Submitted:
01 July 2024
Posted:
02 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Synthetic Approaches with Glycerol as a Green Solvent
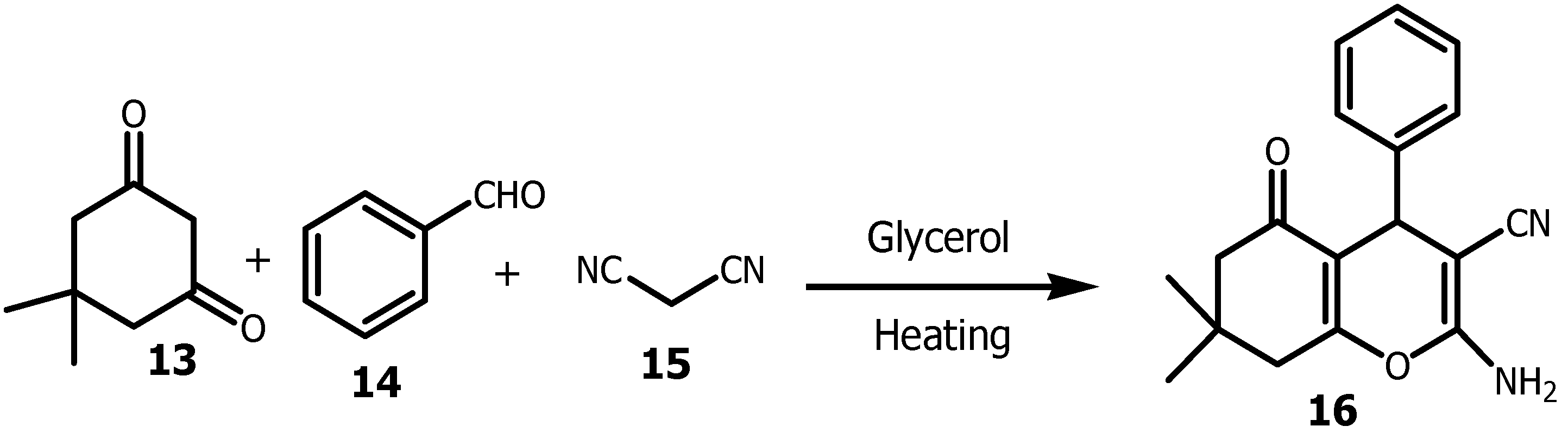
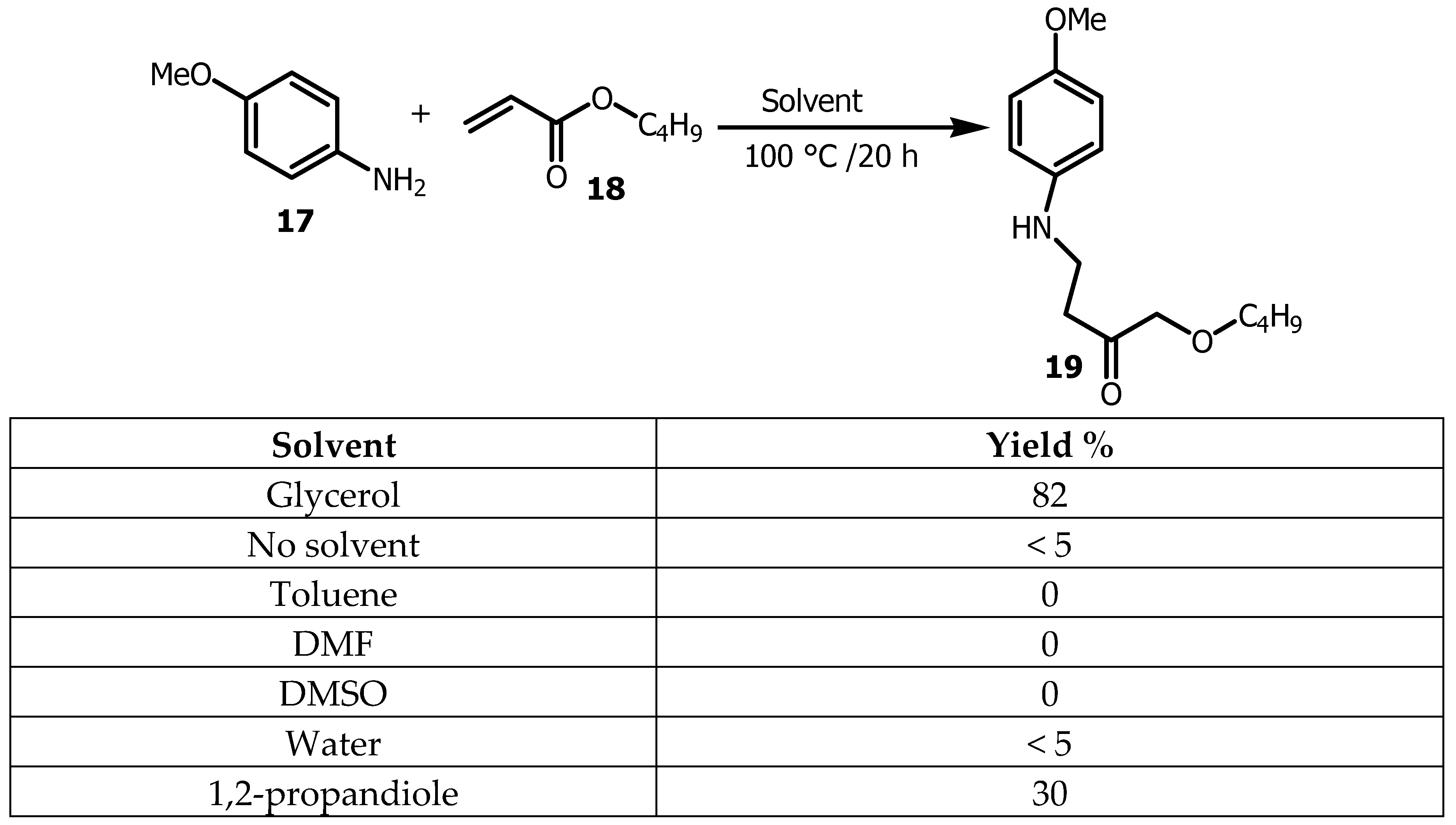
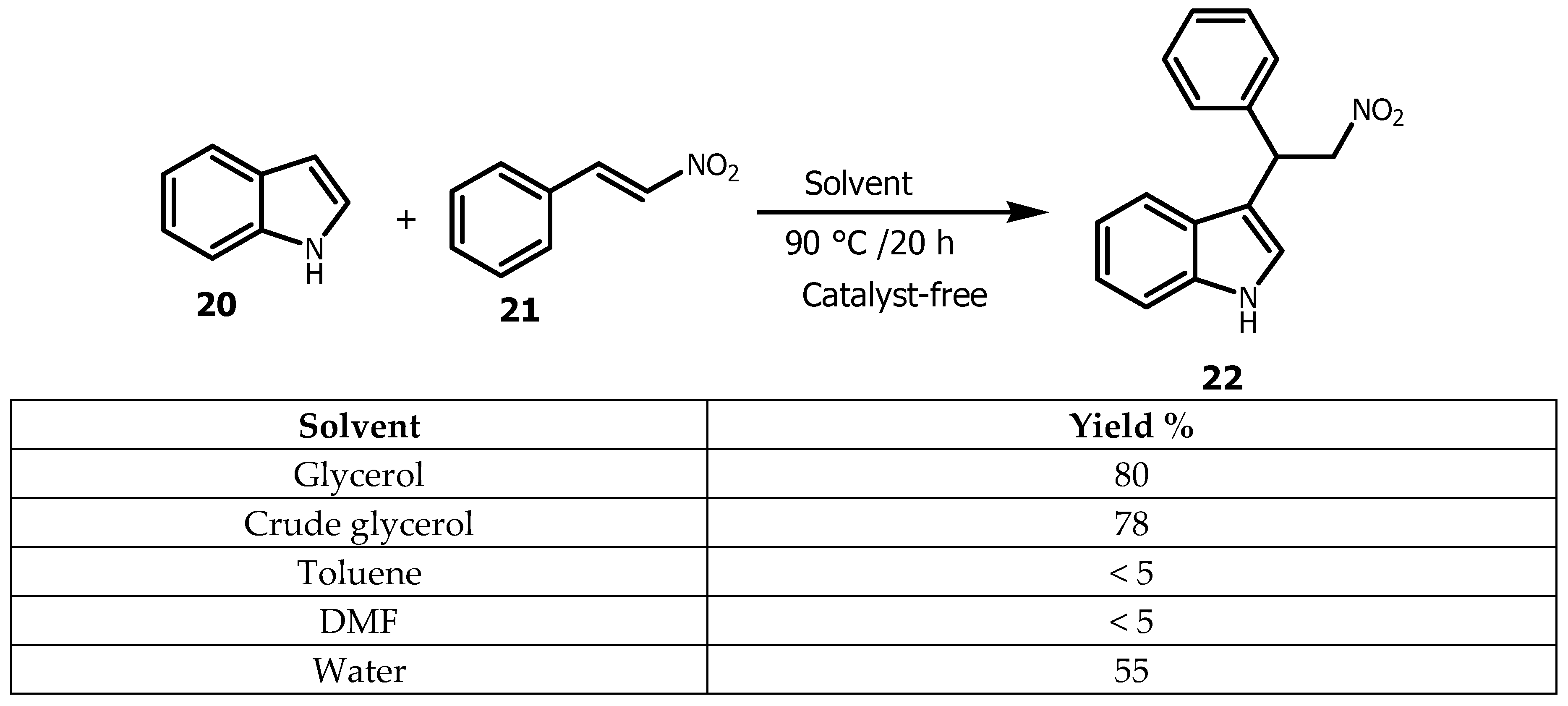
2.1. Glycerol as a Viable Solvent in the Synthesis of Bis (Aryl) Ketones and Aryl Compounds
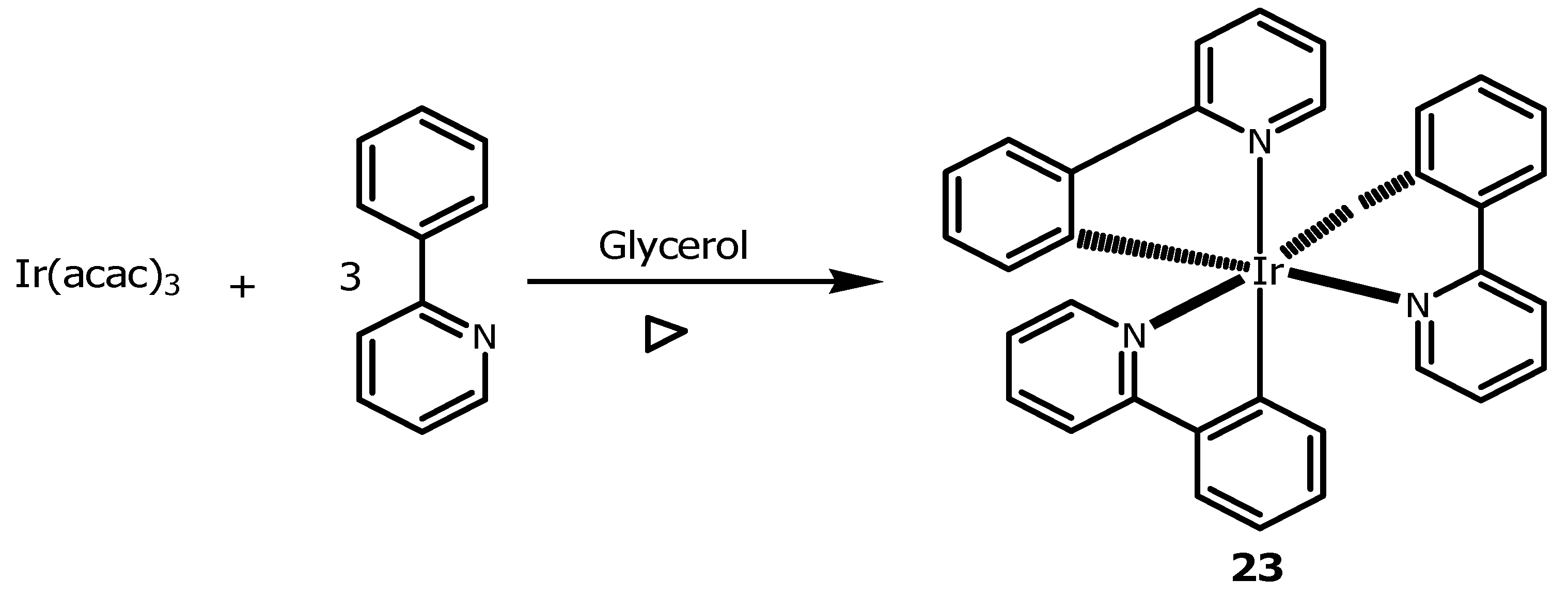
2.2. Glycerol as a Suitable Solvent in the Synthesis of Metallic Complexes
3. Glycerol as a key Synthetic Intermediate
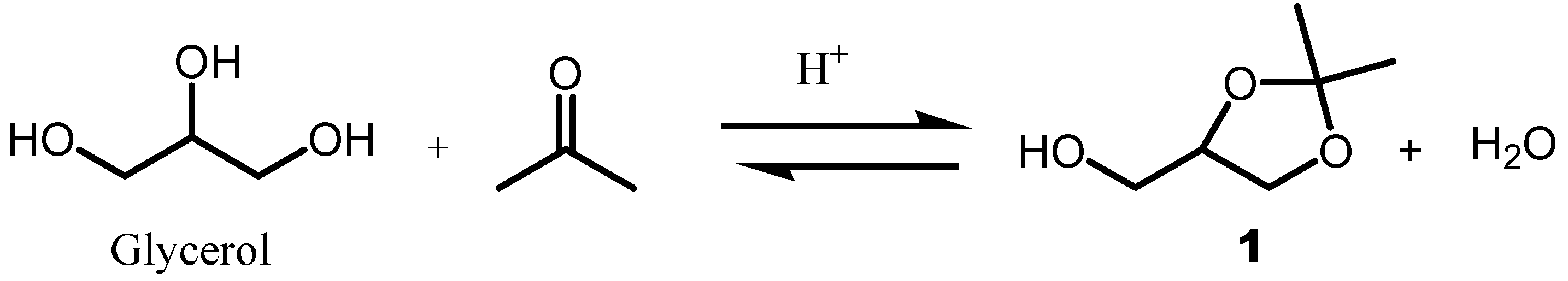
3.1. Conversion of Glycerol to Solketal
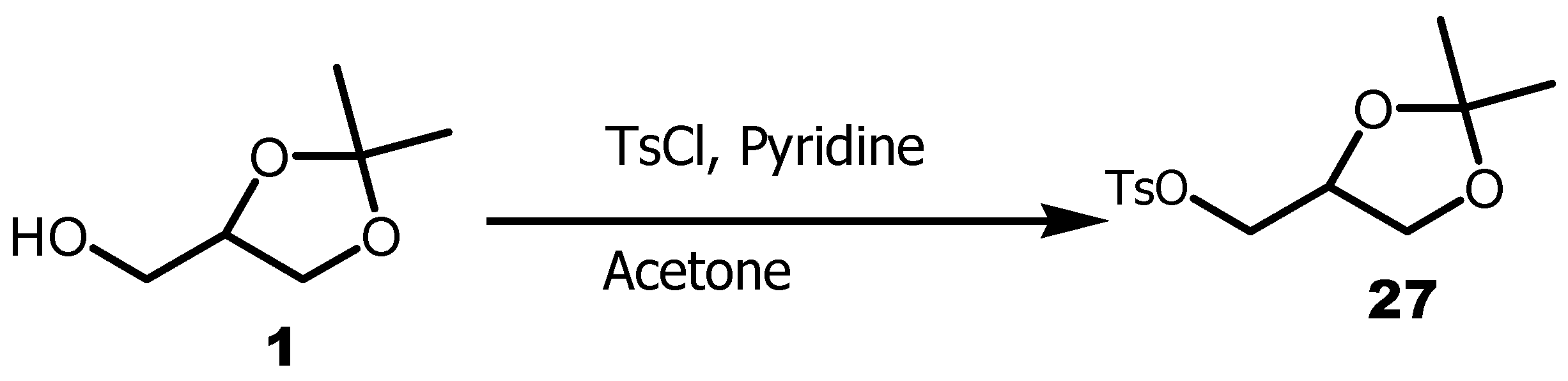
3.2. Conversion of Glycerol to Tosylsolketal
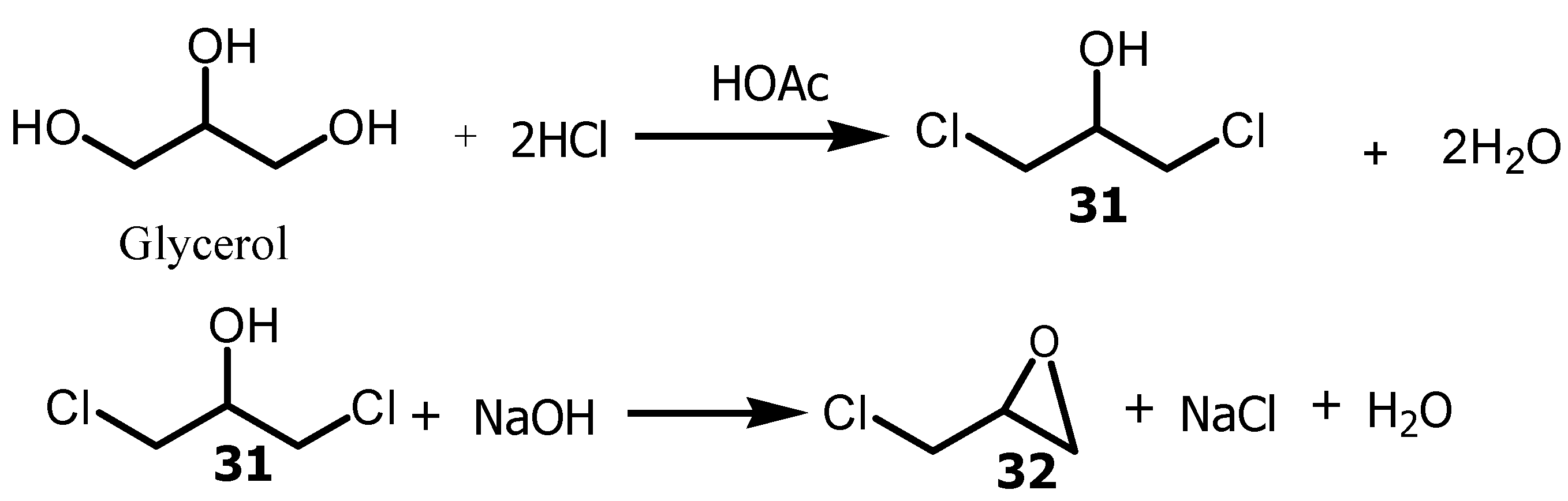
3.3. Conversion of glycerol to Epichlorohydrin
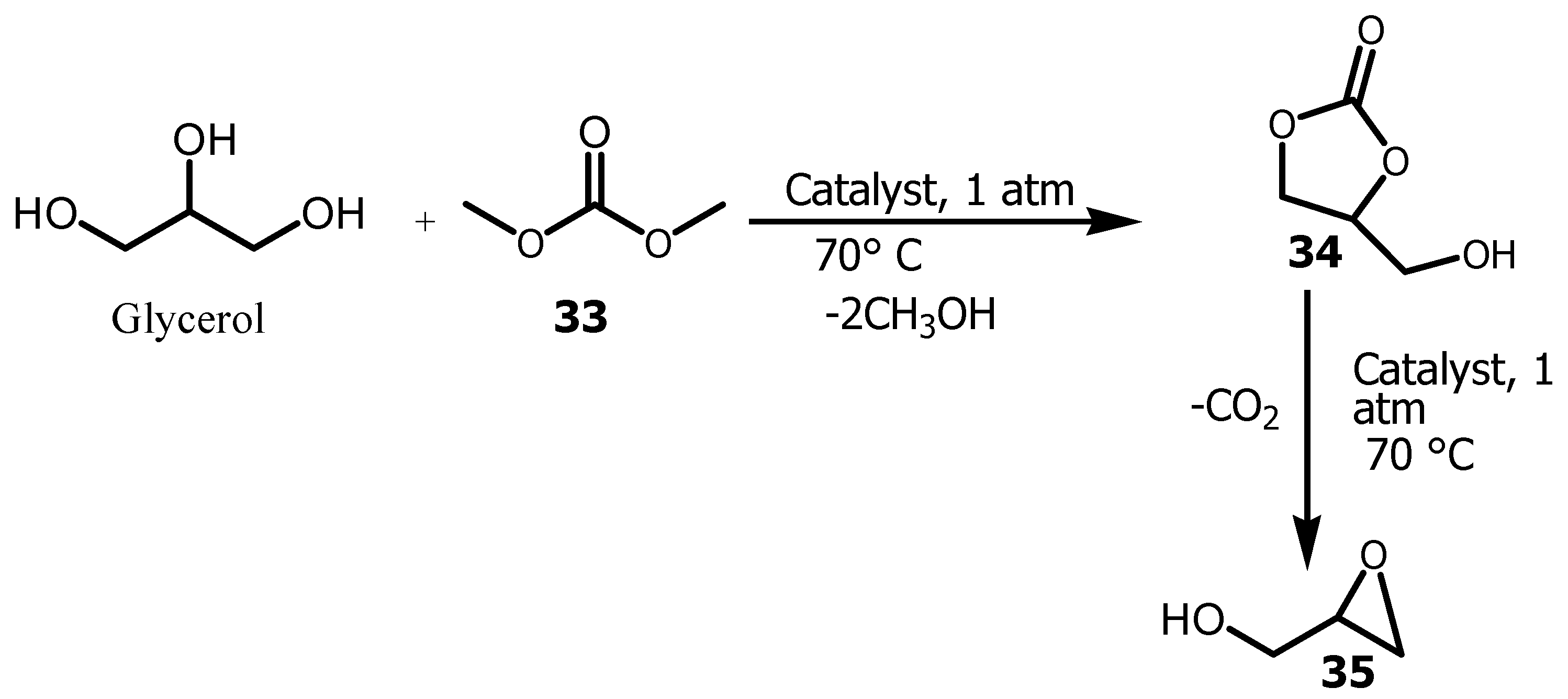
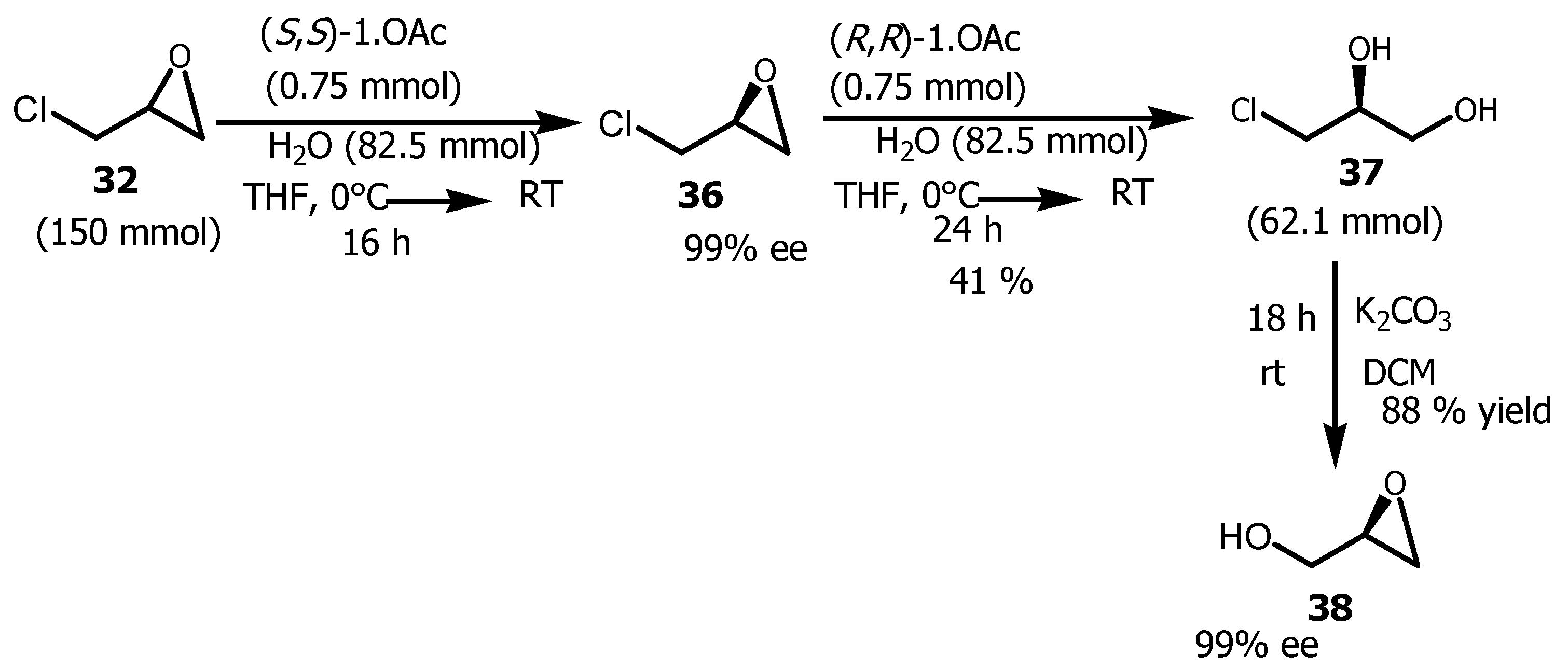
3.4. Conversion of Glycerol to (R)-Glycidol
4. Synthesis of Glycerol Derivatives
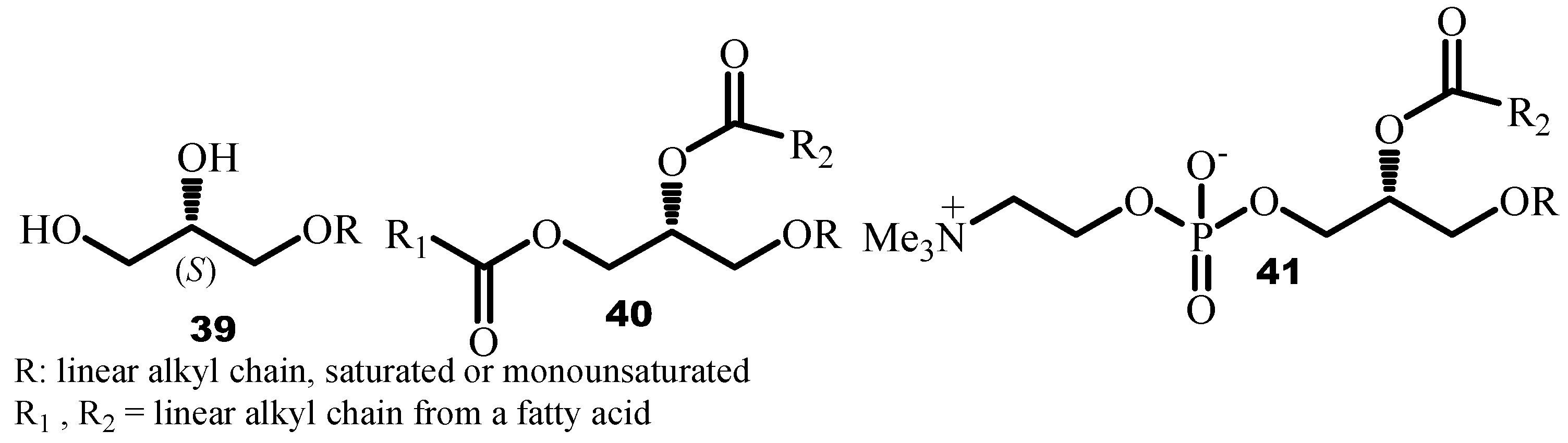
4.1. Synthesis of 1-O-Alkylglycerol from Solketal
4.2. Synthesis of Glycerol Derivatives from Glycidol
4.3. Synthesis of Organochalcogen from TosylSolketal
5. Conclusion
References
- Li L, Korányi, TI, Sels, BF and Pescarmona, PP. Highly-efficient conversion of glycerol to solketal over heterogeneous Lewis acid catalysts Green Chem., 2012, 14, 1611-1619. [CrossRef]
- Coombs, J and Hall, K. Chemicals and polymers from biomass Renewable Energy, 1998, 15, 1-4. [CrossRef]
- Corma, A, Iborra S and Velty, A. Chemical Routes for the Transformation of Biomass into Chemicals. Chem. Rev., 2007, 107, 2411–2502. [CrossRef]
- Lichtenthaler FW and Mondel, S. Perspectives in the use of low molecular weight carbohydrates as organic raw materials Pure Appl. Chem. 1997, 69, 1853–1866. [CrossRef]
- Gallezot, P. Catalytic routes from renewables to fine chemicals Catal. Today, 2007, 121, 76–91. [CrossRef]
- Liang, Y, et Cui, Y, Trushenski, J, Blackburn JW.Converting crude glycerol derived from yellow grease to lipids through yeast fermentation. Bioresour Technol 2010, 101(19): P. 7581-7586.
- Escriba, M, Eras, J, Duran, M, Simon, S, Cristina Butchosa, C, Villorbina, G, Balcells, M, Canela, R. From glycerol to chlorohydrin esters using a solvent-free system. Microwave irradiation versus conventional heating. Tetrahedron, 2009, 65, 10370–103. [CrossRef]
- Luo, X, Ge, X, Cui, S, Li, Y. Value-added processing of crude glycerol into chemicals and polymersBioresour Technol, 2016, 215, 144-154. [CrossRef]
- Pérez-Barrado, E, Pujol, MC, Aguiló, M, Llorca, J, Cesteros, Y, Díaz, F, Pallarès, J, Marsal, LF, Salagre, P. Influence of acid–base properties of calcined MgAl and CaAl layered double hydroxides on the catalytic glycerol etherification to short-chain polyglycerols. Elselvier Sciences Direct, 2015. 264, 547-556.
- Wang, Z, Zhuge, J, Fang, H, Prior, BA. Glycerol production by microbial fermentation: A review Biotechnology Advances, 2001, 19, 201-223. [CrossRef]
- Len, C, Luque, R. Continuous flow transformations of glycerol to valuable products: An overview. Sustain. Chem. Process. 2014, 2, 1–10. [CrossRef]
- García, E, Laca, M, Pérez,E, Garrido, A and Peinado,JN. New Class of Acetal Derived from Glycerin as a Biodiesel Fuel Component Energy Fuels, 2008, 22, 4274–4280. [CrossRef]
- Garcia, JI, Garcia-Marin, H, Pires, E. Glycerol based solvents: Synthesis, properties and applications. Green Chem., 2014,16, 1007-1033. [CrossRef]
- Costa, ICR, Itabaiana, I, Flores, MC, Lourenço, AC, Leite, SGF, Miranda, LS, Leal, ICR, Souza, RO. Biocatalyzed Acetins Production under Continuous-Flow Conditions: Valorization of Glycerol Derived from Biodiesel Industry. J. of Flow Chemistry , 2013, 3 41-45. [CrossRef]
- Krief, A, Provins,L and Froidbise, A.Diastereoselective synthesis of dimethyl cyclopropane-1,1-dicarboxylates from a γ-alkoxy-alkylidene malonate and sulfur and phosphorus ylides.Tetrahedron Letters,1998, 39, 1437–1440. [CrossRef]
- Ray, PC and Roberts, SM.Overcoming intrinsic diastereoselection using polyleucine as a chiral epoxidation catalyst. Tetrahedron Letters. 1999, 40, 1779–1782. [CrossRef]
- Michael, EJ and Teresa, JS. Total synthesis of (R)-glycerol acetonide and the antiepileptic and hypotensive drug (-)-.gamma.-amino-.beta.-hydroxybutyric acid (GABOB): Use of vitamin C as a chiral starting material.Journal of American ChemialSociety. 1980, 102, 6304-6311. [CrossRef]
- Deniau, AL, Mosset, P, Le Bot, D, Legrand, AB. Which alkylglycerols from shark liver oil have anti-tumour activities? Biochimie, 2011, 93, 1-3. [CrossRef]
- Momha, R, Kuete, V, Pagès, JM, Pegnyemb, DE, Mosset, P. Synthesis and Biological Evaluation of Four New Ricinoleic Acid-Derived 1-O-alkylglycerols.Marine Drugs,2020, 18(2), 113. [CrossRef]
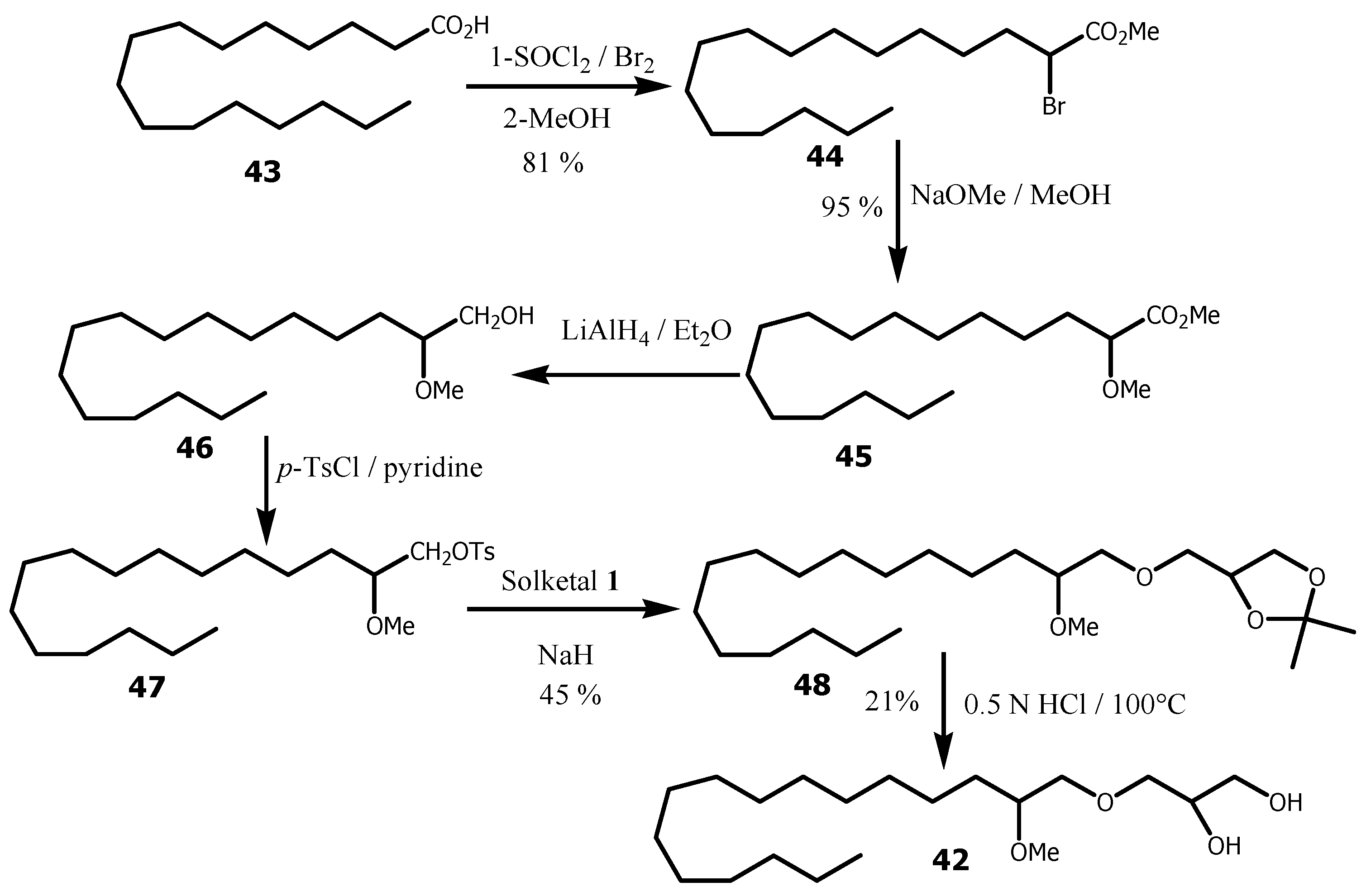
- Momha, R., Pegnyemb, DE, Mosset, P.Synthesis of (Z)-(2′ R)-1-O-(2′-methoxynonadec-10′-enyl)-sn-glycerol, a new analog of bioactive ether lipids.Tetrahedron2012, 68, 2973-2983. [CrossRef]
- Momha, R. Synthesis and therapeutic effects of Alkylglycerols, LAMBERT Academic Publishing, 2020, pp 1-56. ISBN: 978-3-330-02986-6.
- Oh, K, Yamada, K, Asami,T, Yoshizawa, Y. Synthesis of novel brassinosteroid biosynthesis inhibitors based on the ketoconazole scaffold.Bioorganic and Medicinal Chemistry Letters. 2012, 22, 1625-1628. [CrossRef]
- Callam, CS; et al. Computational analysis of the potential energy surfaces of glycerol in the gas and aqueous phases: Effects of level of theory, basis set, and solvation on strongly intramolecularly hydrogen-bonded systems.Journal of American Chemical Society. 2001, 123 (47), 11743-11754; 23. Callam, CS; et al. Computational analysis of the potential energy surfaces of glycerol in the gas and aqueous phases: Effects of level of theory, basis set, and solvation on strongly intramolecularly hydrogen-bonded systems.Journal of American Chemical Society. 2001, 123 (47), 11743-11754. [CrossRef]
- Da Silva, GP, De Lima, CJB,and Contiero,JJCT. Production and productivity of 1, 3-propanediol from glycerol by Klebsiella pneumoniae GLC29. Catalysis Today. 2015, 257, 259-266. [CrossRef]
- Simo MO, Li CJ Green. Chemistry oriented organic synthesis in water. Green Chemistry, 2012, 41, 1415-1427. [CrossRef]
- Capello, C, Fisher,U and Kungerb, uhler.“What Is a Green Solvent? A Comprehensive Framework for the Environmental Assessment of Solvents”. Green Chemistry.2007, 9, 927–934. [CrossRef]
- Leal-Duaso, A, Perez,P, Mayoral, JA, Pires, E and Garcia JI. Glycerol as a source of designer solvents:physicochemical properties of low melting mixtures containing glycerol ethers and ammonium salts. Phys. Chem. Chem. Phys., 2017, 19, 28302-28312. [CrossRef]
- Yin Len Kua, Suyin Gan, Andrew Morris, Hoon Kiat Ng. Ethyl lactate as a potential green solvent to extract hydrophilic (polar) and lipophilic (non-polar) phytonutrients simultaneously from fruit and vegetable by-products. Sustainable Chemistry and Pharmacy 2016, 4, 21-31. [CrossRef]
- Gu, Y and François J. Glycerol as a sustainable solvent for green chemistry.Green Chemistry. 2010, 12, P. 1127–1138. [CrossRef]
- Garcia, JI, Garcia-Marin, H, Mayoral, JA, P. Perez, P. Quantitative structure–property relationships prediction of some physico-chemical properties of glycerol based solvents. Green Chem. 2013, 15, 2283 –2293. [CrossRef]
- Capriati, V, Perna, FM, Salomone, A The Great Beauty” of organolithium chemistry: A land still worth exploring. Dalton Transactions. 2014, 43, 14204-14210. [CrossRef]
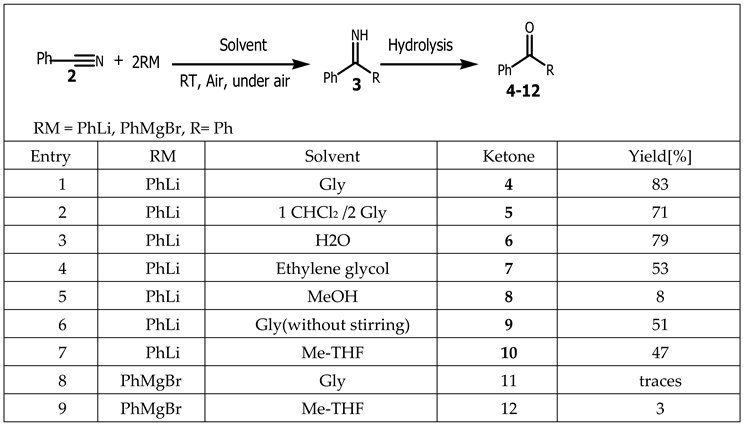
- Maria J, Rodriguez-Alvarez, Joaquin Garcia-lvarez, Marina Uzelac, Michael Fairley, Charles T. O’Hara, and Eva Hevia. Introducing Glycerol as a Sustainable Solvent to Organolithium Chemistry: Ultrafast Chemoselective Addition of Aryllithium Reagents to Nitriles under Air and at Ambient Temperature. Chemistry. A European Journal. 2018, 24, 1720–1725. [CrossRef]
- Butler, RN, Coyne, AG. Organic synthesis reactions on-water at the organic–liquid water interface Organic and Biomolecular Chemistry.2016, 14, 9945-9960. [CrossRef]
- Safaei, HR, Shekouhy, M, Rahmanpur, S, Shirinfeshan, A. Glycerol as a biodegradable and reusable promoting medium for the catalyst-free one pot three component synthesis of 4H-pyrans. Green Chemistry.2012, 14, 1696-1704. [CrossRef]
- Gu Y, Barrault J, Jerome F. Glycerol. as an efficient promoting medium for organic reactions. Advanced Synthesis and catalysis.2008, 350, 2007-2012. [CrossRef]
- Choi, JW, Jang, BK, Cho, NC, Park, JH, Yeon, SK, Ju, EJ, Lee, YS, Han, G, Pae, AN, Kim, DJ, and Park, KD.Synthesis of Amide and Ester Derivatives of Cinnamic Acid and Its Analogs: Evaluation of Their Free Radical Scavenging and Monoamine Oxidase and Cholinesterase Inhibitory Activities.Chemical and Pharmaceutical Bulletin, 2017, 65(11), 1020-1027. [CrossRef]
- Green,O, Smith, NA, Ellis, AB and Burstyn, JN.AgBF4-Impregnated Poly(vinyl phenyl ketone): An Ethylene Sensing Film. Journal of American Chemical Society.2004, 126 (19), 5952–5953. [CrossRef]
- Bianco,A, Cavarischia, C and Guiso, M. The Heck Coupling Reaction Using Aryl Vinyl Ketones: Synthesis of Flavonoids.European Journal of Organic Chemistry. 2004, 13, 2894 – 2898. [CrossRef]
- Schroeder, M, Mathys, M, Ehrensperger, N and Buchel, M. γ-Unsaturated Aldehydes as Potential Lilial Replacers. Chem. Biodiversity, 2014, 11, 1651–1673.
- Hu, L, Lu, X and Deng, L. Catalytic Enantioselective Peroxidation of α,β-Unsaturated Aldehydes for the Asymmetric Synthesis of Biologically Important Chiral Endoperoxides. Journal of American Chemical Society.2015, 137(26), 8400–8403. [CrossRef]
- Breuer, M, Ditrich, K, Habicher,T, Hauer, B, Kesseler, M, Sturmer, R and Zelinski, T. Industrial Methods for the Production of Optically Active Intermediates. Angew. Chem., Int. Ed., 2004, 43, 788–824. [CrossRef]
- Liu, L, Corma, A. Metal Catalysts for Heterogeneous Catalysis: From Single Atoms to Nanoclusters and Nanoparticles. Chemical Reviews. 2018, 118(10), 4981−5079. [CrossRef]
- Roat-Malone, RM. Bioinorganic Chemistry: A Short Course. Wiley: Hoboken, NJ, 2007.
- Lee, JS, Vlaisavljevich, B, Britt, DK, Brown, CM, Haranczyk, M, Neaton, JB, Smit, B, Long, JR, Queen, WL. Understanding Small-Molecule Interactions in Metal−Organic Frameworks: Coupling Experiment with Theory. Advanced Materials. 2015, 27(38), 5785-5796. [CrossRef]
- Chahdoura, F, Favier, I and Gomez, M. Glycerol as Suitable Solvent for Synthesis of Metallic Species and Catalysis.Chemistry A European Journal.2014, 20, 1–11. [CrossRef]
- Albrecht, M. Cyclometalation Using d-Block Transition Metals: Fundamental Aspects and Recent Trends. Chemistry Reviews. 2010, 110, 576 –623. [CrossRef]
- Kobayashi, K, Ishikubo, M, Kanaizuka, K, Kosuge, K, Masaoka, S, Sakai, K, Nozaki, K, Haga, MA. Proton-induced tuning of metal-metal communication in rack-type dinuclear Ru complexes containing benzimidazolyl moieties. Chemistry A European Journal. 2011, 17(25), 6954-6963. [CrossRef]
- Chen, PL, Chao, H, Xu, J, Wang, L, Li, H. Luminescence properties of a di-ruthenium(II) complex with an intramolecular hydrogen bond modulated by DNA and copper(II) ion. Transition Metal Chemistry, 2009, 34, 773– 778. [CrossRef]
- Sprouse,S, King, KA, Spellane, PJ, Watts, RJ. Photophysical effects of metal-carbon .sigma. bonds in ortho-metalated complexes of iridium(III) and rhodium(III). Journal Of The American Chemical Society, 1984, 106, 22, 6647-6653. [CrossRef]
- Yue, SM, Xu, HB, Ma, JF, Su, ZM, Zhang HJ, Design and syntheses of blue luminescent zinc(II) and cadmium(II) complexes with bidentate or tridentate pyridyl-imidazole ligands. Polyhedron, 2006, 25, 635-644. [CrossRef]
- Khatri, PK, Jain, SL. Glycerol ingrained copper: An efficient recyclable catalyst for the N-arylation of amines with aryl halides. Tetrahedron Lett. 2013, 54, 2740 –2743. [CrossRef]
- Gordon, JF, Chittenden. Some aspects of the reaction of glycerol with 2,2-dimethoxypropane. Carbohydrate research. 1983, 121, 316-323. [CrossRef]
- Patrick, C, Nobre, Elton L, Borges, Cristian M, Silva, Angela M, Casaril, Débora M, Martinez Eder J, Lenardão, Diego Alves, Lucielli Savegnago, Gelson Perin. Organochalcogen compounds from glycerol: Synthesis of new antioxidants.Bioorganic & Medecinal Chemistry. 2014, 22, 6242–6249. [CrossRef]
- Irvine, J, MacDonald, J, Soutar, C.Condensation of acetone and benzaldehyde with glycerol. Preparation of glycerol α-methyl ether. Journal of the Chemical Society. 1915, 107, 337. [CrossRef]
- Jackson, K& Jones, J. Synthesis of 3-hexuloses. Canadian Journal of Chemistry. 1969, 47, 2498-2501.
- Michael E, Jung and Teresa J, Shaw. Total Synthesis of (R)-Glycerol Acetonide and the Antiepileptic and Hypotensive Drug (-)-y-Amino-P-hydroxybutyric Acid (GABOB): Use of Vitamin C as a Chiral Starting Material. Journal of the American Chemical Society. 1980, 102(20), 6304–6311. [CrossRef]
- Kong PS, Kheireddine M, Mohd W, Wan A. Conversion of crude and pure glycerol into derivatives : A feasibility evaluation. Renewable and Sustainable Energy Reviews, 2016, 63, pp. 533–555. [CrossRef]
- Smith, AG. Synthetically Useful Reactions of Epoxides. Synthesis 1984, 8, 629-656. [CrossRef]
- Palomo, M, Segura,RL, Mateo, C, Terreni, M, Guisan, JM and Fernández-Lafuente, R. Tetrahedron Asymmetry:, 2005, 16, 869-874. [CrossRef]
- Hanson, RM. The synthetic methodology of nonracemic glycidol and related 2,3-epoxy alcohols Chem. Rev., 1991, 91, 437-475. [CrossRef]
- Pagliaro, M, Ciriminna, R, Kimura, H, Rossi, M and Pina, CD. From Glycerol to Value-Added Products Angew. Chem. Int. Ed. 2007, 46, 4434-4440. [CrossRef]
- Elrasheed, E, Wang,H, Imran, M, Hegazi, SEF, Hassan, M, Eldoma, MA, Hakami, J, Wani, WA, Chaudhary, AA. Nanocatalyst-Assisted Facile One-Pot Synthesis of Glycidol from Glycerol and Dimethyl Carbonate. ACS Omega 2022. [CrossRef]
- Crosby J. Synthesis of optically active compounds: A large scale perspective. Tetrahedron,1991 47, 4789-4846. [CrossRef]
- Schaus, SE, Brandes, BD.; Larrow, JF.; Tokunaga, M, Hansen, KB.; Gould, AE, Furrow, ME, Jacobsen, EN. Highly Selective Hydrolytic Kinetic Resolution of Terminal Epoxides Catalyzed by Chiral (salen)CoIII Complexes. Practical Synthesis of Enantioenriched Terminal Epoxides and 1,2-Diols. J. Am. Chem. Soc. 2002, 124, 1307-1315. [CrossRef]
- Bordier, CG, Sellier, N, Foucault, AP, Le Goffic, F. Purification and characterization of deep sea shark Centrophorus squamosus liver oil 1-O-alkylglycerol ether lipids. Lipids, 1996, 31, 521–528. [CrossRef]
- Baer, E.; Fisher, H. O. L. J. Biol. Chem.1941, 140, 397-410.
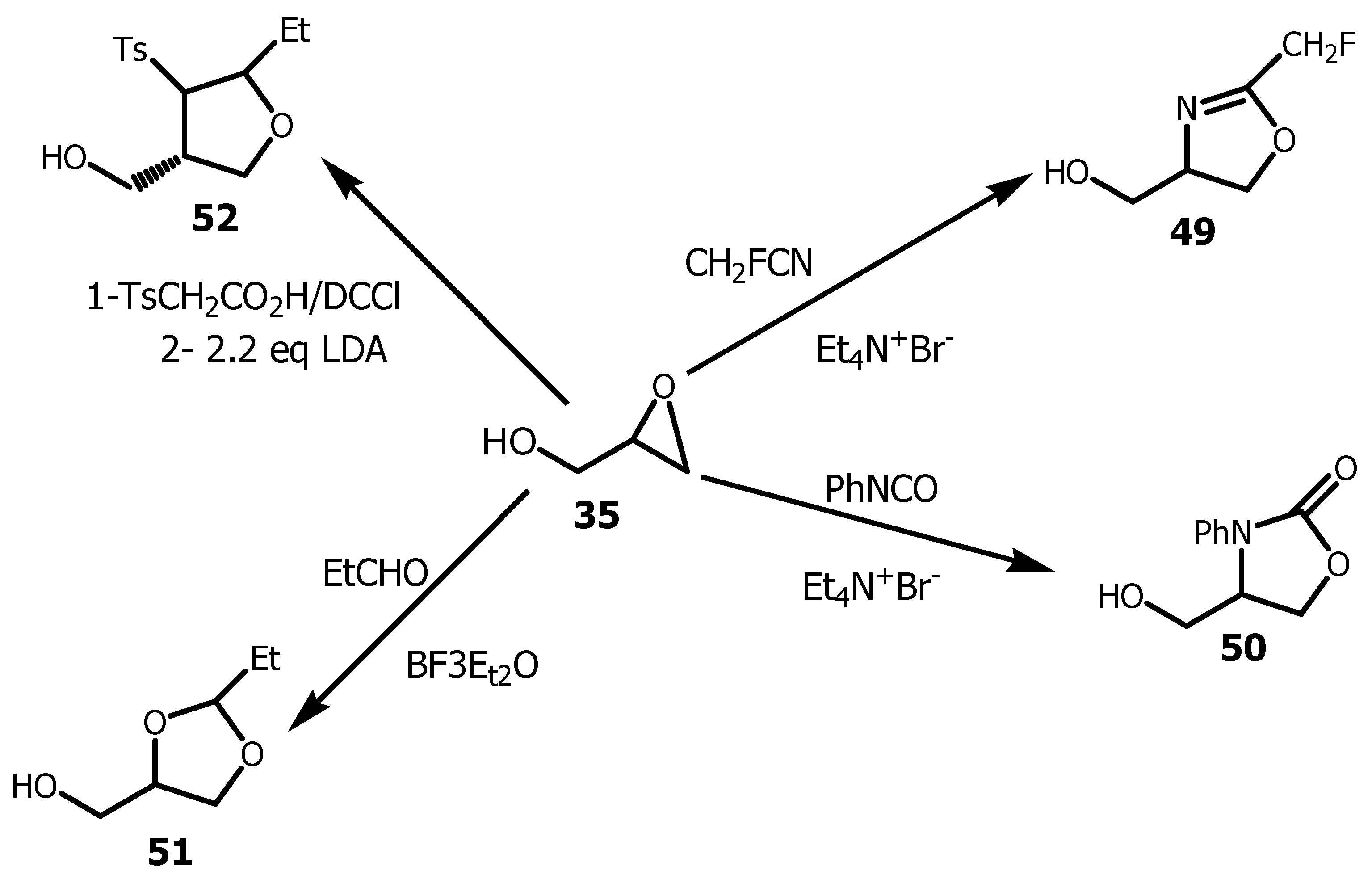
- Momha, R, Pegnyemb, DE, Mosset, P. Synthesis of halogenated 1-O-alkylglycerols from ricinoleic acid derivatives. Synthetic Communications, 2020, 50, 1656-1664. [CrossRef]
- Linman, JW, Long, MJ, Korst, DR, Bethell, FH. J. Lab. Clin. Med.1959, 54, 335–343.
- Brohult, A, Brohult, J, Brohult, S, Joelsson, I. Effect of alkoxyglycerols on the frequency of injuries following radiation therapy for carcinoma of the uterine cervix.Acta Obst. Gynecol. Scand.1977, 56, 441–448. PMID: 602713. [CrossRef]
- Momha, R, Bayiha, GBN, Pegnyemb, DE, Mosset, P. First total synthesis of two 1-O-alkylglycerols based-alkyne analogues of bioactive natural products.ChemistrySelect 2020, 5, 6678-6682. [CrossRef]
- Brohult, A, Brohult, J, Brohult, S. Regression of tumour growth after administration of Alkoxyglycerols. Acta Obstet. Gynecol. Scand.1978, 57, 79–83. [CrossRef]
- Momha, R, Le Bot, D, Mosset, P, Legrand, AB. Anti-Angiogenic and Cytotoxicity Effects of Selachyl Alcohol Analogues. Anticancer Agents in Medicinal Chemestry. 2022, 10, 1913-1920. [CrossRef]
- Ngwenya, BZ & Foster, DM. Enhancement of antibody production by lysophosphatidylcholine and alkylglycerol. Proceedings of the Society for Experimental Biology and Medicine. 1991, 196, 69-75. [CrossRef]
- Brohult, A, Brohult, J, Brohult, S. Effect of irradiation and alkoxyglycerol treatment on the formation of antibodies after Salmonella vaccination. Experientia, 1972, 28, 954–955. [CrossRef]
- Hallgren, B& Ställberg, G. Methoxy-substituted glycerol ethers isolated from Greenland shark liver oil. Acta Chem. Scand.1967, 21, 1519-1529.
- Hallgren, B.; Ställberg, G. Acta Chem. Scand. 1967, 21, 1519-1529.
- Hanson, RM. The synthetic methodology of nonracemic glycidol and related 2,3-epoxy alcohols. Chem. Rev. 1991, 91(4), 437–475. [CrossRef]
- Desai, SP, Taylor, MS. Diarylborinic Acid-Catalyzed Regioselective Ring Openings of Epoxy Alcohols with Pyrazoles, Imidazoles, Triazoles, and Other Nitrogen Heterocycles. Org. Lett., 2021, 23(18), 7049–7054. [CrossRef]
- Fotadar, U, Becu, C; Borremans, F. AM, Anteunis, M J0. Synthetic and conformational aspects of trimethylammonium-methyl substituted 2-oxazolines as potential cholinergics .Tetrahedron, 1978, 34, 3537-44. [CrossRef]
- Farrissey, WJ; Nashu, AMJ. The rearrangement of glycicyl N-phenylcarbamate. Heterocycl. Chem. 1970, 7, 331-333. [CrossRef]
- Barcellos, AM, Abenante, L, Sarro, MT, Leo, ID, Lenardão, EJ, Perin, G, Santi, C. New prospective for redox modulation mediated by organoselenium and organotellurium compounds. Current. Organic Chemistry.2017, 21, 1–18. [CrossRef]
- Mugesh, G, du Mont, WW, Sies, H. Chemistry of biologically important synthetic organoselenium compounds. Chemistry. Review. 2001, 101, 2125–2179. [CrossRef]
- Santoro, S, Azeredo, JB, Nascimento, V, Sancineto, L, Braga, AL, Santi, C. The green side of the moon:Ecofriendly aspects of organoselenium chemistry. RSC Advances.2014, 4, 31521–31535. [CrossRef]
- Mitamura, T, Ogawa, A. Palladium-catalyzed alkynylselenation of acetylenedicarboxylates leading to enyne selenides and application to synthesis of multisubstituted aryl selenides. Tetrahedron Letters, 2010, 51, 3538–3541. [CrossRef]
- Liu, CR., Yang, FL, Jin, YZ, Ma, XT, Cheng, DJ, Li, N, Tian, SK. Catalytic regioselective synthesis of structurally diverse indene derivatives from n-benzylic sulfonamides and disubstituted alkynes. Organic Letters, 2010, 12, 3832–3835. [CrossRef]
- Okoronkwo, AE, Godoi, B, Schumacher, RF, Neto, JSS, Luchese, C, Prigol, M, Nogueira, CW, Zeni, G.Csp3-tellurium copper cross-coupling: Synthesis of alkynyl tellurides a novel class of antidepressive-like compounds. Tetrahedron Lett. 2009, 50, 909–915. [CrossRef]
- Savegnago, L, Borges, VC, Alves, D, Jesse, CR, Rocha, JBT, Nogueira, CW. Evaluation of antioxidant activity and potential toxicity of 1-buthyltelurenyl-2-methylthioheptene. Life Sciences, 2006, 79, 1546–1552. [CrossRef]
- Ávila, DS, Gubert, P, Palma, A, Colle, D, Alves, D, Nogueira, CW, Rocha, JBT, Soares, FAA. An organotellurium compound with antioxidant activity against excitotoxic agents without neurotoxic effects in brain of rats. Brain Resaerch Bulletin 2008, 76, 114–123. [CrossRef]
- Soares, LK, Silva, RB, Peglow, TJ, Silva, MS, Jacob, RG, Alves, D, Perin, G. Selective synthesis of vinyl- or alkynyl chalcogenides from glycerol and their water-soluble derivatives. ChemistrySelect. 2016, 1, 2009–2013. [CrossRef]
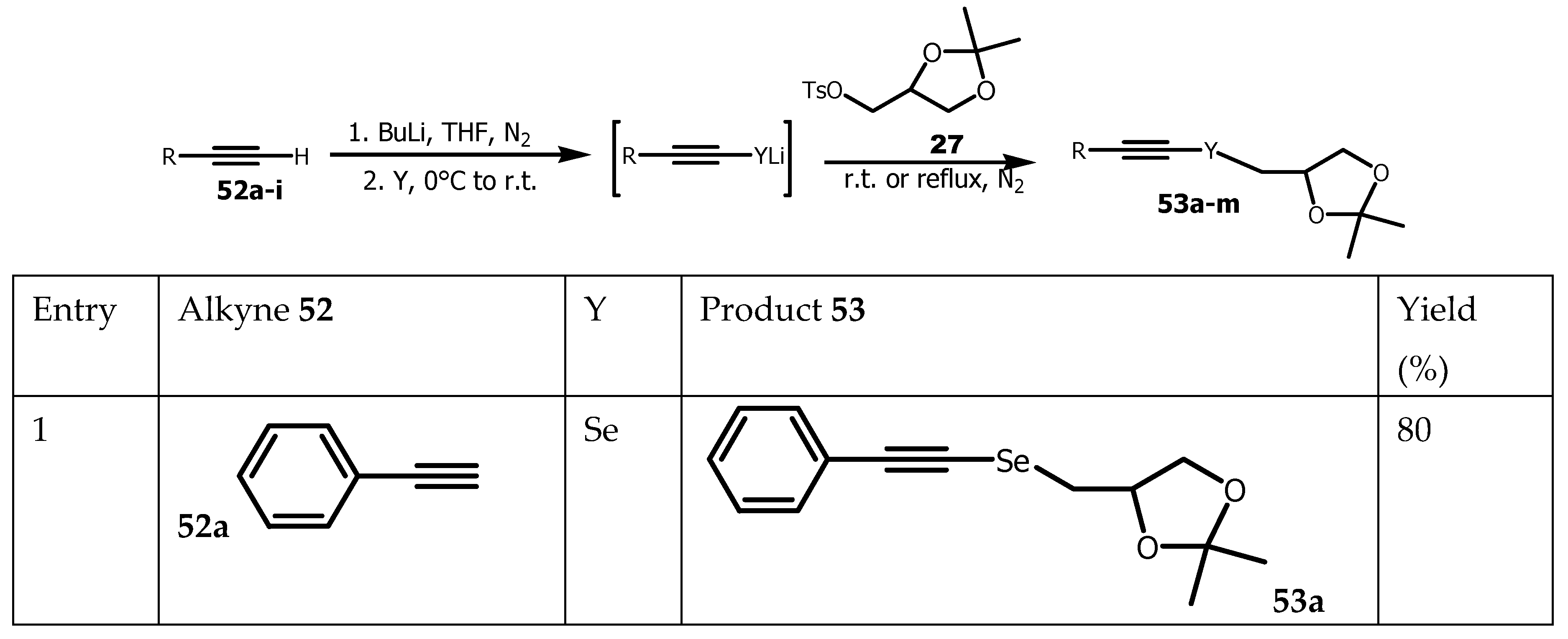
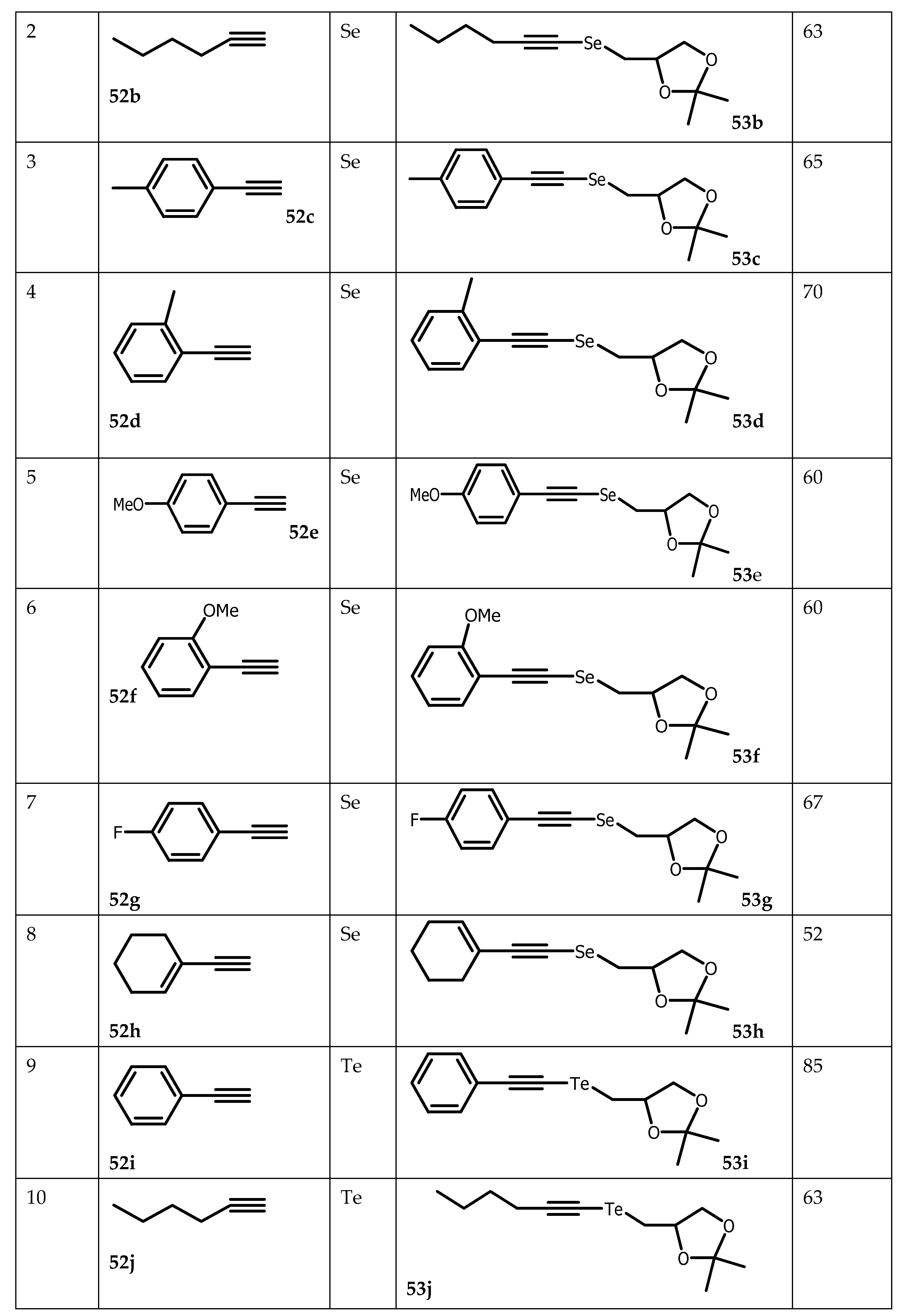
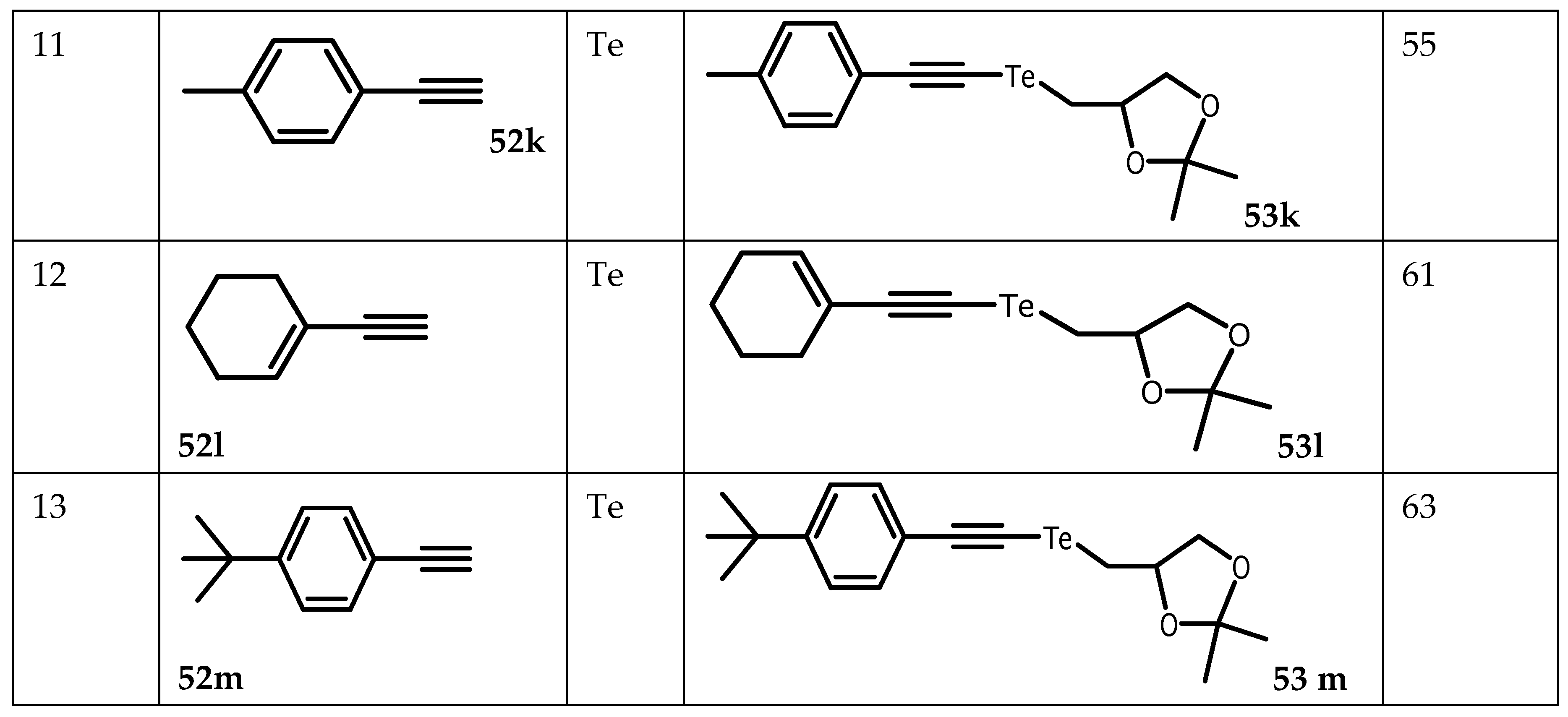
- Lenardão, EJ, Borges, EL, Stach, G, Liane K, Alves, SD, Schumacher, RF, Bagnoli, L, Marini, F and Perin, G Glycerol as Precursor of Organoselanyl and Organotellanyl Alkynes. Molecules. 2017, 22, 391. [CrossRef]
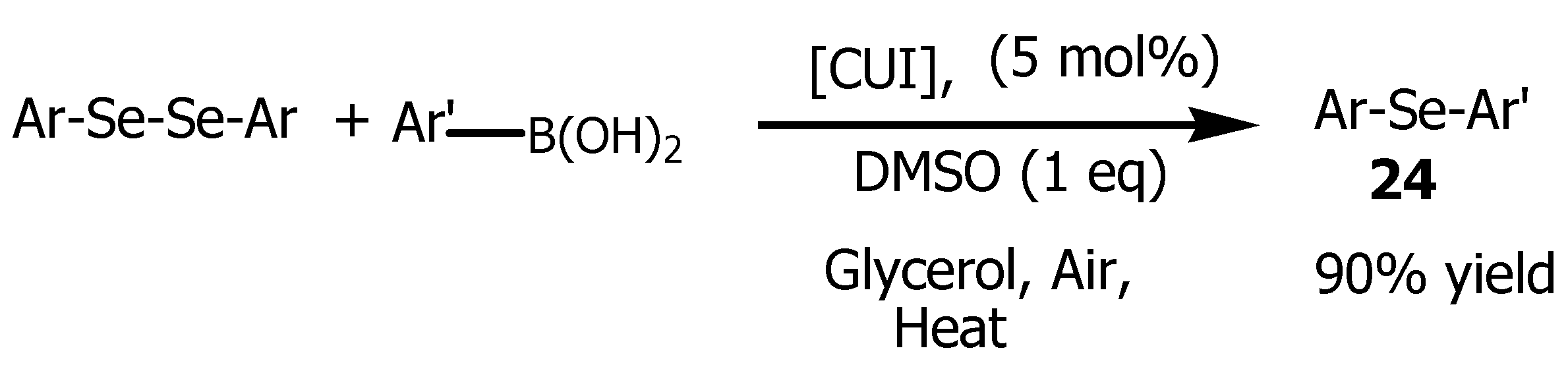
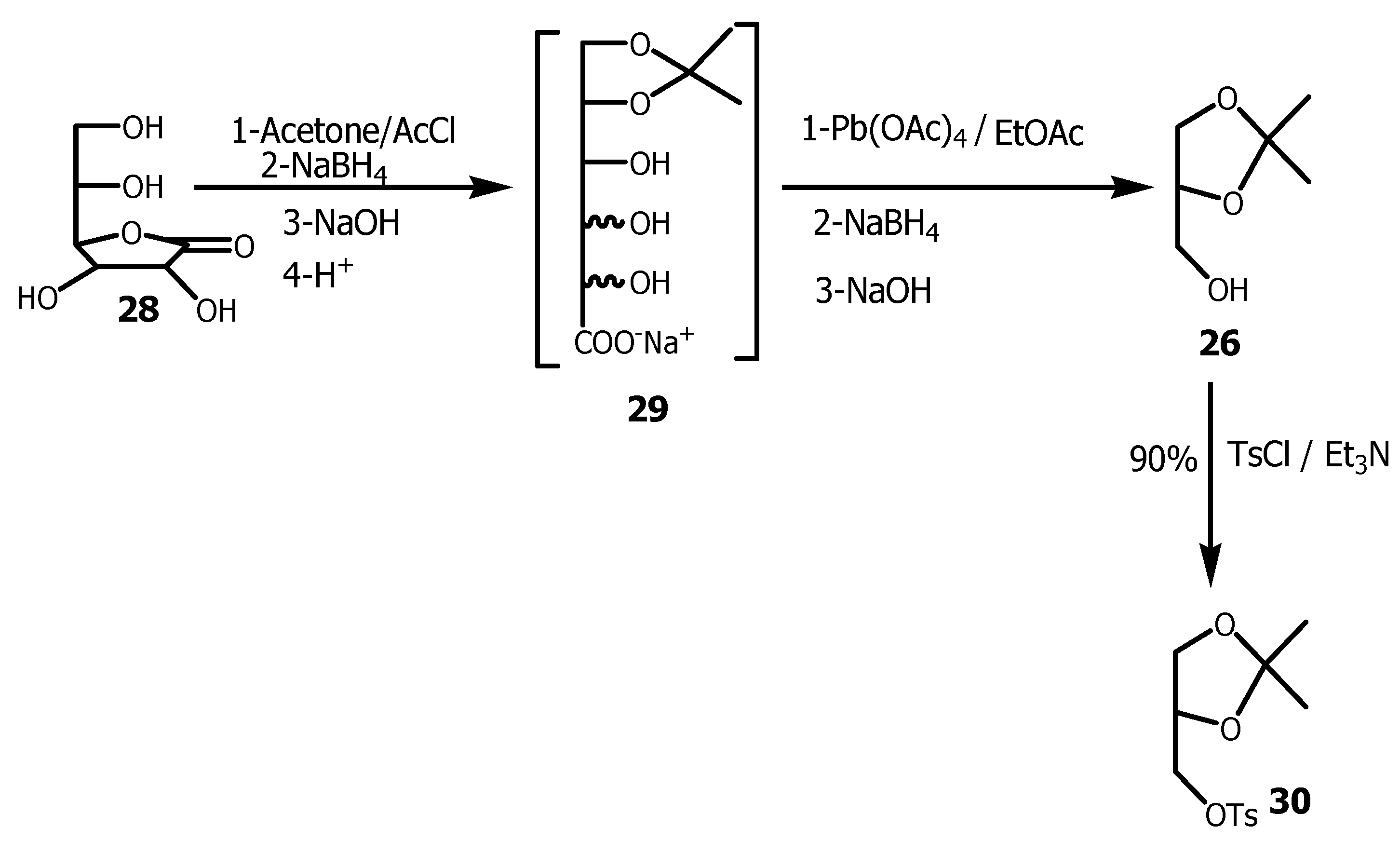
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




