Submitted:
18 June 2024
Posted:
19 June 2024
Read the latest preprint version here
Abstract
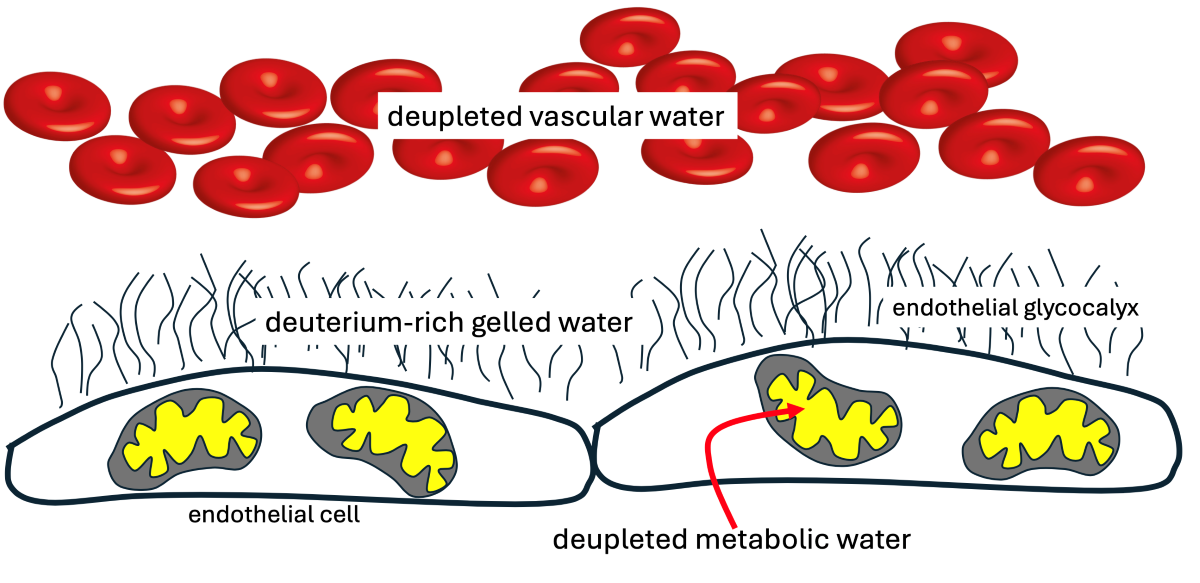
Keywords:
1. Introduction
2. Isomerases and Deuterium
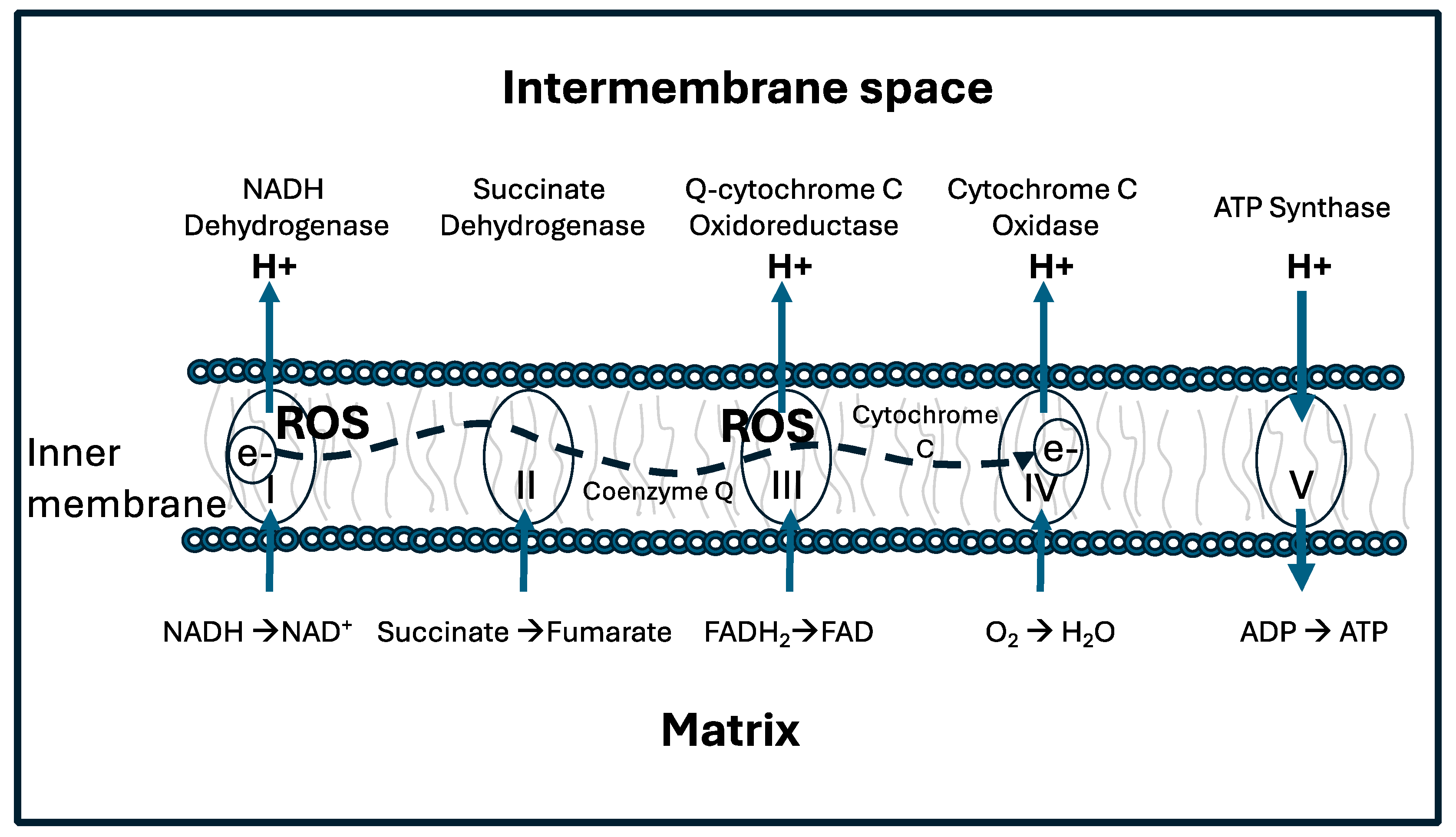
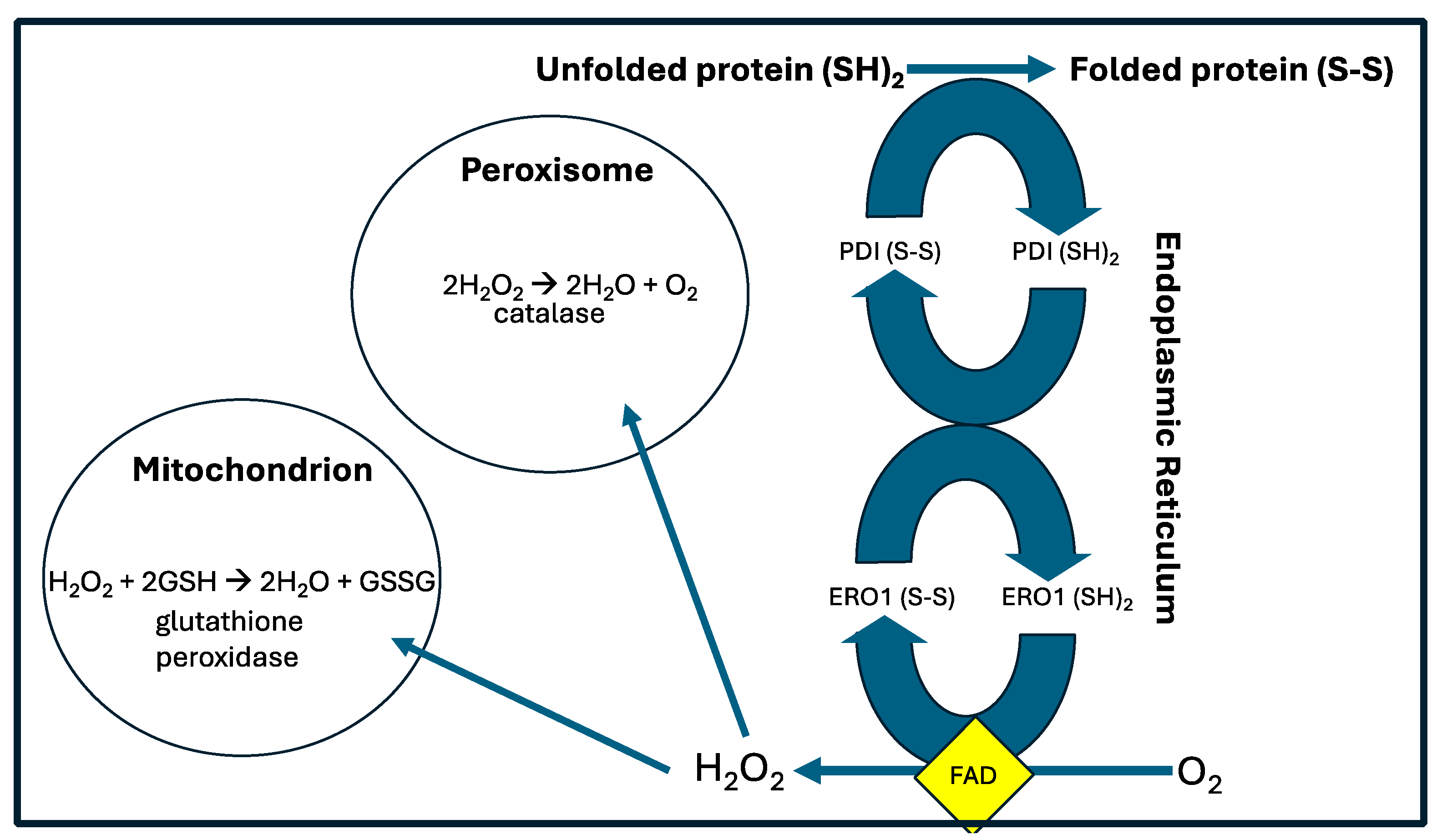
3. Redox Reactions and the “Redox Triangle”
4. Proline
4.1. Cyclic Peptides
4.2. Cyclophilins
4.3. Proline and Collagen
4.4. Collagen Recycling
4.5. Proline Role in Intrinsically Disordered Proteins
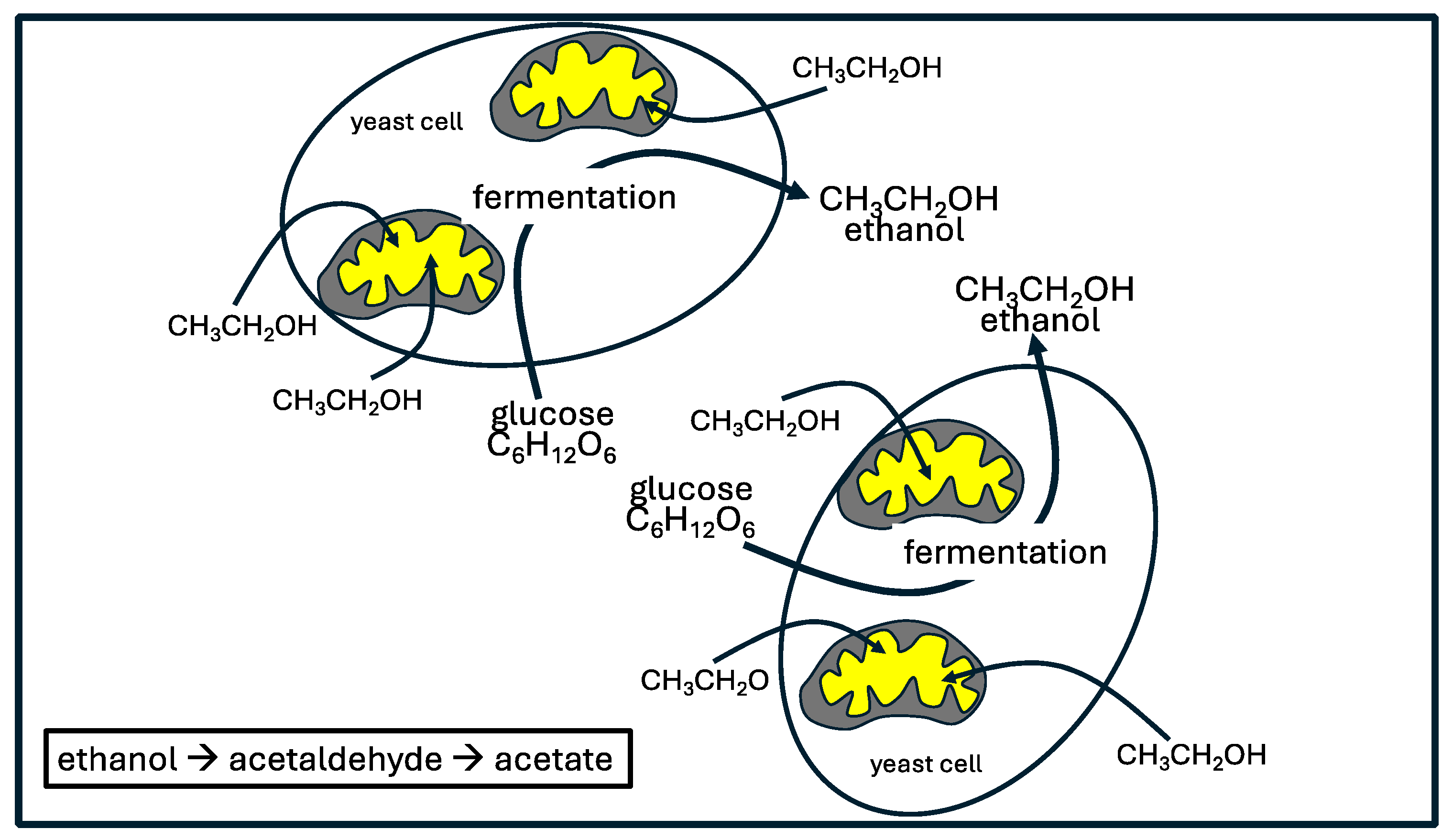
5. Yeast Overgrowth and Serum Ethanol
6. Deuterium and Cancer
6.1. V-ATPase and Microenvironment Proton Deupletion
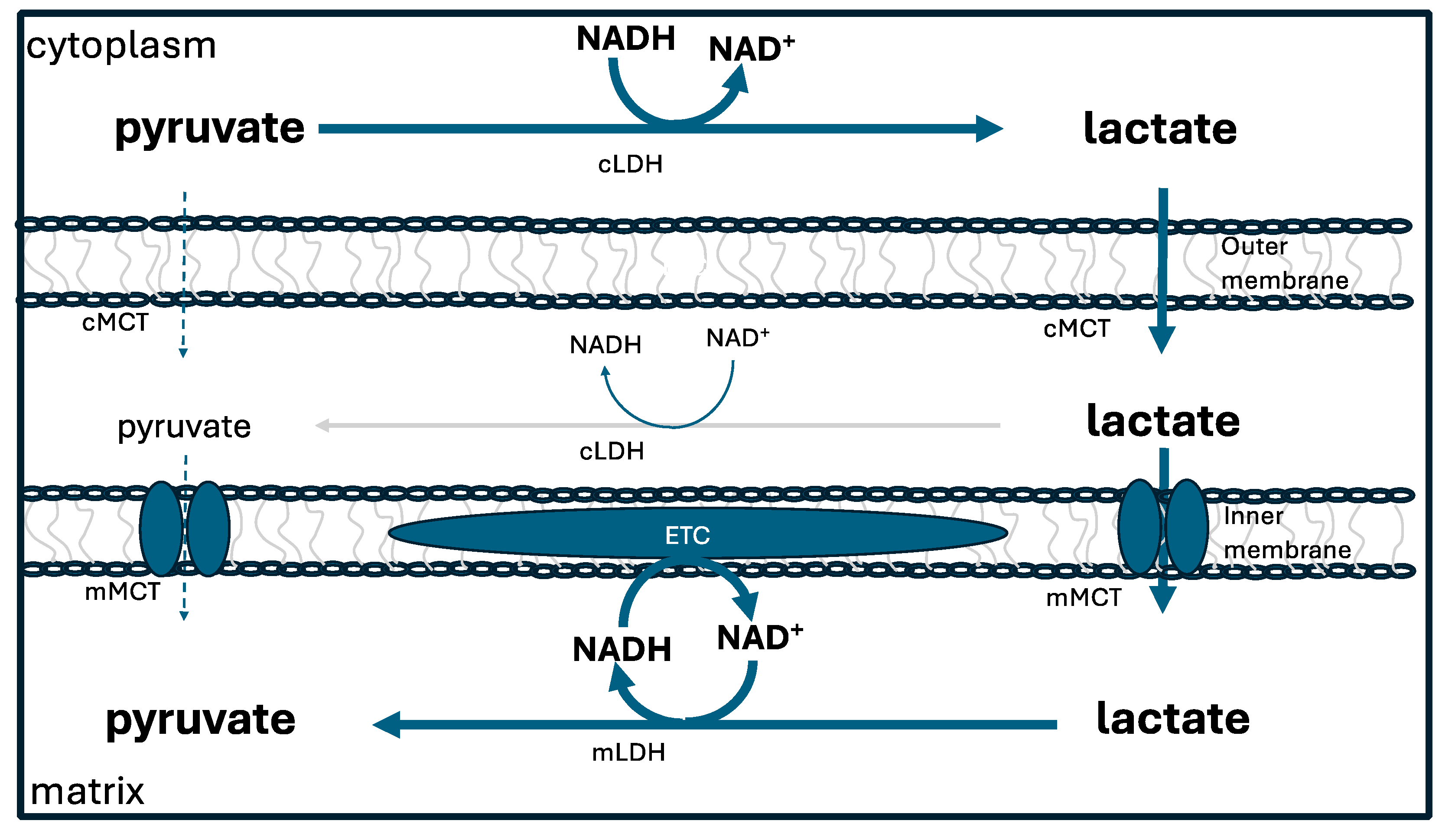
6.2. Lactate
6.3. Succinate Dehydrogenase and Proline Dehydrogenase
6.4. Epidermal Growth Factor and Cancer
6.5. NOX4 and DDW
7. Conclusion
Funding
Acknowledgments
Conflicts of Interest
References
- Boros LG, D’Agostino DP, Katz HE, Roth JP, Meuillet EJ, Somlyai G. Submolecular regulation of cell transformation by deuterium depleting water exchange reactions in the tricarboxylic acid substrate cycle. Med Hypotheses. 2016;87:69-74. [CrossRef]
- Stein LR, Imai S. The dynamic regulation of NAD metabolism in mitochondria. Trends Endocrinol Metab. 2012;23(9):420-8. [CrossRef]
- Olgun, A. Biological effects of deuteronation: ATP synthase as an example. Theor Biol Med Model. 2007;4:9. [CrossRef]
- Murcia Rios A, Vahidi S, Dunn SD, Konermann L. Evidence for a partially stalled γ rotor in F1-ATPase from Hydrogen-Deuterium Exchange Experiments and Molecular Dynamics Simulations. J Am Chem Soc. 2018;140(44):14860-14869. [CrossRef]
- Giulivi C, Zhang K, Arakawa H. Recent advances and new perspectives in mitochondrial dysfunction. Sci Rep 2023; 13: 7977. [CrossRef]
- Nicolson GL. Mitochondrial dysfunction and chronic disease: treatment with natural supplements. Integr Med (Encinitas). 2014;13(4):35-43.
- Jiang C, Liu Y, Wang L, Lu F. Interaction between Heavy Water and Single-Strand DNA: A SERS Study. Molecules. 2022;27(18):6023. PMC9505314. [CrossRef]
- Zlatska A, Gordiienko I, Vasyliev R, Zubov D, Gubar O, Rodnichenko A, Syroeshkin A, Zlatskiy I. In vitro study of deuterium effect on biological properties of human cultured adipose-derived stem cells. ScientificWorldJournal. 2018;2018:5454367. [CrossRef]
- Bar-Peled L, Kory N. Principles and functions of metabolic compartmentalization. Nat Metab. 2022;4(10):1232-1244. [CrossRef]
- Pongratz RL, Kibbey RG, Shulman GI, Cline GW. Cytosolic and mitochondrial malic enzyme isoforms differentially control insulin secretion. J Biol Chem. 2007;282(1):200-7. [CrossRef]
- Navratil AR, Shchepinov MS, Dennis EA. Lipidomics reveals dramatic physiological kinetic isotope effects during the enzymatic oxygenation of polyunsaturated fatty acids ex vivo. J Am Chem Soc. 2018;140(1):235-243. [CrossRef]
- Henkel S, Ertelt M, Sander W. Deuterium and hydrogen tunneling in the hydrogenation of 4-oxocyclohexa-2,5-dienylidene. Chemistry. 2014;20(25):7585-8. [CrossRef]
- Sutcliffe MJ, Scrutton NS. A new conceptual framework for enzyme catalysis. Hydrogen tunnelling coupled to enzyme dynamics in flavoprotein and quinoprotein enzymes. Eur J Biochem. 2002;269(13):3096-102. [CrossRef]
- Hay S, Pudney CR, Scrutton NS. Structural and mechanistic aspects of flavoproteins: probes of hydrogen tunnelling. FEBS J. 2009;276(15):3930-41. [CrossRef]
- Al-Khallaf, H. Isocitrate dehydrogenases in physiology and cancer: biochemical and molecular insight. Cell Biosci. 2017;7:37. [CrossRef]
- Maloney AE, Kopf SH, Zhang Z, McFarlin J, Nelson DB, Masterson AL, Zhang X. Large enrichments in fatty acid 2H/1H ratios distinguish respiration from aerobic fermentation in yeast Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 2024;121(20):e2310771121. [CrossRef]
- Paardekooper LM, Dingjan I, Linders PTA, Staal AHJ, Cristescu SM, Verberk WCEP, van den Bogaart G. Human monocyte-derived dendritic cells produce millimolar concentrations of ROS in phagosomes per second. Front Immunol. 2019;10:1216. [CrossRef]
- Gharibi H, Chernobrovkin AL, Eriksson G, Saei AA, Timmons Z, Kitchener AC, Kalthoff DC, Lidn K, Makarov AA, Zubarev RA. Abnormal (hydroxy)proline deuterium content redefines hydrogen chemical mass. J Am Chem Soc. 2022;144(6):2484-2487. [CrossRef]
- Leadlay PF, Albery WJ, Knowles JR. Energetics of triosephosphate isomerase: deuterium isotope effects in the enzyme-catalyzed reaction. Biochemistry. 1976;15(25):5617-20.
- Lewis CA, Parker SJ, Fiske BP, McCloskey D, Gui DY, Green CR, Vokes NI, Feist AM, Vander Heiden MG, Metallo CM. Tracing compartmentalized NADPH metabolism in the cytosol and mitochondria of mammalian cells. Mol Cell. 2014;55(2):253-63. [CrossRef]
- Sun J, Cui J, He Q, Chen Z, Arvan P, Liu M. Proinsulin misfolding and endoplasmic reticulum stress during the development and progression of diabetes. Mol Aspects Med. 2015;42:105-18. PMC4404191. [CrossRef]
- Pudney CR, Hay S, Scrutton NS. Practical aspects on the use of kinetic isotope effects as probes of flavoprotein enzyme mechanisms. Methods Mol Biol. 2014;1146:161-75.
- Yoboue ED, Sitia R, Simmen T. Redox crosstalk at endoplasmic reticulum (ER) membrane contact sites (MCS) uses toxic waste to deliver messages. Cell Death Dis. 2018;9(3):331. [CrossRef]
- Argyrou A, Blanchard JS. Flavoprotein disulfide reductases: advances in chemistry and function. Prog Nucleic Acid Res Mol Biol. 2004;78:89-142.
- Dunaevsky YE, Tereshchenkova VF, Oppert B, Belozersky MA, Filippova IY, Elpidina EN. Human proline specific peptidases: a comprehensive analysis. Biochim Biophys Acta Gen Subj. 2020;1864(9):129636. [CrossRef]
- Mateos B, Conrad-Billroth C, Schiavina M, Beier A, Kontaxis G, Konrat R, Felli IC, Pierattelli R. The ambivalent role of proline residues in an intrinsically disordered protein: from disorder promoters to compaction facilitators. J Mol Biol. 2020;432(9):3093-3111. [CrossRef]
- Wu WJ, Raleigh DP. Local control of peptide conformation: stabilization of cis proline peptide bonds by aromatic proline interactions. Biopolymers. 1998;45(5):381-94.
- Maguire OR, Taylor B, Higgins EM, Rees M, Cobb SL, Simpkins NS, Hayes CJ, O’Donoghue AC. Unusually high α-proton acidity of prolyl residues in cyclic peptides. Chem Sci. 2020;11(29):7722-7729. [CrossRef]
- Tuccinardi T, Rizzolio F. Editorial: Peptidyl-prolyl isomerases in human pathologies. Front Pharmacol. 2019;10:794. [CrossRef]
- Chen Y, Wu YR, Yang HY, Li XZ, Jie MM, Hu CJ, Wu YY, Yang SM, Yang YB. Prolyl isomerase Pin1: a promoter of cancer and a target for therapy. Cell Death Dis. 2018;9(9):883. [CrossRef] [PubMed]
- Sletten MR, Schoenheimer R. The metabolism of 1(-)-proline studied with the aid of deuterium and isotopic nitrogen. J Biol Chem 1944; 153: 113-132.
- Joo, SH. Cyclic peptides as therapeutic agents and biochemical tools. Biomol Ther (Seoul). 2012;20(1):19-26. [CrossRef]
- Ramadhani D, Maharani R, Gazzali AM, Muchtaridi M. Cyclic peptides for the treatment of cancers: a review. Molecules. 2022;27(14):4428. PMC9317348. [CrossRef]
- Guan J, Li F, Kang D, Anderson T, Pitcher T, Dalrymple-Alford J, Shorten P, Singh-Mallah G. Cyclic glycine-proline (cGP) normalises insulin-like growth factor-1 (IGF-1) function: clinical significance in the ageing brain and in age-related neurological conditions. Molecules. 2023;28(3):1021. [CrossRef]
- Guan J, Krishnamurthi R, Waldvogel HJ, Faull RL, Clark R, Gluckman P. N-terminal tripeptide of IGF-1 (GPE) prevents the loss of TH positive neurons after 6-OHDA induced nigral lesion in rats. Brain Res. 2000;859(2):286-92. [CrossRef]
- Silva-Reis SC, Sampaio-Dias IE, Costa VM, Correia XC, Costa-Almeida HF, Garca-Mera X, Rodrguez-Borges JE. Concise overview of glypromate neuropeptide research: from chemistry to pharmacological applications in neurosciences. ACS Chem Neurosci. 2023;14(4):554-572. [CrossRef]
- Thomson AW, Cameron ID. Immune suppression with cyclosporin A-optimism and caution. Scott Med J. 1981;26(2):139-44. [CrossRef]
- Boyle TJ, Coles RE, Kizilbash AM, Lyerly HK. Effects of cyclosporine on human B-cell lymphoma development in vivo. Surg Oncol. 1992;1(1):79-86. [CrossRef]
- Wang P, Heitman J. The cyclophilins. Genome Biol. 2005;6(7):226. [CrossRef]
- Nigro, P., Pompilio, G., Capogrossi, M. Cyclophilin A: a key player for human disease. Cell Death Dis 2013; 4: e888. [CrossRef]
- Kay EJ, Zanivan S, Rufini A. Proline metabolism shapes the tumor microenvironment: from collagen deposition to immune evasion. Curr Opin Biotechnol. 2023;84:103011. [CrossRef]
- Elrod JW, Molkentin JD. Physiologic functions of cyclophilin D and the mitochondrial permeability transition pore. Circ J. 2013;77(5):1111-22. [CrossRef]
- Castillo EC, Morales JA, Chapoy-Villanueva H, Silva-Platas C, Trevio-Saldaa N, Guerrero-Beltrn CE, Bernal-Ramrez J, Torres-Quintanilla A, Garca N, Youker K, Torre-Amione G, Garca-Rivas G. Mitochondrial hyperacetylation in the failing hearts of obese patients mediated partly by a reduction in SIRT3: the involvement of the mitochondrial permeability transition pore. Cell Physiol Biochem. 2019;53(3):465-479. [CrossRef]
- Endlicher R, Drahota Z, tefkov K, ervinkov Z, Kuera O. The Mitochondrial permeability transition pore-current knowledge of its structure, function, and regulation, and optimized methods for evaluating its functional state. Cells. 2023;12(9):1273. [CrossRef]
- Lam MP, Lau E, Liem DA, Ping P. Cyclophilin D and acetylation: a new link in cardiac signaling. Circ Res. 2013;113(12):1268-9.
- Ansari A, Rahman MS, Saha SK, Saikot FK, Deep A, Kim KH. Function of the SIRT3 mitochondrial deacetylase in cellular physiology, cancer, and neurodegenerative disease. Aging Cell. 2017;16(1):4-16. [CrossRef]
- Li P, Wu G. Roles of dietary glycine, proline, and hydroxyproline in collagen synthesis and animal growth. Amino Acids. 2018;50(1):29-38. [CrossRef]
- Elton DC, Spencer PD, Riches JD, Williams ED. Exclusion zone phenomena in water - a critical review of experimental findings and theories. Int J Mol Sci. 2020;21(14):5041. [CrossRef]
- Pollack, GH. Water, energy and life: Fresh views from the water’s edge. Int J Des Nat Ecodyn. 2010;5(1):27-29. [CrossRef]
- Hwang SG, Hong JK, Sharma A, Pollack GH, Bahng G. Exclusion zone and heterogeneous water structure at ambient temperature. PLoS One. 2018;13(4):e0195057. [CrossRef]
- Kabedev A, Lobaskin V. Structure and elasticity of bush and brush-like models of the endothelial glycocalyx. Sci Rep. 2018;8(1):240. [CrossRef]
- Ninham BW, Battye MJ, Bolotskova PN, Gerasimov RY, Kozlov VA, Bunkin NF. Nafion: New and old insights into structure and function. Polymers (Basel). 2023;15(9):2214. [CrossRef]
- Ikonomidis I, Thymis J, Simitsis P, Koliou GA, Katsanos S, Triantafyllou C, Kousathana F, Pavlidis G, Kountouri A, Polyzogopoulou E, Katogiannis K, Vlastos D, Kostelli G, Triantafyllidi H, Parissis J, Papadavid E, Lekakis J, Filippatos G, Lambadiari V. Impaired endothelial glycocalyx predicts adverse outcome in subjects without overt cardiovascular disease: a 6-year follow-up study. J Cardiovasc Transl Res. 2022;15(4):890-902. [CrossRef]
- Pretorius D, Richter RP, Anand T, Cardenas JC, Richter JR. Alterations in heparan sulfate proteoglycan synthesis and sulfation and the impact on vascular endothelial function. Matrix Biol Plus. 2022;16:100121. [CrossRef]
- Giubertoni G, Feng L, Klein K, Giannetti G, Rutten L, Choi Y, van der Net A, Castro-Linares G, Caporaletti F, Micha D, Hunger J, Deblais A, Bonn D, Sommerdijk N, ari A, Ilie IM, Koenderink GH, Woutersen S. Elucidating the role of water in collagen self-assembly by isotopically modulating collagen hydration. Proc Natl Acad Sci U S A. 2024;121(11):e2313162121. [CrossRef]
- Morgan AA, Rubenstein E. Proline: the distribution, frequency, positioning, and common functional roles of proline and polyproline sequences in the human proteome. PLoS One. 2013;8(1):e53785. [CrossRef]
- Chen XR, Igumenova TI. Regulation of eukaryotic protein kinases by Pin1, a peptidyl-prolyl isomerase. Adv Biol Regul. 2023;87:100938. [CrossRef]
- Ishikawa Y, Boudko S, Bchinger HP. Ziploc-ing the structure: Triple helix formation is coordinated by rough endoplasmic reticulum resident PPIases. Biochim Biophys Acta. 2015;1850(10):1983-93. [CrossRef]
- Paige M, Wang K, Burdick M, Park S, Cha J, Jeffery E, Sherman N, Shim YM. Role of leukotriene A4 hydrolase aminopeptidase in the pathogenesis of emphysema. J Immunol. 2014;192(11):5059-68. [CrossRef]
- Eni-Aganga I, Lanaghan ZM, Balasubramaniam M, Dash C, Pandhare J. Prolidase: a review from discovery to its role in health and disease. Front Mol Biosci. 2021;8:723003. [CrossRef]
- Lupi A, De Riso A, Torre SD, Rossi A, Campari E, Vilarinho L, Cetta G, Forlino A. Characterization of a new PEPD allele causing prolidase deficiency in two unrelated patients: natural-occurrent mutations as a tool to investigate structure-function relationship. J Hum Genet. 2004;49(9):500-506. [CrossRef]
- Stsiapanava A, Olsson U, Wan M, Kleinschmidt T, Rutishauser D, Zubarev RA, Samuelsson B, Rinaldo-Matthis A, Haeggström JZ. Binding of Pro-Gly-Pro at the active site of leukotriene A4 hydrolase/aminopeptidase and development of an epoxide hydrolase selective inhibitor. Proc Natl Acad Sci U S A. 2014;111(11):4227-32. [CrossRef]
- Wright PE, Dyson HJ. Intrinsically unstructured proteins: re-assessing the protein structure-function paradigm. J Mol Biol. 1999;293(2):321-31. [CrossRef]
- Luo Y, Ma B, Nussinov R,, Wei G. Structural insight into tau proteins paradox of intrinsically disordered behavior, self-acetylation activity, and aggregation. Journal of Physical Chemistry Letters 2014; 5: 30263031. [CrossRef]
- Xiao Zhen Zhou, Oliver Kops, Andreas Werner, Pei-Jung Lu, Minhui Shen, Gerlind Stoller, Gerhard Kllertz, Michael Stark, Gunter Fischer, Kun Ping Lu, Pin1-dependent prolyl isomerization regulates dephosphorylation of Cdc25C and tau proteins. Molecular Cell 2000; 6(4): 873-883. 8. [CrossRef]
- He HJ, Wang XS, Pan R, Wang DL, Liu MN, He RQ. The proline-rich domain of tau plays a role in interactions with actin. BMC Cell Biol. 2009;10:81. [CrossRef]
- Gong CX, Iqbal K. Hyperphosphorylation of microtubule-associated protein tau: a promising therapeutic target for Alzheimer disease. Curr Med Chem. 2008;15(23):2321-8. [CrossRef]
- Malter, JS. Pin1 and Alzheimer’s disease. Transl Res. 2023;254:24-33. [CrossRef]
- Blair LJ, Baker JD, Sabbagh JJ, Dickey CA. The emerging role of peptidyl-prolyl isomerase chaperones in tau oligomerization, amyloid processing, and Alzheimer’s disease. J Neurochem. 2015;133(1):1-13. [CrossRef]
- Freitas MA, Marshall AG. Gas phase RNA and DNA ions 2. Conformational dependence of the gas-phase H/D exchange of nucleotide-5’-monophosphates. J Am Soc Mass Spectrom. 2001;12(7):780-5. [CrossRef]
- Robinson JM, Greig MJ, Griffey RH, Mohan V, Laude DA. Hydrogen/deuterium exchange of nucleotides in the gas phase. Anal. Chem. 1998; 70: 3566-3571. [CrossRef]
- Favretto F, Baker JD, Strohker T, Andreas LB, Blair LJ, Becker S, Zweckstetter M. The molecular basis of the interaction of cyclophilinA with α-synuclein. Angew Chem Int Ed Engl. 2020;59(14):5643-5646. [CrossRef]
- Faubert B, Li KY, Cai L, Hensley CT, Kim J, Zacharias LG, Yang C, Do QN, Doucette S, Burguete D, Li H, Huet G, Yuan Q, Wigal T, Butt Y, Ni M, Torrealba J, Oliver D, Lenkinski RE, Malloy CR, Wachsmann JW, Young JD, Kernstine K, De-Berardinis RJ. Lactate metabolism in human lung tumors. Cell. 2017;171(2):358-371.e9. [CrossRef]
- Zewude, RT, Croitoru, K, Das, R, Goldman B, Bogoch II. Auto-brewery syndrome in a 50-year-old woman. CMAJ 2024; 196(21): E724-E727. [CrossRef]
- Tameez Ud Din A, Alam F, Tameez-Ud-Din A, Chaudhary FMD. Auto-brewery syndrome: a clinical dilemma. Cureus. 2020;12(10):e10983. [CrossRef]
- Rabinowitz JD, Enerbck S. Lactate: the ugly duckling of energy metabolism. Nat Metab. 2020;2(7):566-571. [CrossRef]
- Xiao T, Khan A, Shen Y, Chen L, Rabinowitz JD. Glucose feeds the tricarboxylic acid cycle via excreted ethanol in fermenting yeast. Nat Chem Biol 2022; 18: 1380-1387. [CrossRef]
- Zhang X, Wang J, Zubarev RA. Slight deuterium enrichment in water acts as an antioxidant: is deuterium a cell growth regulator? Mol Cell Proteomics. 2020;19(11):1790-1804. PMC7664117. [CrossRef]
- Wang H, Zhu B, Liu C, Fang W, Yang H. [Deuterium-depleted water selectively inhibits nasopharyngeal carcinoma cell proliferation in vitro]. Nan Fang Yi Ke Da Xue Xue Bao. 2012;32(10):1394-9. Chinese.
- Somlyai G, Gy ̈ongyi Z,, Somlyai I, Boros L. LG. Pre-clinical and clinical data confirm the anticancer effect of deuterium depletion. Eur. J. Integr. Med. 2016; 8: 28.
- Yaglova NV, Timokhina EP, Obernikhin SS, Yaglov VV. Emerging role of deuterium/protium disbalance in cell cycle and apoptosis. Int J Mol Sci. 2023;24(4):3107. [CrossRef]
- Yu D, Liu Z. The research progress in the interaction between Candida albicans and cancers. Front Microbiol. 2022;13:988734. [CrossRef]
- Wang X, Zhang W, Wu W, Wu S, Young A, Yan Z. Is Candida albicans a contributor to cancer? A critical review based on the current evidence. Microbiol Res. 2023;272:127370. [CrossRef]
- Seo W, Gao Y, He Y, Sun J, Xu H, Feng D, Park SH, Cho YE, Guillot A, Ren T, Wu R, Wang J, Kim SJ, Hwang S, Liangpunsakul S, Yang Y, Niu J, Gao B. ALDH2 deficiency promotes alcohol-associated liver cancer by activating oncogenic pathways via oxidized DNA-enriched extracellular vesicles. J Hepatol. 2019;71(5):1000-1011. [CrossRef]
- Seitz HK, Stickel F. Acetaldehyde as an underestimated risk factor for cancer development: role of genetics in ethanol metabolism. Genes Nutr. 2010;5(2):121-8. [CrossRef]
- Dohlman AB, Klug J, Mesko M, Gao IH, Lipkin SM, Shen X, Iliev ID. A pan-cancer mycobiome analysis reveals fungal involvement in gastrointestinal and lung tumors. Cell. 2022; 185(20):3807-3822.e12. [CrossRef]
- Teoh F, Pavelka N. How Chemotherapy increases the risk of systemic Candidiasis in cancer patients: current paradigm and future directions. Pathogens. 2016;5(1):6. [CrossRef]
- Li X, Snyder MP. Yeast longevity promoted by reversing aging-associated decline in heavy isotope content. NPJ Aging Mech Dis. 2016;2:16004. [CrossRef]
- Perzov N, Padler-Karavani V, Nelson H, Nelson N. Features of V-ATPases that distinguish them from F-ATPases. FEBS Lett. 2001;504(3):223-8. [CrossRef]
- Kotyk A, Dvorkov M, Koryta J. Deuterons cannot replace protons in active transport processes in yeast. FEBS Lett. 1990;264(2):203-5. [CrossRef]
- Sennoune SR, Bakunts K, Martnez GM, Chua-Tuan JL, Kebir Y, Attaya MN, Martnez-Zaguiln R. Vacuolar H+-ATPase in human breast cancer cells with distinct metastatic potential: distribution and functional activity. Am J Physiol Cell Physiol. 2004;286(6):C1443-52. [CrossRef]
- Jiang X, Wang J, Deng X, Xiong F, Ge J, Xiang B, Wu X, Ma J, Zhou M, Li X, Li Y, Li G, Xiong W, Guo C, Zeng Z. Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol Cancer 2019; 18: 10. [CrossRef]
- Caslin HL, Abebayehu D, Pinette JA, Ryan JJ. Lactate is a metabolic mediator that shapes immune cell fate and function. Front Physiol. 2021;12:688485. [CrossRef]
- Li X, Yang Y, Zhang B, Lin X, Fu X, An Y, Zou Y, Wang JX, Wang Z, Yu T. Lactate metabolism in human health and disease. Signal Transduct Target Ther. 2022;7(1):305. Erratum in: Signal Transduct Target Ther. 2022;7(1):372. [CrossRef]
- Warburg O, Wind F, Negelein E. The metabolism of tumors in the body. J. Gen. Physiol. 1927;8:519-530. [CrossRef]
- DeBerardinis RJ, Chandel NS. We need to talk about the Warburg effect. Nat Metab 2020;2: 127-129. [CrossRef]
- Brooks GA, Dubouchaud H, Brown M, Sicurello JP, Butz CE. Role of mitochondrial lactate dehydrogenase and lactate oxidation in the intracellular lactate shuttle. Proc Natl Acad Sci U S A. 1999;96(3):1129-34. [CrossRef]
- Grimshaw CE, Cleland WW. Deuterium isotope effects on lactate dehydrogenase using L-2-hydroxysuccinamate and effect of an inhibitor in the variable substrate on observed isotope effects. Biochemistry. 1980;19(14):3153-7. [CrossRef]
- Potter M, Newport E, Morten KJ. The Warburg effect: 80 years on. Biochem Soc Trans. 2016;44(5):1499-1505. [CrossRef]
- Barya M, Semeniuk-Wojta A, Rg L, Kraj L, Mayszko M, Stec R. Oncometabolites – a link between cancer cells and tumor microenvironment. Biology (Basel). 2022;11(2):270. [CrossRef]
- Dalla Pozza E, Dando I, Pacchiana R, Liboi E, Scupoli MT, Donadelli M, Palmieri M. Regulation of succinate dehydrogenase and role of succinate in cancer. Semin Cell Dev Biol. 2020;98:4-14. [CrossRef]
- Fedotcheva NI, Kondrashova MN, Litvinova EG, Zakharchenko MV, Khunderyakova NV, Beloborodova NV. Modulation of the activity of succinate dehydrogenase by acetylation with chemicals, drugs, and microbial metabolites. Biophysics 2018; 63: 743-750. [CrossRef]
- Kuo CC, Wu JY, Wu KK. Cancer-derived extracellular succinate: a driver of cancer metastasis. J Biomed Sci 2022; 29: 93. [CrossRef]
- Goncalves RL, Rothschild DE, Quinlan CL, Scott GK, Benz CC, Brand MD. Sources of superoxide/H2O2 during mitochondrial proline oxidation. Redox Biol. 2014;2:901-9. [CrossRef]
- Choudhury D, Rong N, Senthil Kumar HV, Swedick S, Samuel RZ, Mehrotra P, Toftegaard J, Rajabian N, Thiyagarajan R, Podder AK, Wu Y, Shahini S, Seldeen KL, Troen B, Lei P, Andreadis ST. Proline restores mitochondrial function and reverses aging hallmarks in senescent cells. Cell Rep. 2024;43(2):113738. [CrossRef]
- Serrano H, Blanchard JS. Kinetic and isotopic characterization of L-proline dehydrogenase from Mycobacterium tuberculosis. Biochemistry. 2013;52(29):5009-15. [CrossRef]
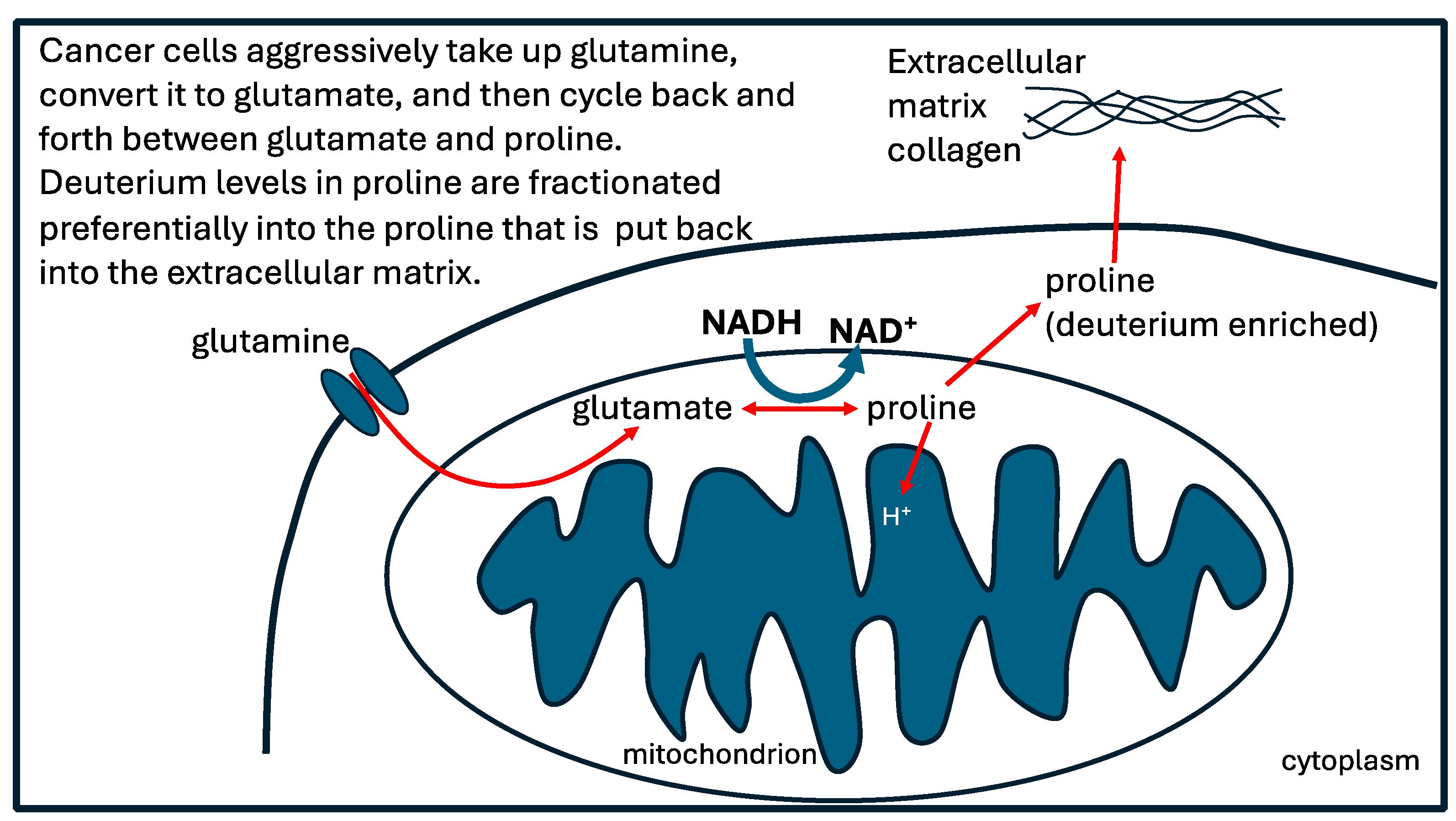
- Phang JM, Liu W, Hancock CN, Fischer JW. Proline metabolism and cancer: emerging links to glutamine and collagen. Current Opinion in Clinical Nutrition and Metabolic Care 2015; 18(1):71-77. [CrossRef]
- Kory N, Uit de Bos J, van der Rijt S, Jankovic N, Gra M, Arp N, Pena IA, Prakash G, Chan SH, Kunchok T, Lewis CA, Sabatini DM. MCART1/SLC25A51 is required for mitochondrial NAD transport. Sci Adv. 2020;6(43):eabe5310. [CrossRef]
- Li Y, Bie J, Zhao L, Song C, Zhang T, Li M, Yang C, Luo J. SLC25A51 promotes tumor growth through sustaining mitochondria acetylation homeostasis and proline biogenesis. Cell Death Differ. 2023;30(8):1916-1930. [CrossRef]
- Kay EJ, Paterson K, Riera-Domingo C, Sumpton D, Dbritz JHM, Tardito S, Boldrini C, Hernandez-Fernaud JR, Athineos D, Dhayade S, Stepanova E, Gjerga E, Neilson LJ, Lilla S, Hedley A, Koulouras G, McGregor G, Jamieson C, Johnson RM, Park M, Kirschner K, Miller C, Kamphorst JJ, Loayza-Puch F, Saez-Rodriguez J, Mazzone M, Blyth K, Zagnoni M, Zanivan S. Cancer-associated fibroblasts require proline synthesis by PYCR1 for the deposition of pro-tumorigenic extracellular matrix. Nat Metab. 2022;4(6):693-710. [CrossRef]
- Sasaki T, Hiroki K, Yamashita Y. The role of epidermal growth factor receptor in cancer metastasis and microenvironment. Biomed Res Int. 2013;2013:546318. [CrossRef]
- Patel A, Unni N, Peng Y. The changing paradigm for the treatment of HER2-positive breast cancer. Cancers (Basel). 2020;12(8):2081. [CrossRef]
- Yang L, Li Y, Zhang Y. Identification of prolidase as a high affinity ligand of the ErbB2 receptor and its regulation of ErbB2 signaling and cell growth. Cell Death Dis. 2014;5(5):e1211. [CrossRef]
- Shanmugasundaram K, Nayak BK, Friedrichs WE, Kaushik D, Rodriguez R, Block K. NOX4 functions as a mitochondrial energetic sensor coupling cancer metabolic reprogram- ming to drug resistance. Nat Commun. 2017;8(1):997. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



