Submitted:
06 July 2024
Posted:
08 July 2024
Read the latest preprint version here
Abstract
Keywords:
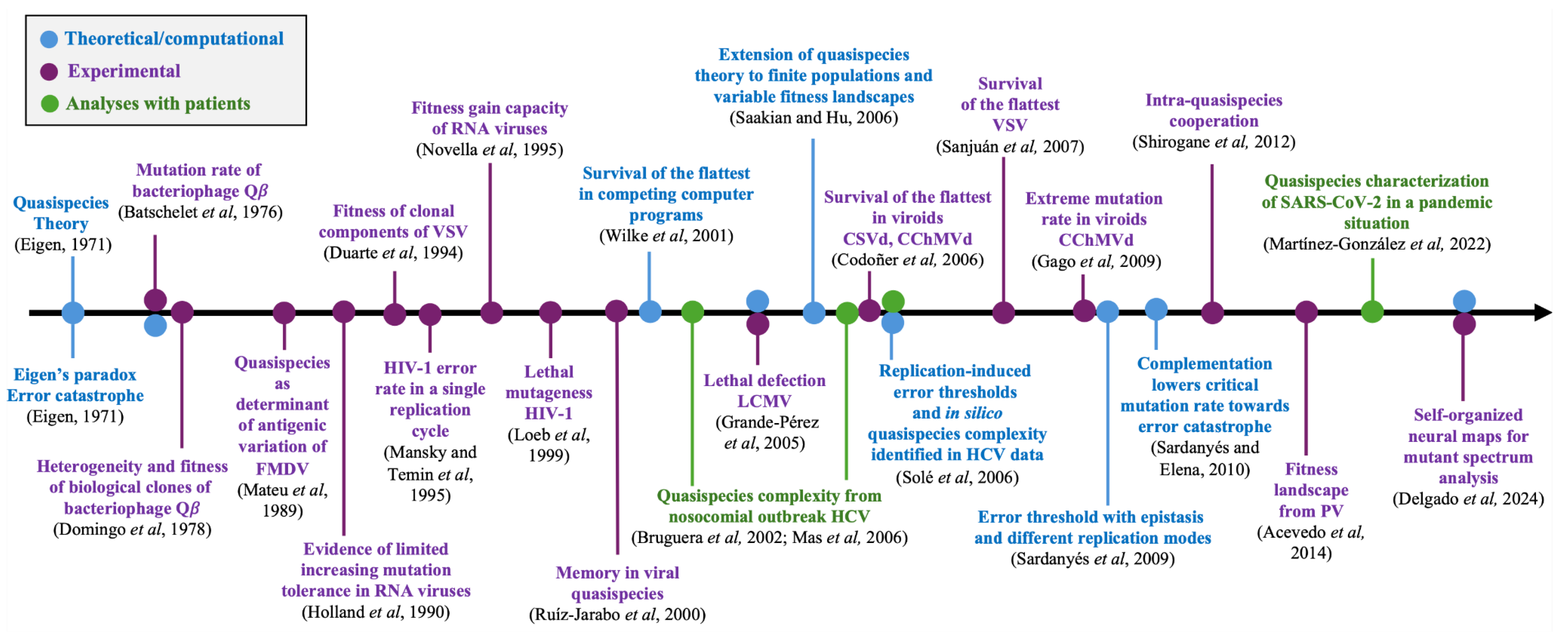
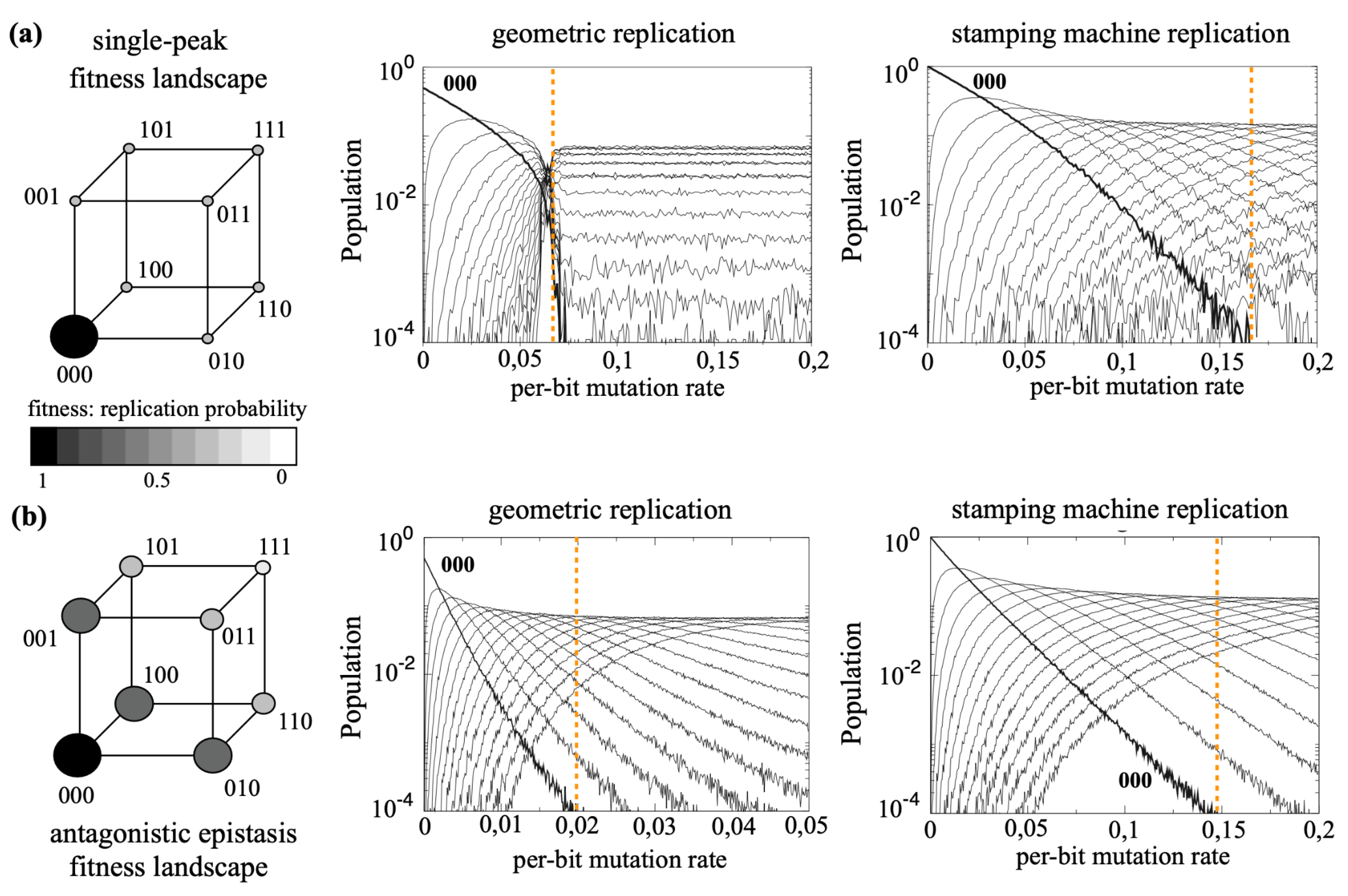
Theoretical Quasispecies: What Has Been Explored and Existing gaps
Quasispecies and RNA Viruses: Genomic Heterogeneity, Adaptation and Clinical Implications of Genetic Information Thresholds
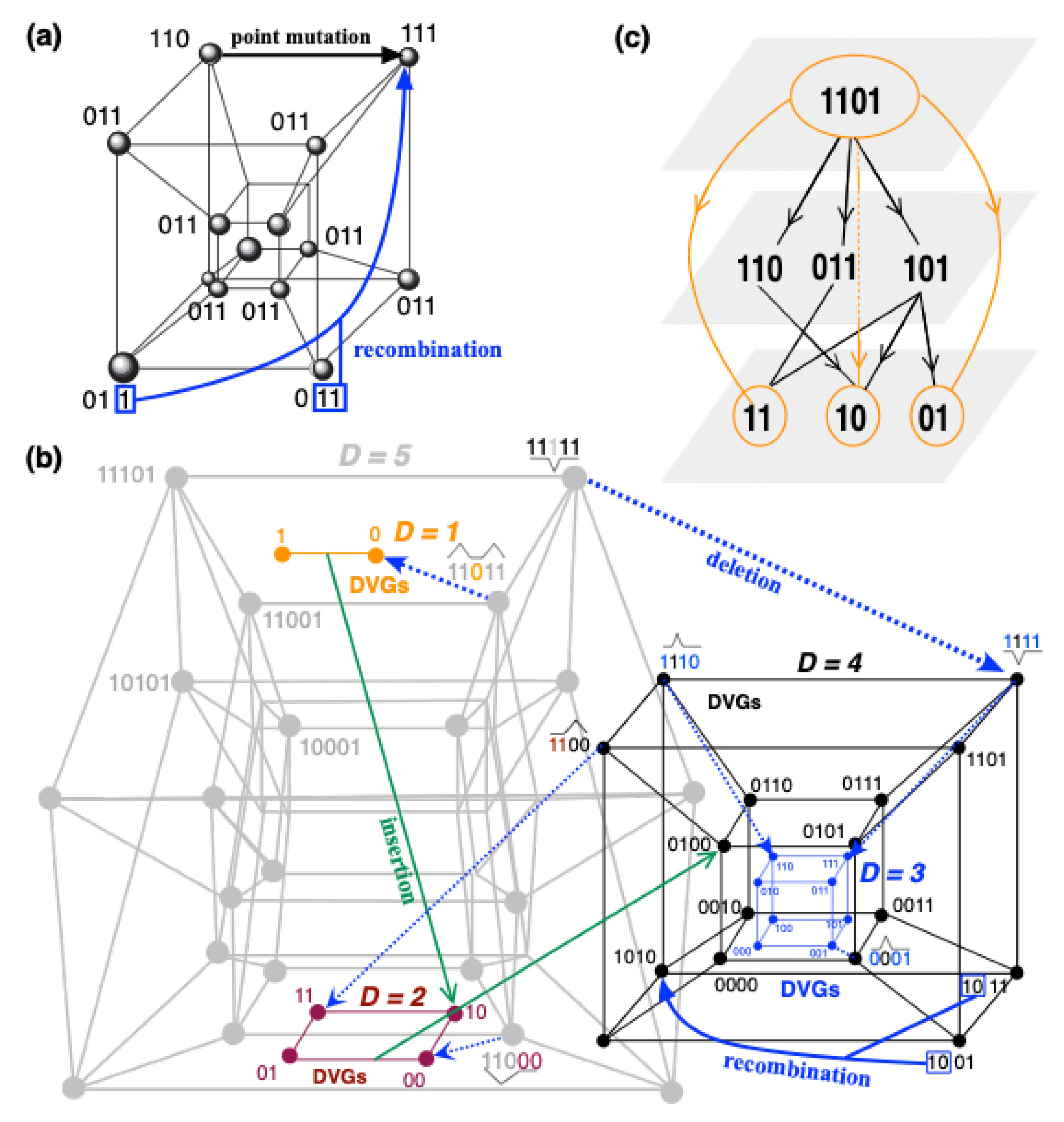
Quasispecies Complexity: From the Hypercube to the Ultracube
Multilayer Models for Multi-Scale Virus Dynamics: Integrating Quasispecies into Virus Epidemiology and Ecology
Impact of Quasispecies Populations in Emerging Viral Diseases
Future Challenges in Experimental and Clinical Quasispecies
Author Contributions
Funding
Acknowledgements
References
- Acevedo, A.; Brodsky, L.; Andino, R. Mutational and fitness landscapes of an RNA virus revealed through population sequencing. Nature 2014, 505, 686. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, J.; Manrubia, S. Effect of spatial competition on the diversity of a quasispecies. Phys. Rev. Lett. 2008, 100, 038106. [Google Scholar] [CrossRef] [PubMed]
- Altemeyer, S.; McCaskill, J. Error threshold for spatially resolved evolution in the quasispecies model. Phys. Rev. Lett. 2001, 86, 5819. [Google Scholar] [CrossRef] [PubMed]
- Batschelet, E.; Domingo, E.; Weissmann, C. The proportion of revertant and mutant phage in a growing population, as a function of mutation and growth rate. Gene 1976, 1, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Ben-Ari, I.; Schinazi, R. A stochastic model for the evolution of a quasispecies. J. Stat. Phys. 2016, 162, 415–425. [Google Scholar] [CrossRef]
- Biebricher, C.; Eigen, M. The error threshold. Virus. Res. 2005, 107, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Biebricher, C.; Eigen, M. What is a quasispecies? Curr. Top. Microbiol. Immunol. 2005, 299, 1–31. [Google Scholar]
- Boccaletti, S.; Bianconi, G.; Criado, R.; del Genio, C.I.; et al. The structure and dynamics of multilayer networks. Phys. Rep. 2014, 544, 1–122. [Google Scholar] [CrossRef] [PubMed]
- Brumera, Y.; Michor, F.; Shakhnovich, E.I. Genetic instability and the quasispecies model. J. Theor. Biol. 2006, 241, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Bull, J.; Sanjuán, R.; Wilke, C.O. (2008). Lethal mutagenesis. In E. Domingo, C.R. Parrish, & J. J. Holland (Eds.), Origin and Evolution of Viruses (pp. 207-218). Elsevier Science.
- Bull, J.J.; Meyers, L.A.; Lachmann, M. Quasispecies made simple. PLoS Comp. Biol. 2005, 1, e61. [Google Scholar] [CrossRef]
- Campagnola, G.; Govindarajan, V.; Pelletier, A.; Canard, B.; et al. The SARS-CoV nsp12 polymerase active site is tuned for large-genome replication. J. Virol. 2022, 96, e0067122. [Google Scholar] [CrossRef] [PubMed]
- Campos, C.; Colomer-Castell, S.; Garcia-Cehic, D.; Gregori, J.; et al. The frequency of defective genomes in Omicron differs from that of the Alpha, Beta and Delta variants. Sci. Rep. 2022, 12, 22571. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, P.; de la Iglesia, F.; Elena, S.F. Distribution of fitness and virulence effects caused by single-nucleotide substitutions in Tobacco etch virus. J. Virol. 2007, 81, 979–984. [Google Scholar] [CrossRef]
- Cellai, D.; López, E.; Zhou, J.; Gleeson, J.P. Percolation in multiplex networks with overlap. Phys. Rev. E 2013, 88, 052811. [Google Scholar] [CrossRef]
- Chao, L.; Rang, C.U.; Wong, L.E. Distribution of spontaneous mutants and inferences about the replication mode of the RNA bacteriophage φ6. J. Virol. 2002, 76, 3276–3281. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Beutler, N.; Basen, G.; Pablo, M.; et al. A single-administration therapeutic interfering particle reduces SARS-CoV-2 viral shedding and pathogenesis in hams. Proc. Natl. Acad. Sci. USA 2022, 119, e2204624119. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Vasen, G.; Pablo, M.; Chen, X.; et al. Identification of a therapeutic interfering particle—A single-dose SARS-CoV-2 antiviral intervention with a high barrier to resistance. Cell 2021, 184, 6022–6036. [Google Scholar] [CrossRef]
- Codoñer, F.M.; Daròs, J.A.; Solé, R.V.; Elena, S.F. The fittest versus the flattest: experimental confirmation of the quasispecies effect with subviral pathogens. PLoS Pathog. 2006, 2, e136. [Google Scholar] [CrossRef] [PubMed]
- Crotty, S.; Cameron, C.; Andino, R. RNA virus error catastrophe: direct molecular test by using ribavirin. Proc. Natl. Acad. Sci. USA 2001, 98, 6895–6900. [Google Scholar] [CrossRef]
- Dapp, M.J.; Patterson, S.E.; Mansky, L.M. Back to the future: revisiting HIV-1 lethal mutagenesis. Trends Microbiol. 2013, 21, 56–62. [Google Scholar] [CrossRef]
- de Ávila, A.; Gallego, I.; Sori, M.E.; Gregori, J.; et al. Lethal mutagenesis of hepatitis C virus induced by favipiravir. PLoS ONE 2016, 11, e0164691. [Google Scholar] [CrossRef]
- de Ávila, A.; Moreno, E.; Perales, C.; Domingo, E. Favipiravir can evoke lethal mutagenesis and extinction of foot-and-mouth disease virus. Virus Res. 2017, 233, 105–112. [Google Scholar] [CrossRef]
- de la Peña, M.; Elena, S.F.; Moya, A. Effect of deleterious mutation-accumulation on the fitness of RNA bacteriophage MS2. Evolution 2000, 54, 686–691. [Google Scholar] [CrossRef]
- Delgado, S.; Somovilla, P.; Ferrer-Orta, C.; Martínez-González, B.; et al. Incipient functional SARS-CoV-2 diversification identified through neural network haplotype maps. Proc. Natl. Acad. Sci. 2024, 121, e2317851121. [Google Scholar] [CrossRef] [PubMed]
- Denhardt, D.; Silver, R.B. An analysis of the clone size distribution of φX174 mutants and recombinants. Virology 1966, 30, 10–19. [Google Scholar] [CrossRef]
- Dickison, M.; Havlin, S.; Stella, H.E. Epidemics on interconnected networks. Phys. Rev. E 2012, 85, 066109. [Google Scholar] [CrossRef] [PubMed]
- Domingo, E. (2001). Quasispecies and RNA Virus Evolution: Principles and Consequences (E. Domingo, Ed.). Landes Bioscience.
- Domingo, E. (2020). Virus as Populations: Composition, Complexity, Dynamics, and Biological Implications (2nd ed.). Academic Press, Elsevier Inc.
- Domingo, E.; García-Crespo, C.; Perales, C. Historical perspective on the discovery of the quasispecies concept. Ann. Rev. Virol. 2021, 8, 51–72. [Google Scholar] [CrossRef] [PubMed]
- Domingo, E.; Holland, J.J.; Webster, R.G. (Eds.). (1999). Origin and Evolution of Viruses. Elsevier Science.
- Domingo, E.; Sabo, D.; Taniguchi, T.; Weissman, C. Nucleotide sequence heterogeneity of an RNA phage population. Cell 1978, 13, 735–744. [Google Scholar] [CrossRef]
- Domingo, E.; Schuster, P.; Elena, S.F.; Perales, C. (Eds.). (2023). Viral Fitness and Evolution: Population Dynamics and Adaptive Mechanisms. Springer International Publishing.
- Domingo-Calap, P.; Cuevas, J.M.; Sanjuán, R. The fitness effects of random mutations in single-stranded DNA and RNA bacteriophages. PLoS Genet. 2009, 5, e1000742. [Google Scholar] [CrossRef]
- Duarte, E.A.; Novella, I.S.; Ledesma, S.; Clarke, D.K.; et al. Subclonal components of consensus fitness in an RNA virus clone. J. Virol. 1994, 68, 4295–4301. [Google Scholar] [CrossRef]
- Eigen, M. Selforganization of matter and the evolution of biological macromolecules. Naturwissenschaften 1971, 58, 465–523. [Google Scholar] [CrossRef] [PubMed]
- Eigen, M.; McCaskill, P.J.; Schuster, P. (1989). Molecular quasi-species (Advances in Chemical Physics. John Wiley & Sons, Inc. ed., Vol. LXXV). I. Prigogine, Stuart A. Rice.
- Eigen, M.; Schuster, P. (1979). The Hypercycle: A Principle of Natural Self-Organization. Springer Berlin Heidelberg.
- Elena, S.; Moya, A. Rate of deleterious mutation and the distribution of its effects on fitness in vesicular stomatitis virus. J. Evol. Biol. 1999, 12, 1078–1088. [Google Scholar] [CrossRef]
- Elena, S.F.; Solé, R.; Saranyés, J. Simple genomes, complex interactions: Epistasis in RNA virus. Chaos 2010, 20, 026106. [Google Scholar] [CrossRef] [PubMed]
- Fernández, G.; Clotet, B.; Martínez, M.A. Fitness landscape of human immunodeficiency virus type 1 protease quasispecies. J. Virol. 2007, 81, 1485–2496. [Google Scholar] [CrossRef]
- Funk, S.; Jansen, V.A.A. Interacting epidemics on overlay networks. Phys. Rev. E 2010, 81, 036118. [Google Scholar] [CrossRef] [PubMed]
- García-Crespo, C.; de Ávila, A.I.; gallego, I.; Soria, M.E.; et al. Synergism between remdesivir and ribavirin leads to SARS-CoV-2 extinction in cell culture. Br. J. Pharmacol. 2024, 1–19. [Google Scholar] [CrossRef] [PubMed]
- García-Villada, L.; Drake, J.W. The three faces of riboviral spontaneous mutation: spectrum, mode of genome replication, and mutation rate. PLoS Genet. 2012, 8, e1002832. [Google Scholar] [CrossRef] [PubMed]
- González-Aparicio, L.; López, C. Selection of nonstandard viral genomes during the evolution of RNA viruses: a virus survival strategy of a pesky inconvenience? Adv. Virus Res. 2024, 119, 39–61. [Google Scholar] [PubMed]
- Gordon, C.J.; Tchesnokov, E.P.; Schinazi, R.F.; Götte, M. Molnupiravir promotes SARS-CoV-2 mutagenesis via the RNA template. J. Biol. Chem. 2021, 297, 100770. [Google Scholar] [CrossRef]
- Grande-Pérez, A.; Lázaro, E.; Lowenstein, P.; Domingo, E.; et al. Suppression of viral infectivity through lethal defection. Proc. Natl. Acad. Sci. USA 2005, 102, 4448–4452. [Google Scholar] [CrossRef]
- Grande-Pérez, A.; Sierra, S.; Castro, M.G.; Domingo, E.; et al. Molecular indetermination in the transition to error catastrophe: Systematic elimination of lymphocytic choriomeningitis virus through mutagenesis does not correlate linearly with large increases in mutant spectrum complexity. Proc. Natl. Acad. Sci. USA 2002, 99, 12938–12943. [Google Scholar] [CrossRef] [PubMed]
- Gregori, J.; Colomer-Castell, S.; Ibañez-Lligoña, M.; García-Cehid, D.; et al. In-host flat-like quasispecies: characterization methods and clinical implications. Microroganisms 2024, 12, 1011. [Google Scholar] [CrossRef] [PubMed]
- Harak, C.; Lohmann, V. Ultrastructure of the replication sites of positive-strand RNA viruses. J. Virol. 2015, 419, 418–433. [Google Scholar] [CrossRef] [PubMed]
- Hillung, J.; Olmo-Uceda, M.J.; Muñoz-Sánchez, J.C.; Elena, S.F. Accumulation dynamics of defective genomes during experimental evolution of two betacoronaviruses. Viruses 2024, 16, 644. [Google Scholar] [CrossRef]
- Holland, J.J.; Domingo, E.; de la Torre, J.; Steinhauer, D.A.; et al. Mutation frequencies at defined single codon sites in vesicular stomatitis virus and poliovirus can be increased only slightly by chemical mutagenesis. J. Virol. 1990, 64, 3960–3962. [Google Scholar] [CrossRef] [PubMed]
- Holland, J.J.; Spindler, K.; Horodyski, F.; Grabau, E.; Nichol, S.; et al. Rapid evolution of RNA genomes. Science 1982, 215, 1577–1585. [Google Scholar] [CrossRef] [PubMed]
- Honjo, M.; Emura, N.; Kawagoe, T.; Sujisaka, J.; et al. Seasonality of interactions between a plant virus and its host during persistent infection in a natural environment. ISME J. 2020, 14, 506–518. [Google Scholar] [CrossRef] [PubMed]
- Jacobi, M.N.; Nordahl, M. Quasispecies and recombination. Theor. Pop. Biol. 2006, 70, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Kabinger, F.; Stiller, C.; Schmitzová, J.; Dienemann, C.; et al. Mechanism of molnupiravir-induced SARS-CoV-2 mutagenesis. Nature Struct. Molec. Biol. 2021, 28, 740–746. [Google Scholar] [CrossRef]
- Kauffman, S.A.; Levin, S. Towards a general theory of adaptive walks on rugged landscapes. J. Theor. Biol. 1987, 128, 11–45. [Google Scholar] [CrossRef]
- Kopek, B.G.; Perkins, G.; Miller, D.J.; Ellisman, M.H.; et al. Three-dimensional analysis of a viral RNA replication complex reveals a virus-induced mini-organelle. PLoS Biol. 2007, 5, e220. [Google Scholar] [CrossRef] [PubMed]
- Lederberg, J. (1993). Viruses and humankind. Intracellular symbiosis and evolutionary competition. In S. S. Morse (Ed.), Emerging Viruses (pp. 3-9). Oxford University Press.
- Li, J.; Browning, S.; Mahal, S.; Oelschlegel, A.; et al. Darwinian evolution of prions in cell culture. Science 2010, 327, 869–872. [Google Scholar] [CrossRef] [PubMed]
- Loeb, L.; Essigmann, J.; Kazazi, F.; Zhang, J.; et al. Lethal mutagenesis of HIV with mutagenic nucleoside analogs. Proc. Natl. Acad. Sci. USA 1999, 96, 1492–1497. [Google Scholar] [CrossRef] [PubMed]
- Mackey, M.C.; Glass, L. Oscillation and chaos in physiological control systems. Science 1977, 197, 287–289. [Google Scholar] [CrossRef]
- Mansky, L.; Temin, H. Lower in vivo mutation rate of human immunodeficiency virus type 1 than that predicted from the fidelity of purified reverse transcriptase. J. Virol. 1995, 69, 5087–5094. [Google Scholar] [CrossRef]
- Martínez, F.; Sardanyés, J.; Elena, S.F.; Daròs, J.A. Dynamics of a plant RNA virus intracellular accumulation: stamping machine vs. geometric replication. Genetics 2011, 188, 637–646. [Google Scholar] [CrossRef]
- Martínez-González, B.; Soria, M.E.; Mínguez, P.; Lorenzo-Redondo, R.; et al. SARS-CoV-2 mutant spectra as variant of concern nurseries: endless variation? Front. Microb. 2024, 15, 1358258. [Google Scholar] [CrossRef]
- Martínez-González, B.; Vázquez-Sirvent, I.; Soria, M.E.; Mínguez, P.; et al. SARS-CoV-2 mutant spectra at different depth levels reveal an overwhelming abundance of low frequency mutations. Pathogens 2022, 11, 662. [Google Scholar] [CrossRef]
- Martínez-González, B.; Vázquez-Sirvent, L.; Soria, M.E.; Mínguez, P.; et al. Vaccine breakthrough infections with SARS-CoV-2 Alpha mirror mutations in Delta Plus, Iota, and Omicron. J. Clin. Inv. 2022, 132, e157700. [Google Scholar] [CrossRef]
- Mas, A.; Ulloa, E.; Bruguera, M.; Furčić, V.; et al. Hepatitis C virus population analysis of a single-source nosocomial outbreak reveals an inverse correlation between viral load and quasispecies complexity. J. Gen. Virol. 2004, 85, 3619–3626. [Google Scholar] [CrossRef]
- Mateu, M.; Martínez, M.A.; Rocha, E.; Andreu, D.; et al. Implications of a quasispecies genome structure: effect of frequent, naturally occurring amino acid substitutions on the antigenicity of foot-and-mouth disease virus. Proc. Natl. Acad. Sci. USA 1989, 86, 5883–5887. [Google Scholar] [CrossRef] [PubMed]
- Mendiola, A.S.; Serrano, M.; Boguñá, M. Epidemic spreading on interconnected networks. Phys. Rev. E 2012, 86, 026106. [Google Scholar] [CrossRef] [PubMed]
- Morens, D.; Fauci, A. Emerging pandemic diseases: how we got to COVID-19. Cell 2000, 183, 837. [Google Scholar] [CrossRef] [PubMed]
- Morse, S.S. (Ed.). (1993). Emerging Viruses. Oxford University Press.
- Novella, I.; Duarte, E.A.; Elena, S.F.; Moya, A.; et al. Exponential increases of RNA virus fitness during large population transmissions. Proc. Natl. Acad. Sci. USA 1992, 92, 5841–5844. [Google Scholar] [CrossRef] [PubMed]
- Nowak, M.; Schuster, P. Error thresholds of replication in finite populations mutation frequencies and the onset of Muller’s ratchet. J. Theor. Biol. 1989, 137, 375–395. [Google Scholar] [CrossRef] [PubMed]
- Olmo-Ulceda, M.J.; Muñoz-Sánchez, J.C.; Lasso-Giraldo, W.; Arnau, V.; et al. DVGfinder: a metasearch engine for identifying defective viral genomes in RNA-Seq data. Viruses 2022, 14, 1114. [Google Scholar] [CrossRef] [PubMed]
- Organization WH. (2003). SARS: status of the outbreak and lessons for the immediate future 2003. WHO. https://www.who.int/csr/media/sars_wha.pdf.
- Ortega-Prieto, A.; Sheldon, J.; Grande-Pérez, A.; Tejero, H. Extinction of hepatitis C virus by ribavirin in hepatoma cells involves lethal mutagenesis. PLoS ONE 2013, 8, e71039. [Google Scholar] [CrossRef]
- Papastavrou, N.; Horning, D.P.; Joyce, G.F. RNA-catalyzed evolution of catalytic RNA. Proc. Natl. Acad. Sci. USA 2024, 121, e2321592121. [Google Scholar] [CrossRef] [PubMed]
- Pastor-Satorras, R.; Vespignani, A. Epidemic spreading in scale-free networks. Phys. Rev. Lett. 2001, 86, 3200. [Google Scholar] [CrossRef]
- Perales, C. Quasispecies dynamics and clinical significance of hepatitis C virus (HCV) antiviral resistance. Int. J. Antimicrob. Agents 2020, 56, 105562. [Google Scholar] [CrossRef]
- Perales, C.; Gallego, I.; de Ávila, A.I.; Soria, M.E.; et al. The increasing impact of lethal mutagenesis of viruses. Future Med. Chem. 2019, 11, 1645–1657. [Google Scholar] [CrossRef] [PubMed]
- Perales, C.; Henry, M.; Domingo, E.; Wain-Hobson, S. Lethal mutagenesis of foot-and-mouth disease virus involves shifts in sequence space. J. Virol. 2011, 85, 12227–12240. [Google Scholar] [CrossRef] [PubMed]
- Pilosof, S.; Porter, M.; Pascual, M.; Kéfi, S. The multilayer nature of ecological networks. Nat Ecol. Evol. 2017, 1, 0101. [Google Scholar] [CrossRef] [PubMed]
- Pocock, M.J.O.; Evans, D.M.; Memmott, J. The robustness and restoration of a network of ecological networks. Science 2012, 335, 973–977. [Google Scholar] [CrossRef] [PubMed]
- Rangel, M.A.; Dolan, P.T.; Taguwa, S.; et al. High-resolution mapping reveals the mechanism and contribution of genome insertions and deletions to RNA virus evolution. Proc. Natl. Acad. Sci. USA 2023, 120, e2304667120. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Jarabo, C.M.; Arias, A.; Baranowski, E.; Escarmís, C.; et al. Memory in viral quasispecies. J. Virol. 2000, 74, 3543–3547. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Jarabo, C.M.; Ly, C.; Domingo, E.; de la Torre, J.C. Lethal mutagenesis of the prototypic arenavirus lymphocytic choriomeningitis virus (LCMV). Virology 2003, 308, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Saakian, D.B.; Hu, C.-K. Exact solution of the Eigen model with general fitness functions and degradation rates. Proc. Natl. Acad. Sci. USA 2006, 103, 4935–4939. [Google Scholar] [CrossRef]
- Saakian, D.B.; Koh, J.M.; Cheong, K.H. Approximate perturbative solutions of quasispecies model with recombination. Phys. Rev. E 2019, 99, 062407. [Google Scholar] [CrossRef]
- Sahneh, F.D.; Scoglio, C. Competitive epidemic spreading over arbitrary multilayer networks. Phys. Rev. E 2014, 89, 062817. [Google Scholar] [CrossRef]
- Sanjuán, R. Mutational fitness effects in RNA and single-stranded DNA viruses: common patterns revealed by site-directed mutagenesis studies. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 1975–1982. [Google Scholar] [CrossRef] [PubMed]
- Sanjuán, R.; Cuevas, J.M.; Holmes, E.C.; Moya, A. Selection for robustness in mutagenized RNA viruses. PLoS Genet. 2007, 3, e93. [Google Scholar] [CrossRef] [PubMed]
- Sanjuán, R.; Moya, A.; Elena, S.F. The distribution of fitness effects caused by single-nucleotide substitutions in an RNA virus. Proc. Natl. Acad. Sci. USA 2004, 101, 8396–8401. [Google Scholar] [CrossRef] [PubMed]
- Sanz, J.; Xia, C.-Y.; Meloni, S.; Moreno, Y. Dynamics of interacting diseases. Phys. Rev. X 2014, 4, 041005. [Google Scholar] [CrossRef]
- Sardanyés, J.; Elena, S.F. Error threshold in RNA quasispecies models with complementation. J. Theor. Biol. 2010, 265, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Sardanyés, J.; Elena, S.F. Quasispecies spatial models for RNA viruses with different replication modes and infection strategies. PLoS ONE 2011, 6, e24884. [Google Scholar] [CrossRef] [PubMed]
- Sardanyés, J.; Elena, S.F.; Solé, R.V. Simple quasispecies models for the survival-of-the-flattest effect: the role of space. J. Theor. Biol. 2008, 250, 560–568. [Google Scholar] [CrossRef]
- Sardanyés, J.; Solé, R.; Elena, S.F. Replication mode and landscape topology differentially affect RNA virus mutational load and robustness. J. Virol. 2009, 83, 12579–12589. [Google Scholar] [CrossRef]
- Sardanyés, J.; Solé, R.V. (2013). Red Queen coevolution on fitness landscapes. In H. Richter & A. Engelbrecht (Eds.), Recent Advances in the Theory and Application of Fitness Landscapes (pp. 301-338). Springer Berlin Heidelberg.
- Schulte, M.B.; Draghi, J.A.; Plotkin, J.B.; Andino, R. Experimentally guided models reveal replication principles that shape the mutation distribution of RNA viruses. eLife 2015, 4, e03753. [Google Scholar] [CrossRef]
- Schuster, P. (1994). How do RNA molecules and viruses explore their worlds? In G. A. Cowan, D. Pines, & D. Meltzer (Eds.), Complexity: Metaphors, Models, and Reality (pp. 383-418). Avalon Publishing.
- Schuster, P. (2016). Quasispecies on fitness landscapes. In Quasispecies: From Theory to Experimental Systems (pp. 61-120). E. Domingo, P. Schuster.
- Schuster, P.; Stadler, P. (2023). Viral Fitness and Evolution. Current Topics in Microbiology and Immunology, (E. Domingo, P. Schuster, S. F. Elena, & C. Perales, Eds.; Vol. 439). Springer International Publishing.
- Schuster, P.; Swetina, J. Stationary mutant distributions and evolutionary optimization. Bull. Math. Biol. 1988, 50, 635–660. [Google Scholar] [CrossRef]
- Shirogane, Y.; Watanabe, S.; Yanagi, Y. Cooperation between different RNA virus genomes produces a new phenotype. Nat Commun 2012, 3, 1235. [Google Scholar] [CrossRef] [PubMed]
- Smolinski, M.S.; Hamburg, M.A.; Lederberg, J. (Eds.) . (2003). Microbial Threats to Health: Emergence, Detection, and Response. National Academies Press.
- Snell, N.J. Novel and re-emerging respiratory infections. Expert Rev. Anti-infect. Ther. 2004, 2, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Solé, R.V. (2000). Signs Of Life: How Complexity Pervades Biology. Basic Books.
- Solé, R.V. Phase transitions in unstable cancer cell populations. Eur. Phys. J. 2003, 35, 117–123. [Google Scholar] [CrossRef]
- Solé, R.V.; Deisboek, T. An error catastrophe in cancer? J. Theor. Biol. 2004, 228, 47–54. [Google Scholar] [CrossRef]
- Solé, R.V.; Elena, S.F. (2018). Viruses as Complex Adaptive Systems, Princeton University Press.
- Solé, R.V.; Sardanyés, J.; Díez, J.; Mas, A. Information catastrophe in RNA viruses through replication thresholds. J. Theor. Biol. 2006, 240, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Solé, R.V.; Sardanyés, J.; Elena, S.F. Phase transitions in virology. Rep. Progr. Phys. 2021, 84, 115901. [Google Scholar] [CrossRef]
- Somovilla, P.; García-Crespo, C.; Martínez-González, B.; Soria, M.E.; et al. Atypical mutational spectrum of SARS-CoV-2 replicating in the presence of ribavirin. Antimicrob. Agents Chemother. 2023, 67, e0131522. [Google Scholar] [CrossRef]
- Stella, M.; Andreazzi, C.; Selakovic, S.; Goudarzi, A. Parasite spreading in spatial ecological multiplex networks. J. Complex Netw. 2017, 5, 486–511. [Google Scholar] [CrossRef]
- Swetina, J.; Schuster, P. Self-replication with errors: a model for polynucleotide replication. Biphys. Chem. 1982, 16, 329–345. [Google Scholar] [CrossRef]
- Tapper, M.L. Emerging viral diseases and infectious disease risks. Haemophilia 2006, 12, 3–7. [Google Scholar] [CrossRef]
- van Nimwegen, E.; Crutchfield, J.P.; Huynen, M. Neutral evolution of mutational robustness. Proc. Natl. Acad. Sci. USA 1999, 96, 9176–9720. [Google Scholar] [CrossRef] [PubMed]
- Vignuzzi, M.; López, C.B. Defective viral genomes are key drivers of the virus–host interaction. Nat. Microbiol. 2019, 4, 1075–1087. [Google Scholar] [CrossRef] [PubMed]
- Wilke, C.O.; Ronnewinkel, C. Dynamic fitness landscapes: expansions for small mutation rates. Phys. A 2001, 290, 475–490. [Google Scholar] [CrossRef]
- Wilke, C.O.; Ronnewinkel, C.; Martinetz, T. Dynamic fitness landscapes in molecular evolution. Phys. Rep. 2001, 349, 395–446. [Google Scholar] [CrossRef]
- Wilke, C.O.; Wang, J.L.; Ofria, C.; Lenski, R.E.; et al. Evolution of digital organisms at high mutation rates leads to survival of the flattest. Nature 2001, 412, 331–333. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.C.Y.; Law, S.K.P.; Huang, Y.; Yuen, K.Y. Coronavirus diversity, phylogeny and interspecies jumping (Short Survey). Exp. Biol. Med. 2009, 234, 1117–1127. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Stapon, A.; Bouda, E.; Garcia-Diaz, M. Fidelity of ribonucleotide incorporation by the SARS-CoV-2 replication complex. J. Mol. Biol. 2023, 435, 167973. [Google Scholar] [CrossRef]
- Zandi, M.; Shafaati, M.; Shapshak, P.; Reza Hashemnia, S.M. Monkeypox virus replication underlying circadian rhythm networks. J. Neurovirol. 2023, 29, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Li, L.; Peng, H.; Luo, Q.; et al. Multiple routes transmitted epidemics on multiplex networks. Phys. Lett. A 2014, 378, 770–776. [Google Scholar] [CrossRef]
- Zhou, T.; Gilliam, N.J.; Li, S.; Spandau, S.; et al. Generation and functional analysis of defective viral genomes during SARS-CoV-2 infection. mBio 2023, 14, e0025023. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



