Submitted:
08 June 2024
Posted:
11 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
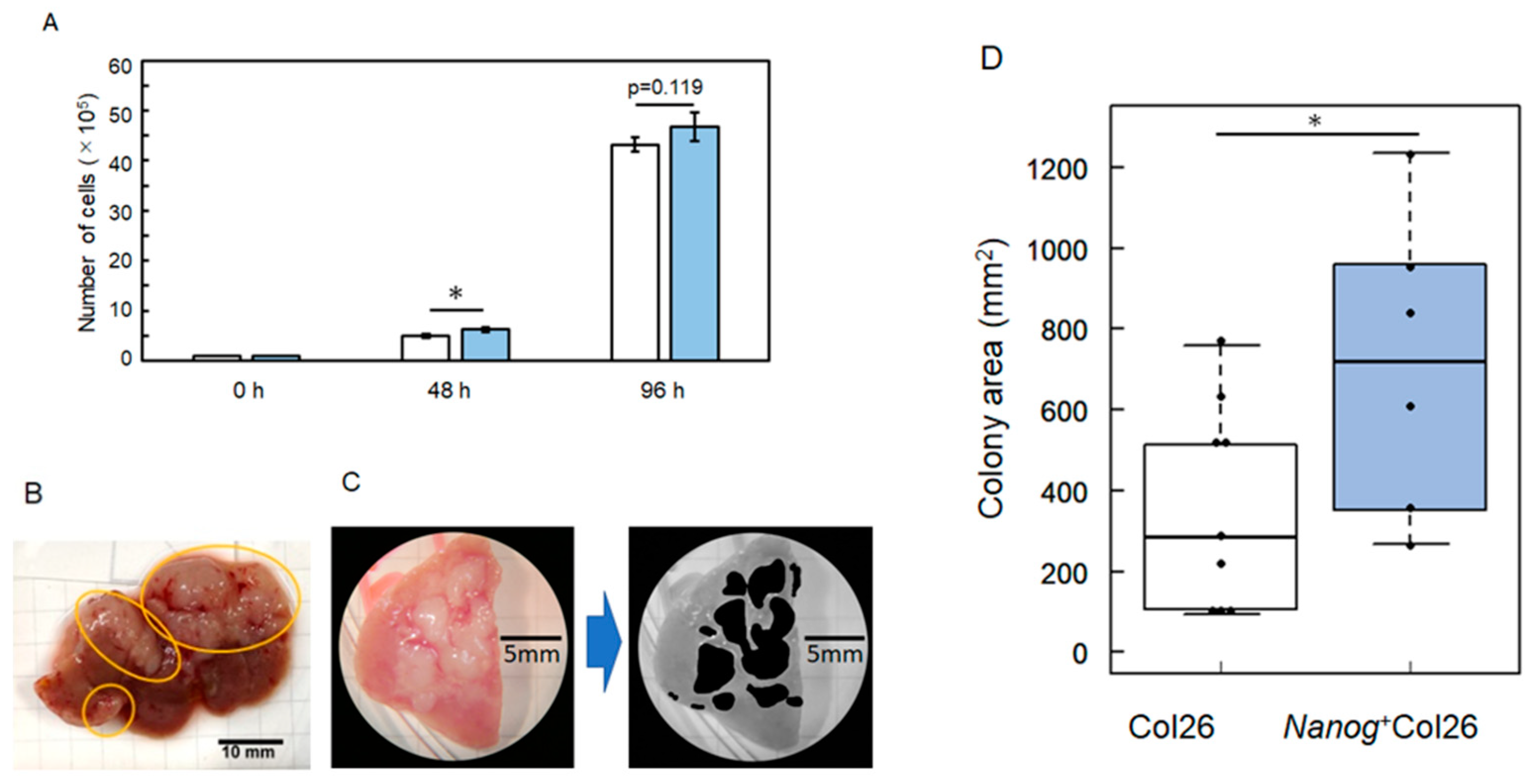
2.1. Properties of Nanog-Overexpressing Colon-26 (Nanog+Colon26) Cells
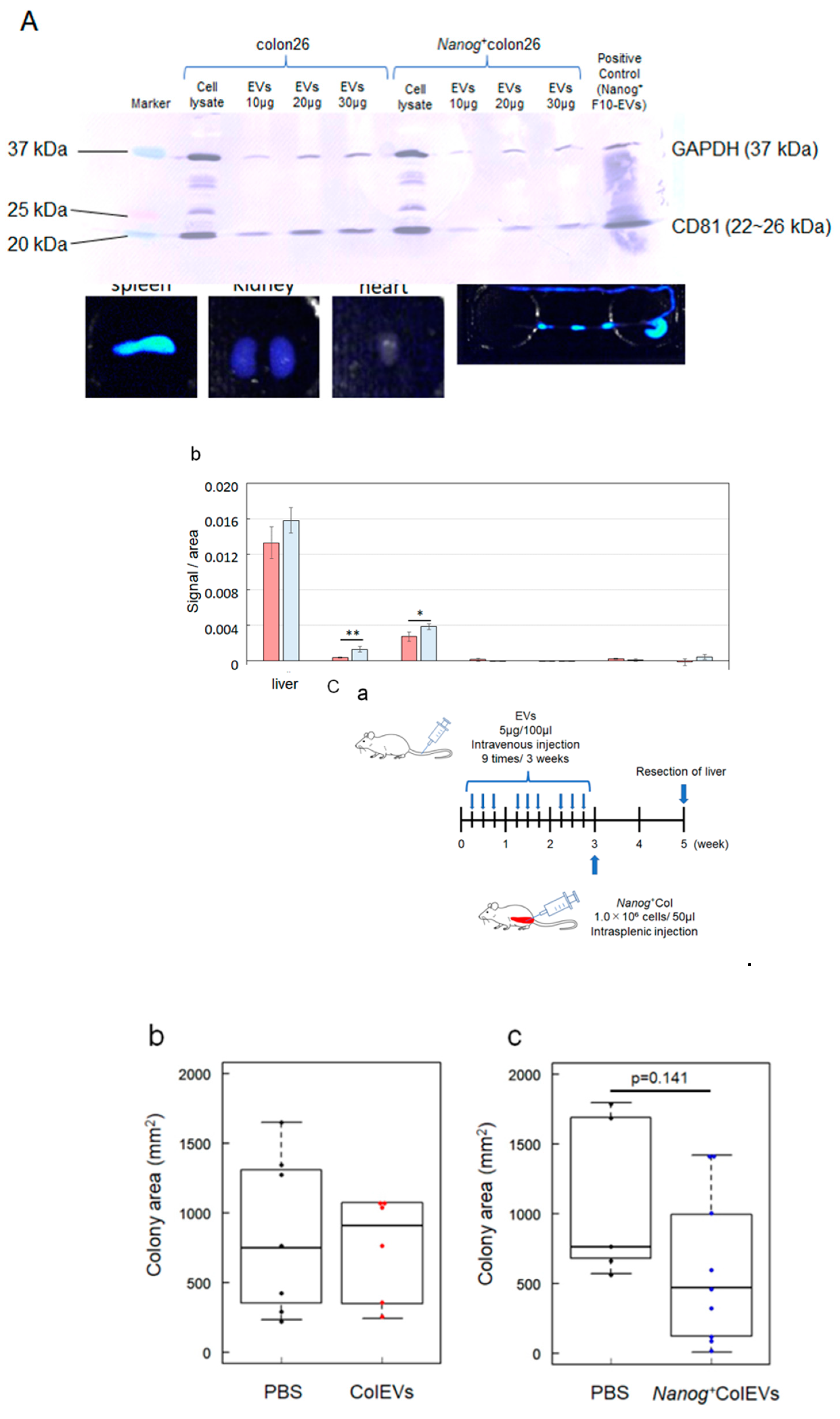
2.2. Metastasis Suppression Effect of Nanog+colon26EVs
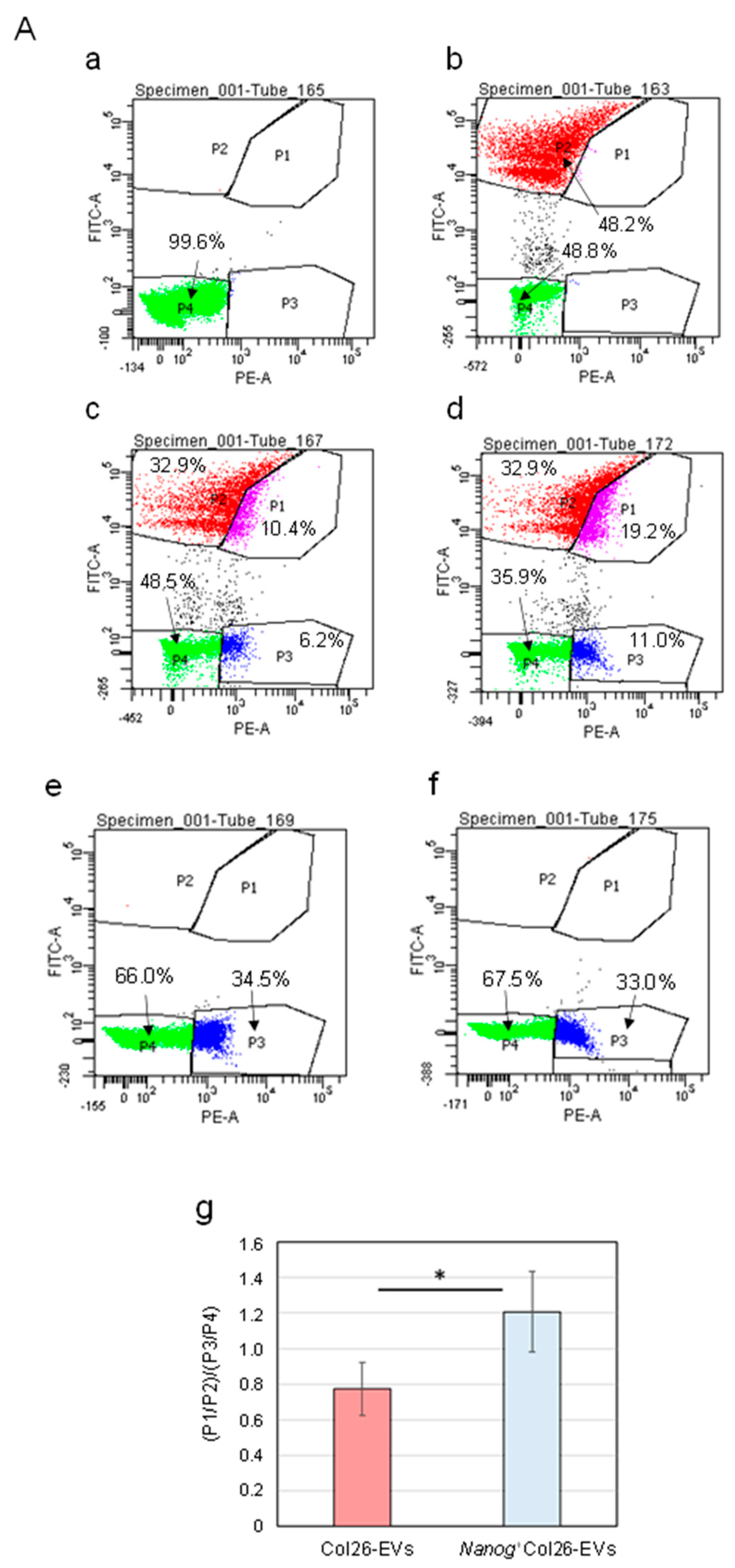
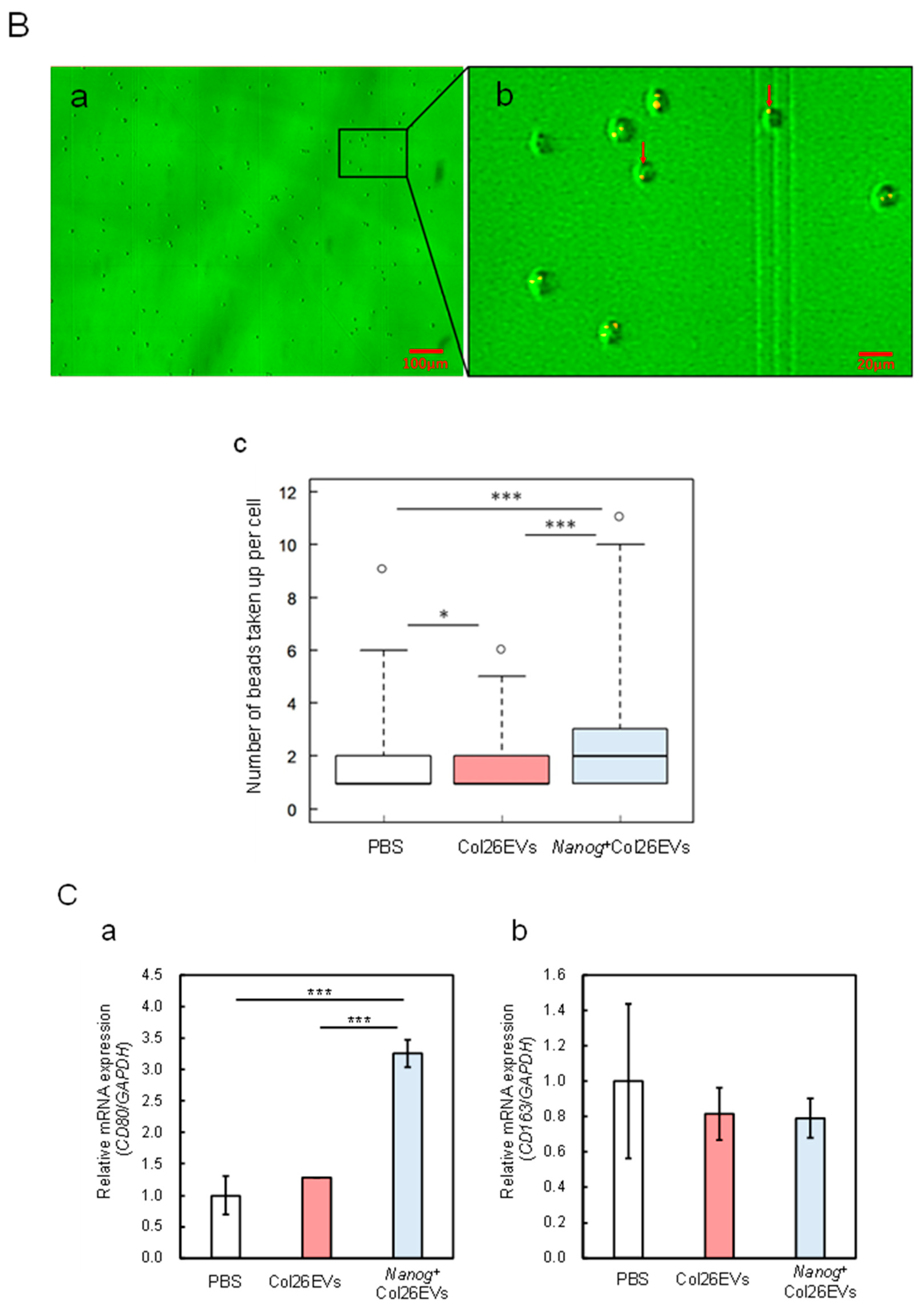
2.3. Effects of Nanog+colon26EVs on Macrophage Phagocytic Function
2.4. Effects on Macrophage Polarization
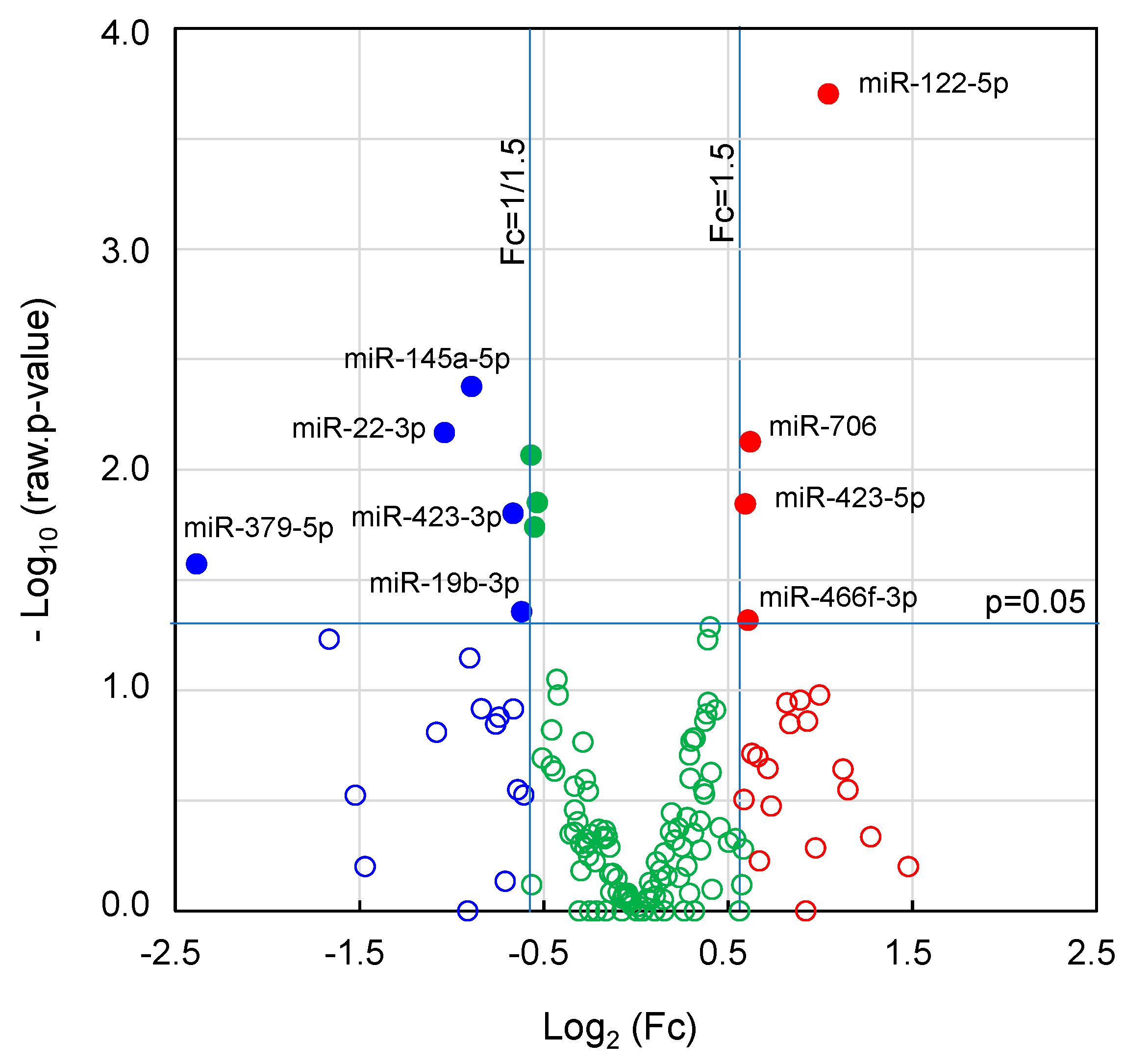
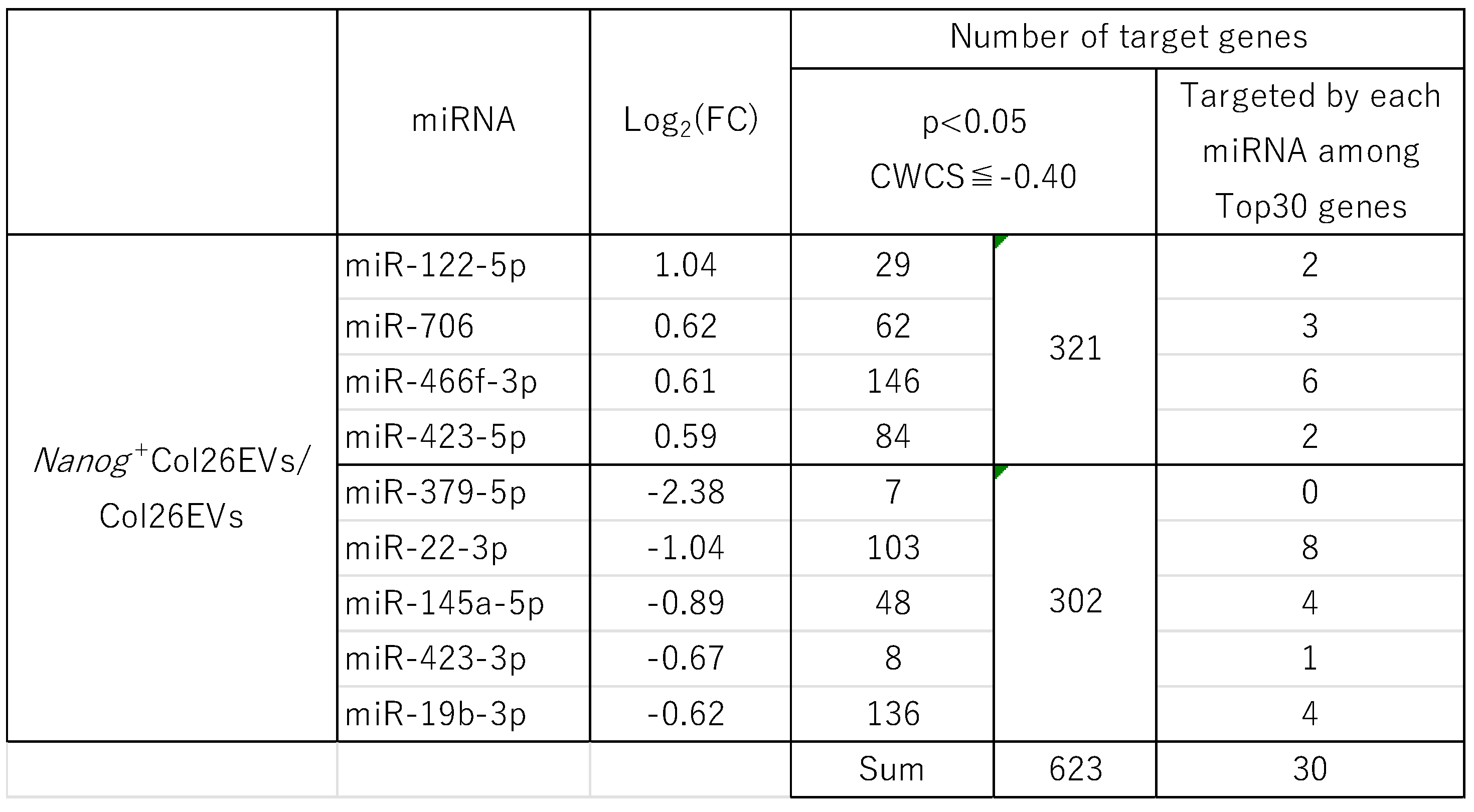
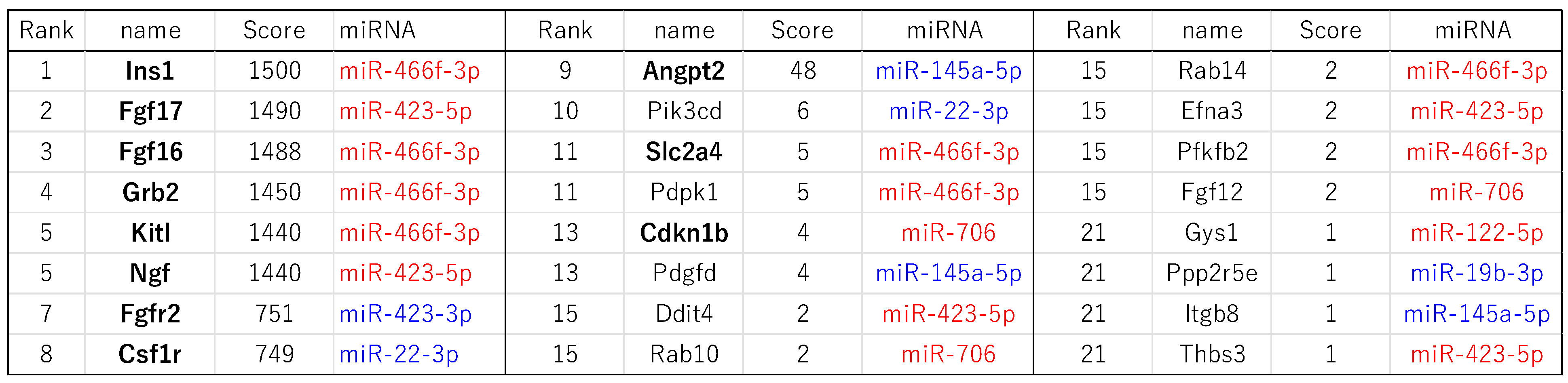
2.5. Expression of miRNAs and Their Target Genes in EVs
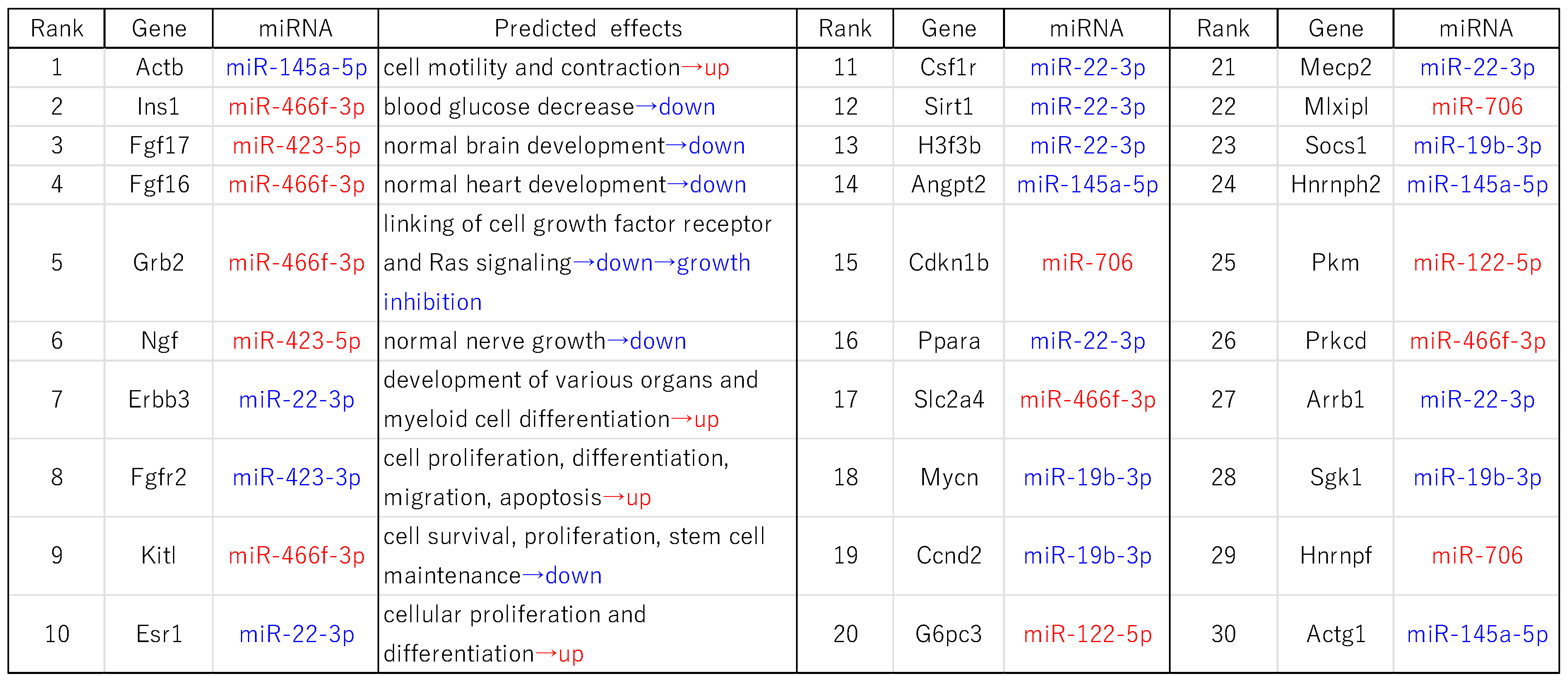
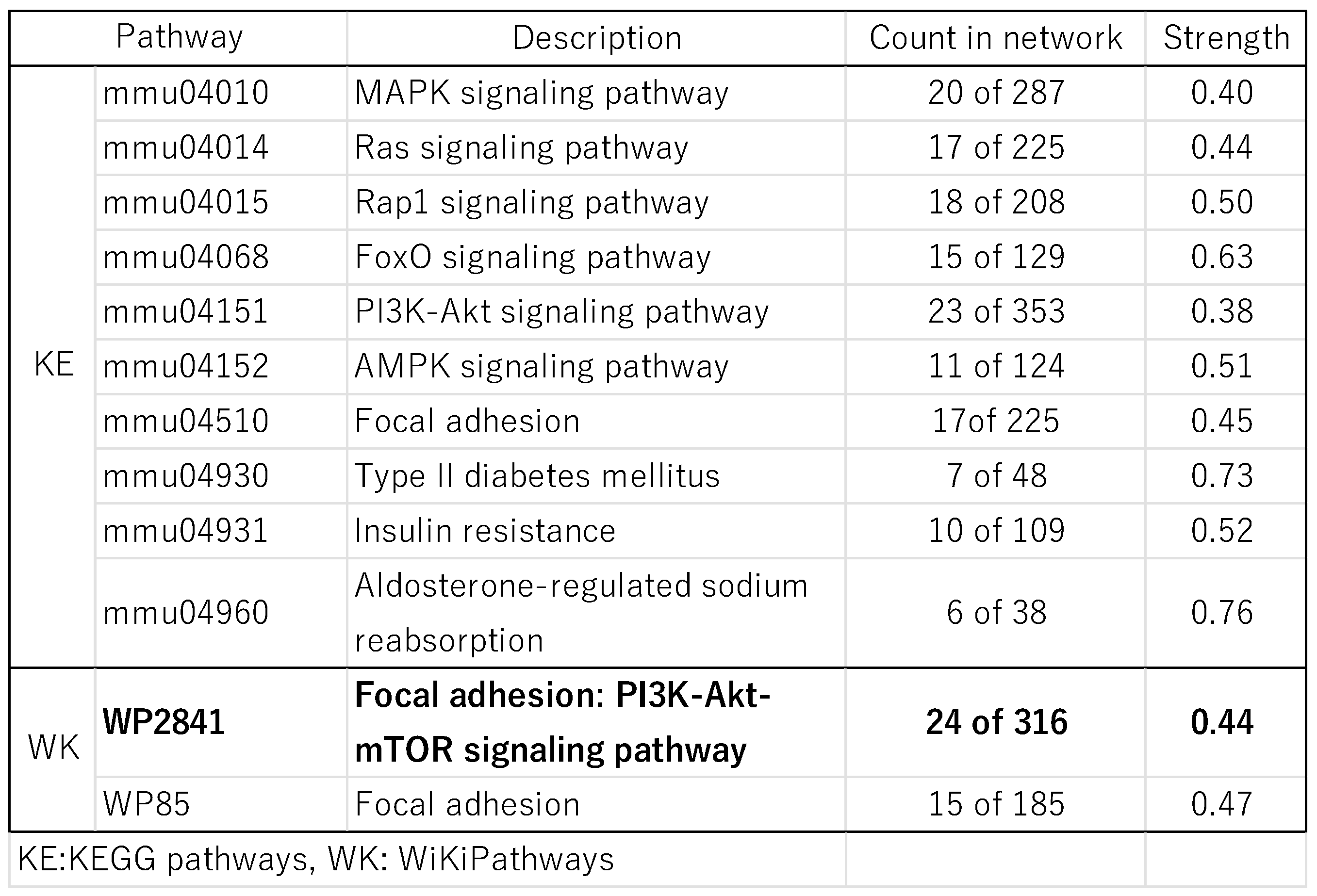
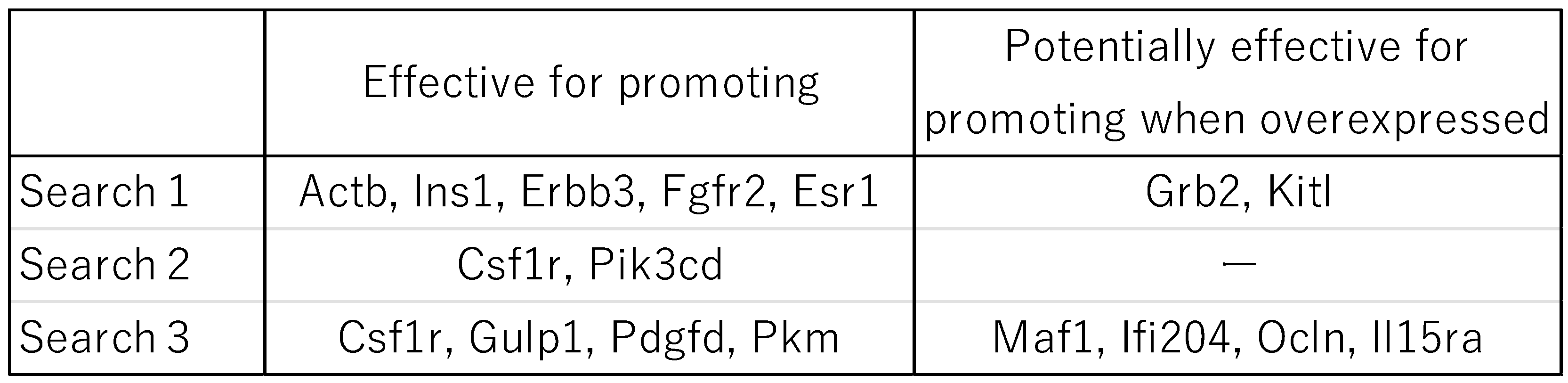
2.6. Factors Involved in the Activation of the Immune System
Results of the First Search
Results of the Second Search
Results of the Third Search
3. Discussion
4. Material and Methods
4.1. Cell Culture
4.2. Animals
4.3. Generation of a Nanog-Overexpressing Colon Cancer Cell Line
4.4. Quantification of Colon Metastasis to Liver
4.5. Preparation of EVs
4.6. Western Analysis of CD81 and Gapdh
4.7. Analysis of the Transfer of EVs to Various Organs in a Mouse
4.8. In Vivo Test of the Effects of EVs on the Metastasis
4.9. Phagocytic Activity Test
4.10. Analysis of miRNAs and Their Target Genes
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kajiyama,H.; Shibata, K.; Terauchi, M.; Yamashita, M.; Ino, K.; Nawa, A.; Kikkawa, Chemoresistance to paclitaxel induces epithelial-mesenchymal transition and enhances metastatic potential for epithelial ovarian carcinoma cells. F. Int. J. Oncol. 2007, 31, 277-283. [CrossRef]
- Wattanawongdon, W.; Hahnvajanawong, C.; Namwat, N.; Kanchanawat, S.; Boonmars, T.; Jearanaikoon, P.; Leelayuwat, C.; Techasen, A.; Seubwai, W. Establishment and characterization of gemcitabine-resistant human cholangiocarcinoma cell lines with multidrug resistance and enhanced invasiveness. Int. J. Oncol. 2015, 47, 398-410. [CrossRef]
- Cao, W.; Wei, W.; Zhan, Z.; Xie, D.; Xie, Y.; Xiao, Q. Regulation of drug resistance and metastasis of gastric cancer cells via the microRNA647-ANK2 axis. Int. J. Mol. Med. 2018, 41, 1958-1966. [CrossRef]
- Speers, C.; Zhao, S.; Liu, M.; Bartelink, H.; Pierce, L.J.; Feng, F.Y. Development and validation of a novel radiosensitivity signature in human breast cancer. Clin. Cancer Res. 2015, 21, 3667-3677. [CrossRef]
- Carlos-Reyes, A.; Muñiz-Lino, M.A.; Romero-Garcia, S.; López-Camarillo, C.; Hernández-de la Cruz, O.N. Biological adaptations of tumor cells to radiation therapy. Front. Oncol. 2021, 11, 718636. [CrossRef]
- Gurung, S.; Perocheau, D.;T ouramanidou, L.; Baruteau, J. The exosome journey: from biogenesis to uptake and intracellular signaling. Cell Commun. Signal. 2021, 19, 47. [CrossRef]
- Mitchell, M.I.; , Ma, J.; Carter, C.L.; Loudig, O. Circulating exosome cargoes contain functionally diverse cancer biomarkers: from biogenesis and function to purification and potential translational utility. Cancers 2022, 14, 3350. [CrossRef]
- Kar, R.; Dhar, R.; Mukherjee, S.; Nag, S.; Gorai, S.; Mukerjee, N.; Mukherjee, R. Vatsa, R.; Jadhav, M.C.; Ghosh, A.; Devi, A.; Krishnan, A.; Thorat, N.D. Exosome-based smart drug delivery tool for cancer theranostics. ACS Biomater. Sci. Eng. 2023, 9, 577-594. https://pubs.acs.org/action/showCitFormats?doi=10.1021/acsbiomaterials.2c01329&ref=pdf. [CrossRef]
- Santos, P.; Almeida, F. Exosome-based vaccines: history, current state, and clinical trials. Front. Immunol. 2021, 12, 711565. [CrossRef]
- Fernández-Delgado, I.; Calzada-Fraile, D.; Sánchez-Madrid, S. Immune regulation by dendritic cell extracellular vesicles in cancer immunotherapy and vaccines. Cancers 2020, 12, 3558. [CrossRef]
- Wang, C.; Huang, X.; Wu, Y.; Wang, J.; Li, F.; Guo, G. Tumor cell-associated exosomes robustly elicit anti-tumor immune responses through modulating dendritic cell vaccines in lung tumor. Int. J. Biol. Sci. 2020, 16(4), 633-643. [CrossRef]
- Elashiry, M.; Elsayed, R.; Cutler, C.W. Exogenous and endogenous dendritic cell-derived exosomes: lessons learned for immunotherapy and disease pathogenesis. Cells 2022, 11, 115. [CrossRef]
- van Neerven, S.M.; Tieken, M.; Vermeulen, L.; Bijlsma, M.F. Bidirectional interconversion of stem and nonstem cancer cell populations: A reassessment of theoretical models for tumor heterogeneity. Mol. Cell. Oncol. 2016, 3, 2, e1098791 . [CrossRef]
- Chen, C.L.; Kumar, D.B.U.; Punj, V.; Xu, J.; Sher, L.; Tahara, S.M.; Hess, S.; Machida, K. NANOG metabolically reprograms tumor-initiating stem-like cells through tumorigenic changes in oxidative phosphorylation and fatty acid metabolism. Cell Metab. 2016, 23, 206–219. [CrossRef]
- Gawlik-Rzemieniewska, N.; Bednarek, I. The role of NANOG transcriptional factor in the development of malignant phenotype of cancer cells. Cancer Biol. Ther. 2016, 17, 1–10. [CrossRef]
- Saito, M. Novel roles of Nanog in cancer cells and their extracellular vesicles. Cells 2022, 11, 3881. [CrossRef]
- Saito, M.; Kishi, R.; Sasai, T.; Hatakenaka, T.; Matsuki, N.; Minagawa, S. Effect of Nanog overexpression on the metastatic potential of a mouse melanoma cell line B16-BL6. Mol. Cell. Biochem. 2021, 476, 2651-2661.
- Hatakenaka, T.; Matsuki, N.; Minagawa, S.; May, C.K.S.; Saito, M., Anti-metastatic function of extracellular vesicles derived from Nanog-overexpressing melanoma. Curr. Oncol. 2022, 29, 1029-1046. [CrossRef]
- Miyazaki, T.; Chung, S.; Sakai, H.; Ohata, H.; Obata, Y.; Shiokawa, D.; Mizoguchi, Y.; Kubo, T.; Ichikawa, H.; Taniguchi, H.; Aoki, K., Soga, T.; Nakagama, H.; Okamoto, K. Stemness and immune evasion conferred by the TDO2-AHR pathway are associated with liver metastasis of colon cancer. Cancer Science 2022, 113, 170–181. [CrossRef]
- Descarpentrie, J.; Araúzo-Bravo, M.J.; He, Z.; François, A.; González, Á.; Garcia-Gallastegi, P.; Badiola, I.; Evrard, S.; Pernot, S.; Creemers, J.W.M.; Abdel-Majid, K. Role of furin in colon cancer stem cells malignant phenotype and expression of LGR5 and NANOG in KRAS and BRAF-mutated colon tumors. Cancers 2022, 14, 1195. [CrossRef]
- Tarique, A.A.; Logan, J.; Thomas, E.; Holt, P.; Sly, P.D.; Fantino, E. Phenotypic, functional, and plasticity features of classical and alternatively activated human macrophages, Am. J. Respir. Cell Mol. Biol., 2015, 53, 676-688. [CrossRef]
- Xiang, W.; Shi, R., Zhang, D.; Kang, X.; Zhang, L.; Yuan, J.; Zhang, X.; Miao, H. Dietary fats suppress the peritoneal seeding of colorectal cancer cells through the TLR4/Cxcl10 axis in adipose tissue macrophages. Signal Transduct. Targeted Ther. 2020, 5, 239. [CrossRef]
- Khoo, C.S.M.; Hatakenaka, T.; Matsuki, N.; Minagawa, S.; Asami, K.; Henmi, T.; Morimoto, A.; Saito, M. Moderate hyperglycemia suppresses melanoma metastasis to the liver. Exp. Anim. 2023, 72(2), 183-192. [CrossRef]
- Deng, W.; Zhu, S.; Zeng, L.; Liu, J.; Kang, R.; Yang, M.; Cao, L.; Wang, H.; Billiar, T.R.; Jiang, J.; Xie, M.; Tang, D. The circadian clock controls immune checkpoint pathway in sepsis. Cell Rep. 2018, 24(2), 366–378. [CrossRef]
- Khoo, C.S.M.; Henmi, T.; Saito, M. Comparative study of metastasis suppression effects of extracellular vesicles derived from anaplastic cell lines, Nanog-overexpressing melanoma, and induced pluripotent stem cells. Int. J. Mol. Sci. 2023, 24, 17206.

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




