Submitted:
19 June 2024
Posted:
20 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. AML NPM1mut and Prognosis
3. AML NPM1mut Treatment Strategies
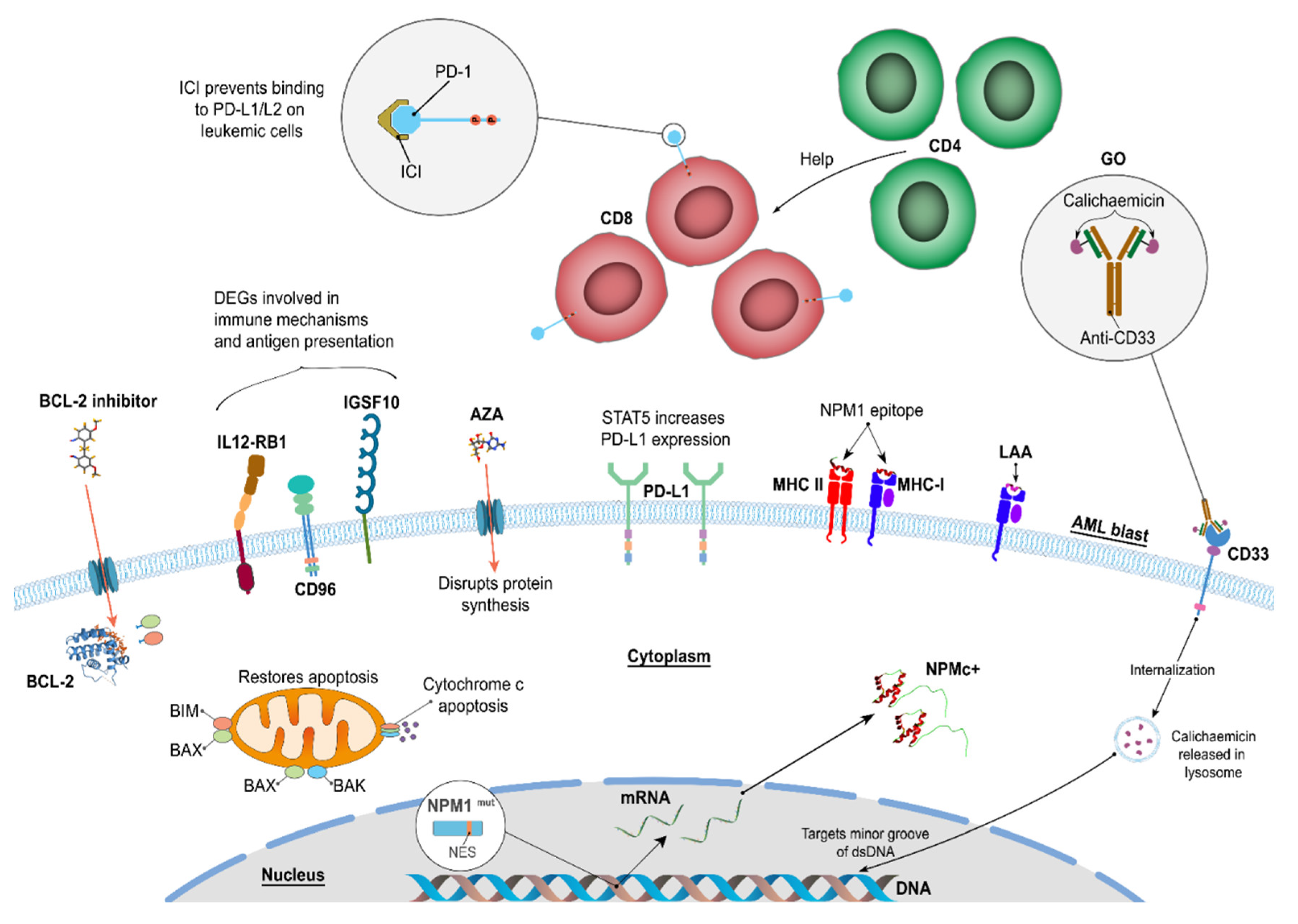
4. Immunogenic Mutation Related Targets
5. Monoclonal Antibody Therapies
5.1. αCD33
5.2. αCD123
5.3. The immune Checkpoint Inhibitors - αPD-1 and αPD-L1
6. Venetoclax and Hypomethylating Agents
7. Discussion
8. Conclusions and Future Developments
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| alloHSCT | allogeneic hematopoietic stem cell transplant | LDAC | low-dose cytarabine |
| AML | Acute myeloid leukemia | LPC/LSC | leukemic progenitor/stem cells |
| APC | Antigen presenting cells | MDS | Myelodysplastic syndrome |
| AZA | 5′-azacitidine | MRD | Minimal residual disease |
| BCL-2 | B-cell leukemia/lymphoma-2 | NPM1 | Nucleophosmin 1 |
| CEBPA | CCAAT/enhancer-binding protein-α | OS | overall survival |
| CR | Complete remission | PD-1 | programmed cell death-1 |
| CT | Chemotherapy | PD-L1 | Programmed cell death 1 ligand 1 |
| FAB | French American British | PDx | PD-1/PD-L1 axis |
| FDA | Food and Drug Administration | PFS | Progression-free survival |
| FKT3-ITD | fms related receptor tyrosine kinase 3-internal tandem duplication | PRAME | Preferentially expressed Antigen in Melanoma |
| GO | Gemtuzumab-Ozogamicin | RHAMM | Receptor for hyaluronan-mediated motility |
| HMA | hypomethylating agents | R/R | Relapsed/refractory |
| ICI | immune checkpoint inhibitor | STAT5 | Signal transducer and activator of transcription |
| LAA | Leukemia associated antigens | WHO | World Health Organization |
References
- Grisendi, S.; Mecucci, C.; Falini, B.; Pandolfi, P.P. Nucleophosmin and cancer. Nat Rev Cancer 2006, 6, 493–505. [Google Scholar] [CrossRef] [PubMed]
- Zarka, J.; Short, N.J.; Kanagal-Shamanna, R.; Issa, G.C. Nucleophosmin 1 Mutations in Acute Myeloid Leukemia. Genes (Basel) 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.; Nassiri, M. Acute Promyelocytic Leukemia: A Review and Discussion of Variant Translocations. Arch Pathol Lab Med 2015, 139, 1308–1313. [Google Scholar] [CrossRef] [PubMed]
- Raimondi, S.C.; Dube, I.D.; Valentine, M.B.; Mirro, J., Jr.; Watt, H.J.; Larson, R.A.; Bitter, M.A.; Le Beau, M.M.; Rowley, J.D. Clinicopathologic manifestations and breakpoints of the t(3;5) in patients with acute nonlymphocytic leukemia. Leukemia 1989, 3, 42–47. [Google Scholar] [PubMed]
- Dumbar, T.S.; Gentry, G.A.; Olson, M.O. Interaction of nucleolar phosphoprotein B23 with nucleic acids. Biochemistry 1989, 28, 9495–9501. [Google Scholar] [CrossRef] [PubMed]
- Cordell, J.L.; Pulford, K.A.; Bigerna, B.; Roncador, G.; Banham, A.; Colombo, E.; Pelicci, P.G.; Mason, D.Y.; Falini, B. Detection of normal and chimeric nucleophosmin in human cells. Blood 1999, 93, 632–642. [Google Scholar] [CrossRef] [PubMed]
- Borer, R.A.; Lehner, C.F.; Eppenberger, H.M.; Nigg, E.A. Major nucleolar proteins shuttle between nucleus and cytoplasm. Cell 1989, 56, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Falini, B.; Mecucci, C.; Tiacci, E.; Alcalay, M.; Rosati, R.; Pasqualucci, L.; La Starza, R.; Diverio, D.; Colombo, E.; Santucci, A. Cytoplasmic nucleophosmin in acute myelogenous leukemia with a normal karyotype. New England Journal of Medicine 2005, 352, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Falini, B.; Martelli, M.P.; Bolli, N.; Bonasso, R.; Ghia, E.; Pallotta, M.T.; Diverio, D.; Nicoletti, I.; Pacini, R.; Tabarrini, A. , et al. Immunohistochemistry predicts nucleophosmin (NPM) mutations in acute myeloid leukemia. Blood 2006, 108, 1999–2005. [Google Scholar] [CrossRef] [PubMed]
- Dillon, R.; Hills, R.; Freeman, S.; Potter, N.; Jovanovic, J.; Ivey, A.; Kanda, A.S.; Runglall, M.; Foot, N.; Valganon, M. , et al. Molecular MRD status and outcome after transplantation in NPM1-mutated AML. Blood 2020, 135, 680–688. [Google Scholar] [CrossRef] [PubMed]
- Cela, I.; Di Matteo, A.; Federici, L. Nucleophosmin in Its Interaction with Ligands. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Falini, B.; Brunetti, L.; Sportoletti, P.; Martelli, M.P. NPM1-mutated acute myeloid leukemia: from bench to bedside. Blood 2020, 136, 1707–1721. [Google Scholar] [CrossRef]
- Dohner, H.; Wei, A.H.; Appelbaum, F.R.; Craddock, C.; DiNardo, C.D.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Godley, L.A.; Hasserjian, R.P. , et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood 2022, 140, 1345–1377. [Google Scholar] [CrossRef] [PubMed]
- Huber, S.; Baer, C.; Hutter, S.; Dicker, F.; Fuhrmann, I.; Meggendorfer, M.; Pohlkamp, C.; Kern, W.; Haferlach, T.; Haferlach, C. , et al. Risk assessment according to IPSS-M is superior to AML ELN risk classification in MDS/AML overlap patients defined by ICC. Leukemia 2023, 37, 2138–2141. [Google Scholar] [CrossRef] [PubMed]
- Shimony, S.; Stahl, M.; Stone, R.M. Acute myeloid leukemia: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol 2023, 98, 502–526. [Google Scholar] [CrossRef] [PubMed]
- Kantarjian, H.M.; Kadia, T.M.; DiNardo, C.D.; Welch, M.A.; Ravandi, F. Acute myeloid leukemia: Treatment and research outlook for 2021 and the MD Anderson approach. Cancer 2021, 127, 1186–1207. [Google Scholar] [CrossRef] [PubMed]
- Verhaak, R.G.; Goudswaard, C.S.; van Putten, W.; Bijl, M.A.; Sanders, M.A.; Hugens, W.; Uitterlinden, A.G.; Erpelinck, C.A.; Delwel, R.; Lowenberg, B. , et al. Mutations in nucleophosmin (NPM1) in acute myeloid leukemia (AML): association with other gene abnormalities and previously established gene expression signatures and their favorable prognostic significance. Blood 2005, 106, 3747–3754. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Gable, A.L.; Nastou, K.C.; Lyon, D.; Kirsch, R.; Pyysalo, S.; Doncheva, N.T.; Legeay, M.; Fang, T.; Bork, P. , et al. The STRING database in 2021: customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res 2021, 49, D605–D612. [Google Scholar] [CrossRef] [PubMed]
- Falini, B.; Martelli, M.P.; Bolli, N.; Sportoletti, P.; Liso, A.; Tiacci, E.; Haferlach, T. Acute myeloid leukemia with mutated nucleophosmin (NPM1): is it a distinct entity? Blood 2011, 117, 1109–1120. [Google Scholar] [CrossRef] [PubMed]
- Papaemmanuil, E.; Gerstung, M.; Bullinger, L.; Gaidzik, V.I.; Paschka, P.; Roberts, N.D.; Potter, N.E.; Heuser, M.; Thol, F.; Bolli, N. , et al. Genomic Classification and Prognosis in Acute Myeloid Leukemia. N Engl J Med 2016, 374, 2209–2221. [Google Scholar] [CrossRef] [PubMed]
- Borrow, J.; Dyer, S.A.; Akiki, S.; Griffiths, M.J. Molecular roulette: nucleophosmin mutations in AML are orchestrated through N-nucleotide addition by TdT. Blood 2019, 134, 2291–2303. [Google Scholar] [CrossRef] [PubMed]
- Heath, E.M.; Chan, S.M.; Minden, M.D.; Murphy, T.; Shlush, L.I.; Schimmer, A.D. Biological and clinical consequences of NPM1 mutations in AML. Leukemia 2017, 31, 798–807. [Google Scholar] [CrossRef] [PubMed]
- Juliusson, G.; Jadersten, M.; Deneberg, S.; Lehmann, S.; Mollgard, L.; Wennstrom, L.; Antunovic, P.; Cammenga, J.; Lorenz, F.; Olander, E. , et al. The prognostic impact of FLT3-ITD and NPM1 mutation in adult AML is age-dependent in the population-based setting. Blood Adv 2020, 4, 1094–1101. [Google Scholar] [CrossRef] [PubMed]
- Schlenk, R.F.; Döhner, K.; Krauter, J.; Fröhling, S.; Corbacioglu, A.; Bullinger, L.; Habdank, M.; Späth, D.; Morgan, M.; Benner, A. Mutations and treatment outcome in cytogenetically normal acute myeloid leukemia. New England Journal of Medicine 2008, 358, 1909–1918. [Google Scholar] [CrossRef] [PubMed]
- Angenendt, L.; Rollig, C.; Montesinos, P.; Ravandi, F.; Juliusson, G.; Recher, C.; Itzykson, R.; Racil, Z.; Wei, A.H.; Schliemann, C. Revisiting coexisting chromosomal abnormalities in NPM1-mutated AML in light of the revised ELN 2022 classification. Blood 2023, 141, 433–435. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, R.; Pianigiani, G.; Sciabolacci, S.; Perriello, V.M.; Marra, A.; Cardinali, V.; Pierangeli, S.; Milano, F.; Gionfriddo, I.; Brunetti, L. , et al. Current status and future perspectives in targeted therapy of NPM1-mutated AML. Leukemia 2022, 36, 2351–2367. [Google Scholar] [CrossRef] [PubMed]
- Schneider, V.; Zhang, L.; Bullinger, L.; Rojewski, M.; Hofmann, S.; Wiesneth, M.; Schrezenmeier, H.; Götz, M.; Botzenhardt, U.; Barth, T.F.E. , et al. Leukemic stem cells of acute myeloid leukemia patients carrying NPM1 mutation are candidates for targeted immunotherapy. Leukemia 2014, 28, 1759–1762. [Google Scholar] [CrossRef] [PubMed]
- Gilliland, D.G.; Griffin, J.D. The roles of FLT3 in hematopoiesis and leukemia. Blood 2002, 100, 1532–1542. [Google Scholar] [CrossRef] [PubMed]
- Levis, M.; Small, D. FLT3: ITDoes matter in leukemia. Leukemia 2003, 17, 1738–1752. [Google Scholar] [CrossRef] [PubMed]
- Shiah, H.S.; Kuo, Y.Y.; Tang, J.L.; Huang, S.Y.; Yao, M.; Tsay, W.; Chen, Y.C.; Wang, C.H.; Shen, M.C.; Lin, D.T. , et al. Clinical and biological implications of partial tandem duplication of the MLL gene in acute myeloid leukemia without chromosomal abnormalities at 11q23. Leukemia 2002, 16, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Dohner, K.; Tobis, K.; Ulrich, R.; Frohling, S.; Benner, A.; Schlenk, R.F.; Dohner, H. Prognostic significance of partial tandem duplications of the MLL gene in adult patients 16 to 60 years old with acute myeloid leukemia and normal cytogenetics: a study of the Acute Myeloid Leukemia Study Group Ulm. J Clin Oncol 2002, 20, 3254–3261. [Google Scholar] [CrossRef] [PubMed]
- Barjesteh van Waalwijk van Doorn-Khosrovani, S.; Erpelinck, C.; van Putten, W.L.; Valk, P.J.; van der Poel-van de Luytgaarde, S.; Hack, R.; Slater, R.; Smit, E.M.; Beverloo, H.B.; Verhoef, G. , et al. High EVI1 expression predicts poor survival in acute myeloid leukemia: a study of 319 de novo AML patients. Blood 2003, 101, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Gregory, T.K.; Wald, D.; Chen, Y.; Vermaat, J.M.; Xiong, Y.; Tse, W. Molecular prognostic markers for adult acute myeloid leukemia with normal cytogenetics. Journal of Hematology & Oncology 2009, 2, 23. [Google Scholar] [CrossRef]
- Döhner, K.; Paschka, P. Intermediate-risk acute myeloid leukemia therapy: current and future. Hematology 2014, 2014, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Döhner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Büchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood, The Journal of the American Society of Hematology 2017, 129, 424–447. [Google Scholar] [CrossRef] [PubMed]
- Preudhomme, C.; Sagot, C.; Boissel, N.; Cayuela, J.M.; Tigaud, I.; de Botton, S.; Thomas, X.; Raffoux, E.; Lamandin, C.; Castaigne, S. , et al. Favorable prognostic significance of CEBPA mutations in patients with de novo acute myeloid leukemia: a study from the Acute Leukemia French Association (ALFA). Blood 2002, 100, 2717–2723. [Google Scholar] [CrossRef] [PubMed]
- Barjesteh van Waalwijk van Doorn-Khosrovani, S.; Erpelinck, C.; Meijer, J.; van Oosterhoud, S.; van Putten, W.L.; Valk, P.J.; Berna Beverloo, H.; Tenen, D.G.; Lowenberg, B.; Delwel, R. Biallelic mutations in the CEBPA gene and low CEBPA expression levels as prognostic markers in intermediate-risk AML. Hematol J 2003, 4, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, S.; Götz, M.; Schneider, V.; Guillaume, P.; Bunjes, D.; Döhner, H.; Wiesneth, M.; Greiner, J. Donor lymphocyte infusion induces polyspecific CD8+ T-cell responses with concurrent molecular remission in acute myeloid leukemia with NPM1 mutation. Journal of Clinical Oncology 2013, 31, e44–e47. [Google Scholar] [CrossRef] [PubMed]
- Jäger, P.; Rautenberg, C.; Kaivers, J.; Kasprzak, A.; Geyh, S.; Baermann, B.-N.; Haas, R.; Germing, U.; Schroeder, T.; Kobbe, G. Allogeneic hematopoietic stem cell transplantation and pre-transplant strategies in patients with NPM1-mutated acute myeloid leukemia: a single center experience. Scientific Reports 2023, 13, 10774. [Google Scholar] [CrossRef] [PubMed]
- Jager, P.; Rautenberg, C.; Kaivers, J.; Kasprzak, A.; Geyh, S.; Baermann, B.N.; Haas, R.; Germing, U.; Schroeder, T.; Kobbe, G. Allogeneic hematopoietic stem cell transplantation and pre-transplant strategies in patients with NPM1-mutated acute myeloid leukemia: a single center experience. Sci Rep 2023, 13, 10774. [Google Scholar] [CrossRef] [PubMed]
- DiNardo, C.D.; Jonas, B.A.; Pullarkat, V.; Thirman, M.J.; Garcia, J.S.; Wei, A.H.; Konopleva, M.; Dohner, H.; Letai, A.; Fenaux, P. , et al. Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. N Engl J Med 2020, 383, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Forghieri, F.; Riva, G.; Lagreca, I.; Barozzi, P.; Bettelli, F.; Paolini, A.; Nasillo, V.; Lusenti, B.; Pioli, V.; Giusti, D. , et al. Neoantigen-Specific T-Cell Immune Responses: The Paradigm of NPM1-Mutated Acute Myeloid Leukemia. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Greiner, J.; Ono, Y.; Hofmann, S.; Schmitt, A.; Mehring, E.; Götz, M.; Guillaume, P.; Döhner, K.; Mytilineos, J.; Döhner, H. Mutated regions of nucleophosmin 1 elicit both CD4+ and CD8+ T-cell responses in patients with acute myeloid leukemia. Blood, The Journal of the American Society of Hematology 2012, 120, 1282–1289. [Google Scholar] [CrossRef] [PubMed]
- Greiner, J.; Schneider, V.; Schmitt, M.; Gotz, M.; Dohner, K.; Wiesneth, M.; Dohner, H.; Hofmann, S. Immune responses against the mutated region of cytoplasmatic NPM1 might contribute to the favorable clinical outcome of AML patients with NPM1 mutations (NPM1mut). Blood 2013, 122, 1087–1088. [Google Scholar] [CrossRef] [PubMed]
- Kuzelova, K.; Brodska, B.; Fuchs, O.; Dobrovolna, M.; Soukup, P.; Cetkovsky, P. Altered HLA Class I Profile Associated with Type A/D Nucleophosmin Mutation Points to Possible Anti-Nucleophosmin Immune Response in Acute Myeloid Leukemia. PLoS One 2015, 10, e0127637. [Google Scholar] [CrossRef] [PubMed]
- Morris, V.S.; Ghazi, H.; Fletcher, D.M.; Guinn, B.A. A Direct Comparison, and Prioritisation, of the Immunotherapeutic Targets Expressed by Adult and Paediatric Acute Myeloid Leukaemia Cells: A Systematic Review and Meta-Analysis. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Guinn, B.A.; Bland, E.A.; Lodi, U.; Liggins, A.P.; Tobal, K.; Petters, S.; Wells, J.W.; Banham, A.H.; Mufti, G.J. Humoral detection of leukaemia-associated antigens in presentation acute myeloid leukaemia. Biochem Biophys Res Commun 2005, 335, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.P.; Sahota, S.S.; Mijovic, A.; Czepulkowski, B.; Padua, R.A.; Mufti, G.J.; Guinn, B.A. Frequent expression of HAGE in presentation chronic myeloid leukaemias. Leukemia 2002, 16, 2238–2242. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.E.; Bonney, S.A.; Lee, C.; Publicover, A.; Khan, G.; Smits, E.L.; Sigurdardottir, D.; Arno, M.; Li, D.; Mills, K.I. , et al. Application of the pMHC Array to Characterise Tumour Antigen Specific T Cell Populations in Leukaemia Patients at Disease Diagnosis. PLoS One 2015, 10, e0140483. [Google Scholar] [CrossRef] [PubMed]
- Hardwick, N.; Buchan, S.; Ingram, W.; Khan, G.; Vittes, G.; Rice, J.; Pulford, K.; Mufti, G.; Stevenson, F.; Guinn, B.A. An analogue peptide from the Cancer/Testis antigen PASD1 induces CD8+ T cell responses against naturally processed peptide. Cancer Immun 2013, 13, 16. [Google Scholar] [PubMed]
- Almshayakhchi, R.; Nagarajan, D.; Vadakekolathu, J.; Guinn, B.A.; Reeder, S.; Brentville, V.; Metheringham, R.; Pockley, A.G.; Durrant, L.; McArdle, S. A Novel HAGE/WT1-ImmunoBody((R)) Vaccine Combination Enhances Anti-Tumour Responses When Compared to Either Vaccine Alone. Front Oncol 2021, 11, 636977. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, M.; Schmitt, A.; Rojewski, M.T.; Chen, J.; Giannopoulos, K.; Fei, F.; Yu, Y.; Gotz, M.; Heyduk, M.; Ritter, G. , et al. RHAMM-R3 peptide vaccination in patients with acute myeloid leukemia, myelodysplastic syndrome, and multiple myeloma elicits immunologic and clinical responses. Blood 2008, 111, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Qazilbash, M.H.; Wieder, E.; Thall, P.F.; Wang, X.; Rios, R.; Lu, S.; Kanodia, S.; Ruisaard, K.E.; Giralt, S.A.; Estey, E.H. , et al. PR1 peptide vaccine induces specific immunity with clinical responses in myeloid malignancies. Leukemia 2017, 31, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Brayer, J.; Lancet, J.E.; Powers, J.; List, A.; Balducci, L.; Komrokji, R.; Pinilla-Ibarz, J. WT1 vaccination in AML and MDS: A pilot trial with synthetic analog peptides. Am J Hematol 2015, 90, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Greiner, J.; Goetz, M.; Schuler, P.J.; Bulach, C.; Hofmann, S.; Schrezenmeier, H.; Dӧhner, H.; Schneider, V.; Guinn, B.A. Enhanced stimulation of antigen-specific immune responses against nucleophosmin 1 mutated acute myeloid leukaemia by an anti-programmed death 1 antibody. Br J Haematol 2022, 198, 866–874. [Google Scholar] [CrossRef] [PubMed]
- Ehninger, A.; Kramer, M.; Röllig, C.; Thiede, C.; Bornhäuser, M.; Von Bonin, M.; Wermke, M.; Feldmann, A.; Bachmann, M.; Ehninger, G. Distribution and levels of cell surface expression of CD33 and CD123 in acute myeloid leukemia. Blood cancer journal 2014, 4, e218–e218. [Google Scholar] [CrossRef]
- Caron, P.C.; Dumont, L.; Scheinberg, D.A. Supersaturating infusional humanized anti-CD33 monoclonal antibody HuM195 in myelogenous leukemia. Clin Cancer Res 1998, 4, 1421–1428. [Google Scholar] [PubMed]
- Raza, A.; Jurcic, J.G.; Roboz, G.J.; Maris, M.; Stephenson, J.J.; Wood, B.L.; Feldman, E.J.; Galili, N.; Grove, L.E.; Drachman, J.G. , et al. Complete remissions observed in acute myeloid leukemia following prolonged exposure to lintuzumab: a phase 1 trial. Leuk Lymphoma 2009, 50, 1336–1344. [Google Scholar] [CrossRef] [PubMed]
- Feldman, E.; Kalaycio, M.; Weiner, G.; Frankel, S.; Schulman, P.; Schwartzberg, L.; Jurcic, J.; Velez-Garcia, E.; Seiter, K.; Scheinberg, D. , et al. Treatment of relapsed or refractory acute myeloid leukemia with humanized anti-CD33 monoclonal antibody HuM195. Leukemia 2003, 17, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Laszlo, G.S.; Estey, E.H.; Walter, R.B. The past and future of CD33 as therapeutic target in acute myeloid leukemia. Blood Reviews 2014, 28, 143–153. [Google Scholar] [CrossRef]
- Gill, S.I. How close are we to CAR T-cell therapy for AML? Best practice & research Clinical haematology 2019, 32, 101104. [Google Scholar]
- Lambert, J.; Pautas, C.; Terre, C.; Raffoux, E.; Turlure, P.; Caillot, D.; Legrand, O.; Thomas, X.; Gardin, C.; Gogat-Marchant, K. , et al. Gemtuzumab ozogamicin for de novo acute myeloid leukemia: final efficacy and safety updates from the open-label, phase III ALFA-0701 trial. Haematologica 2019, 104, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Hills, R.K.; Castaigne, S.; Appelbaum, F.R.; Delaunay, J.; Petersdorf, S.; Othus, M.; Estey, E.H.; Dombret, H.; Chevret, S.; Ifrah, N. , et al. Addition of gemtuzumab ozogamicin to induction chemotherapy in adult patients with acute myeloid leukaemia: a meta-analysis of individual patient data from randomised controlled trials. Lancet Oncol 2014, 15, 986–996. [Google Scholar] [CrossRef] [PubMed]
- Dohner, H.; Weber, D.; Krzykalla, J.; Fiedler, W.; Kuhn, M.W.M.; Schroeder, T.; Mayer, K.; Lubbert, M.; Wattad, M.; Gotze, K. , et al. Intensive chemotherapy with or without gemtuzumab ozogamicin in patients with NPM1-mutated acute myeloid leukaemia (AMLSG 09-09): a randomised, open-label, multicentre, phase 3 trial. Lancet Haematol 2023, 10, e495–e509. [Google Scholar] [CrossRef] [PubMed]
- Schlenk, R.F.; Paschka, P.; Krzykalla, J.; Weber, D.; Kapp-Schwoerer, S.; Gaidzik, V.I.; Leis, C.; Fiedler, W.; Kindler, T.; Schroeder, T. , et al. Gemtuzumab Ozogamicin in NPM1-Mutated Acute Myeloid Leukemia: Early Results From the Prospective Randomized AMLSG 09-09 Phase III Study. J Clin Oncol 2020, 38, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Kapp-Schwoerer, S.; Weber, D.; Corbacioglu, A.; Gaidzik, V.I.; Paschka, P.; Kronke, J.; Theis, F.; Rucker, F.G.; Teleanu, M.V.; Panina, E. , et al. Impact of gemtuzumab ozogamicin on MRD and relapse risk in patients with NPM1-mutated AML: results from the AMLSG 09-09 trial. Blood 2020, 136, 3041–3050. [Google Scholar] [CrossRef] [PubMed]
- Hills, R.K.; Castaigne, S.; Appelbaum, F.R.; Delaunay, J.; Petersdorf, S.; Othus, M.; Estey, E.H.; Dombret, H.; Chevret, S.; Ifrah, N. , et al. Addition of gemtuzumab ozogamicin to induction chemotherapy in adult patients with acute myeloid leukaemia: a meta-analysis of individual patient data from randomised controlled trials. The Lancet Oncology 2014, 15, 986–996. [Google Scholar] [CrossRef] [PubMed]
- Thol, F.; Schlenk, R.F. Gemtuzumab ozogamicin in acute myeloid leukemia revisited. Expert opinion on biological therapy 2014, 14, 1185–1195. [Google Scholar] [CrossRef] [PubMed]
- O’Hear, C.; Inaba, H.; Pounds, S.; Shi, L.; Dahl, G.; Bowman, W.P.; Taub, J.W.; Pui, C.H.; Ribeiro, R.C.; Coustan-Smith, E. Gemtuzumab ozogamicin can reduce minimal residual disease in patients with childhood acute myeloid leukemia. Cancer 2013, 119, 4036–4043. [Google Scholar] [CrossRef] [PubMed]
- Castaigne, S.; Pautas, C.; Terré, C.; Raffoux, E.; Bordessoule, D.; Bastie, J.-N.; Legrand, O.; Thomas, X.; Turlure, P.; Reman, O. Effect of gemtuzumab ozogamicin on survival of adult patients with de-novo acute myeloid leukaemia (ALFA-0701): a randomised, open-label, phase 3 study. The Lancet 2012, 379, 1508–1516. [Google Scholar] [CrossRef]
- Schlenk, R.F.; Paschka, P.; Krzykalla, J.; Weber, D.; Kapp-Schwoerer, S.; Gaidzik, V.I.; Leis, C.; Fiedler, W.; Kindler, T.; Schroeder, T. , et al. Gemtuzumab Ozogamicin in NPM1-Mutated Acute Myeloid Leukemia (AML): Results from the Prospective Randomized AMLSG 09-09 Phase-III Study. Blood 2018, 132, 81. [Google Scholar] [CrossRef]
- Perriello, V.M.; Gionfriddo, I.; Rossi, R.; Milano, F.; Mezzasoma, F.; Marra, A.; Spinelli, O.; Rambaldi, A.; Annibali, O.; Avvisati, G. , et al. CD123 Is Consistently Expressed on NPM1-Mutated AML Cells. Cancers 2021, 13, 496. [Google Scholar] [CrossRef] [PubMed]
- He, S.Z.; Busfield, S.; Ritchie, D.S.; Hertzberg, M.S.; Durrant, S.; Lewis, I.D.; Marlton, P.; McLachlan, A.J.; Kerridge, I.; Bradstock, K.F. A Phase 1 study of the safety, pharmacokinetics and anti-leukemic activity of the anti-CD123 monoclonal antibody CSL360 in relapsed, refractory or high-risk acute myeloid leukemia. Leukemia & lymphoma 2015, 56, 1406–1415. [Google Scholar]
- Pemmaraju, N.; Lane, A.A.; Sweet, K.L.; Stein, A.S.; Vasu, S.; Blum, W.; Rizzieri, D.A.; Wang, E.S.; Duvic, M.; Sloan, J.M. Tagraxofusp in blastic plasmacytoid dendritic-cell neoplasm. New England Journal of Medicine 2019, 380, 1628–1637. [Google Scholar] [CrossRef] [PubMed]
- Daver, N.; Montesinos, P.; Aribi, A.; Marconi, G.; Altman, J.K.; Wang, E.S.; Roboz, G.J.; Burke, P.W.; Gaidano, G.; Walter, R.B. , et al. Broad Activity for the Pivekimab Sunirine (PVEK, IMGN632), Azacitidine, and Venetoclax Triplet in High-Risk Patients with Relapsed/Refractory Acute Myeloid Leukemia (AML). Blood 2022, 140, 145–149. [Google Scholar] [CrossRef]
- Kaur, M.; Drake, A.C.; Hu, G.; Rudnick, S.; Chen, Q.; Phennicie, R.; Attar, R.; Nemeth, J.; Gaudet, F.; Chen, J. Induction and therapeutic targeting of human NPM1c+ myeloid leukemia in the presence of autologous immune system in mice. The Journal of Immunology 2019, 202, 1885–1894. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, J.; Zhang, F.; Liu, P. A perspective of immunotherapy for acute myeloid leukemia: Current advances and challenges. Frontiers in Pharmacology 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Cui, L.; Zhao, X.; Bai, H.; Cai, S.; Wang, G.; Zhao, Z.; Zhao, J.; Chen, S.; Song, J. , et al. Use of Immunotherapy With Programmed Cell Death 1 vs Programmed Cell Death Ligand 1 Inhibitors in Patients With Cancer: A Systematic Review and Meta-analysis. JAMA Oncol 2020, 6, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Han, X. Anti-PD-1/PD-L1 therapy of human cancer: past, present, and future. J Clin Invest 2015, 125, 3384–3391. [Google Scholar] [CrossRef] [PubMed]
- Greiner, J.; Hofmann, S.; Schmitt, M.; Gotz, M.; Wiesneth, M.; Schrezenmeier, H.; Bunjes, D.; Dohner, H.; Bullinger, L. Acute myeloid leukemia with mutated nucleophosmin 1: an immunogenic acute myeloid leukemia subtype and potential candidate for immune checkpoint inhibition. Haematologica 2017, 102, e499–e501. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, R.J.; Loughnan, M.S.; Flynn, E.; Folkman, J. Thalidomide is an inhibitor of angiogenesis. Proc Natl Acad Sci U S A 1994, 91, 4082–4085. [Google Scholar] [CrossRef] [PubMed]
- Guinn, B.A.; Schuler, P.J.; Schrezenmeier, H.; Hofmann, S.; Weiss, J.; Bulach, C.; Gotz, M.; Greiner, J. A Combination of the Immunotherapeutic Drug Anti-Programmed Death 1 with Lenalidomide Enhances Specific T Cell Immune Responses against Acute Myeloid Leukemia Cells. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Greiner, J., Schuler, P.J., Schrezenmeier, H., Weiss, J., Bulach, C., Goetz, M., Guinn, B. Combinations of Different Immunotherapeutics Enhance Specific T Cell Immune Responses Against Leukemic Cells, as well as Leukemic Progenitor and Stem Cells in Acute Myeloid Leukemia In Preparation.
- Huang, Z.W.; Zhang, X.N.; Zhang, L.; Liu, L.L.; Zhang, J.W.; Sun, Y.X.; Xu, J.Q.; Liu, Q.; Long, Z.J. STAT5 promotes PD-L1 expression by facilitating histone lactylation to drive immunosuppression in acute myeloid leukemia. Signal Transduct Target Ther 2023, 8, 391. [Google Scholar] [CrossRef] [PubMed]
- Daver, N.; Garcia-Manero, G.; Basu, S.; Boddu, P.C.; Alfayez, M.; Cortes, J.E.; Konopleva, M.; Ravandi-Kashani, F.; Jabbour, E.; Kadia, T. , et al. Efficacy, Safety, and Biomarkers of Response to Azacitidine and Nivolumab in Relapsed/Refractory Acute Myeloid Leukemia: A Nonrandomized, Open-Label, Phase II Study. Cancer Discov 2019, 9, 370–383. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wong, M.P.M.; Ng, R.K. Aberrant DNA Methylation in Acute Myeloid Leukemia and Its Clinical Implications. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Chin, L.; Wong, C.Y.G.; Gill, H. Targeting and Monitoring Acute Myeloid Leukaemia with Nucleophosmin-1 (NPM1) Mutation. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Lachowiez, C.A.; Loghavi, S.; Zeng, Z.; Tanaka, T.; Kim, Y.J.; Uryu, H.; Turkalj, S.; Jakobsen, N.A.; Luskin, M.R.; Duose, D.Y. , et al. A Phase Ib/II Study of Ivosidenib with Venetoclax +/- Azacitidine in IDH1-Mutated Myeloid Malignancies. Blood Cancer Discov 2023, 4, 276–293. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-C.; Rérolle, D.; Berthier, C.; Hleihel, R.; Sakamoto, T.; Quentin, S.; Benhenda, S.; Morganti, C.; Wu, C.; Conte, L. Actinomycin D targets NPM1c-primed mitochondria to restore PML-driven senescence in AML therapy. Cancer discovery 2021, 11, 3198–3213. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Qin, F.; Yang, L.; Xian, J.; Zou, Q.; Jin, H.; Wang, L.; Zhang, L. Nucleophosmin mutations induce chemosensitivity in THP-1 leukemia cells by suppressing NF-κB activity and regulating Bax/Bcl-2 expression. Journal of Cancer 2016, 7, 2270. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.W.; Davids, M.S.; Pagel, J.M.; Kahl, B.S.; Puvvada, S.D.; Gerecitano, J.F.; Kipps, T.J.; Anderson, M.A.; Brown, J.R.; Gressick, L. Targeting BCL2 with venetoclax in relapsed chronic lymphocytic leukemia. New England Journal of Medicine 2016, 374, 311–322. [Google Scholar] [CrossRef]
- Lachowiez, C.A.; Reville, P.K.; Kantarjian, H.; Jabbour, E.; Borthakur, G.; Daver, N.; Issa, G.; Furudate, K.; Tanaka, T.; Pierce, S. Contemporary outcomes in IDH-mutated acute myeloid leukemia: The impact of co-occurring NPM1 mutations and venetoclax-based treatment. American journal of hematology 2022, 97, 1443–1452. [Google Scholar] [CrossRef]
- Quintás-Cardama, A.; Ravandi, F.; Liu-Dumlao, T.; Brandt, M.; Faderl, S.; Pierce, S.; Borthakur, G.; Garcia-Manero, G.; Cortes, J.; Kantarjian, H. Epigenetic therapy is associated with similar survival compared with intensive chemotherapy in older patients with newly diagnosed acute myeloid leukemia. Blood, The Journal of the American Society of Hematology 2012, 120, 4840–4845. [Google Scholar] [CrossRef]
- Dombret, H.; Seymour, J.F.; Butrym, A.; Wierzbowska, A.; Selleslag, D.; Jang, J.H.; Kumar, R.; Cavenagh, J.; Schuh, A.C.; Candoni, A. International phase 3 study of azacitidine vs conventional care regimens in older patients with newly diagnosed AML with> 30% blasts. Blood, The Journal of the American Society of Hematology 2015, 126, 291–299. [Google Scholar] [CrossRef]
- Veselý, J. Mode of action and effects of 5-azacytidine and of its derivatives in eukaryotic cells. Pharmacology & therapeutics 1985, 28, 227–235. [Google Scholar]
- Wei, A.H.; Montesinos, P.; Ivanov, V.; DiNardo, C.D.; Novak, J.; Laribi, K.; Kim, I.; Stevens, D.A.; Fiedler, W.; Pagoni, M. Venetoclax plus LDAC for newly diagnosed AML ineligible for intensive chemotherapy: a phase 3 randomized placebo-controlled trial. Blood, The Journal of the American Society of Hematology 2020, 135, 2137–2145. [Google Scholar] [CrossRef] [PubMed]
- Wei, A.H.; Strickland Jr, S.A.; Hou, J.-Z.; Fiedler, W.; Lin, T.L.; Walter, R.B.; Enjeti, A.; Tiong, I.S.; Savona, M.; Lee, S. Venetoclax combined with low-dose cytarabine for previously untreated patients with acute myeloid leukemia: results from a phase Ib/II study. Journal of Clinical Oncology 2019, 37, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Dick, J.E.; Bhatia, M.; Gan, O.; Kapp, U.; Wang, J.C. Assay of human stem cells by repopulation of NOD/SCID mice. Stem Cells 1997, 15 Suppl 1, 199-203; discussion 204-197. [CrossRef]
- Becker, H.; Marcucci, G.; Maharry, K.; Radmacher, M.D.; Mrozek, K.; Margeson, D.; Whitman, S.P.; Wu, Y.Z.; Schwind, S.; Paschka, P. , et al. Favorable prognostic impact of NPM1 mutations in older patients with cytogenetically normal de novo acute myeloid leukemia and associated gene- and microRNA-expression signatures: a Cancer and Leukemia Group B study. J Clin Oncol 2010, 28, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Lachowiez, C.A.; Loghavi, S.; Kadia, T.M.; Daver, N.; Borthakur, G.; Pemmaraju, N.; Naqvi, K.; Alvarado, Y.; Yilmaz, M.; Short, N. , et al. Outcomes of older patients with NPM1-mutated AML: current treatments and the promise of venetoclax-based regimens. Blood Adv 2020, 4, 1311–1320. [Google Scholar] [CrossRef]
- Brodska, B.; Otevrelova, P.; Salek, C.; Fuchs, O.; Gasova, Z.; Kuzelova, K. High PD-L1 Expression Predicts for Worse Outcome of Leukemia Patients with Concomitant NPM1 and FLT3 Mutations. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef]
- Ahmadzadeh, M.; Johnson, L.A.; Heemskerk, B.; Wunderlich, J.R.; Dudley, M.E.; White, D.E.; Rosenberg, S.A. Tumor antigen-specific CD8 T cells infiltrating the tumor express high levels of PD-1 and are functionally impaired. Blood 2009, 114, 1537–1544. [Google Scholar] [CrossRef]
- Chen, Y.; Pei, Y.; Luo, J.; Huang, Z.; Yu, J.; Meng, X. Looking for the Optimal PD-1/PD-L1 Inhibitor in Cancer Treatment: A Comparison in Basic Structure, Function, and Clinical Practice. Front Immunol 2020, 11, 1088. [Google Scholar] [CrossRef] [PubMed]
- Orskov, A.D.; Treppendahl, M.B.; Skovbo, A.; Holm, M.S.; Friis, L.S.; Hokland, M.; Gronbaek, K. Hypomethylation and up-regulation of PD-1 in T cells by azacytidine in MDS/AML patients: A rationale for combined targeting of PD-1 and DNA methylation. Oncotarget 2015, 6, 9612–9626. [Google Scholar] [CrossRef] [PubMed]
- Tettamanti, S.; Pievani, A.; Biondi, A.; Dotti, G.; Serafini, M. Catch me if you can: how AML and its niche escape immunotherapy. Leukemia 2022, 36, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Knights, A.J.; Weinzierl, A.O.; Flad, T.; Guinn, B.A.; Mueller, L.; Mufti, G.J.; Stevanovic, S.; Pawelec, G. A novel MHC-associated proteinase 3 peptide isolated from primary chronic myeloid leukaemia cells further supports the significance of this antigen for the immunotherapy of myeloid leukaemias. Leukemia 2006, 20, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Nelde, A.; Schuster, H.; Heitmann, J.S.; Bauer, J.; Maringer, Y.; Zwick, M.; Volkmer, J.P.; Chen, J.Y.; Stanger, A.M.P.; Lehmann, A. , et al. Immune Surveillance of Acute Myeloid Leukemia Is Mediated by HLA-Presented Antigens on Leukemia Progenitor Cells. Blood Cancer Discov 2023, 4, 468–489. [Google Scholar] [CrossRef] [PubMed]
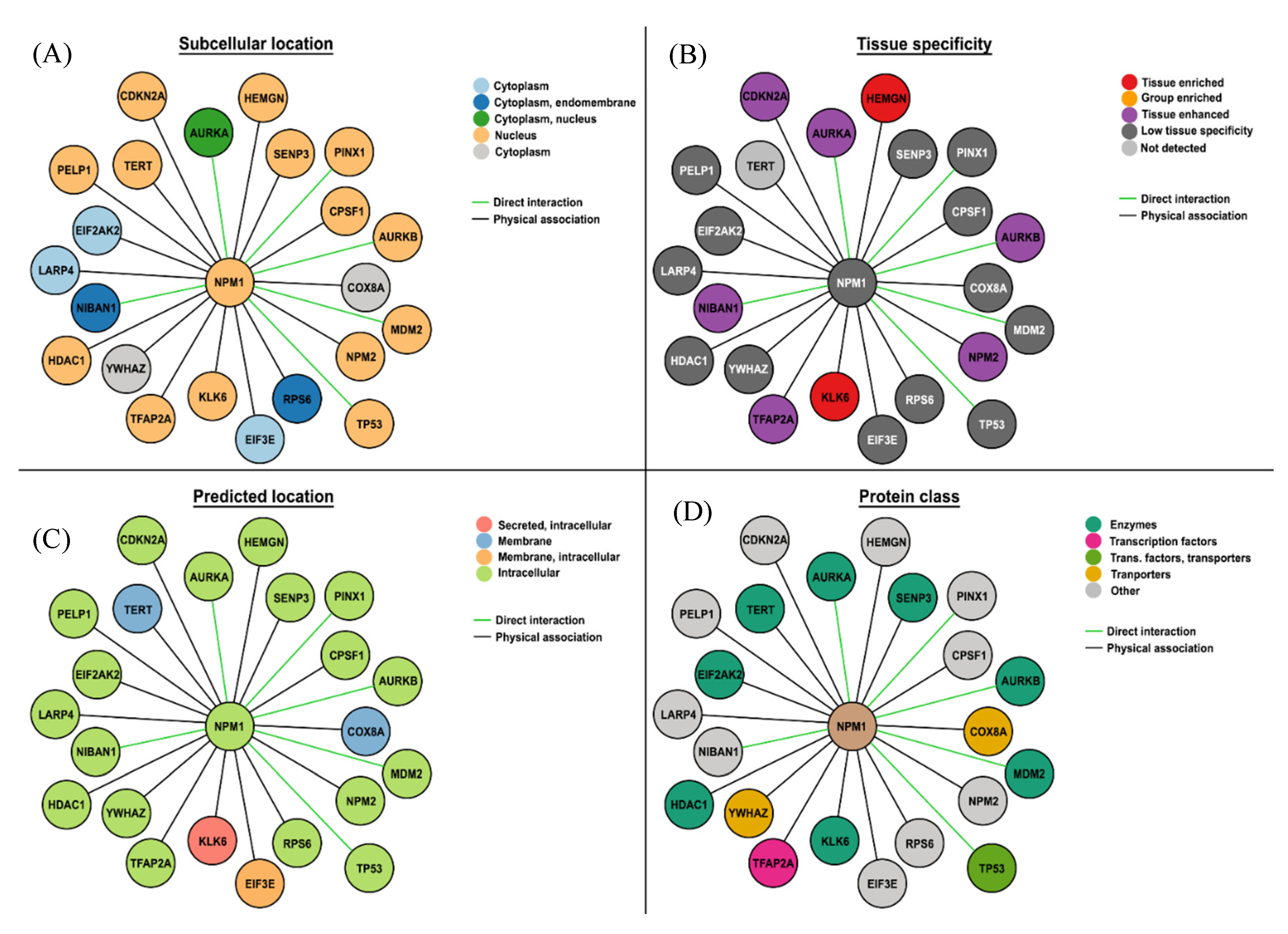
| AML NPM1mut | AML NPM1WT |
| Gatekeeper mutation, association with AML, own WHO subgroup, found in all AML cells, stable (seen again at relapse) | |
| Associated with older AML patients >35 years old, de novo AML and increased frequency in female | |
| Good response to induction therapy, better prognosis in older but not younger AML patients | |
| High WCC | |
| Normal Karyotype | t(8;21), inv(16), t(15;17) |
| FAB M1-M6, more often M4 and M5 | FAB M0 |
| Diseased cells are CD33+ and >90% are CD34− | LSCs are CD34+CD38− |
| FLT3-ITD (2 x more common) | Biallelic CEPBA mutations occur |
| NPM1mut/FLT3-ITD- patients have a better prognosis than NPM1mut FLT3-ITD+ patients | |
| Upregulated HOX genes (A4, A5, A6, A7, A9, A10, B2, B3, B5, B6) | |
| Upregulated HOX-related genes - PBX3 and MEIS1 | |
| LSC express CD96, IL12RB1 |
| Risk | Molecular and Cytogenetics Indicators |
|---|---|
| Poor | FLT3-ITD [28,29]; partial tandem duplication (PTD) [30,31] of the mixed lineage leukemia gene (MLL), and increased expression of the transcription factor ecotropic virus integration site 1 (EVI1) [32]; DNMT3A, MN1, BAALC, EGR-1, AF1q [33] |
| Intermediate | IDH1 (isocitrate dehydrogenase 1 (NADP+), soluble), IDH2 (isocitrate dehydrogenase 2 (NADP+), mitochondrial), and TET2 (tet methylcytosine dioxygenase 2) [34]; t(9;11)(p21.3;q23.3); MLLT3-KMT2A [35] |
| Favourable | Mutations in the transcription factor CEBPA - favorable response to therapy [36,37]; t(8;21), inv(16)/t(16;16), t(15;17) and NPM1mut [35] |
| Treatment | Pre-transplant status | 2-year PFS | 2-year OS |
| 1st line CT: AlloHSCT indicated due to intermediate or poor-risk genetic features (n=27) | MRD- CR | 77% | 81% |
| MRD+ CR | 41% | 71% | |
| Active Disease | 20% | 52% | |
| 2nd line CT: AlloHSCT indicated as salvage therapy in case of molecular persistence, relapse or hematological relapse (n=37) | MRD- CR | 86% | 86% |
| MRD+ CR | 47% | 68% | |
| Active Disease | 24% | 52% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




