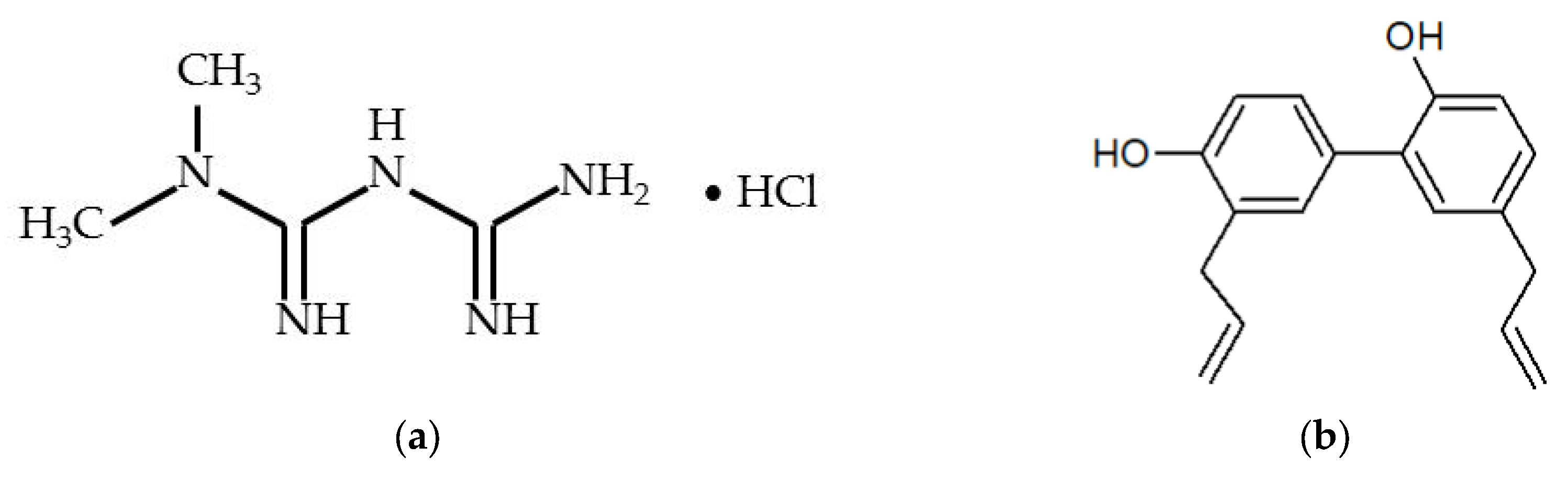Submitted:
05 July 2024
Posted:
09 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
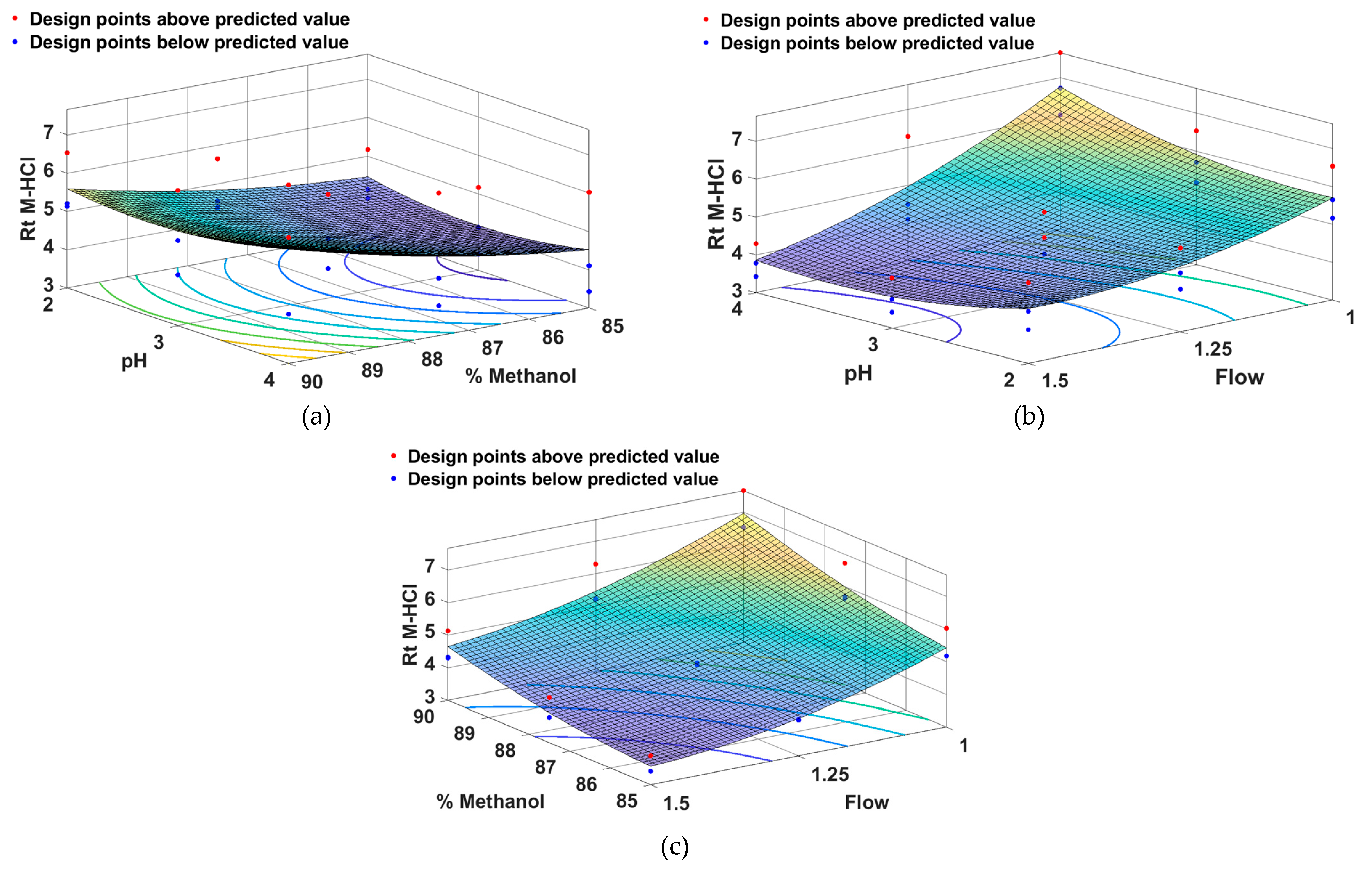
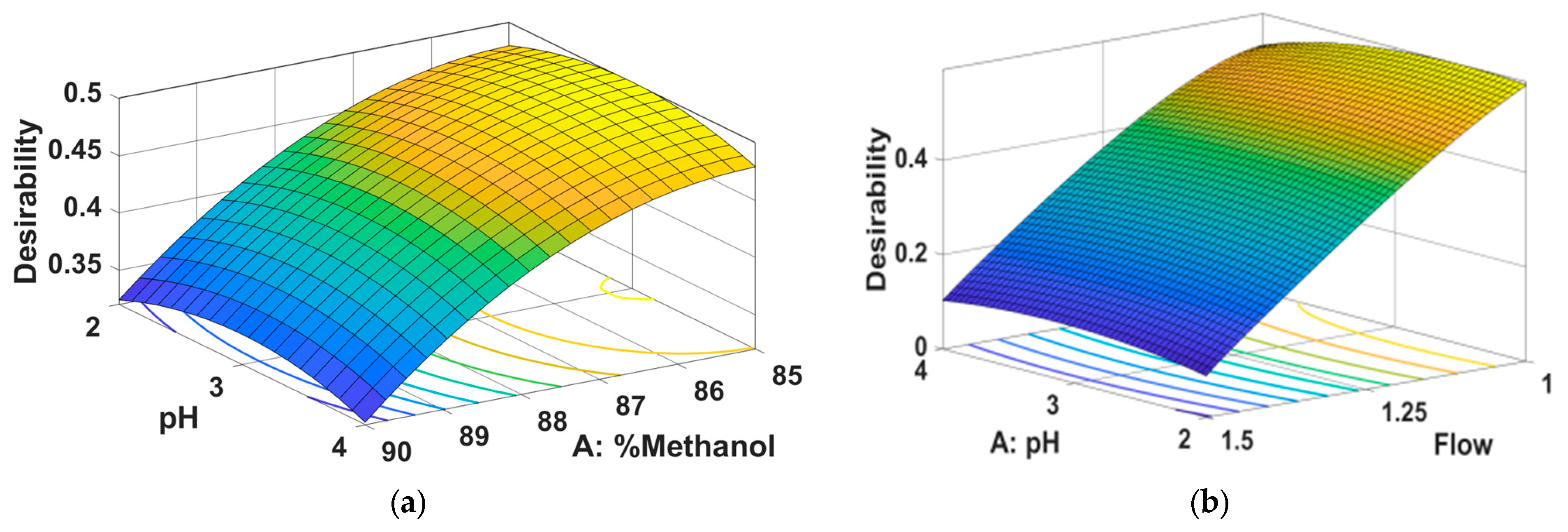
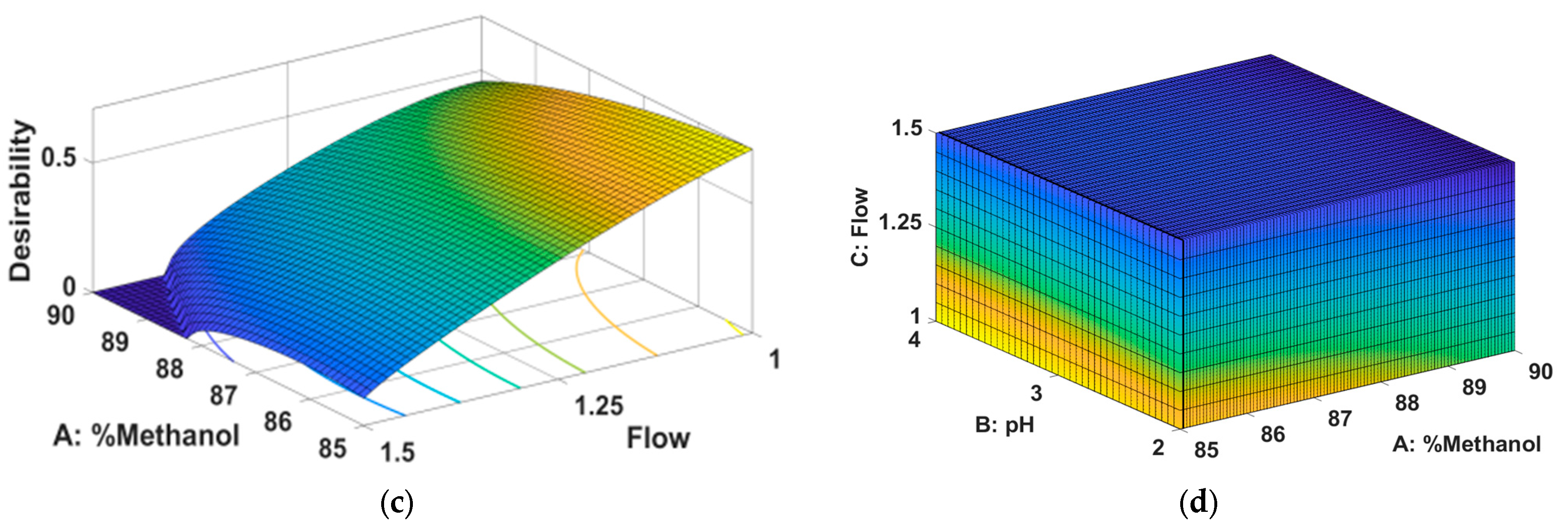
2.1. Method Development and Optimization Using QbD Approach
- Method Optimization
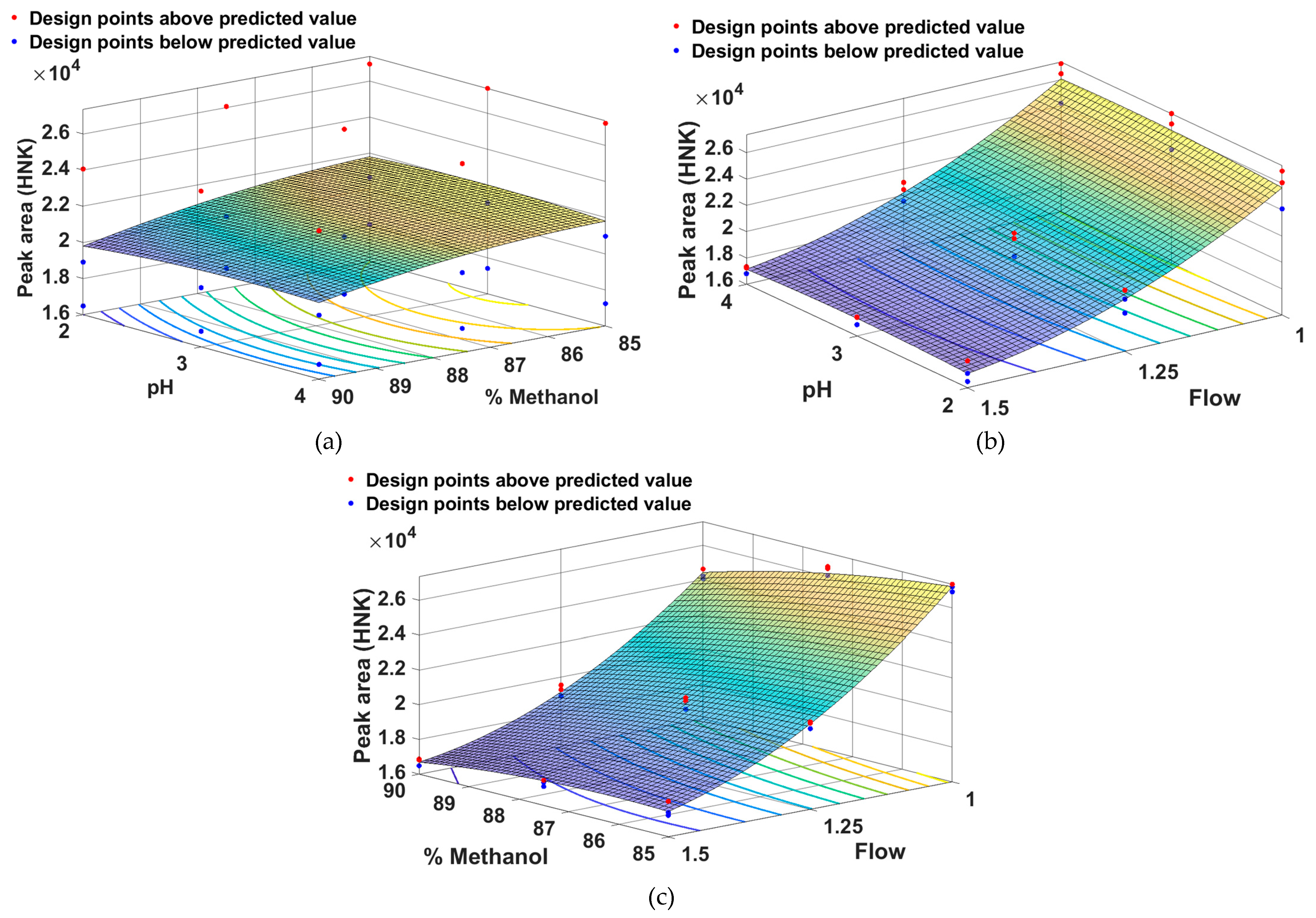
| Peak area (HNK) | Peak area (M-HCl) | Rt (M-HCl) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Coefficient | t-Stat | P-value | Coefficient | t-Stat | P-value | Coefficient | t-Stat | P-value | |
| β0 | –222525.41 | –1.849 | 0.082 | –1904154* | –3.0572 | 0.007 | 214.03 | 2.0114 | 0.060 |
| β1 | 8281.01* | 3.025 | 0.007 | 46999* | 3.3178 | 0.004 | –4.9477 | –2.0444 | 0.056 |
| β2 | –1946.43 | –0.776 | 0.449 | 4826.5 | 0.37163 | 0.715 | –2.6944 | –1.2144 | 0.241 |
| β3 | –143856.08* | –13.76 | 1.207×10–10 | –112896.35* | –2.0866 | 0.049 | 2.9691 | 0.32122 | 0.752 |
| β4 | 44.07 | 1.597 | 0.129 | –54.803 | –0.38366 | 0.706 | 0.0336 | 1.3768 | 0.186 |
| β5 | 810.72* | 7.343 | 1.149×10–6 | 1814.5* | 3.1756 | 0.005 | –0.1829 | –1.874 | 0.078 |
| β6 | –359.50 | –1.303 | 0.210 | 1346.6 | 0.94272 | 0.359 | –1.6527* | –6.7722 | 0.000 |
| β7 | –55.91* | –3.581 | 0.002 | –286.18* | –3.5417 | 0.002 | 0.0305* | 2.2092 | 0.041 |
| β8 | –55.91* | –2.302 | 0.034 | –304.32 | –0.60258 | 0.555 | 0.3233* | 3.7469 | 0.001 |
| β9 | 22597.51* | 14.47 | 5.456×10–11 | –29485* | –3.649 | 0.001 | 5.5324* | 4.0076 | 0.001 |
| Run | Run order | Factor 1 | Factor 2 | Factor 3 | Response 1 | Response 2 | Response 3 |
|---|---|---|---|---|---|---|---|
| Methanol (%) |
pH | Flow (mL/min) |
Peak area (HNK) |
Peak area (M-HCl) |
Rt M-HCl (min) |
||
| 1 | 1 | 85 | 2 | 1 | 26934.4 | 36288.5 | 5.170 |
| 2 | 13 | 87.5 | 2 | 1 | 26051.8 | 35084.3 | 5.651 |
| 3 | 3 | 90 | 2 | 1 | 24065.6 | 29777.44 | 6.537 |
| 4 | 5 | 85 | 2 | 1.25 | 20652.0 | 32147.4 | 4.125 |
| 5 | 17 | 87.5 | 2 | 1.25 | 19963.5 | 30535.5 | 4.555 |
| 6 | 7 | 90 | 2 | 1.25 | 18914.8 | 25817.9 | 5.212 |
| 7 | 9 | 85 | 2 | 1.5 | 18052.4 | 22994.3 | 3.896 |
| 8 | 15 | 87.5 | 2 | 1.5 | 17096.1 | 20943.2 | 4.381 |
| 9 | 11 | 90 | 2 | 1.5 | 16482.0 | 20862.1 | 5.132 |
| 10 | 21 | 85 | 3 | 1 | 27366.9 | 37076.2 | 5.167 |
| 11 | 25 | 87.5 | 3 | 1 | 26577.8 | 36215.17 | 5.702 |
| 12 | 23 | 90 | 3 | 1 | 24623.7 | 30006.2 | 6.535 |
| 13 | 20 | 85 | 3 | 1.25 | 21042.0 | 30104.6 | 4.119 |
| 14 | 27 | 87.5 | 3 | 1.25 | 20627.6 | 31948.4 | 4.553 |
| 15 | 18 | 90 | 3 | 1.25 | 19280.3 | 25998 | 5.228 |
| 16 | 22 | 85 | 3 | 1.5 | 17406.8 | 22245.6 | 3.423 |
| 17 | 26 | 87.5 | 3 | 1.5 | 17451.7 | 21623.1 | 3.770 |
| 18 | 24 | 90 | 3 | 1.5 | 16868.4 | 20969.6 | 4.327 |
| 19 | 2 | 85 | 4 | 1 | 27219.6 | 35458.15 | 6.020 |
| 20 | 14 | 87.5 | 4 | 1 | 26458.6 | 34020.9 | 6.725 |
| 21 | 4 | 90 | 4 | 1 | 24217.1 | 28466 | 7.664 |
| 22 | 6 | 85 | 4 | 1.25 | 20970.1 | 31024.5 | 4.105 |
| 23 | 19 | 87.5 | 4 | 1.25 | 20429.2 | 32885.2 | 4.500 |
| 24 | 8 | 90 | 4 | 1.25 | 19543.6 | 24956.4 | 6.292 |
| 25 | 10 | 85 | 4 | 1.5 | 17241.7 | 23676.9 | 3.426 |
| 26 | 16 | 87.5 | 4 | 1.5 | 17337.4 | 21837 | 3.783 |
| 27 | 12 | 90 | 4 | 1.5 | 16816.4 | 20120.31 | 4.293 |
2.2. Method Validation
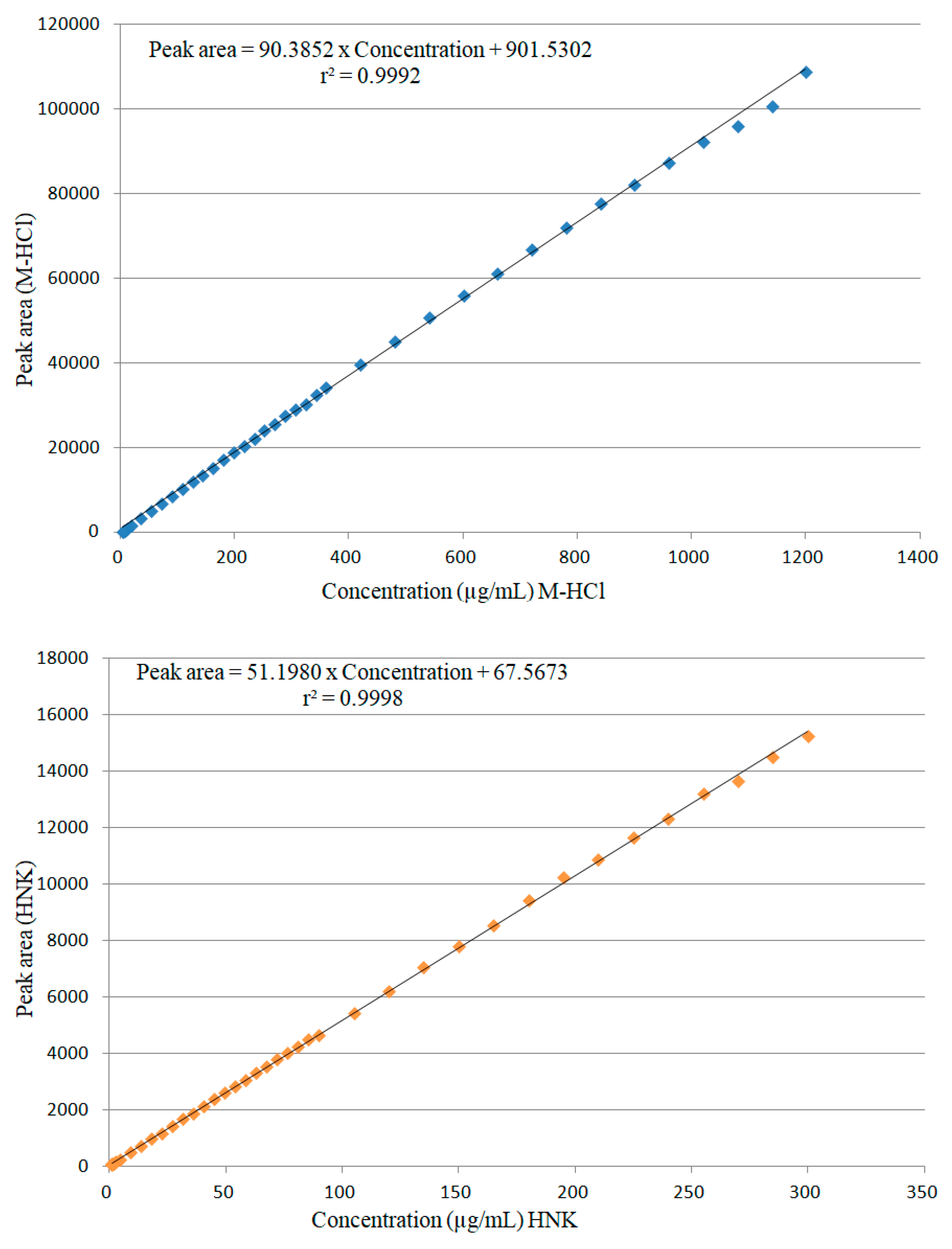
2.2.1. Linearity
2.2.2. Limits of Detection (LOD) and Limits of Quantification (LOQ)
2.2.3. Precision and Accuracy
2.3. Stability Studies for HNK and for M-HCl / HNK Solutions
- for HNK the recovery decreases after 24 hours with 0.46%, 0.72% and 0.93% and after 48 hours with 0.82%, 1.12% and 1.61% (for a concentration of 90 μg/mL), with 1.1%, 1.49% and 1.58% and after 48 hours with 1.37%, 1.66% and 2.16% (for a concentration of 300 μg/mL), in refrigerator, at room temperature and on the water bath respectively;
- for M-HCL / HNK mixture, the recovery of HNK are not changed with more than 1% after 24 hours or 48 hours for any studied concentration (90 or 300 μg/mL), in refrigerator, at room temperature and on the water bath respectively and the recovery of M-HCl are changed with more than 1% after 48 hours only for a concentration of 1200 μg/mL) on the water bath;
- for M-HCL / HNK mixture, in the case of photostability, the recovery are changed with more than 1% after 48 hours for 1200 / 300 µg/mL M-HCl / HNK (unprotected for light) and 1200 M-HCl (protected for light).
2.4. Determination of HNK and M-HCl Content from Oral Dosage Forms
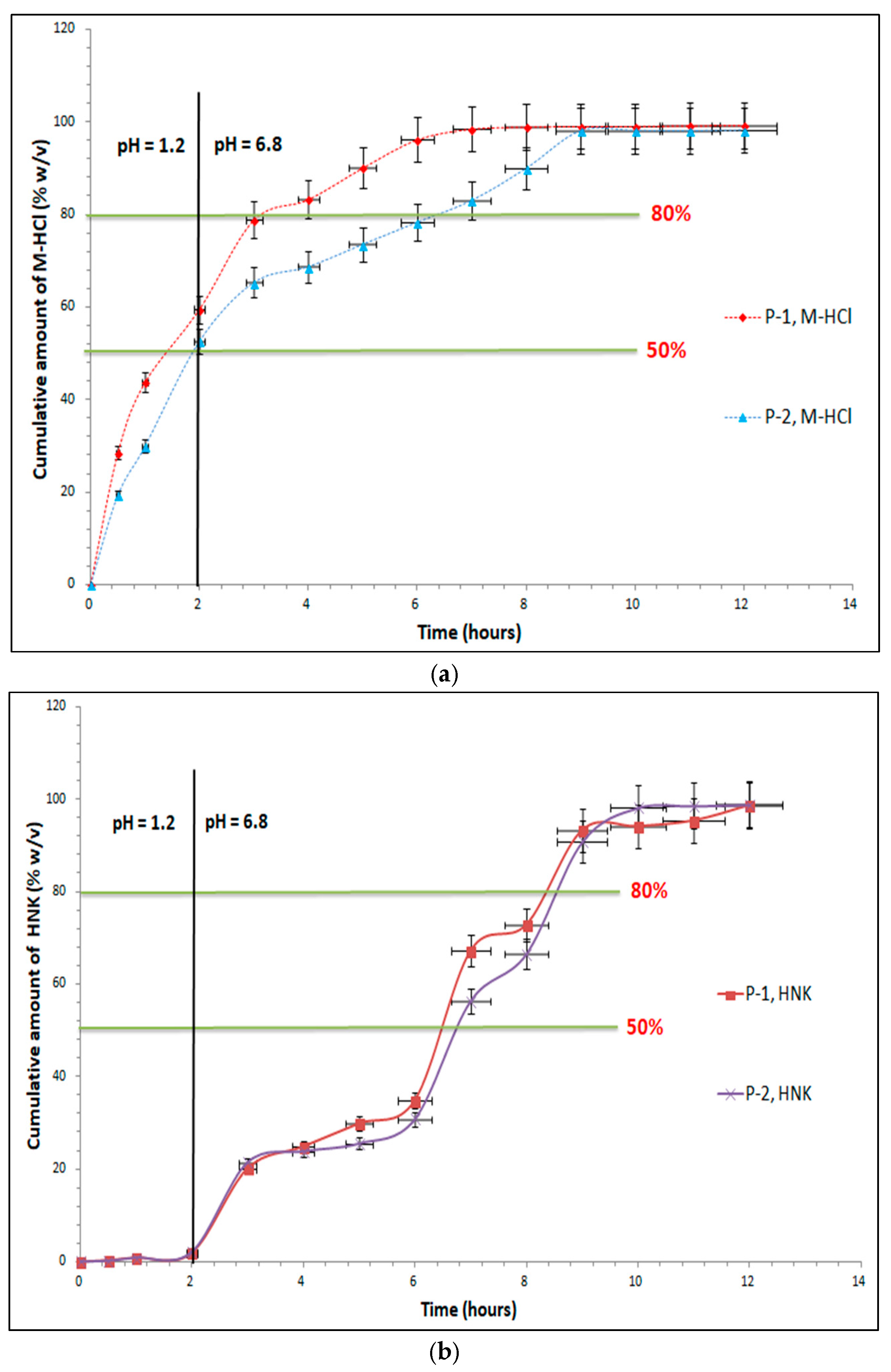
2.5. Dissolution Studies
2.6. Greenness of the Method
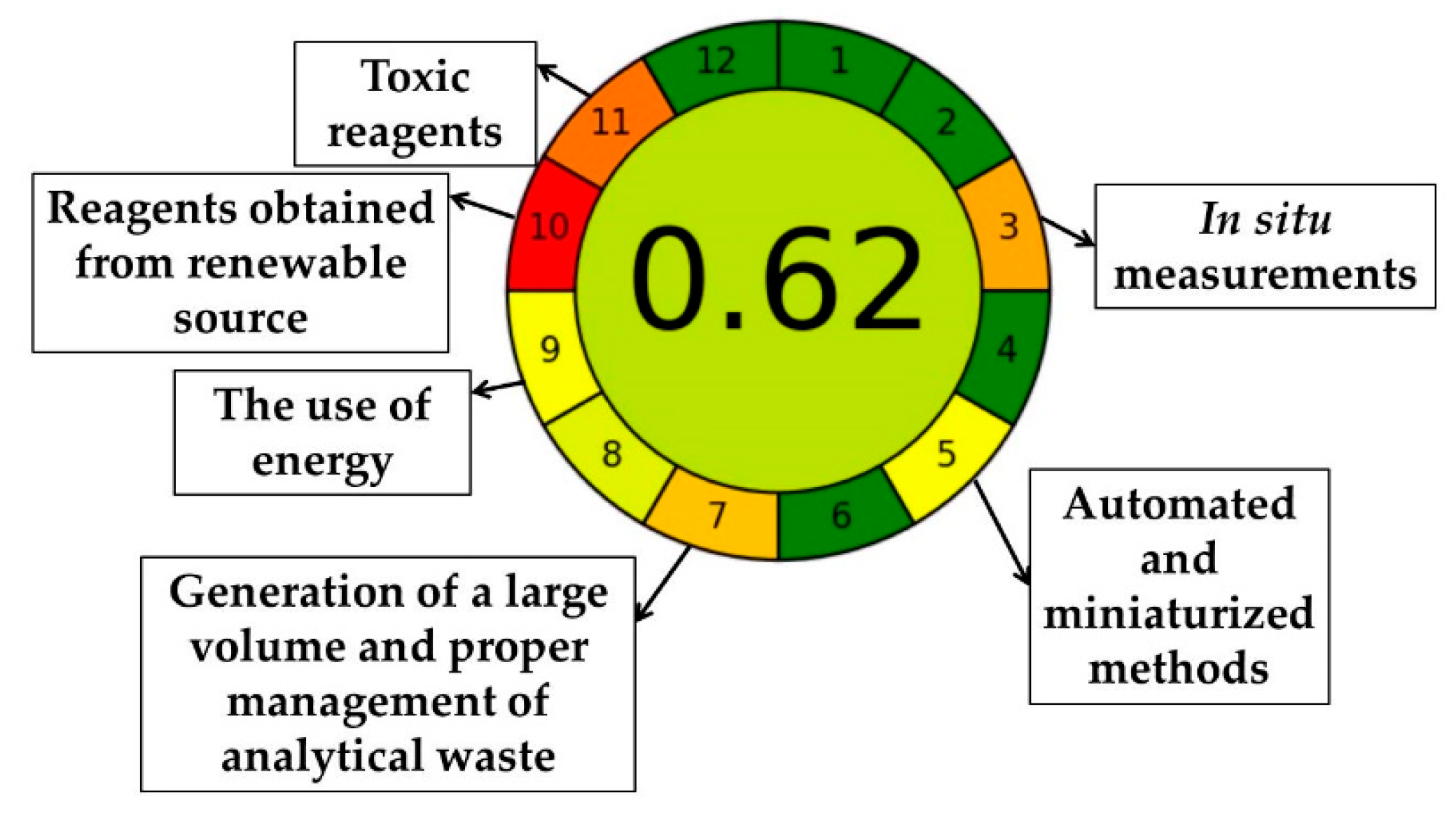
2.6.1. Evaluation of the Greenness of the Proposed Method (Analytical Eco-Scale, AES)
2.6.2. Analytical GREEnness Metric (AGREE)
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Equipment
3.3. Preparation of Standard Solutions (M-HCl Aqueous Solution, HNK Alcoholic Solution and M-HCl / HNK Final Hydroalcoholic Solution)
3.4. Selection and Preparation of Mobile Phase
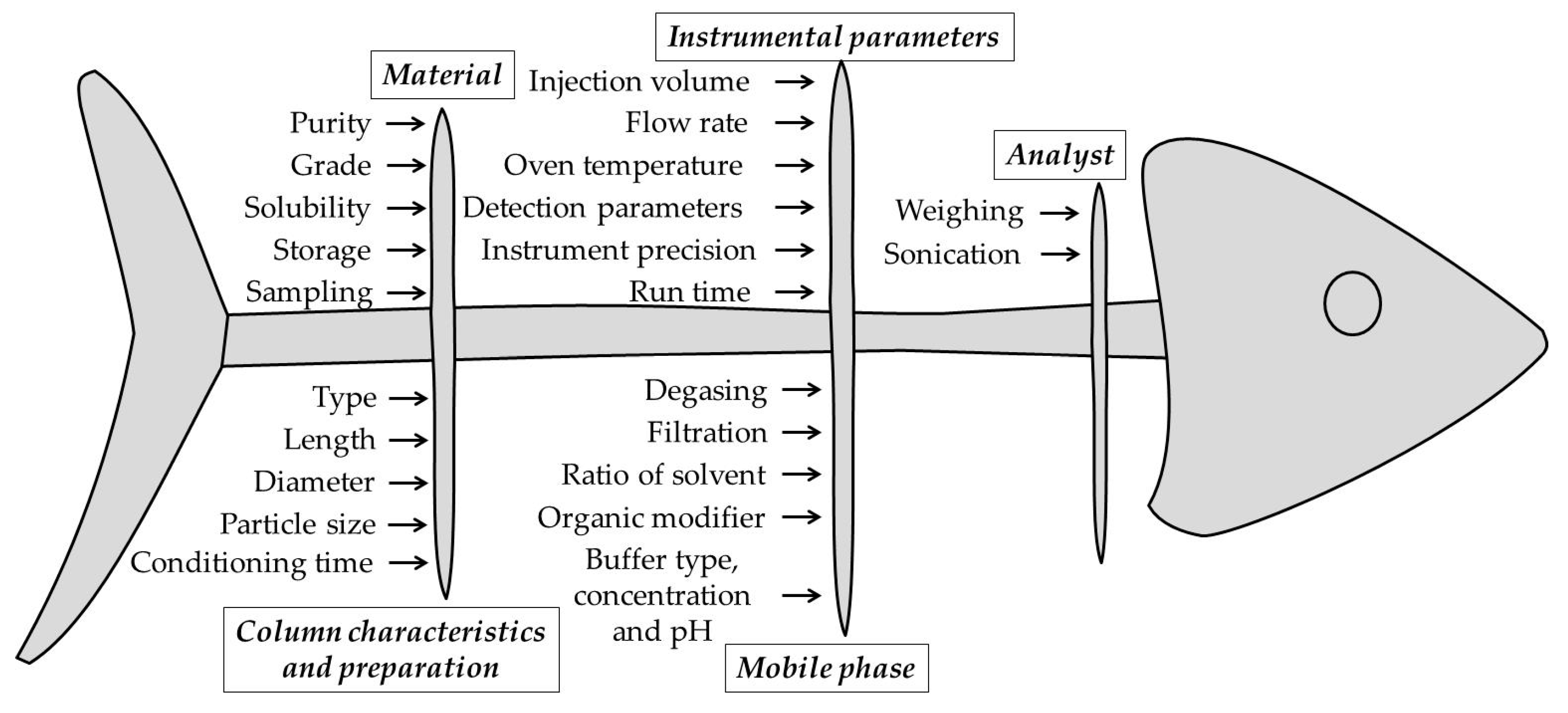
3.5. Method Development and Optimization Using QbD Approach
3.5.1. Method Development
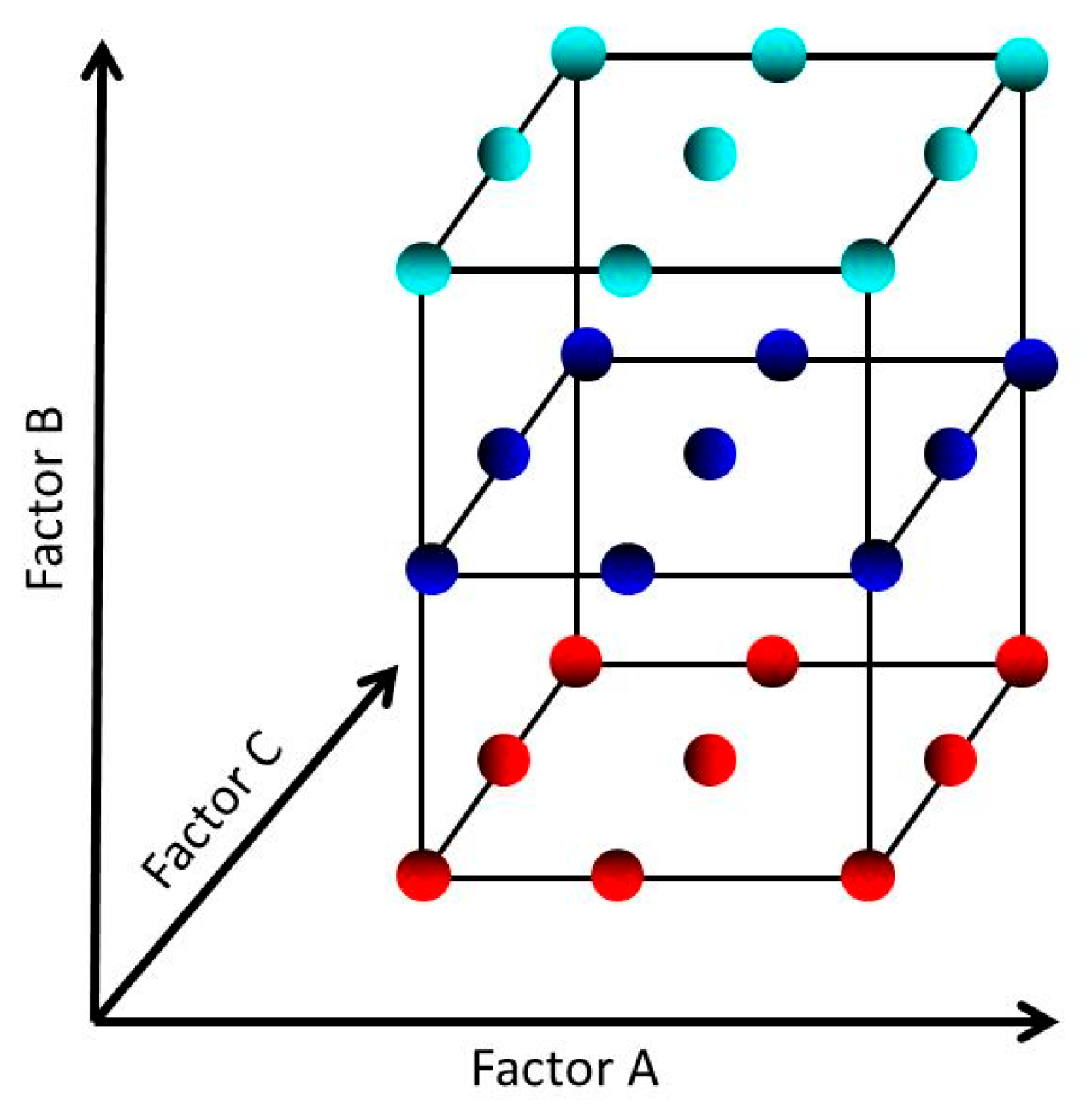
3.5.2. The Model and Treatment Runs for a 3 Factors – 3 Levels Design
3.6. Method Validation
3.7. Stability Studies for HNK Alcoholic Solution and M-HCl / HNK Hydroalcoholic Solutions
3.8. Determination of HNK and M-HCl Content from Oral Dosage Forms
3.9. Dissolution Studies
- C% = percentage release in the dissolution medium;
- CI% = percentage concentration calculated for the first sampling;
- CII% = percentage concentration calculated in the 2 mL taken previously;
- DF = dilution factor (1 or 2);
- PA = peak area (mAU·min);
- Int and S = intercept and slope of the regression line respectively;
- A = declared content (mg);
- tx = current sampling time;
- tx-1 = previous sampling time.
3.10. Greenness of the Method
3.10.1. Evaluation of the Greenness of the Proposed Method (Analytical Eco-Scale, AES)
3.10.2. Analytical GREEnness Metric (AGREE)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations: AES
References
- International Diabetes Federation (IDF). IDF Diabetes Atlas 10th edition, 2021 https://diabetesatlas.org/idfawp/resource-files/2021/07/IDF_Atlas_10th_Edition_2021.pdf (accessed on 28 May 2024).
- U.S. Centers for Disease Control And Prevention (CDC). National Diabetes Statistics Report. https://www.cdc.gov/diabetes/php/data-research/index.html (accessed on 28 May 2024).
- Chatzianagnostou, K.; Gaggini, M.; Suman Florentin, A.; Simonini, L.; Vassalle, C. New Molecules in Type 2 Diabetes: Advancements, Challenges and Future Directions. Int. J. Mol. Sci. 2024, 25, 6218. [Google Scholar] [CrossRef] [PubMed]
- Sha’at, M.; Spac, A.F.; Stoleriu, I.; Bujor, A.; Cretan, M.S.; Hartan, M.; Ochiuz, L. Implementation of QbD Approach to the Analytical Method Development and Validation for the Estimation of Metformin Hydrochloride in Tablet Dosage Forms by HPLC. Pharmaceutics 2022, 14, 1187. [Google Scholar] [CrossRef]
- Sha’at, M.; Ghiciuc, C.M.; Bujor, A.; Timofte, D.V.; Ștefanache, A.; Ochiuz, L. Literature review of oral treatment of type 2 diabetes mellitus. Med. Surg. J. 2020, 124, 482–495. Available online: https://revmedchir.ro/index.php/revmedchir/article/view/2225.
- Petrescu, D.C.; Vicovan, A.G.; Constantinescu, D.; Iftimi, E.; Sha’at, M.; Solcan, C.; Ochiuz, L.; Ghiciuc, C.M.; Ungureanu, L.B. Metformin’s impact on serum imunoglobulins and bronchoalveolar lavage cells in mice with complicated severe asthma. Med. Surg. J. 2024, 128, 316–323. Available online: https://www.revmedchir.ro/index.php/revmedchir/article/view/2961.
- Soukas, A.A.; Hao, H.; Wu, L. Metformin as Anti-Aging Therapy: Is It for Everyone? Trends Endocrinol. Metab. 2019, 30, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Pavaloiu, R.-D.; Sha’at, F.; Bubueanu, C.; Petrescu, M.; Sevcenco, C. Assessment of Pullulan, a Microbial Polysaccharide, as a Matrix for Senotherapeutics Delivery. Chem. Proc. 2023, 13, 23. [Google Scholar] [CrossRef]
- Gedawy, A.; Al-Salami, H.; Dass, C.R. Development and validation of a new analytical HPLC method for simultaneous determination of the antidiabetic drugs, metformin and gliclazide. J. Food Drug. Anal. 2019, 27, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Kant, R.; Bodla, R.B.; Kapoor, G.; Bhutani, R. Optimization of a single HPLC-PDA method for quantifying metformin, gliclazide, pioglitazone, dapagliflozin, empagliflozin, saxagliptin, linagliptin and teneligliptin using central composite design. Bioorg. Chem. 2019, 91, 103111. [Google Scholar] [CrossRef]
- Sebaiy, M.M.; El-Adl, S.M.; Baraka, M.M.; Hassan, A.A. Rapid RP-HPLC method for simultaneous estimation of metformin, pioglitazone, and glimepiride in human plasma. Acta Chromatogr. 2020, 32, 16–21. [Google Scholar] [CrossRef]
- Shende, M.A.; Budde, B.R. Novel RP-HPLC method development and validation for simultaneous estimation of metformin, voglibose and pioglitazone in bulk and triple fixed drug combinations pharmaceutical dosage form. J. Drug Deliv. Ther. 2019, 9, 30–37. [Google Scholar] [CrossRef]
- Mahrouse, M.A.; Lamie, N.T. Experimental design methodology for optimization and robustness determination in ion pair RP-HPLC method development: Application for the simultaneous determination of metformin hydrochloride, alogliptin benzoate and repaglinide in tablets. Microchem. J. 2019, 147, 691–706. [Google Scholar] [CrossRef]
- Raza, A.; Murtaza, S.H.; Hanif, S.; Iqbal, J.; Ali, I.; Aftab, T.; Shakir, R.; Bedar, R.; Ali Syed, M. Validation of a rapid and economical RP-HPLC method for simultaneous determination of metformin hydrochloride and sitagliptin phosphate monohydrate: Greenness evaluation using AGREE score. Pak. J. Pharm. Sci. 2022, 35, 15–21. [Google Scholar] [PubMed]
- Balamurugan, K.; Kirtimaya, M. Quality by design based development and validation of RP-HPLC method for simultaneous estimation of sitagliptin and metformin in bulk and pharmaceutical dosage forms. Int. J. Pharm. Investig. 2020, 10, 512–518. [Google Scholar]
- Shakoor, A.; Ahmed, M.; Ikram, R.; Hussain, S.; Tahir, A.; Jan, B.M.; Adnan, A. Stability-indicating RP-HPLC method for simultaneous determination of metformin hydrochloride and vildagliptin in tablet and biological samples. Acta Chromtogr. 2020, 32, 39–43. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines and HealthCare. European Pharmacopoeia, 11th ed.; Council of Europe: Strasbourg, France, 2023. [Google Scholar]
- Trifan, A.; Bostănaru, A.-C.; Luca, S.V.; Temml, V.; Akram, M.; Herdlinger, S.; Kulinowski, Ł.; Skalicka-Woźniak, K.; Granica, S.; Czerwińska, M.E.; et al. Honokiol and Magnolol: Insights into Their Antidermatophytic Effects. Plants 2021, 10, 2522. [Google Scholar] [CrossRef] [PubMed]
- Ricordi, C.; Pacifici, F.; Lanzoni, G.; Palamara, A.T.; Garaci, E.; Della-Morte, D. Dietary and Protective Factors to Halt or Mitigate Progression of Autoimmunity, COVID-19 and Its Associated Metabolic Diseases. Int. J. Mol. Sci. 2021, 22, 3134. [Google Scholar] [CrossRef]
- Lee, A.Y.; Christensen, S.M.; Duong, N.; Tran, Q.-A.; Xiong, H.M.; Huang, J.; James, S.; Vallabh, D.; Talbott, G.; Rose, M.; et al. Sirt3 Pharmacologically Promotes Insulin Sensitivity through PI3/AKT/mTOR and Their Downstream Pathway in Adipocytes. Int. J. Mol. Sci. 2022, 23, 3740. [Google Scholar] [CrossRef] [PubMed]
- Bibi, T.; Bano, S.; ud Din, F.; Ali, H.; Khan, S. Preparation, characterization, and pharmacological application of oral honokiol-loaded solid lipid nanoparticles for diabetic neuropathy. Int. J. Pharm. 2023, 645, 123399. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Jiang, W.; Ye, C.; Hu, T.; Yu, Q.; Meng, M.; Sun, L.; Liang, J.; Chen, Y. Honokiol Attenuates High Glucose-Induced Peripheral Neuropathy via Inhibiting Ferroptosis and Activating AMPK/SIRT1/PGC-1α Pathway in Schwann Cells. Phytother. Res. 2023, 37, 5787–5802. [Google Scholar] [CrossRef]
- Islam, M.R.; Islam, F.; Nafady, M.H.; Akter, M.; Mitra, S.; Das, R.; Urmee, H.; Shohag, S.; Akter, A.; Chidambaram, K.; et al. Natural Small Molecules in Breast Cancer Treatment: Understandings from a Therapeutic Viewpoint. Molecules 2022, 27, 2165. [Google Scholar] [CrossRef]
- Mottaghi, S.; Abbaszadeh, H. Natural Lignans Honokiol and Magnolol as Potential Anticarcinogenic and Anticancer Agents. A Comprehensive Mechanistic Review. Nutr. Cancer 2022, 74, 761–778. [Google Scholar] [CrossRef]
- Fried, L.E.; Arbiser, J.L. Honokiol, a multifunctional antiangiogenic and antitumor agent. Antioxid. Redox Signal. 2009, 11, 1139–1148. [Google Scholar] [CrossRef]
- Ong, C.P.; Lee, W.L.; Tang, Y.Q.; Yap, W.H. Honokiol: A Review of Its Anticancer Potential and Mechanisms. Cancers 2020, 12, 48. [Google Scholar] [CrossRef] [PubMed]
- Rauf, A.; Khan, S.A.; Olatunde, A.; Imran, M.; Alhumaydhi, F.A.; Aljohani, A.S.M.; Uddin, M.S.; Mitra, S.; Emran, T.B.; Khayrullin, M.; et al. Honokiol: A review of its pharmacological potential and therapeutic insights. Phytomedicine 2021, 90, 153647. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yuan, Z.; Wang, Y.; Wang, W.; Shi, J. Recent advances of honokiol: pharmacological activities, manmade derivatives and structure-activity relationship. Eur. J. Med. Chem. 2024, 272, 116471. [Google Scholar] [CrossRef]
- Khatoon, F.; Ali, S.; Kumar, V.; Elasbali, A.M.; Alhassan, H.H.; Alharethi, S.H.; Islam, A.; Hassan, M.I. Pharmacological Features, Health Benefits and Clinical Implications of Honokiol. J. Biomol. Struct. Dyn. 2022, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.; Li, Q.; Xiong, S.; Yang, Y.; Yang, Y.; Huang, C. Delivery strategies, structural modification, and pharmacological mechanisms of honokiol: a comprehensive review. Chem. Biodivers. 2024, 21, e202302032. [Google Scholar] [CrossRef] [PubMed]
- Cheresh, P.; Kim, S.-J.; Jablonski, R.; Watanabe, S.; Lu, Z.; Chi, M.; Helmin, K.A.; Gius, D.; Budinger, G.R.S.; Kamp, D.W. SIRT3 Overexpression Ameliorates Asbestos-Induced Pulmonary Fibrosis, mt-DNA Damage, and Lung Fibrogenic Monocyte Recruitment. Int. J. Mol. Sci. 2021, 22, 6856. [Google Scholar] [CrossRef]
- Zhang, W.; Li, B.; Lv, Y.; Wei, S.; Zhang, S.; Hu, Y. Transcriptomic analysis shows the antifungal mechanism of honokiol against Aspergillus flavus. Int. J. Food Microbiol. 2022, 384, 109972. [Google Scholar] [CrossRef]
- Usach, I.; Alaimo, A.; Fernández, J.; Ambrosini, A.; Mocini, S.; Ochiuz, L.; Peris, J.-E. Magnolol and Honokiol: Two Natural Compounds with Similar Chemical Structure but Different Physicochemical and Stability Properties. Pharmaceutics 2021, 13, 224. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Jung, U.J. Honokiol Improves Insulin Resistance, Hepatic Steatosis, and Inflammation in Type 2 Diabetic db/db Mice. Int. J. Mol. Sci. 2019, 20, 2303. [Google Scholar] [CrossRef]
- Sarrica, A.; Kirika, N.; Romeo, M.; Salmona, M.; Diomede, L. Safety and toxicology of magnolol and honokiol. Planta Med. 2018, 84, 1151–1164. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhai, T.; Chen, Y. Effects of Honokiol on CYP450 Activity and Transporter mRNA Expression in Type 2 Diabetic Rats. Int. J. Mol. Sci. 2018, 19, 815. [Google Scholar] [CrossRef]
- Yu, Y.; Li, M.; Su, N.; Zhang, Z.; Zhao, H.; Yu, H.; Xu, Y. Honokiol protects against renal ischemia/reperfusion injury via the suppression of oxidative stress, iNOS, inflammation and STAT3 in rats. Mol. Med. Rep. 2016, 13, 1353–1360. [Google Scholar] [CrossRef]
- Bunel, V.; Antoine, M.-H.; Stévigny, C.; Nortier, J.; Duez, P. New in vitro insights on a cell death pathway induced by magnolol and honokiol in aristolochic acid tubulotoxicity. Food Chem. Toxicol. 2016, 87, 77–87. [Google Scholar] [CrossRef]
- Liu, H.-T.; Wang, T.-E.; Hsu, Y.-T.; Chou, C.-C.; Huang, K.-H.; Hsu, C.-C.; Liang, H.-J.; Chang, H.-W.; Lee, T.-H.; Tsai, P.-S. Nanoparticulated Honokiol Mitigates Cisplatin-Induced Chronic Kidney Injury by Maintaining Mitochondria Antioxidant Capacity and Reducing Caspase 3-Associated Cellular Apoptosis. Antioxidants 2019, 8, 466. [Google Scholar] [CrossRef]
- Khushnud, T.; Mousa, S.A. Potential role of naturally derived polyphenols and their nanotechnology delivery in cancer. Mol. Biotechnol. 2013, 55, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Quan, Y.; Park, W.; Jin, J.; Kim, W.; Park, S.K.; Kang, K.P. Sirtuin 3 Activation by Honokiol Decreases Unilateral Ureteral Obstruction-Induced Renal Inflammation and Fibrosis via Regulation of Mitochondrial Dynamics and the Renal NF-κB-TGF-β1/Smad Signaling Pathway. Int. J. Mol. Sci. 2020, 21, 402. [Google Scholar] [CrossRef] [PubMed]
- Kataoka, S.; Umemura, A.; Okuda, K.; Taketani, H.; Seko, Y.; Nishikawa, T.; Yamaguchi, K.; Moriguchi, M.; Kanbara, Y.; Arbiser, J.L.; et al. Honokiol Acts as a Potent Anti-Fibrotic Agent in the Liver through Inhibition of TGF-β1/SMAD Signaling and Autophagy in Hepatic Stellate Cells. Int. J. Mol. Sci. 2021, 22, 13354. [Google Scholar] [CrossRef]
- Benedé, J.L.; Rodriguez, E.; Chisvert, A.; Salvador, A. Rapid and Simple Determination of Honokiol and Magnolol in Cosmetic Products by Liquid Chromatography with Ultraviolet Detection. Anal. Lett. 2021, 54, 1510–1521. [Google Scholar] [CrossRef]
- Zhuang, Q.; Pan, R.; Liu, X.; Xu, W.; Wang, H.; Zhang, X.; Lai, X.; Wang, H.; Zhang, L.; Jiang, J. A Validated Ultra-HPLC–MS/MS Method for Determination of Honokiol in Human Plasma and its Application to a Clinical Pharmacokinetic Study. Bioanalysis 2019, 11, 1085–1098. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.D.; Liu, X.L. HPLC analysis of magnolol and honokiol in Magnoliae Cortex after solvent sublation. Acta Chromatogr. 2008, 20, 147–156. [Google Scholar] [CrossRef]
- Wu, X.; Chen, X.; Hu, Z. High-performance liquid chromatographic method for simultaneous determination of honokiol and magnolol in rat plasma. Talanta 2003, 59, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ww, A.; Wei, X.J.C.; Biointerfaces, S.B. Evaluation of anticancer activity of honokiol by complexation with hydroxypropyl-β-cyclodextrin. Colloids Surf. B Biointerf. 2020, 196, 111298. [Google Scholar] [CrossRef]
- Zheng, X.; Kan, B.; Gou, M.; Fu, S.; Zhang, J.; Men, K.; Chen, L.; Luo, F.; Zhao, Y.; Zhao, X.; et al. Preparation of mpeg-pla nanoparticle for honokiol delivery in vitro. Int. J. Pharm. 2010, 386, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Godugu, C.; Doddapaneni, R.; Singh, M. Honokiol nanomicellar formulation produced increased oral bioavailability and anticancer effects in triple negative breast cancer (TNBC). Colloids Surf. B 2017, 153, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.W.; Gong, C.Y.; Shi, S.; Fu, S.Z.; Men, K.; Zeng, S. Self-assembled honokiol-loaded micelles based on poly(caprolactone)-poly(ethylene glycol)-poly(caprolactone) copolymer. Int. J. Pharm. 2009, 369, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.; Chen, J.; Lei, Q.; Tan, G.; Zou, Y.; Peng, G.; Xie, J.; Li, W. Rapid determination of multiple components in herbal medicine using a single reference compound by high-performance liquid chromatography at equal absorption wavelength: case study of Magnoliae officinalis cortex. Sep. Sci. Plus 2024, 7, 2300071. [Google Scholar] [CrossRef]
- Lata, E.; Fulczyk, A.; Ott, P.G.; Kowalska, T.; Sajewicz, M.; Moricz, A.M. Thin-Layer Chromatographic Quantification of Magnolol and Honokiol in Dietary Supplements and Selected Biological Properties of These Preparations. J. Chromatogr. A 2020, 1625, 461230. [Google Scholar] [CrossRef]
- Chu, C.; Li, J.; Wang, S.; Weng, L.; Jiang, L.; Zhang, H.; Liu, C.; Yan, J. A simple and sensitive dispersive micro-solid-phase extraction coupled with high-performance liquid chromatography for quantification of honokiol and magnolol in complex matrices. J. AOAC Int. 2020, 103, 1406–1411. [Google Scholar] [CrossRef]
- Report from the EMA-FDA QbD pilot program, EMA/213746/2017, European Medicines Agency, US Food & Drug Administration, 2017, https://www.fda.gov/media/104371/download (accessed on 22 April 2024).
- Kumar. M.; Pant, A.; Chopra, S.; Bhatia A. AQbD enabled method development and quantification of asiaticoside in foam-based formulations. Accredit. Qual. Assur 2024, 2024, 1-16. [CrossRef]
- Attimarad, M.; Alali, M.J.; Alali, H.A.; Alabdulmuhsin, D.H.; Alnajdi, A.K.; Venugopala, K.N.; Nair, A.B. Design of Experimental Approach for Development of Rapid High Performance Liquid Chromatographic Process for Simultaneous Estimation of Metoprolol, Telmisartan, and Amlodipine from Formulation: Greenness and Whiteness Evaluation. Molecules 2024, 29, 1087. [Google Scholar] [CrossRef] [PubMed]
- International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH Topic Q2 (R1). Validation of Analytical Procedures: Text and Methodology. Step 5. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/ich-q-2-r1-validation-analytical-procedures-text-methodology-step-5_en.pdf (accessed on 15 March 2024).
- International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH Topic Q2 (R2). Validation of Analytical Procedures: Text and Methodology, European Medicines Agency. Step 5, Committee for Medicinal Products for Human Use, EMA/CHMP/ICH/82072/2006, December 2023. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/ich-q2r2-guideline-validation-analytical-procedures-step-5-revision-1_en.pdf (accessed on 15 March 2024).
- Kumar, N.; Sangeetha, D. Analytical method development by using QbD—An emerging approach for robust analytical method development. J. Pharm. Sci. Res. 2020, 12, 1298–1305. Available online: https://www.jpsr.pharmainfo.in/Documents/Volumes/vol12issue10/jpsr12102009.pdf.
- Sandhu, P.S.; Beg, S.; Katare, O.P.; Singh, B. QbD-Driven Development and Validation of a HPLC Method for Estimation of Tamoxifen Citrate with Improved Performance. J. Chromatogr. Sci. 2016, 54, 1373–1384. [Google Scholar] [CrossRef] [PubMed]
- Palakurthi, A.K.; Dongala, T.; Katakam, L.N.R. QbD based development of HPLC method for simultaneous quantification of Telmisartan and Hydrochlorothiazide impurities in tablets dosage form. Pract. Lab. Med. 2020, 21, e00169. [Google Scholar] [CrossRef]
- Jadhav, M.L.; Tambe, S.R. Implementation of QbD Approach to the Analytical Method Development and Validation for the Estimation of Propafenone Hydrochloride in Tablet Dosage Form. Chromatogr. Res. Int. 2013, 2013, 676501. [Google Scholar] [CrossRef]
- Martínez, J.; Cortés, J.F.; Miranda, R. Green Chemistry Metrics, A Review. Processes 2022, 10, 1274. [Google Scholar] [CrossRef]
- Romanian Pharmacopoeia Commission National Medicines Agency. Romanian Pharmacopoeia, 10th ed.; Medical Publishing House: Bucharest, Romania, 1993. https://scholar.google.com/scholar_lookup?title=Romanian+Pharmacopoeia&author=Romanian+Pharmacopoeia+Commission+National+Medicines+Agency&publication_year=1993.
- Gałuszka, A.; Migaszewski, Z.M.; Konieczka, P.; Namieśnik, J. Analytical Eco-Scale for assessing the greenness of analytical procedures. TrAC Trends Anal. Chem. 2012, 37, 61–72. [Google Scholar] [CrossRef]
- Mlinarić, Z.; Turković, L.; Begović, I.; Nigović, B.; Sertić, M. Rapid Capillary Electrophoresis Method for Simultaneous Determination of Abemaciclib, Ribociclib, and Palbociclib in Pharmaceutical Dosage Forms: A Green Approach. Molecules 2022, 27, 7603. [Google Scholar] [CrossRef]
- Tobiszewski, M. Metrics for green analytical chemistry. Anal. Methods 2016, 8, 2993–2999. [Google Scholar] [CrossRef]
- Three-level full factorial designs, In: NIST/SEMATECH e-Handbook of Statistical Methods; https://www.itl.nist.gov/div898/handbook/pri/section3/pri339.htm (accessed on 6 March 2024).
- Pena-Pereira, F.; Wojnowski, W.; Tobiszewski, M. AGREE—Analytical GREEnness metric approach and software. Anal. Chem. 2020, 92, 10076–10082. [Google Scholar] [CrossRef]
- Chandrasekaram, K.; Alias, Y.; Mohamad, S. Dispersive Membrane Microextraction of Substituted Phenols from Honey Samples and a Brief Outlook on Its Sustainability Using Analytical Eco-Scale and Analytical GREEnness Metric Approach. Membranes 2022, 12, 649. [Google Scholar] [CrossRef] [PubMed]

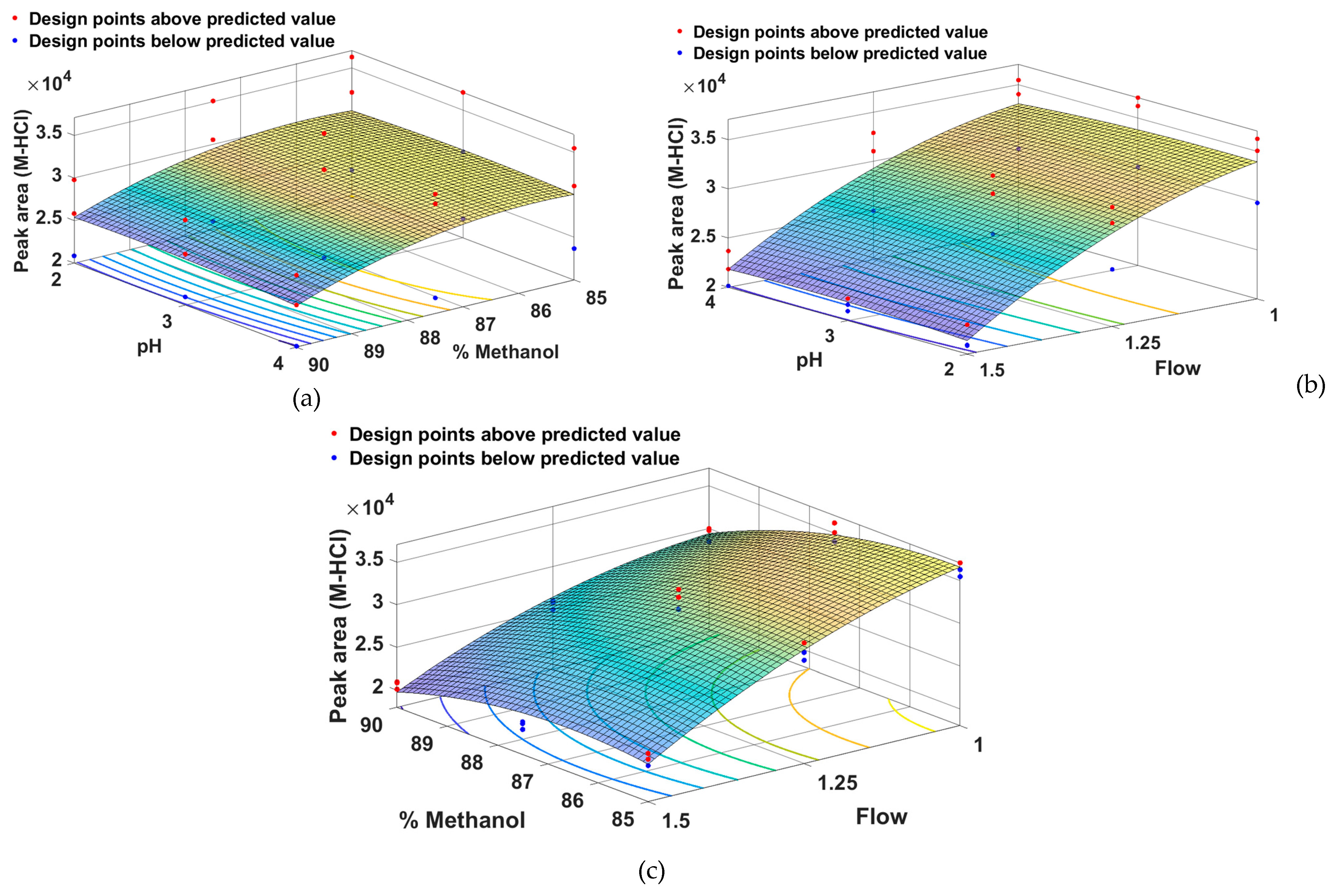
| Run | Run order | Factor 1 | Factor 2 | Factor 3 | Response 1 | Response 2 | Response 3 |
|---|---|---|---|---|---|---|---|
| Methanol (%) |
pH | Flow (mL/min) |
Peak area (HNK) |
Peak area (M-HCl) |
Rt M-HCl (min) |
||
| 1 | 1 | 85 | 2 | 1 | 26934.4 | 36288.5 | 5.170 |
| 2 | 13 | 87.5 | 2 | 1 | 26051.8 | 35084.3 | 5.651 |
| 3 | 3 | 90 | 2 | 1 | 24065.6 | 29777.44 | 6.537 |
| 4 | 5 | 85 | 2 | 1.25 | 20652.0 | 32147.4 | 4.125 |
| 5 | 17 | 87.5 | 2 | 1.25 | 19963.5 | 30535.5 | 4.555 |
| 6 | 7 | 90 | 2 | 1.25 | 18914.8 | 25817.9 | 5.212 |
| 7 | 9 | 85 | 2 | 1.5 | 18052.4 | 22994.3 | 3.896 |
| 8 | 15 | 87.5 | 2 | 1.5 | 17096.1 | 20943.2 | 4.381 |
| 9 | 11 | 90 | 2 | 1.5 | 16482.0 | 20862.1 | 5.132 |
| 10 | 21 | 85 | 3 | 1 | 27366.9 | 37076.2 | 5.167 |
| 11 | 25 | 87.5 | 3 | 1 | 26577.8 | 36215.17 | 5.702 |
| 12 | 23 | 90 | 3 | 1 | 24623.7 | 30006.2 | 6.535 |
| 13 | 20 | 85 | 3 | 1.25 | 21042.0 | 30104.6 | 4.119 |
| 14 | 27 | 87.5 | 3 | 1.25 | 20627.6 | 31948.4 | 4.553 |
| 15 | 18 | 90 | 3 | 1.25 | 19280.3 | 25998 | 5.228 |
| 16 | 22 | 85 | 3 | 1.5 | 17406.8 | 22245.6 | 3.423 |
| 17 | 26 | 87.5 | 3 | 1.5 | 17451.7 | 21623.1 | 3.770 |
| 18 | 24 | 90 | 3 | 1.5 | 16868.4 | 20969.6 | 4.327 |
| 19 | 2 | 85 | 4 | 1 | 27219.6 | 35458.15 | 6.020 |
| 20 | 14 | 87.5 | 4 | 1 | 26458.6 | 34020.9 | 6.725 |
| 21 | 4 | 90 | 4 | 1 | 24217.1 | 28466 | 7.664 |
| 22 | 6 | 85 | 4 | 1.25 | 20970.1 | 31024.5 | 4.105 |
| 23 | 19 | 87.5 | 4 | 1.25 | 20429.2 | 32885.2 | 4.500 |
| 24 | 8 | 90 | 4 | 1.25 | 19543.6 | 24956.4 | 6.292 |
| 25 | 10 | 85 | 4 | 1.5 | 17241.7 | 23676.9 | 3.426 |
| 26 | 16 | 87.5 | 4 | 1.5 | 17337.4 | 21837 | 3.783 |
| 27 | 12 | 90 | 4 | 1.5 | 16816.4 | 20120.31 | 4.293 |
| Peak area (HNK) | Peak area (M-HCl) | Rt (M-HCl) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Coefficient | t-Stat | P-value | Coefficient | t-Stat | P-value | Coefficient | t-Stat | P-value | |
| β0 | –222525.41 | –1.849 | 0.082 | –1904154* | –3.0572 | 0.007 | 214.03 | 2.0114 | 0.060 |
| β1 | 8281.01* | 3.025 | 0.007 | 46999* | 3.3178 | 0.004 | –4.9477 | –2.0444 | 0.056 |
| β2 | –1946.43 | –0.776 | 0.449 | 4826.5 | 0.37163 | 0.715 | –2.6944 | –1.2144 | 0.241 |
| β3 | –143856.08* | –13.76 | 1.207×10–10 | –112896.35* | –2.0866 | 0.049 | 2.9691 | 0.32122 | 0.752 |
| β4 | 44.07 | 1.597 | 0.129 | –54.803 | –0.38366 | 0.706 | 0.0336 | 1.3768 | 0.186 |
| β5 | 810.72* | 7.343 | 1.149×10–6 | 1814.5* | 3.1756 | 0.005 | –0.1829 | –1.874 | 0.078 |
| β6 | –359.50 | –1.303 | 0.210 | 1346.6 | 0.94272 | 0.359 | –1.6527* | –6.7722 | 0.000 |
| β7 | –55.91* | –3.581 | 0.002 | –286.18* | –3.5417 | 0.002 | 0.0305* | 2.2092 | 0.041 |
| β8 | –55.91* | –2.302 | 0.034 | –304.32 | –0.60258 | 0.555 | 0.3233* | 3.7469 | 0.001 |
| β9 | 22597.51* | 14.47 | 5.456×10–11 | –29485* | –3.649 | 0.001 | 5.5324* | 4.0076 | 0.001 |
| Peak area HNK | Peak area M-HCl | Retention time M-HCl | |
|---|---|---|---|
| 0.9974 | 0.9686 | 0.9763 | |
| 0.9961 | 0.9520 | 0.9637 | |
| 239 | 1237.1 | 0.211 |
| %Methanol | pH | Flow | Peak area HNK | Peak area M-HCl | Retention time M-HCl | Desirability |
|---|---|---|---|---|---|---|
| 85 | 3 | 1 | 27379.17 | 36786.70 | 5.21 | 0.82 |
| Optimization | maximize | maximize | minimize | |||
| Priority | 3 | 2 | 1 | |||
| Parameters | Values |
|---|---|
| Stationary phase (column) | Thermoscientific ODS Hypersyl TM (250 x 4.6 mm, 5 μm) |
| Mobile phase | 0.02 M acetate buffer (pH = 3) / methanol (15/85, v/v) |
| Flow rate | 1 mL/min |
| Column temperature | 35 °C |
| Injection volume | 20 μL |
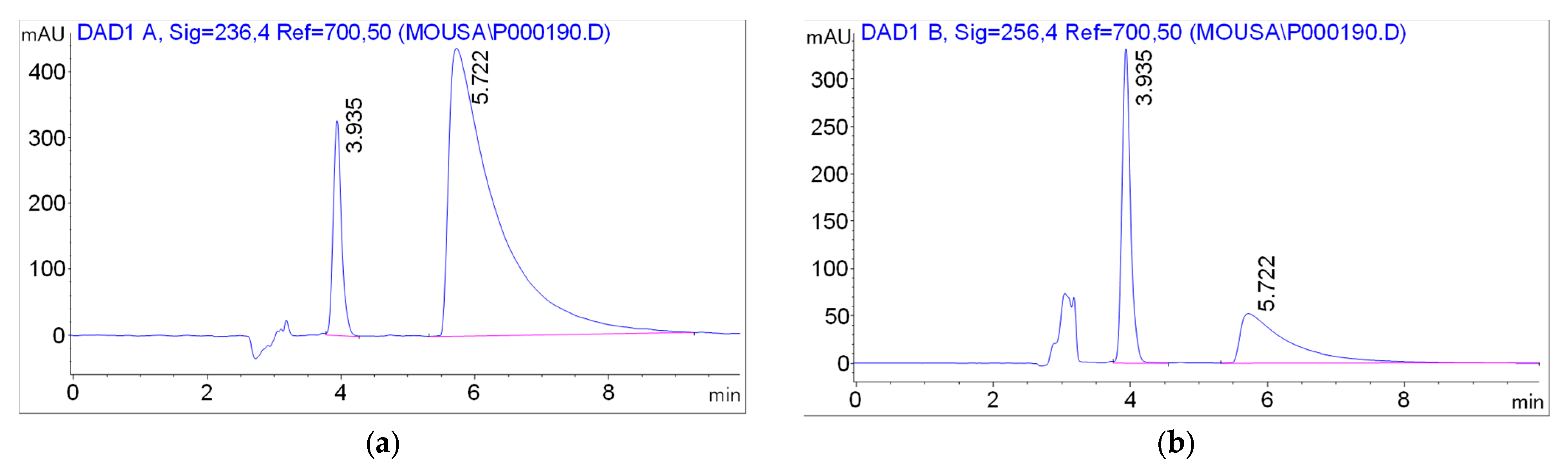
| Detection wavelength | 256 nm (HNK), 236 nm (M-HCl) |
| Retention time | 3.935 ± 0.197 min (HNK), 5.722 ± 0.286 min (M-HCl) |
| No. | Peak area (M-HCl) | Statistics | Peak area (HNK) | Statistics |
|---|---|---|---|---|
| 1 | 37175.5 | Mean = 37119.97 SD = 125.0813 RSD = 0.3370% |
5190.1 | Mean = 5193.02 SD = 20.6052 RSD = 0.3968% |
| 2 | 37104.6 | 5184.2 | ||
| 3 | 37341.9 | 5230.4 | ||
| 4 | 37009.9 | 5177.7 | ||
| 5 | 37030.6 | 5201.1 | ||
| 6 | 37045.3 | 5174.6 |
| Concentration (µg/mL) |
Method precision | Intermediate precision | ||
|---|---|---|---|---|
| Peak area | Statistics | Peak area | Statistics | |
| M-HCl | ||||
| 320 | 29784.7 | Mean = 29804.13 | 29784.6 | Mean = 29826.10 |
| 29834.6 | SD = 26.7171 | 30014.3 | SD = 171.2635 | |
| 29793.1 | RSD = 0.0896% | 29679.4 | RSD = 0.5742% | |
| 400 | 37009.9 | Mean = 37028.60 | 36820.3 | Mean = 37080.00 |
| 37030.6 | SD = 17.7845 | 37145.6 | SD = 233.9041 | |
| 37045.3 | RSD = 0.0480% | 37274.1 | RSD = 0.6308% | |
| 480 | 44285.4 | Mean = 44393.60 | 44217.5 | Mean = 44319.43 |
| 44453.2 | SD = 98.8652 | 44246.6 | SD = 152.0501 | |
| 44442.2 | RSD = 0.2114% | 44494.2 | RSD = 0.3431% | |
| HNK | ||||
| 80 | 4140.8 | Mean = 4160.87 | 4204.2 | Mean = 4175.57 |
| 4168.1 | SD = 17.6024 | 4155.9 | SD = 25.3678 | |
| 4173.7 | RSD = 0.4230% | 4166.6 | RSD = 0.6075% | |
| 100 | 5177.7 | Mean = 5184.47 | 5222.9 | Mean = 5235.40 |
| 5201.1 | SD = 14.4880 | 5224.1 | SD = 20.6201 | |
| 5174.6 | RSD = 0.2795% | 5259.2 | RSD = 0.3939% | |
| 120 | 6218.7 | Mean = 6235.57 | 6356.2 | Mean = 6352.67 |
| 6245.7 | SD = 14.7056 | 6365.1 | SD = 14.5260 | |
| 6242.3 | RSD = 0.2358% | 6336.7 | RSD = 0.2287% | |
| Method accuracy | |||||
|---|---|---|---|---|---|
| M-HCl | HNK | ||||
| Concentration (µg/mL) |
Peak area | Recovery (%) | Concentration (µg/mL) |
Peak area | Recovery (%) |
| 320 | 29818.6 | 99.98 | 80 | 4176.3 | 100.31 |
| 29820.9 | 99.99 | 4171.2 | 100.19 | ||
| 29876.7 | 100.18 | 4150.9 | 99.70 | ||
| 400 | 37212.2 | 100.43 | 100 | 5206.8 | 100.38 |
| 36989.2 | 99.82 | 5188.8 | 100.03 | ||
| 37184.9 | 100.36 | 5202.9 | 100.30 | ||
| 480 | 44374.6 | 100.20 | 120 | 6210.5 | 99.98 |
| 44199.5 | 99.80 | 6209.9 | 99.98 | ||
| 44300.2 | 100.03 | 6225.4 | 100.23 | ||
| Mean recovery (%) = 100.09 Minimum (%) = 99.80 Maximum (%) = 100.43 |
Mean recovery (%) = 100.12 Minimum (%) = 99.70 Maximum (%) = 100.38 |
||||
| No. | CR (μg/mL) |
Mean peak area (n = 3) |
CM (μg/mL) |
Recovery (%) |
RE (%) |
|---|---|---|---|---|---|
| 1 | 108 | 10394.13 | 105.02 | 97.24 | 2.76 |
| 2 | 126 | 12188.93 | 124.88 | 99.11 | 0.89 |
| 3 | 144 | 13793.67 | 142.64 | 99.06 | 0.94 |
| 4 | 162 | 15542.87 | 161.99 | 99.99 | 0.01 |
| 5 | 180 | 17427.33 | 182.84 | 101.58 | 1.58 |
| 6 | 198 | 19208.90 | 202.55 | 102.30 | 2.30 |
| 7 | 216 | 20630.83 | 218.28 | 101.06 | 1.06 |
| 8 | 234 | 22268.60 | 236.40 | 101.03 | 1.03 |
| 9 | 252 | 24214.30 | 257.93 | 102.35 | 2.35 |
| 10 | 270 | 25772.73 | 275.17 | 101.91 | 1.91 |
| 11 | 288 | 27801.57 | 297.62 | 103.34 | 3.34 |
| 12 | 306 | 29199.47 | 313.08 | 102.31 | 2.31 |
| 13 | 324 | 30397.33 | 326.33 | 100.72 | 0.72 |
| 14 | 342 | 32738.03 | 352.23 | 102.99 | 2.99 |
| 15 | 360 | 34506.30 | 371.80 | 103.28 | 3.28 |
| 16 | 420 | 39979.80 | 432.35 | 102.94 | 2.94 |
| 17 | 480 | 45267.90 | 490.86 | 102.26 | 2.26 |
| 18 | 540 | 50976.57 | 554.02 | 102.60 | 2.60 |
| 19 | 600 | 56196.80 | 611.77 | 101.96 | 1.96 |
| 20 | 660 | 61372.30 | 669.03 | 101.37 | 1.37 |
| 21 | 720 | 67165.27 | 733.13 | 101.82 | 1.82 |
| 22 | 780 | 72354.53 | 790.54 | 101.35 | 1.35 |
| 23 | 840 | 78040.73 | 853.45 | 101.60 | 1.60 |
| 24 | 900 | 82284.63 | 900.40 | 100.04 | 0.04 |
| 25 | 960 | 87452.63 | 956.58 | 99.75 | 0.25 |
| 26 | 1020 | 92566.40 | 1014.16 | 99.43 | 0.57 |
| 27 | 1080 | 96253.60 | 1054.95 | 97.68 | 2.32 |
| 28 | 1140 | 100993.77 | 1107.40 | 97.14 | 2.86 |
| 29 | 1200 | 109041.37 | 1196.43 | 99.70 | 0.30 |
| Recovery (Minimum = 97.14%, Mean = 100.96%, Maximum = 103.34%), Mean percent error = 1.71% | |||||
| No. | CR (μg/mL) |
Mean peak area (n = 3) |
CM (μg/mL) |
Recovery (%) |
RE (%) |
|---|---|---|---|---|---|
| 1 | 27 | 1419.27 | 26.40 | 97.78 | 2.22 |
| 2 | 31.5 | 1680.17 | 31.50 | 99.99 | 0.01 |
| 3 | 36 | 1872.47 | 35.25 | 97.93 | 2.07 |
| 4 | 40.5 | 2153.70 | 40.75 | 100.61 | 0.61 |
| 5 | 45 | 2404.13 | 45.64 | 101.42 | 1.42 |
| 6 | 49.5 | 2626.03 | 49.97 | 100.95 | 0.95 |
| 7 | 54 | 2842.05 | 54.19 | 100.35 | 0.35 |
| 8 | 58.5 | 3063.24 | 58.51 | 100.02 | 0.02 |
| 9 | 63 | 3307.77 | 63.29 | 100.46 | 0.46 |
| 10 | 67.5 | 3530.37 | 67.64 | 100.20 | 0.20 |
| 11 | 72 | 3788.09 | 72.67 | 100.93 | 0.93 |
| 12 | 76.5 | 4010.05 | 77.00 | 100.66 | 0.66 |
| 13 | 81 | 4260.62 | 81.90 | 101.11 | 1.11 |
| 14 | 85.5 | 4493.72 | 86.45 | 101.11 | 1.11 |
| 15 | 90 | 4674.54 | 89.98 | 99.98 | 0.02 |
| 16 | 105 | 5430.71 | 104.75 | 99.76 | 0.24 |
| 17 | 120 | 6221.86 | 120.21 | 100.17 | 0.17 |
| 18 | 135 | 7065.04 | 136.67 | 101.24 | 1.24 |
| 19 | 150 | 7810.87 | 151.24 | 100.83 | 0.83 |
| 20 | 165 | 8554.37 | 165.76 | 100.46 | 0.46 |
| 21 | 180 | 9418.55 | 182.64 | 101.47 | 1.47 |
| 22 | 195 | 10240.67 | 198.70 | 101.90 | 1.90 |
| 23 | 210 | 10880.40 | 211.20 | 100.57 | 0.57 |
| 24 | 225 | 11661.63 | 226.46 | 100.65 | 0.65 |
| 25 | 240 | 12343.23 | 239.77 | 99.90 | 0.10 |
| 26 | 255 | 13216.40 | 256.82 | 100.71 | 0.71 |
| 27 | 270 | 13671.23 | 265.71 | 98.41 | 1.59 |
| 28 | 285 | 14516.93 | 282.23 | 99.03 | 0.97 |
| 29 | 300 | 15251.47 | 296.57 | 98.86 | 1.14 |
| Recovery (Minimum = 97.78%, Mean = 100.26%, Maximum = 101.90%), Mean percent error = 0.83% | |||||
| Time (hours) |
Refrigerator (2 – 8°C) | Room temperature (20 – 25 °C) | 37 °C | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Peak area | CC (µg/mL) |
R (%) | Peak area | CC (µg/mL) |
R (%) | Peak area | CC (µg/mL) |
R (%) | |
| 90 µg/mL | |||||||||
| 0 | 4663.1 | 89.76 | 99.73 | 4663.1 | 89.76 | 99.73 | 4663.1 | 89.76 | 99.73 |
| 24 | 4654.2 | 89.59 | 99.54 | 4642.4 | 89.36 | 99.28 | 4632.7 | 89.17 | 99.07 |
| 48 | 4637.8 | 89.27 | 99.18 | 4623.6 | 88.99 | 98.88 | 4601.1 | 88.55 | 98.39 |
| 300 µg/mL | |||||||||
| 0 | 15314.6 | 297.81 | 99.27 | 15314.6 | 297.81 | 99.27 | 15314.6 | 297.81 | 99.27 |
| 24 | 15257.8 | 296.70 | 98.90 | 15198.8 | 295.54 | 98.51 | 15184.2 | 295.26 | 98.42 |
| 48 | 15215.9 | 295.88 | 98.63 | 15172.1 | 295.02 | 98.34 | 15095.4 | 293.52 | 97.84 |
| Time (hours) |
M-HCl / HNK |
Refrigerator (2 – 8°C) | Room temperature (20 – 25 °C) | 37 °C | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Peak area | CC (µg/mL) |
R (%) | Peak area | CC (µg/mL) |
R (%) | Peak area | CC (µg/mL) |
R (%) | ||
| 360 / 90 µg/mL (M-HCl / HNK) | ||||||||||
| 0 | M-HCl | 33527.3 | 360.96 | 100.27 | 33527.3 | 360.96 | 100.27 | 33527.3 | 360.96 | 100.27 |
| HNK | 4712.4 | 90.72 | 100.80 | 4712.4 | 90.72 | 100.80 | 4712.4 | 90.72 | 100.80 | |
| 24 | M-HCl | 33492.6 | 360.58 | 100.16 | 33399.6 | 359.55 | 99.88 | 33362.4 | 359.14 | 99.76 |
| HNK | 4704.8 | 90.57 | 100.64 | 4694.1 | 90.37 | 100.41 | 4687.6 | 90.24 | 100.27 | |
| 48 | M-HCl | 33396.1 | 359.51 | 99.86 | 33287.5 | 358.31 | 99.53 | 33215.8 | 357.52 | 99.31 |
| HNK | 4698.5 | 90.45 | 100.50 | 4674.6 | 89.98 | 99.98 | 4664.2 | 89.78 | 99.76 | |
| 1200 / 300 µg/mL (M-HCl / HNK) | ||||||||||
| 0 | M-HCl | 55005.7 | 1197.19 | 99.77 | 55005.7 | 1197.19 | 99.77 | 55005.7 | 1197.19 | 99.77 |
| HNK | 7772.2 | 300.97 | 100.32 | 7772.2 | 300.97 | 100.32 | 7772.2 | 300.97 | 100.32 | |
| 24 | M-HCl | 54941.9 | 1195.78 | 99.65 | 54860.8 | 1193.98 | 99.50 | 54782.5 | 1192.25 | 99.35 |
| HNK | 7766.1 | 300.74 | 100.25 | 7742.7 | 299.82 | 99.94 | 7717.8 | 298.85 | 99.62 | |
| 48 | M-HCl | 54812.3 | 1192.91 | 99.41 | 54603.4 | 1188.29 | 99.02 | 54386.2 | 1183.48 | 98.62 |
| HNK | 7748.6 | 300.05 | 100.02 | 7717.9 | 298.85 | 99.62 | 7691.4 | 297.82 | 99.27 | |
| Time (hours) |
M-HCl / HNK |
Room temperature (20 – 25 °C) | 37 °C | ||||
|---|---|---|---|---|---|---|---|
| Peak area | CC (µg/mL) |
R (%) | Peak area | CC (µg/mL) |
R (%) | ||
| 1200 / 300 µg/mL (M-HCl / HNK), unprotected for light | |||||||
| 0 | M-HCl | 55098.6 | 1199.25 | 99.94 | 55098.6 | 1199.25 | 99.94 |
| HNK | 7760.3 | 300.51 | 100.17 | 7760.3 | 300.51 | 100.17 | |
| 24 | M-HCl | 54930.5 | 1195.53 | 99.63 | 54846.3 | 1193.66 | 99.47 |
| HNK | 7721.2 | 298.98 | 99.66 | 7695.3 | 297.97 | 99.32 | |
| 48 | M-HCl | 54747.4 | 1191.48 | 99.29 | 54537.9 | 1186.84 | 98.90 |
| HNK | 7686.9 | 297.64 | 99.21 | 7642.8 | 295.92 | 98.64 | |
| 1200 / 300 µg/mL (M-HCl / HNK), protected for light | |||||||
| 0 | M-HCl | 55098.6 | 1199.25 | 99.94 | 55098.6 | 1199.25 | 99.94 |
| HNK | 7760.3 | 300.51 | 100.17 | 7760.3 | 300.51 | 100.17 | |
| 24 | M-HCl | 54922.5 | 1195.35 | 99.61 | 54784.8 | 1192.30 | 99.36 |
| HNK | 7746.1 | 299.95 | 99.98 | 7707.5 | 298.45 | 99.48 | |
| 48 | M-HCl | 54777.4 | 1192.14 | 99.34 | 54561.1 | 1187.35 | 98.95 |
| HNK | 7718.6 | 298.88 | 99.63 | 7687.2 | 297.65 | 99.22 | |
| Pharmaceutical product | Pharmaceutical form | Country manufacturer | A (mg) |
Mm (g) |
a (g) |
PA | C (%) |
C1 (%) |
|---|---|---|---|---|---|---|---|---|
| CP – 1 | prolonged-release tablet | France | 500 | 0.7270 | 0.2938 | 36925.8 | 98.62 | 98.59 |
| CP – 2 | tablet | Romania | 850 | 1.0146 | 0.2388 | 37033.9 | 99.91 | 99.20 |
| CP – 3 | film-coated tablet | France | 1000 | 1.1238 | 0.2282 | 36976.7 | 98.28 | 97.78 |
| CP – 4 | film-coated tablet | Germany | 1000 | 1.1065 | 0.2243 | 37093.1 | 98.76 | 98.48 |
| CP – 5 | prolonged-release tablet | France | 1000 | 1.4549 | 0.2931 | 37056.1 | 99.28 | 98.95 |
| Pharmaceutical product | Pharmaceutical form | Country manufacturer | A (mg) |
Mm (g) |
a (g) |
PA | C (%) |
|---|---|---|---|---|---|---|---|
| HNK – 1 | capsules | UK | 150 | 0.1471 | 0.1002 | 5083.6 | 97.84 |
| HNK – 2 | capsules | Romania | 150 | 0.1513 | 0.1000 | 5048.5 | 100.14 |
| HNK – 3 | capsules | EU | 250 | 0.2445 | 0.1004 | 5099.9 | 97.70 |
| HNK – 4 | capsules | Romania | 250 | 0.2497 | 0.1002 | 5104.2 | 100.07 |
| Pharmaceutical product | Pharmaceutical form | A (mg) |
Mm (g) |
a (g) |
PA | C (%) |
|---|---|---|---|---|---|---|
| P – 1 | prolonged-release tablet | 400 (M-HCl) 100 (HNK) |
0.8126 | 0.8040 | 36567.1 (M-HCl) 5069.2 (HNK) |
99.62 (M-HCl) 100.63 (HNK) |
| P – 2 | prolonged-release tablet | 400 (M-HCl) 100 (HNK) |
0.8098 | 0.8000 | 37004.3 (M-HCl) 5043.4 (HNK) |
100.99 (M-HCl) 100.26 (HNK) |
| Development method | Number of pictograms* | Hazard** | Penalty points (PP)*** |
|---|---|---|---|
| REAGENTS | |||
| Methanol | 3 | 2 | 6 |
| Acetic acid (glacial) | 2 | 2 | 4 |
| SAMPLE PREPARATION | |||
| Dilution with ethanol:water (2:1) | 3 | 2 | 6 |
| INSTRUMENTS | |||
| Liquid chromatography (LC) | ≤ 1.5 kWh per sample | 1 | |
| UV-VIS detection | < 0.1 kWh per sample | 0 | |
| Ultrasound | < 0.1 kWh per sample | 0 | |
| WASTE | |||
| 1-10 mL (g) waste / sample | 3 | ||
| No treatment waste | 3 | ||
| Occupational hazard | Analytical process hermetization | - | |
| Total penalty points | 23 | ||
| Analytical Eco-Scale Score | 77 | ||
| Principle AGREE | Observations |
|---|---|
| Principle 1 | Sampling procedure: remote sensing without sample damage |
| Principle 2 | Sample size: 0.1 mL |
| Principle 3 | Analytical device positioning: At-line |
| Principle 4 | No of major steps: 3 or fewer |
| Principle 5 | Automated and miniaturized method: manual, none or miniaturized |
| Principle 6 | Derivatization: none |
| Principle 7 | Waste generated and proper management: 10 mL |
| Principle 8 | Number of analytes/run and samples analysed per hour: 2 and 6 |
| Principle 9 | Most energy – intensive technique used: LC (HPLC) |
| Principle 10 | Source of reagents: none of the reagents are from bio-based sources |
| Principle 11 | Toxic reagents / solvents: Yes (methanol) / 8.5 mL per run |
| Principle 12 | Operator safety: none |
| Run | Run order | Coded factors level | ||
|---|---|---|---|---|
| Factor 1 | Factor 2 | Factor 3 | ||
| 1 | 1 | -1 | -1 | -1 |
| 2 | 13 | 0 | -1 | -1 |
| 3 | 3 | +1 | -1 | -1 |
| 4 | 5 | -1 | -1 | 0 |
| 5 | 17 | 0 | -1 | 0 |
| 6 | 7 | +1 | -1 | 0 |
| 7 | 9 | -1 | -1 | +1 |
| 8 | 15 | 0 | -1 | +1 |
| 9 | 11 | +1 | -1 | +1 |
| 10 | 21 | -1 | 0 | -1 |
| 11 | 25 | 0 | 0 | -1 |
| 12 | 23 | +1 | 0 | -1 |
| 13 | 20 | -1 | 0 | 0 |
| 14 | 27 | 0 | 0 | 0 |
| 15 | 18 | +1 | 0 | 0 |
| 16 | 22 | -1 | 0 | +1 |
| 17 | 26 | 0 | 0 | +1 |
| 18 | 24 | +1 | 0 | +1 |
| 19 | 2 | -1 | +1 | -1 |
| 20 | 14 | 0 | +1 | -1 |
| 21 | 4 | +1 | +1 | -1 |
| 22 | 6 | -1 | +1 | 0 |
| 23 | 19 | 0 | +1 | 0 |
| 24 | 8 | +1 | +1 | 0 |
| 25 | 10 | -1 | +1 | +1 |
| 26 | 16 | 0 | +1 | +1 |
| 27 | 12 | +1 | +1 | +1 |
| Level of factor | ||||
| Parameter | Low (–1) | Intermediate (0) | High (+1) | |
| A: % Methanol | 85 | 87.5 | 90 | |
| B: pH | 2 | 3 | 4 | |
| C: Flow (mL/min) | 1 | 1.25 | 1.5 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).