Submitted:
03 June 2024
Posted:
03 June 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Schiff Based Isatin-Thiosemicarbazones Synthesis
2.2.2. Preparations of Biodiesel-Diesel Blends
2.2.3. Differential Scanning Calorimetry (DSC)
2.2.4. Thermogravimetric Analysis (TGA)
2.2.5. Fourier Transform Infrared Spectroscopy (FT- IR)
2.2.6. DPPH. Method for Antioxidant Activity
2.2.7. Kinetics of Isatin-Thiosemicarbazone Derivatives
3. Results and Discussion
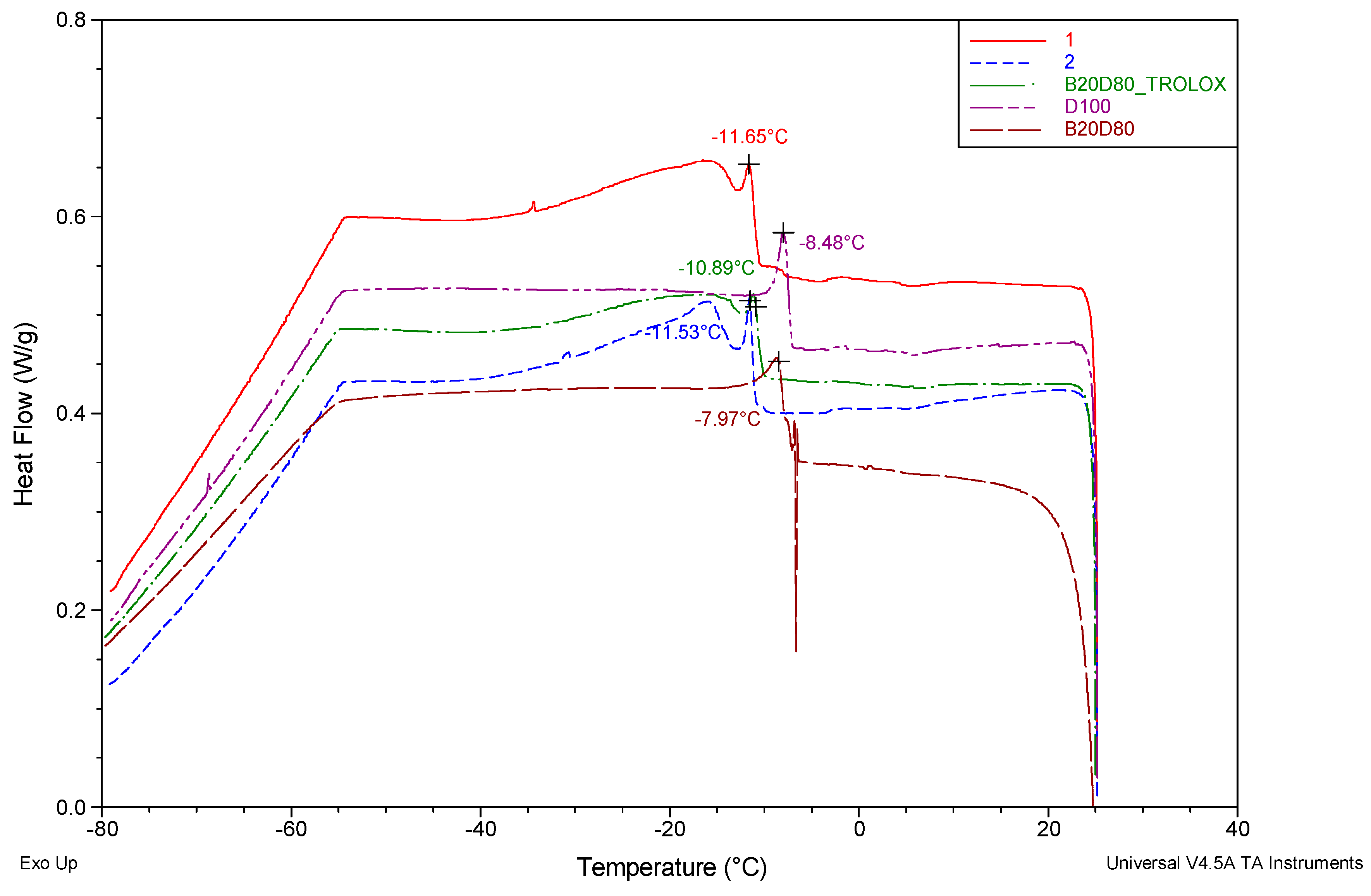
3.1. Differential Scanning Calorimetry (DSC)
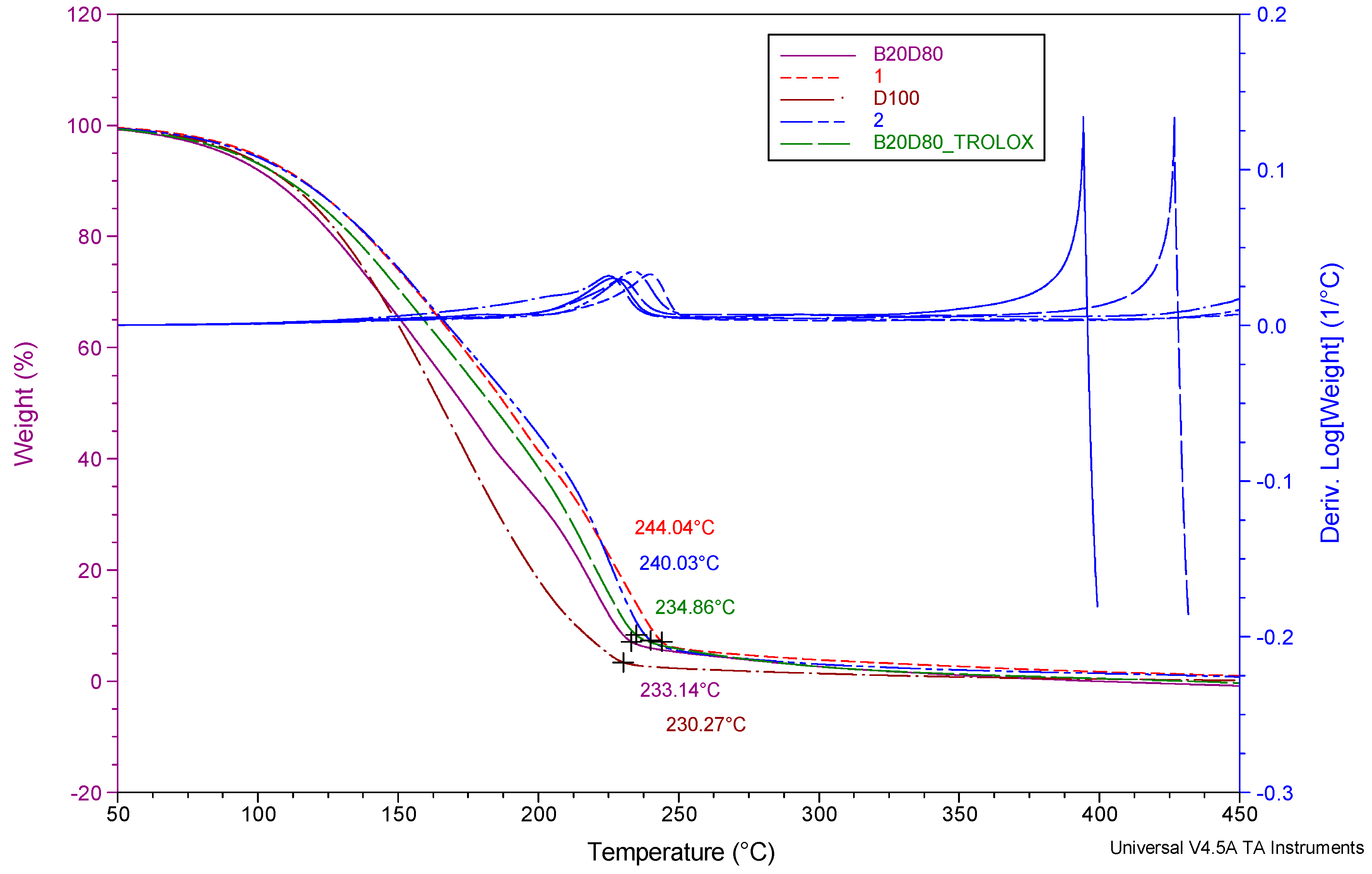
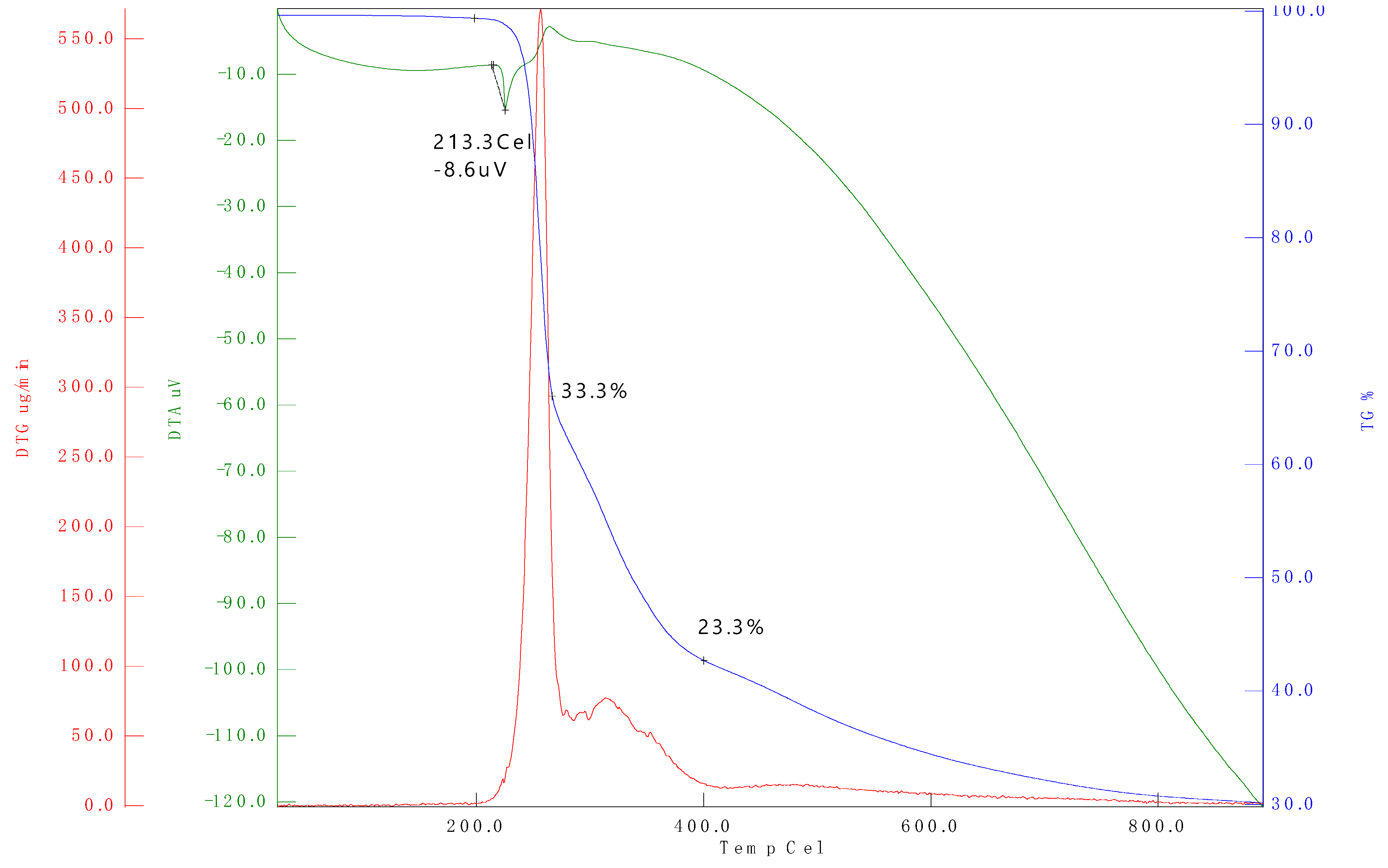
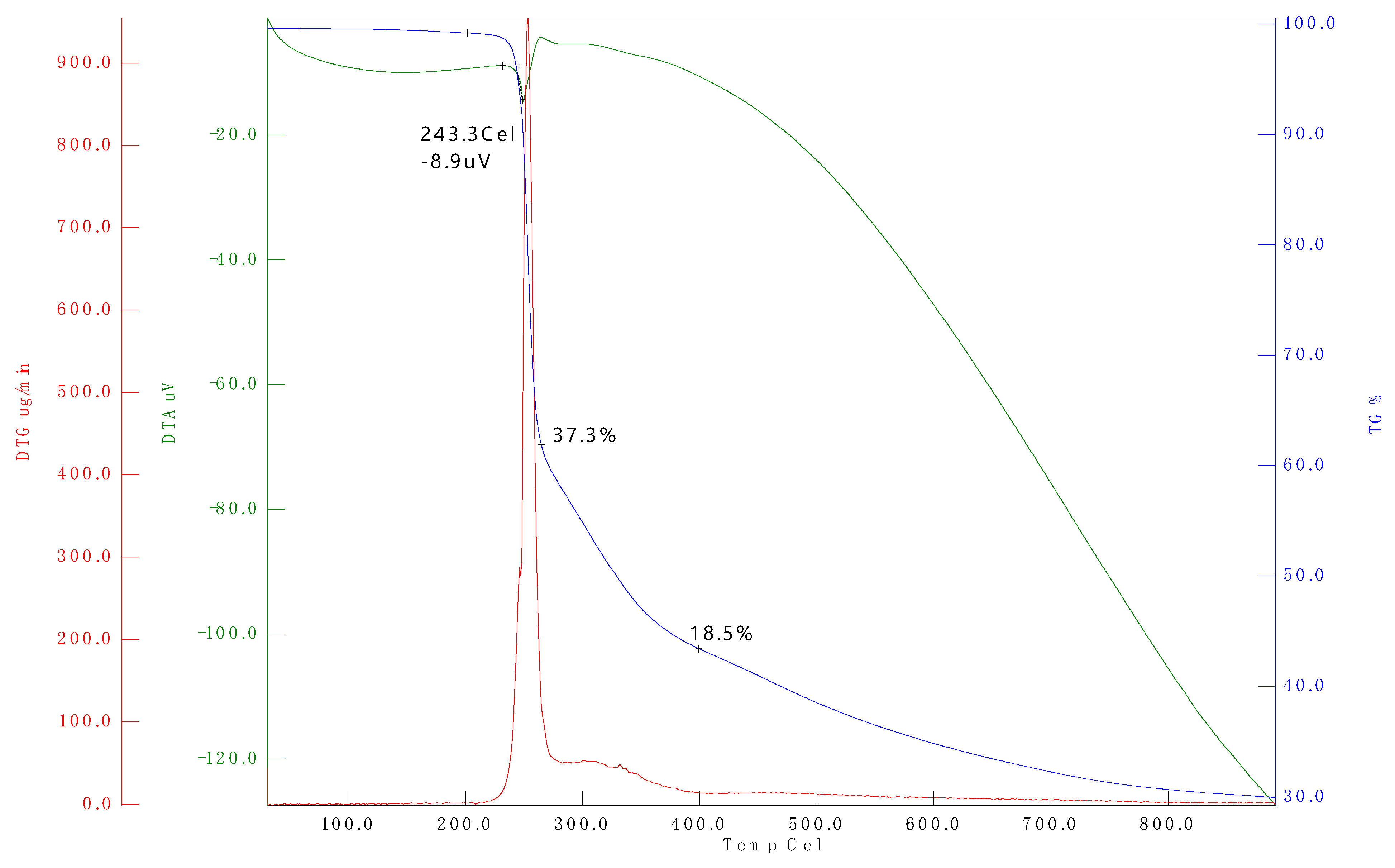
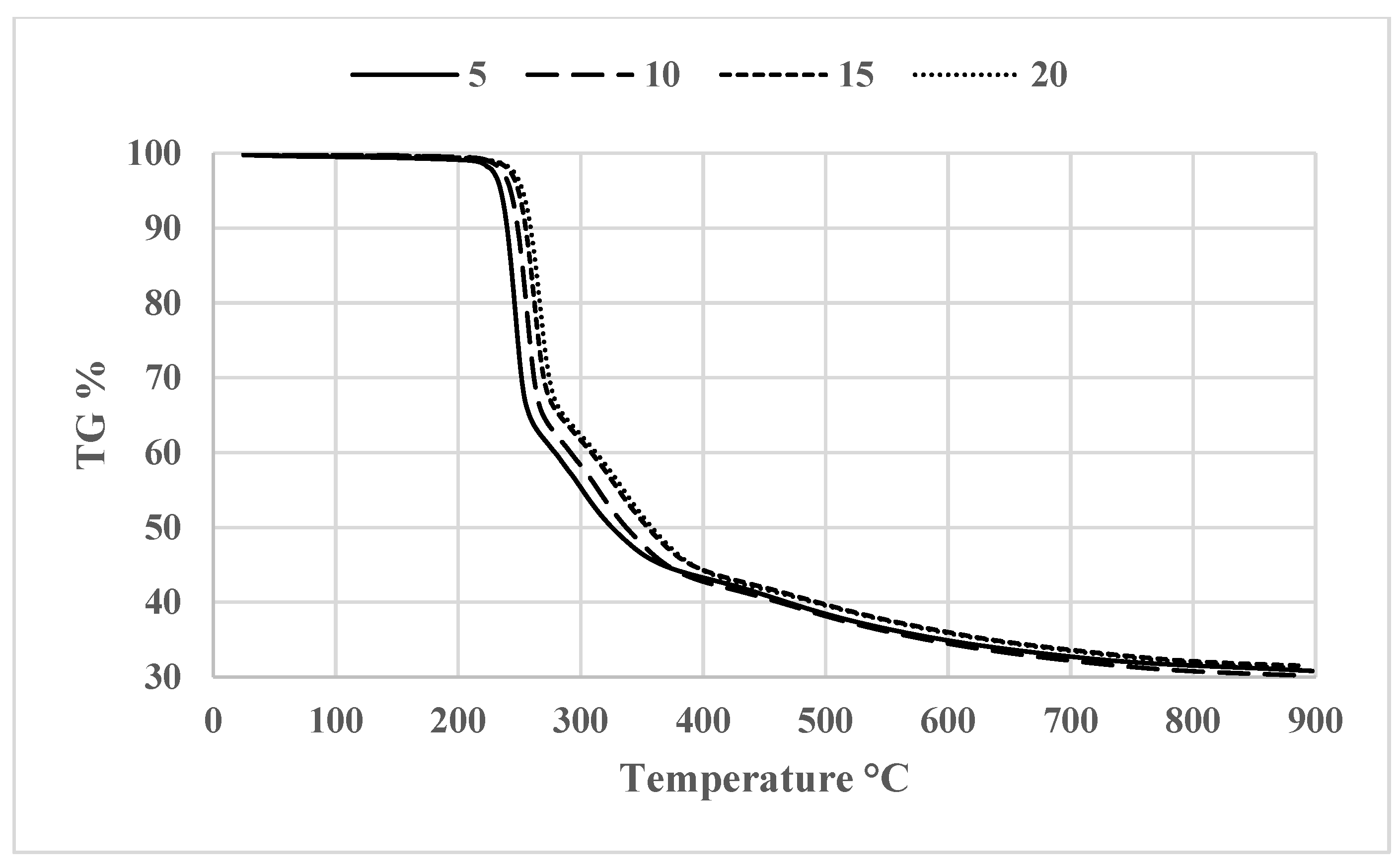
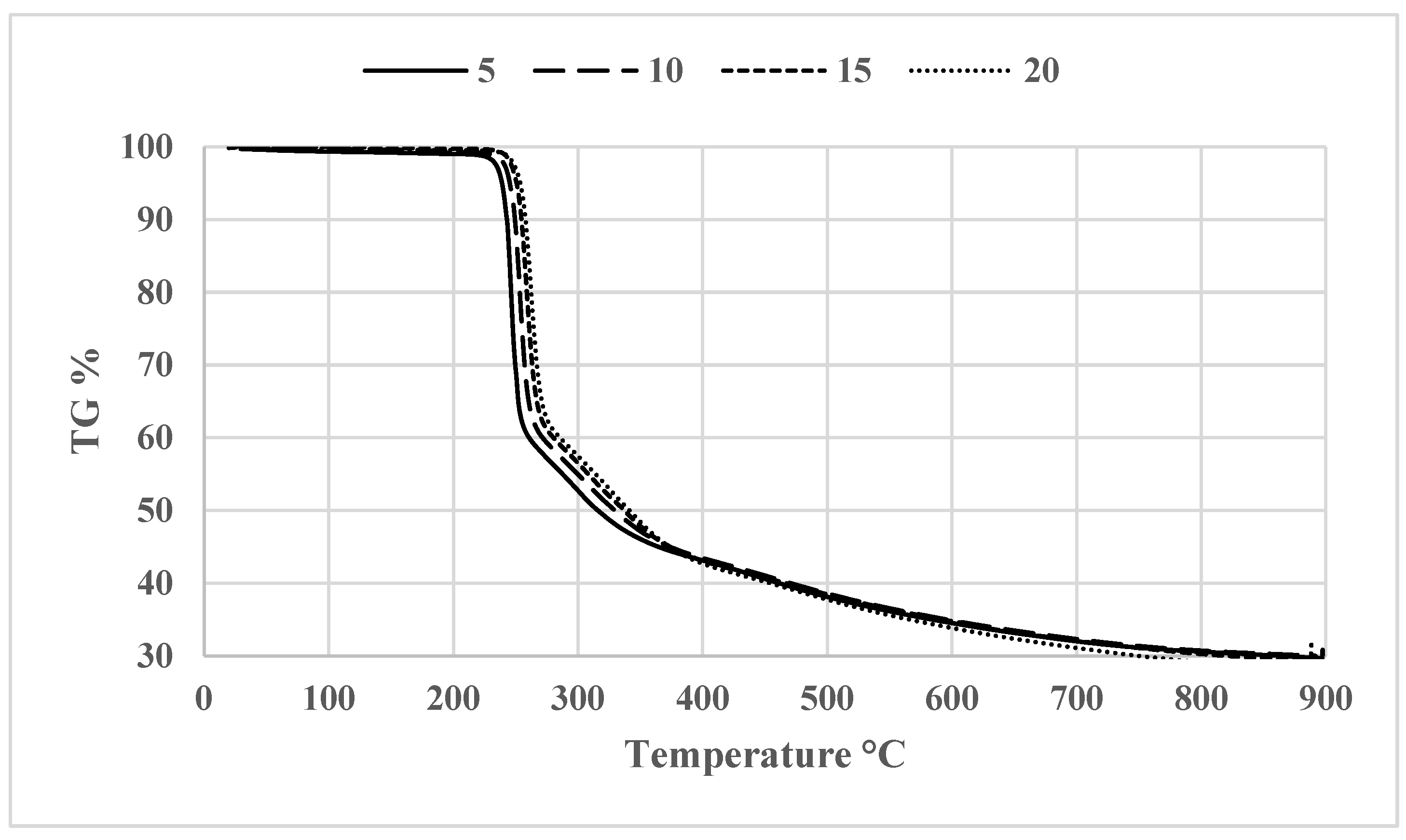
3.2. Thermogravimetric Analysis (TGA)
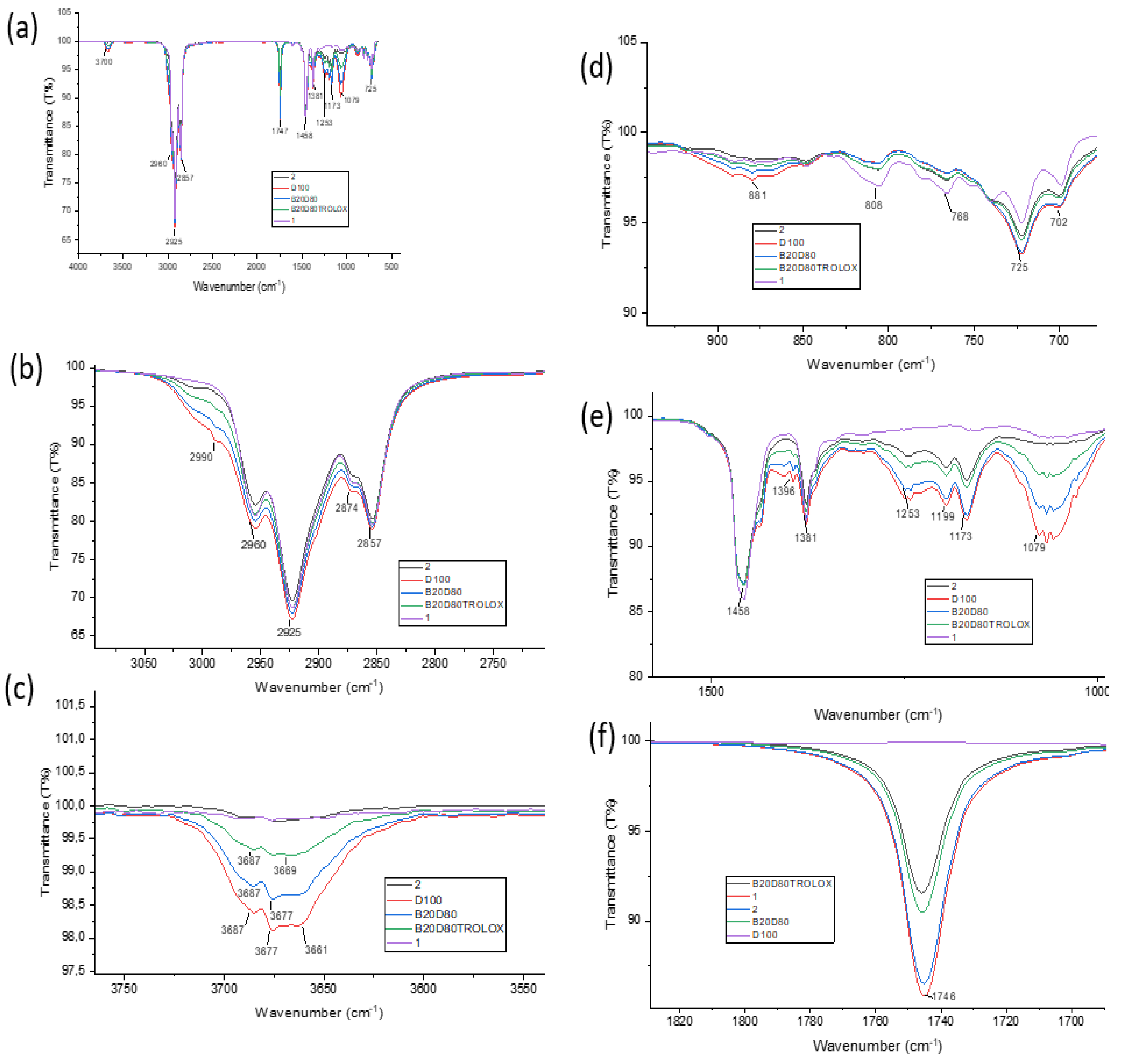
3.3. Fourier Transform Infrared Spectroscopy (FT- IR)
3.4. DPPH. Free Radical Scavenger Effect for Isatin-Thiosemicarbazones
3.5. Thermal Characterization of Isatin-Thiosemicarbazones
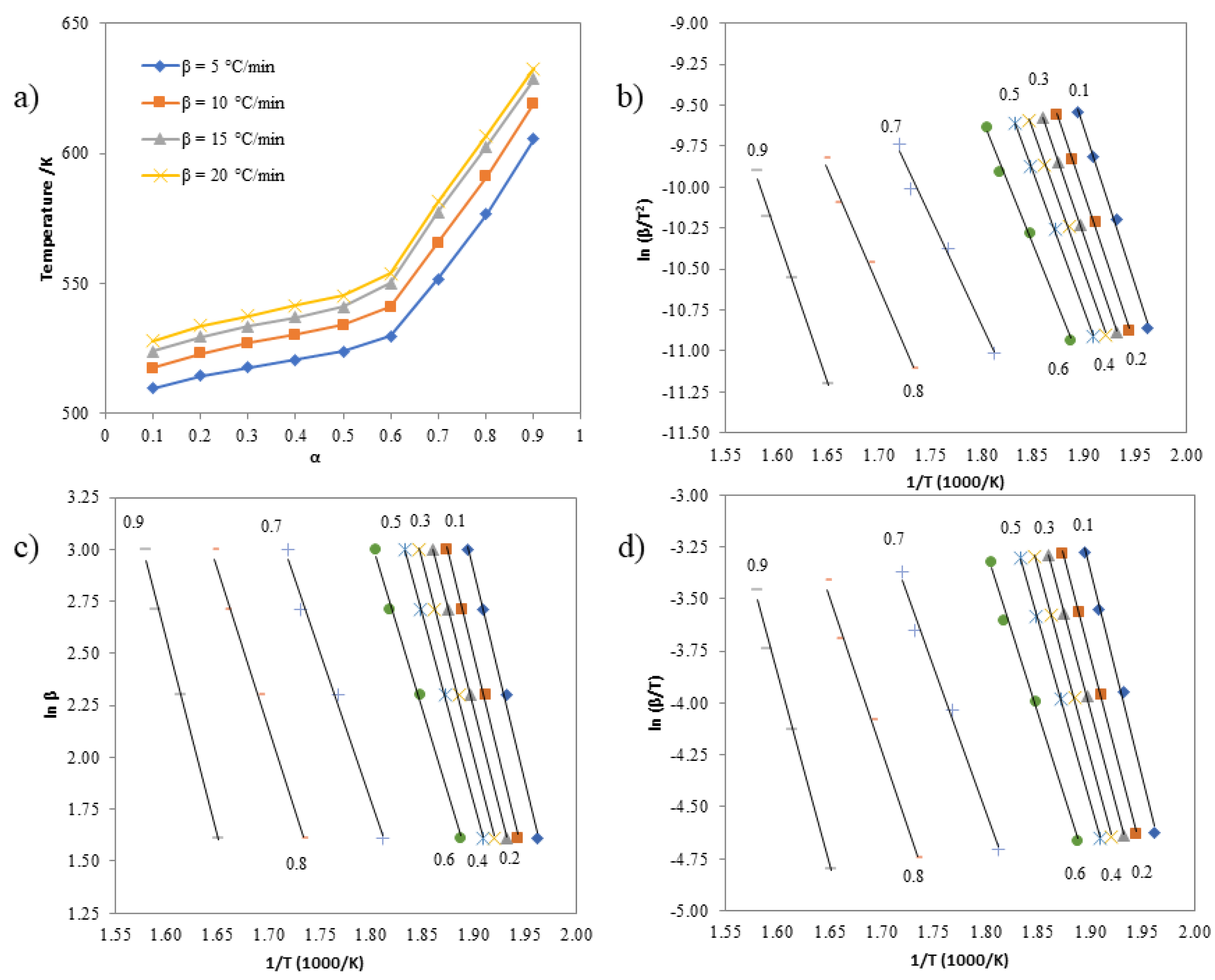
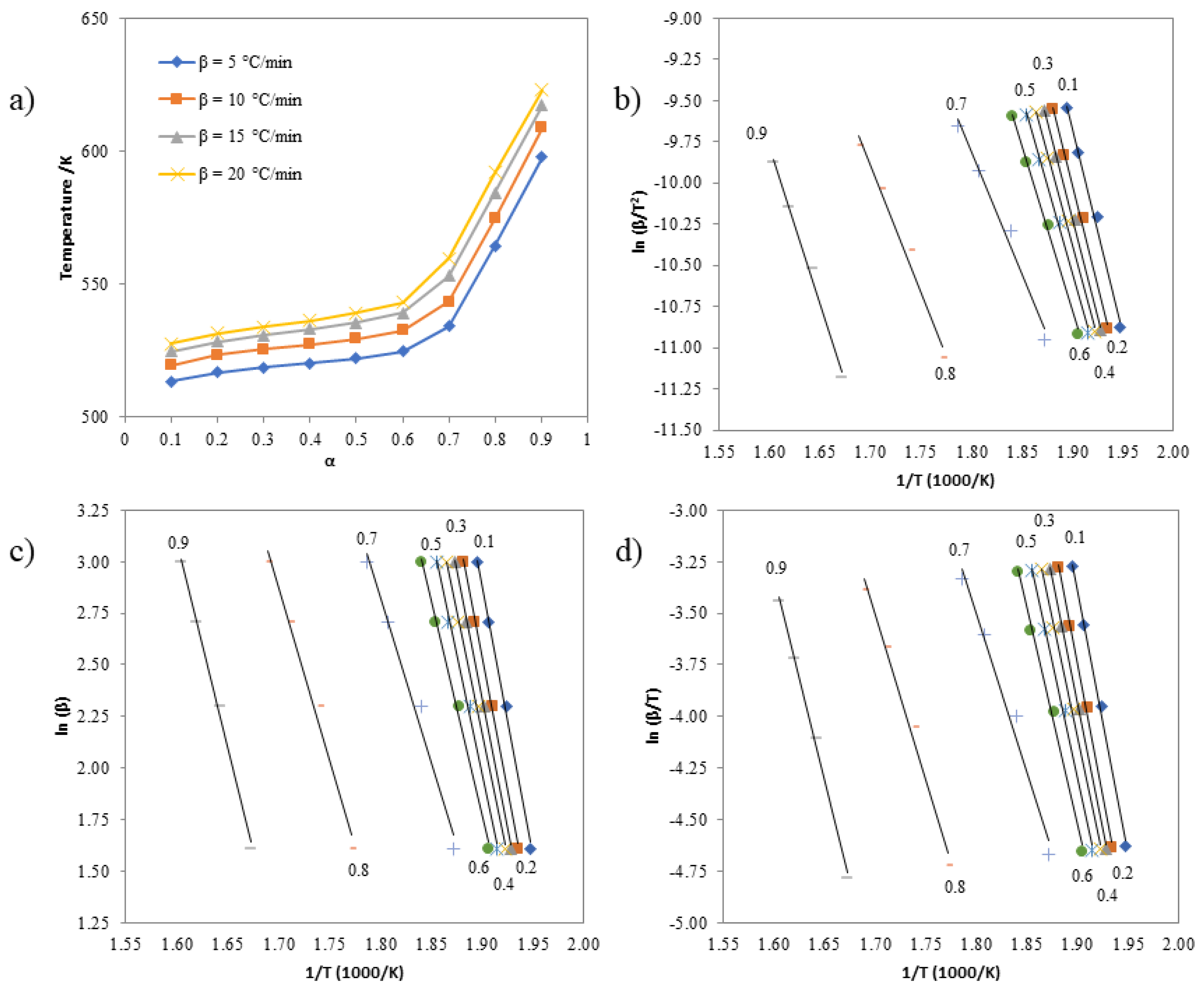
3.6. Investigating the Kinetics of Isatin-Thiosemicarbazones
5. Conclusions
6. Patents
Author Contributions
Funding
Data Availability Statement
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Atabani, A.E.; Silitonga, A.S.; Badruddin, I.A.; Mahlia, T.M.I.; Masjuki, H.H.; Mekhilef, S. A Comprehensive Review on Biodiesel as an Alternative Energy Resource and Its Characteristics. Renewable and Sustainable Energy Reviews 2012, 16, 2070–2093. [Google Scholar] [CrossRef]
- Srivastava, A.; Prasad, R. Triglycerides-Based Diesel Fuels. Renewable & sustainable energy reviews 2000, 4, 111–133. [Google Scholar] [CrossRef]
- Hosseinzadeh-bandbafha, H.; Kumar, D.; Singh, B.; Shahbeig, H. Biodiesel Antioxidants and Their Impact on the Behavior of Diesel Engines : A Comprehensive Review. Fuel Processing Technology 2022, 232, 107264. [Google Scholar] [CrossRef]
- Ravi Krishna, E.; Muralidhar Reddy, P.; Sarangapani, M.; Hanmanthu, G.; Geeta, B.; Shoba Rani, K.; Ravinder, V. Synthesis of N4 Donor Macrocyclic Schiff Base Ligands and Their Ru (II), Pd (II), Pt (II) Metal Complexes for Biological Studies and Catalytic Oxidation of Didanosine in Pharmaceuticals. Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy 2012, 97, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Kumar, V.; Sham, R. Stability of Biodiesel – A Review. Renewable and Sustainable Energy Reviews 2016, 62, 866–881. [Google Scholar] [CrossRef]
- Fang, H.L.; McCormick, R.L. Spectroscopic Study of Biodiesel Degradation Pathways. SAE Technical Papers 2006, 776–790. [Google Scholar] [CrossRef]
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural Polyphenols: An Overview. International Journal of Food Properties 2017, 20, 1689–1699. [Google Scholar] [CrossRef]
- Rizwanul Fattah, I.M.; Masjuki, H.H.; Kalam, M.A.; Hazrat, M.A.; Masum, B.M.; Imtenan, S.; Ashraful, A.M. Effect of Antioxidants on Oxidation Stability of Biodiesel Derived from Vegetable and Animal Based Feedstocks. Renewable and Sustainable Energy Reviews 2014, 30, 356–370. [Google Scholar] [CrossRef]
- Pullen, J.; Saeed, K. An Overview of Biodiesel Oxidation Stability. Renewable and Sustainable Energy Reviews 2012, 16, 5924–5950. [Google Scholar] [CrossRef]
- Rashedul, H.K.; Masjuki, H.H.; Kalam, M.A.; Teoh, Y.H.; How, H.G.; Rizwanul Fattah, I.M. Effect of Antioxidant on the Oxidation Stability and Combustion-Performance-Emission Characteristics of a Diesel Engine Fueled with Diesel-Biodiesel Blend. Energy Conversion and Management 2015, 106, 849–858. [Google Scholar] [CrossRef]
- Uddin, M.N.; Ahmed, S.S.; Alam, S.M.R. REVIEW: Biomedical Applications of Schiff Base Metal Complexes. Journal of Coordination Chemistry 2020, 73, 3109–3149. [Google Scholar] [CrossRef]
- Koteswara Rao, N.S.R.R.M.M.; Ram Reddy, M.G. Studies on the Synthesis, Characterisation and Antimicrobial Activity of New Co(II), Ni(II) and Zn(II) Complexes of Schiff Base Derived from Ninhydrin and Glycine. Biology of Metals 1990, 3, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Kalem, E.; Ağar, E. Schiff Bazlarinin Biyolojik Aktivitesi Biological Activity of Schiff Bases Öz : Abstract . 2021, 8, 57–76. [Google Scholar]
- Chen, Y.; Mi, Y.; Li, Q.; Dong, F.; Guo, Z. Synthesis of Schiff Bases Modified Inulin Derivatives for Potential Antifungal and Antioxidant Applications. International Journal of Biological Macromolecules 2020, 143, 714–723. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Wang, R.; Komatsu, K.; Bonaz-Krause, P.; Zyrianov, Y.; McKenna, C.E.; Csipke, C.; Tokes, Z.A.; Lien, E.J. Synthesis, Biological Evaluation, and Quantitative Structure -Activity Relationship Analysis of New Schiff Bases of Hydroxysemicarbazide as Potential Antitumor Agents. Journal of Medicinal Chemistry 2002, 45, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Duff, B.; Reddy Thangella, V.; Creaven, B.S.; Walsh, M.; Egan, D.A. Anti-Cancer Activity and Mutagenic Potential of Novel Copper(II) Quinolinone Schiff Base Complexes in Hepatocarcinoma Cells. European Journal of Pharmacology 2012, 689, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Jarrahpour, A.; Sheikh, J.; Mounsi, I. El; Juneja, H.; Hadda, T. Ben Computational Evaluation and Experimental in Vitro Antibacterial, Antifungal and Antiviral Activity of Bis-Schiff Bases of Isatin and Its Derivatives. Medicinal Chemistry Research 2013, 22, 1203–1211. [Google Scholar] [CrossRef]
- Singh, K.; Raparia, S.; Surain, P. Co(II), Ni(II), Cu(II) and Zn(II) Complexes of 4-(4-Cyanobenzylideneamino)-3-Mercapto-5-Oxo-1,2,4-Triazine: Synthesis, Characterization and Biological Studies. Medicinal Chemistry Research 2015, 24, 2336–2346. [Google Scholar] [CrossRef]
- Tople, M.S.; Patel, N.B.; Patel, P.P.; Purohit, A.C.; Ahmad, I.; Patel, H. An in Silico-in Vitro Antimalarial and Antimicrobial Investigation of Newer 7-Chloroquinoline Based Schiff-Bases. Journal of Molecular Structure 2023, 1271, 134016. [Google Scholar] [CrossRef]
- Petrović, Z.D.; Orović, J.; Simijonović, D.; Petrović, V.P.; Marković, Z. Experimental and Theoretical Study of Antioxidative Properties of Some Salicylaldehyde and Vanillic Schiff Bases. RSC Advances 2015, 5, 24094–24100. [Google Scholar] [CrossRef]
- Bharti, S.K.; Nath, G.; Tilak, R.; Singh, S.K. Synthesis, Anti-Bacterial and Anti-Fungal Activities of Some Novel Schiff Bases Containing 2,4-Disubstituted Thiazole Ring. European Journal of Medicinal Chemistry 2010, 45, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Bal, T.R.; Anand, B.; Yogeeswari, P.; Sriram, D. Synthesis and Evaluation of Anti-HIV Activity of Isatin β-Thiosemicarbazone Derivatives. Bioorganic and Medicinal Chemistry Letters 2005, 15, 4451–4455. [Google Scholar] [CrossRef] [PubMed]
- Cvijetić, I.N.; Herlah, B.; Marinković, A.; Perdih, A.; Bjelogrlić, S.K. Phenotypic Discovery of Thiocarbohydrazone with Anticancer Properties and Catalytic Inhibition of Human DNA Topoisomerase IIα. Pharmaceuticals 2023, 16, 341. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, R.B.; de Souza-Fagundes, E.M.; Soares, R.P.P.; Andrade, A.A.; Krettli, A.U.; Zani, C.L. Synthesis and Antimalarial Activity of Semicarbazone and Thiosemicarbazone Derivatives. European Journal of Medicinal Chemistry 2008, 43, 1983–1988. [Google Scholar] [CrossRef] [PubMed]
- Fayed, E.A.; Ragab, A.; Ezz Eldin, R.R.; Bayoumi, A.H.; Ammar, Y.A. In Vivo Screening and Toxicity Studies of Indolinone Incorporated Thiosemicarbazone, Thiazole and Piperidinosulfonyl Moieties as Anticonvulsant Agents. Bioorganic Chemistry 2021, 116, 105300. [Google Scholar] [CrossRef] [PubMed]
- Jacob, Í.T.T.; Gomes, F.O.S.; de Miranda, M.D.S.; de Almeida, S.M.V.; da Cruz-Filho, I.J.; Peixoto, C.A.; da Silva, T.G.; Moreira, D.R.M.; de Melo, C.M.L.; de Oliveira, J.F.; et al. Anti-Inflammatory Activity of Novel Thiosemicarbazone Compounds Indole-Based as COX Inhibitors. Pharmacological Reports 2021, 73, 907–925. [Google Scholar] [CrossRef] [PubMed]
- Yakan, H. Preparation, Structure Elucidation, and Antioxidant Activity of New Bis(Thiosemicarbazone) Derivatives. Turkish Journal of Chemistry 2020, 44, 1085–1099. [Google Scholar] [CrossRef] [PubMed]
- Pelosi, G.; Bisceglie, F.; Bignami, F.; Ronzi, P.; Schiavone, P.; Re, M.C.; Casoli, C.; Pilotti, E. Antiretroviral Activity of Thiosemicarbazone Metal Complexes. Journal of Medicinal Chemistry 2010, 53, 8765–8769. [Google Scholar] [CrossRef] [PubMed]
- Yakan, H.; Muğlu, H.; Türkeş, C.; Demir, Y.; Erdoğan, M.; Çavuş, M.S.; Beydemir, Ş. A Novel Series of Thiosemicarbazone Hybrid Scaffolds: Design, Synthesis, DFT Studies, Metabolic Enzyme Inhibition Properties, and Molecular Docking Calculations. Journal of Molecular Structure 2023, 1280. [Google Scholar] [CrossRef]
- Qin, Y.; Xing, R.; Liu, S.; Li, K.; Meng, X.; Li, R.; Cui, J.; Li, B.; Li, P. Novel Thiosemicarbazone Chitosan Derivatives: Preparation, Characterization, and Antifungal Activity. Carbohydrate Polymers 2012, 87, 2664–2670. [Google Scholar] [CrossRef]
- Khan, S.A.; Kumar, P.; Joshi, R.; Iqbal, P.F.; Saleem, K. Synthesis and in Vitro Antibacterial Activity of New Steroidal Thiosemicarbazone Derivatives. European Journal of Medicinal Chemistry 2008, 43, 2029–2034. [Google Scholar] [CrossRef]
- Andreani, A.; Burnelli, S.; Granaiola, M.; Leoni, A.; Locatelli, A.; Morigi, R.; Rambaldi, M.; Varoli, L.; Cremonini, M.A.; Placucci, G.; et al. New Isatin Derivatives with Antioxidant Activity. European Journal of Medicinal Chemistry 2010, 45, 1374–1378. [Google Scholar] [CrossRef]
- Elsaman, T.; Mohamed, M.S.; Eltayib, E.M.; Abdel-aziz, H.A.; Abdalla, A.E.; Munir, M.U.; Mohamed, M.A. Isatin Derivatives as Broad-Spectrum Antiviral Agents: The Current Landscape. Medicinal Chemistry Research 2022, 31, 244–273. [Google Scholar] [CrossRef]
- Chohan, Z.H.; Pervez, H.; Rauf, A.; Khan, K.M.; Supuran, C.T. Isatin-Derived Antibacterial and Antifungal Compounds and Their Transition Metal Complexes. Journal of Enzyme Inhibition and Medicinal Chemistry 2004, 19, 417–423. [Google Scholar] [CrossRef]
- Guo, H. Isatin Derivatives and Their Anti-Bacterial Activities. European Journal of Medicinal Chemistry 2019, 164, 678–688. [Google Scholar] [CrossRef]
- Tangadanchu, V.K.R.; Sui, Y.F.; Zhou, C.H. Isatin-Derived Azoles as New Potential Antimicrobial Agents: Design, Synthesis and Biological Evaluation. Bioorganic and Medicinal Chemistry Letters 2021, 41, 128030. [Google Scholar] [CrossRef]
- Jiang, D.; Wang, G.Q.; Liu, X.; Zhang, Z.; Feng, L.S.; Liu, M.L. Isatin Derivatives with Potential Antitubercular Activities. Journal of Heterocyclic Chemistry 2018, 55, 1263–1279. [Google Scholar] [CrossRef]
- Nain, S. Recent Advancement in Synthesis of Isatin as Anticonvulsant Agents: A Review. Medicinal Chemistry 2014, 4. [Google Scholar] [CrossRef]
- Van Den Berg, R.; Haenen, G.R.M.M.; Van Den Berg, H.; Bast, A. Applicability of an Improved Trolox Equivalent Antioxidant Capacity (TEAC) Assay for Evaluation of Antioxidant Capacity Measurements of Mixtures. Food Chemistry 1999, 66, 511–517. [Google Scholar] [CrossRef]
- Lúcio, M.; Nunes, C.; Gaspar, D.; Ferreira, H.; Lima, J.L.F.C.; Reis, S. Antioxidant Activity of Vitamin E and Trolox: Understanding of the Factors That Govern Lipid Peroxidation Studies in Vitro. Food Biophysics 2009, 4, 312–320. [Google Scholar] [CrossRef]
- Volkan, Y.M. Investigation of The Evaluation of Diasporic Bauxite from Islahiye Region in Alumina Production, Sakarya University, 2022.
- Muğlu, H. Synthesis, Characterization, and Antioxidant Activity of Some New N 4-Arylsubstituted-5-Methoxyisatin-β-Thiosemicarbazone Derivatives. Research on Chemical Intermediates 2020, 46, 2083–2098. [Google Scholar] [CrossRef]
- Wang, X.; Dai, M.; Xie, Y.; Han, J.; Ma, Y.; Chen, C. Experimental Investigation of Evaporation Characteristics of Biodiesel-Diesel Blend Droplets with Carbon Nanotubes and Nanoceria as Nanoadditives. Applied Surface Science 2020, 505, 144186. [Google Scholar] [CrossRef]
- Dunn, R.O. Effect of Antioxidants on the Oxidative Stability of Methyl Soyate ( Biodiesel ) B. 2005, 86, 1071–1085. [Google Scholar] [CrossRef]
- Maru, M.M.; Lucchese, M.M.; Legnani, C.; Quirino, W.G.; Balbo, A.; Aranha, I.B.; Costa, L.T.; Vilani, C.; de Sena, L.Á.; Damasceno, J.C.; et al. Biodiesel Compatibility with Carbon Steel and HDPE Parts. Fuel Processing Technology 2009, 90, 1175–1182. [Google Scholar] [CrossRef]
- Aniza, R.; Chen, W.H.; Kwon, E.E.; Bach, Q.V.; Hoang, A.T. Lignocellulosic Biofuel Properties and Reactivity Analyzed by Thermogravimetric Analysis (TGA) toward Zero Carbon Scheme: A Critical Review. Energy Conversion and Management: X 2024, 22, 100538. [Google Scholar] [CrossRef]
- Kutuk, H.; Turkoz, N. Microwave-Assisted Synthesis of Disulfides. Phosphorus, Sulfur and Silicon and the Related Elements 2011, 186, 1515–1522. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT - Food Science and Technology 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Yakan, H.; Cakmak, S.; Kutuk, H.; Yenigun, S.; Ozen, T. Synthesis, Characterization, Antioxidant, and Antibacterial Activities of New 2,3-Dimethoxy and 3-Acetoxy-2-Methyl Benzamides. Research on Chemical Intermediates 2020, 46, 2767–2787. [Google Scholar] [CrossRef]
- Haji, I.; Yildız, K. Decomposition Kinetics of Diasporitic Bauxite from Gaziantep Region. In Proceedings of the 9th International Scientific Research Congress, Ankara, 2021; pp. 439–447. [Google Scholar]
- Angelopoulos, P, Samouhos, M, Taxiarchou, M. Thermal Decomposition Kinetics of Greek Diasporic Bauxite. Proceedings of OPMR 2016- Opportunities in Processing of Metal Resources in South East Europe 2016, 191–200.
- Küçük, F.; Yildiz, K. The Decomposition Kinetics of Mechanically Activated Alunite Ore in Air Atmosphere by Thermogravimetry. Thermochimica Acta 2006, 448, 107–110. [Google Scholar] [CrossRef]
- Çavuş, M.S.; Yakan, H.; Muğlu, H.; Bakır, T. Novel Carbohydrazones Including 5-Substituted Isatin: Synthesis, Characterization, and Quantum-Chemical Studies on the Relationship between Electronic and Antioxidant Properties. Journal of Physics and Chemistry of Solids 2020, 140. [Google Scholar] [CrossRef]
- Pitucha, M.; Ramos, P.; Wojtunik-Kulesza, K.; Głogowska, A.; Stefańska, J.; Kowalczuk, D.; Monika, D.; Augustynowicz-Kopeć, E. Thermal Analysis, Antimicrobial and Antioxidant Studies of Thiosemicarbazone Derivatives. Journal of Thermal Analysis and Calorimetry 2023, 148, 4223–4234. [Google Scholar] [CrossRef]
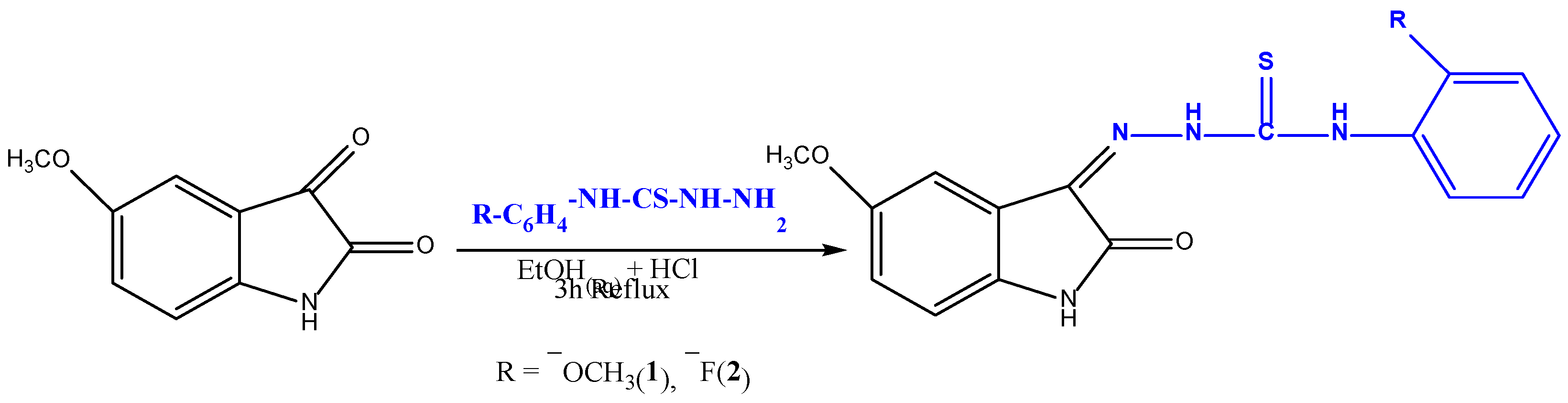
| Compound code | R | Molecular formula | Molecular weight | Melting point (°C) | Yield % |
|---|---|---|---|---|---|
| 1 | 2-OCH3 | C17H16N4O3S | 356.41 | 218–220 | 87 |
| 2 | 2-F | C16H13FN4O2S | 344.37 | 234–235 | 82 |
| Sample | Biodiesel (%) | Diesel (%) |
|---|---|---|
| D100 | - | 100 |
| B20D80 | 20 | 80 |
| B20D80TROLOX | 20 | 80 |
| B20D80_2 | 20 | 80 |
| B20D80_1 | 20 | 80 |
| Sample | Crystallizations onset temperature (℃) |
|---|---|
| D100 | 7.97 |
| B20D80 | 8.48 |
| B20D80TROLOX | 10.89 |
| B20D80_2 | 11.53 |
| B20D80_1 | 11.65 |
| Sample name | Temperature range (°C) | Max, degradation temperature (°C) (Tonset) |
Mass loss (%) |
|---|---|---|---|
| D100 | 25-250 | 99.36 | 11.52 |
| B20D80 | 25-250 | 99.12 | 7.72 |
| B20D80TROLOX | 25-250 | 99.27 | 7.71 |
| B20D80_2 | 25-250 | 99.24 | 8.94 |
| B20D80_1 | 25-250 | 98.56 | 10.20 |
| Compound | ν(O−H) | ν(C−H) Aromatic |
ν(C−H) Aliphatic |
ν(C=O) | ν(N−H) | ν(C−N) | ν(C−O) |
|---|---|---|---|---|---|---|---|
| B20D80_2 | – | 2925 | 2874 | 1746 | 1458 | 1199 | 1079 |
| B20D80_1 | – | 2925 | 2874 | 1746 | 1458 | 1173 | 1079 |
| B20D80 TROLOX | 3687 | 2925 | 2857 | 1746 | – | – | 1079 |
| B20D80 | 3687 | 2925 | 2857 | 1746 | – | – | 1079 |
| D100 | 3687 | 2925 | 2857 | 1746 | – | – | 1079 |
| α | Activation Energies (kJ mol-1) | ||
|---|---|---|---|
| Kissinger | Ozawa | Boswell | |
| 0.1 | 163.05 | 171.68 | 167.37 |
| 0.2 | 155.18 | 163.89 | 159.53 |
| 0.3 | 152.00 | 160.77 | 156.38 |
| 0.4 | 146.72 | 155.54 | 151.13 |
| 0.5 | 143.70 | 152.59 | 148.15 |
| 0.6 | 130.64 | 139.65 | 135.14 |
| 0.7 | 114.96 | 124.38 | 119.67 |
| 0.8 | 123.40 | 133.24 | 128.32 |
| 0.9 | 153.22 | 163.51 | 158.36 |
| α | Activation Energies (kJ mol-1) | ||
|---|---|---|---|
| Kissinger | Ozawa | Boswell | |
| 0.1 | 211.74 | 220.40 | 216.07 |
| 0.2 | 206.48 | 215.19 | 210.83 |
| 0.3 | 199.63 | 208.38 | 204.01 |
| 0.4 | 190.60 | 199.38 | 194.99 |
| 0.5 | 182.53 | 191.35 | 186.94 |
| 0.6 | 169.41 | 178.29 | 173.85 |
| 0.7 | 125.89 | 134.98 | 130.44 |
| 0.8 | 129.56 | 139.17 | 134.36 |
| 0.9 | 160.42 | 170.57 | 165.49 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




