Submitted:
06 June 2024
Posted:
10 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
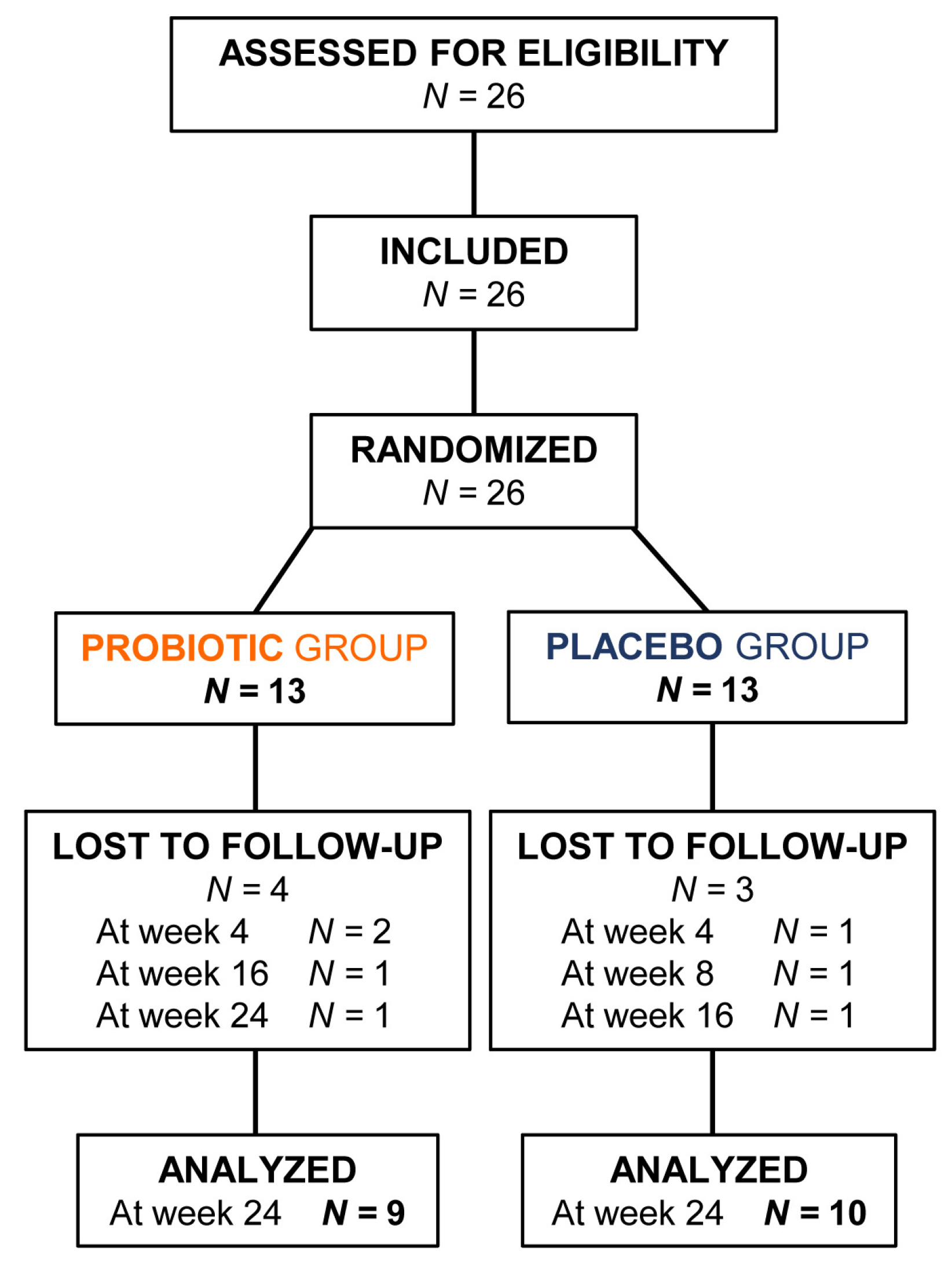
2.1. Study Design and Ethics
2.2. Study Population
2.3. Randomization and Intervention
2.4. Outcomes
2.5. Statistical Analysis
2.6. Gut and Skin Microbiota Study
3. Results
3.1. Baseline Descriptive Data
3.2. Number of AA Plaques
3.3. Affected Scalp Surface Area (SALT Scale)
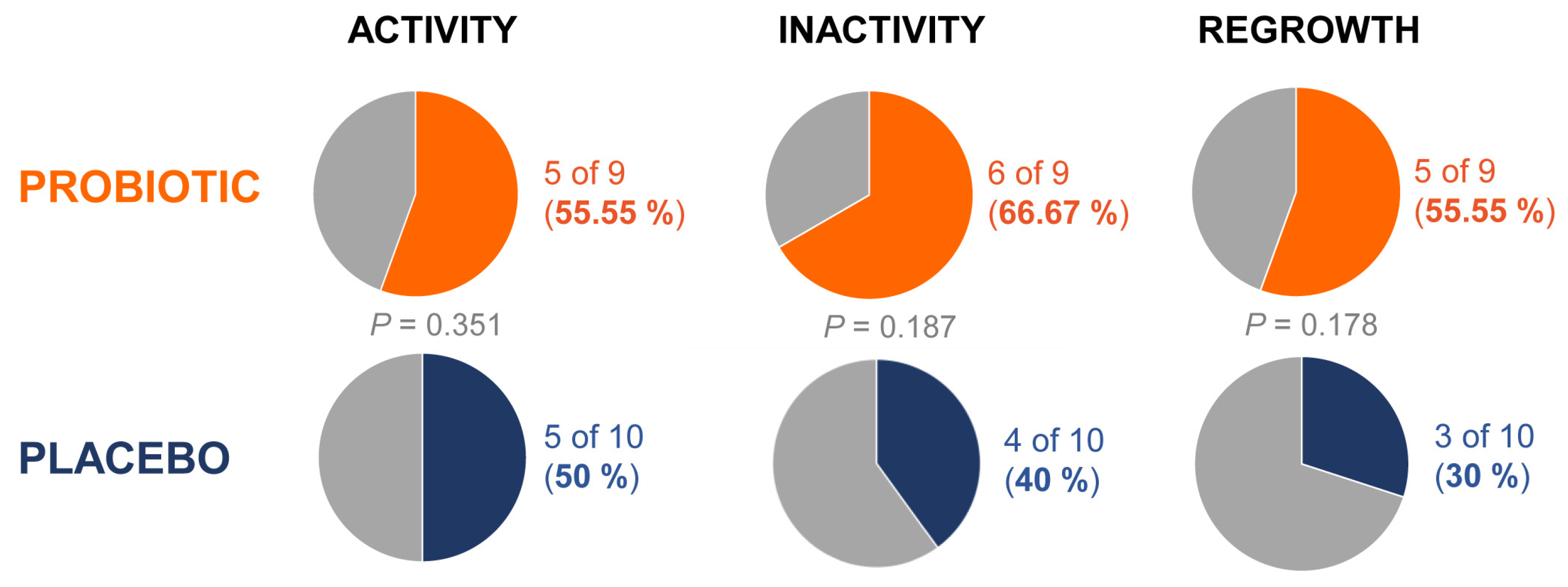
3.4. Activity, Inactivity and Regrowth Scores
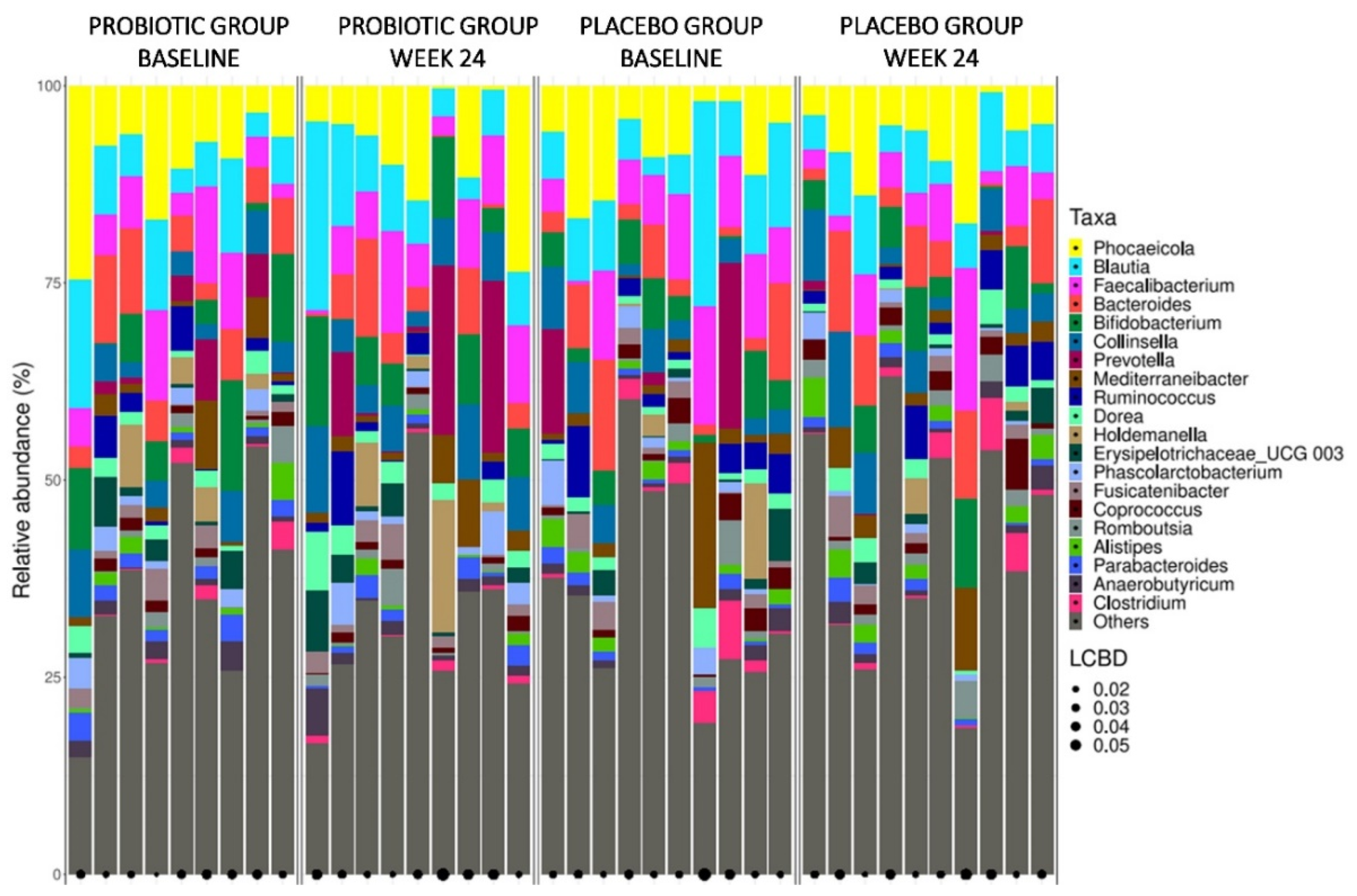
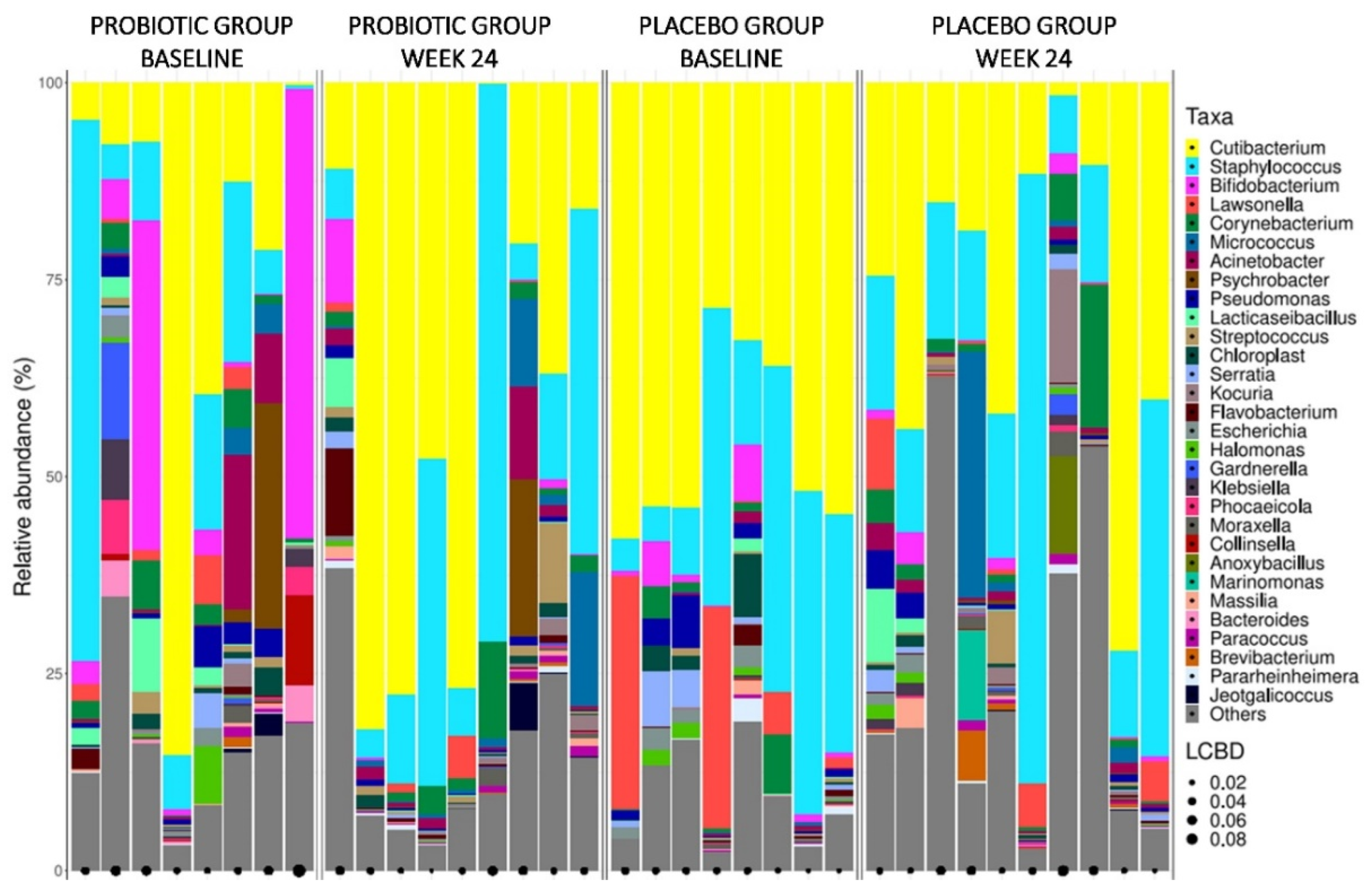
3.5. Microbiota Analysis
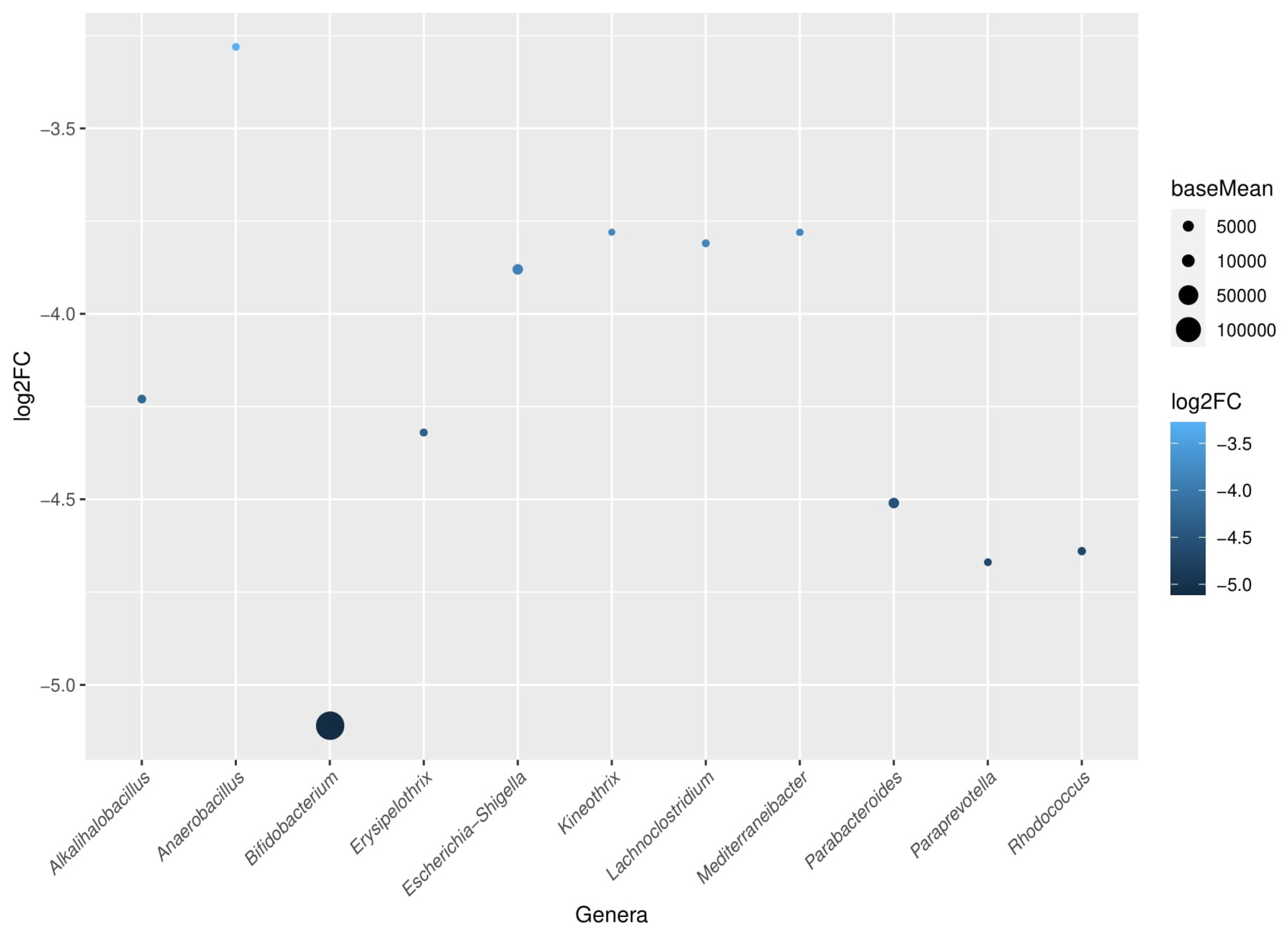
3.5.1. Gut Microbiota
3.5.1. Skin Microbiota
3.6. Safety
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rajabi, F.; Drake, L.A.; Senna, M.M.; Rezaei, N. Alopecia Areata: A Review of Disease Pathogenesis. Br J Dermatol 2018, 179, 1033–1048. [CrossRef]
- Suchonwanit, P.; Kositkuljorn, C.; Pomsoong, C. Alopecia Areata: An Autoimmune Disease of Multiple Players. Immunotargets Ther 2021, 10, 299–312. [CrossRef]
- Simakou, T.; Butcher, J.P.; Reid, S.; Henriquez, F.L. Alopecia Areata: A Multifactorial Autoimmune Condition. J Autoimmun 2019, 98, 74–85. [CrossRef]
- Minokawa, Y.; Sawada, Y.; Nakamura, M. Lifestyle Factors Involved in the Pathogenesis of Alopecia Areata. Int J Mol Sci 2022, 23, 1038. [CrossRef]
- Meah, N.; Wall, D.; York, K.; Bhoyrul, B.; Bokhari, L.; Sigall, D.A.; Bergfeld, W.F.; Betz, R.C.; Blume-Peytavi, U.; Callender, V.; et al. The Alopecia Areata Consensus of Experts (ACE) Study: Results of an International Expert Opinion on Treatments for Alopecia Areata. J Am Acad Dermatol 2020, 83, 123–130. [CrossRef]
- Mahmud, M.R.; Akter, S.; Tamanna, S.K.; Mazumder, L.; Esti, I.Z.; Banerjee, S.; Akter, S.; Hasan, M.R.; Acharjee, M.; Hossain, M.S.; et al. Impact of Gut Microbiome on Skin Health: Gut-Skin Axis Observed through the Lenses of Therapeutics and Skin Diseases. Gut Microbes 2022, 14, 2096995. [CrossRef]
- O’Neill, C.A.; Monteleone, G.; McLaughlin, J.T.; Paus, R. The Gut-Skin Axis in Health and Disease: A Paradigm with Therapeutic Implications. Bioessays 2016, 38, 1167–1176. [CrossRef]
- Navarro-López, V.; Núñez-Delegido, E.; Ruzafa-Costas, B.; Sánchez-Pellicer, P.; Agüera-Santos, J.; Navarro-Moratalla, L. Probiotics in the Therapeutic Arsenal of Dermatologists. Microorganisms 2021, 9, 1513. [CrossRef]
- Sánchez-Pellicer, P.; Navarro-Moratalla, L.; Núñez-Delegido, E.; Ruzafa-Costas, B.; Agüera-Santos, J.; Navarro-López, V. Acne, Microbiome, and Probiotics: The Gut-Skin Axis. Microorganisms 2022, 10, 1303. [CrossRef]
- De Pessemier, B.; Grine, L.; Debaere, M.; Maes, A.; Paetzold, B.; Callewaert, C. Gut-Skin Axis: Current Knowledge of the Interrelationship between Microbial Dysbiosis and Skin Conditions. Microorganisms 2021, 9, 353. [CrossRef]
- Borde, A.; Åstrand, A. Alopecia Areata and the Gut-the Link Opens up for Novel Therapeutic Interventions. Expert Opin Ther Targets 2018, 22, 503–511. [CrossRef]
- Moreno-Arrones, O.M.; Serrano-Villar, S.; Perez-Brocal, V.; Saceda-Corralo, D.; Morales-Raya, C.; Rodrigues-Barata, R.; Moya, A.; Jaen-Olasolo, P.; Vano-Galvan, S. Analysis of the Gut Microbiota in Alopecia Areata: Identification of Bacterial Biomarkers. J Eur Acad Dermatol Venereol 2020, 34, 400–405. [CrossRef]
- Lu, J.; Zhang, P.; Hu, R.; Qi, S.; Zhao, Y.; Miao, Y.; Han, Y.; Zhou, L.; Yang, Q. Gut Microbiota Characterization in Chinese Patients with Alopecia Areata. J Dermatol Sci 2021, 102, 109–115. [CrossRef]
- Rangu, S.; Lee, J.-J.; Hu, W.; Bittinger, K.; Castelo-Soccio, L. Understanding the Gut Microbiota in Pediatric Patients with Alopecia Areata and Their Siblings: A Pilot Study. JID Innov 2021, 1, 100051. [CrossRef]
- Pinto, D.; Sorbellini, E.; Marzani, B.; Rucco, M.; Giuliani, G.; Rinaldi, F. Scalp Bacterial Shift in Alopecia Areata. PLoS One 2019, 14, e0215206. [CrossRef]
- Juhász, M.L.W.; Chen, S.; Khosrovi-Eghbal, A.; Ekelem, C.; Landaverde, Y.; Baldi, P.; Mesinkovska, N.A. Characterizing the Skin and Gut Microbiome of Alopecia Areata Patients. SKIN The Journal of Cutaneous Medicine 2020, 4, 23–30. [CrossRef]
- Won, E.J.; Jang, H.H.; Park, H.; Kim, S.J. A Potential Predictive Role of the Scalp Microbiome Profiling in Patients with Alopecia Areata: Staphylococcus Caprae, Corynebacterium, and Cutibacterium Species. Microorganisms 2022, 10, 864. [CrossRef]
- Lee, S.; Lee, H.; Lee, C.H.; Lee, W.-S. Comorbidities in Alopecia Areata: A Systematic Review and Meta-Analysis. J Am Acad Dermatol 2019, 80, 466-477.e16. [CrossRef]
- Navarro-López, V.; Ramírez-Boscá, A.; Ramón-Vidal, D.; Ruzafa-Costas, B.; Genovés-Martínez, S.; Chenoll-Cuadros, E.; Carrión-Gutiérrez, M.; Horga de la Parte, J.; Prieto-Merino, D.; Codoñer-Cortés, F.M. Effect of Oral Administration of a Mixture of Probiotic Strains on SCORAD Index and Use of Topical Steroids in Young Patients With Moderate Atopic Dermatitis: A Randomized Clinical Trial. JAMA Dermatol 2018, 154, 37–43. [CrossRef]
- Navarro-López, V.; Martínez-Andrés, A.; Ramírez-Boscá, A.; Ruzafa-Costas, B.; Núñez-Delegido, E.; Carrión-Gutiérrez, M.A.; Prieto-Merino, D.; Codoñer-Cortés, F.; Ramón-Vidal, D.; Genovés-Martínez, S.; et al. Efficacy and Safety of Oral Administration of a Mixture of Probiotic Strains in Patients with Psoriasis: A Randomized Controlled Clinical Trial. Acta Derm Venereol 2019, 99, 1078–1084. [CrossRef]
- Salem, I.; Ramser, A.; Isham, N.; Ghannoum, M.A. The Gut Microbiome as a Major Regulator of the Gut-Skin Axis. Front Microbiol 2018, 9, 1459. [CrossRef]
- Rebello, D.; Wang, E.; Yen, E.; Lio, P.A.; Kelly, C.R. Hair Growth in Two Alopecia Patients after Fecal Microbiota Transplant. ACG Case Rep J 2017, 4, e107. [CrossRef]
- Xie, W.-R.; Yang, X.-Y.; Xia, H.H.-X.; Wu, L.-H.; He, X.-X. Hair Regrowth Following Fecal Microbiota Transplantation in an Elderly Patient with Alopecia Areata: A Case Report and Review of the Literature. World J Clin Cases 2019, 7, 3074–3081. [CrossRef]
- Chen, L.; Li, H.; Li, J.; Chen, Y.; Yang, Y. Lactobacillus rhamnosus GG treatment improves intestinal permeability and modulates microbiota dysbiosis in an experimental model of sepsis. Int J Mol Med 2019, 43, 1139–1148. [CrossRef]
- Panpetch, W.; Hiengrach, P.; Nilgate, S.; Tumwasorn, S.; Somboonna, N.; Wilantho, A.; Chatthanathon, P.; Prueksapanich, P.; Leelahavanichkul, A. Additional Candida albicans administration enhances the severity of dextran sulfate solution induced colitis mouse model through leaky gut-enhanced systemic inflammation and gut-dysbiosis but attenuated by Lactobacillus rhamnosus L34. Gut Microbes 2020, 11, 465–480. [CrossRef]
- Jia, L.; Wu, R.; Han, N.; Fu, J.; Luo, Z.; Guo, L.; Su, Y.; Du, J.; Liu Y. Porphyromonas gingivalis and Lactobacillus rhamnosus GG regulate the Th17/Treg balance in colitis via TLR4 and TLR2. Clin Transl Immunology 2020, 9, e1213. [CrossRef]
- Potrykus, M.; Czaja-Stolc, S.; Stankiewicz, M.; Kaska, Ł.; Małgorzewicz, S. Intestinal Microbiota as a Contributor to Chronic Inflammation and Its Potential Modifications. Nutrients 2021, 13, 3839. [CrossRef]
- Olsen, E.; Hordinsky, M.; McDonald-Hull, S.; Price, V.; Roberts, J.; Shapiro, J.; Stenn, K. Alopecia Areata Investigational Assessment Guidelines. National Alopecia Areata Foundation. J Am Acad Dermatol 1999, 40, 242–246. [CrossRef]
- Waśkiel, A.; Rakowska, A.; Sikora, M.; Olszewska, M.; Rudnicka, L. Trichoscopy of Alopecia Areata: An Update. J Dermatol 2018, 45, 692–700. [CrossRef]
- Sánchez-Pellicer, P.; Navarro-Moratalla, L.; Núñez-Delegido, E.; Agüera-Santos, J.; Navarro-López, V. How Our Microbiome Influences the Pathogenesis of Alopecia Areata. Genes (Basel) 2022, 13, 1860. [CrossRef]
- Gupta, A.K.; Carviel, J.L.; Foley, K.A.; Shear, N.H.; Piraccini, B.M.; Piguet, V.; Tosti, A. Monotherapy for Alopecia Areata: A Systematic Review and Network Meta-Analysis. Skin Appendage Disord 2019, 5, 331–337. [CrossRef]
- Yee, B.E.; Tong, Y.; Goldenberg, A.; Hata, T. Efficacy of Different Concentrations of Intralesional Triamcinolone Acetonide for Alopecia Areata: A Systematic Review and Meta-Analysis. J Am Acad Dermatol 2020, 82, 1018–1021. [CrossRef]
| Placebo | Probiotic | |
|---|---|---|
| Sex (woman)* | 11 (85%) | 7 (54%) |
| Age (years)† | 43.4 [34.7 – 52.2] | 35.5 [29.9 – 41.1] |
| Allergies and intolerances* | 3 (23%) | 2 (15%) |
| Preexisting diseases* | 6 (46%) | 6 (46%) |
| TRICOSCOPY | ||
| Activity Scorew0 × AA plaques w0† | 10.4 [5.8 – 14.9] | 10.2 [6.4 – 14.0] |
| Inactivity Score w0 / AA plaques w0† | 0.7 [0.4 – 1.0] | 0.4 [0.2 – 0.6] |
| Regrowth Score w0 / AA plaques w0† | 0.8 [0.4 – 1.1] | 0.4 [0.2 – 0.6] |
| AA plaquesw0† | 3.3 [1.8 – 4.8] | 4.0 [2.7 – 5.1] |
| SALT scale | ||
| S1* | 8 (61.5%) | 6 (46.1%) |
| S2* | 4 (30.8%) | 7 (53.8%) |
| S3* | 0 (0.0%) | 0 (0.0%) |
| S4* | 1 (7.7%) | 0 (0.0%) |
| Activity | Inactivity | Regrowth | |
|---|---|---|---|
| Probiotic | -5.8 [(-19.2) – 7.7] | 4.6 [0.38 – 8.8] | 4.4 [0.1 – 8.7] |
| Placebo | -3.0 [(-8.6) – 2.6] | 2.2 [(-0.8) – 5.3] | 2.0 [(-1.0) – 5.1] |
| P-value † | 0.67 | 0.31 | 0.31 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




