzens und der grossen Gefässstämme der Salmoniden. Anat Hefte 1902, 19, 579–688. [Google Scholar] [CrossRef]
- Bremer, J.L. Part I. An interpretation of the development of the heart. Part II. The left aorta of reptiles. Am J Anat 1928, 42, 307–369. [Google Scholar] [CrossRef]
- Patten, B.M. The formation of the cardiac loop in the chick. Am J Anat 1922, 30, 373–397. [Google Scholar] [CrossRef]
- Senior, H.D. The development of the heart in shad (Alosa Sapadissima, Wilson). Am J Anat 1909, 9, 211–262. [Google Scholar] [CrossRef]
- Tschermak, A. 1909. Physiologische Untersuchungen am embryonalen Fischherzen. Sitzgsber Akad Wiss Wien, Math-naturwiss Kl 3 1909, 118, 17. [Google Scholar]
- Robertson, J.I. The development of the heart and vascular system of Lepidosiren paradoxa. J Cell Sci 1913, 59, 53–132. [Google Scholar] [CrossRef]
- Tschermak, A. Über das Verhalten des embryonalen Fischherzens gegenüber dem konstanten Strom. Z Exper Med 1929, 68, 452–474. [Google Scholar] [CrossRef]
- Icardo, J.M.; Guerrero, A.; Durán, AC.; Domezain, A.; Colvee, E.; Sana-Coma, V. The development of the sturgeon heart. Anat Embryol 2004, 208, 439–449. [Google Scholar] [CrossRef]
- Rodríguez, C,; Sans-Coma, V. ; Grimes, A.C.; Fernández, B.; Arqué, J.M.; Durán, A.C. Embryonic development of the bulbus arteriosus of the primitive heart of jawed vertebrates. Zool Anz 2013, 252, 359–366. [Google Scholar] [CrossRef]
- Cooke, J. Developmental mechanism and evolutionary origin of vertebrate left/right asymmetries. Biol Rev 2004, 79, 377–407. [Google Scholar] [CrossRef]
- Palmer, A.R. Symmetry breaking and the evolution of development. Science 2004, 306, 828–833. [Google Scholar] [CrossRef] [PubMed]
- Ramsdell, A.F. Left-right asymmetry and congenital cardiac defects: getting to the heart of the matter in vertebrate left-right axis determination. Dev Biol 2005, 288, 1–20. [Google Scholar] [CrossRef]
- Stainier, D.Y.R. , Fishman, M.C. The zebrafish as a model system to study cardiovascular development. Trends Cardiovasc Med 1994, 4, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.A. , Uribe, V. Getting to the heart of left-right asymmetry: contributions from the zebrafish model. J Cardiovasc Dev Dis 2021, 8, 64. [Google Scholar] [CrossRef]
- Lombardo, V.A.; Heise, M.; Moghtadaei, M.; Bornhorst, D.; Männer, J.; Abdelilah-Seyfried, S. Morphogenetic control of zebrafish cardia looping by Bmp signaling. Development 2019, 146, dev180091. [Google Scholar] [CrossRef]
- Tessadori, F.; Tsingos, E.; Colizzi, E.S.; Kruse, F.; van den Brink, S.C.; van den Boogaard, M.; Christoffels, V.M.; Merks, R.M.H.; Bakkers, J. Twisting of the zebrafish heart tube during cardiac looping is a tbx5-dependent and tissue-intrinsic process. Elife 2021, 10, e61733. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, G.C.; Lo, C.W. Left-right patterning in congenital heart disease and beyond. Am J Genet C Semin Med Genet 2020, 184, 90–96. [Google Scholar] [CrossRef]
- Nakano, H.; Fajardo, V.M.; Nakano, A. The role of glucose in physiological and pathological heart formation. Dev Biol 2021, 475, 222–233. [Google Scholar] [CrossRef]
- Rahman, T.; Zhang, H.; Fan, J.; Wan, Q. Cell chirality in cardiovascular development and disease. APL Bioeng 2020, 4, 031503. [Google Scholar] [CrossRef]
- Desgrange, A.; Le Garrec, J.F.; Meilac, S.M. Left-right asymmetry in heart development and disease: forming the right loop. Development 2018, 145, dev162776. [Google Scholar] [CrossRef]
- Kilner PJ, Yang GZ, Wilkes AJ, Mohladdin RH, Firmin DN, Yacoub MH. Asymmetric redirection of flow through the heart. Nature 2000, 404, 759–761. [Google Scholar] [CrossRef]
- Kilner, P.J.; Yang, G.Z.; Firmin, D.N. Morphodynamics of flow through sinuous curvatures of the heart. Biorheology 2002, 39, 409–417. [Google Scholar] [PubMed]
- Amodeo, A.; Olivero, M.; Versacci, P.; Marino, B. Spiral shapes in heart and shells: when form and function do matter. Eur J Cardio-Thorac Surg 2012, 41, 473–475. [Google Scholar] [CrossRef] [PubMed]
- Hiermeier, F.; Männer, J. Kinking and torsion can significantly improve the efficiency of valveless pumping in periodically compressed tubular conduits. Implications for understanding of the form-function relationship of embryonic heart tubes. J Cardiovasc Dev Dis 2017, 4, 19. [Google Scholar] [CrossRef] [PubMed]
- Sherrid, M.V.; Männer, J.; Swistel, D.G.; Olivotto, I.; Halpern, D.G. On the cardiac loop and its failing: left ventricular outflow tract obstruction. J Am Heart Ass 2020, 9, e014857. [Google Scholar] [CrossRef] [PubMed]
- Männer, J. When does the human embryonic heart start beating? A review of contemporary and historical sources of knowledge about the onset of blood circulation in man. J Cardiovasc Dev Dis 2022, 9, 187. [Google Scholar] [CrossRef] [PubMed]
- Simões-Costa, M.S.; Vasconcelos, M.; Sampaio, A.C.; Cravo, R.M.; Linhares, V.L.; Hochgreb, T.; Yan, C.Y.I.; Davidson, B.; Xavier-Neto, J. The evolutionary origin of cardiac chambers. Dev Biol 2005, 277, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Männer, J.; Wessel, A.; Yelbuz, T.M. How does the tubular embryonic heart work? Looking for the physical mechanism driving unidirectional blood flow in the valveless embryonic heart tube. Dev Dyn 2010, 239, 1035–1046. [Google Scholar] [CrossRef]
- Abu Issa, R.; Kirby, M.L. Heart field: from mesoderm to heart tube. Ann Rev Cell Dev Biol 2007, 23, 45–68. [Google Scholar] [CrossRef]
- Meilhac, S.M.; Lescroart, F.; Blanpain, C.; Buckingham, M.E. Cardiac cell lineages that form the heart. Cold Spring Harb Perspect Med 2014, 4, a013888. [Google Scholar] [CrossRef]
- Burggren, W.W. Cardiac design in lower vertebrates: what can phylogeny reveal about ontogeny? Experentia 1988, 44, 919–930. [Google Scholar] [CrossRef] [PubMed]
- Bettex, D.A.; Prêtre, R.; Chassot, P.G. Is our heart a well- designed pump? The heart along evolution. Eur Heart J 2014, 35, 2322–2332. [Google Scholar] [CrossRef] [PubMed]
- Ziermann, J.M.; Freitas, R.; Diogo, R. Muscle development in the shark Scyliorhinus canicula: implications for the evolution of the gnathostome head and paired appendage musculature. Front Zool 2017, 14, 31. [Google Scholar] [CrossRef] [PubMed]
- Männer, J. On the form problem of embryonic heart loops, its geometrical solutions, and a new biophysical concept of cardiac looping. Ann Anat 2013, 195, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Kunz, Y. Morphologische Studien über die embryonale und postembryonale Entwicklung bei Teleostiern mit besonderer Berücksichtigung des Dottersystems und der Leber. Revue Suisse de Zoologie 1964, 71, 445–525. [Google Scholar] [CrossRef]
- Kunz-Ramsay, Y. Developmental biology of Teleost fishes. Springer: New York, U.S.A., 2013.
- Singelman, C.; Holtzman, N.G. Analysis of postembryonic heart development and maturation in the zebrafish, Danio rerio. Dev Dyn 2012, 241, 1993–2004. [Google Scholar] [CrossRef] [PubMed]
- Pernkopf, E.; Wirtinger, W. Die Transposition der Herzostien – ein Versuch der Erklärung dieser Erscheinung. I. Teil. Die Pheronomie der Herzentwicklung. Z Anat Entwickl Gesch 1933, 100, 563–711. [Google Scholar] [CrossRef]
- Männer, J. The anatomy of cardiac looping: a step towards the understanding of the morphogenesis of several forms of congenital cardiac malformations. Clin Anat 2009, 22, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Icardo, J.M.; Colvee, E.; Schorna, S.; Lauriano, E.R.; Fudge, D.S.; Glover, C.N.; Zaccone, G. Morphological analysis of the hagfish heart I: The ventricle, the arterial connection and the ventral aorta. J Morphol 2015, 277, 326–340. [Google Scholar] [CrossRef]
- Icardo, J.M. Heart morphology and anatomy. In The cardiovascular system: morphology, control and function. Fish Physiology Vol. 36 Part A., Gamperl, A.K.; Gillis, T.E., Farrell, A.P., Eds.; Brauner, C.J. Eds.: Academic Press, 2017; pp. 1–54. [Google Scholar]
- Muramatsu, B.; Suzuki, D.G.; Suzuki, M.; Higashiyama, H. Gross anatomy of the Pacific hagfish, Eptatretus burgeri, with special reference to the coelomic viscera. Anat Rec 2024, 307, 155–171. [Google Scholar] [CrossRef]
- Santer, R.M. 1985. Morphology and innervation of the fish heart. Adv Anat Embryol Cell Biol 1954, 89. [Google Scholar]
- Richardson, M.K.; Admiraal, J.; Wright, G.M. Developmental anatomy of lampreys. Biol Rev 2010, 85, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Anthony, J.; Millot, J.; Robineau, D. Le cœur et l’aorte ventrale de Latimeria chalumnae (Poisson coelacanthidé). CR Acad Sci 1965, 261, 223–226. [Google Scholar]
- Millot, J.; Anthony, J.; Robineau, D. Anatomie de Latimeria chalumnae. Apparell digestif – apparel respiratoire – apparel urogenital – glandes endocrines – apparel circulatoire – teguments – écailles – conclusion générales. CNRS 1978, 3, 198. [Google Scholar]
- Gregory, J.A.; Graham, J.B.; Cech, J.J. Jr.; Dalton, N. Michaels, J. Lai, N.C. Pericardial and peritoneal canal relationships to cardiac function in the white sturgeon (Acipenser transmontanus). Comp Biochem Physiol A 2004, 138, 203–213. [Google Scholar] [CrossRef]
- Boas, J.E.V. Über Herz und Arterienbogen bei Ceratodus und Protopterus. Morphol JB 1880, 6, 321–354. [Google Scholar]
- Home, E. XXVII. Additions to an account of the anatomy of Squalus maximus, contained in a former paper; with observations on the structure of the branchial artery. Phil Trans Royal Soc 1813, 103, 227–241. [Google Scholar]
- Liem, K.F. Tetrapod parallelisms and other features in the functional morphology of the blood vascular system of Fluta alba Zuiew (pisces: teleostei). J Morphol 1961, 108, 131–143. [Google Scholar] [CrossRef]
- Lai, N.C.; Graham, J.B.; Dalton, N.; Shabetai, R.; Bhargava, V. Echocardiographic and hemodynamic determinations of the ventricular filling pattern in some teleost fishes. Physiol Zool 1998, 71, 157–167. [Google Scholar] [CrossRef]
- Ludwig, W. Das Rechts-Links-Problem im Tierreich und beim Menschen. Springer: Berlin, Germany, 1932.
- Mercola, M. Embryological basis for cardiac left-right asymmetry. Sem Cell Dev Biol 1999, 10, 109–116. [Google Scholar] [CrossRef]
- Kathiriya IS, Srivastava D. Left-right asymmetry and cardiac looping: implications for cardiac development and congenital heart disease. Am J Med Genet (Semin Med Genet) 2000, 97, 271–279. [Google Scholar] [CrossRef]
- Fishman, M.C.; Chien, K.R. Fashioning the vertebrate heart: earliest embryonic decisions. Development 1999, 124, 2099–2117. [Google Scholar] [CrossRef] [PubMed]
- Blum, M.; Ott, T. Animal left-right asymmetry. Current Biol 2018, 28, R293–R305. [Google Scholar] [CrossRef] [PubMed]
- Soares, K.D.A.; Toledo-Piza, M. Branching patterns of the afferent branchial arteries and their phylogenetic significance in rays (Batoidea). Sci Rep 2021, 11, 23236. [Google Scholar] [CrossRef] [PubMed]
- Van Mierop, L.H.S.; Kutsche, L.M. Comparative anatomy and embryology of the ventricles and arterial pole of the vertebrate heart. In: Nora, J.J.; Takao, A. Eds. Congenital heart disease. Causes and Processes, Futura, New York, U.S.A. 1984; pp.459-479.
- Zaccone, G.; Grimes, A.C.; Farrell, A.P.; Dabrowski, K.; Marino, F. Morphology, innervation and its phylogenetic step in the heart of the longnose gar Lepisosteus Osseus. Acta Zool 2012, 93, 381–389. [Google Scholar] [CrossRef]
- Tota, B. 1999. Heart. Chapter 10 In: Hamlett, W.C. Ed., Sharks, skated, and rays. The biology of elasmobranch fish. Johns Hopkins University Press, Baltimore, U.S.A. 1999; pp. 238-272.
- Icardo, J.M.; Colvee, E.; Cerra, M.C.; Tota, B. Structure of the conus arteriosus of the sturgeon (Acipenser naccarii) heart I: The conus valves and subendocardium. Anat Rec 2002, 267, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Szidon, J.; Lahiri, S.; Lev, M.; Fishman, A.P. Heart and circulation of the African lungfish. Circulation Res 1969, 25, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Hatta, A.P. Über die Entwicklung des Gefäßsystems des Neunauges, Lampetra mitsukurii Hatta. Zool JB (Anat.) 1923, 44, 1–264. [Google Scholar]
- Piavis, G.W. Embryology. In Biology of lampreys. Hardisty, M.W.; Potter, I.C., Ed.; Academic Press: London, 1971; pp. 361–400. [Google Scholar]
- Icardo, J.M. Conus arteriosus of the teleost heart: dismissed, but not missed. Anat Rec 2006, 288A, 900–908. [Google Scholar] [CrossRef]
- Grimes, A.C.; Kirby, M.L. The outflow tract of the heart of fishes: anatomy, genes, and evolution. J Fish Biol 2009, 74, 983–1036. [Google Scholar] [CrossRef]
- Lorenzale, M.; Lopez-Unzu, M.A.; Rodríguez, C.; Fernández, B.; Durán, A.C.; Sans-Coma, V. The anatomical components of the cardiac outflow tract of chondrichthyans and actinopterygians. Biol Rev 2018, 93, 1604–1619. [Google Scholar] [CrossRef]
- Drechsel, J. Myokardauffaserungsversuche am Fisch- und Vogelherzen. Z Anat Entwickl Gesch 1935, 104, 403–423. [Google Scholar] [CrossRef]
- Victor, S.; Nayak, V.M.; Rajasingh, R. Evolution of the ventricles. Tex Heart Inst 1999, 26, 168–175. [Google Scholar]
- Schäfer, K.J. Untersuchungen zur Frage der bilateral asymmetrischen Morphologie des adulten Zebrafischherzens. Doctoral thesis, UMG, Georg-August-University, Goettingen, May 31 2023.
- Sánchez-Quintana, D.; García-Martínez, V.; Climent, V.; Hurlé, J.M. Myocardial fiber and connective tissue architecture in the fish heart ventricle. J Exp Zool 1996, 275, 112–124. [Google Scholar] [CrossRef]
- Ballard, W.W. Stages and rates of normal development in the holostean fish, Amia calva. J Exp Zool 1986, 238, 337–354. [Google Scholar] [CrossRef]
- Clowes, C; Boylan, M.G.S.; Ridge, L.A.; Barnes, E.; Wright, J.A.; Hentges, K.E. The functional diversity of essential genes required for mammalian cardiac development. Genesis 2014, 52, 713–737. [CrossRef]
- Sabin, F.R. Origin and development of the primitive vessels of the chick and of the pig. Contr Embryol Carnegie Inst 1917, 6, 61–124. [Google Scholar]
- Jones, E.A.V. The initiation of blood flow and flow induced events in the early vascular development. Semin Cell Dev Biol 2011, 22, 1028–1035. [Google Scholar] [CrossRef]
- Kalogirou, S.; Malissovas, N.; Moro, E.; Argenton, F.; Stainier, D.Y.R.; Beis, D. Intracardiac flow dynamics regulate atrioventricular valve morphogenesis. Cardiovasc Res 2014, 104, 49–60. [Google Scholar] [CrossRef]
- Berndt, C.; Poschmann, G.; Stühler, K.; Holmgren, A.; Bräutigam, L. Zebrafish heart development is regulated via glutaredoxin 2 dependent migration and survival of neural crest cells. Redox Biol 2014, 2, 673–678. [Google Scholar] [CrossRef]
- Forouhar, A.S.; Liebling, M.; Hickerson, A.; Nasiraei-Moghaddam, A.; Tsai, H.J.; Hove, J.R.; Fraser, S.E.; Dickinson, M.E.; Gharib, M. The embryonic vertebrate heart tube is a dynamic suction pump. Science 2006, 312, 751–753. [Google Scholar] [CrossRef]
- Bisphoric, N.H. Evolution of the heart from bacteria to man. Ann NY Acad Sci 2005, 1047, 13–29. [Google Scholar] [CrossRef]
- Johansen, K. Air-breathing fishes. Scientific American 1968, 219, 102–111. [Google Scholar] [CrossRef]
- Grant, R.P. The morphogenesis of corrected transposition and other anomalies of cardiac polarity. Circulation 1964, 29, 71–83. [Google Scholar] [CrossRef]
- Meredith, M.A.; Hutchins, G.M.; Moore, G.W. Role of the left interventricular sulcus in formation of the interventricular septum and crista supraventricularis in normal human cardiogenesis. Anat Rec 1979, 194, 417–428. [Google Scholar] [CrossRef]
- Männer, J. Cardiac looping in the chick embryo: a morphological review with special reference to terminological and biomechanical aspects of the looping process. Anat Rec 2000, 259, 248–262. [Google Scholar] [CrossRef]
- Moorman, A.F.M.; Christoffels, V.M. Cardiac chamber formation: development, genes, and evolution. Physiol Rev 2003, 83, 1223–1267. [Google Scholar] [CrossRef]
- Lewis, F.T.; Abbott, M.E. Reversed torsion of the human heart. Anat Rec 1915, 9, 103–105. [Google Scholar] [CrossRef]
- Lewis, F.T.; Abbott, M.E. Reversed torsion of the ventricular bend of the embryonic heart in the explanation of certain forms of cardiac anomaly. Bull Int Assoc Med Mus 1916, 6, 111–115. [Google Scholar]
- De la Cruz, M.V.; Anselmi, G.; Cisneros, F.; Reinhold, M.; Portillo, B.; Espino-Vela, J. An embryologic explanation for the corrected transposition of the great vessels: additional description of the main anatomic features of this malformation and its varieties. Am Heart J 1959, 57, 104–117. [Google Scholar] [CrossRef]
- Van Praagh, R.; Van Praagh, S.; Vlad, P.; Keith, J.D. Anatomic types of congenital dextrocardia: diagnostic and embryologic implications. Am J Cardiol 1964, 13, 510–531. [Google Scholar] [CrossRef]
- Anderson, R.H.; Webb, S.; Brown, N.A. Defective lateralisation in children with congenitally malformed hearts. Cardiol Young 1998, 8, 512–531. [Google Scholar] [CrossRef]
- Stainier, D.Y.R.; Lee, R.K.; Fishman, M.C. Cardiovascular development in the zebrafish. I. Myocardial fate map and heart tube formation. Development 1993, 119, 31–40. [Google Scholar] [CrossRef]
- Yang GZ, Merrifield R, Masood S, Kilner PJ. Flow and myocardial interaction: an imaging perspective. Phil Trans R Soc B 2007, 362, 1329–1341. [Google Scholar] [CrossRef]
- Sandblom, E.; Axelsson, M. The venous circulation: a piscine perspective. Comp Biochem Physiol A 2007, 148, 785–801. [Google Scholar] [CrossRef]
- Farrell, A.P. From hagfish to tuna: a perspective on cardiac function in fish. Physiol Zool 1991, 64, 1137–1164. [Google Scholar] [CrossRef]
- Henein, M.Y.; Gibson, D.G. 1999. Normal long axis function. Heart 1999, 81, 111–113. [Google Scholar] [CrossRef]
- Carlsson, M.; Ugander, M.; Mosen, H.; Buhre, T.; Arheden, H. Atrioventricular plane displacement is the major contributor to left ventricular pumping in healthy, athletes, and patients with dilated cardiomyopathy. AJP Heart Circ Physiol 2007, 292, H1452–H1459. [Google Scholar] [CrossRef]
- Carlsson, M.; Ugander, M.; Heiberg, E.; Arheden, H. The quantitative relationship between longitudinal and radial function in left, right, and total heart pumping in humans. AJP Heart Circ Physiol 2007, 293, H636–H644. [Google Scholar] [CrossRef]
- Steding-Ehrenborg, K.; Carlsson, M.; Stephensen, S.; Arheden, H. Atrial aspiration from pulmonary and caval veins is caused by ventricular contraction and secures 70% of the total stroke volume independent of resting heart rate and heart size. Clin Physiol Funct Imaging 2013, 33, 233–240. [Google Scholar] [CrossRef]
- Arutunyan, A.H. Atrioventricular plane displacement is the sole mechanism of atrial and ventricular refill. AJP Heart Circ Physiol 2015, 308, H1317–H1320. [Google Scholar] [CrossRef]
- Watanabe, H.; Sugiura, S.; Hisada, T. The looped heart does not save energy by maintaining the momentum of blood flowing in the ventricle. AJP Heart Circ Physiol 2008, 294, H2191–H2196. [Google Scholar] [CrossRef]
- Kilner, PJ. 2010. Letter to the editor: “Postulated functional advantages of a looped as opposed to a linearly arranged heart”. AJP Heart Circ Physiol 298:H726. [CrossRef]
- Maksuti, E.; Bjällmark, A.; Broomé, M. Modelling the heart with the atrioventricular plane as a piston unit. Medical Engineering Physics 2015, 37, 87–92. [Google Scholar] [CrossRef]
- Jordan, H.J.; Hirsch, G.C. Übungen aus der vergleichenden Physiologie. Springer: Berlin, Germany; 1927.
- Biering, P. Untersuchungen über das Kreislaufsystem bei den Weichtieren II. Z Vergl Physiol 1929, 10, 465–484. [Google Scholar] [CrossRef]
- Schwartzkopff, J. Über die Leistung des isolierten Herzens der Weinbergschnecke (Helix pomatia L.) im künstlichen Kreislauf. Z Vergl Physiol 1954, 36, 543–594. [Google Scholar] [CrossRef]
- Jones, H.D. Circulatory pressures in Helix pomatia. Comp Biochem Physiol A 1971, 39, 289–295. [Google Scholar] [CrossRef]
- von Skramlik, E. Weichtieren. In Ergebnisse der Biologie. Frisch, K.; Ruhland, W., Koehler, O., Stubbe, H., Eds.; Springer: Berlin, Germany, 1941. [Google Scholar]
- Van Aardt, W.J.; Vosloo, A. Modifications to an optocardiographic method for measuring of heart rate in a range of invertebrate species. S Afr J Zool 1996, 32, 97–100. [Google Scholar]
- Nopp, H. Temperaturbezogene Regulationen des Sauerstoffverbrauchs und der Herzschlagrate bei einigen Pulmonaten. Z Vergl Physiol 1965, 50, 641–659. [Google Scholar] [CrossRef]
- Wünnenberg, W. Diurnal rhythms of the heart rate in the snail Helix pomatia L. Comp Biochem Physiol A 1991, 99, 415–417. [Google Scholar] [CrossRef]
- Romero, S.M.; Hoffmann, A. Heart rate and behavioral patterns of Megalobulimus sanctipauli (mollusca, gastropoda, pulmonata). Braz J Med Biol Res 1991, 24, 223–227. [Google Scholar]
- Renwrantz, L.; Spielvogel, F. Heart rate and hemocyte number as stress indicators in disturbed hibernating vineyard snails, Helix pomatia. Comp Biochem Physiol Part A. [CrossRef]
- Noll, A. Untersuchungen über das Kreislaufsystem bei den Weichtieren III. Z Vergl Physiol 1929, 10, 761–777. [Google Scholar] [CrossRef]
- Ugander, M.; Carlsson, M.; Arheden, H. Short axis epicardial volume change is a measure of cardiac left ventricular short axis function, which is independent of myocardial wall thickess. AJP Heart Circ Physiol 2010, 298, H530–H535. [Google Scholar] [CrossRef] [PubMed]
- Ishimatsu, A.; Itazawa, Y. Differences in blood oxygen levels in the outflow vessels of the heart of an air-breathing fish, Channa argus: do separate blood streams exist in a teleostean heart? J Comp Physiol 1983, 149, 435–440. [Google Scholar] [CrossRef]
- Munshi, J.S.D.; Olson, K.R.; Roy, P.K.; Ghosh, U. Scanning electron microscopy of the heart of the climbing perch. J Fish Biol 2001, 59, 1170–1180. [Google Scholar] [CrossRef]
- Park, M.H.; Imbrie-Moore, A.M.; Zhu, Y.; Wilkerson, R.J.; Wang, H.; Park, G.H.; Wu, C.A.; Pandya, P.K.; Mullis, D.M.; Marin-Cuartas, M.; Woo, Y.J. The critical biomechanics of aortomitral angle and systolic anterior motion: engineering native ex vivo simulation. Ann Biomed Engin 2023, 51, 794–805. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



