Submitted:
04 July 2024
Posted:
04 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
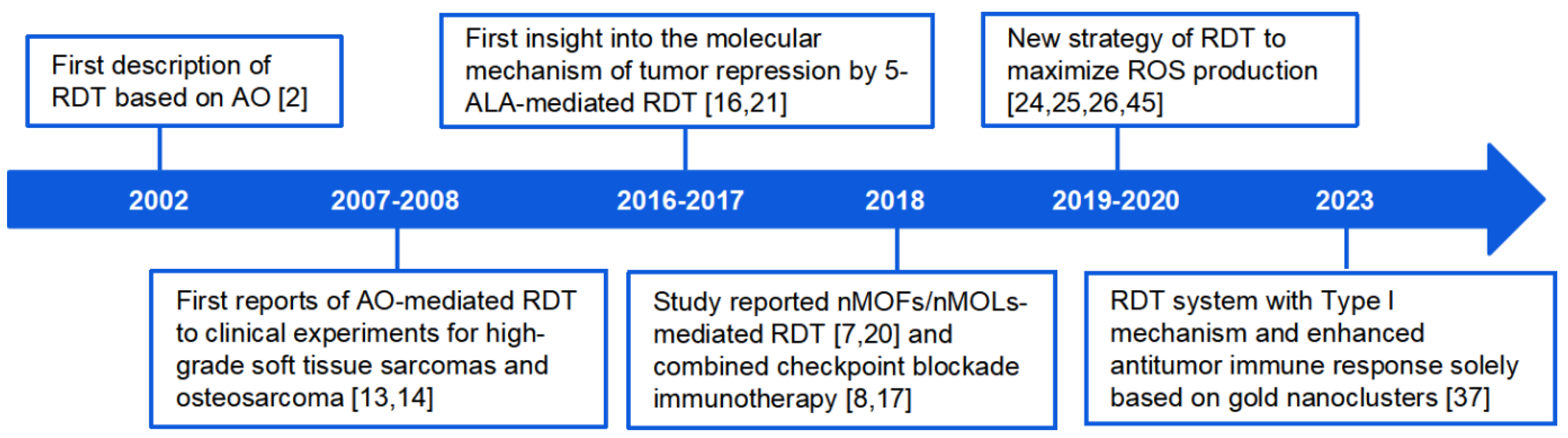
2. Radio-Dynamic Therapy (RDT) and Its Problems
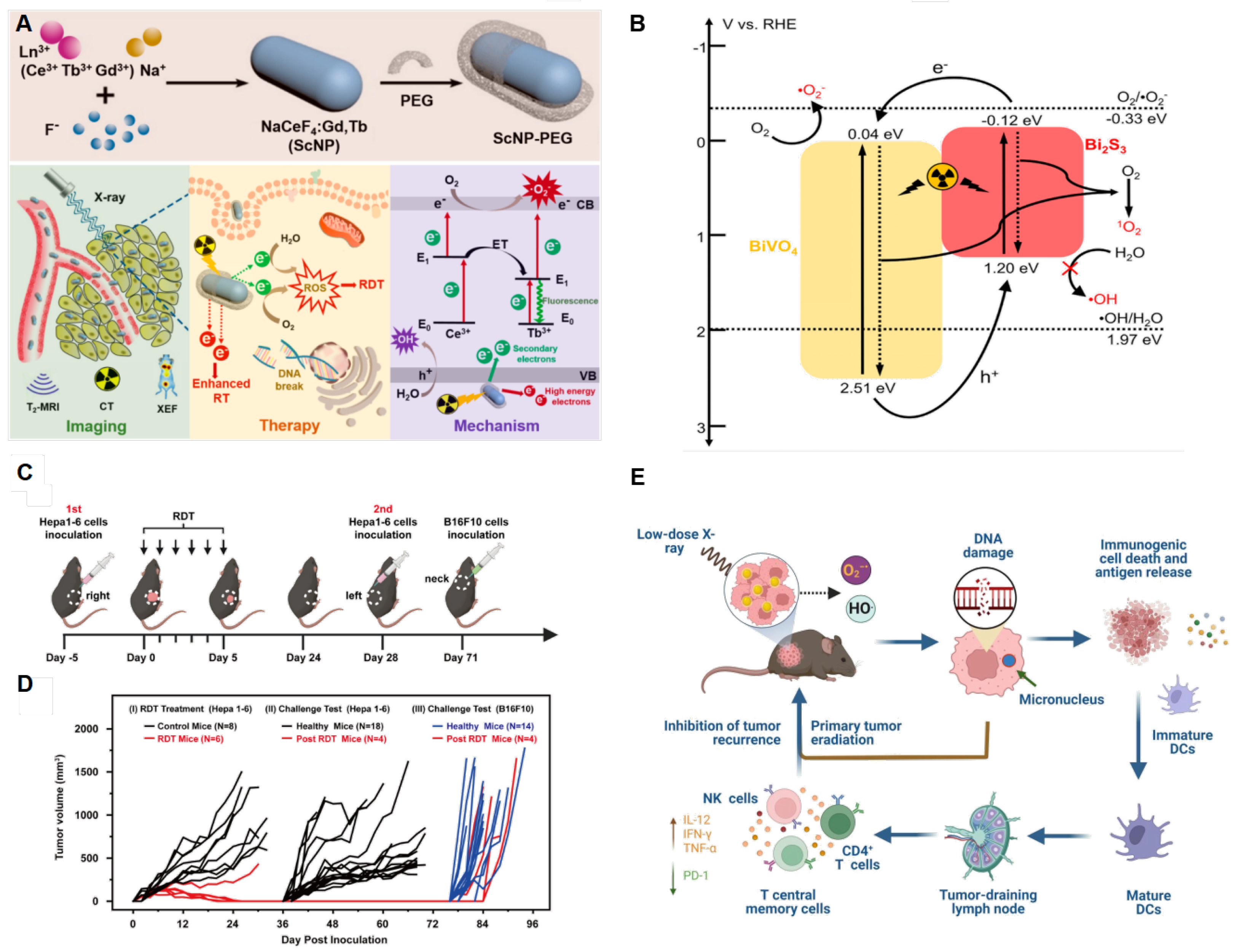
3. Innovative Single-Component RDT Systems
4. RDT Mechanisms
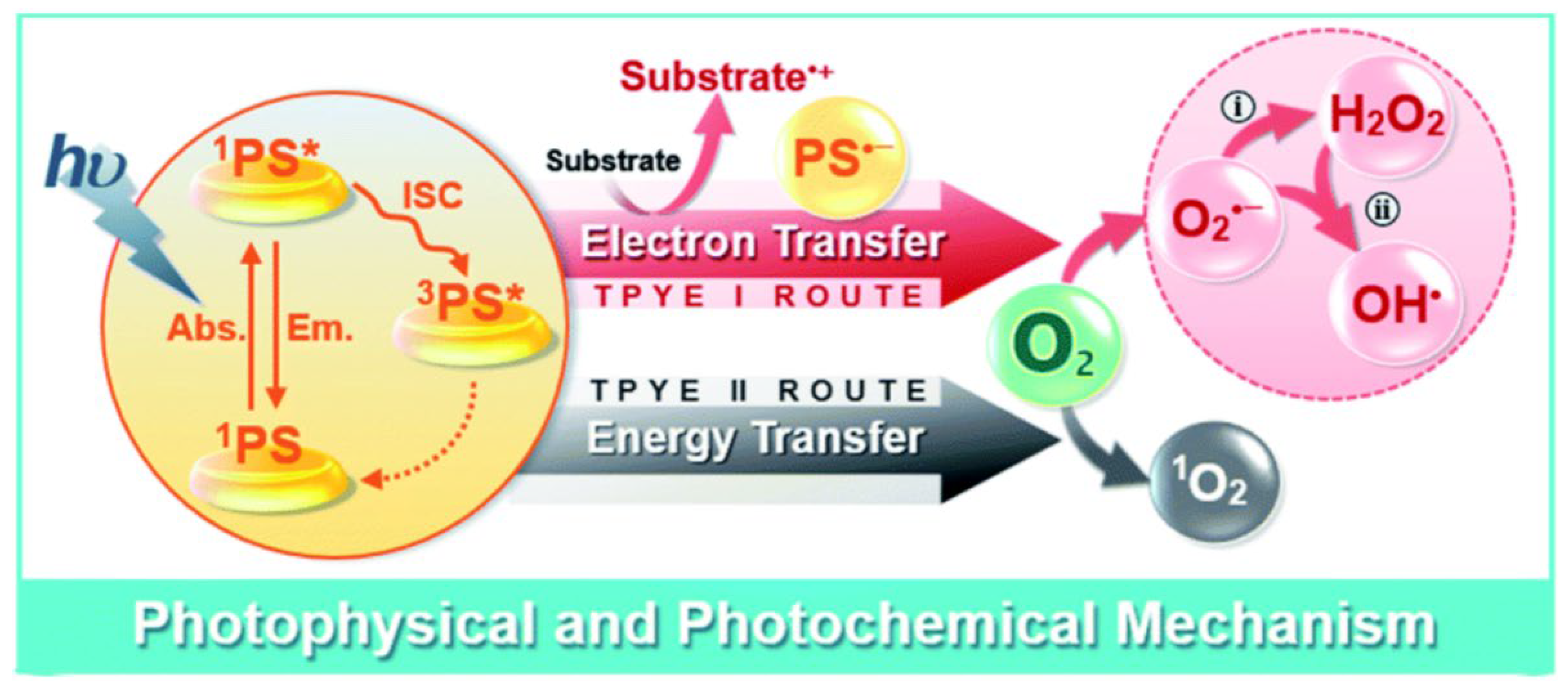
4.1. ROS Generation in RDT
4.2. The Biomedical Mechanism behind RDT
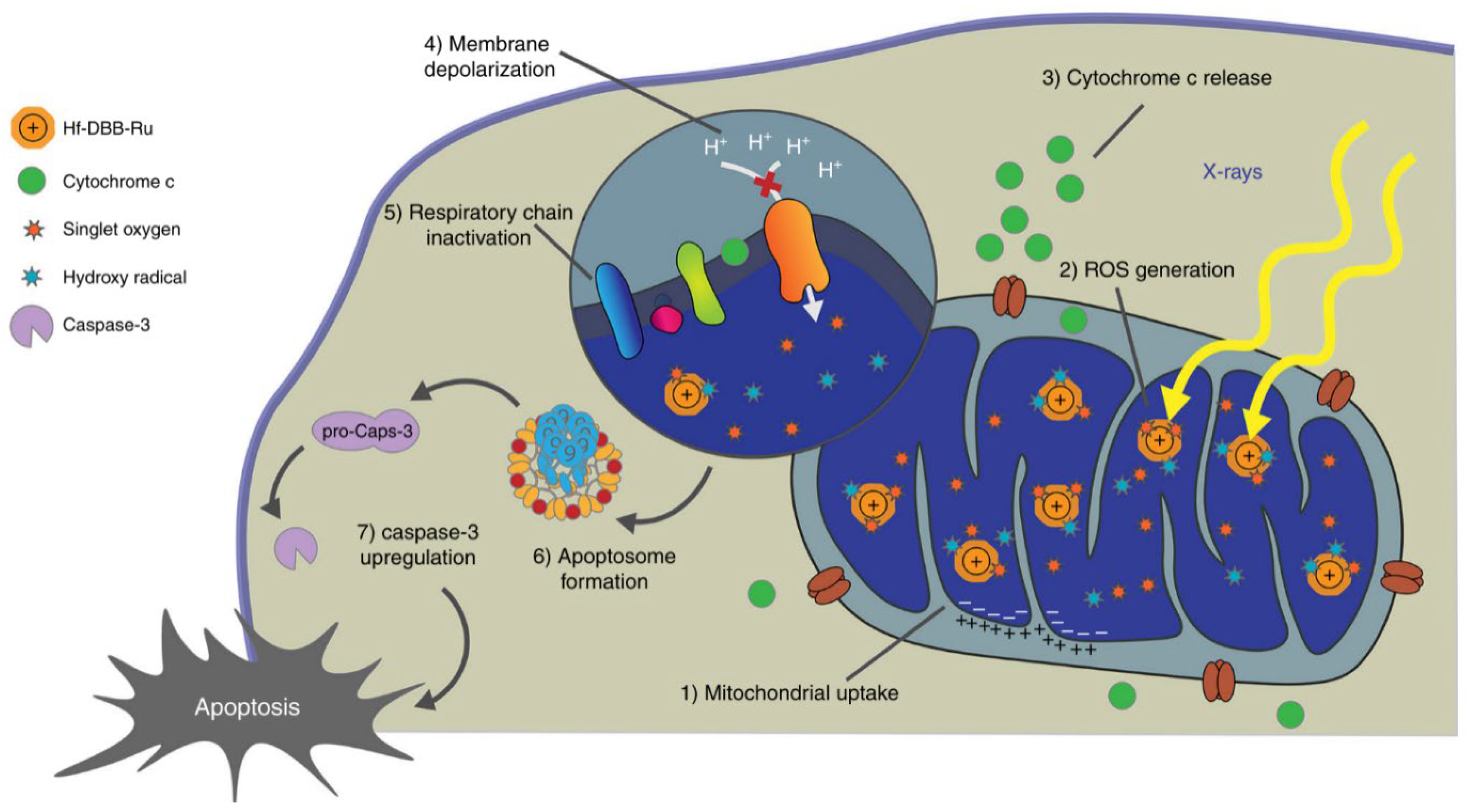
4.2.1. The Cytotoxic Effect of ROS
4.2.2. The Disturbance of Cell Cycle
4.2.3. The Effect of Directly Killing Tumor Cells
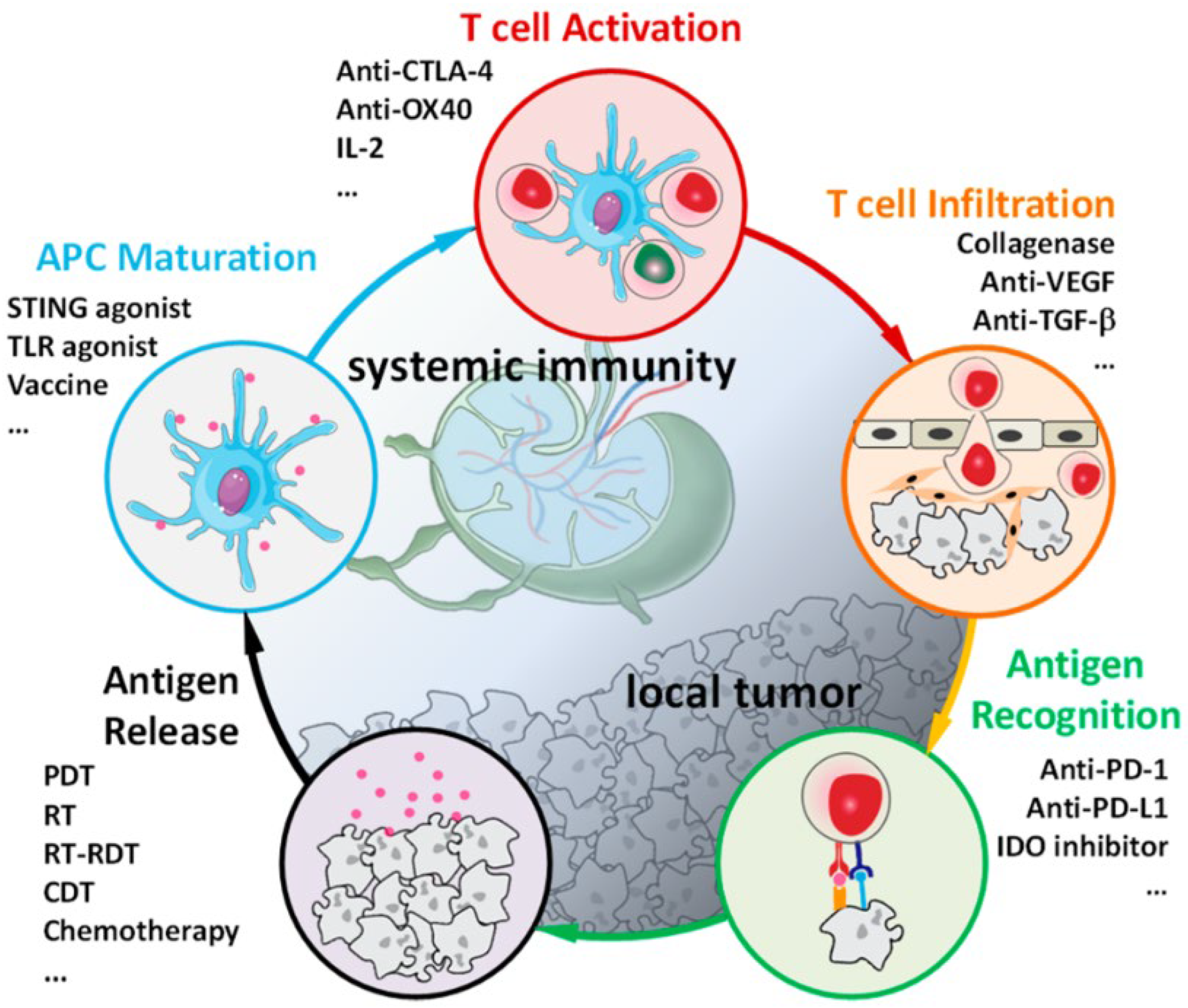
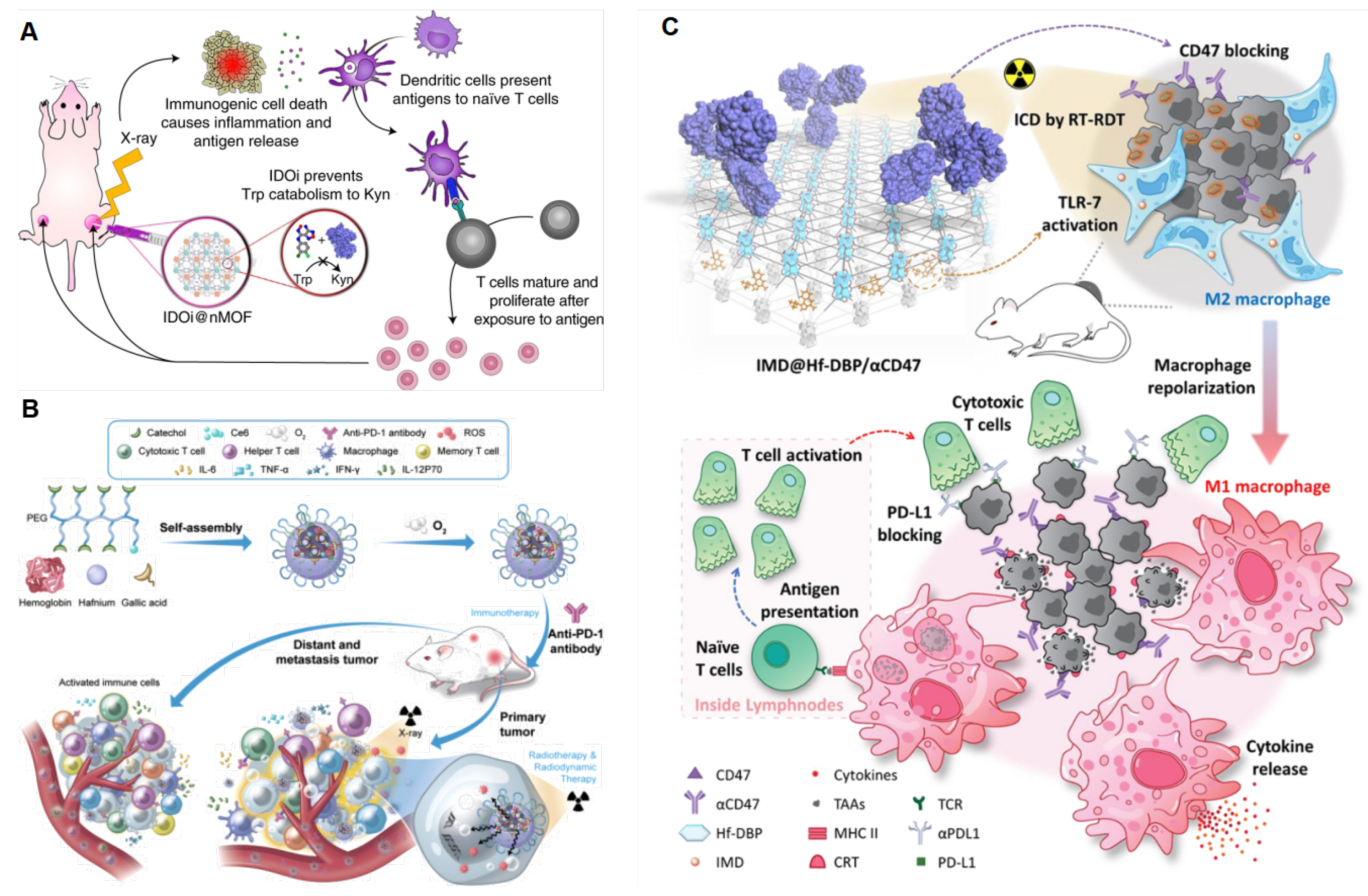
4.2.4. Activation of the Antitumor Immune Response
4.2.5. Activation of the Antitumor Immune Response
5. Combination Therapy
6. Conclusions
Author Contributions
Funding Information
Conflicts of Interest
References
- Dolmans, D.E.J.G.J.; Fukumura, D.; Jain, R.K. Photodynamic therapy for cancer. Nature Reviews Cancer. 2003, 3, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Hashiguchi, S.; Katsuyuki, K.; Murata, H.; et al. Acridine Orange Excited by Low-Dose Radiation Has a Strong Cytocidal Effect on Mouse Osteosarcoma. Oncology. 2002, 62, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, T.; Takahashi, J.; Nagasawa, S.; Doi, M.; Moriyama, A.; Iwahashi, H. DNA Strand Break Properties of Protoporphyrin IX by X-ray Irradiation against Melanoma. International Journal of Molecular Sciences. 2020, 21, 2302. [Google Scholar] [CrossRef] [PubMed]
- Kamkaew, A.; Chen, F.; Zhan, Y.; Majewski, R.L.; Cai, W. Scintillating Nanoparticles as Energy Mediators for Enhanced Photodynamic Therapy. ACS Nano. 2016, 10, 3918–3935. [Google Scholar] [CrossRef] [PubMed]
- Ni, K.; Lan, G.; Lin, W. Nanoscale Metal–Organic Frameworks Generate Reactive Oxygen Species for Cancer Therapy. ACS central science. 2020, 6, 861–868. [Google Scholar] [CrossRef]
- Ni, K.; Luo, T.; Nash, G.T.; Lin, W. Nanoscale Metal–Organic Frameworks for Cancer Immunotherapy. Accounts of Chemical Research. 2020, 53, 1739–1748. [Google Scholar] [CrossRef]
- Ni, K.; Lan, G.; Veroneau, S.S.; Duan, X.; Song, Y.; Lin, W. Nanoscale metal-organic frameworks for mitochondria-targeted radiotherapy-radiodynamic therapy. Nature Communications. 2018, 9, 4321. [Google Scholar] [CrossRef] [PubMed]
- Ni, K.; Lan, G.; Chan, C.; et al. Nanoscale metal-organic frameworks enhance radiotherapy to potentiate checkpoint blockade immunotherapy. Nature Communications. 2018, 9, 2351. [Google Scholar] [CrossRef]
- Sun, W.; Luo, L.; Feng, Y.; et al. Aggregation-Induced Emission Gold Clustoluminogens for Enhanced Low-Dose X-ray-Induced Photodynamic Therapy. Angewandte Chemie International Edition. 2020, 59, 9914–9921. [Google Scholar] [CrossRef]
- Liu, T.; Yang, K.; Liu, Z. Recent advances in functional nanomaterials for X-ray triggered cancer therapy. Progress in Natural Science: Materials International. 2020, 30, 567–576. [Google Scholar] [CrossRef]
- Wang, G.D.; Nguyen, H.T.; Chen, H.; et al. X-Ray Induced Photodynamic Therapy: A Combination of Radiotherapy and Photodynamic Therapy. Theranostics. 2016, 6, 2295–2305. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, A.L.; Harmon, A.C.; et al. 177Lu-Labeled Eu-Doped Mesoporous SiO2 Nanoparticles as a Theranostic Radiopharmaceutical for Colorectal Cancer. ACS applied nano materials. 2020, 3, 8691–8701. [Google Scholar] [CrossRef]
- Kusuzaki, K.; Murata, H.; Matsubara, T.; et al. Acridine orange could be an innovative anticancer agent under photon energy. PubMed. 2007, 21, 205–214. [Google Scholar]
- Kusuzaki, K.; Murata, H.; Matsubara, T.; et al. Review. Acridine orange could be an innovative anticancer agent under photon energy. PubMed. 2007, 21, 205–214. [Google Scholar]
- Nakamura, T.; Kusuzaki, K.; Matsubara, T.; Matsumine, A.; Murata, H.; Uchida, A. A new limb salvage surgery in cases of high-grade soft tissue sarcoma using photodynamic surgery, followed by photo- and radiodynamic therapy with acridine orange. Journal of Surgical Oncology. 2008, 97, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Kusuzaki, K.; Matsubara, T.; et al. Long-term clinical outcome in patients with high-grade soft tissue sarcoma who were treated with surgical adjuvant therapy using acridine orange after intra-lesional or marginal resection. Photodiagnosis and Photodynamic Therapy. 2018, 23, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, J.; Misawa, M.; Iwahashi, H. Combined treatment with X-ray irradiation and 5-aminolevulinic acid elicits better transcriptomic response of cell cycle-related factors than X-ray irradiation alone. International journal of radiation biology. 2016, 92, 774–789. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; He, C.; Guo, N.; et al. Low-dose X-ray radiotherapy–radiodynamic therapy via nanoscale metal–organic frameworks enhances checkpoint blockade immunotherapy. Nature Biomedical Engineering. 2018, 2, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Ni, K.; Luo, T.; Culbert, A.; Kaufmann, M.; Jiang, X.; Lin, W. Nanoscale Metal–Organic Framework Co-delivers TLR-7 Agonists and Anti-CD47 Antibodies to Modulate Macrophages and Orchestrate Cancer Immunotherapy. Journal of the American Chemical Society. 2020, 142, 12579–12584. [Google Scholar] [CrossRef]
- Lan, G.; Ni, K.; Xu, R.; et al. Nanoscale Metal-Organic Layers for Deeply Penetrating X-ray-Induced Photodynamic Therapy. Angewandte Chemie. 2017, 129, 12270–12274. [Google Scholar] [CrossRef]
- Lan, G.; Ni, K.; Veroneau, S.S.; Song, Y.; Lin, W. Nanoscale Metal–Organic Layers for Radiotherapy–Radiodynamic Therapy. Journal of the American Chemical Society. 2018, 140, 16971–16975. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, J.; Iwahashi, H. Introduction to 5-Aminolevulinic Acid-Protoporphyrin IXMediated Radiodynamic Therapy (RDT). Clinics in oncology. 2017, 2, 1330. [Google Scholar]
- Takahashi, J.; Murakami, M.; Mori, T.; Iwahashi, H. Verification of radiodynamic therapy by medical linear accelerator using a mouse melanoma tumor model. Scientific Reports. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, J.; Nagasawa, S.; Ikemoto, M.J.; Sato, C.; Sato, M.; Iwahashi, H. Verification of 5-Aminolevurinic Radiodynamic Therapy Using a Murine Melanoma Brain Metastasis Model. International Journal of Molecular Sciences. 2019, 20, 5155. [Google Scholar] [CrossRef]
- Zhong, X.; Wang, X.; Zhan, G.; et al. NaCeF4:Gd,Tb Scintillator as an X-ray Responsive Photosensitizer for Multimodal Imaging-Guided Synchronous Radio/Radiodynamic Therapy. Nano Letters. 2019, 19, 8234–8244. [Google Scholar] [CrossRef] [PubMed]
- Ni, K.; Lan, G.; Song, Y.; Hao, Z.; Lin, W. Biomimetic nanoscale metal–organic framework harnesses hypoxia for effective cancer radiotherapy and immunotherapy. Chemical science. 2020, 11, 7641–7653. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liu, S.; Wang, L.; et al. BiVO4@Bi2S3 Heterojunction Nanorods with Enhanced Charge Separation Efficiency for Multimodal Imaging and Synergy Therapy of Tumor. ACS applied bio materials. 2020, 3, 5080–5092. [Google Scholar] [CrossRef]
- Han, R.; Zhao, M.; Wang, Z.; et al. Super-efficient in Vivo Two-Photon Photodynamic Therapy with a Gold Nanocluster as a Type I Photosensitizer. ACS nano. 2019, 14, 9532–9544. [Google Scholar] [CrossRef]
- Zhuang, Z.; Dai, J.; Yu, M.; et al. Type I photosensitizers based on phosphindole oxide for photodynamic therapy: Apoptosis and autophagy induced by endoplasmic reticulum stress. Chemical Science. 2020, 11, 3405–3417. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Hu, Y.; Du, H.; Wang, H. Intracellular gold nanoparticle aggregation and their potential applications in photodynamic therapy. Chemical Communications. 2014, 50, 7287. [Google Scholar] [CrossRef]
- Hu, J.; Tang, Y.; Elmenoufy, A.H.; Xu, H.; Cheng, Z.; Yang, X. Nanocomposite-Based Photodynamic Therapy Strategies for Deep Tumor Treatment. Small. 2015, 11, 5860–5887. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.D.; Luo, Z.; Chen, J.; et al. Ultrasmall Au10−12(SG)10−12Nanomolecules for High Tumor Specificity and Cancer Radiotherapy. Advanced Materials. 2014, 26, 4565–4568. [Google Scholar] [CrossRef] [PubMed]
- Amirhosein Kefayat, Ghahremani, F.; Hasan Motaghi, Alireza Amouheidari. Ultra-small but ultra-effective: Folic acid-targeted gold nanoclusters for enhancement of intracranial glioma tumors’ radiation therapy efficacy. Nanomedicine 2019, 16, 173–184. [CrossRef] [PubMed]
- Jia, T.T.; Yang, G.; Mo, S.J.; et al. Atomically Precise Gold–Levonorgestrel Nanocluster as a Radiosensitizer for Enhanced Cancer Therapy. ACS Nano. 2019, 13, 8320–8328. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Wang, X.; Zeng, S.; Ramamurthy, G.; Burda, C.; Basilion, J.P. Targeted Gold Nanocluster-Enhanced Radiotherapy of Prostate Cancer. Small. 2019, 15, 1900968. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, X.; Jiang, Y.; et al. Glutathione-Depleting Gold Nanoclusters for Enhanced Cancer Radiotherapy through Synergistic External and Internal Regulations. ACS Applied Materials & Interfaces. 2018, 10, 10601–10606. [Google Scholar] [CrossRef]
- Zhang, X.D.; Chen, J.; Luo, Z.; et al. Enhanced Tumor Accumulation of Sub-2 nm Gold Nanoclusters for Cancer Radiation Therapy. Advanced Healthcare Materials. 2013, 3, 133–141. [Google Scholar] [CrossRef]
- Zhu, S.; Yan, F.; Yang, L.; et al. Low-dose X-ray radiodynamic therapy solely based on gold nanoclusters for efficient treatment of deep hypoxic solid tumors combined with enhanced antitumor immune response. Theranostics. 2023, 13, 1042–1058. [Google Scholar] [CrossRef]
- Yang, B.; Chen, Y.; Shi, J. Reactive Oxygen Species (ROS)-Based Nanomedicine. Chemical Reviews. 2019, 119, 4881–4985. [Google Scholar] [CrossRef]
- D’Autréaux, B.; Toledano, M.B. ROS as signalling molecules: Mechanisms that generate specificity in ROS homeostasis. Nature Reviews Molecular Cell Biology. 2007, 8, 813–824. [Google Scholar] [CrossRef]
- Nakayama, M.; Sasaki, R.; Ogino, C.; et al. Titanium peroxide nanoparticles enhanced cytotoxic effects of X-ray irradiation against pancreatic cancer model through reactive oxygen species generation in vitro and in vivo. Radiation Oncology. 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Price, M.; Reiners, J.J.; Santiago, A.M.; Kessel, D. Monitoring Singlet Oxygen and Hydroxyl Radical Formation with Fluorescent Probes During Photodynamic Therapy. Photochemistry and Photobiology. 2009, 85, 1177–1181. [Google Scholar] [CrossRef]
- Wang, J.; Guo, Y.; Liu, B.; et al. Detection and analysis of reactive oxygen species (ROS) generated by nano-sized TiO2 powder under ultrasonic irradiation and application in sonocatalytic degradation of organic dyes. Ultrasonics Sonochemistry. 2011, 18, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Misawa, M.; Takahashi, J. Generation of reactive oxygen species induced by gold nanoparticles under x-ray and UV Irradiations. Nanomedicine: Nanotechnology, Biology and Medicine. 2011, 7, 604–614. [Google Scholar] [CrossRef]
- Zhang, Q.; Xiao, G.; Sun, Q.; et al. Investigation of the Mechanisms of Radio-Dynamic Therapy. Mathews Journal of Cancer Science. 2020, 5, 10024. [Google Scholar] [CrossRef]
- Fan, W.; Lu, N.; Shen, Z.; et al. Generic synthesis of small-sized hollow mesoporous organosilica nanoparticles for oxygen-independent X-ray-activated synergistic therapy. Nature Communications. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Winterbourn, C.C. Reconciling the chemistry and biology of reactive oxygen species. Nature Chemical Biology. 2008, 4, 278–286. [Google Scholar] [CrossRef]
- Tew, L.S.; Cai, M.T.; Lo, L.W.; Khung, Y.L.; Chen, N.T. Pollen-Structured Gold Nanoclusters for X-ray Induced Photodynamic Therapy. Materials. 2018, 11, 1170. [Google Scholar] [CrossRef]
- Ghelli Luserna di Rora’, A.; Iacobucci, I.; Martinelli, G. The cell cycle checkpoint inhibitors in the treatment of leukemias. Journal of Hematology & Oncology. 2017, 10, 1–14. [Google Scholar] [CrossRef]
- Duronio, R.J.; Xiong, Y. Signaling Pathways that Control Cell Proliferation. Cold Spring Harbor Perspectives in Biology. 2013, 5, a008904. [Google Scholar] [CrossRef]
- Rhind, N.; Russell, P. Signaling Pathways that Regulate Cell Division. Cold Spring Harbor Perspectives in Biology. 2012, 4, a005942. [Google Scholar] [CrossRef] [PubMed]
- Dickens, L.S.; Powley, I.R.; Hughes, M.A.; MacFarlane, M. The “complexities” of life and death: Death receptor signalling platforms. Experimental Cell Research. 2012, 318, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Favaloro, B.; Allocati, N.; Graziano, V.; Di Ilio, C.; De Laurenzi, V. Role of Apoptosis in disease. Aging. 2012, 4, 330–349. [Google Scholar] [CrossRef] [PubMed]
- McIlwain, D.R.; Berger, T.; Mak, T.W. Caspase functions in cell death and disease. Cold Spring Harbor perspectives in biology. 2013, 5, a008656. [Google Scholar] [CrossRef] [PubMed]
- Shamas-Din, A.; Kale, J.; Leber, B.; Andrews, D.W. Mechanisms of Action of Bcl-2 Family Proteins. Cold Spring Harbor Perspectives in Biology. 2013, 5, a008714. [Google Scholar] [CrossRef] [PubMed]
- Gorbet, M.J.; Ranjan, A. Cancer immunotherapy with immunoadjuvants, nanoparticles, and checkpoint inhibitors: Recent progress and challenges in treatment and tracking response to immunotherapy. Pharmacology & Therapeutics 2020, 207, 107456. [Google Scholar] [CrossRef]
- Kuwahara, Y.; Tomita, K.; Urushihara, Y.; Sato, T.; Kurimasa, A.; Fukumoto, M. Association between radiation-induced cell death and clinically relevant radioresistance. Histochemistry and cell biology. 2018, 150, 649–659. [Google Scholar] [CrossRef]
- D’Arcy, M.S. Cell death: A review of the major forms of apoptosis, necrosis and autophagy. Cell Biology International. 2019, 43, 582–592. [Google Scholar] [CrossRef]
- Martin, S.J.; Henry, C.M. Distinguishing between apoptosis, necrosis, necroptosis and other cell death modalities. Methods. 2013, 61, 87–89. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Y.; Xia, S.; et al. Gasdermin E suppresses tumor growth by activating anti-tumor immunity. Nature. 2020, 579, 415–420. [Google Scholar] [CrossRef]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; et al. Molecular mechanisms of cell death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death & Differentiation. 2018, 25, 486–541. [Google Scholar] [CrossRef]
- Riegman, M.; Sagie, L.; Galed, C.; et al. Ferroptosis occurs through an osmotic mechanism and propagates independently of cell rupture. Nature Cell Biology. 2020, 22, 1042–1048. [Google Scholar] [CrossRef] [PubMed]
- Ni, K.; Lan, G.; Chan, C.; et al. Ultrathin metal-organic layer-mediated radiotherapy-radiodynamic therapy enhances immunotherapy of metastatic cancers. PubMed. 2019, 1, 1331–1353. [Google Scholar]
- Formenti, S.C.; Demaria, S. Systemic effects of local radiotherapy. The Lancet Oncology. 2009, 10, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Ruiz, M.E.; Vanpouille-Box, C.; Melero, I.; Formenti, S.C.; Demaria, S. Immunological Mechanisms Responsible for Radiation-Induced Abscopal Effect. Trends in Immunology. 2018, 39, 644–655. [Google Scholar] [CrossRef]
- Abuodeh, Y.; Venkat, P.; Kim, S. Systematic review of case reports on the abscopal effect. Current Problems in Cancer. 2016, 40, 25–37. [Google Scholar] [CrossRef]
- Song, G.; Cheng, L.; Chao, Y.; Yang, K.; Liu, Z. Emerging Nanotechnology and Advanced Materials for Cancer Radiation Therapy. Advanced Materials. 2017, 29, 1700996. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Dai, M.; Liu, Q.; Xiu, R. Oxygen carriers and cancer chemo- and radiotherapy sensitization: Bench to bedside and back. Cancer Treatment Reviews. 2007, 33, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; Wilson, W.R. Exploiting tumour hypoxia in cancer treatment. Nature Reviews Cancer. 2004, 4, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Barker, H.E.; Paget, J.T.E.; Khan, A.A.; Harrington, K.J. The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence. Nature Reviews Cancer. 2015, 15, 409–425. [Google Scholar] [CrossRef]
- Sang, W.; Xie, L.; Wang, G.; et al. Oxygen-Enriched Metal-Phenolic X-Ray Nanoprocessor for Cancer Radio-Radiodynamic Therapy in Combination with Checkpoint Blockade Immunotherapy. Advanced Science. 2020, 8, 2003338. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kwon, N.; Guo, T.; Liu, Z.; Yoon, J. Innovative Strategies for Hypoxic-Tumor Photodynamic Therapy. Angewandte Chemie International Edition. 2018, 57, 11522–11531. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Yu, H.; Dong, Y.; et al. Photoactivation switch from type II to type I reactions by electron-rich micelles for improved photodynamic therapy of cancer cells under hypoxia. Journal of Controlled Release: Official Journal of the Controlled Release Society. 2011, 156, 276–280. [Google Scholar] [CrossRef] [PubMed]
- Gilson, R.C.; Black, K.C.L.; Lane, D.D.; Achilefu, S. Hybrid TiO2 -Ruthenium Nano-photosensitizer Synergistically Produces Reactive Oxygen Species in both Hypoxic and Normoxic Conditions. Angewandte Chemie (International Ed in English). 2017, 56, 10717–10720. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Zhang, J.; Wan, Y.; et al. Dual Fenton Catalytic Nanoreactor for Integrative Type-I and Type-II Photodynamic Therapy Against Hypoxic Cancer Cells. ACS applied bio materials. 2019, 2, 3854–3860. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, K.; Bu, W.; et al. Marriage of Scintillator and Semiconductor for Synchronous Radiotherapy and Deep Photodynamic Therapy with Diminished Oxygen Dependence. Angewandte Chemie. 2015, 54, 1770–1774. [Google Scholar] [CrossRef]
- Lv, Z.; Wei, H.; Li, Q.; et al. Achieving efficient photodynamic therapy under both normoxia and hypoxia using cyclometalated Ru(II) photosensitizer through type I photochemical process. Chemical Science. 2018, 9, 502–512. [Google Scholar] [CrossRef] [PubMed]
- Teng, K.; Chen, W.; Niu, L.; Fang, W.; Cui, G.; Yang, Q. BODIPY-Based Photodynamic Agents for Exclusively Generating Superoxide Radical over Singlet Oxygen. Angewandte Chemie. 2021, 60, 19912–19920. [Google Scholar] [CrossRef]
- Misra, H.P. Generation of Superoxide Free Radical during the Autoxidation of Thiols. Journal of Biological Chemistry. 1974, 249, 2151–2155. [Google Scholar] [CrossRef]
- Kaewmati, P.; Somsook, E.; Dhital, R.N.; Sakurai, H. Aerobic oxygenation of phenylboronic acid promoted by thiol derivatives under gold-free conditions: A warning against gold nanoparticle catalysis. Tetrahedron letters. 2012, 53, 6104–6106. [Google Scholar] [CrossRef]
- Ibrahim-Hashim, A.; Estrella, V. Acidosis and cancer: From mechanism to neutralization. Cancer and Metastasis Reviews 2019, 38, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.; Gordijo, C.R.; Abbasi, A.Z.; et al. Multifunctional Albumin–MnO2Nanoparticles Modulate Solid Tumor Microenvironment by Attenuating Hypoxia, Acidosis, Vascular Endothelial Growth Factor and Enhance Radiation Response. ACS Nano. 2014, 8, 3202–3212. [Google Scholar] [CrossRef] [PubMed]
- Maiti, D.; Zhong, J.; Zhang, Z.; et al. Polyoxomolybdate (POM) nanoclusters with radiosensitizing and scintillating properties for low dose X-ray inducible radiation-radiodynamic therapy. Nanoscale Horizons. 2020, 5, 109–118. [Google Scholar] [CrossRef]
- Chen, D.S.; Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature. 2017, 541, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Ngwa, W.; Irabor, O.C.; Schoenfeld, J.D.; Hesser, J.; Demaria, S.; Formenti, S.C. Using immunotherapy to boost the abscopal effect. Nature Reviews Cancer. 2018, 18, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Jiang, W.; Kim, B.Y.; Zhang, C.; Fu, Y.X.; Weissman, I.L. Phagocytosis checkpoints as new targets for cancer immunotherapy. Nature reviews Cancer. 2019, 19, 568–586. [Google Scholar] [CrossRef]
- Demaria, O.; Cornen, S.; Daëron, M.; Morel, Y.; Medzhitov, R.; Vivier, E. Harnessing innate immunity in cancer therapy. Nature. 2019, 574, 45–56. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



