Submitted:
07 June 2024
Posted:
10 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Animals, General Preparations, and Peptides Potency In Vitro
5.2. Drugs and Solutions
5.3. Circular Dichroism
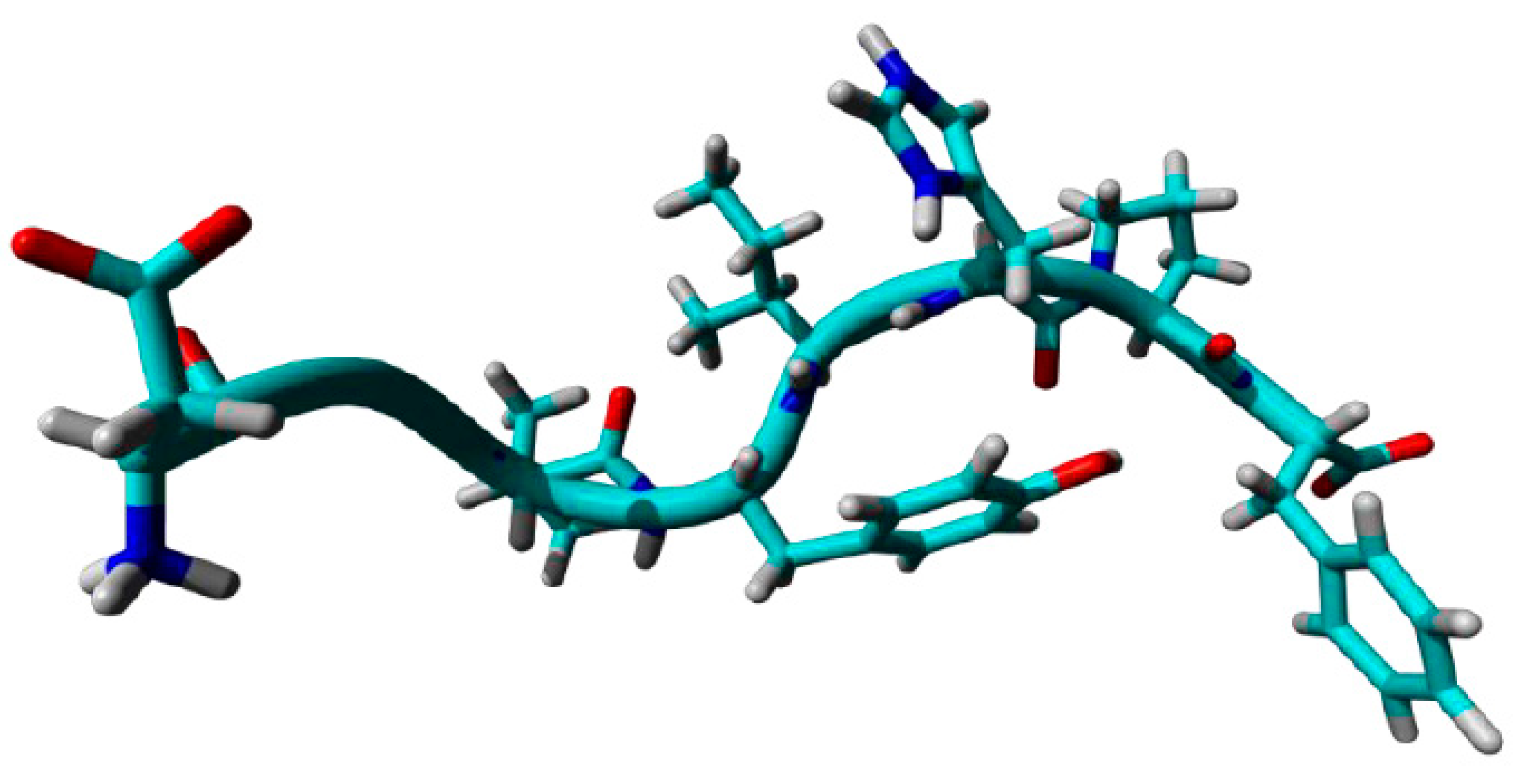
5.4. Molecular Dynamics
5.5. Statistical Analysis
Institution Review Board Statements
Supplementary Materials
Author Contributions
Funding
Informed consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aboye, T.; Meeks, C.; Majumder, S.; Shekhtman, A.; Rodgers, K.; Camarero, J. Design of a MCoTI-based cyclotide with angiotensin (1-7)-like activity. Molecules 2016, 21, 152. [Google Scholar]
- Barany, G.; Merrifield. R. B. The Peptides, 1980, 2, 1-284, Gross, E , Meinhofer, J.(eds), Academic Press Inc., New York.
- Barros, A. J.; Ito, C. M.; Makino, E. N.; Cembranelli, F. A. M.; Moraes, F. C.; Souza, S. E. G.; Oliveira. L.; Shimuta, I. S.; Nakaie, C. R. Factors regulating tachyphylaxis triggered by N-terminal-modified angiotensin II analogs”. Biological Chemistry 2009, 390. https://www.degruyter.com/doi/10.1515/BC.2009.143.
- Bernhofer, M., Dallago, C.; Karl, T.; satagopam, V.; Heinzinger, M.; Littmann, M.; Olenyl, T.; Qiu, J.; Schütze, K.; Yachdav, G.; Ashkenazy, H.; Ben-Tal, N.; Bromberg, Y.; Goldberg, T.; Kajan, L.; O´Donoghue, S.; Sander, C,. Schafferhans, A.; Vriend, G.; Mirdita, M.; Gawron, P.; Gu, W.; Jarosz, Y.; Trefois, C.; Steinegger, M.; Schneider, R.; Bost, B. PredictProtein – Predictng protein structure and function for 29 years. Nucleic Acid Res. 2021, 49(W1):W540m. [CrossRef]
- Carstens, B.B,.; Swedberg, J.; Berecki, G.; Adams, D.J.; Craik, D.J.; Clark, R.J. Effects of linker sequence modifications on the structure, stability, and biological activity of a cyclic α-Conotoxin. Biopolymers 2016, 106, 864–75.
- Chowdhury, S.;M.; Talukder, S.A.; Khan, A.; M, Afrin, N.; Ali, M.A.; Islam, R.; Parves. R, Al Mamun, A.; Sufian, M.A.; Hossain, M.N.; Hossain, M.A.; Halim, M.A.; Antiviral peptides as promising therapeutics against SARS-CoV-2. J. Phys. Chem. 2020. 124, 9785–9792.
- Correa, S.A,; Zalcberg, H.; Han, S.W.;, Oliveira, L.; Costa-Neto, C.M.; Paiva, A.C.; Shimuta. S. I.; Aliphatic amino acids in helix VI of the AT1 receptor play a relevant role in agonist binding and activity. Regul. Pept. 2002 106 , 33–38.
- De Vivo, M.; Matteo, M.; Giovanni, B.; Cavalli, A. Role of molecular dynamics and related methods in drug discovery. J. Med. Chem. 2016, 2016 59, 4035–4061. [Google Scholar]
- Ferreira, M.M.L.; Souza, S.E.G.; Silva, C.C.; Souza, L. E. A.; Bicev, R. N.; Silva, E. R.; Nakaie, C. R.; Pyroglutamination-induced changes in the physicochemical features of a CXCR4 chemokine peptide: kinetic and structural snalysis. Biochemistry. 2023 5; 62, 2530-2540. [CrossRef]
- Hunyady, L., Baukal, A. J.; Balla, T.; Catt. K. J.; Independence of type I angiotensin II receptor endocytosis from G protein coupling and signal transduction. J. Biol. Chem. 1994, 269, 24798–804.
- Iismaa, T.P., Shine, J.. G protein-coupled receptors;. Curr. Opin. Cell. Biol. 1992. 4, 195–202.
- Inoue, I, Nakajima, T.; Williams, C. S.; Quackenbush, J.; Puryear, R.; Powers, M., Cheng, T.; Ludwig, E. H.; Sharma, A. M.,; Hata, A.; Jeunemaitre, X.; Lalouel, J. M.; A Nucleotide substitution in the promoter of human angiotensinogen is associated with essential hypertension and affects basal transcription in vitro. J. Clin. Invest. 1997. 99, 1786–97.
- Kawamura, S,; Kakuta, Y,; Tanaka, Ii.; Hikichi, K.; Kuhara. S.; Yamasaki. N.; Kimura, M. Glycine-15 in the bend between two α-helices can explain the thermostability of DNA binding protein HU from Bacillus Stearothermophilus. Biochemistry 1996 35, 1195–1200.
- Krieger, G.; Darden, T.; Nabuurs, S. B.; Finkelstein, A.; Vriend, G. Making optimal use of empirical energy functions: force-field parameterization in crystal space. Proteins 2004, 57, 678–683. [Google Scholar] [CrossRef]
- Krieger, E.; Vriend. New Ways to Boost Molecular Dynamics Simulations. J. Comput. Chem. 2015, 36, 996–1007. [Google Scholar]
- Leeb-Lundberg, L. M.; Fredrik, D. S.K .; Maria E. L.; Fathy, L. B.; The human B1 bradykinin receptor exhibits high ligand-independent, constitutive ctivity. J. Biol. Chem. 2001 276, 8785–892.
- Leeb-Lundberg, L.; M, Marceau. F,; Müller-Esterl W.; Pettibone, D.J.; Zuraw, B.L.; International union of pharmacology XLV. Classification of the inin receptor family: from molecular mecanism to pathophysiological consequences. Pharmacol. Rev. 2005.57, 27–77.
- Lincoff, J.; Sukanya, S., Head-Gordon, T. Comparing generalized fnsemble methods for sampling of systems with many degrees of freedom. J. Chem. Phys. 2016, 145, 174107.
- Lopes, D. D. ; Vieira, R. F. F. ; Malavolta, L. ; Poletti, E. F., Shimuta, S. I. ; Paiva, A. C. M. ; Schreier, S. ; Oliveira, L. ; Nakaie, C. R Short peptide constructs mimic agonist sites of AT1R and BK receptors. Amino Acids 2013, 44, 835–846.
- Marchetto, R.; Schreier, S.; Nakaie, C, R,; A novel spin-labeled amino acid derivative for use in peptide synthesis: (9-fluorenylmethyloxycarbonyl)-2,2,6,6-tetramethylpiperidine-N-Oxyl-4-amino-4-carboxylic acid. J. Amer. Chem. Soc., 1993, 115; 11042–1143.
- Monnot C, Bihoreau C, Conchon S, Curnow KM, Corvol P, Clauser E. 1996. “Polar Residues in the Transmembrane Domains of the Type 1 Angiotensin II Receptor Are Required for Binding and Coupling”. Journal of Biological Chemistry 271(3): 1507–13.
- Morrone, J. A., Perez, A.; MacCallum, J.; Dill, K.A. . 2017. Computed binding of peptides to proteins with MELD-accelerated molecular dynamics.. J. Chem. Theory. Comput. 2017. 13, 870–76.
- Nakaie, C.R.; Goissis, G.; Schreier, S.; Paiva, A.C.M. pH dependence of ESR spectra of nitroxide containing ionizable groups. Braz; J. Med. Biol. Res, 1981 14, 173-180.
- Nakaie, C.R.; Schreier, S; Paiva, A.C.M. Synthesis and properties of spin-labeled angiotensin derivatives.. Biochim. Biophys. Acta, 1983, 742, 63-71.
- Nakaie, C.R.; Silva, E.G.; Cilli, E.M.; Marchetto, R.; Schreier, S.; Paiva, T.B.; Paiva, A.C.M. Synthesis and pharamacological properties of TOAC-labeled angiotensin and bradykinin analogs. Peptides 2002, 23, 65–70. [Google Scholar]
- Oliveira, L., Paiva, A.C.M., Sander, C., Vriend. G. A common step for signal transduction in G protein-coupled receptors. Trend. Pharmacol. Sciences 1994, 15, 170–172.
- Oliveira, L.; Costa-Neto, C.M.; Nakaie, C.R.; Schreier, S.; Shimuta, S. I., Paiva, A.C.; The Angiotensin II AT 1 receptor structure-activity correlations in the light of rhodopsin structure. Physiol. Rev. 2007, 87, 565–592.
- Peach, M. J.; Dostal, D. E. The angiotensin II receptor and the cctions of angiotensin II. J. Cardiov. Pharmacol. 1990, 16, S25–30. [Google Scholar]
- Pignatari, G. C.; Rozenfeld, R.; Ferro, E. S.;, Oliveira, L.; Paiva, A.C.; Devi, L.A.; A role for transmembrane domains V and VI in ligand binding and maturation of the angiotensin II AT1 receptor. Biol. Chem. 2006, 387, https://www.degruyter.com/view/j/bchm.2006.387-3/bc.2006.036/bc.2006.036.xml.
- Prekash, A.; Sprenger, K.G., Pfaendtner, J.; Essential slow degrees of freedom in protein-surface simulations: a metadynamics investigation. Biochem. Biophys. Res. 2018, 498, 274–281.
- Sandberg, K., Ji, H.; Catt, K.J.; Regulation of angiotensin II receptors in rat Brain during dietary sodium changes. Hypertension 1994. 23, I137–I137.
- Schreier, S.; Barbosa, S.R.; Casallanovo, F.; Vieira, R.F.; Cilli, E.M.; Paiva, A.C.; Nakaie, C.R. Conformational basis for the biological activity of TOAC-labeled angiotensin II and bradykinin: electron paramagnetic resonance, circular dichroism, and fluorescence studies. Biopolymers 2004, 74, 389–402. [Google Scholar]
- Schreier, S.; Bozelli, Jr, J. C., Marin, N., Vieira, R. F. F., Nakaie, C. R. .The spin label amino acid TOAC and its uses in studies of peptides: chemical, physicochemical, spectroscopic, and conformational aspects. Biophys. Rev. 2012, 45–66.
- Senthilkumar, B. D.; Meshach, P., Srinivasan, E.;, Rajasekaran. R. Structural stability among hybrid antimicrobial peptide Cecropin A (1–8)–Magainin 2(1–12) and its analogs: a computational approach. J. Cluster Sci. 2017, 28, 2549–63.
- Silva, R. L., Papakyriakou, A., Carmona, A. K., Spyroulias, G. A., Sturrock, E. D., Bersanetti, P.A.; Nakaie, C. R. Peptide inhibitors of angiotensin-I converting enzyme based on angiotensin (1-7) with selectivity for the C-terminal domain. Bioorg. Chem. 2022, 129, 106204.
- Sharma, A K., Persichetti, J, Tale, E., Prelvukaj, G, Cropley T, Choudhury, R. A Computational examination of the binding interactions of amyloid β and human Cystatin C. J. Theor. Comput. Chem. 2018.17, 1850001.
- Tigerstedt, R.; Bergman, P. Q.; Niere Und Kreislauf . Skandinavisches Archiv Für Physiologie 1898, 8, 223–271.
- Timmermans, P.B.; Wong, P. C.; Chiu, A.T.; Herblin, W.F.; Smith, R.D.;. New perspectives in angiotensin system control. J. Human Hypert. 1993 . S19-31.
- Toniolo, C.; Crisma, M.; Formaggio, F. TOAC, a nitroxide spin-labeled, achiral C (alpha)-tetrasubstituted alpha-amino acid is an excellent tool in material science and biochemistry. Biopolymers, 1998, 47, 153-158.
- Verlander, M. 2001.Solid-phase synthesis: a practical guide, Edit. Kates, E. and Albericio, F., Marcel Dekker: New York. 2000. 826, ISBN 0-8247-0359-6.
- Yan, B.X.; Sun, Y. Q,. Glycine residues provide flexibility for enzyme active sites. J. Biol. Chem. 1997, 272, 3190–3194.
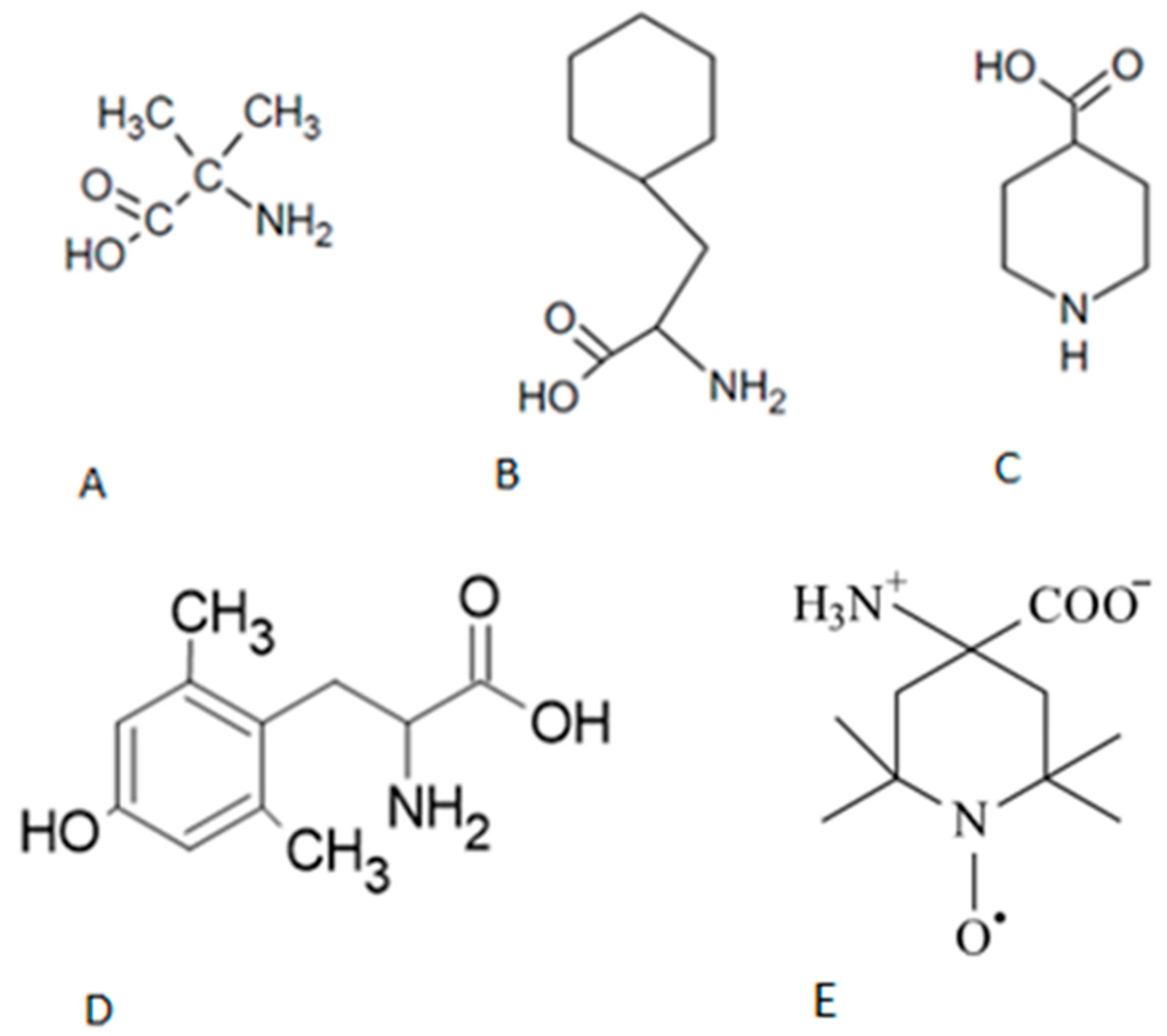
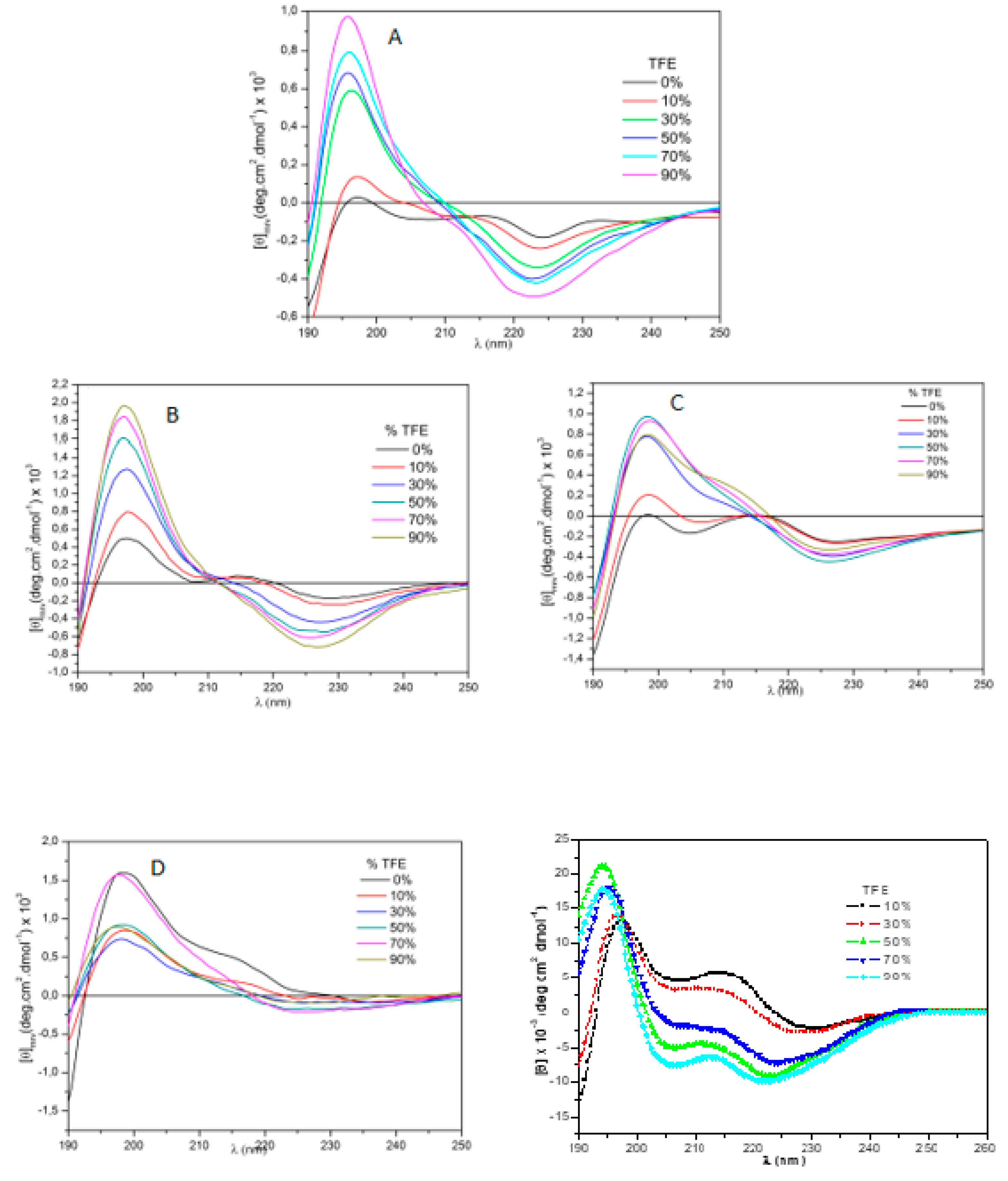
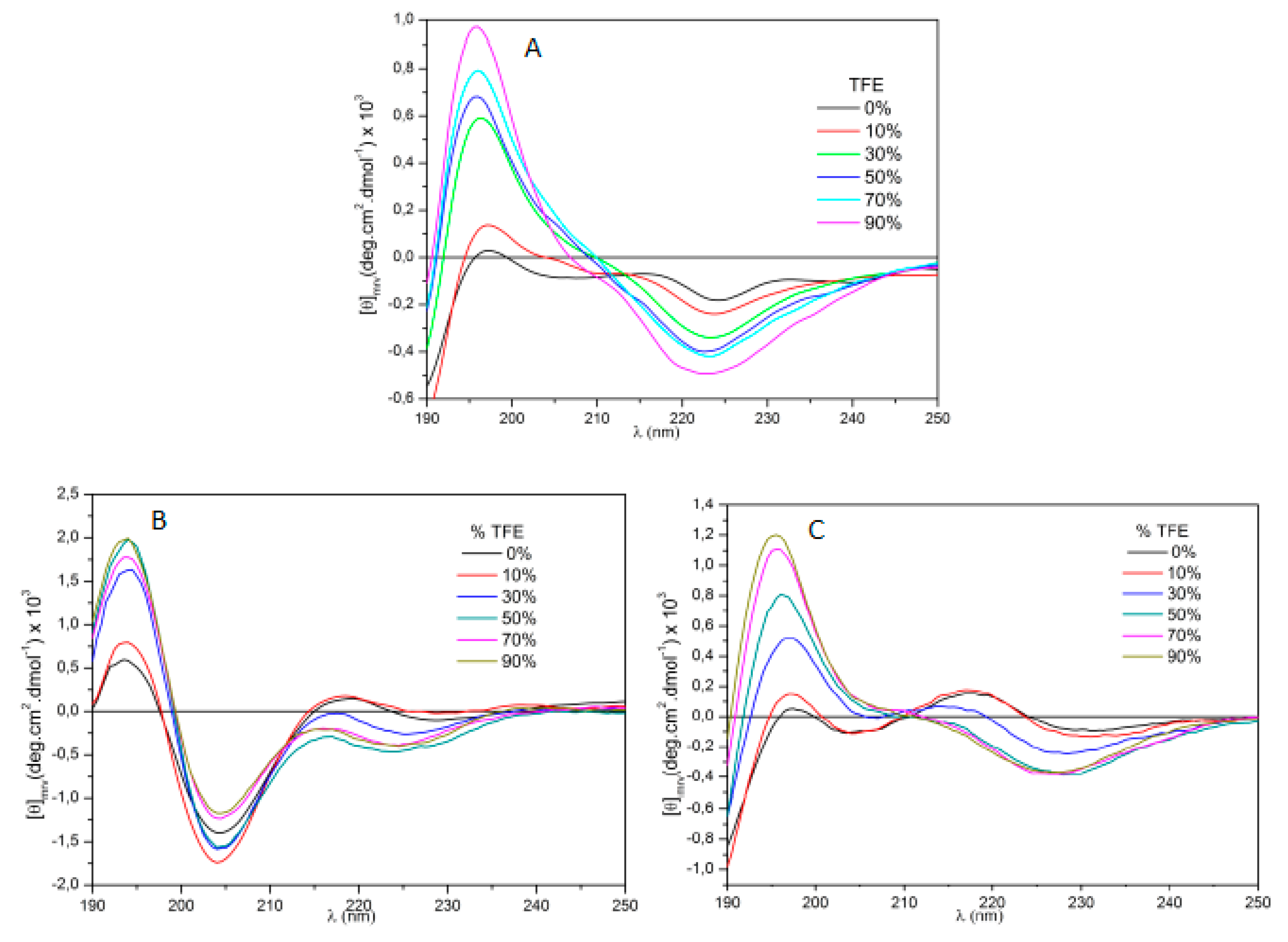

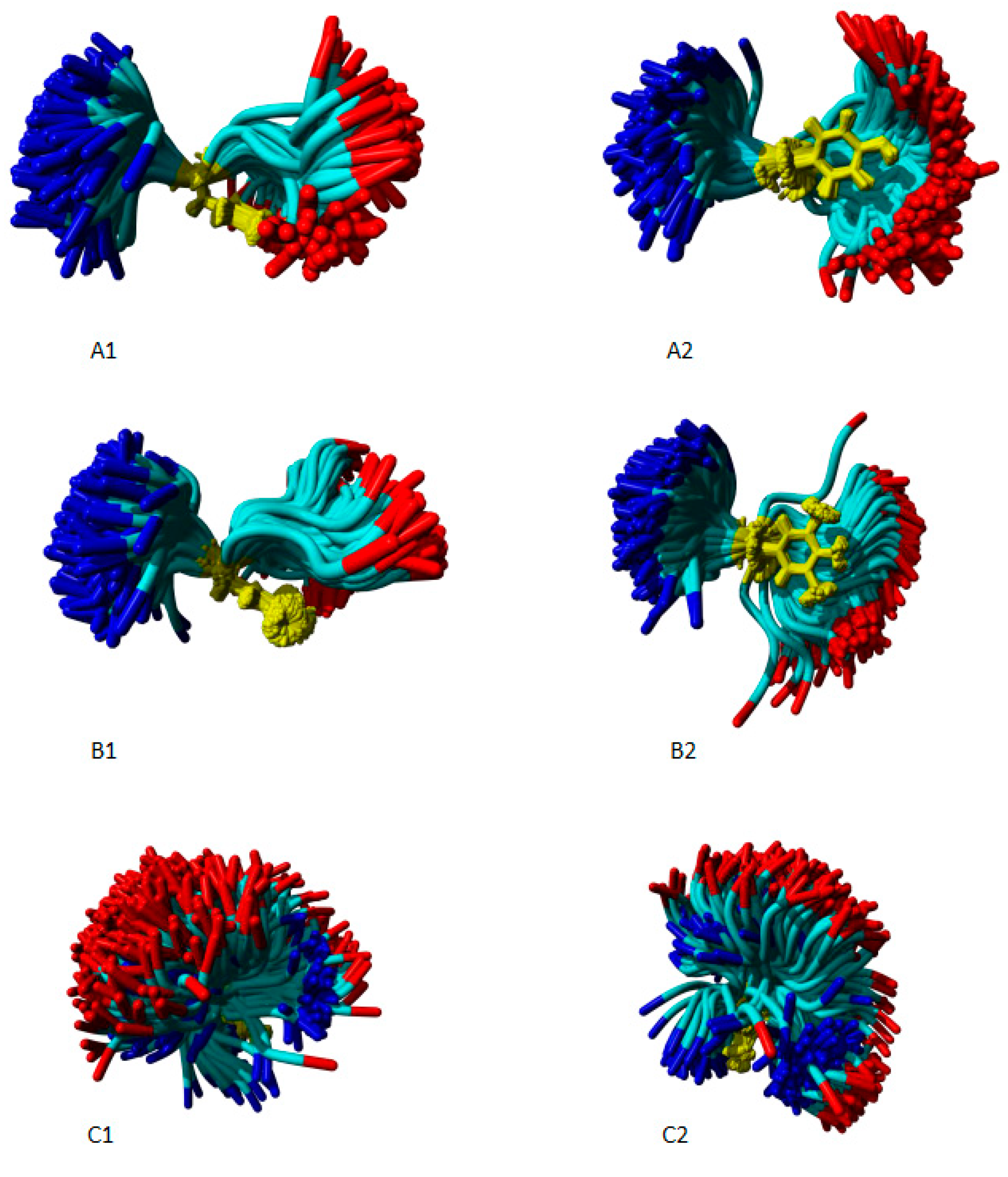
| # | Peptides | pD2 | % of AngII activity |
|---|---|---|---|
| 1 | AngII | 8,90 ± 0,01 | 10100,0 |
| 2 | AIB3-AngII | 7,42 ± 0,06 | 3,3 |
| 3 | CHA3-AngII | 7,25 ± 0,01 | 2,2 |
| 4 | IAP3-AngII | Inactive | -- |
| 5 | TOAC3-AngII | Inactive | -- |
| 6 | DMT4-AngII | 8,54 ± 0,03 | 43,6 |
| 7 | Phe4-AngII* | 7,97 ± 0,05 | 11,7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




