Submitted:
18 June 2024
Posted:
19 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental
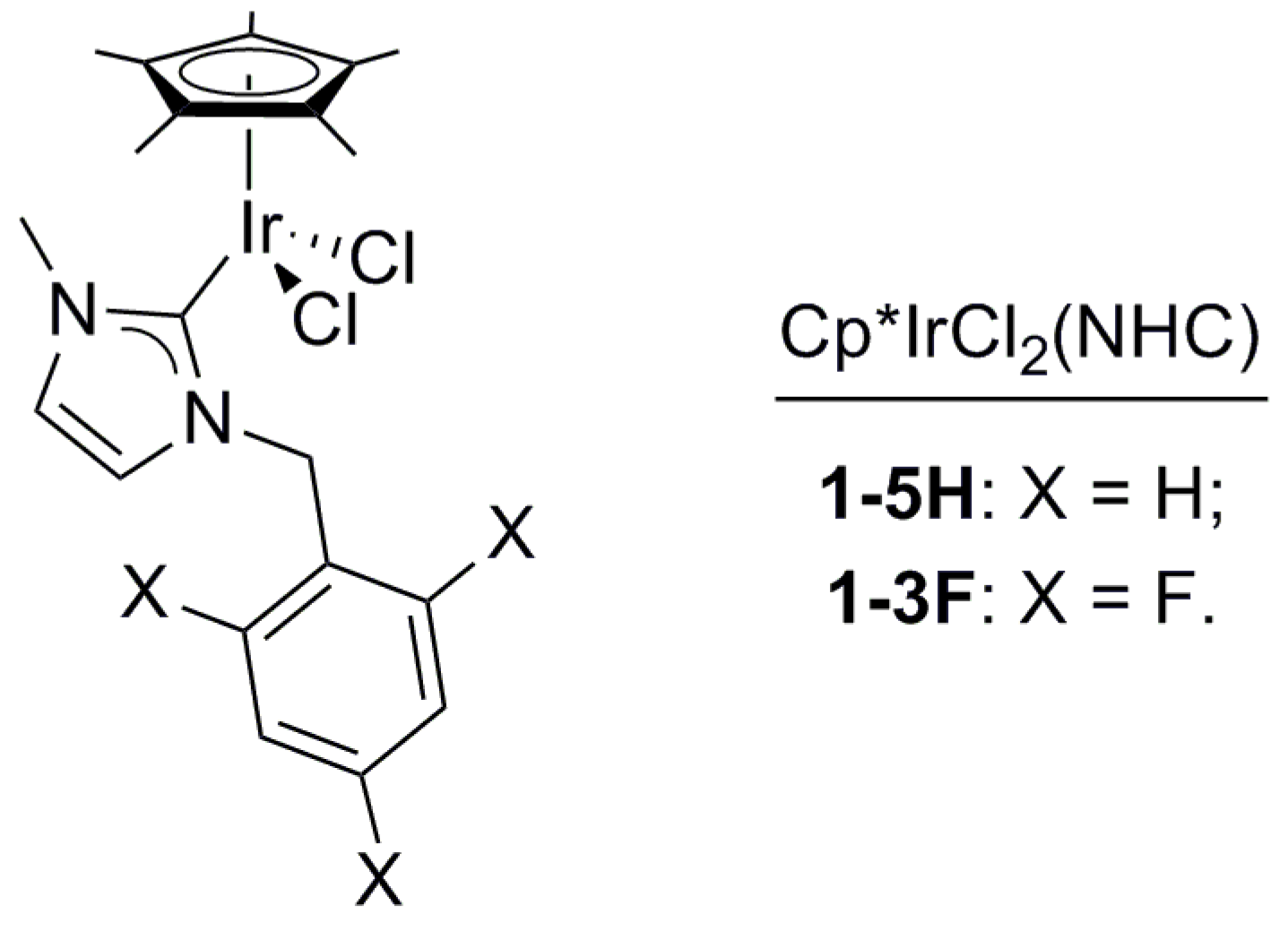
2.1. Reactions of 1,3-Propanediol in the Presence of Cp*IrCl2(NHC) Complexes 1-5H or 1-3F
2.1.1. Reaction of 1,3-PDO in the Presence of Cp*IrCl2(NHC) Complexes 1-5H or 1-3F, and K2CO3, in Ionic Liquid: Screening Reaction Conditions
2.1.2. Reaction of 1,3-PDO in the Presence of 1-5H or 1-3F, and K2CO3, at 120 or 150°C, in N1,8,8,8NTf2, and at [1,3-PDO]:[Ir] ≅75.0: Recycling Experiments
2.1.3. Reaction of 1,3-PDO in the Presence of 1-5H or 1-3F, and K2CO3, at 150°C, in N1,8,8,8NTf2, and Water, at [1,3-PDO]:[Ir] ≅75.0
2.1.4. Reaction of 1,3-PDO in the Presence of 1-5H or 1-3F, and K2CO3, in 1,3-PDO
2.1.5. Reaction of 1,3-PDO in the Presence of 1-5H or 1-3F, and K2CO3, at 120°C, in Toluene, at [1,3-PDO]:[Ir] ≅75.0
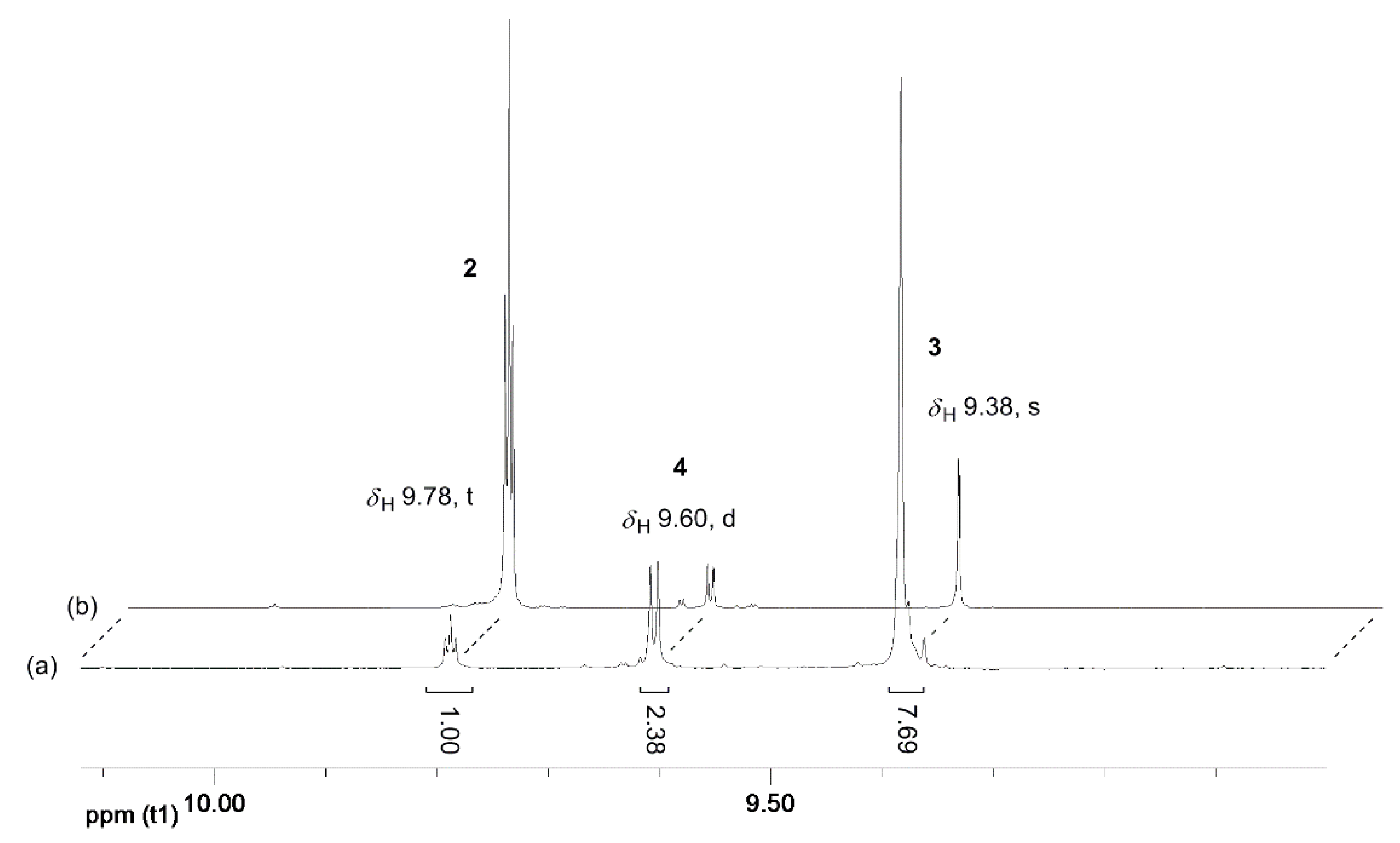
2.2.1. H NMR Analysis of REACTION product Solutions: General Procedure
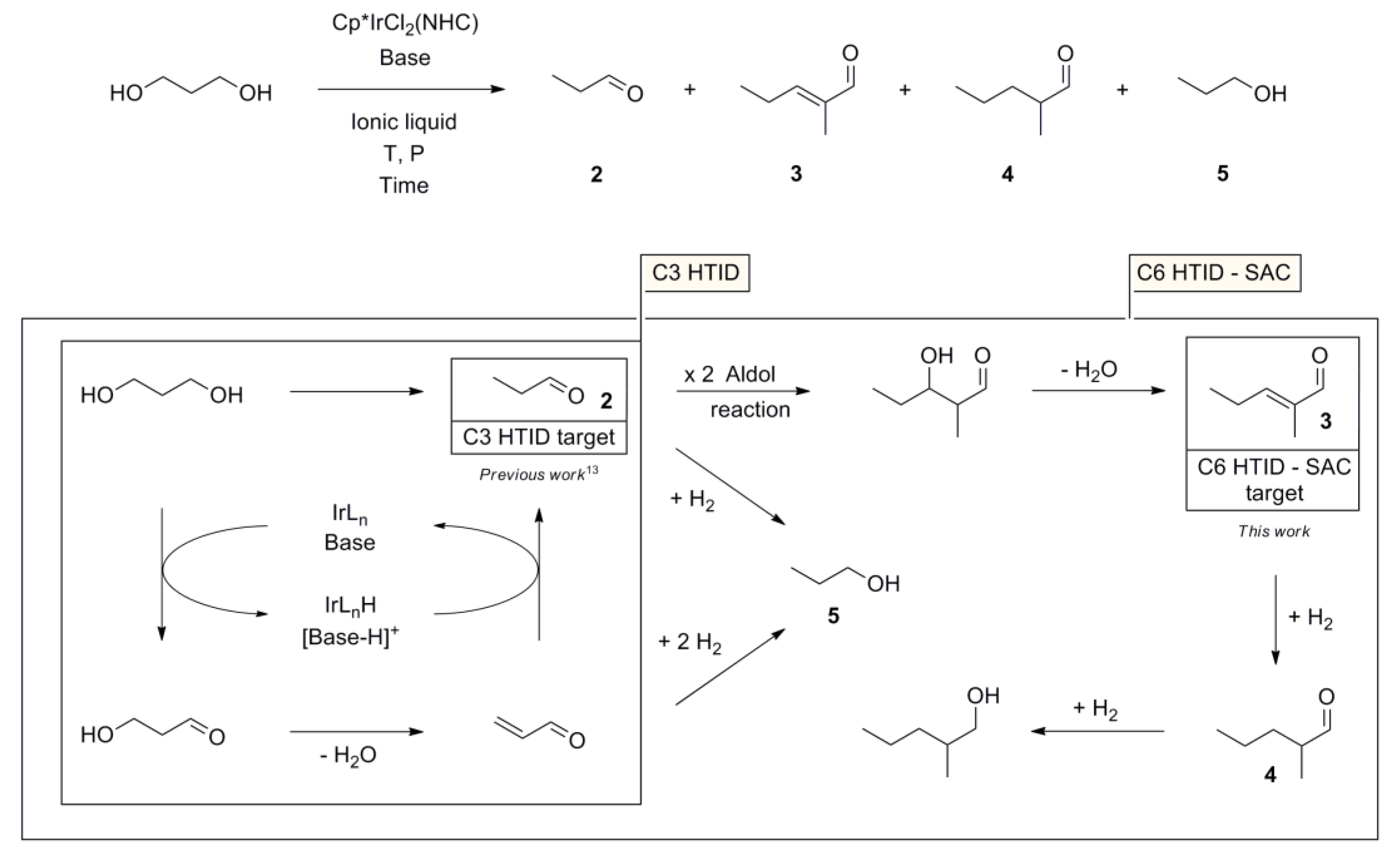
3. Results and Discussion
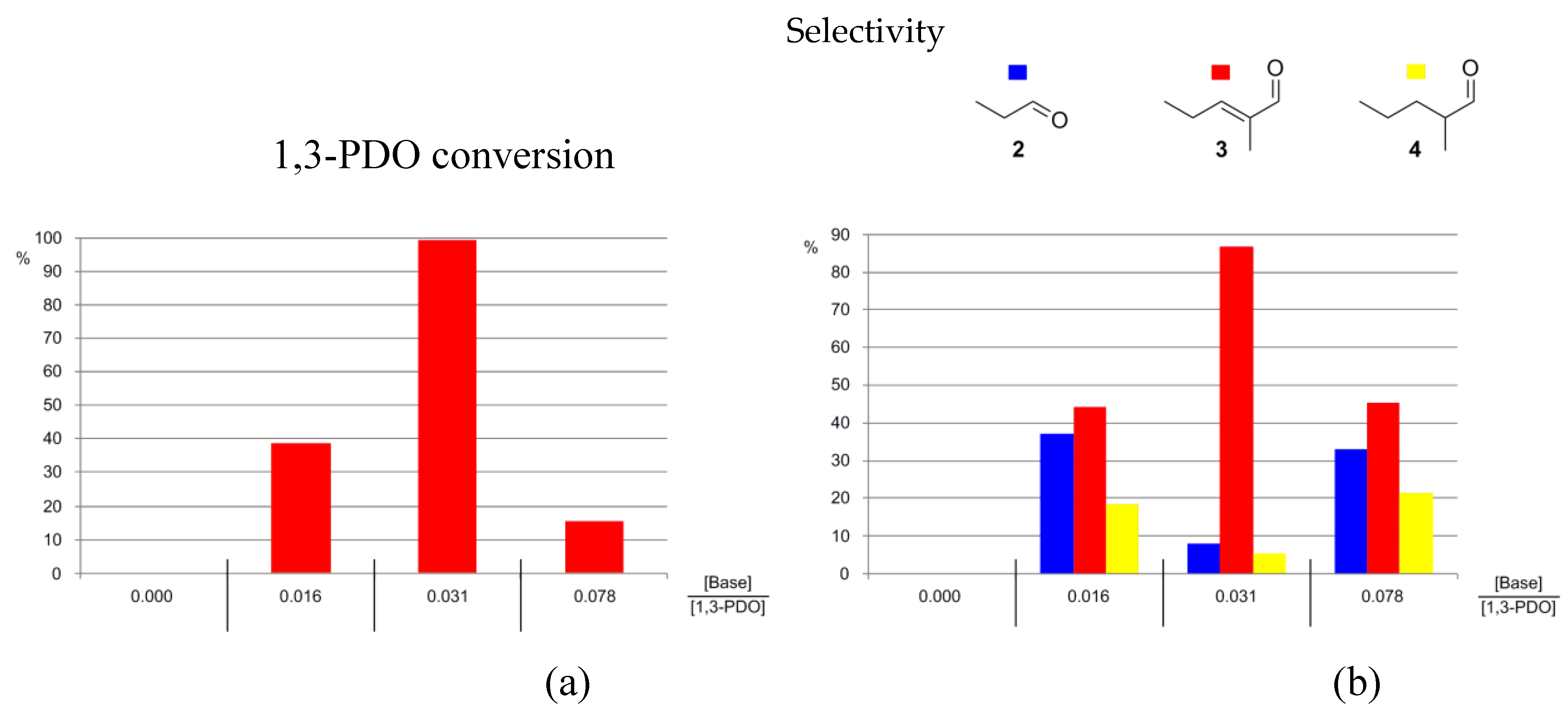
3.1. The Effects of Altering the Base
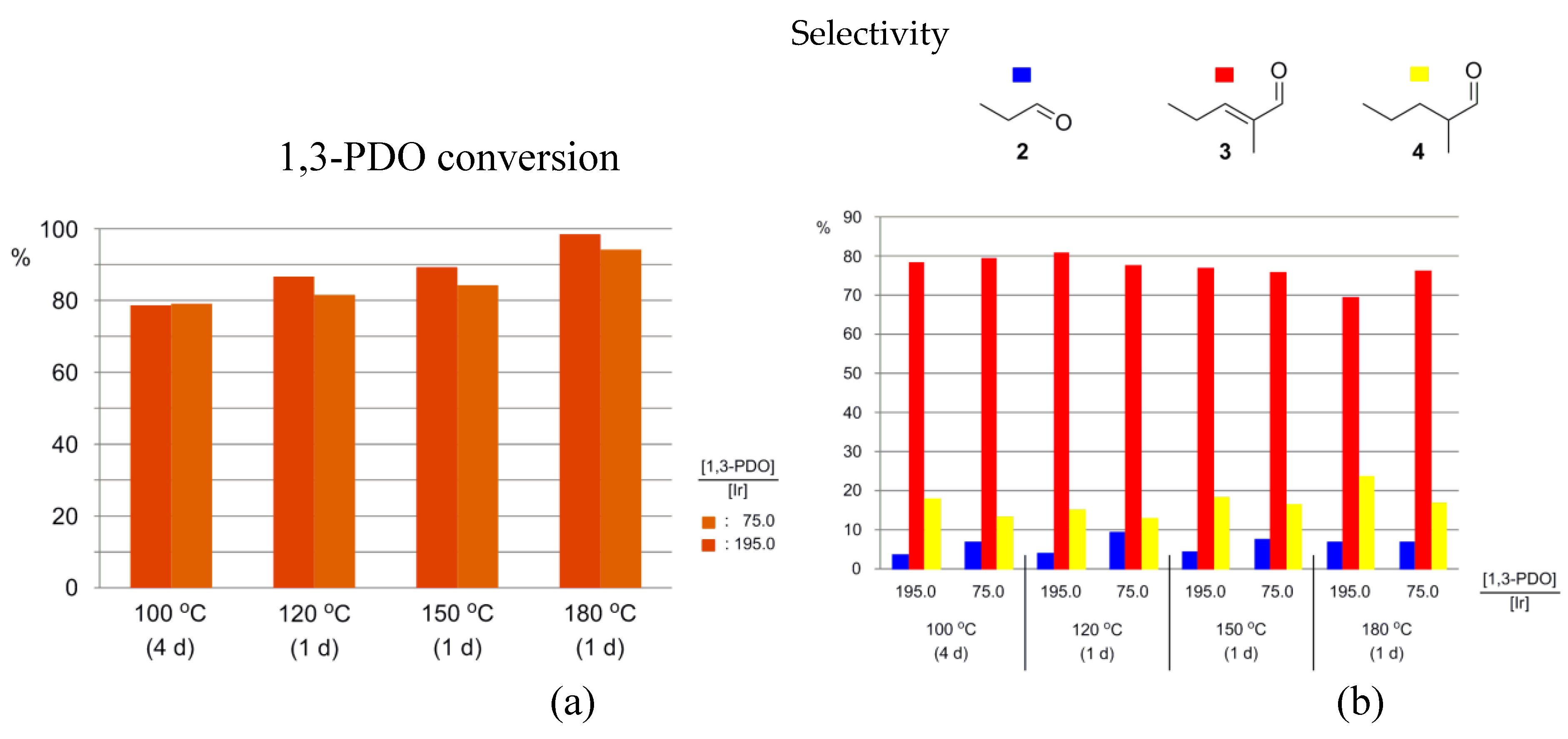
3.2. The Effect of Catalyst Loading and Temperature
3.2.1. In the Presence of 1-5H
3.2.2. In the Presence of 1-3F
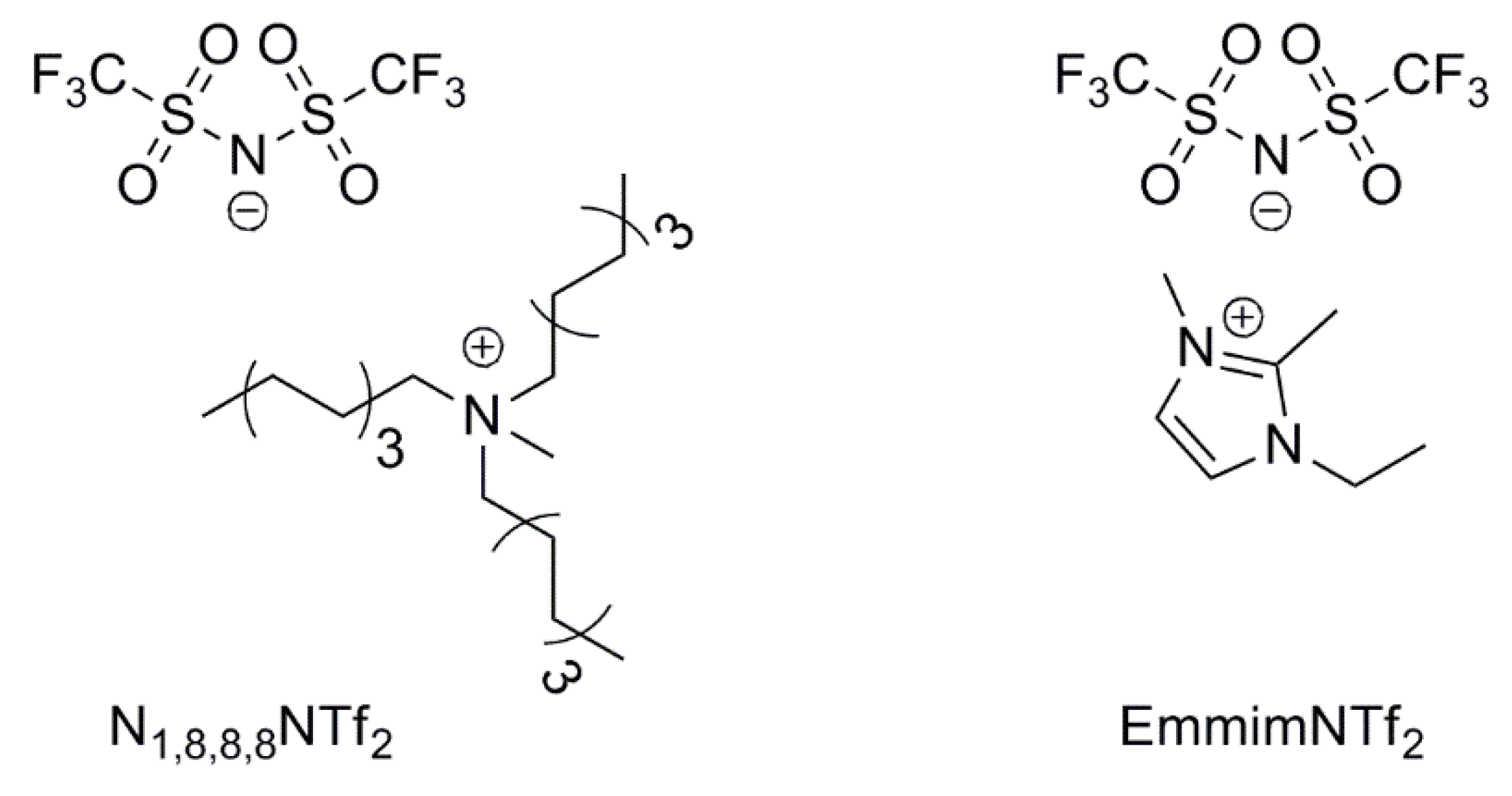
3.3. Ionic Liquid Effect
3.4. 1,3-PDO and Water Effect
| Entry | Catalyst | 1,3-PDO conversiona | 2 | 3 | 4 | TOFb |
| [%] | [s−1](×103) | |||||
| 1 | Ir-5H | 77 | 23.8 | 68.2 | 8.1 | 0.63 |
| 2 | Ir-3F | 71 | 11.2 | 75.6 | 13.2 | 0.63 |
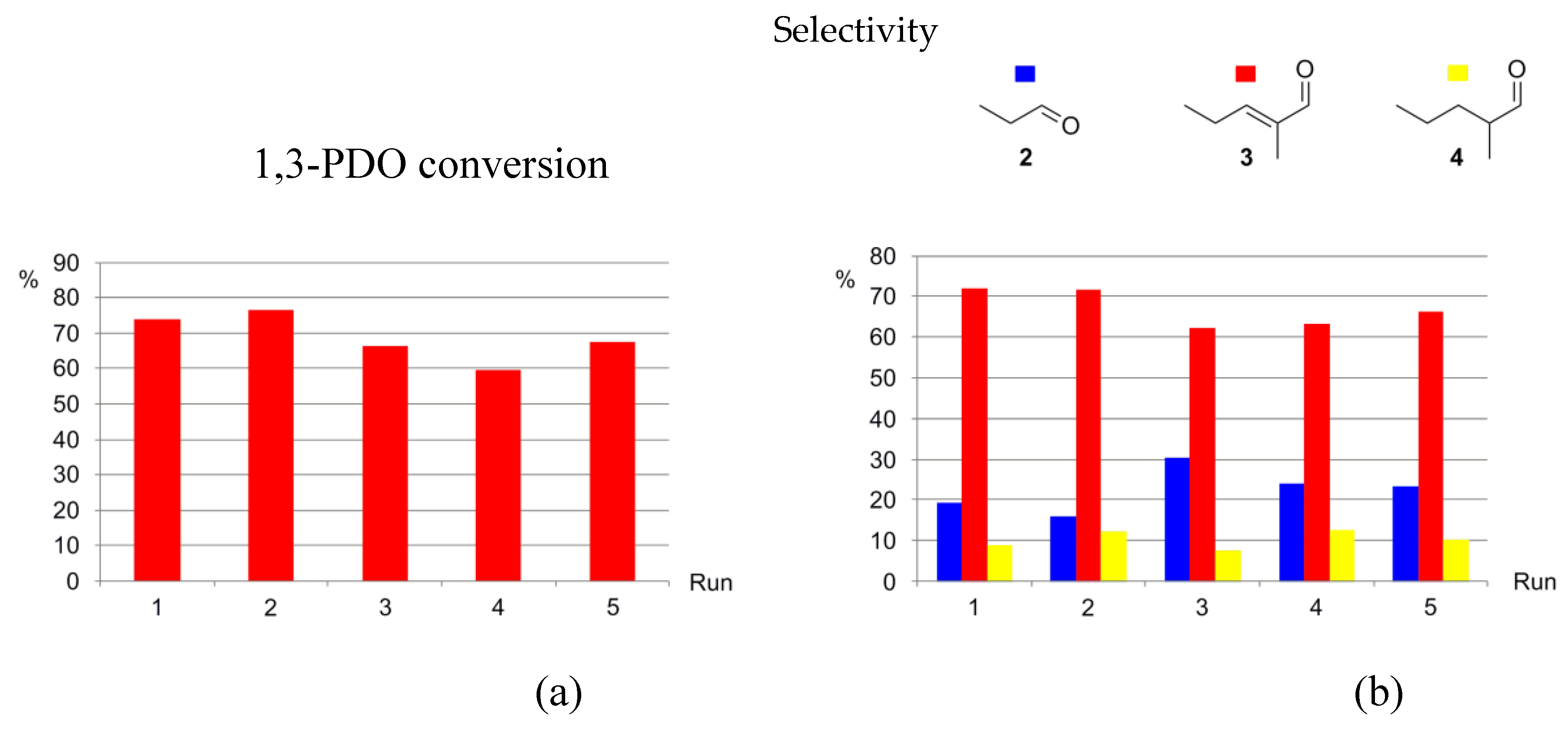
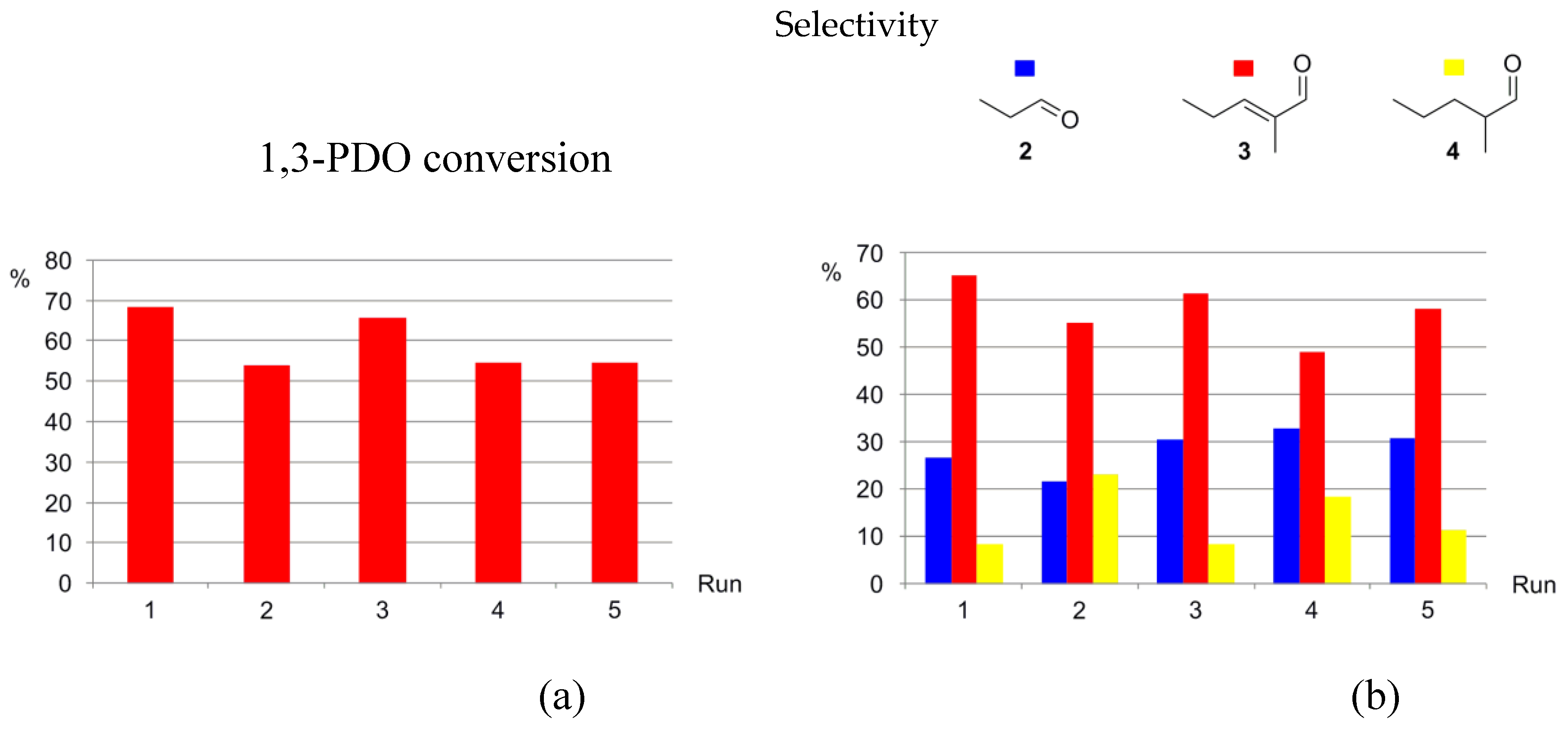
3.5. Catalysts Recycling
4. Conclusions
Supplementary Materials
Acknowledgments
References
- Zimmerman, J. B.; Anastas, P. T.; Erythropel, H. C.; Leitner, W. Designing for a green chemistry future, Science, 2020, 367, 397–400. [CrossRef]
- Marr, A. C.; S. F. Liu, S. F. Combining bio- and chemo-catalysis: from enzymes to cells, from petroleum to biomass. Trends in Biotechnol. 2011, 29, 199 – 204. [CrossRef]
- J. Pradima, M.; Rajeswari Kulkarni, A. Review on enzymatic synthesis of value added products of glycerol, a by-product derived from biodiesel production, Resource-Efficient Technologies, 2017, 3, 2017, 394–405. [CrossRef]
- Zhu, Y.; Wang, Y.; Gao, H.; Wang, H.; Wan, Z.; Jiang, Y.; Xin, F.; Zhang, W.; Jiang, M. Current advances in microbial production of 1,3-propanediol, Biofuels, Bioprod. Bioref. 2021, 15, 1566–1583. [CrossRef]
- Nimbalkar, P. R.; Dharne, M. S. A review on microbial 1, 3-propanediol production: Emerging strategies, key hurdles and attainable solutions to re-establish its commercial interest, Ind. Crops Prod. 2024, 209, 117961. [CrossRef]
- Liu, S. F.; Rebros, M.; Stephens, G.; Marr, A. C. Adding Value to Renewables: A One Pot Process Combining Microbial Cells and Hydrogen Transfer Catalysis to Utilise Waste Glycerol from Biodiesel Production. Chem. Commun. 2009, 2308 – 2310. [CrossRef]
- Liu, X.-H.; Rebros, M.; Dolejš, I.; Marr, A. C. Designing Ionic Liquids for the Extraction of Alcohols from Fermentation Broth: Phosphonium Alkanesulfonates, Solvents for Diol Extraction. ACS Sustainable Chem. Eng., 2017, 5, 8260–8268. [CrossRef]
- Dolejš, I.; Líšková, M.; Krasňan, V.; Markošová, K.; Rosenberg, M.; Lorenzini, F.; Marr, A. C.; Rebroš, M. Production of 1,3-propanediol from pure and crude glycerol using immobilized clostridium butyricum. Catalysts, 2019, 9, 317. [CrossRef]
- Bothwell, K. M.; Krasňan, V.; Lorenzini, F.; Rebroš, M.; Marr, A. C.; Marr, P. C. Utilizing Crude Waste Glycerol in the Biorefinery: Glycerol Gels for in Situ Substrate Delivery to Whole Cell Biocatalysts. ACS Sustainable Chem. Eng. 2019, 7, 9948–9956. [CrossRef]
- Gérardy, R.; Debecker, D. P.; Estager, J.; Luis, P.; Monbaliu, J.-C. M. Continuous Flow Upgrading of Selected C2–C6 Platform Chemicals Derived from Biomass, Chem. Rev. 2020, 120, 7219–7347. [CrossRef]
- da Silva Ruy, A. D.; de Brito Alves, R. M.; Hewer, T. L. R.; de Aguiar Pontes, D.; Leonardo Sena Gomes Teixeira , Pontes, L. A. M. Catalysts for glycerol hydrogenolysis to 1,3-propanediol: A review of chemical routes and market, Catalysis Today, 2021, 381, 243–253. [CrossRef]
- Ma, L.; Liu, H.; He, D. Recent Progress in Catalyst Development of the Hydrogenolysis of Biomass-Based Glycerol into Propanediols—A Review. Bioengineering 2023, 10, 1264. [CrossRef]
- Marr, A. C. Organometallic hydrogen transfer and dehydrogenation catalysts for the conversion of bio-renewable alcohols. Catal. Sci. Technol., 2012, 2, 279–287. 10.1039/C1CY00338K.
- Kumar, A.; Gao, C. Homogeneous (De)hydrogenative Catalysis for Circular Chemistry – Using Waste as a Resource, ChemCatChem, 2021, 13, 1105–1134. [CrossRef]
- Amination and Dehydration of 1,3-Propanediol by Hydrogen Transfer: Reactions of a Bio-Renewable Platform Chemical. S. D. Lacroix, A. Pennycook, S. Liu, T. T. Eisenhart, A. C. Marr, Catal. Sci. Technol., 2012, 2, 288 – 290. [CrossRef]
- Wang, Y.-M.; Lorenzini, F.; Rebros, M.; Saunders, G. C.; Marr, A. C. Combining Bio- and Chemo-Catalysis for the Conversion of Bio-Renewable Alcohols: Homogeneous Iridium Catalysed Hydrogen Transfer Initiated Dehydration of 1,3-Propanediol to Aldehydes. Green Chem. 2016, 18, 1751 – 1761. [CrossRef]
- Ma, Y.; Wang, Y.-M.; Morgan, P.; Liu, X.-H.; Saunders, G. C.; Lorenzini, F.; Marr, A. C. Designing effective homogeneous catalysis for glycerol valorisation: selective synthesis of a value-added aldehyde from 1,3-propanediol via hydrogen transfer catalysed by a highly recyclable, fluorinated Cp*Ir(NHC) catalyst. Catal. Today. 2018, 307, 248-259. [CrossRef]
- Bothwell, K. M.; Lorenzini, F.; Mathers, E.; Marr, P. C.; Marr, A. C. Basic Ionic Liquid Gels for Catalysis: Application to the Hydrogen Borrowing Mediated Dehydration of 1,3-Propanediol. ACS Sustainable Chem. Eng., 2019, 7, 2686–2690. [CrossRef]
- Kirchhecker, S.; Spiegelberg, B.; de Vries, J.G. Homogenous Iridium Catalysts for Biomass Conversion. In Iridium Catalysts for Organic Reactions; Oro, L.A., Claver, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; Volume 69, pp. 341–395.
- Aida, T.; Nagasawa, T.; Yamazaki, Y. WO Pat., 065851, 2008.
- Tanaka, S.; Fukuda, K.; Asada, T. WO Pat., 029033, 2004.
- Godwin, A. D., Schlosberg, R. H., Hershkowitz, F., Matturro, M. G., Kiss, G., Nadler, K. C., Buess, P. L., Miller, R. C., Allen, P. W., Deckman, H. W., Caers, R., Mozeleski, E. J., Reynolds, R. P. U.S. Pat. 6,307,093, 2001.
- Blanc, P. Y.; Perret, A.; Teppa, F. Etude de l’évolution du propanal et du butanal en milieu aqueux alcalin à température peu élevée. Helv. Chim. Acta, 1964, 47, 567-575. https://doi.org/10.1002/hlca.19640470224.
- Schoenleben, W.; Hoffmann, H.; Lengsfeld, W.; Mueller, H. DE 2727330, 1977.
- Mehnert, C. P.; Dispenziere, N. C.; Cook, R. A. Preparation of C9-aldehyde via aldol condensation reactions in ionic liquid media. Chem. Commun., 2002, 1610-1611. [CrossRef]
- Mehnert, C. P.; Dispenziere, N. C.; Schlosberg, R. H. US Pat., 6,552,232, 2003.
- Kelly, G. F.; King, F.; Kett, M. Waste elimination in condensation reactions of industrial importance. Green Chem., 2002, 4, 392–399. [CrossRef]
- Mestres, R. A green look at the aldol reaction. Green Chem., 2004, 6, 583–603. [CrossRef]
- Stevens, J. G.; Bourne, R. A.; Poliakoff, M. The continuous self aldol condensation of propionaldehyde in supercritical carbon dioxide: a highly selective catalytic route to 2-methylpentenal. Green Chem., 2009, 11, 409-416. [CrossRef]
- Spivey, J. J.; Gogate, M. R. Research Triangle Institute, US EPA Grant, Pollution Prevention in Industrial Condensation Reactions, 1996.
- Matsui, K.; Kawanami, H.; Ikushima, Y.; Hayashi. H. Control of self-aldol condensation by pressure manipulation under compressed CO2. Chem. Commun., 2003, 2502-2503. [CrossRef]
- Sharma, S. K.; Parikh, P. A.; Jasra, R. V. Solvent free aldol condensation of propanal to 2-methylpentenal using solid base catalysts. J. Mol. Catal. A: Chem., 2007, 278, 135-144. [CrossRef]
- Aratake, S.; Itoh, T.; Okano, T.; Usui, T.; Shoji, M.; Hayashi, Y. Small organic molecule in enantioselective, direct aldol reaction “in water”. Chem. Commun., 2007, 2524-2526. [CrossRef]
- Corberán, R.; Sanaú, M.; Peris, E. Highly Stable Cp*−Ir(III) Complexes with N-Heterocyclic Carbene Ligands as C−H Activation Catalysts for the Deuteration of Organic Molecules. J. Am. Chem. Soc. 2006, 128, 3974–3979. [CrossRef]
- Wei, J.; Ma, T.; Ma, X.; Guan, W.; Liu, Q.; Yang, J. Study on thermodynamic properties and estimation of polarity of ionic liquids {[Cnmmim][NTf2] (n = 2, 4)}.
- RSC Adv., 2014, 4, 30725–30732. [CrossRef]
- Fraile, J. M.; García, J. I.; Herrerías, C. I.; Mayoral, J. A.; Carrié, D.; Vaultier, M. Enantioselective cyclopropanation reactions in ionic liquids. Tetrahedron: Asymmetry, 2001, 12, 1891–1894. [CrossRef]
- Hart, R.; Pollet, P.; Hahne, D. J.; John, E.; Llopis-Mestre, V.; Blasucci, V.; Huttenhower, H.; Leitner, W.; Eckerta, C. A.; Liotta, C. L. Benign coupling of reactions and separations with reversible ionic liquids, Tetrahedron, 2010, 66, 1082–1090.


| Entry | Solvent | Catalyst | [1,3-PDO]:[Ir] | Base | [Base]:[1,3-PDO] | T | Time | 1,3-PDO conversiona | 2 | 3 | 4 | TOFb |
| [°C] | [d] | [%] | [%] | [%] | [%] | [s−1] (×103) | ||||||
| 1 | N1,8,8,8NTf2 | Ir-5H | 71.9 | K2CO3 | 0.0310 | 100 | 4 | 79 | 7.1 | 79.4 | 13.5 | 0.16 |
| 2 | N1,8,8,8NTf2 | Ir-5H | 193.8 | K2CO3 | 0.0312 | 100 | 4 | 79 | 3.9 | 78.1 | 18.0 | 0.44 |
| 3 | N1,8,8,8NTf2 | Ir-5H | 72.2 | K2CO3 | 0.0304 | 120 | 1 | 82 | 9.3 | 77.5 | 13.2 | 0.17 |
| 4 | N1,8,8,8NTf2 | Ir-5H | 193.5 | K2CO3 | 0.0307 | 120 | 1 | 87 | 4.0 | 80.6 | 15.3 | 0.49 |
| 5 | N1,8,8,8NTf2 | Ir-5H | 72.2 | K2CO3 | 0.0306 | 150 | 1 | 84 | 7.6 | 75.8 | 16.7 | 0.23 |
| 6 | N1,8,8,8NTf2 | Ir-5H | 189.4 | K2CO3 | 0.0306 | 150 | 1 | 89 | 4.6 | 76.9 | 18.5 | 0.65 |
| 7 | N1,8,8,8NTf2 | Ir-5H | 69.5 | K2CO3 | 0.0312 | 180 | 1 | 94 | 6.9 | 76.3 | 16.8 | 0.76 |
| 8 | N1,8,8,8NTf2 | Ir-5H | 194.8 | K2CO3 | 0.0309 | 180 | 1 | 98 | 6.9 | 69.4 | 23.6 | 2.23 |
| 9 | EmmimNTf2 | Ir-5H | 72.4 | K2CO3 | 0.0301 | 100 | 7 | 77 | 7.5 | 79.2 | 13.3 | 0.16 |
| 10 | EmmimNTf2 | Ir-5H | 191.9 | K2CO3 | 0.0301 | 100 | 7 | 77 | 10.2 | 80.3 | 9.5 | 0.43 |
| 11 | EmmimNTf2 | Ir-5H | 77.0 | K2CO3 | 0.0302 | 120 | 4 | 60 | 8.1 | 78.4 | 13.5 | 0.10 |
| 12 | EmmimNTf2 | Ir-5H | 187.4 | K2CO3 | 0.0311 | 120 | 4 | 44 | 1.8 | 82.2 | 15.9 | 0.33 |
| 13 | EmmimNTf2 | Ir-5H | 76.5 | K2CO3 | 0.0307 | 150 | 3 | 85 | 19.7 | 67.1 | 13.2 | 0.25 |
| 14 | EmmimNTf2 | Ir-5H | 194.3 | K2CO3 | 0.0305 | 150 | 3 | 68 | 6.2 | 75.5 | 18.3 | 0.51 |
| 15 | EmmimNTf2 | Ir-5H | 71.7 | K2CO3 | 0.0309 | 180 | 1 | 88 | 43.8 | 42.4 | 13.8 | 0.41 |
| 16 | EmmimNTf2 | Ir-5H | 196.1 | K2CO3 | 0.0307 | 180 | 1 | 93 | 53.0 | 37.3 | 9.7 | 2.15 |
| 17 | N1,8,8,8NTf2 | No catalyst | / | K2CO3 | 0.0318 | 120 | 1 | 1 | 1.0 | 1.0 | 1.0 | / |
| 18 | EmmimNTf2 | No catalyst | / | K2CO3 | 0.0307 | 150 | 3 | / | / | / | / | / |
| 19 | No IL | Ir-5H | 179.8 | K2CO3 | 0.0311 | 120 | 1 | 52 | 9.8 | 60.4 | 29.8 | 1.07 |
| 20 | Toluene | Ir-5H | 177.8 | K2CO3 | 0.0315 | 120 | 1 | 70 | 92.6 | 7.4 | 0.0 | 1.45 |
| 21 | 1,3-PDOc | Ir-5H | 191.6 | K2CO3 | 0.0305 | 120 | 3 | 138 | 30.5 | 65.2 | 4.3 | 1.02 |
| 22 | N1,8,8,8NTf2 | Ir-5H | 180.1 | No base | / | 150 | 3 | 0 | / | / | / | 0.00 |
| 23 | N1,8,8,8NTf2 | Ir-5H | 183.0 | K2CO3 | 0.0151 | 150 | 3 | 23 | 34.0 | 55.4 | 10.5 | 0.16 |
| 24 | N1,8,8,8NTf2 | Ir-5H | 185.7 | K2CO3 | 0.0762 | 150 | 3 | 12 | 36.2 | 44.6 | 19.2 | 0.08 |
| Entry | Solvent | [1,3-PDO]:[Ir] | Base | [Base]:[1,3-PDO] | T | Time | 1,3-PDO conversiona | 2 | 3 | 4 | TOFb |
| [°C] | [d] | [%] | [%] | [%] | [%] | [s−1] (×103) | |||||
| 1 | N1,8,8,8NTf2 | 78.2 | K2CO3 | 0.0309 | 100 | 7 | 73 | 44.2 | 50.0 | 5.8 | 0.09 |
| 2 | N1,8,8,8NTf2 | 215.5 | K2CO3 | 0.0296 | 100 | 7 | 97 | 46.5 | 48.4 | 5.1 | 0.35 |
| 3 | N1,8,8,8NTf2 | 76.2 | K2CO3 | 0.0304 | 120 | 5 | 99 | 42.4 | 53.4 | 4.2 | 0.18 |
| 4 | N1,8,8,8NTf2 | 196.0 | K2CO3 | 0.0318 | 120 | 5 | 99 | 49.8 | 47.3 | 3.0 | 0.45 |
| 5 | N1,8,8,8NTf2 | 77.6 | K2CO3 | 0.0310 | 150 | 3 | 99 | 8.3 | 82.1 | 9.7 | 0.30 |
| 6 | N1,8,8,8NTf2 | 204.6 | K2CO3 | 0.0310 | 150 | 3 | 99 | 8.0 | 86.6 | 5.4 | 0.79 |
| 7 | N1,8,8,8NTf2 | 77.2 | K2CO3 | 0.0306 | 180 | 1 | 99 | 45.2 | 30.8 | 24.0 | 0.89 |
| 8 | N1,8,8,8NTf2 | 212.0 | K2CO3 | 0.0303 | 180 | 1 | 99 | 32.2 | 55.9 | 11.9 | 2.45 |
| 9 | EmmimNTf2 | 77.7 | K2CO3 | 0.0302 | 100 | 7 | 99 | 44.7 | 50.4 | 4.9 | 0.13 |
| 10 | EmmimNTf2 | 187.7 | K2CO3 | 0.0309 | 100 | 7 | 99 | 18.4 | 70.9 | 10.7 | 0.31 |
| 11 | EmmimNTf2 | 86.1 | K2CO3 | 0.0279 | 120 | 5 | 98 | 22.5 | 64.8 | 12.7 | 0.20 |
| 12 | EmmimNTf2 | 212.2 | K2CO3 | 0.0310 | 120 | 5 | 99 | 12.2 | 76.0 | 11.9 | 0.49 |
| 13 | EmmimNTf2 | 79.1 | K2CO3 | 0.0300 | 150 | 3 | 99 | 55.4 | 35.7 | 8.9 | 0.31 |
| 14 | EmmimNTf2 | 210.7 | K2CO3 | 0.0308 | 150 | 3 | 99 | 46.9 | 47.9 | 5.2 | 0.81 |
| 15 | EmmimNTf2 | 77.7 | K2CO3 | 0.0312 | 180 | 1 | 99 | 44.1 | 41.4 | 14.5 | 0.90 |
| 16 | EmmimNTf2 | 209.0 | K2CO3 | 0.0310 | 180 | 1 | 99 | 28.1 | 63.5 | 8.4 | 2.42 |
| 17 | No IL | 207.1 | K2CO3 | 0.0322 | 120 | 5 | 78 | 56.5 | 43.5 | 0.0 | 0.37 |
| 18 | Toluene | 204.6 | K2CO3 | 0.0308 | 120 | 3 | 76 | 90.9 | 9.1 | 0.0 | 0.60 |
| 19 | 1,3-PDOc | 207.1 | K2CO3 | 0.0307 | 150 | 3 | / | 23.5 | 71.1 | 5.4 | 1.41 |
| 20 | N1,8,8,8NTf2 | 203.5 | No base | / | 150 | 3 | 0 | / | / | / | 0.00 |
| 21 | N1,8,8,8NTf2 | 200.4 | K2CO3 | 0.0148 | 150 | 3 | 39 | 37.2 | 44.2 | 18.6 | 0.30 |
| 22 | N1,8,8,8NTf2 | 178.6 | K2CO3 | 0.0769 | 150 | 3 | 16 | 33.0 | 45.5 | 21.5 | 0.11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




