Submitted:
05 July 2024
Posted:
08 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Seed Preparation and Growth Conditions
2.2. Biometric Parameters
2.3. Photosynthetic Parameters
2.4. Oxidative Stress Parameters
2.5. Statistical Analysis
3. Results
3.1. The Impact of SBC on Plant Growth
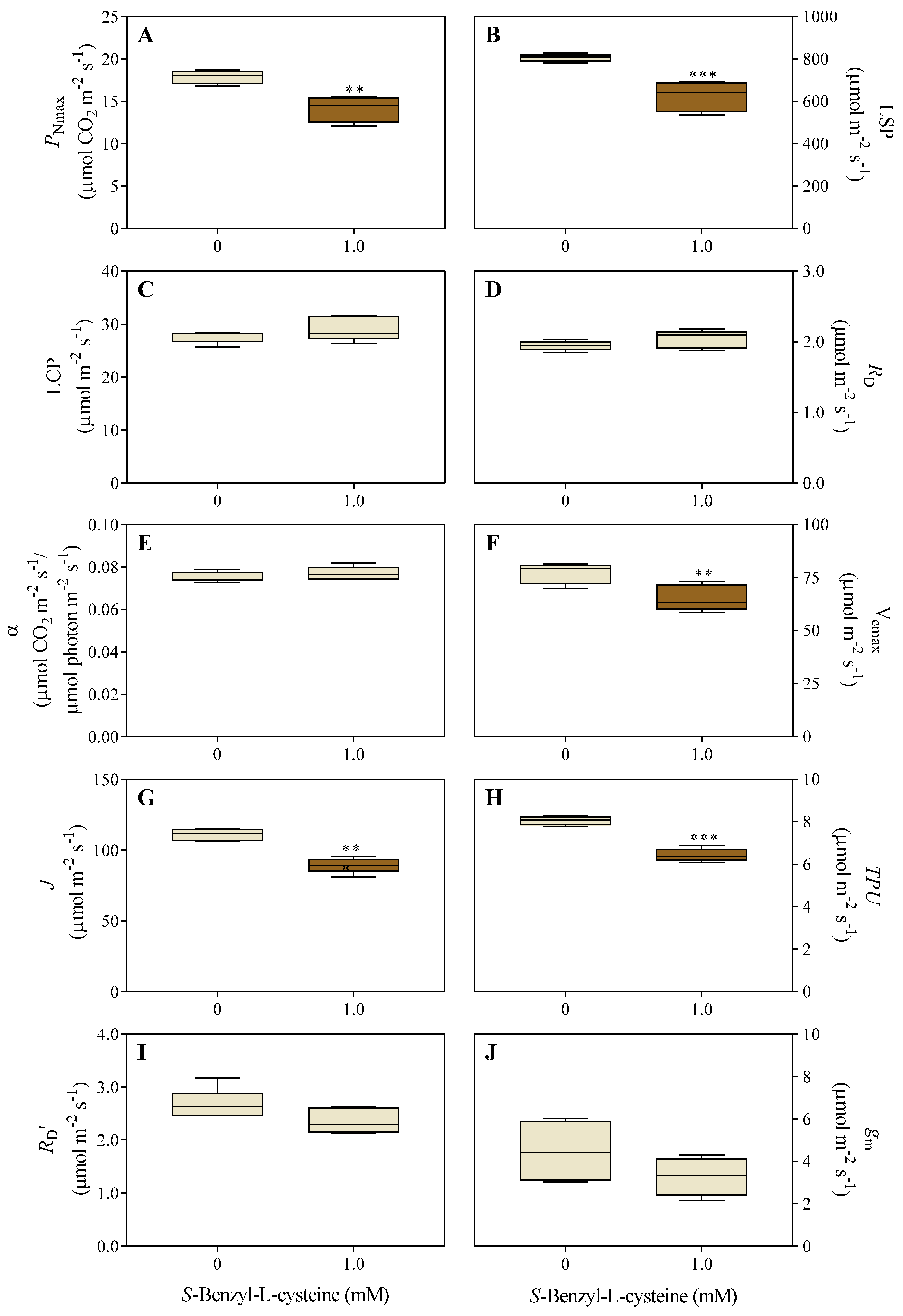
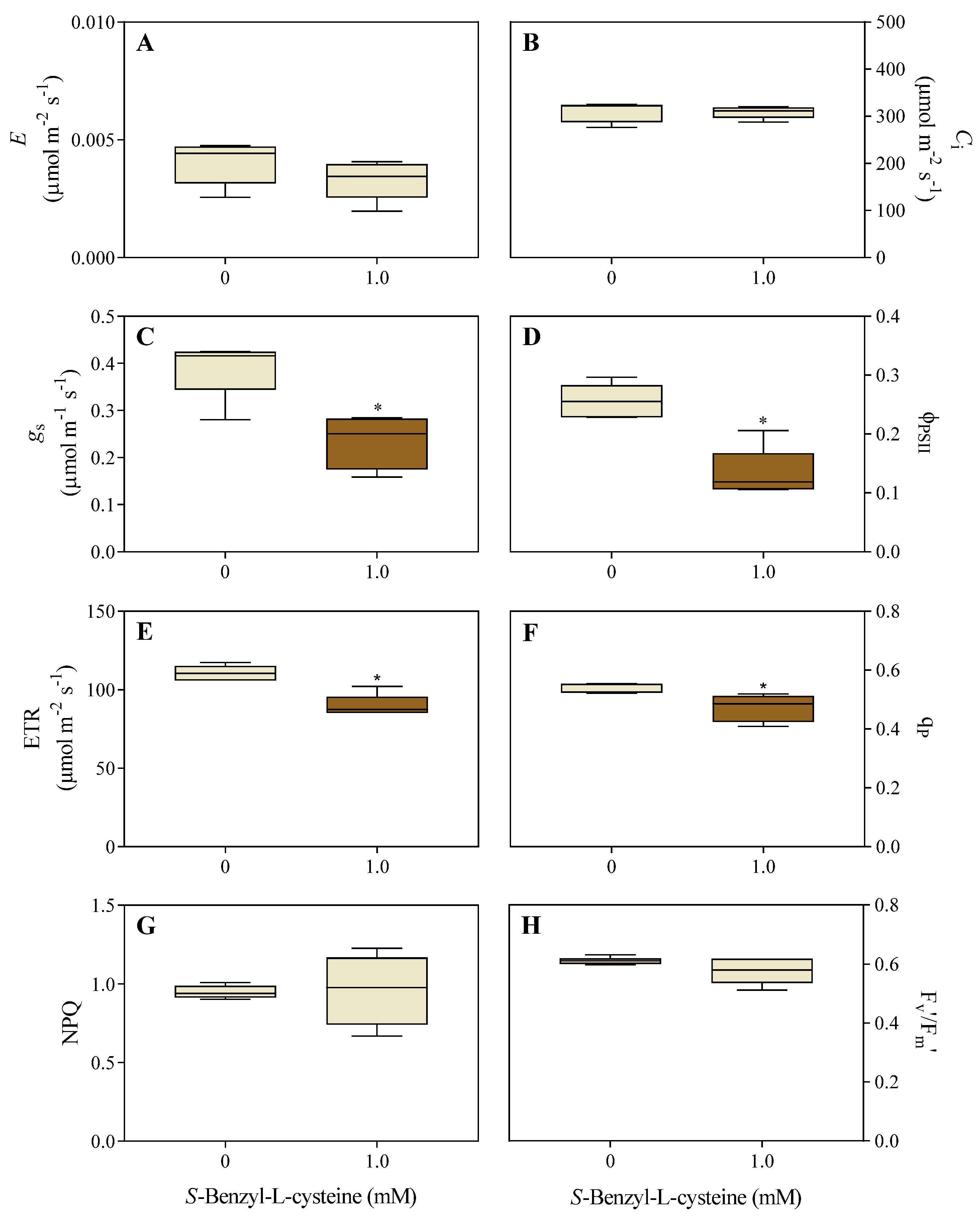
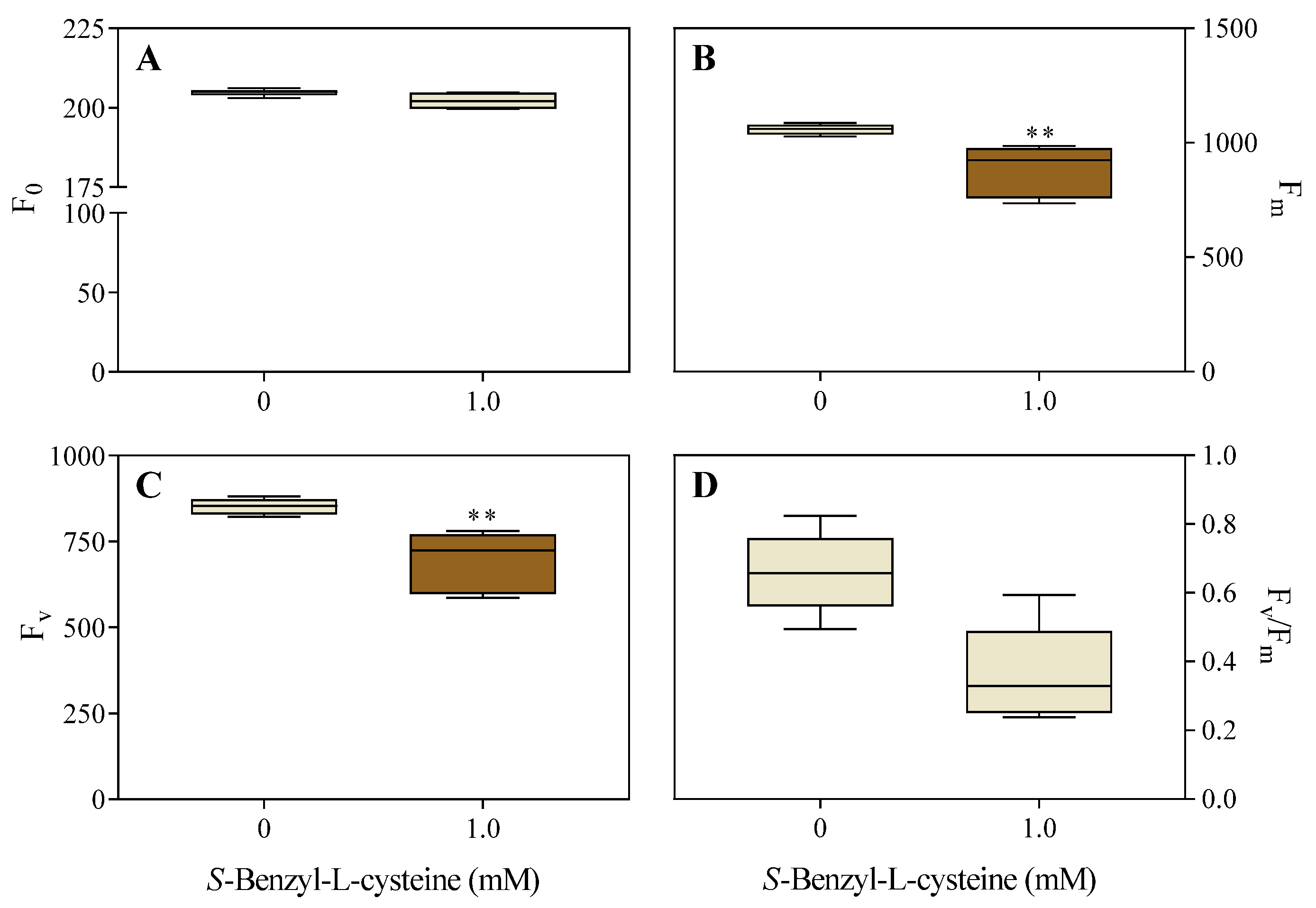
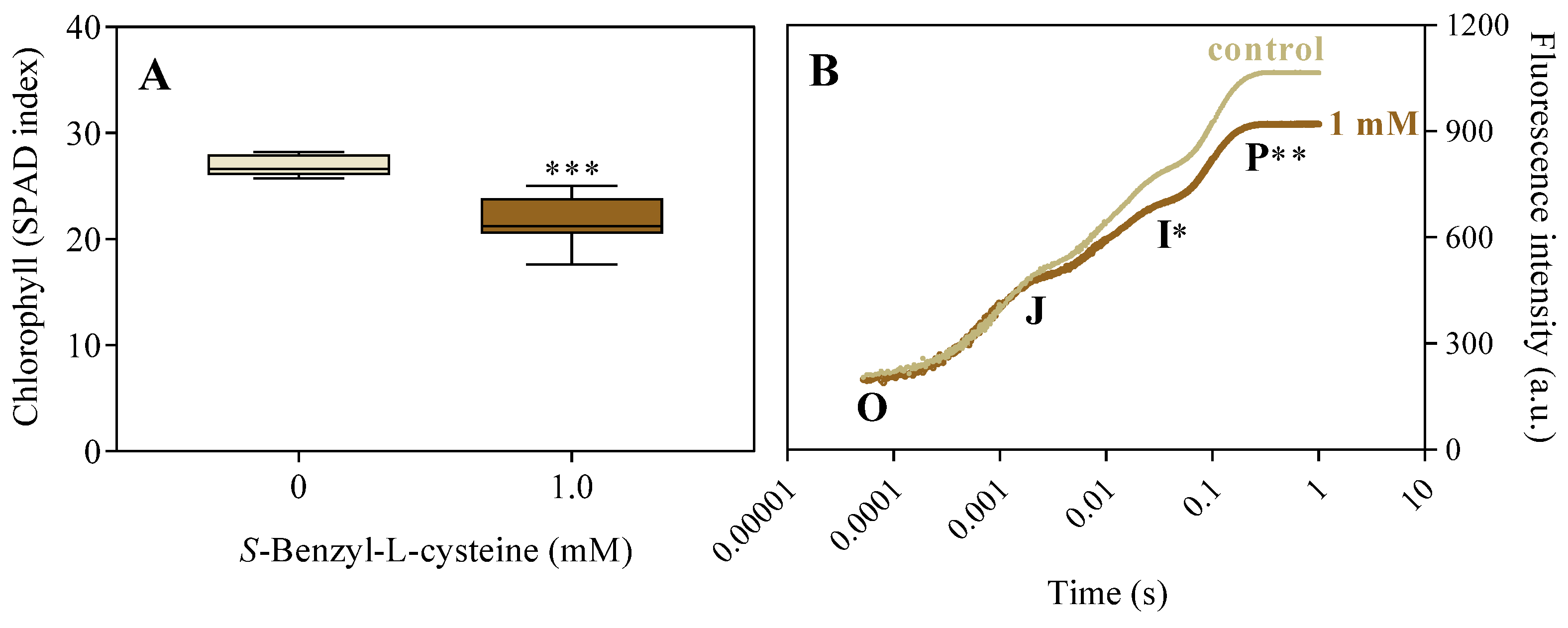
3.2. The Impact of SBC on Photosynthesis
3.3. The Impact of SBC and Oxidative Stress
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marschner, P. Marschner’s Mineral Nutrition of Higher Plants; Marschner, P., Ed.; Third edit.; Elsevier, 2012; ISBN 9780123849052.
- Li, Q.; Gao, Y.; Yang, A. Sulfur Homeostasis in Plants. Int. J. Mol. Sci. 2020, 21, 1–16. [CrossRef]
- Jez, J.M.; Ravilious, G.E.; Herrmann, J. Structural Biology and Regulation of the Plant Sulfation Pathway. Chem. Biol. Interact. 2016, 259, 31–38. [CrossRef]
- Huang, X.Y.; Li, M.; Luo, R.; Zhao, F.J.; Salt, D.E. Epigenetic Regulation of Sulfur Homeostasis in Plants. J. Exp. Bot. 2019, 70, 4171–4182. [CrossRef]
- Koprivova, A.; Kopriva, S. Sulfation Pathways in Plants. Chem. Biol. Interact. 2016, 259, 23–30. [CrossRef]
- Kopriva, S.; Malagoli, M.; Takahashi, H. Sulfur Nutrition: Impacts on Plant Development, Metabolism, and Stress Responses. J. Exp. Bot. 2019, 70, 4069–4073. [CrossRef]
- Jez, J.M. Structural Biology of Plant Sulfur Metabolism: From Sulfate to Glutathione. J. Exp. Bot. 2019, 70, 4089–4103. [CrossRef]
- Jez, J.M.; Dey, S. The Cysteine Regulatory Complex from Plants and Microbes: What Was Old Is New Again. Curr. Opin. Struct. Biol. 2013, 23, 302–310. [CrossRef]
- Heap, I. International Herbicide-Resistant Weed Database Available online: https://www.weedscience.org/Home.aspx (accessed on 1 January 2024).
- Warrilow, A.G.S.; Hawkesford, M.J. Modulation of Cyanoalanine Synthase and O-Acetylserine (Thiol) Lyases A and B Activity by β-Substituted Alanyl and Anion Inhibitors. J. Exp. Bot. 2002, 53, 439–445.
- Foletto-Felipe, M. de P. O-Acetilserina(Tiol) Liase: Estudos in Silico, in Vitro e in Vivo, Universidade Estadual de Maringá, 2021.
- Pagnoncelli, F. de B.; Trezzi, M.M.; Brum, B.; Vidal, R.A.; Portes, Á.F.; Scalcon, E.L.; Machado, A. Morning Glory Species Interference on the Development and Yield of Soybeans. Bragantia 2017, 76, 470–479. [CrossRef]
- Pazuch, D.; Trezzi, M.M.; Guimarães, A.C.D.; Barancelli, M.V.J.; Pasini, R.; Vidal, R.A. Evolução Da Tolerância Ao Glyphosate Em Populações de Corda-de-Viola. Planta Daninha 2017, 35. [CrossRef]
- Chan, K.X.; Wirtz, M.; Phua, S.Y.; Estavillo, G.M.; Pogson, B.J. Balancing Metabolites in Drought: The Sulfur Assimilation Conundrum. Trends Plant Sci. 2013, 18, 18–29. [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The Water Culture Method for Growing Plants without Soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 32.
- Lobo, F. de A.; de Barros, M.P.; Dalmagro, H.J.; Dalmolin, Â.C.; Pereira, W.E.; de Souza, É.C.; Vourlitis, G.L.; Rodríguez Ortíz, C.E. Fitting Net Photosynthetic Light-Response Curves with Microsoft Excel - a Critical Look at the Models. Photosynthetica 2013, 51, 445–456. [CrossRef]
- Ye, Z.P. A New Model for Relationship between Irradiance and the Rate of Photosynthesis in Oryza Sativa. Photosynthetica 2007, 45, 637–640. [CrossRef]
- Sharkey, T.D.; Bernacchi, C.J.; Farquhar, G.D.; Singsaas, E.L. Fitting Photosynthetic Carbon Dioxide Response Curves for C3 Leaves. Plant, Cell Environ. 2007, 30, 1035–1040. [CrossRef]
- Marchiosi, R.; de Souza Bido, G.; Böhm, P.A.F.; Soares, A.R.; da Silva, H.A.; Ferro, A.P.; de Lourdes Lucio Ferrarese, M.; Ferrarese-Filho, O. Photosynthetic Response of Soybean to L-DOPA and Aqueous Extracts of Velvet Bean. Plant Growth Regul. 2016, 80, 171–182. [CrossRef]
- Stirbet, A.; Lazár, D.; Kromdijk, J. Chlorophyll a Fluorescence Induction: Can Just a One-Second Measurement Be Used to Quantify Abiotic Stress Responses? Photosynthetica 2018, 56, 86–104. [CrossRef]
- Jambunathan, N. Determination and Detection of Reactive Oxygen Species (ROS), Lipid Peroxidation, and Electrolyte Leakage in Plants. In Plant stress tolerance: Methods in molecular biology; Sunkar, R., Ed.; Springer, 2010; pp. 291–297 ISBN 978-1-60761-701-3.
- Heath, R.L.; Packer, L. Photoperoxidation in Isolated Chloroplast. I. Kinetics and Stoichiometry of Fatty Acids Peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198.
- Buege, J.A.; Aust, S.D. Biomembranes - Part C: Biological Oxidations. Methods Enzymol. 1978, 52, 302–310.
- Azevedo, R.A.; Alas, R.M.; Smith, R.J.; Lea, P.J. Response of Antioxidant Enzymes to Transfer from Elevated Carbon Dioxide to Air and Ozone Fumigation, in the Leaves and Roots of Wild-Type and a Catalase-Deficient Mutant of Barley. Physiol. Plant. 1998, 104, 280–292. [CrossRef]
- Giannopolitis, C.N.; Ries, S.K. Superoxide Dismutases Occurrence in Higher Plants. Plant Physiol. 1977, 59, 309–314. [CrossRef]
- Tománková, K.; Luhová, L.; Petřivalský, M.; Peč, P.; Lebeda, A. Biochemical Aspects of Reactive Oxygen Species Formation in the Interaction between Lycopersicon Spp. and Oidium neolycopersici. Physiol. Mol. Plant Pathol. 2006, 68, 22–32. [CrossRef]
- Dos Santos, W.D.; Ferrarese, M.L.L.; Nakamura, C. V.; Mourão, K.S.M.; Mangolin, C.A.; Ferrarese-Filho, O. Soybean (Glycine max) Root Lignification Induced by Ferulic Acid. The Possible Mode of Action. J. Chem. Ecol. 2008, 34, 1230–1241. [CrossRef]
- Foletto-Felipe, M. de P.; Abrahão, J.; Siqueira-Soares, R. de C.; Contesoto, I. de C.; Grizza, L.H.E.; de Almeida, G.H.G.; Constantin, R.P.; Philippsen, G.S.; Seixas, F.A.V.; Bueno, P.S.A.; et al. Inhibition of O-Acetylserine (Thiol) Lyase as a Promising New Mechanism of Action for Herbicides. Plant Physiol. Biochem. 2023, 204, 108127. [CrossRef]
- Lunde, C.; Zygadlo, A.; Simonsen, H.T.; Nielsen, P.L.; Blennow, A.; Haldrup, A. Sulfur Starvation in Rice: The Effect on Photosynthesis, Carbohydrate Metabolism, and Oxidative Stress Protective Pathways. Physiol. Plant. 2008, 134, 508–521. [CrossRef]
- Bucher, S.F.; Bernhardt–Römermann, M.; Römermann, C. Chlorophyll Fluorescence and Gas Exchange Measurements in Field Research: An Ecological Case Study. Photosynthetica 2018, 56, 1161–1170. [CrossRef]
- Da˛Browski, P.; Baczewska-Dąbrowska, A.H.; Kalaji, H.M.; Goltsev, V.; Paunov, M.; Rapacz, M.; Wójcik-Jagła, M.; Pawluśkiewicz, B.; Bąba, W.; Brestic, M. Exploration of Chlorophyll a Fluorescence and Plant Gas Exchange Parameters as Indicators of Drought Tolerance in Perennial Ryegrass. Sensors (Switzerland) 2019, 19, 2736. [CrossRef]
- Parizotto, A. V.; Marchiosi, R.; Bubna, G.A.; Bevilaqua, J.M.; Ferro, A.P.; Ferrarese, M.L.L.; Ferrarese-Filho, O. Benzoxazolin-2-(3H)-One Reduces Photosynthetic Activity and Chlorophyll Fluorescence in Soybean. Photosynthetica 2017, 55, 386–390. [CrossRef]
- Zhou, Y.H.; Yu, J.Q. Allelochemicals and Photosynthesis. In Allelopathy: A Physiological Process with Ecological Implications; 2006; pp. 127–139 ISBN 9781402042805.
- Bortolo, T. da S.C.; Marchiosi, R.; Viganó, J.; Siqueira-Soares, R. de C.; Ferro, A.P.; Barreto, G.E.; Bido, G. de S.; Abrahão, J.; dos Santos, W.D.; Ferrarese-Filho, O. Trans-Aconitic Acid Inhibits the Growth and Photosynthesis of Glycine max. Plant Physiol. Biochem. 2018, 132, 490–496. [CrossRef]
- Everard, J.D.; Cucci, R.; Kann, S.C.; Flore, J.A.; Loescher, W.H. Gas Exchange and Carbon Partitioning in the Leaves of Celery (Apium Graveolens L.) at Various Levels of Root Zone Salinity. Plant Physiol. 1994, 106, 281–292. [CrossRef]
- Murchie, E.H.; Lawson, T. Chlorophyll Fluorescence Analysis: A Guide to Good Practice and Understanding Some New Applications. J. Exp. Bot. 2013, 64, 3983–3998. [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll Fluorescence - A Practical Guide. J. Exp. Bot. 2000, 51, 659–668. [CrossRef]
- Hussain, M.I.; Reigosa, M.J. Allelochemical Stress Inhibits Growth, Leaf Water Relations, PSII Photochemistry, Non-Photochemical Fluorescence Quenching, and Heat Energy Dissipation in Three C 3 Perennial Species. J. Exp. Bot. 2011, 62, 4533–4545. [CrossRef]
- Lazár, D. The Polyphasic Chlorophyll a Fluorescence Rise Measured under High Intensity of Exciting Light. Funct. Plant Biol. 2006, 33, 9–30. [CrossRef]
- Farquhar, G.D.; Caemmerer, S.; Berry, J.A. A Biochemical Model of Photosynthetic CO2 Assimilation in Leaves of C3 Species. Planta 1980, 149, 78-90–90.
- Sharkey, T.D. Photosynthesis in Intact Leaves of C3 Plants: Physics, Physiology and Rat Limitation. Bot. Rev. 1985, 5, 53–105.
- Sharkey, T.D. What Gas Exchange Data Can Tell Us about Photosynthesis. Plant Cell Environ. 2016, 39, 1161–1163. [CrossRef]
- Long, S.P.; Bernacchi, C.J. Gas Exchange Measurements, What Can They Tell Us about the Underlying Limitations to Photosynthesis? Procedures and Sources of Error. J. Exp. Bot. 2003, 54, 2393–2401. [CrossRef]
- Ashraf, M.; Harris, P.J.C. Photosynthesis under Stressful Environments: An Overview. Photosynthetica 2013, 51, 163–190. [CrossRef]
- Octobre, G.; Delprat, N.; Doumèche, B.; Leca-Bouvier, B. Herbicide Detection: A Review of Enzyme- and Cell-Based Biosensors. Environ. Res. 2024, 249, 118330. [CrossRef]
- Fuerst, E.P.; Norman, M.A. Interactions of Herbicides with Photosynthetic Electron Transport. Weed Sci. 1991, 39, 458–464. [CrossRef]
- Krieger-Liszkay, A. Singlet Oxygen Production in Photosynthesis. J. Exp. Bot. 2005, 56, 337–346. [CrossRef]
- Das, K.; Roychoudhury, A. Reactive Oxygen Species (ROS) and Response of Antioxidants as ROS-Scavengers during Environmental Stress in Plants. Front. Environ. Sci. 2014, 2, 1–13. [CrossRef]
- Apel, K.; Hirt, H. Reactive Oxygen Species: Metabolism, Oxidative Stress, and Signal Transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [CrossRef]
- Sher, A.; Mudassir Maqbool, M.; Iqbal, J.; Nadeem, M.; Faiz, S.; Noor, H.; Hamid, Y.; Yuan, X.; Pingyi, G. The Growth, Physiological and Biochemical Response of Foxtail Millet to Atrazine Herbicide. Saudi J. Biol. Sci. 2021, 28, 6471–6479. [CrossRef]
- Jiang, L.; Yang, H. Prometryne-Induced Oxidative Stress and Impact on Antioxidant Enzymes in Wheat. Ecotoxicol. Environ. Saf. 2009, 72, 1687–1693. [CrossRef]

| SBC (mM) | Root | Stem | Leaf | |
|---|---|---|---|---|
| Length (cm) | 0 | 21.22 ± 0.97 | 6.02 ± 0.67 | 5.60 ± 0.16 |
| 1.0 | 14.87 ± 1.10* | 3.42 ± 0.21* | 4.02 ± 0.19* | |
| Fresh mass (g) | 0 | 0.36 ± 0.02 | 0.30 ± 0.02 | 0.79 ± 0.05 |
| 1.0 | 0.16 ± 0.02* | 0.12 ± 0.01* | 0.34 ± 0.03* | |
| Dry mass (g) | 0 | 0.02 ± 0.001 | 0.02 ± 0.002 | 0.10 ± 0.007 |
| 1.0 | 0.01 ± 0.001* | 0.01 ± 0.001* | 0.05 ± 0.004* |
| SBC (mM) | Root | Leaf | |
|---|---|---|---|
| Reactive oxygen species | 0 | 0.40 ± 0.068 | 4.40 ± 0.543 |
| (fluorescence μg−1) | 1.0 | 0.62 ± 0.035* | 6.61 ± 0.623* |
| Conjugated dienes | 0 | 1.79 ± 0.042 | 5.24 ± 0.414 |
| (μmol g−1) | 1.0 | 2,89 ± 0.163* | 9.27 ± 0.639* |
| Malondialdehyde | 0 | 1.90 ± 0.067 | 19.7 ± 2.041 |
| (nmol g−1) | 1.0 | 2.48 ± 0.055* | 21.9 ± 1.707 |
| Superoxide dismutase | 0 | 0.01 ± 0.001 | 0.06 ± 0.004 |
| (U mg−1) | 1.0 | 0.01 ± 0.002 | 0.05 ± 0.003 |
| Catalase | 0 | 0.02 ± 0.004 | 0.13 ± 0.006 |
| (μmol min−1 mg−1) | 1.0 | 0.03 ± 0.004 | 0.14 ± 0.006 |
| Peroxidase | 0 | 0.10 ± 0.011 | 0.12 ± 0.018 |
| (μmol min−1 g−1) | 1.0 | 0.13 ± 0.003* | 0.16 ± 0.027 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




