Submitted:
28 June 2024
Posted:
01 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
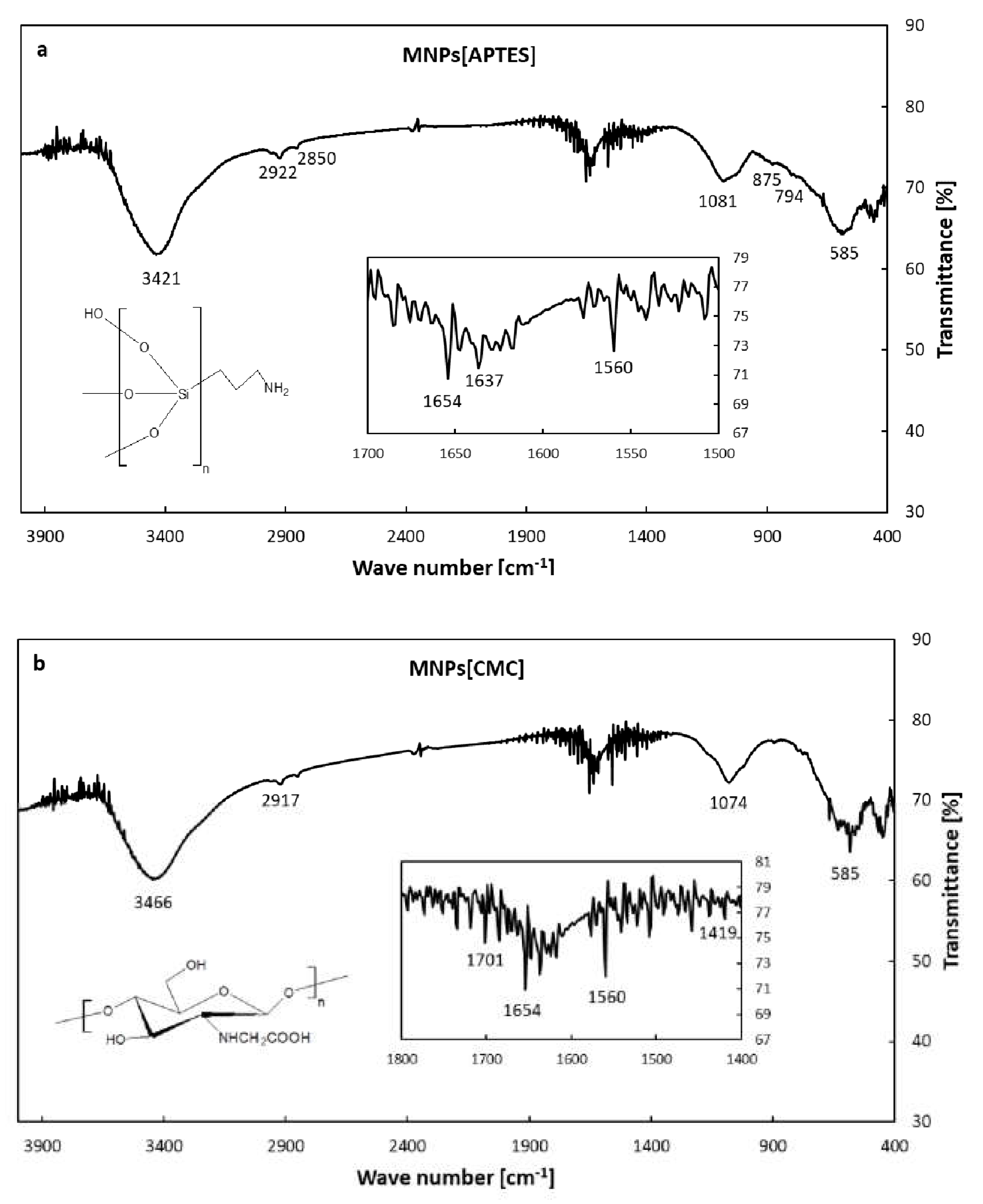
2.1. Characterization with FT-IR and Transmission Electron Microscope (TEM) Imaging of APTES- and CMC-Modified Nanoparticles (MNPs[APTES] and MNPs[CMC])
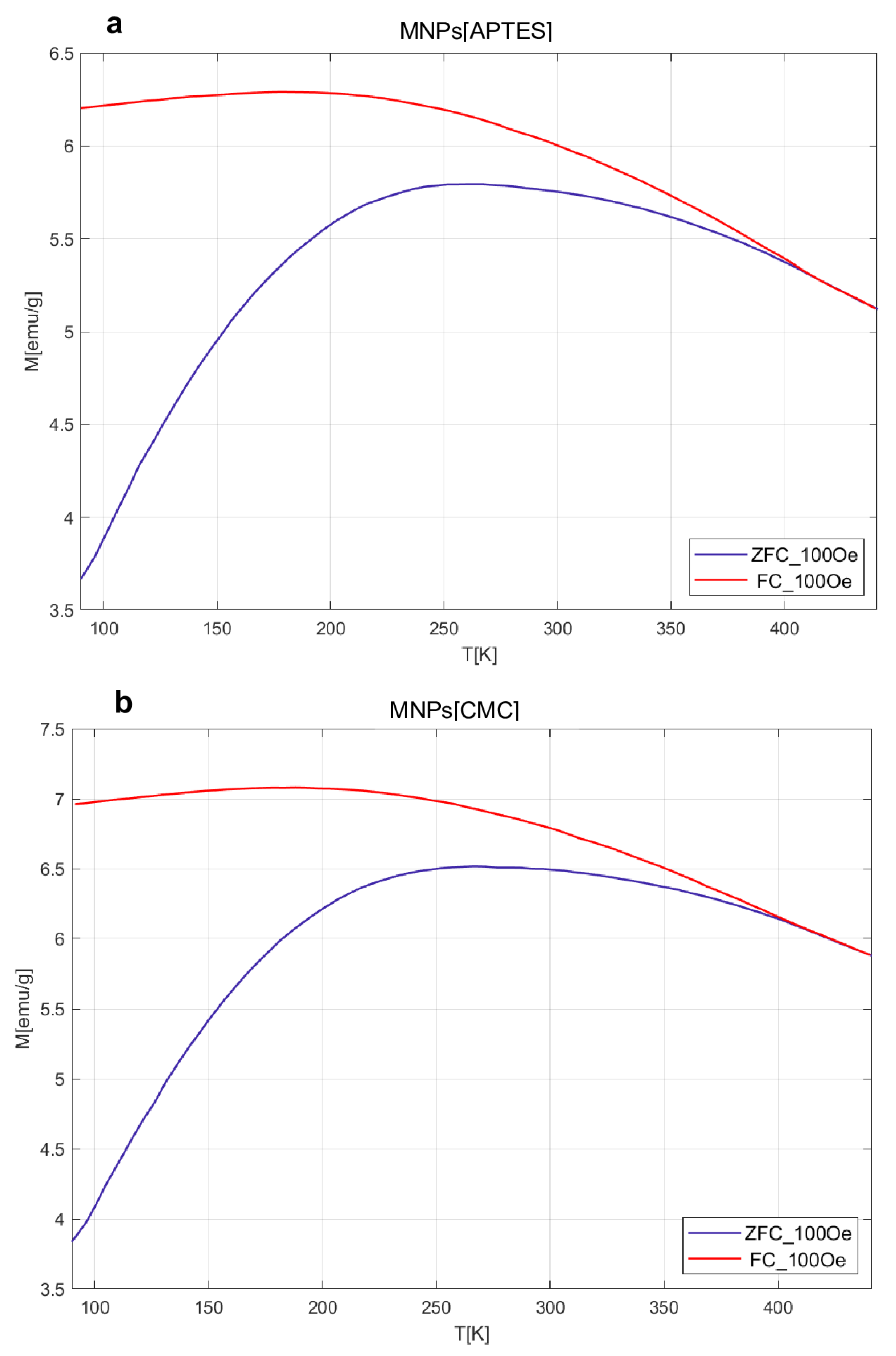
2.2. Magnetic VSM Properties Measurements of MNPs[APTES] and MNPs[CMC]
2.3. Hyperthermic Properties of MNPs[APTES] and MNPs[CMC]
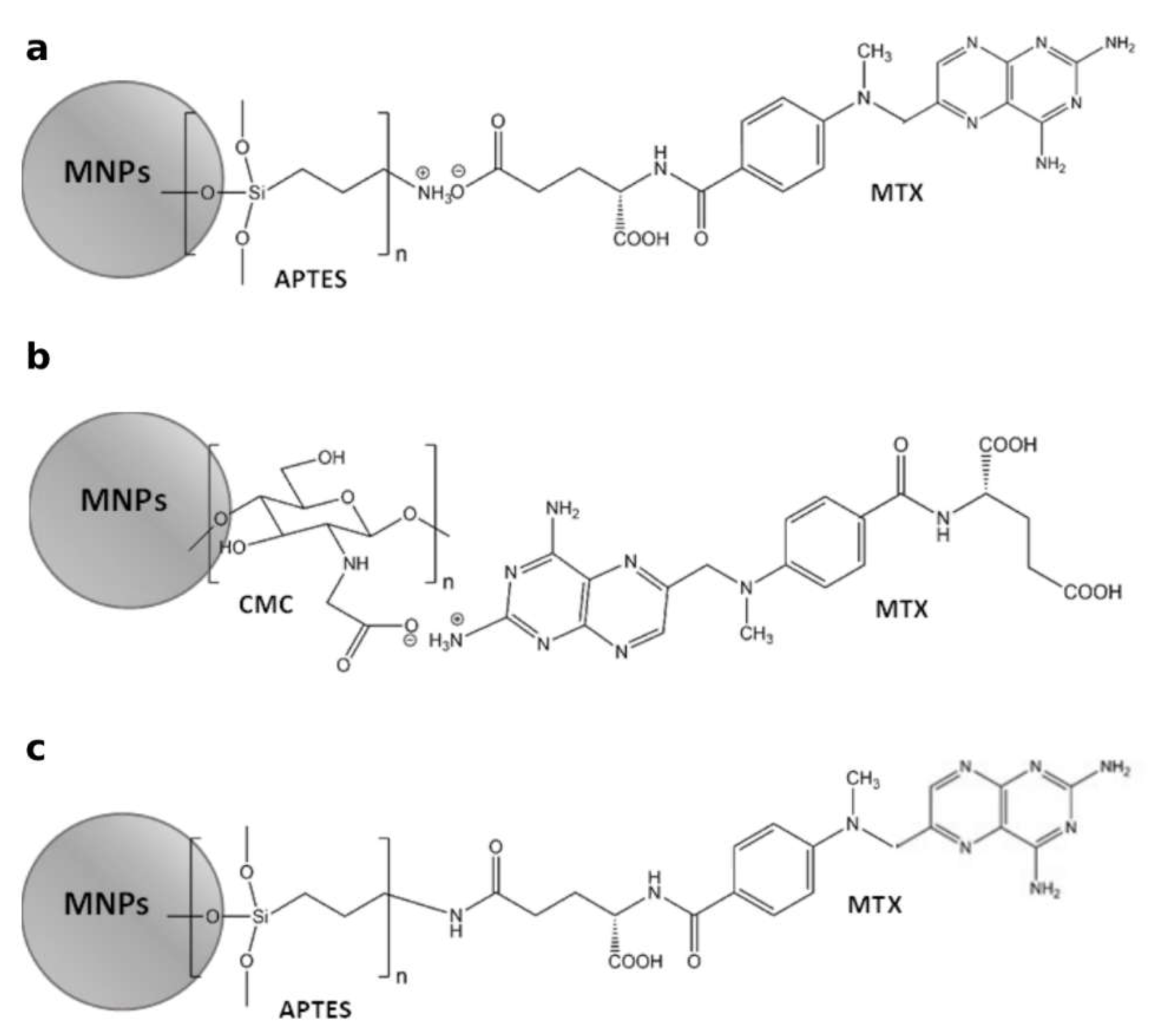
2.4. Capability of MTX Loading
2.5. DLS and Zeta Potential Measurements of MNPs[APTES], MNPs[CMC] and MTX-Functionalized Nanoparticles (MNPs[APTES]MTX and MNPs[CMC]MTX)
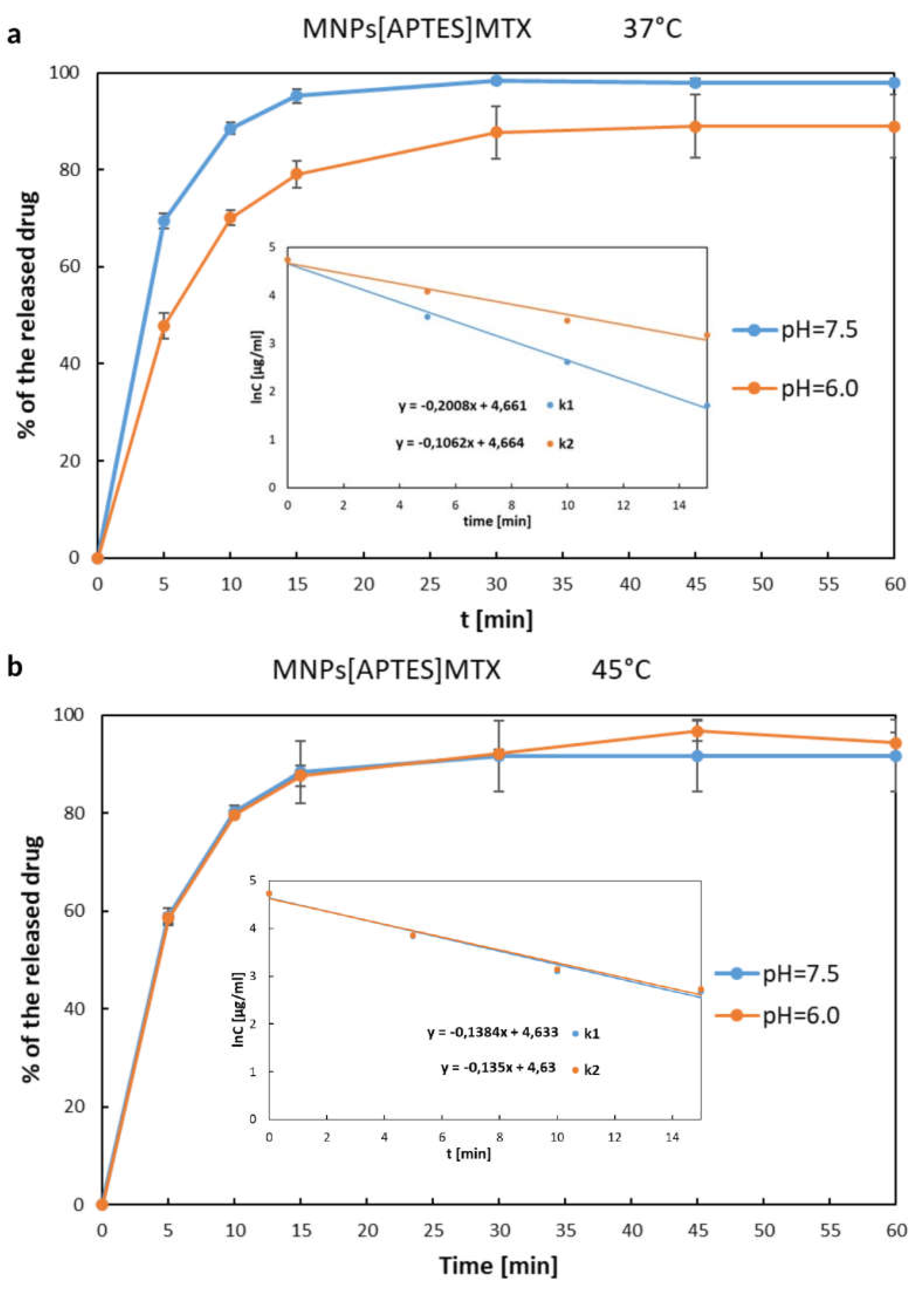
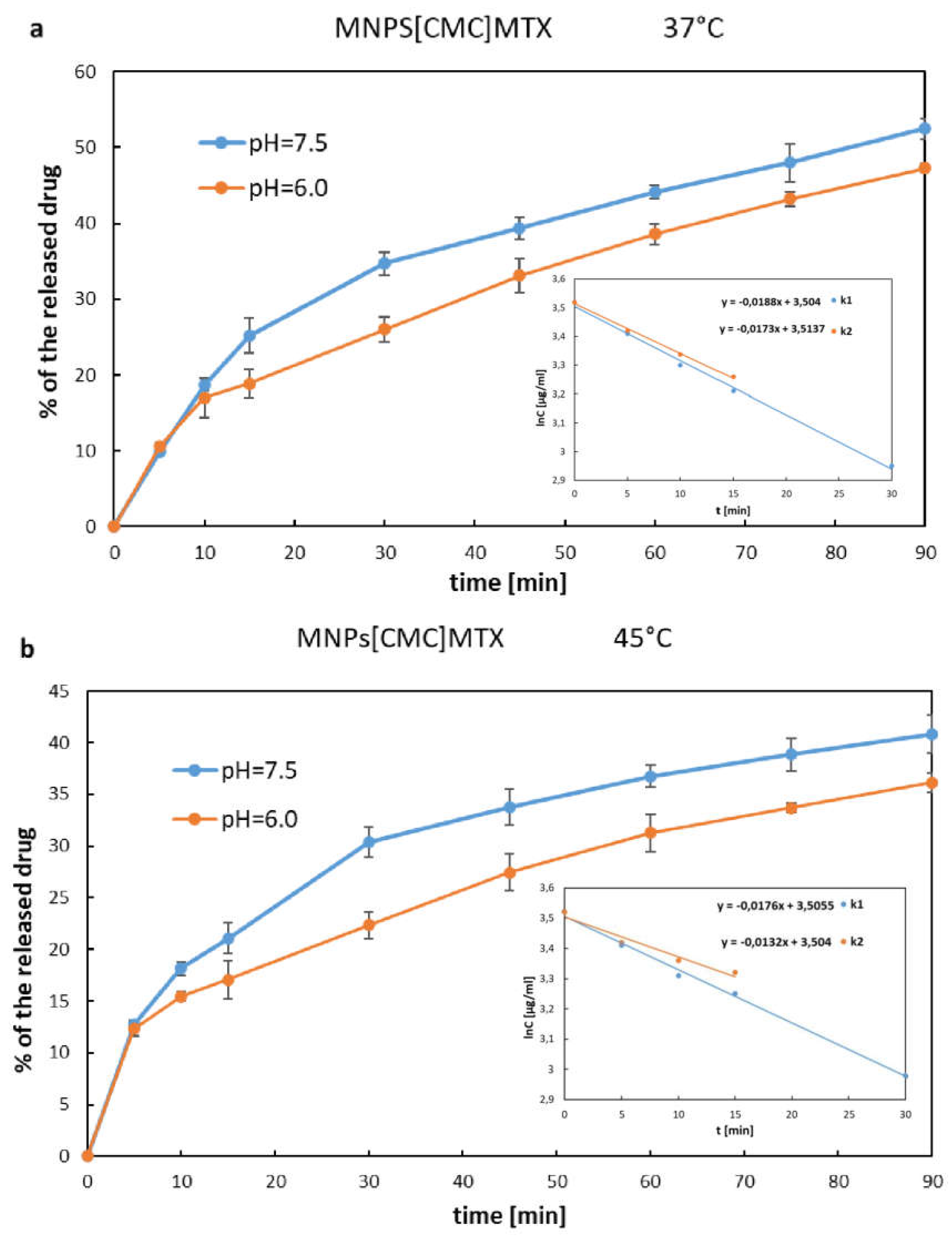
2.6. Investigation of MTX Release from MNPs[APTES]MTX and MNPs[CMC]MTX (Ionic Bond)
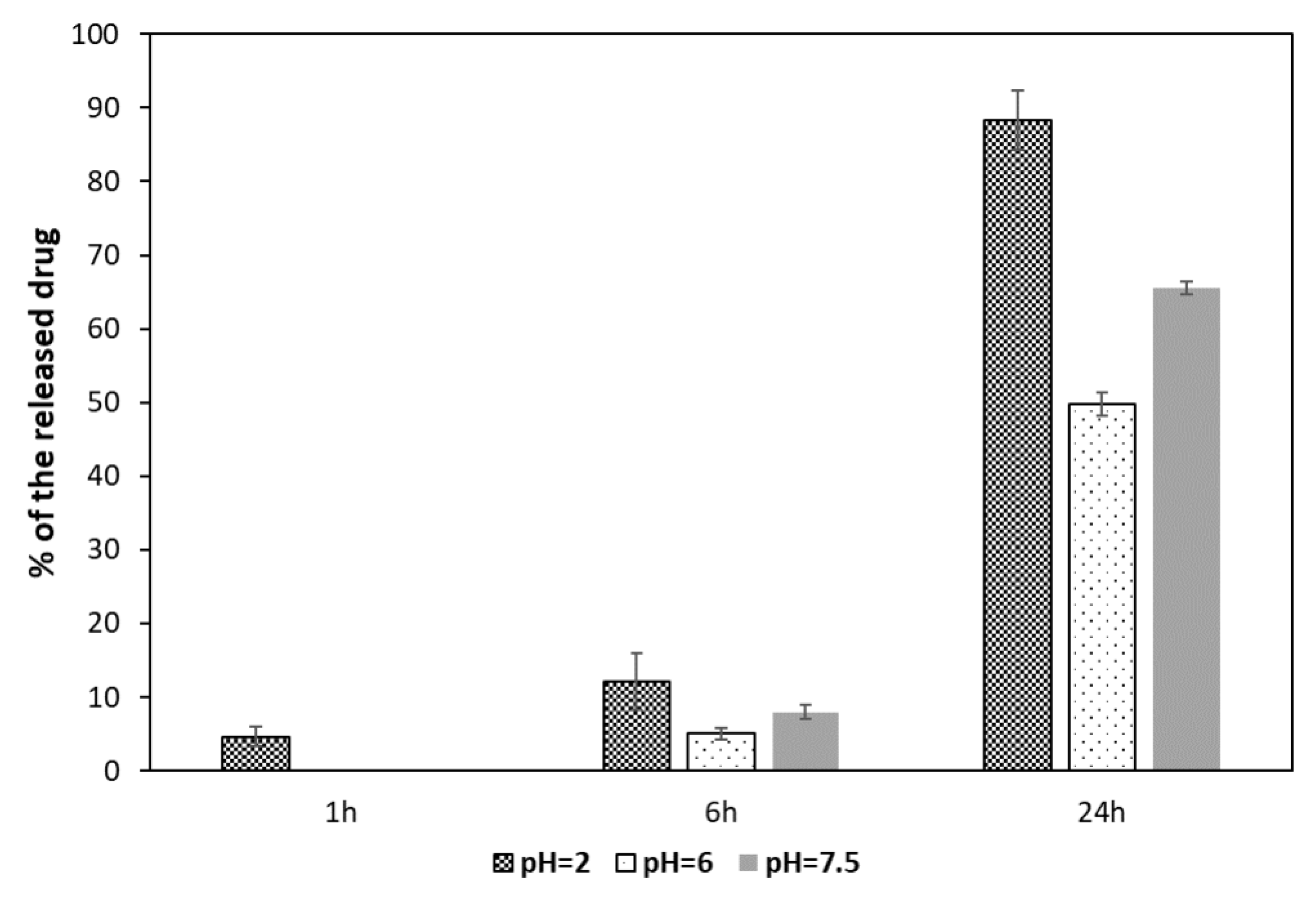
2.7. Investigation of MTX Release from MNPs[APTES]MTX (Amide Bond)
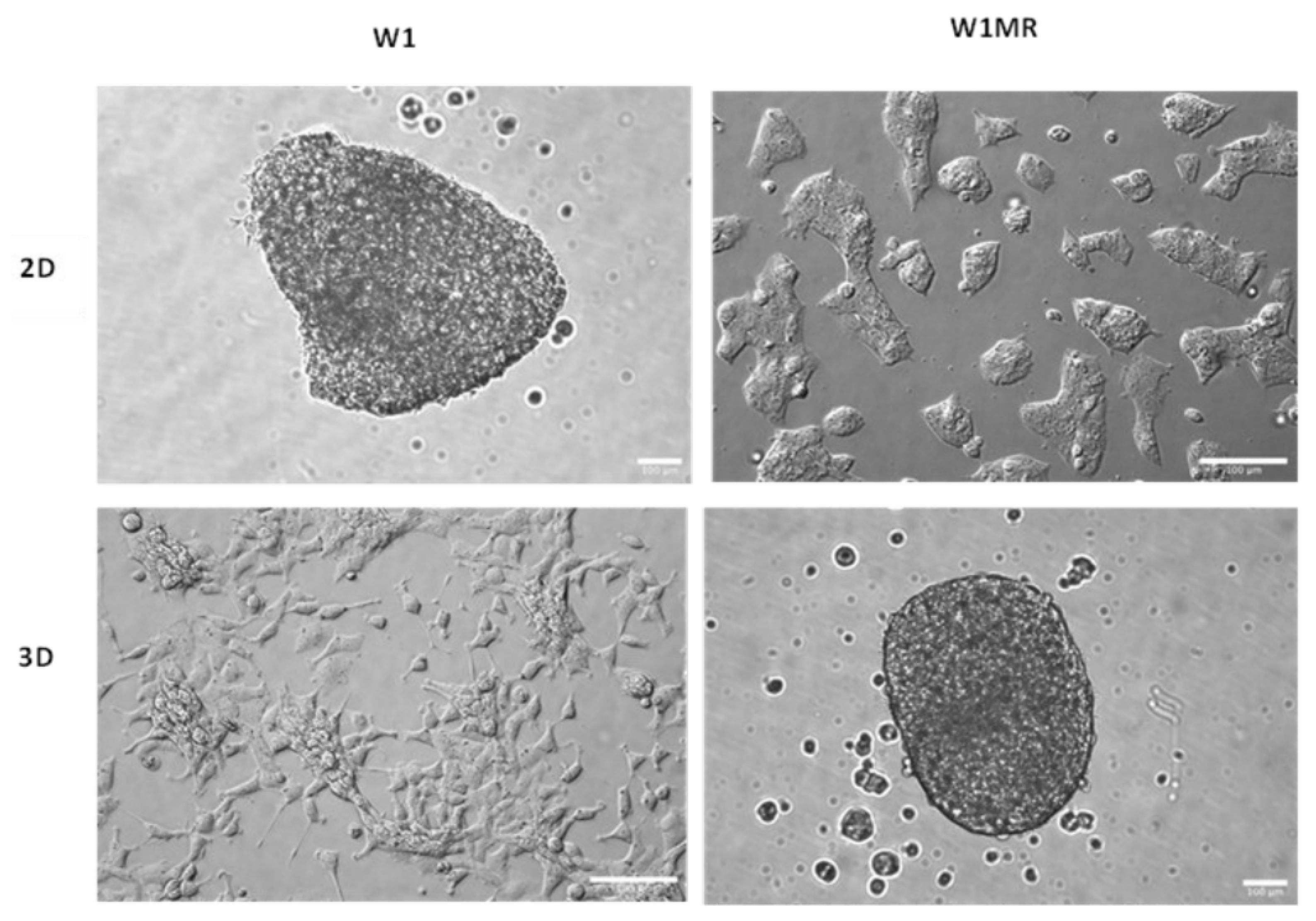
2.8. The Morphology of Cell Model Grown in a 2D and 3D Condition
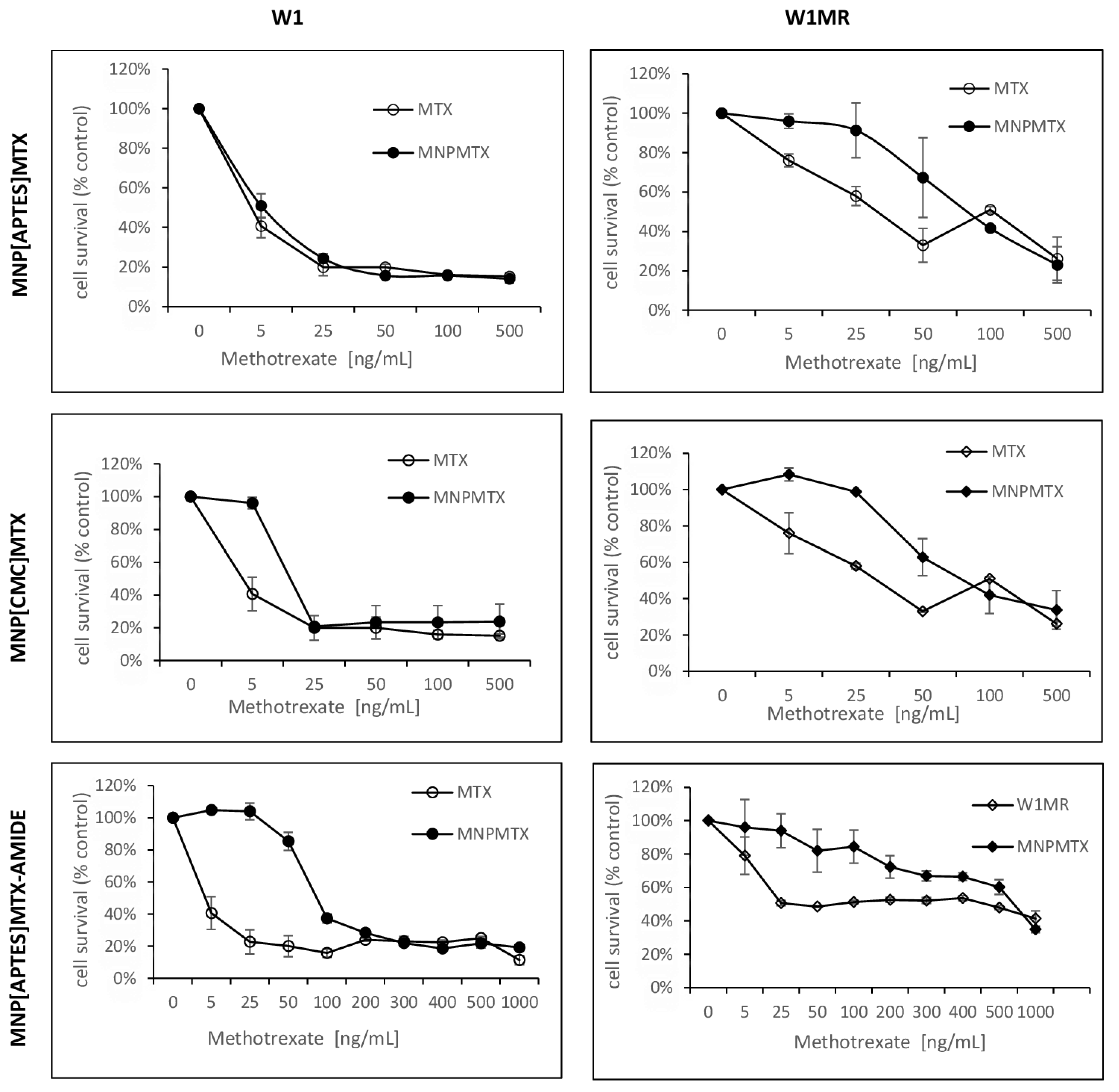
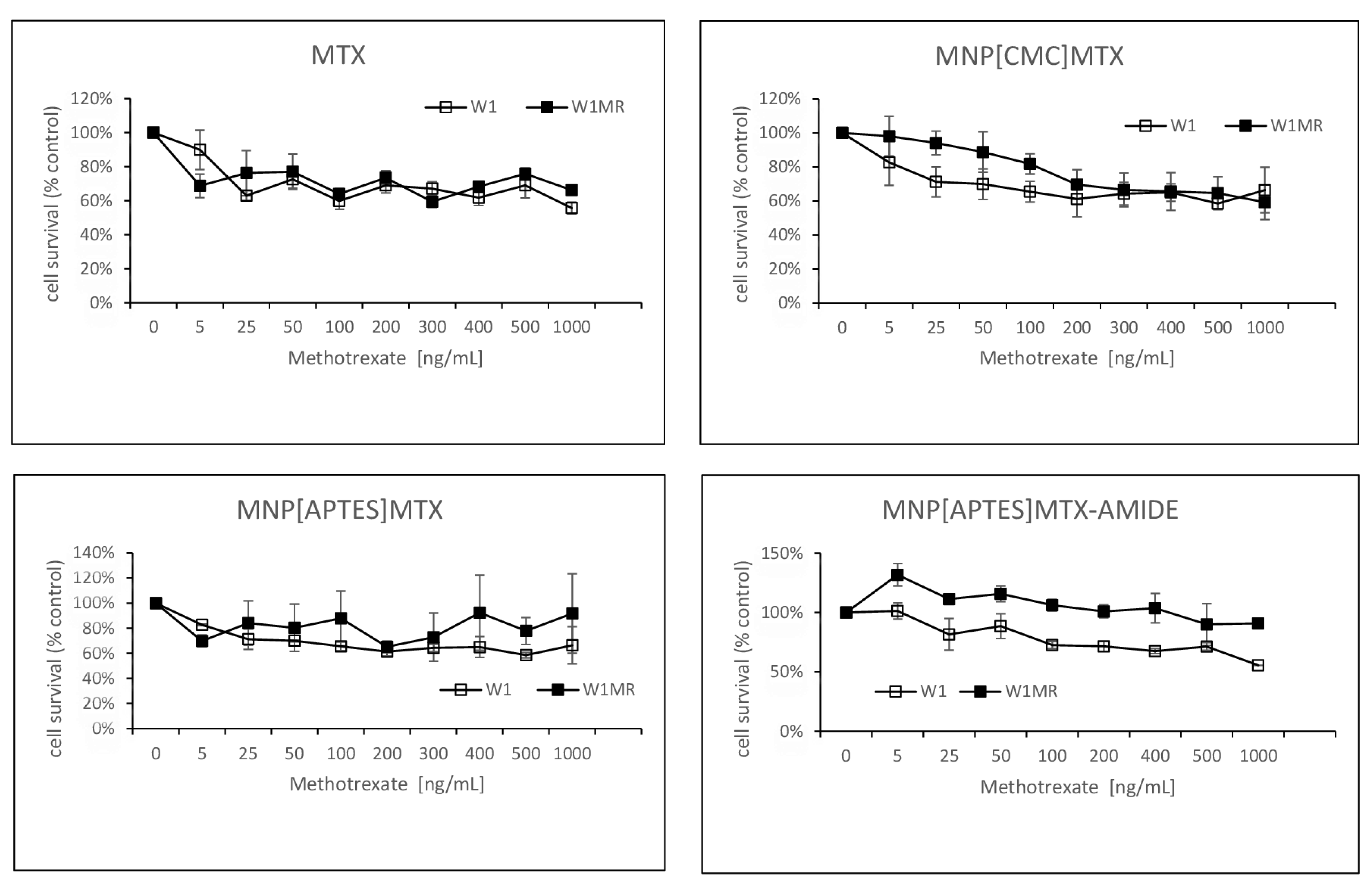
2.9. The Analysis of Methotrexate-Sensitive and Methotrexate–Resistant Cell Lines Viability to Methotrexate Conjugated with Nanoparticles in 2D Culture Condition
2.10. The Analysis of Methotrexate-Sensitive and Methotrexate–Resistant Cell Lines Viability to Methotrexate Conjugated with Nanoparticles in 3D Culture Methods
3. Discussion
4. Materials and Methods
4.1. Synthesis of Magnetic Fe3O4 Nanoparticles (MNPs)
4.2. Surface Modification of Nanoparticles with (3- Aminopropyl)trimethoxysilane (APTES) and N-Carboxymethyl Chitosan (CMC)
4.3. Characterization with FT-IR and Transmission Electron Microscope (TEM) Imaging of APTES- and CMC-Modified Nanoparticles (MNPs[APTES] and CMC[APTES])
4.4. Magnetic VSM Properties Measurements of MNPs[APTES] and MNPs[CMC]
4.5. Hyperthermic Properties of MNPs[APTES] and MNPs[CMC]
4.6. Attaching Methotrexate (MTX) to the APTES-Coated and CMC-Coated MNPs by Ionic Bonds
4.7. Attaching Methotrexate (MTX) to the APTES-Coated MNPs by Amide Bond
4.8. DLS and Zeta Potential Measurements of MNPs, MNPs[APTES], MNPs[CMC] and MTX-Functionalized Nanoparticles (MNPs[APTES]MTX and MNPs[CMC]MTX)
4.9. Investigation of MTX Release from MNPs[APTES]MTX and MNPs[CMC]MTX (Ionic bond)
4.10. Investigation of MTX Release from MNPs[APTES]MTX (Amide Bond)
4.11. Cell Lines and Cell Culture
4.13. MTT Assay
5. Conclusions
Contribution
Acknowledgments
Competing Interests
References
- Bao, Y.; Sherwood, J.A.; Sun, Z. Magnetic Iron Oxide Nanoparticles as T 1 Contrast Agents for Magnetic Resonance Imaging. J. Mater. Chem. C 2018, 6, 1280–1290. [Google Scholar] [CrossRef]
- Huang, J.; Zhong, X.; Wang, L.; Yang, L.; Mao, H. Improving the Magnetic Resonance Imaging Contrast and Detection Methods with Engineered Magnetic Nanoparticles. Theranostics 2012, 2, 86–102. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Wei, Y. For Better or Worse, Iron Overload by Superparamagnetic Iron Oxide Nanoparticles as a MRI Contrast Agent for Chronic Liver Diseases. Chem. Res. Toxicol. 2017, 30, 73–80. [Google Scholar] [CrossRef]
- Russell, E.; Dunne, V.; Russell, B.; Mohamud, H.; Ghita, M.; McMahon, S.J.; Butterworth, K.T.; Schettino, G.; McGarry, C.K.; Prise, K.M. Impact of Superparamagnetic Iron Oxide Nanoparticles on in Vitro and in Vivo Radiosensitisation of Cancer Cells. Radiat Oncol 2021, 16, 104. [Google Scholar] [CrossRef] [PubMed]
- Palzer, J.; Eckstein, L.; Slabu, I.; Reisen, O.; Neumann, U.P.; Roeth, A.A. Iron Oxide Nanoparticle-Based Hyperthermia as a Treatment Option in Various Gastrointestinal Malignancies. Nanomaterials 2021, 11, 3013. [Google Scholar] [CrossRef] [PubMed]
- Obaidat, I.M.; Narayanaswamy, V.; Alaabed, S.; Sambasivam, S.; Muralee Gopi, C.V.V. Principles of Magnetic Hyperthermia: A Focus on Using Multifunctional Hybrid Magnetic Nanoparticles. Magnetochemistry 2019, 5, 67. [Google Scholar] [CrossRef]
- Mu, X.; Li, J.; Yan, S.; Zhang, H.; Zhang, W.; Zhang, F.; Jiang, J. siRNA Delivery with Stem Cell Membrane-Coated Magnetic Nanoparticles for Imaging-Guided Photothermal Therapy and Gene Therapy. ACS Biomater. Sci. Eng. 2018, 4, 3895–3905. [Google Scholar] [CrossRef] [PubMed]
- Uskoković, V.; Tang, S.; Wu, V.M. Targeted Magnetic Separation of Biomolecules and Cells Using Earthicle-Based Ferrofluids. Nanoscale 2019, 11, 11236–11253. [Google Scholar] [CrossRef] [PubMed]
- El-Boubbou, K. Magnetic Iron Oxide Nanoparticles as Drug Carriers: Clinical Relevance. Nanomedicine 2018, 13, 953–971. [Google Scholar] [CrossRef]
- Bukowski, K.; Kciuk, M.; Kontek, R. Mechanisms of Multidrug Resistance in Cancer Chemotherapy. IJMS 2020, 21, 3233. [Google Scholar] [CrossRef]
- Duan, C.; Yu, M.; Xu, J.; Li, B.-Y.; Zhao, Y.; Kankala, R.K. Overcoming Cancer Multi-Drug Resistance (MDR): Reasons, Mechanisms, Nanotherapeutic Solutions, and Challenges. Biomedicine & Pharmacotherapy 2023, 162, 114643. [Google Scholar] [CrossRef]
- Fan, J.; To, K.K.W.; Chen, Z.-S.; Fu, L. ABC Transporters Affects Tumor Immune Microenvironment to Regulate Cancer Immunotherapy and Multidrug Resistance. Drug Resistance Updates 2023, 66, 100905. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.R.; Pattni, B.S.; Abouzeid, A.H.; Torchilin, V.P. Nanopreparations to Overcome Multidrug Resistance in Cancer. Advanced Drug Delivery Reviews 2013, 65, 1748–1762. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, J.I.; Williams, R.T.; Henderson, M.J.; Norris, M.D.; Haber, M. ABC Transporters as Mediators of Drug Resistance and Contributors to Cancer Cell Biology. Drug Resistance Updates 2016, 26, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Mansoori, B.; Mohammadi, A.; Davudian, S.; Shirjang, S.; Baradaran, B. The Different Mechanisms of Cancer Drug Resistance: A Brief Review. Adv Pharm Bull 2017, 7, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Correia, A.L.; Bissell, M.J. The Tumor Microenvironment Is a Dominant Force in Multidrug Resistance. Drug Resistance Updates 2012, 15, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Rahmanian, M.; Seyfoori, A.; Ghasemi, M.; Shamsi, M.; Kolahchi, A.R.; Modarres, H.P.; Sanati-Nezhad, A.; Majidzadeh-A, K. In-Vitro Tumor Microenvironment Models Containing Physical and Biological Barriers for Modelling Multidrug Resistance Mechanisms and Multidrug Delivery Strategies. Journal of Controlled Release 2021, 334, 164–177. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.; Saggar, J.K.; Yu, M.; Wang, M.; Tannock, I.F. Mechanisms of Drug Resistance Related to the Microenvironment of Solid Tumors and Possible Strategies to Inhibit Them. The Cancer Journal 2015, 21, 254–262. [Google Scholar] [CrossRef]
- Muz, B.; De La Puente, P.; Azab, F.; Azab, A.K. The Role of Hypoxia in Cancer Progression, Angiogenesis, Metastasis, and Resistance to Therapy. HP 2015, 83. [Google Scholar] [CrossRef] [PubMed]
- Padera, T.P.; Meijer, E.F.J.; Munn, L.L. The Lymphatic System in Disease Processes and Cancer Progression. Annu. Rev. Biomed. Eng. 2016, 18, 125–158. [Google Scholar] [CrossRef] [PubMed]
- Turley, S.J.; Cremasco, V.; Astarita, J.L. Immunological Hallmarks of Stromal Cells in the Tumour Microenvironment. Nat Rev Immunol 2015, 15, 669–682. [Google Scholar] [CrossRef]
- Westhoff, M.A.; Zhou, S.; Bachem, M.G.; Debatin, K.M.; Fulda, S. Identification of a Novel Switch in the Dominant Forms of Cell Adhesion-Mediated Drug Resistance in Glioblastoma Cells. Oncogene 2008, 27, 5169–5181. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Mayea, Y.; Mir, C.; Masson, F.; Paciucci, R.; LLeonart, M.E. Insights into New Mechanisms and Models of Cancer Stem Cell Multidrug Resistance. Seminars in Cancer Biology 2020, 60, 166–180. [Google Scholar] [CrossRef] [PubMed]
- Nowacka, M.; Sterzynska, K.; Andrzejewska, M.; Nowicki, M.; Januchowski, R. Drug Resistance Evaluation in Novel 3D in Vitro Model. Biomedicine & Pharmacotherapy 2021, 138, 111536. [Google Scholar] [CrossRef]
- Kapałczyńska, M.; Kolenda, T.; Przybyła, W.; Zajączkowska, M.; Teresiak, A.; Filas, V.; Ibbs, M.; Bliźniak, R.; Łuczewski, Ł.; Lamperska, K. 2D and 3D Cell Cultures – a Comparison of Different Types of Cancer Cell Cultures. aoms 2016. [Google Scholar] [CrossRef] [PubMed]
- Weaver, V.M.; Lelièvre, S.; Lakins, J.N.; Chrenek, M.A.; Jones, J.C.R.; Giancotti, F.; Werb, Z.; Bissell, M.J. Β4 Integrin-Dependent Formation of Polarized Three-Dimensional Architecture Confers Resistance to Apoptosis in Normal and Malignant Mammary Epithelium. Cancer Cell 2002, 2, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Pampaloni, F.; Reynaud, E.G.; Stelzer, E.H.K. The Third Dimension Bridges the Gap between Cell Culture and Live Tissue. Nat Rev Mol Cell Biol 2007, 8, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Kato, M.; Shiue, L.; Shively, J.E.; Ares, M.; Lin, R.-J. Cell Type and Culture Condition–Dependent Alternative Splicing in Human Breast Cancer Cells Revealed by Splicing-Sensitive Microarrays. Cancer Research 2006, 66, 1990–1999. [Google Scholar] [CrossRef] [PubMed]
- Świerczewska, M.; Sterzyńska, K.; Ruciński, M.; Andrzejewska, M.; Nowicki, M.; Januchowski, R. The Response and Resistance to Drugs in Ovarian Cancer Cell Lines in 2D Monolayers and 3D Spheroids. Biomedicine & Pharmacotherapy 2023, 165, 115152. [Google Scholar] [CrossRef]
- Lee, J.; Cuddihy, M.J.; Kotov, N.A. Three-Dimensional Cell Culture Matrices: State of the Art. Tissue Engineering Part B: Reviews 2008, 14, 61–86. [Google Scholar] [CrossRef] [PubMed]
- Vinci, M.; Gowan, S.; Boxall, F.; Patterson, L.; Zimmermann, M.; Court, W.; Lomas, C.; Mendiola, M.; Hardisson, D.; Eccles, S.A. Advances in Establishment and Analysis of Three-Dimensional Tumor Spheroid-Based Functional Assays for Target Validation and Drug Evaluation. BMC Biol 2012, 10, 29. [Google Scholar] [CrossRef] [PubMed]
- Marushima, H. Three-Dimensional Culture Promotes Reconstitution of the Tumor-Specific Hypoxic Microenvironment under TGFβ Stimulation. Int J Oncol 2011. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Yin, J.-J.; Wamer, W.G.; Zeng, M.; Lo, Y.M. Reactive Oxygen Species-Related Activities of Nano-Iron Metal and Nano-Iron Oxides. Journal of Food and Drug Analysis 2014, 22, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Sabouri, Z.; Sabouri, M.; Moghaddas, S.S.T.H.; Darroudi, M. Design and Preparation of Amino-Functionalized Core-Shell Magnetic Nanoparticles for Photocatalytic Application and Investigation of Cytotoxicity Effects. J Environ Health Sci Engineer 2022, 21, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Ghutepatil, P.R.; Salunkhe, A.B.; Khot, V.M.; Pawar, S.H. APTES (3-Aminopropyltriethoxy Silane) Functionalized MnFe2O4 Nanoparticles: A Potential Material for Magnetic Fluid Hyperthermia. Chem. Pap. 2019, 73, 2189–2197. [Google Scholar] [CrossRef]
- Comanescu, C. Recent Advances in Surface Functionalization of Magnetic Nanoparticles. Coatings 2023, 13, 1772. [Google Scholar] [CrossRef]
- Wildeboer, R.R.; Southern, P.; Pankhurst, Q.A. On the Reliable Measurement of Specific Absorption Rates and Intrinsic Loss Parameters in Magnetic Hyperthermia Materials. J. Phys. D: Appl. Phys. 2014, 47, 495003. [Google Scholar] [CrossRef]
- Lachowicz, D.; Górka, W.; Kmita, A.; Bernasik, A.; Żukrowski, J.; Szczerba, W.; Sikora, M.; Kapusta, C.; Zapotoczny, S. Enhanced Hyperthermic Properties of Biocompatible Zinc Ferrite Nanoparticles with a Charged Polysaccharide Coating. J. Mater. Chem. B 2019, 7, 2962–2973. [Google Scholar] [CrossRef]
- Kmita, A.; Lachowicz, D.; Żukrowski, J.; Gajewska, M.; Szczerba, W.; Kuciakowski, J.; Zapotoczny, S.; Sikora, M. One-Step Synthesis of Long Term Stable Superparamagnetic Colloid of Zinc Ferrite Nanorods in Water. Materials 2019, 12, 1048. [Google Scholar] [CrossRef] [PubMed]
- Maguire, C.M.; Rösslein, M.; Wick, P.; Prina-Mello, A. Characterisation of Particles in Solution - a Perspective on Light Scattering and Comparative Technologies. Sci Technol Adv Mater 2018, 19, 732–745. [Google Scholar] [CrossRef] [PubMed]
- De Milito, A.; Fais, S. Tumor Acidity, Chemoresistance and Proton Pump Inhibitors. Future Oncol. 2005, 1, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, M.; Alhadlaq, H.A.; Khan, M.A.M.; Akhtar, Mohd.J. Selective Killing of Cancer Cells by Iron Oxide Nanoparticles Mediated through Reactive Oxygen Species via P53 Pathway. J Nanopart Res, 2013; 15, 1225. [CrossRef]
- Jahanbani, J.; Ghotbi, M.; Shahsavari, F.; Seydi, E.; Rahimi, S.; Pourahmad, J. Selective Anticancer Activity of Superparamagnetic Iron Oxide Nanoparticles (SPIONs) against Oral Tongue Cancer Using in Vitro Methods: The Key Role of Oxidative Stress on Cancerous Mitochondria. J Biochem Mol Toxicol 2020, 34. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Farach-Carson, M.C.; Jia, X. Three-Dimensional in Vitro Tumor Models for Cancer Research and Drug Evaluation. Biotechnology Advances 2014, 32, 1256–1268. [Google Scholar] [CrossRef] [PubMed]
- West, G.W.; Weichselbaum, R.; Little, J.B. Limited Penetration of Methotrexate into Human Osteosarcoma Spheroids as a Proposed Model for Solid Tumor Resistance to Adjuvant Chemotherapy. Cancer Res 1980, 40, 3665–3668. [Google Scholar] [PubMed]
- Massart, R. Preparation of Aqueous Magnetic Liquids in Alkaline and Acidic Media. IEEE Trans. Magn. 1981, 17, 1247–1248. [Google Scholar] [CrossRef]
- Cao, H.; He, J.; Deng, L.; Gao, X. Fabrication of Cyclodextrin-Functionalized Superparamagnetic Fe3O4/Amino-Silane Core–Shell Nanoparticles via Layer-by-Layer Method. Applied Surface Science 2009, 255, 7974–7980. [Google Scholar] [CrossRef]
- Bruvera, I.J.; Mendoza Zélis, P.; Pilar Calatayud, M.; Goya, G.F.; Sánchez, F.H. Determination of the Blocking Temperature of Magnetic Nanoparticles: The Good, the Bad, and the Ugly. Journal of Applied Physics 2015, 118, 184304. [Google Scholar] [CrossRef]
- Zhang, H.; Zeng, D.; Liu, Z. The Law of Approach to Saturation in Ferromagnets Originating from the Magnetocrystalline Anisotropy. Journal of Magnetism and Magnetic Materials 2010, 322, 2375–2380. [Google Scholar] [CrossRef]
- Nowak-Jary, J.; Gronczewska, E.; Worobiec, W. Hampered Binding to Blood Serum Proteins and the Biological Activity of Antimicrobial Peptide Containing N3-(4-Methoxyfumaroyl)-L-2,3-Diaminopropanoic Acid Immobilized on Magnetic Nanoparticles. Pharm Chem J 2018, 51, 985–994. [Google Scholar] [CrossRef]
- Sterzyńska, K.; Kaźmierczak, D.; Klejewski, A.; Świerczewska, M.; Wojtowicz, K.; Nowacka, M.; Brązert, J.; Nowicki, M.; Januchowski, R. Expression of Osteoblast-Specific Factor 2 (OSF-2, Periostin) Is Associated with Drug Resistance in Ovarian Cancer Cell Lines. IJMS 2019, 20, 3927. [Google Scholar] [CrossRef] [PubMed]


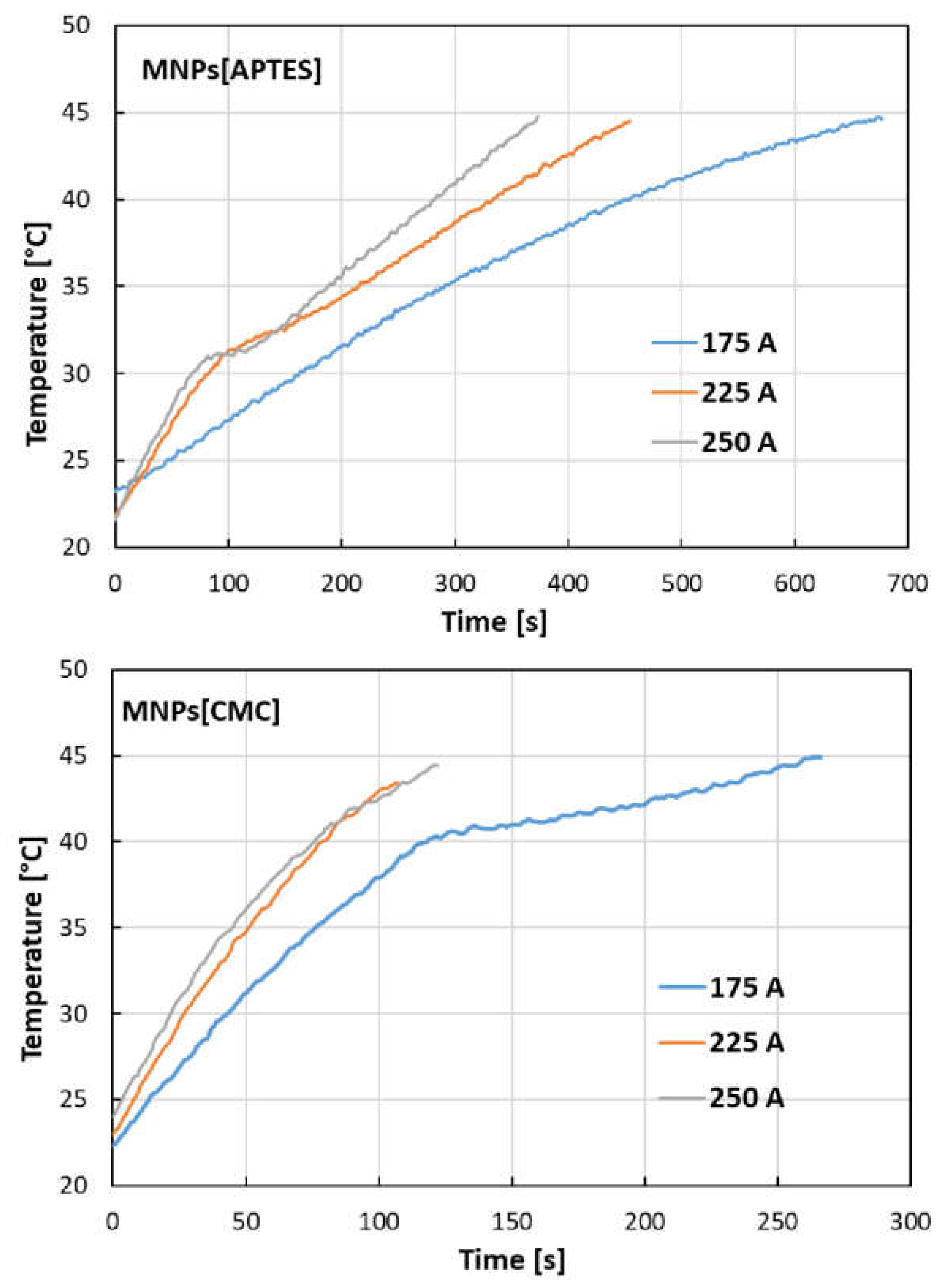
| Aqueous solution of MNPs[APTES] nanoparticles, C = 4 mg MNPs/ml | |||||
| I = 175 A | I = 225 A | I = 250 A | |||
| H [kA/m] | 27.64 | H [kA/m] | 33.83 | H [kA/m] | 36.41 |
| f [kHz] | 356 | f [kHz] | 357 | f [kHz] | 356 |
| SAR (W/g] | 59 | SAR (W/g] | 114 | SAR (W/g] | 154 |
| H [kA/m] | 0.22 | ILP [nH2/kg] | 0.28 | ILP [nH2/kg] | 0.33 |
| Aqueous solution of MNPs[CMC] nanoparticles, C = 4 mg MNPs/ml | |||||
| SAR (W/g] | I = 225 A | I = 250 A | |||
| ILP [nH2/kg] | 27.64 | H [kA/m] | 33.83 | H [kA/m] | 36.41 |
| f [kHz] | 356 | f [kHz] | 357 | f [kHz] | 356 |
| SAR (W/g] | 208 | SAR (W/g] | 314 | SAR (W/g] | 364 |
| ILP [nH2/kg] | 0.76 | ILP [nH2/kg] | 0.77 | ILP [nH2/kg] | 0.77 |
| MNPs[APTES]MTX | ||||
| 37°C | 45°C | |||
| pH = 7.5 | pH = 6.0 | pH = 7.5 | pH = 6.0 | |
| 0 – 60 min I - order kinetics |
k = 0.2008 [min-1] | k = 0.1602 [min-1] | k = 0.1384 [min-1] | k = 0.1350 [min-1] |
| MNPs[CMC]MTX | ||||
| 37°C | 45°C | |||
| pH = 7.5 | pH = 6.0 | pH = 7.5 | pH = 6.0 | |
| 0 – 30 min I - order kinetics |
k = 0.0188 [min-1] | k = 0.0176 [min-1] | ||
| 0 – 15 min I - order kinetics |
k = 0.0173 [min-1] | k = 0.0132 [min-1] | ||
| 30 – 90 min 0 – order kinetics |
k0 = 0.197 [µg/ml×min-1] |
k0 = 0.152 [µg/ml×min-1] |
||
| 15 – 90 min 0 – order kinetics |
k0 = 0.176 [µg/ml×min-1] |
k0 = 0.134 [µg/ml×min-1] |
||
| W1 | W1MR | |
|---|---|---|
| MTX | 4,29 SD ± 0,68 1 |
430,69 SD± 25,47 100,4 ↑*** |
| MNPs[APTES]MTX | 7,74 SD± 2,60 1 |
448,69 SD± 16,69 58,0↑*** |
| MNPs[CMC]MTX | 17,10 SD± 0,22 1 |
409,62 SD± 7,36 24,0↑*** |
| MNPs[APTES]MTXAMID | 86,48 SD± 2,33 1 |
697,05 SD± 55,25 8,1↑*** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




