Submitted:
14 June 2024
Posted:
14 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
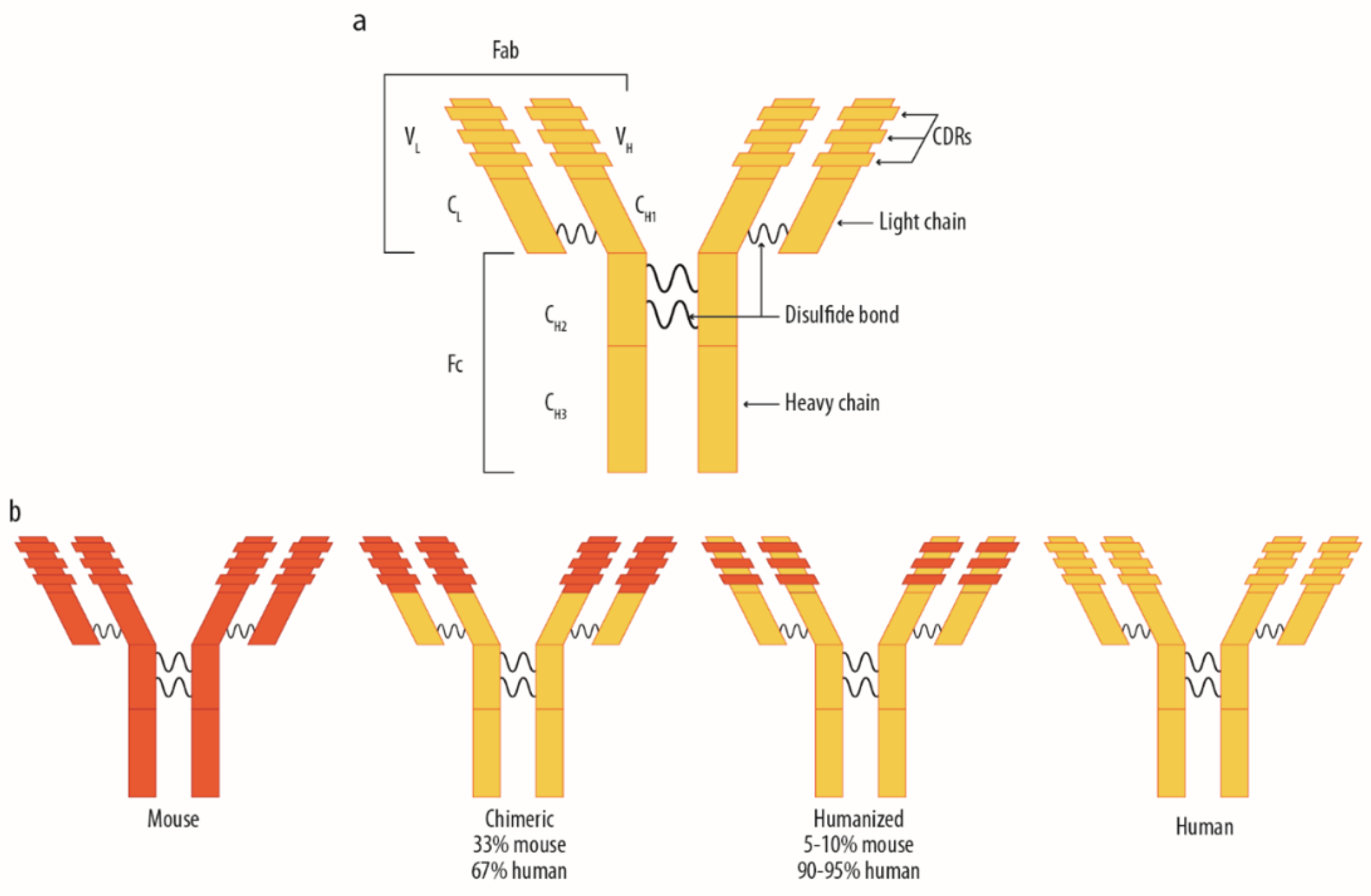
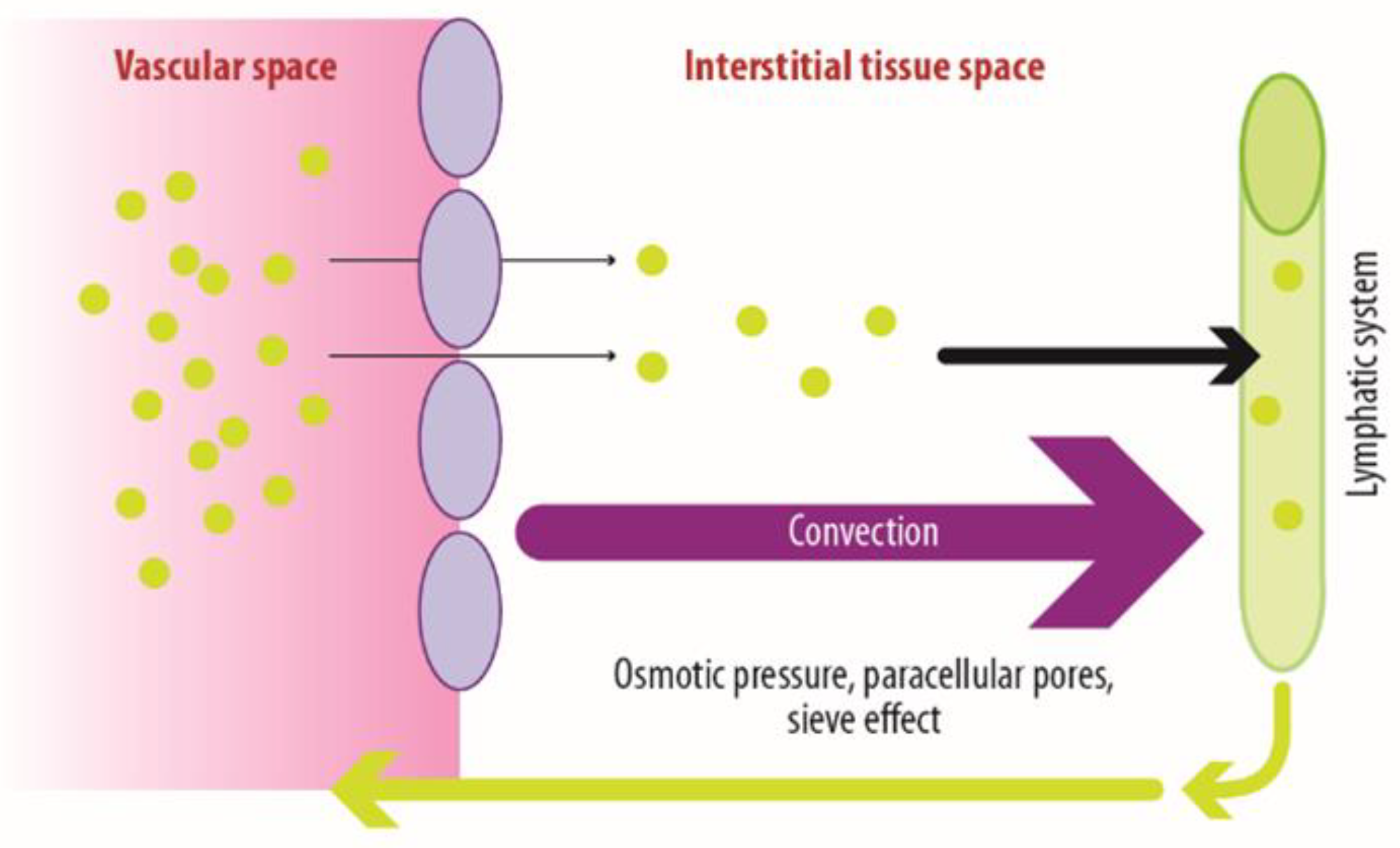
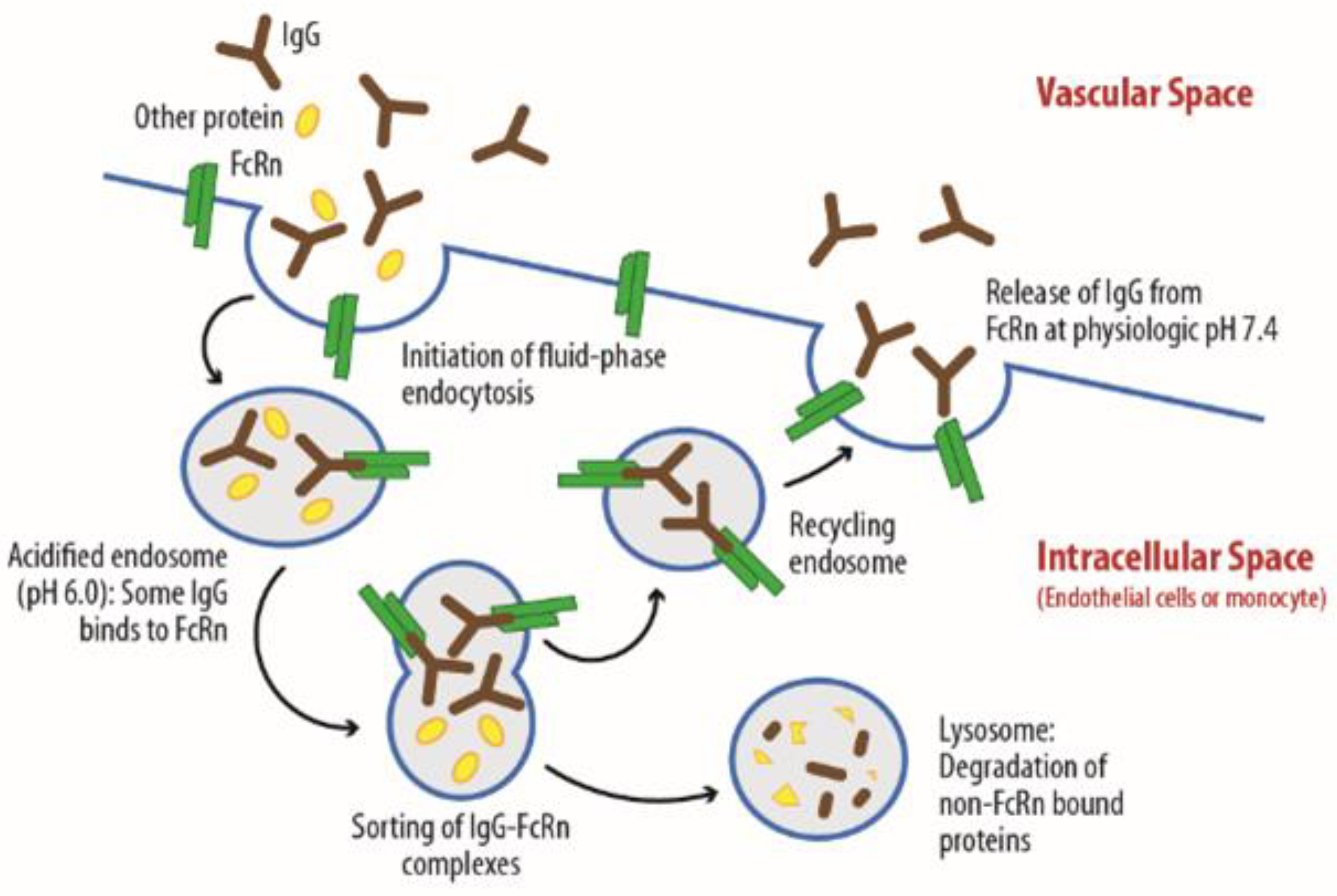
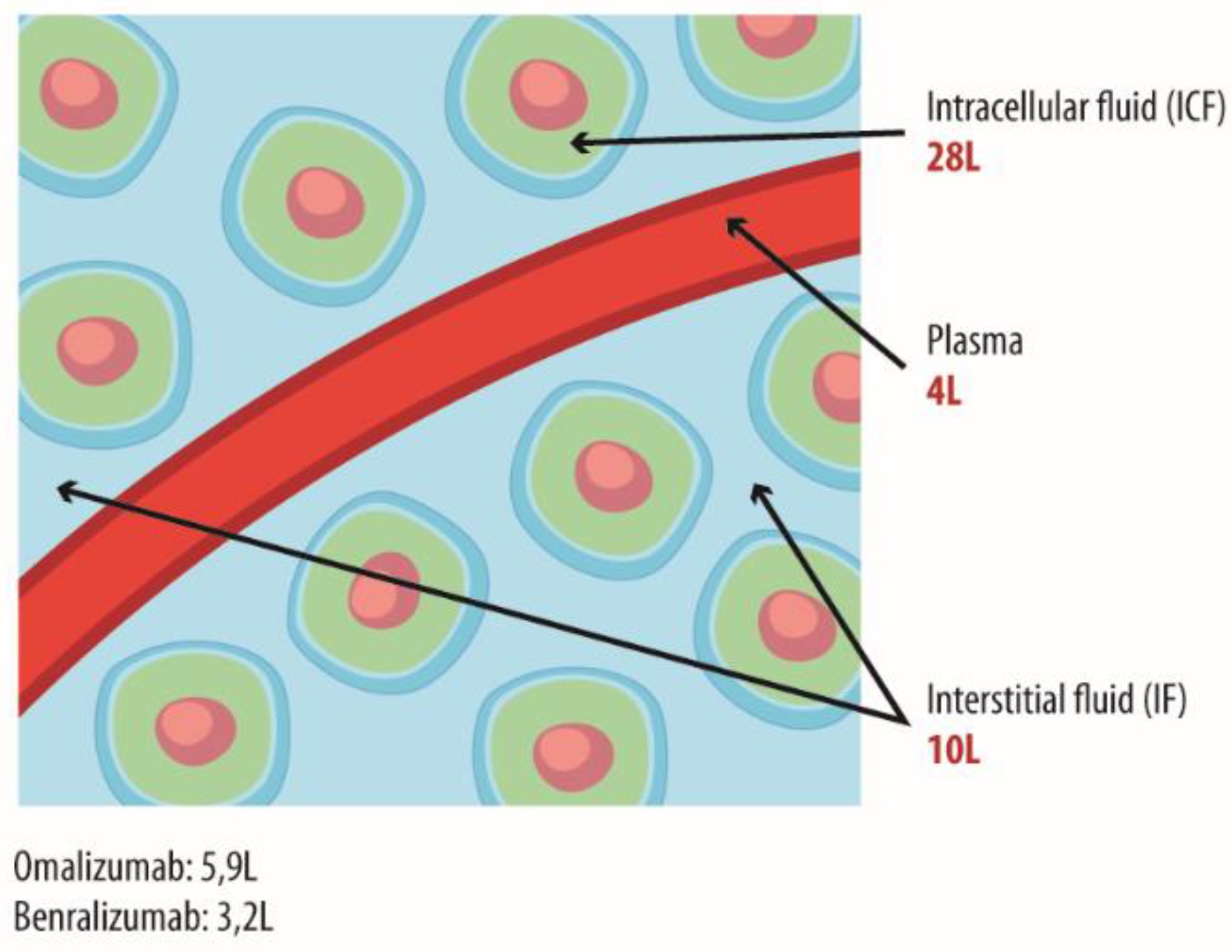
1.1. The Use of Monoclonal Antibodies for Therapeutic Purposes: Distribution Mechanisms and Related Factors
1.2. Mechanism of Action of Therapeutic IgGs for Severe Asthma
1.3. Benralizumab in Severe Asthma: Mechanism of Action and Kinetics
1.3.1. Mechanism of Action
1.3.2. Kinetics of Benralizumab
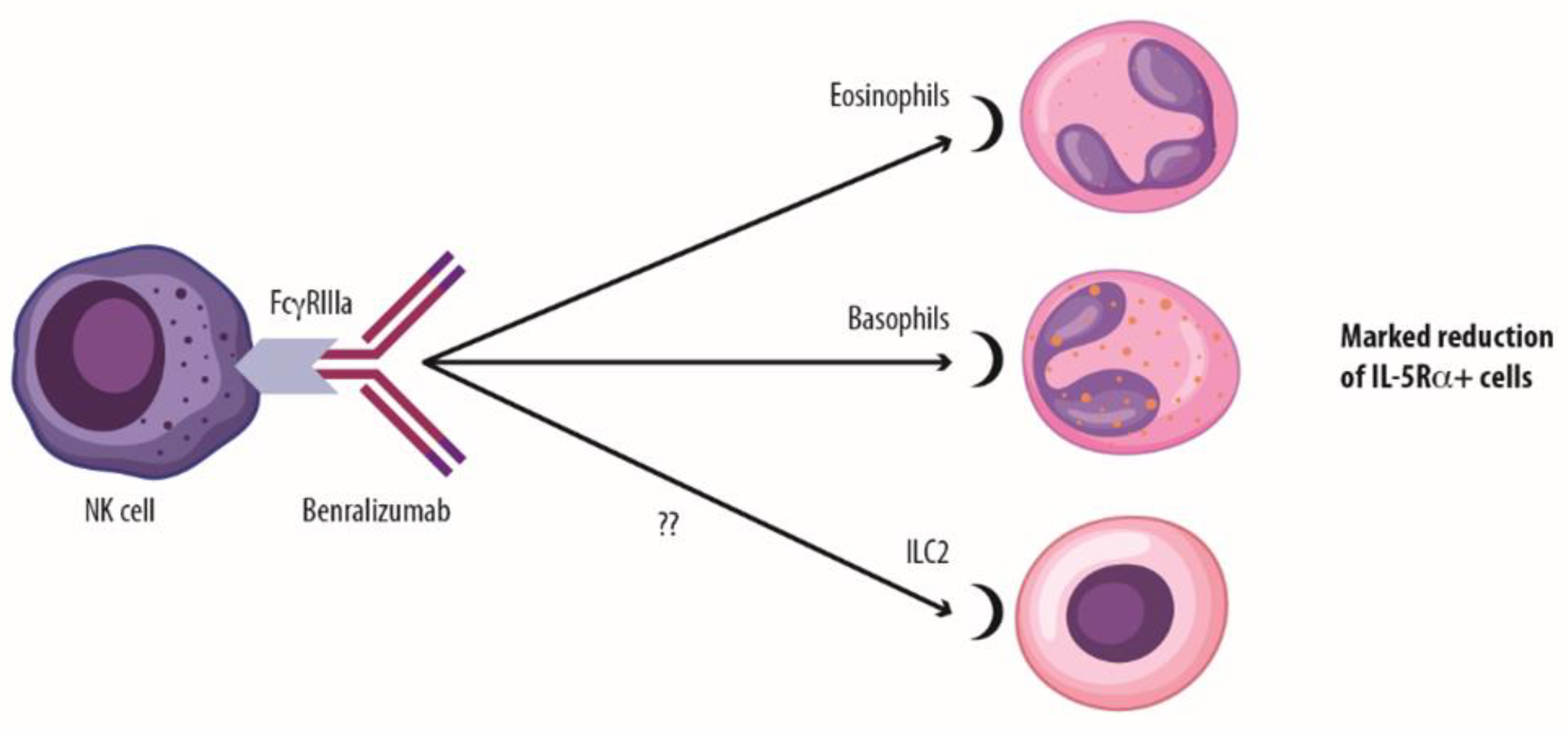
1.4. A Potential third Mechanism of Action of Benralizumab?
1.5. Looking over the Fence: New Insights on Benralizumab Activity
1.6. Benralizumab and Oral Corticosteroids Sparing Effect: As the Mechanism of Action Entails a Clinical Advantage
1.7. Concluding and Innovative Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of interest
Abbreviations
| ADCC | Antibody-dependent cell-mediated cytotoxicity |
| ADCP | Antibody-dependent cellular phagocytosis |
| AER | Annual exacerbation rate |
| CDC | Complement-dependent cytotoxicity |
| EC 90% | 90% effective concentrations |
| EGPA | Eosinophilic granulomatosis with polyangiitis |
| IgG | IgG antibody |
| IL; ILC2; NK | Interleukin; innate type 2 lymphoid cells; natural killer |
| Q4W/Q8W | Every 4 weeks/every 8 weeks |
| S.S. | Steady state |
| Th | T-helper lymphocyte |
| Vd | Distribution volume |
References
- Rönnebjerg, L.; Axelsson, M.; Kankaanranta, H.; Backman, H.; Rådinger, M.; Lundbäck, B.; Ekerljung, L. Severe Asthma in a General Population Study: Prevalence and Clinical Characteristics. J Asthma Allergy. 2021, 14, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- (Global Initiative for Asthma (GINA). Difficult-to-treat & Severe Asthma in adolescent and adult patients – Diagnosis and Management. 2023. Available from: https://ginasthma.org/severeasthma).
- Shaw, D.E.; Sousa, A.R.; Fowler, S.J.; Fleming, L.J.; Roberts, G.; Corfield, J.; Pandis, I.; Bansal, A.T.; Bel, E.H.; Auffray, C.; Compton, C.H.; Bisgaard, H.; Bucchioni, E.; Caruso, M.; Chanez, P.; Dahlén, B.; Dahlen, S.E.; Dyson, K.; Frey, U.; Geiser, T.; Gerhardsson de Verdier, M.; Gibeon, D.; Guo, Y.K.; Hashimoto, S.; Hedlin, G.; Jeyasingham, E.; Hekking, P.P.; Higenbottam, T.; Horváth, I.; Knox, A.J.; Krug, N.; Erpenbeck, V.J.; Larsson, L.X.; Lazarinis, N.; Matthews, J.G.; Middelveld, R.; Montuschi, P.; Musial, J.; Myles, D.; Pahus, L.; Sandström, T.; Seibold, W.; Singer, F.; Strandberg, K.; Vestbo, J.; Vissing, N.; von Garnier, C.; Adcock, I.M.; Wagers, S.; Rowe, A.; Howarth, P.; Wagener, A.H.; Djukanovic, R.; Sterk, P.J.; Chung, K.F.; U-BIOPRED Study Group. Clinical and inflammatory characteristics of the European U-BIOPRED adult severe asthma cohort. Eur Respir J. 2015, 46, 1308–1321. [Google Scholar] [CrossRef] [PubMed]
- Poznanski, S.M.; Mukherjee, M.; Zhao, N.; Huang, C.; Radford, K.; Ashkar, A.A.; Nair, P. Asthma exacerbations on benralizumab are largely non-eosinophilic. Allergy. 2021, 76, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Perez de Llano, L.; Tran, T.N.; Al-Ahmad, M.; Alacqua, M.; Bulathsinhala, L.; Busby, J.; Canonica, G.W.; Carter, V.; Chaudhry, I.; Christoff, G.C.; Cosio, B.G.; Costello, R.W.; FitzGerald, J.M.; Heaney, L.G.; Heffler, E.; Iwanaga, T.; Jackson, D.J.; Kerkhof, M.; Rhee, C.K.; Menzies-Gow, A.N.; Murray, R.; Papadopoulos, N.G.; Papaioannou, A.L.; Pfeffer, P.E.; Popov, T.A.; Price, C.A.; Sadatsafavi, M.; Tohda, Y.; Wang, E.; Wechsler, M.; Zangrilli, J.G.; Price, D.B. Characterization of eosinophilic and non-eosinophilic severe asthma phenotypes and proportion of patients with these phenotypes in the International Severe Asthma Registry (ISAR). Am J Respir Crit Care Med. 2020, 201, A4525. [Google Scholar] [CrossRef]
- Buhl R, Humbert M, Bjermer L, et al. Severe eosinophilic asthma: a roadmap to consensus. Eur Respir J 2017, 49, 1700634. [CrossRef]
- Mukherjee, M.; Huang, C.; Venegas-Garrido, C.; Zhang, K.; Bhalla, A.; Ju, X.; O’Byrne, P.M.; Svenningsen, S.; Sehmi, R.; Nair, P. Benralizumab Normalizes Sputum Eosinophilia in Severe Asthma Uncontrolled by Anti-IL-5 Antibodies: A Single-Blind, Placebo-controlled Clinical Trial. Am J Respir Crit Care Med. 2023, 208, 1330–1335. [Google Scholar] [CrossRef] [PubMed]
- Grayson, M.H.; Feldman, S.; Prince, B.T.; Patel, P.J.; Matsui, E.C.; Apter, A.J. Advances in asthma in 2017: Mechanisms, biologics, and genetics. J Allergy Clin Immunol. 2018, 142, 1423–1436. [Google Scholar] [CrossRef] [PubMed]
- Scioscia, G.; Tondo, P.; Nolasco, S.; Pelaia, C.; Carpagnano, G.E.; Caiaffa, M.F.; Valenti, G.; Maglio, A.; Papia, F.; Triggiani, M.; Crimi, N.; Pelaia, G.; Vatrella, A.; Foschino Barbaro, M.P.; Crimi, C. Benralizumab in patients with severe eosinophilic asthma: a multicentre real-life experience. J Clin Med. 2023, 12, 4362. [Google Scholar] [CrossRef] [PubMed]
- Di Bona, D.; Crimi, C.; D’Uggento, A.M.; Benfante, A.; Caiaffa, M.F.; Calabrese, C.; Campisi, R.; Carpagnano, G.E.; Ciotta, D.; D’Amato, M.; Pelaia, C.; Pelaia, G.; Pellegrino, S.; Scichilone, N.; Scioscia, G.; Ribecco, N.; Spadaro, G.; Valenti, G.; Vatrella, A.; Crimi, N.; Macchia, L. Effectiveness of benralizumab in severe eosinophilic asthma: Distinct sub-phenotypes of response identified by cluster analysis. Clin Exp Allergy. 2022, 52, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Roan, F.; Obata-Ninomiya, K.; Ziegler, S.F. Epithelial cell-derived cytokines: more than just signaling the alarm. J Clin Invest. 2019, 129, 1441–1451. [Google Scholar] [CrossRef]
- Kolbeck R, Kozhich A, Koike M, Peng L, Andersson CK, Damschroder MM, Reed JL, Woods R, Dall’acqua WW, Stephens GL, Erjefalt JS, Bjermer L, Humbles AA, Gossage D, Wu H, Kiener PA, Spitalny GL, Mackay CR, Molfino NA, Coyle AJ. MEDI-563, a humanized anti-IL-5 receptor alpha mAb with enhanced antibody-dependent cell-mediated cytotoxicity function. J Allergy Clin Immunol. 2010, 125, 1344–1353. [CrossRef]
- Bleecker ER, FitzGerald JM, Chanez P, Papi A, Weinstein SF, Barker P, Sproule S, Gilmartin G, Aurivillius M, Werkström V, Goldman M; SIROCCO study investigators. Efficacy and safety of benralizumab for patients with severe asthma uncontrolled with high-dosage inhaled corticosteroids and long-acting β2-agonists (SIROCCO): a randomised, multicentre, placebo-controlled phase 3 trial. Lancet. 2016, 388, 2115–2127. [CrossRef]
- FitzGerald JM, Bleecker ER, Nair P, Korn S, Ohta K, Lommatzsch M, Ferguson GT, Busse WW, Barker P, Sproule S, Gilmartin G, Werkström V, Aurivillius M, Goldman M; CALIMA study investigators. Benralizumab, an anti-interleukin-5 receptor α monoclonal antibody, as add-on treatment for patients with severe, uncontrolled, eosinophilic asthma (CALIMA): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2016, 388, 2128–2141. [CrossRef]
- Nair P, Wenzel S, Rabe KF, Bourdin A, Lugogo NL, Kuna P, Barker P, Sproule S, Ponnarambil S, Goldman M; ZONDA Trial Investigators. Oral Glucocorticoid-Sparing Effect of Benralizumab in Severe Asthma. N Engl J Med. 2017, 376, 2448–2458. [CrossRef]
- Busse WW, Bleecker ER, FitzGerald JM, Ferguson GT, Barker P, Sproule S, Olsson RF, Martin UJ, Goldman M; BORA study investigators. Long-term safety and efficacy of benralizumab in patients with severe, uncontrolled asthma: 1-year results from the BORA phase 3 extension trial. Lancet Respir Med. 2019, 7, 46–59. [CrossRef]
- Cameli, P.; Aliani, M.; Altieri, E.; Bracciale, P.; Brussino, L.; Caiaffa, M.F.; Canonica, G.W.; Caruso, C.; Centanni, S.; D’Amato, M.; De Michele, F.; Del Giacco, S.; Di Marco, F.; Pelaia, G.; Rogliani, P.; Romagnoli, M.; Schino, P.; Schroeder, J.W.; Senna, G.; Vultaggio, A.; Benci, M.; Boarino, S.; Menzella, F. Sustained Effectiveness of Benralizumab in Naïve and Biologics-Experienced Severe Eosinophilic Asthma Patients: Results from the ANANKE Study. J Asthma Allergy. 2024, 17, 273–290. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, J.E.; Hearn, A.P.; Dhariwal, J.; d’Ancona, G.; Douiri, A.; Roxas, C.; Fernandes, M.; Green, L.; Thomson, L.; Nanzer, A.M.; Kent, B.D.; Jackson, D.J. Real-World Effectiveness of Benralizumab in Severe Eosinophilic Asthma. Chest. 2021, 159, 496–506. [Google Scholar] [CrossRef]
- Korn, S.; Bourdin, A.; Chupp, G.; Cosio, B.G.; Arbetter, D.; Shah, M.; Gil, E.G. Integrated Safety and Efficacy Among Patients Receiving Benralizumab for Up to 5 Years. J Allergy Clin Immunol Pract. 2021, 9, 4381–4392. [Google Scholar] [CrossRef]
- Canonica, G.W.; Blasi, F.; Carpagnano, G.E.; Guida, G.; Heffler, E.; Paggiaro, P.; Allegrini, C.; Antonelli, A.; Aruanno, A.; Bacci, E.; Bagnasco, D.; Beghè, B.; Bonavia, M.; Bonini, M.; Brussino, L.; Caiaffa, M.F.; Calabrese, C.; Camiciottoli, G.; Caminati, M.; Caruso, C.; Cavallini, M.; Chieco Bianchi, F.; Conte, M.E.; Corsico, A.G.; Cosmi, L.; Costantino, M.; Costanzo, G.; Crivellaro, M.; D’Alò, S.; D’Amato, M.; Detoraki, A.; Di Proietto, M.C.; Facciolongo, N.C.; Ferri, S.; Fierro, V.; Foschino, M.P.; Latorre, M.; Lombardi, C.; Macchia, L.; Milanese, M.; Montagni, M.; Parazzini, E.M.; Parente, R.; Passalacqua, G.; Patella, V.; Pelaia, G.; Pini, L.; Puggioni, F.; Ricciardi, L.; Ridolo, E.; Rolo, J.; Scichilone, N.; Scioscia, G.; Senna, G.; Solidoro, P.; Varricchi, G.; Vianello, A.; Yacoub, M.R.; Yang, B. Severe Asthma Network Italy Definition of Clinical Remission in Severe Asthma: A Delphi Consensus. J Allergy Clin Immunol Pract. 2023, 11, 3629–3637. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, G.; Lu, H.; Li, H.; Tang, M.; Tong, A. Development of therapeutic antibodies for the treatment of diseases. Mol Biomed. 2022, 3, 35. [Google Scholar] [CrossRef]
- Ryman, J.T.; Meibohm, B. Pharmacokinetics of Monoclonal Antibodies. CPT Pharmacometrics Syst Pharmacol. 2017, 6, 576–588. [Google Scholar] [CrossRef] [PubMed]
- Matera, M.G.; Calzetta, L.; Rogliani, P.; Cazzola, M. Monoclonal antibodies for severe asthma: Pharmacokinetic profiles. Respir Med. 2019, 153, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Borrok MJ, Wu Y, Beyaz N, Yu XQ, Oganesyan V, Dall’Acqua WF, Tsui P. pH-dependent binding engineering reveals an FcRn affinity threshold that governs IgG recycling. J Biol Chem. 2015, 290, 4282–4290. [CrossRef] [PubMed]
- Gorzelak-Pabis, P.; Luczak, E.; Wojdan, K.; Antosik, K.; Borowiec, M.; Broncel, M.; Chalubinski, M. Endothelial integrity may be regulated by a specific antigen via an IgE-mediated mechanism. Postepy Hig Med Dosw (Online). 2017, 71, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Matucci A, Vivarelli E, Perlato M, Mecheri V, Accinno M, Cosmi L, Parronchi P, Rossi O, Vultaggio A. EGPA Phenotyping: Not Only ANCA, but Also Eosinophils. Biomedicines. 2023, 11, 776. [CrossRef]
- Maggi, L.; Rossettini, B.; Montaini, G.; Matucci, A.; Vultaggio, A.; Mazzoni, A.; Palterer, B.; Parronchi, P.; Maggi, E.; Liotta, F.; Annunziato, F.; Cosmi, L. Omalizumab dampens type 2 inflammation in a group of long-term treated asthma patients and detaches IgE from FcεRI. Eur J Immunol. 2018, 48, 2005–2014. [Google Scholar] [CrossRef] [PubMed]
- Harb, H.; Chatila, T.A. Mechanisms of Dupilumab. Clin Exp Allergy. 2020, 50, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Vatrella, A.; Busceti, M.T.; Gallelli, L.; Terracciano, R.; Savino, R.; Pelaia, G. Severe eosinophilic asthma: from the pathogenic role of interleukin-5 to the therapeutic action of mepolizumab. Drug Des Devel Ther. 2017, 11, 3137–3144. [Google Scholar] [CrossRef]
- Menzies-Gow, A.; Wechsler, M.E.; Brightling, C.E. Unmet need in severe, uncontrolled asthma: can anti-TSLP therapy with tezepelumab provide a valuable new treatment option? Respir Res. 2020, 21, 268. [Google Scholar] [CrossRef]
- Gauvreau, G.M.; Sehmi, R.; Ambrose, C.S.; Griffiths, J.M. Thymic stromal lymphopoietin: its role and potential as a therapeutic target in asthma. Expert Opin Ther Targets. 2020, 24, 777–792. [Google Scholar] [CrossRef]
- Takai, T. TSLP expression: cellular sources, triggers, and regulatory mechanisms. Allergol Int. 2012, 61, 3–17. [Google Scholar] [CrossRef]
- Dagher, R.; Kumar, V.; Copenhaver, A.M.; Gallagher, S.; Ghaedi, M.; Boyd, J.; Newbold, P.; Humbles, A.A.; Kolbeck, R. Novel mechanisms of action contributing to benralizumab’s potent anti-eosinophilic activity. Eur Respir J. 2022, 59, 2004306. [Google Scholar] [CrossRef] [PubMed]
- Ghazi, A.; Trikha, A.; Calhoun, W.J. Benralizumab--a humanized mAb to IL-5Rα with enhanced antibody-dependent cell-mediated cytotoxicity--a novel approach for the treatment of asthma. Expert Opin Biol Ther. 2012, 12, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Matucci, A.; Maggi, E.; Vultaggio, A. Eosinophils, the IL-5/IL-5Rα axis, and the biologic effects of benralizumab in severe asthma. Respir Med. 2019, 160, 105819. [Google Scholar] [CrossRef] [PubMed]
- Sehmi, R.; Lim, H.F.; Mukherjee, M.; Huang, C.; Radford, K.; Newbold, P.; Boulet, L.P.; Dorscheid, D.; Martin, J.G.; Nair, P. Benralizumab attenuates airway eosinophilia in prednisone-dependent asthma. J Allergy Clin Immunol. 2018, 141, 1529–1532. [Google Scholar] [CrossRef] [PubMed]
- Vultaggio, A.; Accinno, M.; Vivarelli, E.; Mecheri, V.; Maggiore, G.; Cosmi, L.; Parronchi, P.; Rossi, O.; Maggi, E.; Gallo, O.; Matucci, A. Blood CD62Llow inflammatory eosinophils are related to the severity of asthma and reduced by mepolizumab. Allergy. 2023, 78, 3154–3165. [Google Scholar] [CrossRef] [PubMed]
- Mesnil, C.; Raulier, S.; Paulissen, G.; Xiao, X.; Birrell, M.A.; Pirottin, D.; Janss, T.; Starkl, P.; Ramery, E.; Henket, M.; Schleich, F.N.; Radermecker, M.; Thielemans, K.; Gillet, L.; Thiry, M.; Belvisi, M.G.; Louis, R.; Desmet, C.; Marichal, T.; Bureau, F. Lung-resident eosinophils represent a distinct regulatory eosinophil subset. J Clin Invest. 2016, 126, 3279–3295. [Google Scholar] [CrossRef] [PubMed]
- Matucci, A.; Nencini, F.; Maggiore, G.; Chiccoli, F.; Accinno, M.; Vivarelli, E.; Bruno, C.; Locatello, L.G.; Palomba, A.; Nucci, E.; Mecheri, V.; Perlato, M.; Rossi, O.; Parronchi, P.; Maggi, E.; Gallo, O.; Vultaggio, A. High proportion of inflammatory CD62Llow eosinophils in blood and nasal polyps of severe asthma patients. Clin Exp Allergy. 2023, 53, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.E.; Knight, J.; Liu, Q.; Shelar, A.; Stewart, E.; Wang, X.; Yan, X.; Sanders, J.; Visness, C.; Gill, M.; Gruchalla, R.; Liu, A.H.; Kattan, M.; Khurana Hershey, G.K.; Togias, A.; Becker, P.M.; Altman, M.C.; Busse, W.W.; Jackson, D.J.; Montgomery, R.R.; Chupp, G.L. Activated sputum eosinophils associated with exacerbations in children on mepolizumab. J Allergy Clin Immunol. 2024. [CrossRef]
- www.ema.europa.eu/en/documents/product-information/fasenra-epar-product-information_en.pdf.
- Chia, Y.L.; Yan, L.; Yu, B.; Wang, B.; Barker, P.; Goldman, M.; Roskos, L. Relationship Between Benralizumab Exposure and Efficacy for Patients With Severe Eosinophilic Asthma. Clin Pharmacol Ther. 2019, 106, 383–390. [Google Scholar] [CrossRef]
- Martínez-Moragón E, García-Moguel I, Nuevo J, Resler G; ORBE study investigators. Real-world study in severe eosinophilic asthma patients refractory to anti-IL5 biological agents treated with benralizumab in Spain (ORBE study). BMC Pulm Med. 2021, 21, 417. [CrossRef]
- Padilla-Galo, A.; Moya Carmona, I.; Ausín, P.; Carazo Fernández, L.; García-Moguel, I.; Velasco-Garrido, J.L.; Andújar-Espinosa, R.; Casas-Maldonado, F.; Martínez-Moragón, E.; Martínez Rivera, C.; Vera Solsona, E.; Sánchez-Toril López, F.; Trisán Alonso, A.; Blanco Aparicio, M.; Valverde-Monge, M.; Valencia Azcona, B.; Palop Cervera, M.; Nuevo, J.; Sánchez Tena, J.; Resler, G.; Luzón, E.; Levy Naon, A. Achieving clinical outcomes with benralizumab in severe eosinophilic asthma patients in a real-world setting: orbe II study. Respir Res. 2023, 24, 235. [Google Scholar] [CrossRef]
- Vultaggio, A.; Aliani, M.; Altieri, E.; Bracciale, P.; Brussino, L.; Caiaffa, M.F.; Cameli, P.; Canonica, G.W.; Caruso, C.; Centanni, S.; D’Amato, M.; De Michele, F.; Del Giacco, S.; Di Marco, F.; Menzella, F.; Pelaia, G.; Rogliani, P.; Romagnoli, M.; Schino, P.; Senna, G.; Benci, M.; Boarino, S.; Schroeder, J.W. Long-term effectiveness of benralizumab in severe eosinophilic asthma patients treated for 96-weeks: data from the ANANKE study. Respir Res. 2023, 24, 135. [Google Scholar] [CrossRef]
- Liu, L.Y.; Sedgwick, J.B.; Bates, M.E.; Vrtis, R.F.; Gern, J.E.; Kita, H.; Jarjour, N.N.; Busse, W.W.; Kelly, E.A. Decreased expression of membrane IL-5 receptor alpha on human eosinophils: II. IL-5 down-modulates its receptor via a proteinase-mediated process. J Immunol. 2002, 169, 6459–6466. [Google Scholar] [CrossRef]
- Gregory, B.; Kirchem, A.; Phipps, S.; Gevaert, P.; Pridgeon, C.; Rankin, S.M.; Robinson, D.S. Differential regulation of human eosinophil IL-3, IL-5, and GM-CSF receptor alpha-chain expression by cytokines: IL-3, IL-5, and GM-CSF down-regulate IL-5 receptor alpha expression with loss of IL-5 responsiveness, but up-regulate IL-3 receptor alpha expression. J Immunol. 2003, 170, 5359–5366. [Google Scholar] [CrossRef]
- Matucci, A.; Vultaggio, A.; Danesi, R. The use of intravenous versus subcutaneous monoclonal antibodies in the treatment of severe asthma: a review. Respir Res. 2018, 19, 154. [Google Scholar] [CrossRef]
- Vultaggio, A.; Perlato, M.; Nencini, F.; Vivarelli, E.; Maggi, E.; Matucci, A. How to prevent and mitigate hypersensitivity reactions to biologicals induced by anti-drug antibodies? Front Immunol. 2021, 12, 765747. [Google Scholar] [CrossRef] [PubMed]
- Bergantini, L.; Pianigiani, T.; d’Alessandro, M.; Gangi, S.; Cekorja, B.; Bargagli, E.; Cameli, P. The effect of anti-IL5 monoclonal antibodies on regulatory and effector T cells in severe eosinophilic asthma. Biomed Pharmacother. 2023, 166, 115385. [Google Scholar] [CrossRef]
- Pini L, Bagnasco D, Beghè B, Braido F, Cameli P, Caminati M, Caruso C, Crimi C, Guarnieri G, Latorre M, Menzella F, Micheletto C, Vianello A, Visca D, Bondi B, El Masri Y, Giordani J, Mastrototaro A, Maule M, Pini A, Piras S, Zappa M, Senna G, Spanevello A, Paggiaro P, Blasi F, Canonica GW, On Behalf Of The Sani Study Group. Unlocking the Long-Term Effectiveness of Benralizumab in Severe Eosinophilic Asthma: A Three-Year Real-Life Study. J Clin Med. 2024, 13, 3013. [CrossRef]
- Pelaia, C.; Crimi, C.; Benfante, A.; Caiaffa, M.F.; Calabrese, C.; Carpagnano, G.E.; Ciotta, D.; D’Amato, M.; Macchia, L.; Nolasco, S.; Pelaia, G.; Pellegrino, S.; Scichilone, N.; Scioscia, G.; Spadaro, G.; Valenti, G.; Vatrella, A.; Crimi, N. Therapeutic Effects of Benralizumab Assessed in Patients with Severe Eosinophilic Asthma: Real-Life Evaluation Correlated with Allergic and Non-Allergic Phenotype Expression. J Asthma Allergy. 2021, 14, 163–173. [Google Scholar] [CrossRef]
- Lepretre, F.; Gras, D.; Chanez, P.; Duez, C. Natural killer cells in the lung: potential role in asthma and virus-induced exacerbation? Eur Respir Rev. 2023, 32, 230036. [Google Scholar] [CrossRef]
- Sabogal Piñeros, Y.S.; Bal, S.M.; Dijkhuis, A.; Majoor, C.J.; Dierdorp, B.S.; Dekker, T.; Hoefsmit, E.P.; Bonta, P.I.; Picavet, D.; van der Wel, N.N.; Koenderman, L.; Sterk, P.J.; Ravanetti, L.; Lutter, R. Eosinophils capture viruses, a capacity that is defective in asthma. Allergy. 2019, 74, 1898–1909. [Google Scholar] [CrossRef] [PubMed]
- Murrison, L.B.; Brandt, E.B.; Myers, J.B.; Hershey, G.K.K. Environmental exposures and mechanisms in allergy and asthma development. J Clin Invest. 2019, 129, 1504–1515. [Google Scholar] [CrossRef] [PubMed]
- Vantaggiato, L.; Cameli, P.; Bergantini, L.; d’Alessandro, M.; Shaba, E.; Carleo, A.; Di Giuseppe, F.; Angelucci, S.; Sebastiani, G.; Dotta, F.; Bini, L.; Bargagli, E.; Landi, C. Serum Proteomic Profile of Asthmatic Patients after Six Months of Benralizumab and Mepolizumab Treatment. Biomedicines. 2022, 10, 761. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.M. Clinical Pharmacology of Corticosteroids. Respir Care. 2018, 63, 655–670. [Google Scholar] [CrossRef] [PubMed]
- Bourdin, A.; Husereau, D.; Molinari, N.; Golam, S.; Siddiqui, M.K.; Lindner, L.; Xu, X. Matching-adjusted comparison of oral corticosteroid reduction in asthma: Systematic review of biologics. Clin Exp Allergy. 2020, 50, 442–452. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lepretre, F.; Gras, D.; Chanez, P.; Duez, C. Natural killer cells in the lung: potential role in asthma and virus-induced exacerbation? Eur Respir Rev. 2023, 32, 230036. [Google Scholar] [CrossRef]
- Gutiérrez-Vera, C.; García-Betancourt, R.; Palacios, P.A.; Müller, M.; Montero, D.A.; Verdugo, C.; Ortiz, F.; Simon, F.; Kalergis, A.M.; González, P.A.; Saavedra-Avila, N.A.; Porcelli, S.A.; Carreño, L.J. Natural killer T cells in allergic asthma: implications for the development of novel immunotherapeutical strategies. Front Immunol. 2024, 15, 1364774. [Google Scholar] [CrossRef]
- Björkström, N.K.; Strunz, B.; Ljunggren, H.G. Natural killer cells in antiviral immunity. Nat Rev Immunol. 2022, 22, 112–123. [Google Scholar] [CrossRef]
- Waggoner, S.N.; Reighard, S.D.; Gyurova, I.E.; Cranert, S.A.; Mahl, S.E.; Karmele, E.P.; McNally, J.P.; Moran, M.T.; Brooks, T.R.; Yaqoob, F.; Rydyznski, C.E. Roles of natural killer cells in antiviral immunity. Curr Opin Virol. 2016, 16, 15–23. [Google Scholar] [CrossRef]
- Caminati, M.; Marcon, A.; Guarnieri, G.; Miotti, J.; Bagnasco, D.; Carpagnano, G.E.; Pelaia, G.; Vaia, R.; Maule, M.; Vianello, A.; Senna, G. Benralizumab Efficacy in Late Non-Responders to Mepolizumab and Variables Associated with Occurrence of Switching: A Real-Word Perspective. J Clin Med. 2023, 12, 1836. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



