Submitted:
19 June 2024
Posted:
19 June 2024
You are already at the latest version
Abstract
Keywords:
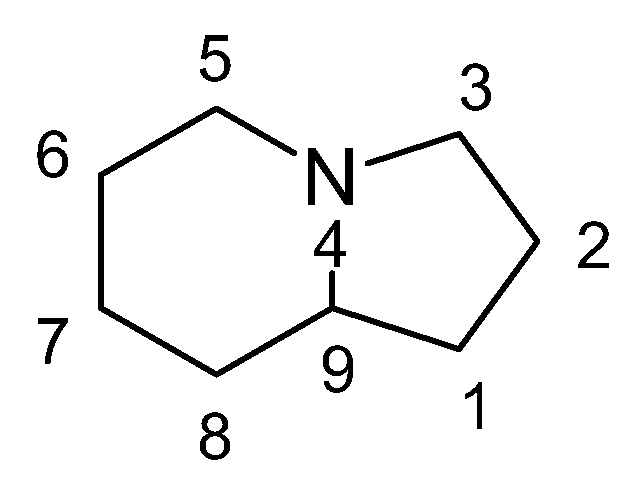
1. Indolizidines: Structure, Sources, and Bioactivities
2. Bioactive Metabolites from Microorganisms
3. Known Actinomycetical Indolizidine-Producers
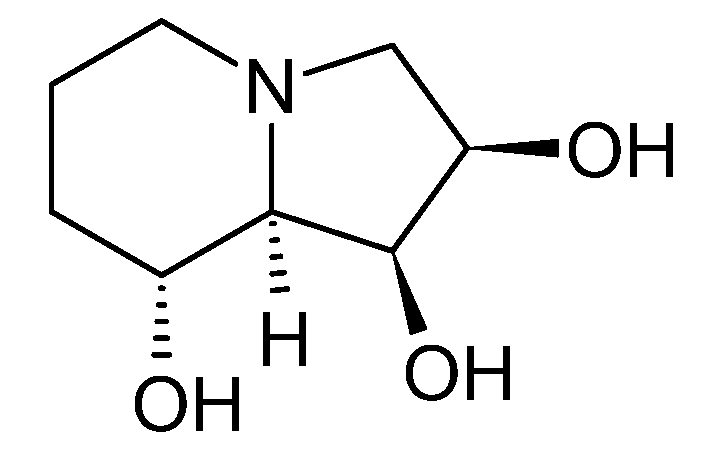
3.1. Cyclizidine M146791 from Streptomyces sp. NCIB 11649
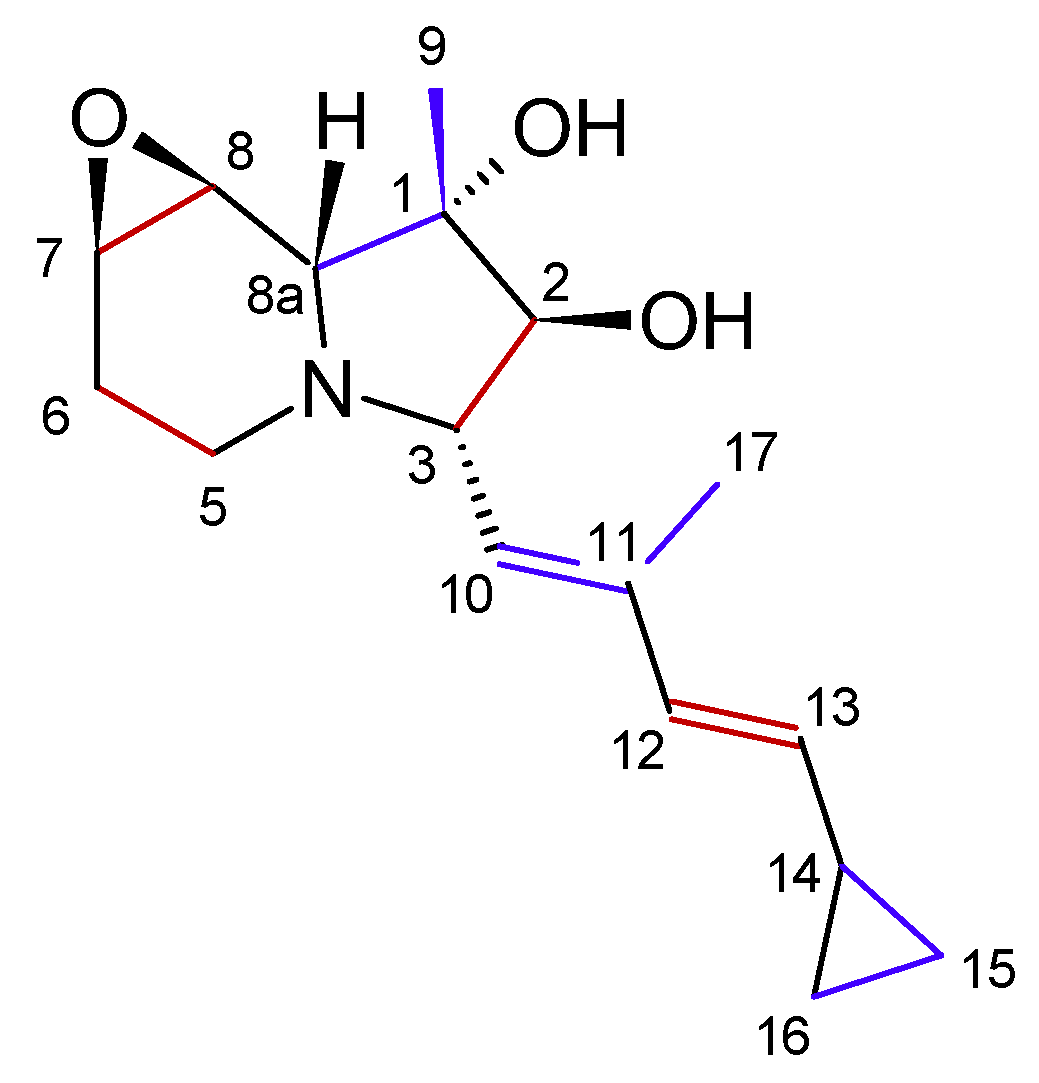
3.1.1. Structure and Isolation
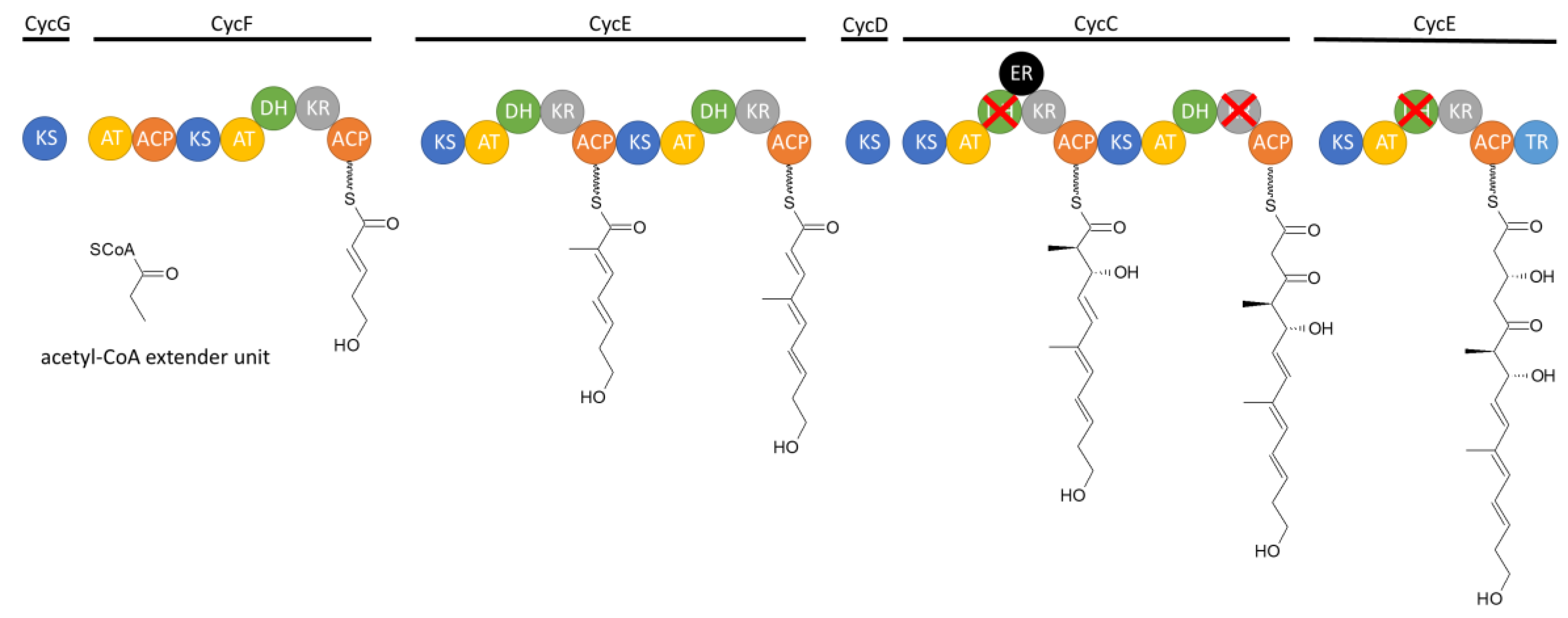
3.1.2. Biosynthesis
3.1.3. Bioactivity
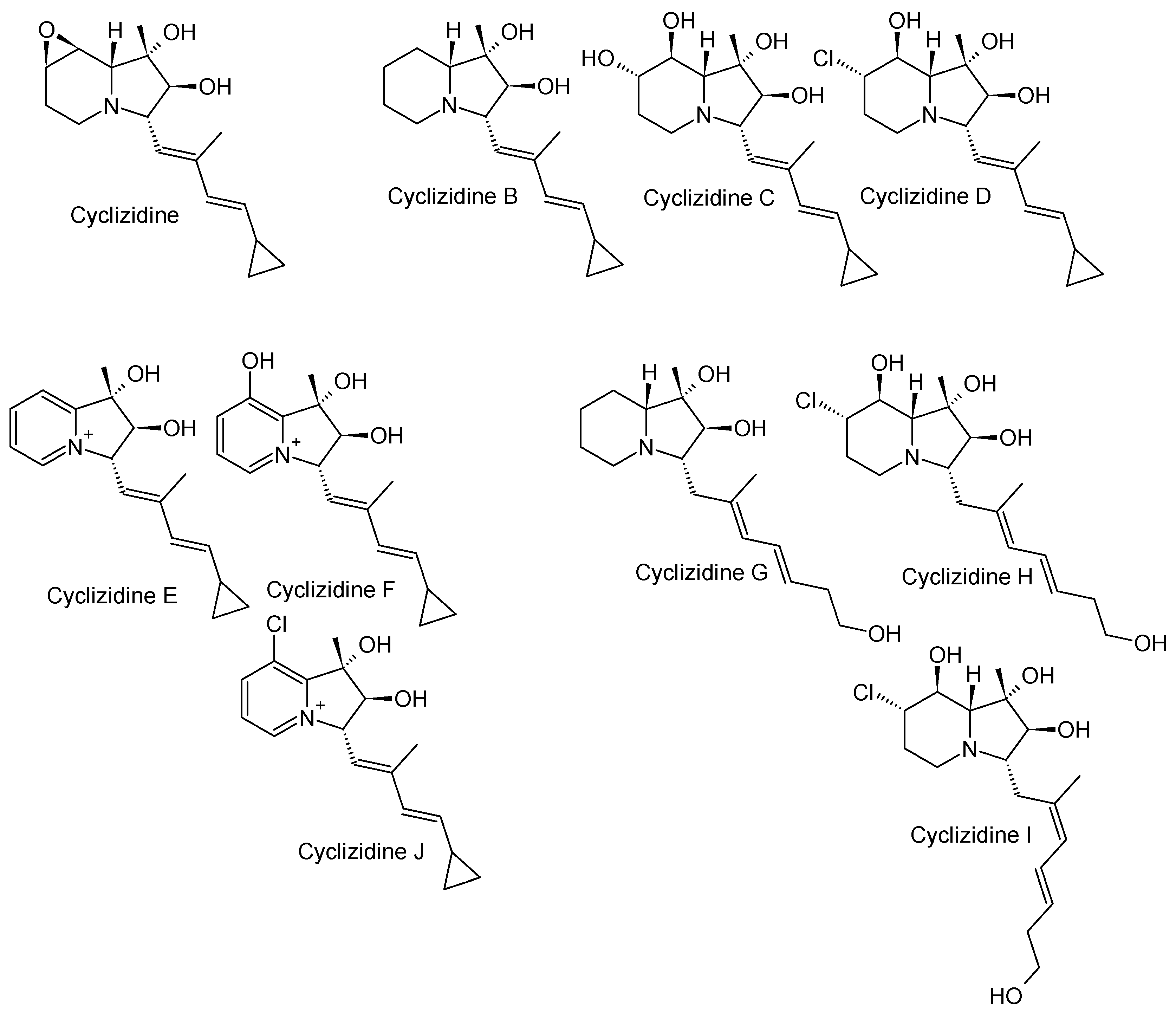
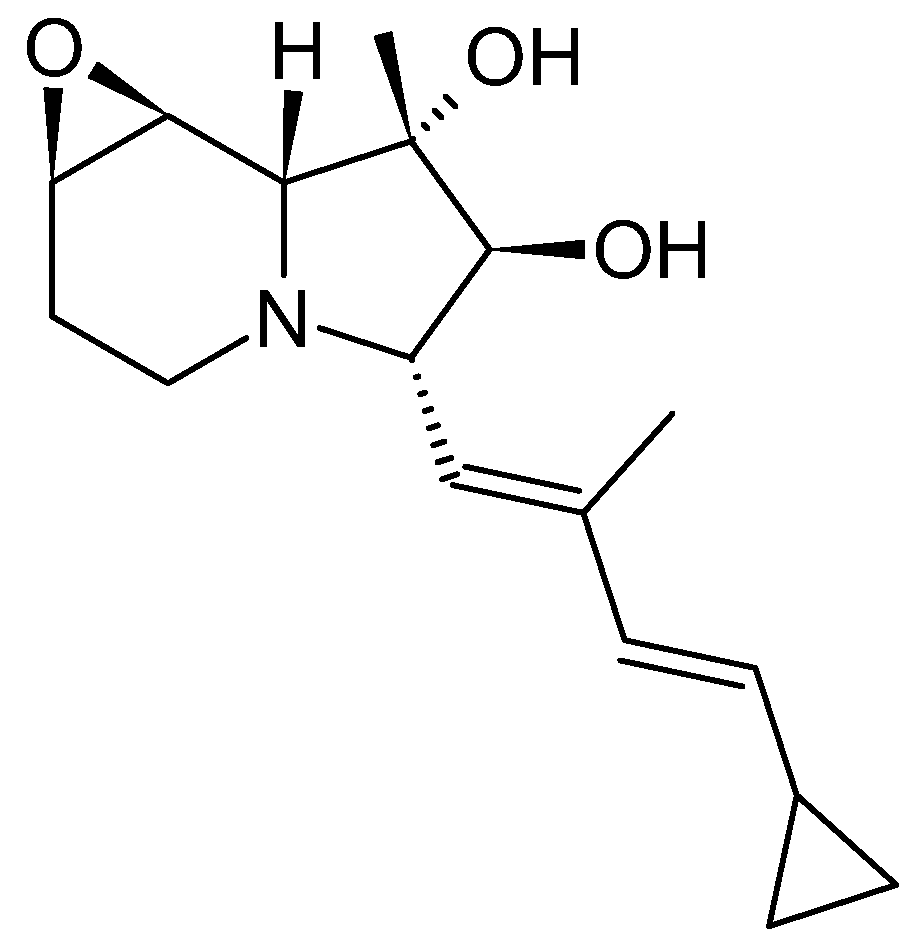
3.2. Cyclizidines from Streptomyces sp. HNA39
3.2.1. Structure and Isolation
3.2.2. Bioactivity
3.3. Cyclizidines from Saccharopolyspora sp. RL78
3.3.1. Structure and Isolation
3.3.2. Bioactivity
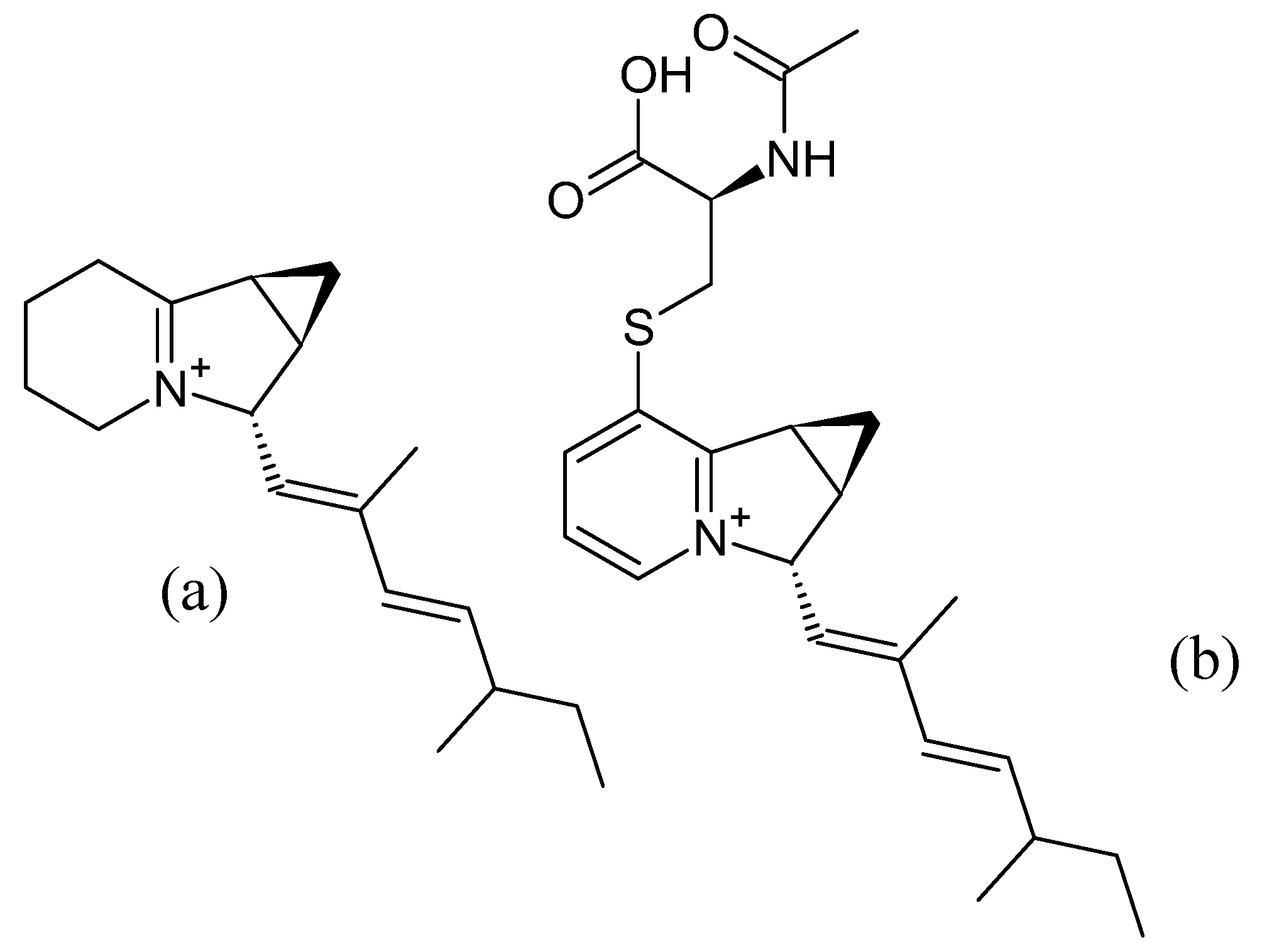
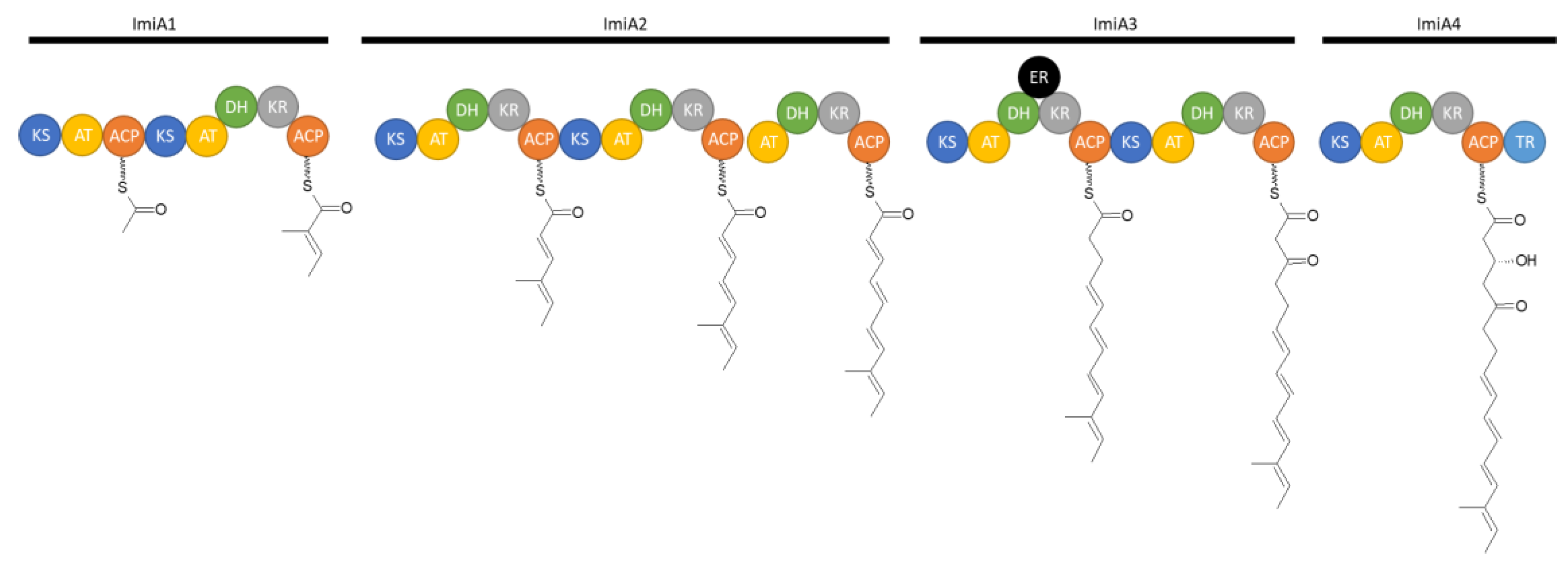
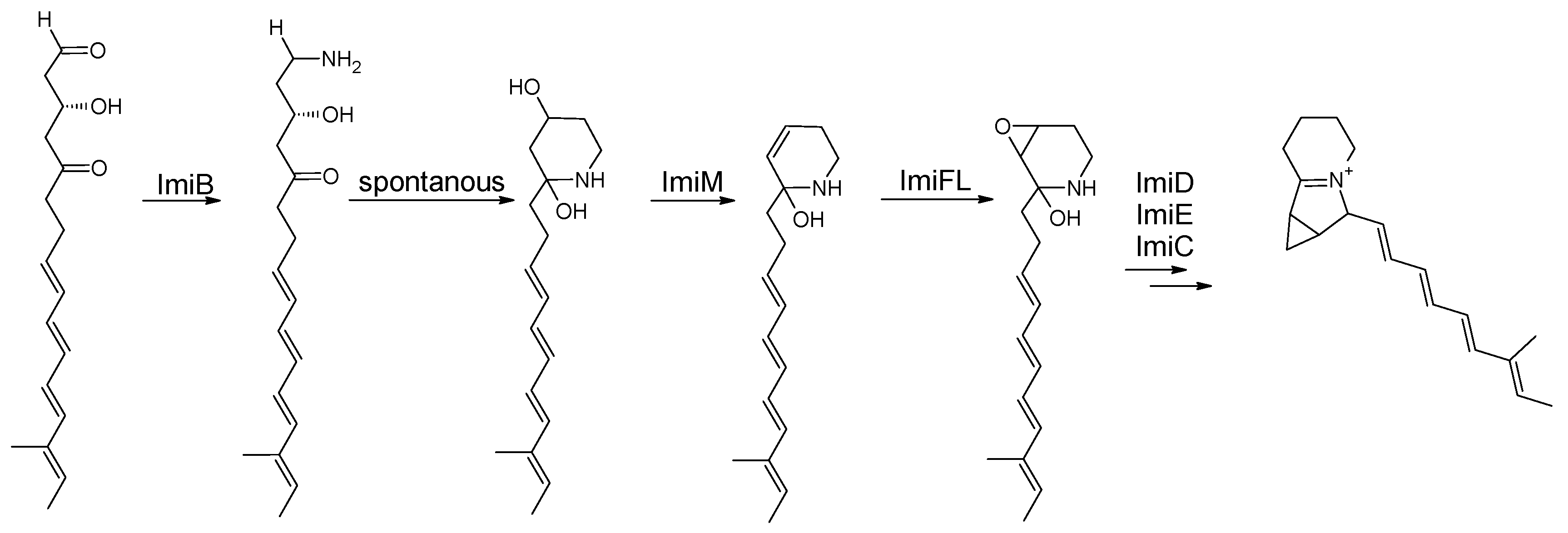
3.4. Iminimycins from Streptomyces griseus
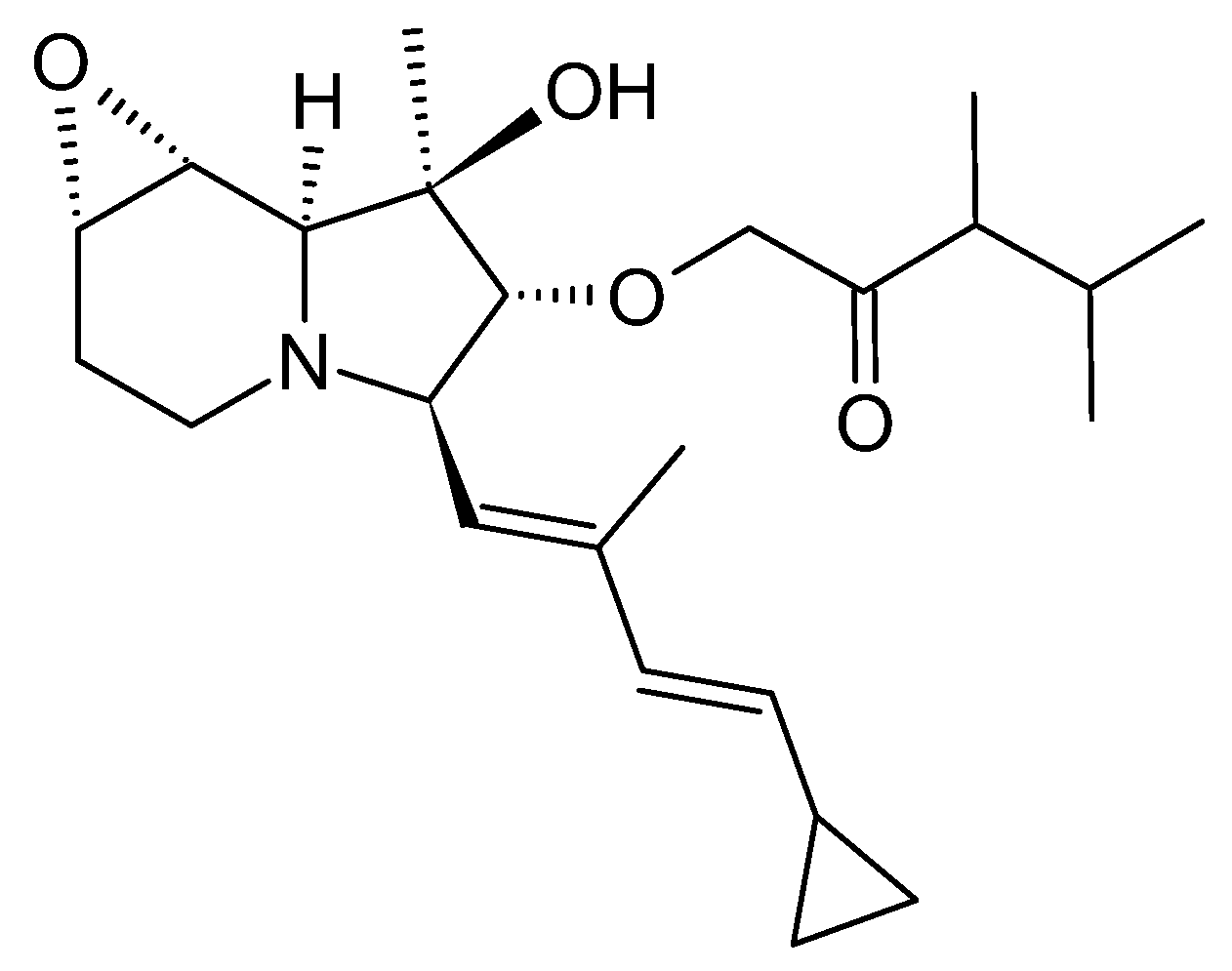
3.4.1. Structure and Isolation
3.4.2. Biosynthesis
3.4.3. Bioactivity
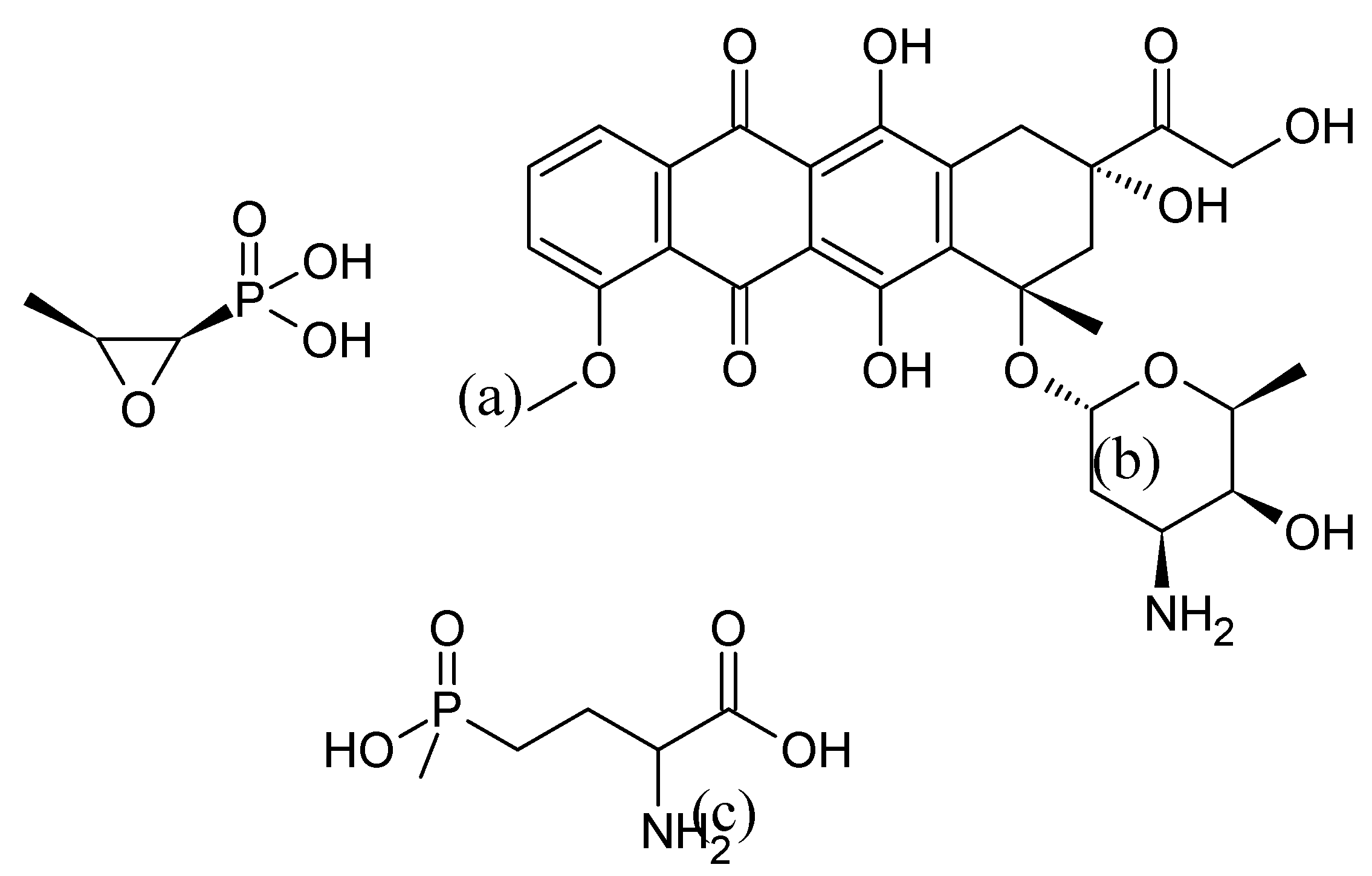
3.5. Indolizomycin from Streptomyces sp. SK2-52
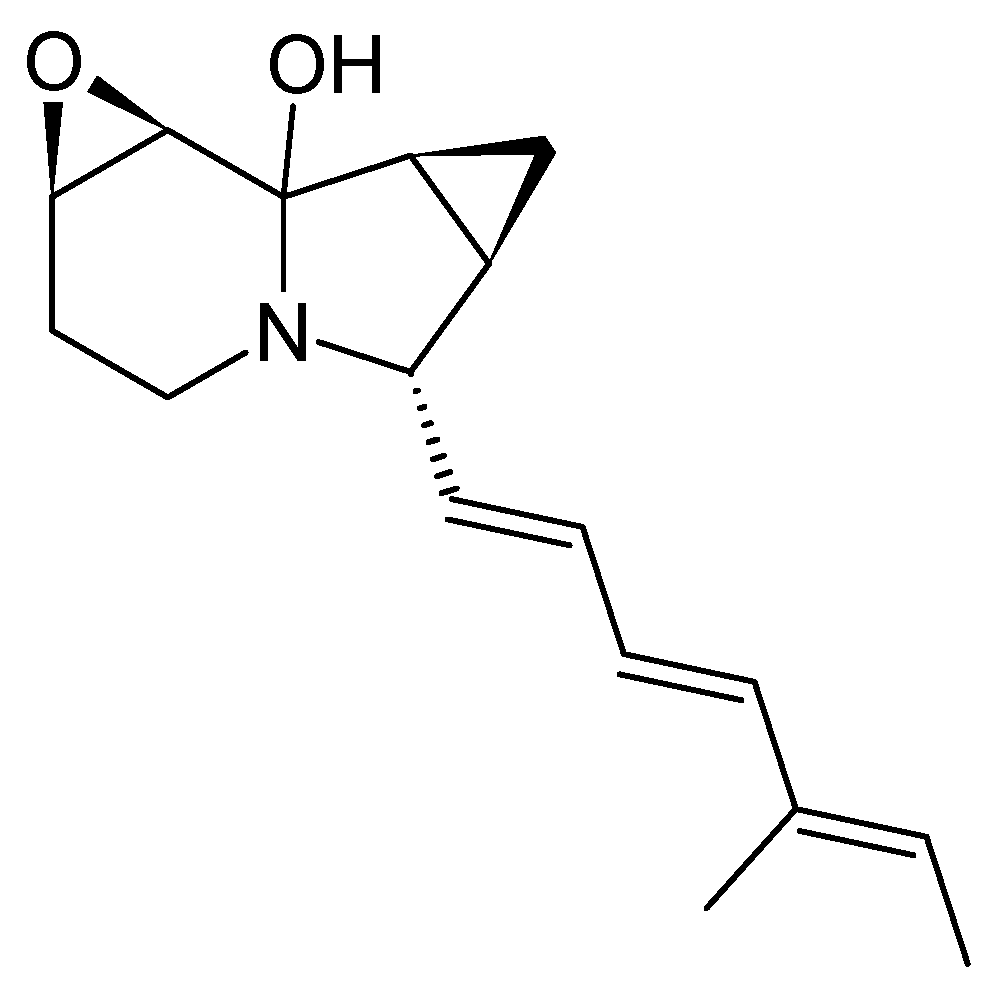
3.5.1. Structure and Isolation
3.5.2. Bioactivity
4. Conclusion and Future Prospects
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- J. Zhang et al., “Biologically active indolizidine alkaloids,” Med Res Rev, vol. 41, no. 2. [CrossRef]
- M. Dhiman, R. R. Parab, S. L. Manju, D. C. Desai, and G. B. Mahajan, “Antifungal Activity of Hydrochloride Salts of Tylophorinidine and Tylophorinine,” Nat Prod Commun, vol. 7, no. 9, pp. 1171–1172, Sep. 2012. [CrossRef]
- M. Kubo et al., “Antimalarial phenanthroindolizine alkaloids from Ficus septica,” Chem Pharm Bull (Tokyo), vol. 64, no. 7, pp. 957–960, Jul. 2016. [CrossRef]
- C. W. Yang, W. L. Chen, P. L. Wu, H. Y. Tseng, and S. J. Lee, “Anti-inflammatory mechanisms of phenanthroindolizidine alkaloids,” Mol Pharmacol, vol. 69, no. 3, pp. 749–758, Mar. 2006. [CrossRef]
- S. Min, Y. Li, C. Nzabanita, H. Liu, M. Ting-Yan, and Y.-Z. Li, “Locoweed endophytes: A review,” 2018. [CrossRef]
- J. Y. Sun et al., “Suppressive effects of swainsonine on C6 glioma cell in vitro and in vivo,” Phytomedicine, vol. 16, no. 11, pp. 1070–1074, Nov. 2009. [CrossRef]
- L. Sun, X. Jin, L. Xie, G. Xu, Y. Cui, and Z. Chen, “Swainsonine represses glioma cell proliferation, migration and invasion by reduction of miR-92a expression,” BMC Cancer, vol. 19, no. 1, pp. 1–10, Mar. 2019. [CrossRef]
- J. Y. Sun, M. Z. Zhu, S. W. Wang, S. Miao, Y. H. Xie, and J. B. Wang, “Inhibition of the growth of human gastric carcinoma in vivo and in vitro by swainsonine,” Phytomedicine, vol. 14, no. 5, pp. 353–359, May 2007. [CrossRef]
- J. Hamaguchi et al., “Swainsonine reduces 5-fluorouracil tolerance in the multistage resistance of colorectal cancer cell lines,” Mol Cancer, vol. 6, no. 1, pp. 1–9, Sep. 2007. [CrossRef]
- H. Zhu et al., “New indolizidine- and pyrrolidine-type alkaloids with anti-angiogenic activities from Anisodus tanguticus,” Biomedicine & Pharmacotherapy, vol. 167, p. 115481, Nov. 2023. [CrossRef]
- M. E. Pyne et al., “A yeast platform for high-level synthesis of tetrahydroisoquinoline alkaloids,” Nat Commun, vol. 11, no. 1, Dec. 2020. [CrossRef]
- Y. J. Jiang, J. Q. Li, H. J. Zhang, W. J. Ding, and Z. J. Ma, “Cyclizidine-type alkaloids from Streptomyces sp. HNA39,” J Nat Prod, vol. 81, no. 2, pp. 394–399, Feb. 2018. [CrossRef]
- W. Huang, S. J. Kim, J. Liu, and W. Zhang, “Identification of the polyketide biosynthetic machinery for the indolizidine alkaloid cyclizidine,” Org Lett, vol. 17, no. 21, pp. 5344–5347, Nov. 2015. [CrossRef]
- M. Izumikawa, T. Hosoya, M. Takagi, and K. Shin-Ya, “A new cyclizidine analog JBIR-102 from Saccharopolyspora sp. RL78 isolated from mangrove soil,” Journal of Antibiotics, vol. 65, no. 1, pp. 41–43, Jan. 2012. [CrossRef]
- H. Tsutsumi et al., “Identification and analysis of the biosynthetic gene cluster for the indolizidine alkaloid iminimycin in Streptomyces griseus,” ChemBioChem, Dec. 2021. [CrossRef]
- Y. Takahashi and T. Nakashima, “Actinomycetes, an inexhaustible source of naturally occurring antibiotics,” Antibiotics, vol. 7, no. 2, pp. 1–17, May 2018. [CrossRef]
- T. O. Rogers and J. Birnbaum, “Biosynthesis of fosfomycin by Streptomyces fradiae.,” Antimicrob Agents Chemother, vol. 5, no. 2, pp. 121–32, Feb. 1974.
- “WHO EML 22nd List (2021) | Enhanced Reader.”.
- J. V. McGowan, R. Chung, A. Maulik, I. Piotrowska, J. M. Walker, and D. M. Yellon, “Anthracycline chemotherapy and cardiotoxicity,” Cardiovasc Drugs Ther, vol. 31, no. 1, pp. 63–75, Feb. 2017. [CrossRef]
- C. J. Thompson and H. Seto, “Bialaphos,” Genetics and Biochemistry of Antibiotic Production, pp. 197–222, Jan. 1995. [CrossRef]
- S. Khan et al., “Biodiversity of actinomycetes and their secondary metabolites: A comprehensive review,” J. Adv. Biomed. & Pharm. Sci. J. Adv. Biomed. & Pharm. Sci, vol. 6, pp. 36–48, 2023, Accessed: Apr. 12, 2024. [Online]. Available: http://jabps.journals.ekb.eg.
- M. S. M. Selim, S. A. Abdelhamid, and S. S. Mohamed, “Secondary metabolites and biodiversity of actinomycetes,” Journal of Genetic Engineering and Biotechnology, vol. 19, no. 1, p. 72, Dec. 2021. [CrossRef]
- C. E. Ezeobiora, N. H. Igbokwe, D. H. Amin, N. V. Enwuru, C. F. Okpalanwa, and U. E. Mendie, “Uncovering the biodiversity and biosynthetic potentials of rare actinomycetes,” Future Journal of Pharmaceutical Sciences 2022 8:1, vol. 8, no. 1, pp. 1–19, Mar. 2022. [CrossRef]
- Menezes et al., “Bioactive metabolites from terrestrial and marine actinomycetes,” Molecules 2023, Vol. 28, Page 5915, vol. 28, no. 15, p. 5915, Aug. 2023. [CrossRef]
- S. Gomi et al., “Isolation and structure of a new antibiotic, indolizomycin, produced by a strain SK2-52 obtained by interspecies fusion treatment,” J Antibiot (Tokyo), vol. 37, no. 11, pp. 1491–1494, 1984. [CrossRef]
- A. Freer, D. Gardner, D. Greatbanks, J. P. Poyser, and G. A. Sim, “Structure of cyclizidine (antibiotic M146791) : X -ray crystal structure of an indolizidinediol metabolite bearing a unique cyclopropyl side-chain,” J Chem Soc Chem Commun, vol. 0, no. 20, pp. 1160–1162, Jan. 1982. [CrossRef]
- F. J. Leeper, S. E. Shaw, and P. Satish, “Biosynthesis of the indolizidine alkaloid cyclizidine: incorporation of singly and doubly labelled precursors,” Can J Chem, vol. 72, no. 1, pp. 131–141, 2011. [CrossRef]
- J. Staunton, “The extraordinary enzymes involved in erythromycin biosynthesis,” Angewandte Chemie International Edition in English, vol. 30, no. 10, pp. 1302–1306, Oct. 1991. [CrossRef]
- C. T. Walsh, S. E. O’Connor, and T. L. Schneider, “Polyketide-nonribosomal peptide epothilone antitumor agents: the EpoA, B, C subunits,” J Ind Microbiol Biotechnol, vol. 30, no. 8, pp. 448–455, Aug. 2003. [CrossRef]
- Nivina, K. P. Yuet, J. Hsu, and C. Khosla, “Evolution and diversity of assembly-line polyketide synthases,” Chem Rev, vol. 119, no. 24, pp. 12524–12547, Dec. 2019. [CrossRef]
- S. Hanessian, U. Soma, S. Dorich, and B. Deschênes-Simard, “Total synthesis of (+)-ent -cyclizidine: Absolute configurational confirmation of antibiotic M146791,” Org Lett, vol. 13, no. 5, pp. 1048–1051, Mar. 2011. [CrossRef]
- S. Li, Y. J. Jiang, Z. Ma, and N. Wang, “Complete genome sequence of Streptomyces sp. HNA39, a new cyclizidine producer isolated from a South China Sea sediment,” Mar Genomics, vol. 70, Aug. 2023. [CrossRef]
- X. W. Cheng, J. Q. Li, Y. J. Jiang, H. Z. Liu, and C. Huo, “A new indolizinium alkaloid from marine-derived Streptomyces sp. HNA39,” J Asian Nat Prod Res, 2020. [CrossRef]
- P. Skehan et al., “New colorimetric cytotoxicity assay for anticancer-drug screening,” JNCI: Journal of the National Cancer Institute, vol. 82, no. 13, pp. 1107–1112, Jul. 1990. [CrossRef]
- M. Takagi and K. Shin-Ya, “New species of actinomycetes do not always produce new compounds with high frequency,” The Journal of Antibiotics, vol. 64, no. 10, pp. 699–701, Jul. 2011. [CrossRef]
- Schatz, E. Bugle, and S. A. Waksman, “Streptomycin, a substance exhibiting antibiotic activity against gram-positive and gram-negative bacteria,” Proceedings of the Society for Experimental Biology and Medicine, vol. 55, no. 1, pp. 66–69, Nov. 2016. [CrossRef]
- T. Nakashima et al., “Iminimycin A, the new iminium metabolite produced by Streptomyces griseus OS-3601,” The Journal of Antibiotics 2016 69:8, vol. 69, no. 8, pp. 611–615, Jan. 2016. [CrossRef]
- T. Nakashima et al., “Absolute configuration of iminimycin B, a new indolizidine alkaloid, from S. griseus OS-3601,” Tetrahedron Lett, vol. 57, no. 30, pp. 3284–3286, Jul. 2016. [CrossRef]
- H. Tsutsumi et al., “Identification and analysis of the biosynthetic gene cluster for the indolizidine alkaloid iminimycin in Streptomyces griseus,” ChemBioChem, vol. 22, Dec. 2021. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).