Submitted:
03 June 2024
Posted:
05 June 2024
Read the latest preprint version here
Abstract
Keywords:
INTRODUCTION
METHODOLOGY
Ethical Statement:
Source of Animal Models:
Collection of the Samples:
Inclusion Criteria for Animal Models:
Exclusion Criteria:
Material:
Place and Date of the Study:
Type of Study:
Methods:
Preclinical Trials (Animal Testing) Procedure:
Randomized Human Clinical Trials Phases 1/2 Procedure:
Determination of Humoral Immunity of LNP-mRNA H1N1, H5N1 Flu Vaccine:
Estimation of Cell Mediated Immunity Using Flow Cytometer:

STATISTICS
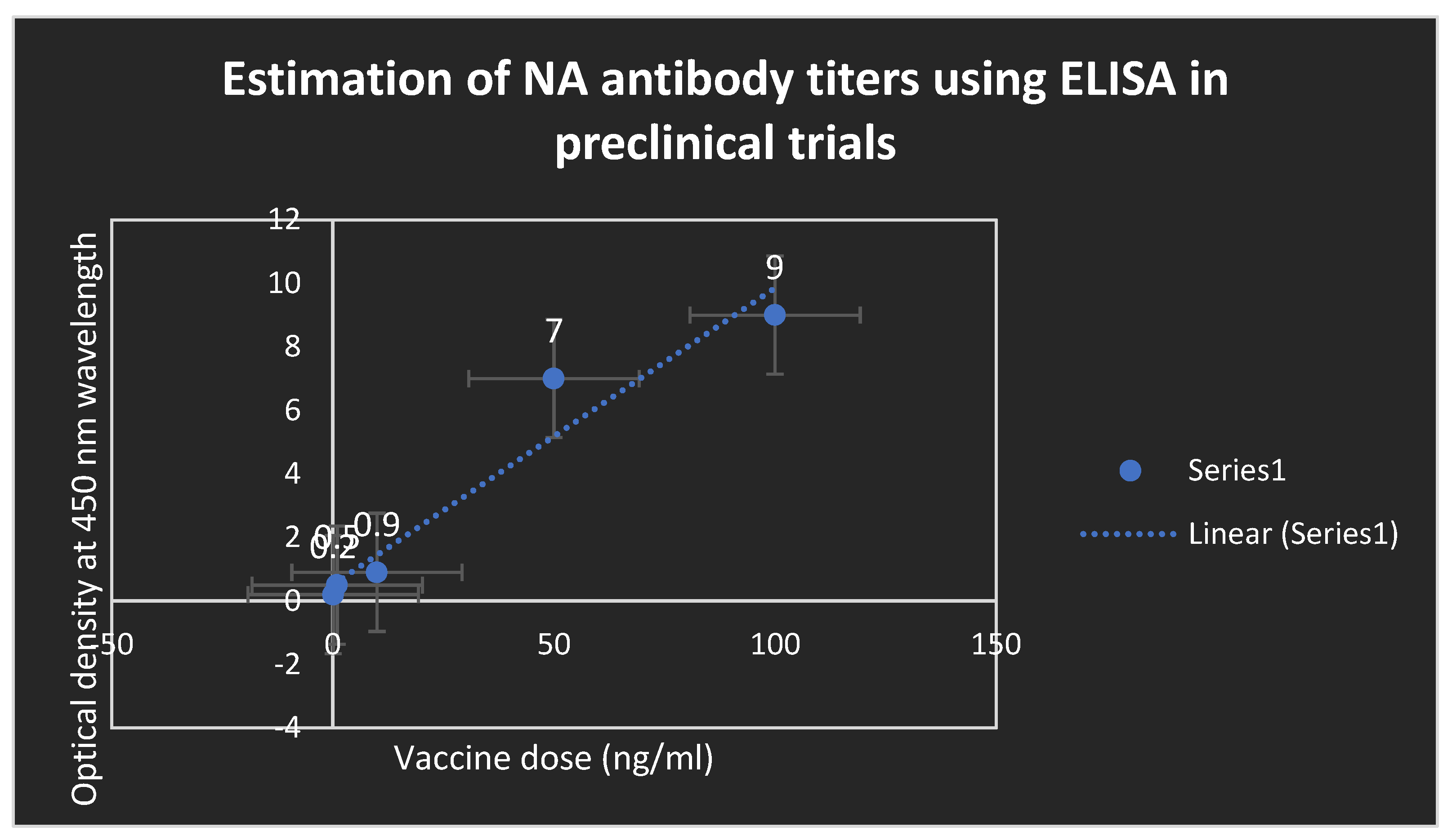
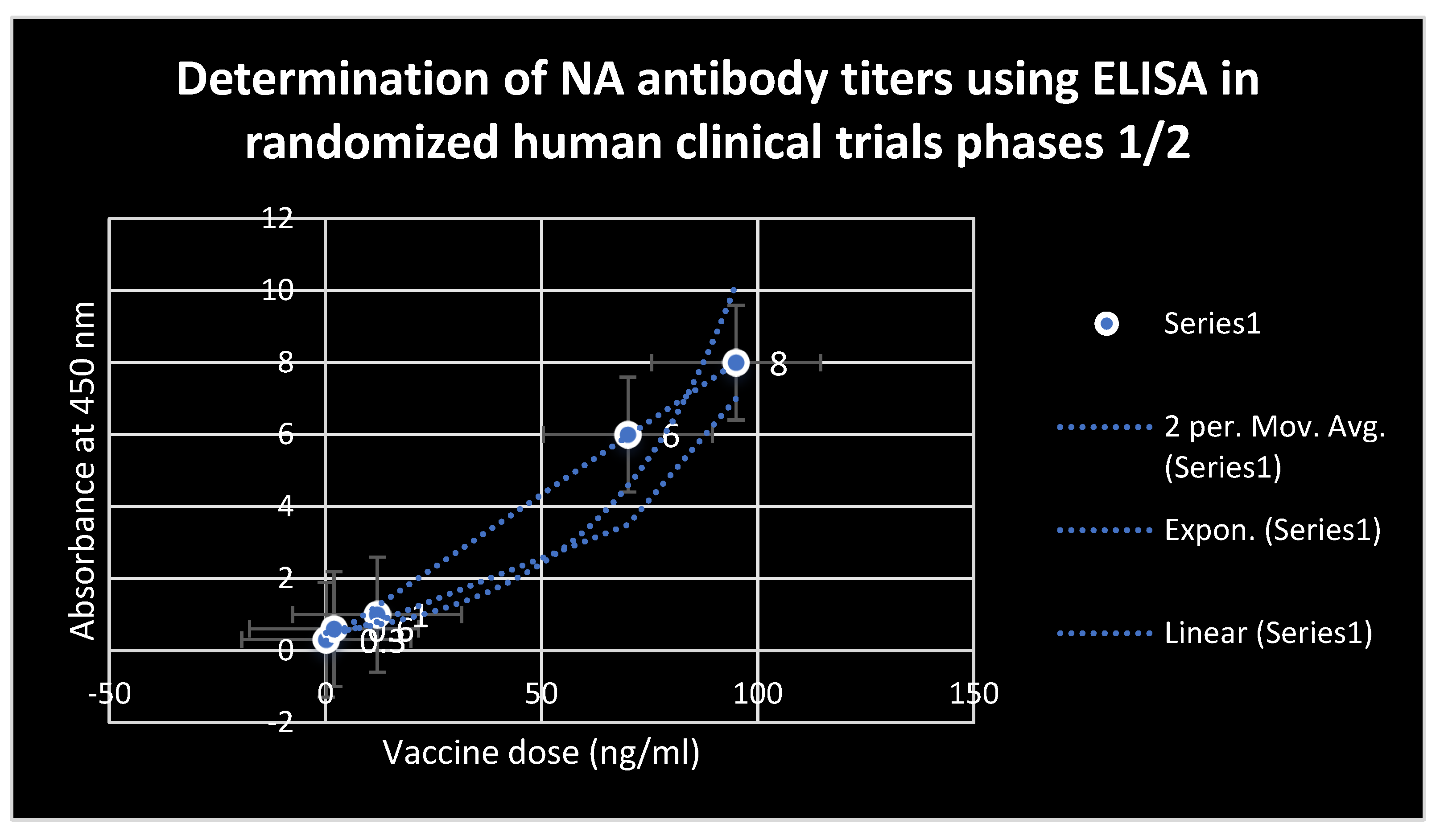
RESULTS

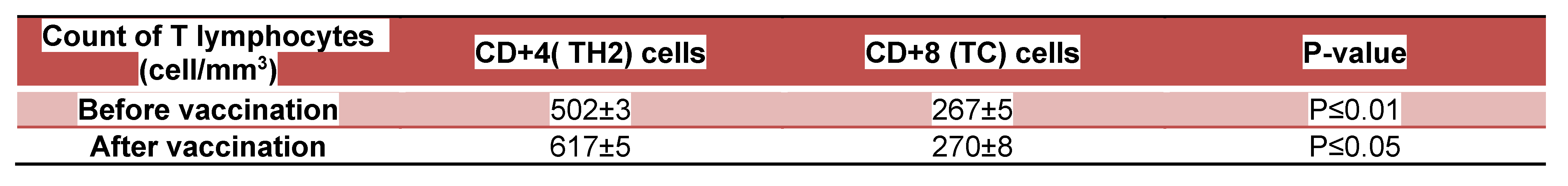


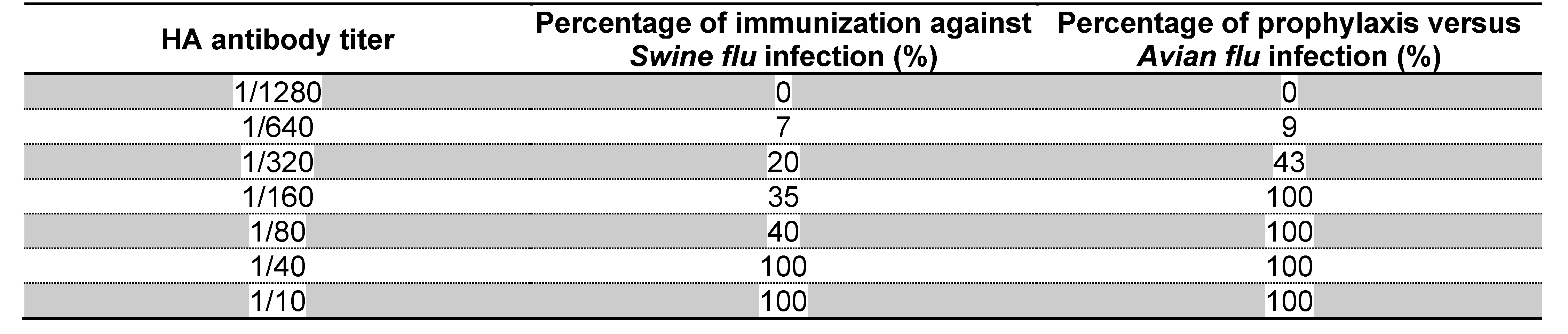
DISCUSSION
CONCLUSION
Funding
Data availability
Consent for publication
Consent to participate
Ethical consideration
Conflict of interest and competing interests
References
- Minozzi S, Lytras T, Gianola S, Gonzalez-Lorenzo M, Castellini G, Galli C, Cereda D, Bonovas S, Pariani E, Moja L. Comparative efficacy and safety of vaccines to prevent seasonal influenza: A systematic review and network meta-analysis. EClinicalMedicine. 2022 Mar 25;46:101331. [CrossRef] [PubMed] [PubMed Central]
- Veroniki AA, Thirugnanasampanthar SS, Konstantinidis M, Dourka J, Ghassemi M, Neupane D, Khan P, Nincic V, Corry M, Robson R, Parker A, Soobiah C, Sinilaite A, Doyon-Plourde P, Gil A, Siu W, Moqueet N, Stevens A, English K, Florez ID, Yepes-Nuñez JJ, Hutton B, Muller M, Moja L, Straus S, Tricco AC. Trivalent and quadrivalent seasonal influenza vaccine in adults aged 60 and older: a systematic review and network meta-analysis. BMJ Evid Based Med. 2024 Apr 10:bmjebm-2023-112767. [CrossRef] [PubMed]
- Demicheli V, Jefferson T, Di Pietrantonj C, Ferroni E, Thorning S, Thomas RE, Rivetti A. Vaccines for preventing influenza in the elderly. Cochrane Database Syst Rev. 2018 Feb 1;2(2):CD004876. [CrossRef] [PubMed] [PubMed Central]
- T, Rivetti A, Di Pietrantonj C, Demicheli V. Vaccines for preventing influenza in healthy children. Cochrane Database Syst Rev. 2018 Feb 1;2(2):CD004879. [CrossRef] [PubMed] [PubMed Central]
- de Fougerolles TR, Baïssas T, Perquier G, Vitoux O, Crépey P, Bartelt-Hofer J, Bricout H, Petitjean A. Public health and economic benefits of seasonal influenza vaccination in risk groups in France, Italy, Spain and the UK: state of play and perspectives. BMC Public Health. 2024 May 3;24(1):1222. [CrossRef] [PubMed] [PubMed Central]
- Ryan J, Zoellner Y, Gradl B, Palache B, Medema J. Establishing the health and economic impact of influenza vaccination within the European Union 25 countries. Vaccine. 2006 Nov 17;24(47-48):6812-22. [CrossRef] [PubMed]
- Müller D, Szucs TD. Influenza vaccination coverage rates in 5 European countries: a population-based cross-sectional analysis of the seasons 02/03, 03/04 and 04/05. Infection. 2007 Oct;35(5):308-19. [CrossRef] [PubMed]
- Ruef, C. Influenza vaccination in Europe--still a long way to go. Infection. 2007 Oct;35(5):299. [CrossRef] [PubMed]
- Influenza vaccine 2011-2012. Med Lett Drugs Ther. 2011 Oct 17;53(1375):81-3. [PubMed]
- Katayose M, Hosoya M, Haneda T, Yamaguchi H, Kawasaki Y, Sato M, Wright PF. The effectiveness of trivalent inactivated influenza vaccine in children over six consecutive influenza seasons. Vaccine. 2011 Feb 17;29(9):1844-9. [CrossRef] [PubMed]
- Heikkinen T, Heinonen S. Effectiveness and safety of influenza vaccination in children: European perspective. Vaccine. 2011 Oct 6;29(43):7529-34. [CrossRef] [PubMed]
- Wen S, Wu Z, Zhong S, Li M, Shu Y. Factors influencing the immunogenicity of influenza vaccines. Hum Vaccin Immunother. 2021 Aug 3;17(8):2706-2718. [CrossRef] [PubMed] [PubMed Central]
- Feldstein LR, Matrajt L, Elizabeth Halloran M, Keitel WA, Longini IM Jr; H5N1 Vaccine Working Group. Extrapolating theoretical efficacy of inactivated influenza A/H5N1 virus vaccine from human immunogenicity studies. Vaccine. 2016 Jul 19;34(33):3796-802. [CrossRef] [PubMed] [PubMed Central]
- Jackson LA, Campbell JD, Frey SE, Edwards KM, Keitel WA, Kotloff KL, Berry AA, Graham I, Atmar RL, Creech CB, Thomsen IP, Patel SM, Gutierrez AF, Anderson EL, El Sahly HM, Hill H, Noah DL, Bellamy AR. Effect of Varying Doses of a Monovalent H7N9 Influenza Vaccine With and Without AS03 and MF59 Adjuvants on Immune Response: A Randomized Clinical Trial. JAMA. 2015 Jul 21;314(3):237-46. [CrossRef] [PubMed]
- Mulligan MJ, Bernstein DI, Winokur P, Rupp R, Anderson E, Rouphael N, Dickey M, Stapleton JT, Edupuganti S, Spearman P, Ince D, Noah DL, Hill H, Bellamy AR; DMID 13-0032 H7N9 Vaccine Study Group. Serological responses to an avian influenza A/H7N9 vaccine mixed at the point-of-use with MF59 adjuvant: a randomized clinical trial. JAMA. 2014 Oct 8;312(14):1409-19. [CrossRef] [PubMed]
- Belshe RB, Frey SE, Graham IL, Anderson EL, Jackson LA, Spearman P, Edupuganti S, Mulligan MJ, Rouphael N, Winokur P, Dolor RJ, Woods CW, Walter EB, Chen WH, Turley C, Edwards KM, Creech CB, Hill H, Bellamy AR; National Institute of Allergy and Infectious Diseases–Funded Vaccine and Treatment Evaluation Units. Immunogenicity of avian influenza A/Anhui/01/2005(H5N1) vaccine with MF59 adjuvant: a randomized clinical trial. JAMA. 2014 Oct 8;312(14):1420-8. [CrossRef] [PubMed]
- Chada KE, Forshee R, Golding H, Anderson S, Yang H. A systematic review and meta-analysis of cross-reactivity of antibodies induced by oil-in-water emulsion adjuvanted influenza H5N1 virus monovalent vaccines. Vaccine. 2017 May 31;35(24):3162-3170. [CrossRef] [PubMed]
- Jordan K, Murchu EO, Comber L, Hawkshaw S, Marshall L, O'Neill M, Teljeur C, Harrington P, Carnahan A, Pérez-Martín JJ, Robertson AH, Johansen K, Jonge J, Krause T, Nicolay N, Nohynek H, Pavlopoulou I, Pebody R, Penttinen P, Soler-Soneira M, Wichmann O, Ryan M. Systematic review of the efficacy, effectiveness and safety of cell-based seasonal influenza vaccines for the prevention of laboratory-confirmed influenza in individuals ≥18 years of age. Rev Med Virol. 2023 May;33(3):e2332. [CrossRef] [PubMed]
- O Murchu E, Comber L, Jordan K, Hawkshaw S, Marshall L, O'Neill M, Ryan M, Teljeur C, Carnahan A, Pérez JJ, Robertson AH, Johansen K, Jonge J, Krause T, Nicolay N, Nohynek H, Pavlopoulou I, Pebody R,Penttinen P, Soler-Soneira M, Wichmann O, Harrington P. Systematic review of the efficacy, effectiveness and safety of MF59® adjuvanted seasonal influenza vaccines for the prevention of laboratory-confirmed influenza in individuals ≥18 years of age. Rev Med Virol. 2023 May;33(3):e2329. [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




