Submitted:
26 June 2024
Posted:
27 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Hypotheses Explaining Anosmia
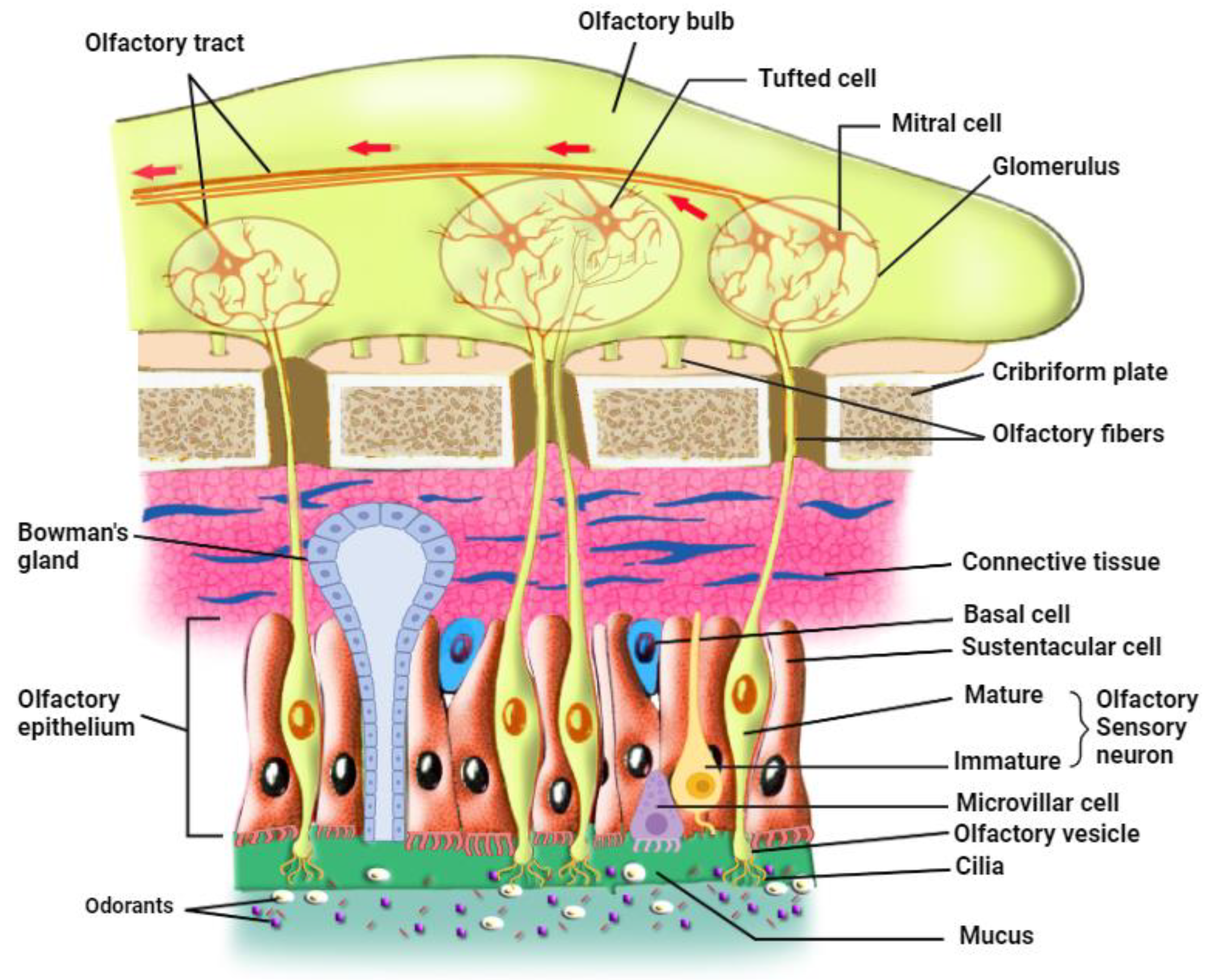
3. The Organization of the Olfactory Epithelium
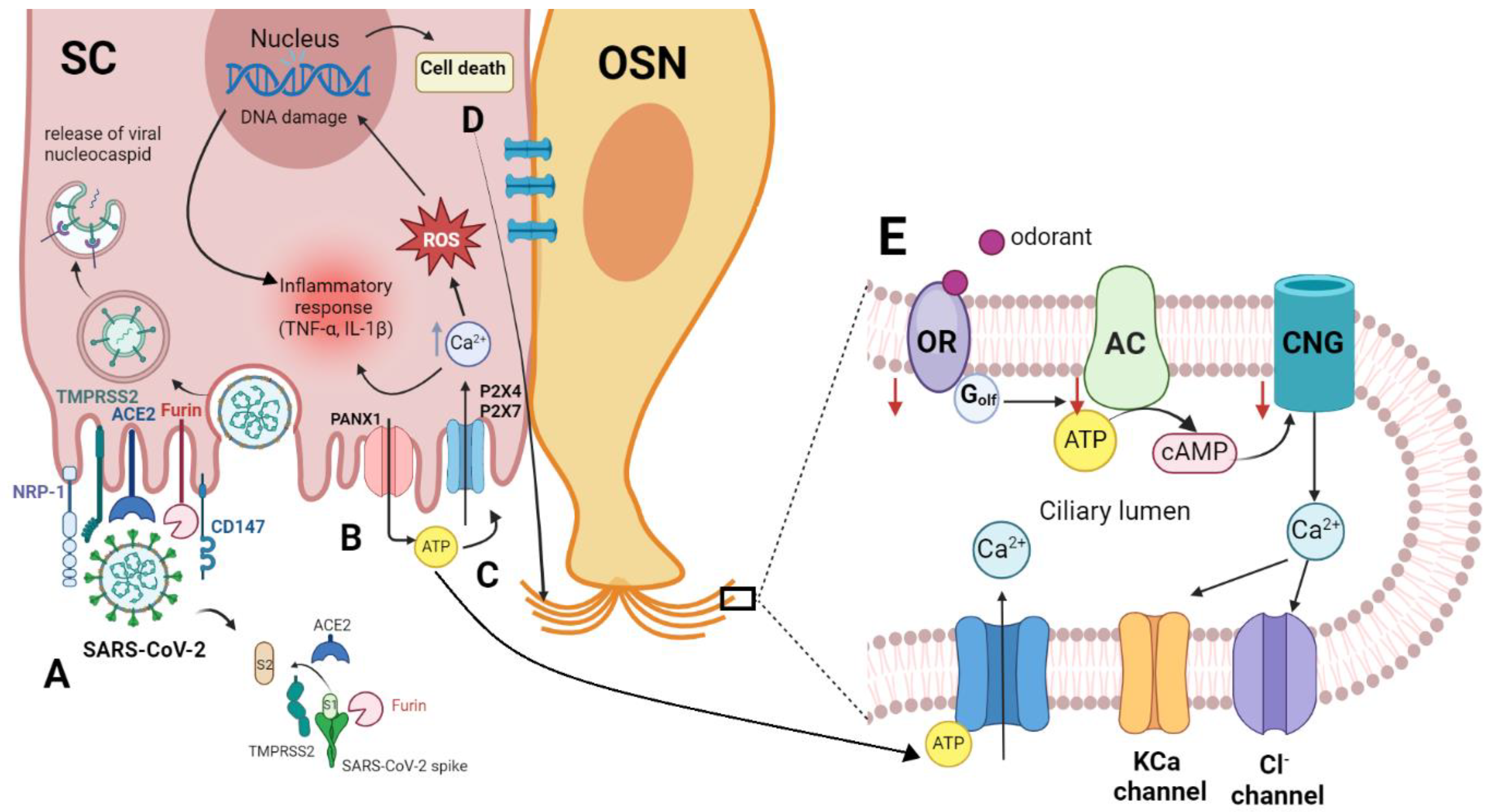
4. ACE2 and TMPRSS2 are the Main Entry Receptors for SARS-CoV-2
5. Neuropilin (NRP1) and Basigin (CD147) are Potential Receptors for Virus Entry
6. Olfactory Receptors Downregulation May be Responsible for Olfactory Dysfunction
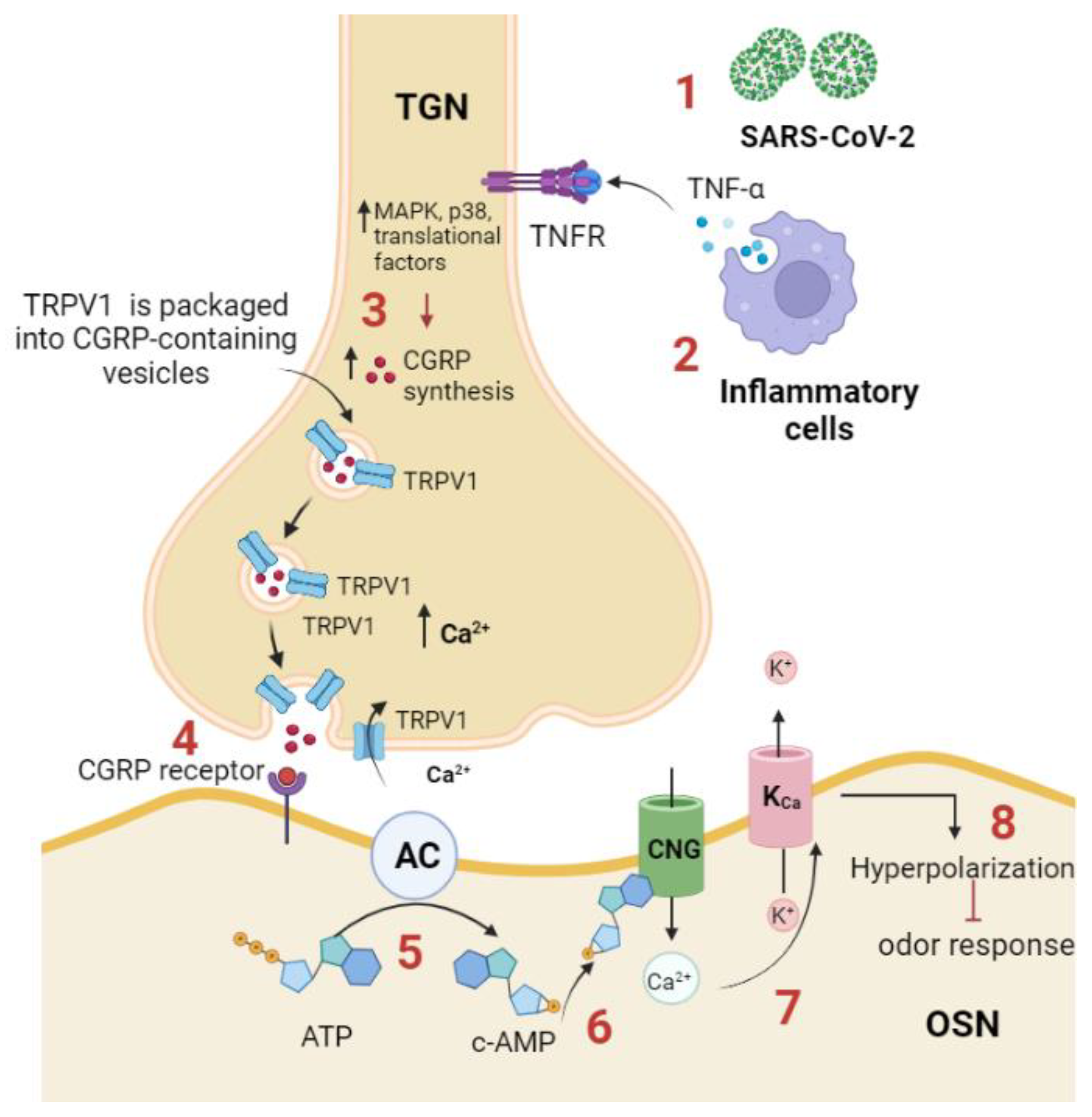
7. Transient Receptor Potential Vanilloid (TRPV1) Trafficking May Lead to Odor Inhibition
8. Purinergic Receptors Involvement in Olfactory Signaling (P2X and P2Y Receptors)
9. Interferon Gamma and Interleukins Receptors in Sustentacular Cells
10. Role of Epithelial Sodium Channel (ENaC) in COVID-19 Induced Anosmia
11. Physiological Role of Oscillatory Calcium Transients in Sustentacular Cells and Signal Transduction through Olfactory Sensory Neurons
12. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brann DH, Tsukahara T, Weinreb C, Lipovsek M, Van den Berge K, Gong B. Non-neuronal expression of SARS-CoV-2 entry genes in the olfactory system suggests mechanisms underlying COVID-19-associated anosmia. Sci Adv. 2020; 6(31): eabc5801.
- Saniasiaya J, Islam MA, Abdullah B. Prevalence of olfactory dysfunction in coronavirus disease 2019 (COVID-19): a meta-analysis of 27,492 patients. Laryngoscope. 2021; 131(4): 865-78.
- Chen, S.; Wang, S. The immune mechanism of the nasal epithelium in COVID-19–related olfactory dysfunction. Front. Immunol. 2023, 14, 1045009. [CrossRef]
- Hoffman, H.J.; Rawal, S.; Li, C.-M.; Duffy, V.B. New chemosensory component in the U.S. National Health and Nutrition Examination Survey (NHANES): first-year results for measured olfactory dysfunction. Rev. Endocr. Metab. Disord. 2016, 17, 221–240. [CrossRef]
- Reichert, J.L.; Schöpf, V. Olfactory Loss and Regain: Lessons for Neuroplasticity. Neurosci. 2018, 24, 22–35. [CrossRef]
- Malik, B.; Elkaddi, N.; Turkistani, J.; I Spielman, A.; Ozdener, M.H. Mammalian Taste Cells Express Functional Olfactory Receptors. Chem. Senses 2019, 44, 289–301. [CrossRef]
- Landis BN, Stow NW, Lacroix JS, Hugentobler M, Hummel T. Olfactory disorders: the patients’ view. Rhinology. 2009; 47(4):454-9.
- Dicpinigaitis PV. Post-viral anosmia (loss of sensation of smell) did not begin with COVID-19! Lung. 2021; 199(3):237-8.
- Doty, R.L. Olfactory dysfunction in COVID-19: pathology and long-term implications for brain health. Trends Mol. Med. 2022, 28, 781–794. [CrossRef]
- Cabrera, C.I.; Hicks, K.; Rodriguez, K.; Stuyt, J.A.G.; McComsey, G.A.; D'Anza, B. Comparison of the incidence of smell and taste disorders between influenza and COVID-19. Am. J. Otolaryngol. 2024, 45, 104176. [CrossRef]
- McWilliams, M.P.; Coelho, D.H.; Reiter, E.R.; Costanzo, R.M. Recovery from Covid-19 smell loss: Two-years of follow up. Am. J. Otolaryngol. 2022, 43, 103607–103607. [CrossRef]
- Lechien JR, Chiesa-Estomba CM, Beckers E, Mustin V, Ducarme M, Journe F, et al. Prevalence and 6-month recovery of olfactory dysfunction: a multicentre study of 1363 COVID-19 patients. J Intern Med. 2021; 290(2):451-461.
- Tan HQ, Pendolino AL, Andrews PJ, Choi D. Prevalence of olfactory dysfunction and quality of life in hospitalised patients 1 year after SARS-CoV-2 infection: a cohort study. BMJ open. 2022; 12(1):e054598.
- Liu, X.; Chiu, S.H.; Tan, C.Y.R.; Chua, A.J.K. Long-term recovery from COVID-19 olfactory and gustatory dysfunction: a longitudinal study. Singap. Med J. 2024. [CrossRef]
- Wise J. Covid-19: Symptomatic infection with omicron variant is milder and shorter than with delta, study reports. BMJ: Brit Med J (Online). 2022; 377.
- Verma AK, Zheng J, Meyerholz DK, Perlman S. SARS-CoV-2 infection of sustentacular cells disrupts olfactory signaling pathways. JCI insight. 2022; 7(24).
- Zazhytska, M.; Kodra, A.; Hoagland, D.A.; Frere, J.; Fullard, J.F.; Shayya, H.; McArthur, N.G.; Moeller, R.; Uhl, S.; Omer, A.D.; et al. Non-cell-autonomous disruption of nuclear architecture as a potential cause of COVID-19-induced anosmia. Cell 2022, 185, 1052–1064.e12. [CrossRef]
- Donadoni, M.; Kaminski, R.; Liao, S.; Al Janabi, S.; Margolskee, R.F.; Ozdener, M.H.; Sariyer, I.K. Coronavirus infection in chemosensory cells. J. NeuroVirology 2023, 29, 35–44. [CrossRef]
- de Melo, G.D.; Lazarini, F.; Levallois, S.; Hautefort, C.; Michel, V.; Larrous, F.; Verillaud, B.; Aparicio, C.; Wagner, S.; Gheusi, G.; et al. COVID-19–related anosmia is associated with viral persistence and inflammation in human olfactory epithelium and brain infection in hamsters. Sci. Transl. Med. 2021, 13. [CrossRef]
- Di Stadio A, Bernitsas E, La Mantia I, Brenner MJ, Ralli M, Vaira LA, et al. Targeting neuroinflammation to alleviate chronic olfactory dysfunction in long covid: a role for investigating disease-modifying therapy (dmt)? Life. 2023; 13(1):226.
- Butowt, R.; Bilinska, K.; von Bartheld, C.S. Olfactory dysfunction in COVID-19: new insights into the underlying mechanisms. Trends Neurosci. 2023, 46, 75–90. [CrossRef]
- Shelton JF, Shastri AJ, Fletez-Brant K, Aslibekyan S, Auton A. The UGT2A1/UGT2A2 locus is associated with COVID-19-related loss of smell or taste. Nat Genet. 2022; 54(2):121-4.
- Tsukahara T, Brann DH, Datta SR. Mechanisms of SARS-CoV-2-associated anosmia. Physiol Rev. 2023; 103(4):2759-66.
- Butowt R, von Bartheld CS. Timing and cause of olfactory deciliation in COVID-19. Physiol Rev. 2024; 104(2): 589-90.
- Bryche B, St Albin A, Murri S, Lacôte S, Pulido C, Ar Gouilh M. Massive transient damage of the olfactory epithelium associated with infection of sustentacular cells by SARS-CoV-2 in golden Syrian hamsters. Brain Behav Immun. 2020; 89:579-86.
- Gupta K, Mohanty SK, Mittal A, Kalra S, Kumar S, Mishra T, et al. The cellular basis of loss of smell in 2019-nCoV-infected individuals. Brief Bioinformatics. 2021; 22(2):873-81.
- Chesler AT, Zou DJ, Le Pichon CE, Peterlin ZA, Matthews GA, Pei X, et al. AG protein/cAMP signal cascade is required for axonal convergence into olfactory glomeruli. Proc Natl Acad Sci U.S.A. 2007; 104(3): 1039-44.
- Rotermund, N.; Schulz, K.; Hirnet, D.; Lohr, C. Purinergic Signaling in the Vertebrate Olfactory System. Front. Cell. Neurosci. 2019, 13, 112. [CrossRef]
- Han, A.Y.; Mukdad, L.; Long, J.L.; Lopez, I.A. Anosmia in COVID-19: Mechanisms and Significance. Chem. Senses 2020, 45, 423–428. [CrossRef]
- Fodoulian L, Tuberosa J, Rossier D, Boillat M, Kan C, Pauli V, et al. SARS-CoV-2 receptors and entry genes are expressed in the human olfactory neuroepithelium and brain. Iscience. 2020; 23(12).
- Hegg, C.C.; Irwin, M.; Lucero, M.T. Calcium store-mediated signaling in sustentacular cells of the mouse olfactory epithelium. Glia 2009, 57, 634–644. [CrossRef]
- Hassenklöver, T.; Schwartz, P.; Schild, D.; Manzini, I. Purinergic Signaling Regulates Cell Proliferation of Olfactory Epithelium Progenitors. STEM CELLS 2009, 27, 2022–2031. [CrossRef]
- Rafols, J.A.; Getchell, T.V. Morphological relations between the receptor neurons, sustentacular cells and Schwann cells in the olfactory mucosa of the salamander. Anat. Rec. 1983, 206, 87–101. [CrossRef]
- Breunig, E.; Czesnik, D.; Piscitelli, F.; Di Marzo, V.; Manzini, I.; Schild, D. Endocannabinoid Modulation in the Olfactory Epithelium. In Sensory and Metabolic Control of Energy Balance; Springer: Berlin/Heidelberg, Germany, 2010; pp. 139–145. [CrossRef]
- Dooley, R.; Mashukova, A.; Toetter, B.; Hatt, H.; Neuhaus, E.M. Purinergic receptor antagonists inhibit odorant-mediated CREB phosphorylation in sustentacular cells of mouse olfactory epithelium. BMC Neurosci. 2011, 12, 86–86. [CrossRef]
- Zhang, C.; Finger, T.E.; Restrepo, D. Mature olfactory receptor neurons express connexin 43. J. Comp. Neurol. 2000, 426, 1–12. [CrossRef]
- DeVries, S.H.; Qi, X.; Smith, R.; Makous, W.; Sterling, P. Electrical Coupling between Mammalian Cones. Curr. Biol. 2002, 12, 1900–1907. [CrossRef]
- Boesveldt S, Postma EM, Boak D, Welge-Luessen A, Schöpf V, Mainland JD, et al. Anosmia—a clinical review. Chemical senses. 2017; 42(7): 513-23.
- Getchell, M.L.; Getchell, T.V. Fine structural aspects of secretion and extrinsic innervation in the olfactory mucosa. Microsc. Res. Tech. 1992, 23, 111–127. [CrossRef]
- Ronnett, G.V.; Moon, C. G Proteins and Olfactory Signal Transduction. Annu. Rev. Physiol. 2002, 64, 189–222. [CrossRef]
- Bilinska, K.; Jakubowska, P.; Von Bartheld, C.S.; Butowt, R. Expression of the SARS-CoV-2 Entry Proteins, ACE2 and TMPRSS2, in Cells of the Olfactory Epithelium: Identification of Cell Types and Trends with Age. ACS Chem. Neurosci. 2020, 11, 1555–1562. [CrossRef]
- Dong, M.; Zhang, J.; Ma, X.; Tan, J.; Chen, L.; Liu, S.; Xin, Y.; Zhuang, L. ACE2, TMPRSS2 distribution and extrapulmonary organ injury in patients with COVID-19. Biomed. Pharmacother. 2020, 131, 110678–110678. [CrossRef]
- Jia, H.P.; Look, D.C.; Shi, L.; Hickey, M.; Pewe, L.; Netland, J.; Farzan, M.; Wohlford-Lenane, C.; Perlman, S.; McCray, P.B., Jr. ACE2 Receptor Expression and Severe Acute Respiratory Syndrome Coronavirus Infection Depend on Differentiation of Human Airway Epithelia. J. Virol. 2005, 79, 14614–14621. [CrossRef]
- Ragia G, Manolopoulos VG. Inhibition of SARS-CoV-2 entry through the ACE2/TMPRSS2 pathway: a promising approach for uncovering early COVID-19 drug therapies. Eur J Clin Pharmacol. 2020; 76:1623-30.
- Li K, Meyerholz DK, Bartlett JA, McCray Jr PB. The TMPRSS2 inhibitor nafamostat reduces SARS-CoV-2 pulmonary infection in mouse models of COVID-19. MBio. 2021; 12(4): e00970-21.
- Singh, H.; Choudhari, R.; Nema, V.; Khan, A.A. ACE2 and TMPRSS2 polymorphisms in various diseases with special reference to its impact on COVID-19 disease. Microb. Pathog. 2021, 150, 104621–104621. [CrossRef]
- Letko M, Marzi A, Munster V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat Microbiol. 2020; 5(4):562-9.
- Chupp G, Spichler-Moffarah A, Søgaard OS, Esserman D, Dziura J, Danzig L, et al. A phase 2 randomized, double-blind, placebo-controlled trial of oral camostat mesylate for early treatment of COVID-19 outpatients showed shorter illness course and attenuation of loss of smell and taste. MedRxiv. 2022; 2022. [CrossRef]
- Daly JL, Simonetti B, Klein K, Chen KE, Williamson MK, Anto’n-Pla’garo C, et al. Neuropilin-1 is a host factor for SARS-CoV-2 infection. Science. 2020; 370 (6518):861-5. [CrossRef]
- Mayi, B.S.; Leibowitz, J.A.; Woods, A.T.; Ammon, K.A.; Liu, A.E.; Raja, A. The role of Neuropilin-1 in COVID-19. PLOS Pathog. 2021, 17, e1009153. [CrossRef]
- Gentzsch, M.; Rossier, B.C. A Pathophysiological Model for COVID-19: Critical Importance of Transepithelial Sodium Transport upon Airway Infection. Function 2020, 1, zqaa024. [CrossRef]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003, 426, 450–454. [CrossRef]
- Ge J, Wang R, Ju B, Zhang Q, Sun J, Chen P, et al. Antibody neutralization of SARS-CoV-2 through ACE2 receptor mimicry. Nat Commun. 2021; 12(1):250.
- Hou YJ, Okuda K, Edwards CE, Martinez DR, Asakura T, Dinnon KH, et al. SARS-CoV-2 reverse genetics reveals a variable infection gradient in the respiratory tract. Cell. 2020; 182(2):429-46.
- Wang KE, Chen W, Zhang Z, Deng Y, Lian JQ, Du P, et al. CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal Transduct Target Ther. 2020; 5(1):283.
- Franciosi, M.L.M.; Lima, M.D.M.; Schetinger, M.R.C.; Cardoso, A.M. Possible role of purinergic signaling in COVID-19. Mol. Cell. Biochem. 2021, 476, 1–8. [CrossRef]
- Nadeali Z, Mohammad-Rezaei F, Aria H, Nikpour P. Possible role of pannexin 1 channels and purinergic receptors in the pathogenesis and mechanism of action of SARS-CoV-2 and therapeutic potential of targeting them in COVID-19. Life Sci. 2022; 297:120482.
- Cantuti-Castelvetri L, Ojha R, Pedro LD, Djannatian M, Franz J, Kuivanen S, et al. Neuropilin-1 facilitates SARS-CoV-2 cell entry and provides a possible pathway into the central nervous system. BioRxiv. 2020; 2020-06.
- Burks SM, Rosas-Hernandez H, Ramirez-Lee MA, Cuevas E, Talpos JC. Can SARS-CoV-2 infect the central nervous system via the olfactory bulb or the blood-brain barrier? Brain Behav Immun. 2021; 95: 7-14.
- Li ZL, Buck M. Neuropilin-1 assists SARS-CoV-2 infection by stimulating the separation of spike protein S1 and S2. Biophys J. 2021; 120(14):2828-37.
- Abebe EC, Ayele TM, Muche ZT, Dejenie TA, Neuropilin 1: A Novel entry factor for SARS-CoV-2 infection and a potential therapeutic target. Biol Targets Ther. 2021; 15: 143-52.
- Alshawaf, E.; Hammad, M.M.; Marafie, S.K.; Ali, H.; Al-Mulla, F.; Abubaker, J.; Mohammad, A. Discovery of natural products to block SARS-CoV-2 S-protein interaction with Neuropilin-1 receptor: A molecular dynamics simulation approach. Microb. Pathog. 2022, 170, 105701–105701. [CrossRef]
- Davies J, Randeva HS, Chatha K, Hall M, Spandidos DA, Karteris E, et al. Neuropilin 1 as a new potential SARS CoV 2 infection mediator implicated in the neurologic features and central nervous system involvement of COVID 19. Mol Med Rep. 2020; 22(5) :4221-6.
- Kang, N.; Koo, J. Olfactory receptors in non-chemosensory tissues. BMB Rep. 2012, 45, 612–622. [CrossRef]
- Bienenstock J, Kunze WA, Forsythe P. Disruptive physiology: olfaction and the microbiome–gut–brain axis. Biol Rev. 2017; 93(1): 390-403.
- Ye Q, Zhou J, He Q, Li RT, Yang G, Zhang Y, et al. SARS-CoV-2 infection in the mouse olfactory system. Cell Discov. 2021; 7(1):49.
- Finlay JB, Brann DH, Abi-Hachem R, Jang DW, Oliva AD, Ko T, et al. Persistent post-COVID-19 smell loss is associated with inflammatory infiltration and altered olfactory epithelial gene expression. BioRxiv. 2022; 2022-04. [CrossRef]
- Mori, I.; Goshima, F.; Imai, Y.; Kohsaka, S.; Sugiyama, T.; Yoshida, T.; Yokochi, T.; Nishiyama, Y.; Kimura, Y. Olfactory receptor neurons prevent dissemination of neurovirulent influenza A virus into the brain by undergoing virus-induced apoptosis. J. Gen. Virol. 2002, 83, 2109–2116. [CrossRef]
- Du, Q.; Liao, Q.; Chen, C.; Yang, X.; Xie, R.; Xu, J. The Role of Transient Receptor Potential Vanilloid 1 in Common Diseases of the Digestive Tract and the Cardiovascular and Respiratory System. Front. Physiol. 2019, 10, 1064. [CrossRef]
- Ahmed, M.K.; Takumida, M.; Ishibashi, T.; Hamamoto, T.; Hirakawa, K. Expression of transient receptor potential vanilloid (TRPV) families 1, 2, 3 and 4 in the mouse olfactory epithelium. Rhinol. J. 2009, 47, 242–247. [CrossRef]
- Messlinger K, Neuhuber W, May A. Activation of the trigeminal system as a likely target of SARS-CoV-2 may contribute to anosmia in COVID-19. Cephalalgia. 2022; 42(2):176-80.
- Torabi, A.; Mohammadbagheri, E.; Akbari Dilmaghani, N.; Bayat, A.-H.; Fathi, M.; Vakili, K.; Alizadeh, R.; Rezaeimirghaed, O.; Hajiesmaeili, M.; Ramezani, M.; et al. Proinflammatory Cytokines in the Olfactory Mucosa Result in COVID-19 Induced Anosmia. ACS Chem. Neurosci. 2020, 11, 1909–1913. [CrossRef]
- Bowen, E.J.; Schmidt, T.W.; Firm, C.S.; Russo, A.F.; Durham, P.L. Tumor necrosis factor-α stimulation of calcitonin gene-related peptide expression and secretion from rat trigeminal ganglion neurons. J. Neurochem. 2006, 96, 65–77. [CrossRef]
- Meng J, Wang J, Steinhoff M, Dolly JO. TNFα induces co-trafficking of TRPV1/TRPA1 in VAMP1-containing vesicles to the plasmalemma via Munc18–1/syntaxin1/SNAP-25 mediated fusion. Sci Rep. 2016; 6(1):21226.
- Daiber, P.; Genovese, F.; Schriever, V.A.; Hummel, T.; Möhrlen, F.; Frings, S. Neuropeptide receptors provide a signalling pathway for trigeminal modulation of olfactory transduction. Eur. J. Neurosci. 2013, 37, 572–582, Epub 2012 Dec 3. PMID: 23205840. [CrossRef]
- Madrid, R.; Delgado, R.; Bacigalupo, J. Cyclic AMP Cascade Mediates the Inhibitory Odor Response of Isolated Toad Olfactory Receptor Neurons. J. Neurophysiol. 2005, 94, 1781–1788. [CrossRef]
- Hassenklöver, T.; Kurtanska, S.; Bartoszek, I.; Junek, S.; Schild, D.; Manzini, I. Nucleotide-induced Ca2+ signaling in sustentacular supporting cells of the olfactory epithelium. Glia 2008, 56, 1614–1624. [CrossRef]
- Gao, L.; Cao, L.; Qiu, Y.; Su, Z.; Burnstock, G.; Xiang, Z.; He, C. Blocking P2X receptors can inhibit the injury-induced proliferation of olfactory epithelium progenitor cells in adult mouse. Int. J. Pediatr. Otorhinolaryngol. 2010, 74, 747–751. [CrossRef]
- Simões JL, Basso HF, Kosvoski GC, Gavioli J, Marafon F, Assmann CE, et al. Targeting purinergic receptors to suppress the cytokine storm induced by SARS-CoV-2 infection in pulmonary tissue. Int Immunopharmacol. 2021; 100:108150.
- Puchałowicz, K.; Tarnowski, M.; Baranowska-Bosiacka, I.; Chlubek, D.; Dziedziejko, V. P2X and P2Y Receptors—Role in the Pathophysiology of the Nervous System. Int. J. Mol. Sci. 2014, 15, 23672–23704. [CrossRef]
- Jia, C.; Doherty, J.; Crudgington, S.; Hegg, C. Activation of purinergic receptors induces proliferation and neuronal differentiation in Swiss Webster mouse olfactory epithelium. Neuroscience 2009, 163, 120–128. [CrossRef]
- Jia, C.; Cussen, A.; Hegg, C. ATP differentially upregulates fibroblast growth factor 2 and transforming growth factor alpha in neonatal and adult mice: effect on neuroproliferation. Neuroscience 2011, 177, 335–346. [CrossRef]
- Rethinavel, H.S.; Ravichandran, S.; Radhakrishnan, R.K.; Kandasamy, M. COVID-19 and Parkinson’s disease: Defects in neurogenesis as the potential cause of olfactory system impairments and anosmia. J. Chem. Neuroanat. 2021, 115, 101965–101965. [CrossRef]
- Conti P, Younes A. Coronavirus COV-19/SARS-CoV-2 affects women less than men: clinical response to viral infection. J Biol Regul Homeost Agents. 2020; 34(2): 339-43.
- Rawish, E.; Langer, H.F. Platelets and the Role of P2X Receptors in Nociception, Pain, Neuronal Toxicity and Thromboinflammation. Int. J. Mol. Sci. 2022, 23, 6585. [CrossRef]
- Hegg, C.C.; Greenwood, D.; Huang, W.; Han, P.; Lucero, M.T. Activation of Purinergic Receptor Subtypes Modulates Odor Sensitivity. J. Neurosci. 2003, 23, 8291–8301. [CrossRef]
- Yu, Y.; Zhang, C. Purinergic signaling negatively regulates activity of an olfactory receptor in an odorant-dependent manner. Neuroscience 2014, 275, 89–101. [CrossRef]
- Bush, C.F.; Jones, S.V.; Lyle, A.N.; Minneman, K.P.; Ressler, K.J.; Hall, R.A. Specificity of Olfactory Receptor Interactions with Other G Protein-coupled Receptors. J. Biol. Chem. 2007, 282, 19042–19051. [CrossRef]
- Yang Q, Tang J, Cao J, Liu F, Fu M, Xue B, et al. SARS-CoV-2 infection activates CREB/CBP in cellular cyclic AMP-dependent pathways. J Med Virol. 2023; 95(1): e28383.
- Watt, W.C.; Sakano, H.; Lee, Z.-Y.; E Reusch, J.; Trinh, K.; Storm, D.R. Odorant Stimulation Enhances Survival of Olfactory Sensory Neurons via MAPK and CREB. Neuron 2004, 41, 955–967. [CrossRef]
- Gallo, O.; Locatello, L.G.; Mazzoni, A.; Novelli, L.; Annunziato, F. The central role of the nasal microenvironment in the transmission, modulation, and clinical progression of SARS-CoV-2 infection. Mucosal Immunol. 2021, 14, 305–316. [CrossRef]
- Ding H, Wang G, Yu Z, Sun H, Wang L. Role of interferon-gamma (IFN-γ) and IFN-γ receptor 1/2 (IFNγR1/2) in regulation of immunity, infection, and cancer development: IFN-γ-dependent or independent pathway. Biomed Pharmacother. 2022; 155:113683.
- Aronsson, F.; Robertson, B.; Ljunggren, H.-G.; Kristensson, K. Invasion and Persistence of the Neuroadapted Influenza Virus A/WSN/33 in the Mouse Olfactory System. Viral Immunol. 2003, 16, 415–423. [CrossRef]
- Hekiert, A.M.; Kofonow, J.M.; Doghramji, L.; Kennedy, D.W.; Chiu, A.G.; Palmer, J.N.; Leid, J.G.; Cohen, N.A. Biofilms Correlate with TH1 Inflammation in ihe Sinonasal Tissue of Patients with Chronic Rhinosinusitis. Otolaryngol. Neck Surg. 2009, 141, 448–453. [CrossRef]
- Pozharskaya, T.; Lane, A.P. Interferon gamma causes olfactory dysfunction without concomitant neuroepithelial damage. Int. Forum Allergy Rhinol. 2013, 3, 861–865. [CrossRef]
- Chen, M.; Reed, R.R.; Lane, A.P. Chronic Inflammation Directs an Olfactory Stem Cell Functional Switch from Neuroregeneration to Immune Defense. Cell Stem Cell 2019, 25, 501–513.e5. [CrossRef]
- Costela-Ruiz VJ, Illescas-Montes R, Puerta-Puerta JM, Ruiz C, Melguizo-Rodríguez L. SARS-CoV-2 infection: the role of cytokines in COVID-19 disease. Cytokine Growth Factor Rev. 2020; 54: 62-75.
- Leist SR, Dinnon KH, Schäfer A, Longping VT, Okuda K, Hou YJ, et al. A mouse-adapted SARS-CoV-2 induces acute lung injury and mortality in standard laboratory mice. Cell. 2020; 183(4):1070-85.
- Cazzolla, A.P.; Lovero, R.; Muzio, L.L.; Testa, N.F.; Schirinzi, A.; Palmieri, G.; Pozzessere, P.; Procacci, V.; Di Comite, M.; Ciavarella, D.; et al. Taste and Smell Disorders in COVID-19 Patients: Role of Interleukin-6. ACS Chem. Neurosci. 2020, 11, 2774–2781. [CrossRef]
- Blanco-Melo D, Nilsson-Payant BE, Liu WC, Uhl S, Hoagland D, Møller R, et al. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell. 2020; 181(5): 1036-45.
- Ward, J.D.; Cornaby, C.; Schmitz, J.L. Indeterminate QuantiFERON Gold Plus Results Reveal Deficient Interferon Gamma Responses in Severely Ill COVID-19 Patients. J. Clin. Microbiol. 2021, 59, e0081121. [CrossRef]
- Anand, P.; Puranik, A.; Aravamudan, M.; Venkatakrishnan, A.; Soundararajan, V. SARS-CoV-2 strategically mimics proteolytic activation of human ENaC. eLife 2020, 9. [CrossRef]
- Rochelle, L.G.; Li, D.C.; Ye, H.; Lee, E.; Talbot, C.R.; Boucher, R.C.; Collawn, J.F.; Matalon, S.; Abdulnour-Nakhoul, S.; Nakhoul, H.N.; et al. Distribution of ion transport mRNAs throughout murine nose and lung. Am. J. Physiol. Cell. Mol. Physiol. 2000, 279, L14–L24. [CrossRef]
- Petrik, D.; Myoga, M.H.; Grade, S.; Gerkau, N.J.; Pusch, M.; Rose, C.R.; Grothe, B.; Götz, M. Epithelial Sodium Channel Regulates Adult Neural Stem Cell Proliferation in a Flow-Dependent Manner. Cell Stem Cell 2018, 22, 865–878.e8. [CrossRef]
- Brown, E.F.; Mitaera, T.; Fronius, M. COVID-19 and Liquid Homeostasis in the Lung—A Perspective through the Epithelial Sodium Channel (ENaC) Lens. Cells 2022, 11, 1801. [CrossRef]
- Grant SN, Lester HA. Regulation of epithelial sodium channel activity by SARS-CoV-1 and SARS-CoV-2 proteins. Biophys j. 2021;120(14):2805-2813.
- Ozdener MH, Mahavadi S, Mummalaneni S, Lyall V. Relationship between ENaC regulators and SARS-CoV-2 virus receptor (ACE2) expression in cultured adult human fungiform (HBO) taste cells. Nutrients. 2022; 14(13):2703.
- Vogalis, F.; Hegg, C.C.; Lucero, M.T. Electrical Coupling in Sustentacular Cells of the Mouse Olfactory Epithelium. J. Neurophysiol. 2005, 94, 1001–1012. [CrossRef]
- Firestein, S. How the olfactory system makes sense of scents. Nature 2001, 413, 211–218. [CrossRef]
- Rebholz H, Braun RJ, Ladage D, Knoll W, Kleber C, Hassel AW. Loss of olfactory function-early indicator for covid-19, other viral infections and neurodegenerative disorders. Front Neurol. 2020; 11:569333.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



