Submitted:
06 June 2024
Posted:
10 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Biosecurity Standards Used
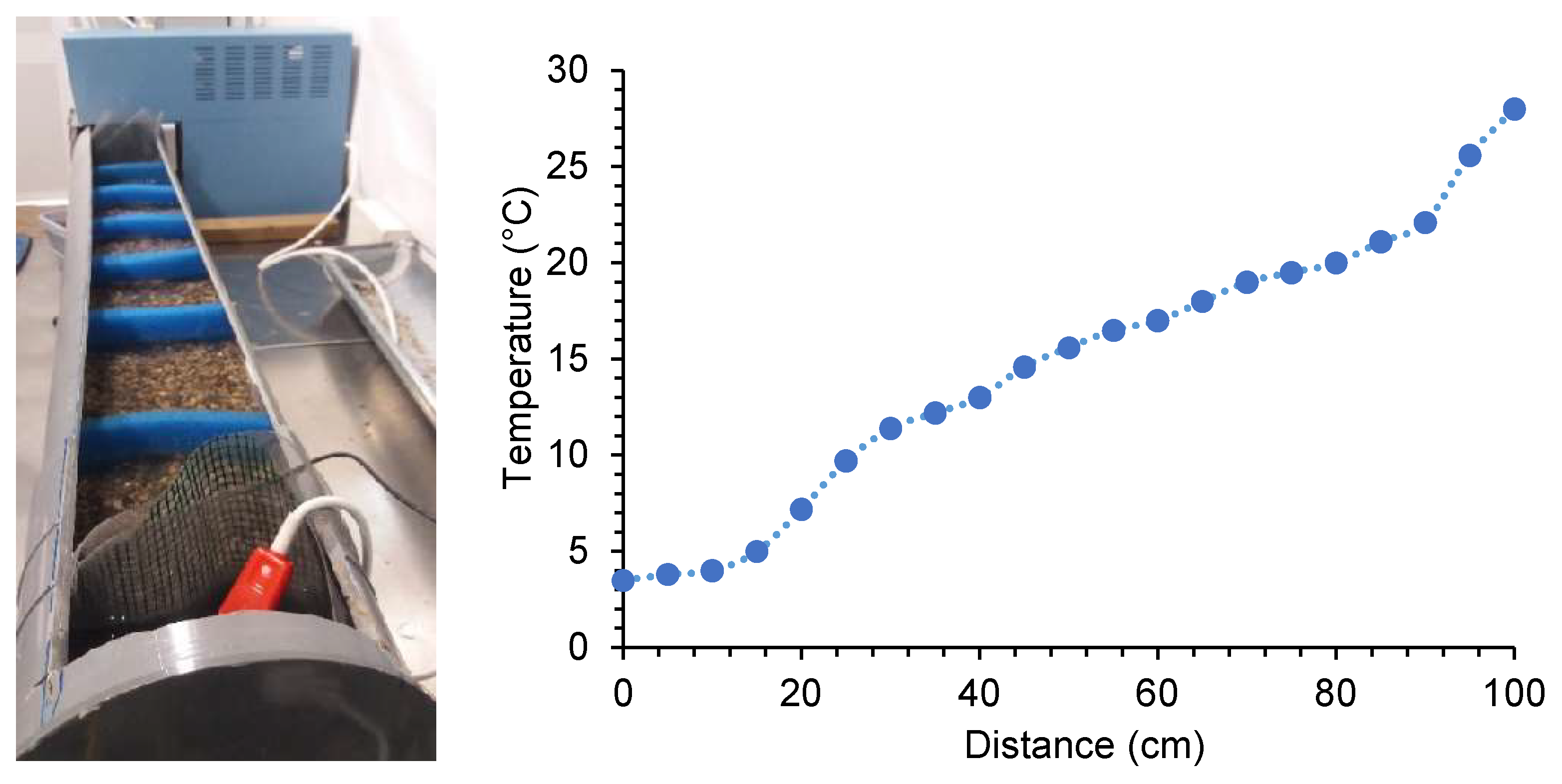
2.2. Experimental Procedure
2.2.1. Critical Thermal Maximum (CTmax)
2.2.2. Cold Tolerance
2.2.3. Preferred Temperature (Tp)
2.3. Statistical Analysis
3. Results
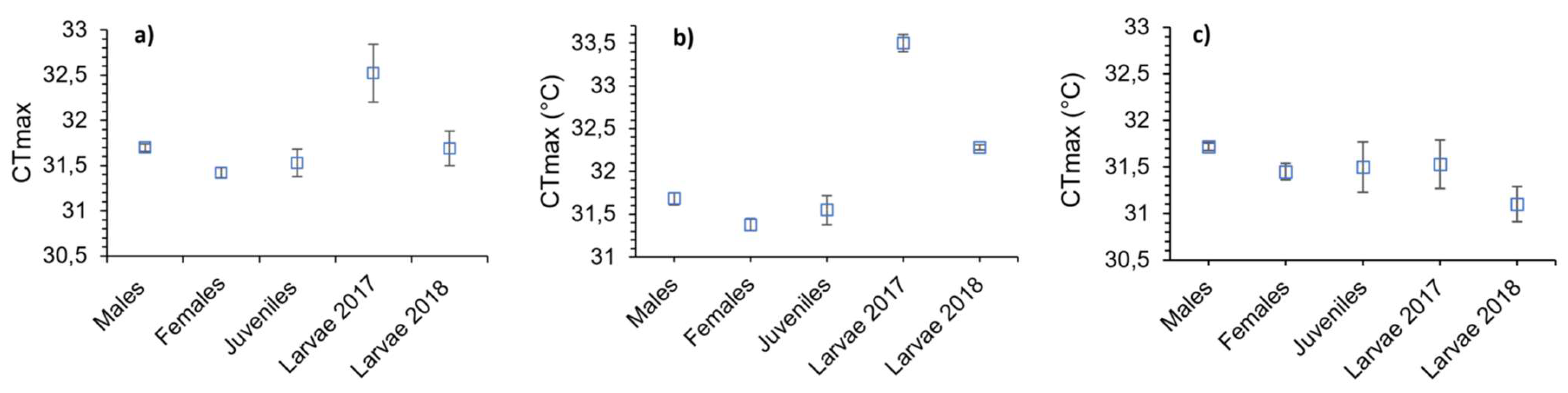
3.1. Critical Thermal Maximum (CTmax)
3.2. Cold Tolerance
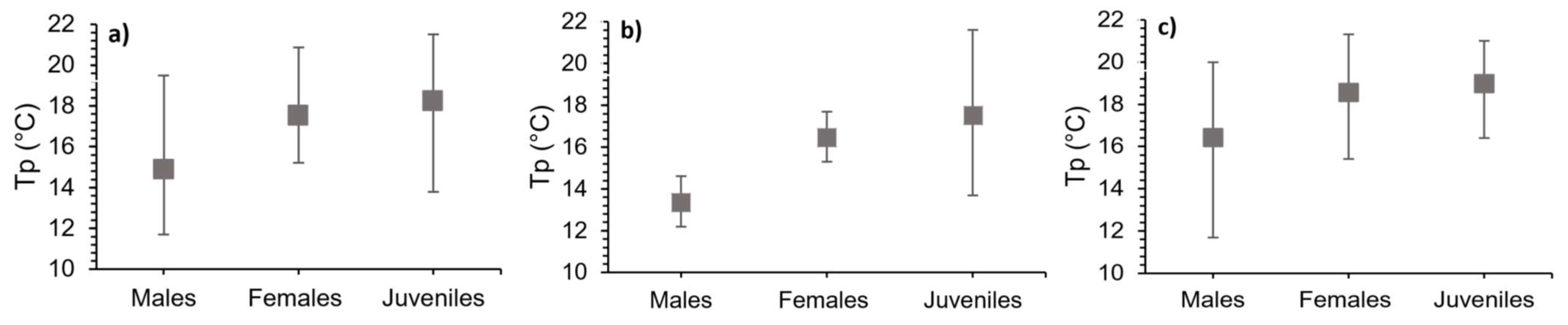
3.3. Preferred Temperature (Thermopreferendum–Tp)
4. Discussion
4.1. Thermal Tolerance of Calotriton arnoldi Species
4.2. Preferred Body Temperature Range (Tp) of Montseny Newt in the Laboratory
4.3. Ecological Implications and Management Measures
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- IPCC. Global Warming of 1.5°C. An IPCC Special Report on the impacts of global warming of 1.5°C above pre-industrial levels and related global greenhouse gas emission pathways, in the context of strengthening the global response to the threat of climate change, sustainable development, and efforts to eradicate poverty. [asson-Delmotte, V., P. Zhai, H.-O. Pörtner, D. Roberts, J. Skea, P.R. Shukla, A. Pirani, W. Moufouma-Okia, C. Péan, R. Pidcock, S. Connors, J.B.R. Matthews, Y. Chen, X. Zhou, M.I. Gomis, E. Lonnoy, T. Maycock, M. Tignor, and T. Waterfield (eds.). Cambridge University Press: Cambridge, UK; New York, NY, USA, p. 616. [CrossRef]
- Morgan, R.; Finnøen, M.H.; Jensen, H.; Pélabon, C.; Jutfelt, F. Low potential for evolutionary rescue from climate change in a tropical fish. Proc. Natl. Acad. Sci. USA 2020, 117, 33365–33372. [Google Scholar] [CrossRef] [PubMed]
- Malhi, Y.; Franklin, J.; Seddon, N.; Solan, M.; Turner, M.G.; Field, C.B.; Knowlton, N. Climate change and ecosystems: threats, opportunities and solutions. Phil. Trans. R. Soc. B 2020, 375, 20190104. [Google Scholar] [CrossRef]
- Tejedo, M.; Duarte, H.; Gutiérrez-Pesquera, L.M.; Beltrán, J.F.; Katzenberger, M.; Marangoni, F.; Navas, C.A. Nicieza, A.C.; Reylea, R.A.; Rezende, E.L.; et al. El estudio de las tolerancias térmicas para el examen de hipótesis biogeográficas y de la vulnerabilidad de los organismos ante el calentamiento global. Ejemplos en anfíbios. Bol. Asoc. Herpetol. Esp.
- Parmesan, C.; Morecroft, M.D.; Trisurat, Y. Climate Change 2022: Impacts, Adaptation and Vulnerability. [Research Report] GIEC. 2022. ffhal-03774939f. https://hal.science/hal-03774939 .
- Parmesan, C. Ecological and Evolutionary Responses to Recent Climate Change. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 637–669. [Google Scholar] [CrossRef]
- Parmesan, C.; Hanley, M. Plants and climate change: complexities and surprises. Ann. Bot. 2015, 16, 849–864. [Google Scholar] [CrossRef]
- Parmesan, C.; Singer, M.C. Mosaics of climatic stress across species' ranges: tradeoffs cause adaptive evolution to limits of climatic tolerance. Philosophical Transactions of the Royal Society B: Biological Sciences 2021, 377, 20210003. [Google Scholar] [CrossRef]
- Enriquez-Urzelai, U.; Bernardo, N.; Moreno-Rueda, G.; Montori, A.; Llorente, G. Are amphibians tracking their climatic niches in response to climate warming? A test with Iberian amphibians. Clim. Change 2019, 154, 289–301. [Google Scholar] [CrossRef]
- Brown, C.J.; O'Connor, M.I.; Poloczanska, E.S.; Schoeman, D.S.; Buckley, L.B.; Burrows, M.T.; Duarte, C.M.; Halpern, B.S.; Pandolfi, J.M.; Parmesan, C.; et al. Ecological and methodological drivers of species’ distribution and phenology responses to climate change. Glob. Change Biol. 2016, 22, 1548–1560. [Google Scholar] [CrossRef] [PubMed]
- Dillon, M.E.; Wang, G.; Huey, R.B. Global metabolic impacts of recent climate warming. Nature 2010, 467, 704–706. [Google Scholar] [CrossRef]
- Paaijmans, K.P.; Heinig, R.L.; Seliga, R.A.; Blanford, J.I.; Blanford, S.; Murdock, C.C.; Thomas, M.B. Temperature variation makes ectotherms more sensitive to climate change. Glob. Change Biol. 2013, 19, 2373–2380. [Google Scholar] [CrossRef]
- Spotila, J.R. Role of Temperature and Water in the Ecology of Lungless Salamanders. Ecological Monographs 1972, 42, 95–125. [Google Scholar] [CrossRef]
- Gvoždík, L. Does reproduction influence temperature preferences in newts? Canadian Journal of Zoology 2005, 83, 1038–1044. [Google Scholar] [CrossRef]
- Gvozdik, L. Plasticity of preferred body temperatures as means of coping with climate change? Biology Letters 2012, 8, 262–265. [Google Scholar] [CrossRef]
- Gvoždík, L.; Kristín, P. Economic thermoregulatory response explains mismatch between thermal physiology and behaviour in newts. J. Exp. Biol. 2017, 220, 1106–1111. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, J.; Arntzen, J. Potential impact of climate warming on the distribution of the Golden-striped salamander, Chioglossa lusitanica, on the Iberian Peninsula. Biodivers. Conserv. 2002, 11, 2167–2176. [Google Scholar] [CrossRef]
- Moreno-Rueda, G.; Pleguezuelos, J.M.; Pizarro, M.; Montori, A. Northward Shifts of the Distributions of Spanish Reptiles in Association with Climate Change. Conserv. Biol. 2012, 26, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, V.H. Critical thermal maximum in salamanders. Physiol. Zool. 1961, 34, 92–125. [Google Scholar] [CrossRef]
- Kearney, M.; Shine, R.; Porter, W.P. The potential for behavioural thermoregulation to buffer ‘cold-blooded’ animals against climate warming. Proc. Natl. Acad. Sci. USA 2009, 106, 3835–3840. [Google Scholar] [CrossRef] [PubMed]
- Angilletta, M.J.; Niewiarowski, P.H.; Navas, C.A. The evolution of thermal physiology in ectotherms. J. Therm. Biol. 2002, 27, 249–268. [Google Scholar] [CrossRef]
- Montori, A.; Amat, F. Surviving on the edge: present and future effects of climate warming on the common frog (Rana temporaria) population in the Montseny massif (NE Iberia). PeerJ 2023, 11, e14527. [Google Scholar] [CrossRef]
- IUCN. The IUCN Red List of Threatened Species. Version 2023-1. 2023. Available online: https://www.iucnredlist.org (accessed on 12 May 2024).
- Carranza, S.; Amat, F. Taxonomy, biogeography and evolution of Euproctus (Amphibia: Salamandridae), with the resurrection of the genus Calotriton and the description of a new endemic species from the Iberian Peninsula. Zool. J. Linn. Soc. 2005, 145, 555–582. [Google Scholar] [CrossRef]
- Valbuena-Ureña, E.; Amat, F.; Carranza, S. Integrative phylogeography of Calotriton newts (Amphibia, Salamandridae), with special remarks on the conservation of the endangered Montseny brook newt (Calotriton arnoldi). PLoS ONE 2013, 8, e62542. [Google Scholar] [CrossRef]
- Guinart, D.; Solórzano, S.; Amat, F.; Grau, J.; Fernández-Guiberteau, D.; Montori, A. Habitat Management of the Endemic and Critical Endangered Montseny Brook Newt (Calotriton arnoldi). Land 2022, 11, 449. [Google Scholar] [CrossRef]
- Talavera, A.; Palmada-Flores, M.; Burriel-Carranza, B.; Valbuena-Ureña, E.; Mochales-Riaño, G.; Adams, D.C.; Tejero-Cicuéndez, H.; Soler-Membrives, A.; Amat, F.; Guinart, D.; et al. Genomic insights into the Montseny brook newt (Calotriton arnoldi), a Critically Endangered glacial relict. iScience 2024, 27, 108665. [Google Scholar] [CrossRef] [PubMed]
- Peñuelas, J.; Boada, M. A global change-induced biome shift in the Montseny mountains (NE Spain). Glob. Change Biol. 2003, 9, 131–140. [Google Scholar] [CrossRef]
- Layne, J.R.J.; Claussen, D. The time courses of CTmax and CTmin acclimation in the salamander Desmognathus fuscus. J. Therm. Biol. 1982, 7, 139–141. [Google Scholar] [CrossRef]
- Šamajová, P.; Gvoždík, L. The influence of temperature on diving behaviour in the alpine newt, Triturus alpestris. J. Therm. Biol. 2009, 34, 401–405. [Google Scholar] [CrossRef]
- Trochet, A.; Dupoué, A.; Souchet, J.; Bertrand, R.; Deluen, M.; Murarasu, S.; Calvez, O.; Martinez-Silvestre, A.; Verdaguer-Foz, I.; Darnet, E.; et al. Variation of preferred body temperatures along an altitudinal gradient: a multi-species study. J. Therm. Biol. 2018, S0306456518301736. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Guiberteau, D.; Montori, A.; Pérez-Sorribes, L.; Carranza, S. Protocols Sanitaris per a les Activitats que Impliquin la Interacció Directa o Indirecta amb les Poblacions D’amfibis al Medi Natural; LIFETM Docs: Barcelona, Spain, 2020; 15p, Available online: https://lifetritomontseny.eu/documents/107349215/342993610/Bones_PractiquesWEB.pdf/11a7c63b-cd53-0e37-0cf5-737c18daded8?t=1602242474228 (accessed on 24 January 2023).
- Martínez-Silvestre, A.; Graciá, E.; Giménez, A.; Cadenas, V.; García de la Fuente, M.I.; Thumsová, B.; Bosch, J.; Bioseguridad en trabajos de campo con reptiles y anfibios. Bol. Asoc. Herpetol. Esp. 2023, 34, 3–16. Available online: http://www.herpetologica.org/BAHE/34/BAHE34[I_1158].pdf (accessed on 23 June 2024).
- Lutterschmidt, W.I.; Hutchison, V.H. The onset of spasms as the definitive end point. Can. J. Zool. 1997, 75, 1553–1560. [Google Scholar] [CrossRef]
- Fox, J.; Weisberg, S. An {R} Companion to Applied Regression, Third Edition. Thousand Oaks CA: Sage Publications. Inc. 2019, 608 pages https://socialsciences.mcmaster.ca/jfox/Books/Companion/.
- R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, 2019. https://www.R-project.org/.
- Giraudoux, P. pgirmess: Spatial Analysis and Data Mining for Field Ecologists. R package version 1.6.9. 2018. https://cran.r-project.org/web/packages/pgirmess/index.html.
- Tran, D.V.; Phan, D.V.; Lau, K.A.; Nishikawa, A. Ecological niche variation drives morphological differentiation in tropical stream-dwelling newts from Indochina. Journal of Zoology, 2024; Early View (29 April 2024). [Google Scholar] [CrossRef]
- Strickland, J.C.; Pinheiro, A.P.; Cecala, K.K.; Dorcas, M.E. Relationship between Behavioral Thermoregulation and Physiological Function in Larval Stream Salamanders. J. Herpetol. 2016, 50, 239–244. [Google Scholar] [CrossRef]
- Cupp, P.V. Thermal Tolerance of Five Salientian Amphibians during Development and Metamorphosis. Herpetologica 1980, 36, 234–244. [Google Scholar]
- Bury, R.B. Low thermal tolerances of stream amphibians in the Pacific Northwest: Implications for riparian and forest management. Appl. Herpetol. 2008, 5, 63–74. [Google Scholar] [CrossRef]
- Bury, R.B. Low thermal tolerances of stream amphibians in the Pacific Northwest: Implications for riparian and forest management. Appl. Herpetol. 2008, 5, 63–74. [Google Scholar] [CrossRef]
- Costanzo, J.P.; Lee, R.E. Avoidance and tolerance of freezing in ectothermic vertebrates. J. Exp. Biol. 2013, 216, 1961–1967. [Google Scholar] [CrossRef] [PubMed]
- Vo, P.; Gridi-Papp, M. Low temperature tolerance, cold hardening and acclimation in tadpoles of the neotropical túngara frog (Engystomops pustulosus). J. Therm. Biol. 2017, 66, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Sanabria, E.A.; Rodríguez, C.Y.; Vergara, C.; Ontivero, E.; Banchig, M.; Navas, A.L.; Herrera-Morata, M.; Quiroga, L.B. Thermal ecology of the post-metamorphic Andean toad (Rhinella spinulosa) at elevation in the monte desert, Argentina. Journal of Thermal Biology 2015, 52, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Montori, A.; Campeny, R. Situación actual de las poblaciones de tritón pirenaico, Euproctus asper, en el macizo del Montseny. Boletín de la Asociación Herpetológica Española 1991, 2, 10–12. [Google Scholar]
- Amat, F.; Montori, A. El tritó del Montseny Calotriton arnoldi i els amfibis i rèptils del Parc Natural i Reserva de la Biosfera del Montseny. Granollers, Museu de Ciències Naturals de Granollers Eds. 2023. p. 196.
- Katzenberger, M.J. Thermal Tolerance and Sensitivity of Amphibian Larvae from Paleartic and Neotropical communities. Trabajo de Fin de Master. 2009. Universidad de Lisboa.
- Levinton, J.S. The latitudinal compensation hypothesis: growth data and a model of latitudinal growth differentiation based upon energy budgets. I. Interspecific comparison of Ophryotrocha (Polychaeta: dorvilleidae). Biol. Bull. 1983, 165, 686–698. [Google Scholar]
- Fangue, N.A.; Podrabsky, J.E.; Crawshaw, L.I.; Schulte, P.M. Countergradient variation in temperature preference in populations of killifish, Fundulus heteroclitus. Physiol. Biochem. Zool. 2009, 82, 776–786. [Google Scholar] [CrossRef] [PubMed]
- Montori, A. Is the Pyrenean newt (Calotriton asper) a thermoconformer? Cloacal and water temperature in two different thermal periods in a Pre-Pyrenean stream population. Basic and Applied Herpetology 2024, (accepted; in press). [Google Scholar] [CrossRef]
- Montori, A.; Llorente, G.A.; Richter-Boix, A. Habitat features affecting the small-scale distribution and longitudinal migration patterns of Calotriton asper in a pre-Pyrenean population. Amphibia-Reptilia 2008, 29, 371–381. [Google Scholar] [CrossRef]
- Hadamová, M.; Gvoždík, L. Seasonal Acclimation of Preferred Body Temperatures Improves the Opportunity for Thermoregulation in Newts. Physiol. Biochem. Zool. 2011, 84, 166–174. [Google Scholar] [CrossRef]
- Amat, F.; Roig, J.M. Amfibis al límit, conservació de Rana temporaria i Euproctus asper al límit meridional de la seva distribució, al Parc Natural del Montseny. Simposi Sobre del Declivi de les Poblacions d'Amfibis. Lleida, 2004 marzo de 2004.
- Ledesma, J.L.J.; Montori, A.; Altava-Ortiz, V.; Barrera-Escoda, A.; Cunillera, J.; Àvila, A. Future hydrological constraints of the Montseny brook newt (Calotriton arnoldi) under changing climate and vegetation cover. Ecol. Evol. 2009, ece3.5506. [Google Scholar] [CrossRef]
- de Pous, P.; Montori, A.; Amat, F.; Sanuy, D. Range contraction and loss of genetic variation of the Pyrenean endemic newt Calotriton asper due to climate change. Reg. Environ. Change 2016, 16, 995–1009. [Google Scholar] [CrossRef]
- Avila, A.; Fernández-Martínez, M. El canvi climàtic al Montseny: anàlisi de les tendències de variació de la temperatura de l’aire i l’aigua a la vall de la Castanya en les tres darreres dècades. IX Trobada D’estudiosos Del Montseny, 2018; 454–464, Diputació de Barcelona. [Google Scholar]

| Western population | |||||||
|---|---|---|---|---|---|---|---|
| Weight (g) | CTmax (°C) | S.E | Min (°C) | Max (°C) | R2 | p-value | |
| Males | 5.70 | 31.68 | 0.07 | 31.4 | 31.8 | -0.39 | 0.44 |
| Females | 7.03 | 31.38 | 0.07 | 31.1 | 31.5 | -0.52 | 0.29 |
| Juveniles | 2.33 | 31.55 | 0.17 | 31.0 | 32.1 | -0.26 | 0.61 |
| Larvae 2017 | 0.75 | 33.50 | 0.10 | 33.0 | 33.6 | 0.67 | 0.14 |
| Larvae 2018 | - | 32.28 | 0.03 | 32.2 | 32.4 | - | - |
| Eastern population | |||||||
| Males | 6.12 | 31.72 | 0.04 | 31.6 | 31.8 | -0.74 | 0.09 |
| Females | 4.33 | 31.45 | 0.09 | 31,0 | 31.6 | -0.51 | 0.30 |
| Juveniles | 2.35 | 31.50 | 0.27 | 30.2 | 31.9 | -0.21 | 0.69 |
| Larvae 2017 | 1.40 | 31.53 | 0.26 | 30.5 | 32.2 | -0.30 | 0.56 |
| Larvae 2018 | - | 31.10 | 0.19 | 30.5 | 31.5 | - | - |
| Western population | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Weight (g) | SVL(cm) | Tp (°C) | SE | Min | Max | R2 Weight- Tp | p-value | R2SVL- Tp | p-value | |
| Males | 5,15 | 5,95 | 13,35 | 0,50 | 12,2 | 14,6 | -0,40 | 0,42 | -0,15 | 0,78 |
| Females | 4,73 | 6,13 | 16,45 | 0,48 | 15,3 | 17,7 | -0,76 | 0,08 | -0,01 | 0,98 |
| Juveniles | 2,08 | 4,48 | 17,51 | 1,46 | 13,7 | 21,6 | 0,68 | 0,13 | 0,76 | 0,08 |
| Eastern population | ||||||||||
| Males | 5,63 | 6,05 | 16,43 | 1,58 | 11,7 | 20,0 | -0,07 | 0,89 | 0,56 | 0,24 |
| Females | 3,98 | 5,61 | 18,56 | 1,13 | 15,4 | 21,3 | -0,29 | 0,57 | -0,03 | 0,95 |
| Juveniles | 2,2 | 4,32 | 18,97 | 0,98 | 16,4 | 21,0 | -0,12 | 0,82 | -0,06 | 0,92 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




