Submitted:
30 May 2024
Posted:
31 May 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Collection of Experimental Chemical Mixtures Data from the Literature
Collection of Drug Combinations
Collection of ChemIDPlus Single Chemicals
Creation of Binary Mixture Dataset
Generating Virtual Mixtures: Assumptions and Methods
6. Hybrid Neural Network (HNN) Method for the Prediction of Chemical Mixture Toxicity
6.1. Dose-Dependent Relationship of the Chemical Mixtures Using the HNN
Molecular Structural Feature Descriptors Using SMILES of the Chemicals
SMILES Preprocessing
Descriptor Calculation
Binary Classification Criteria
- very highly toxic (<0.1 mg/L)
- highly toxic (0.1-1 mg/L)
- moderately toxic (>1-10 mg/L)
- slightly toxic (>10-100 mg/L)
- practically nontoxic (>100 mg/L)
Developing Binary and Multiclass Classification Models using other Machine Learning Methods
Developing Regression Models using other Machine Learning Methods
Ensemble Model
Robust Model Evaluation
Binary and Multiclass Classification Model Evaluation
Regression Model Evaluation
Compound Out
Reproducibility
Results and Discussion
- I.
- Dose-Dependent Toxicity Assessment of Chemical Mixtures using HNN and other Machine Learning Methods
Machine Learning Model Performance using Literature Derived Experimental Mixtures Data
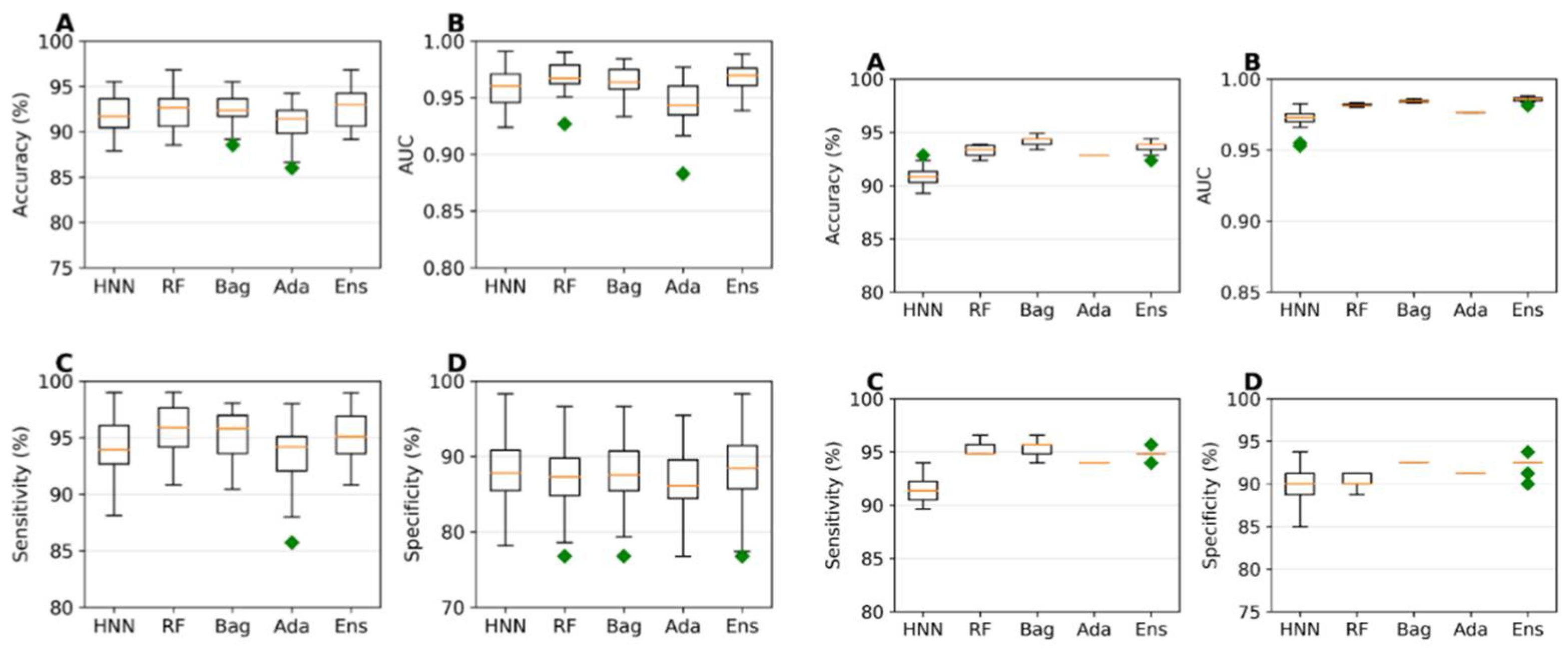
I.1. Mixture Toxicity Prediction Using Binary Classification
I.2. Mixture Toxicity Prediction Using Multiclass Classification
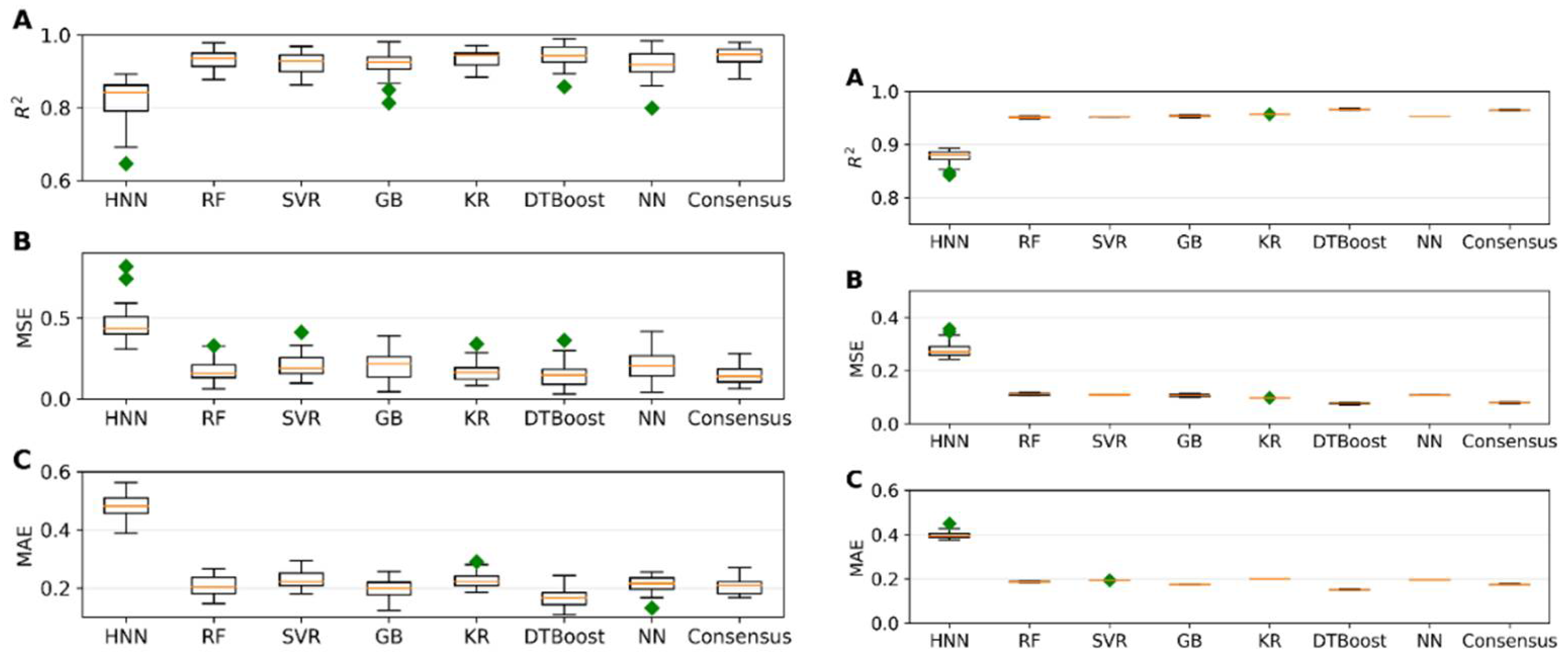
I.3. Mixture Toxicity Prediction Using Regression Models
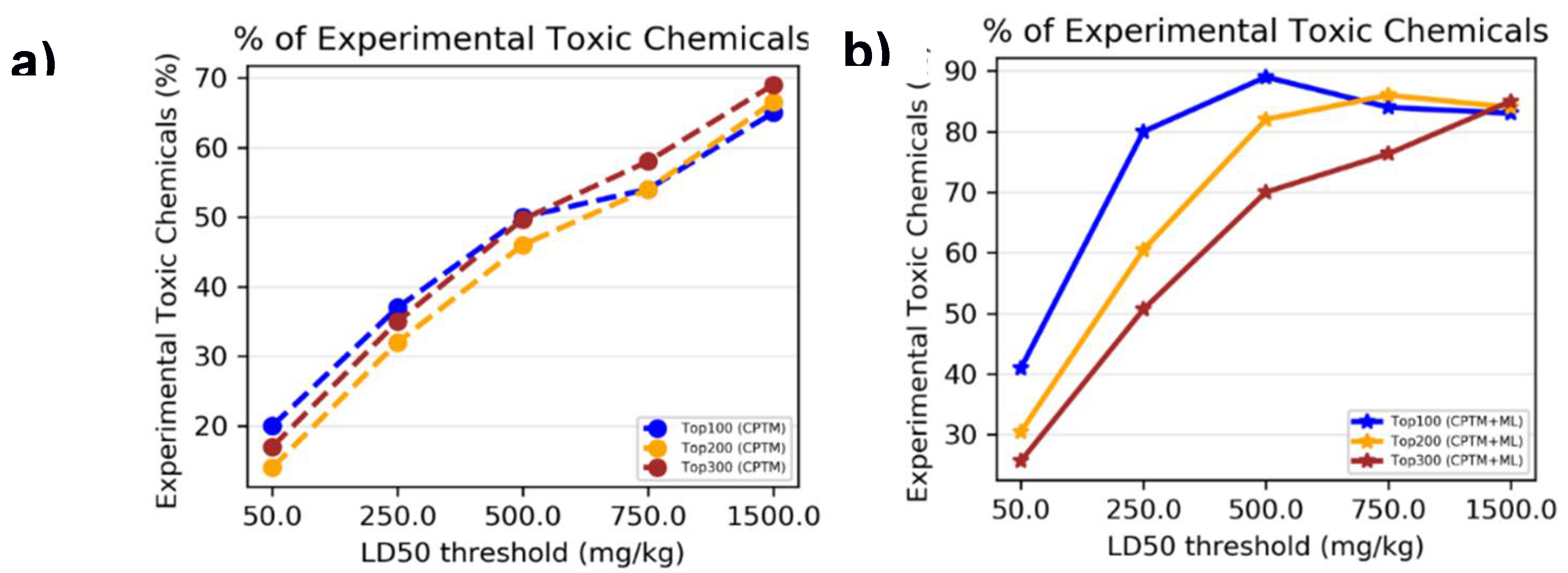
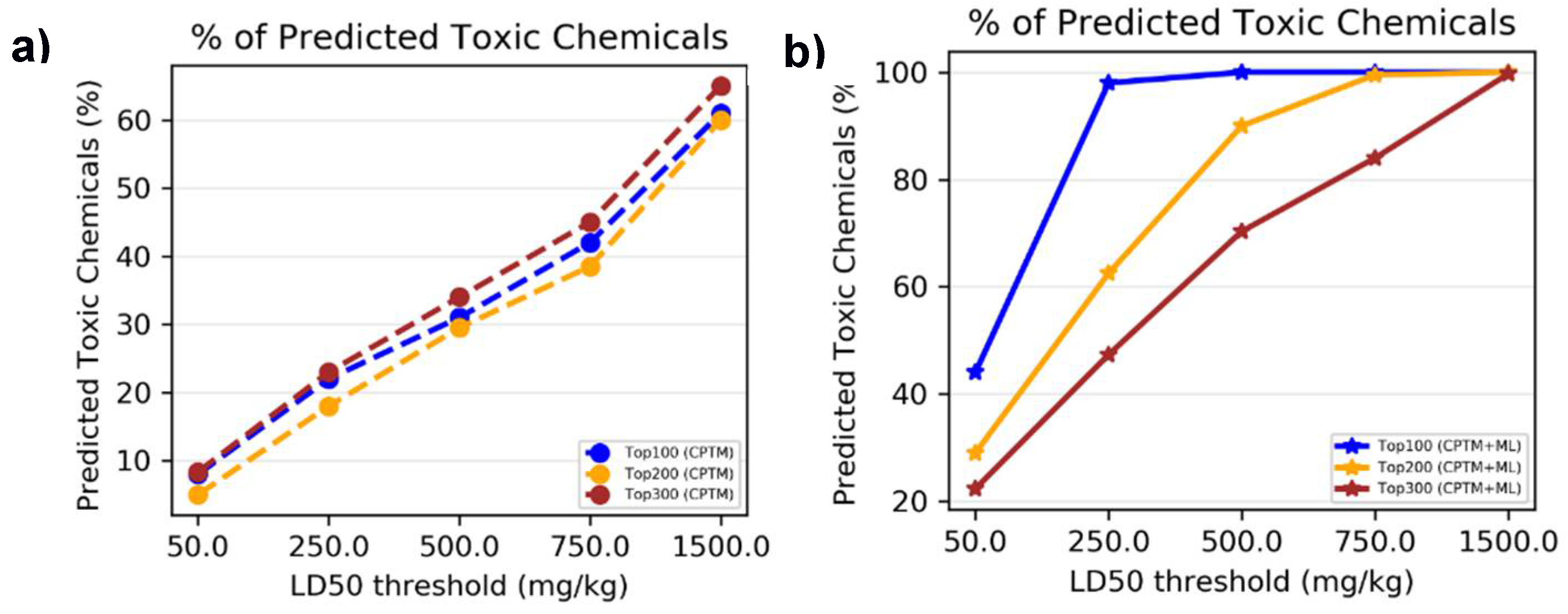
I.4. Comparison of Mixture toxicity Prediction with Existing Literature
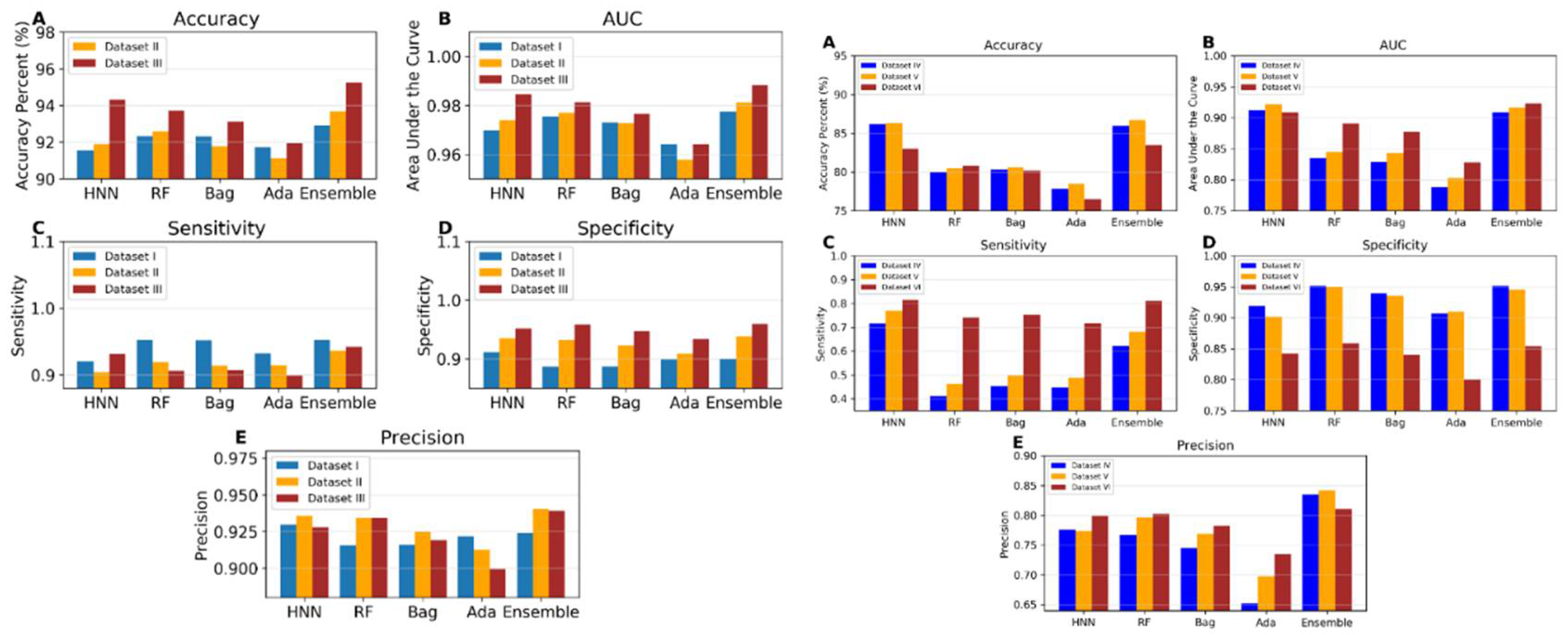
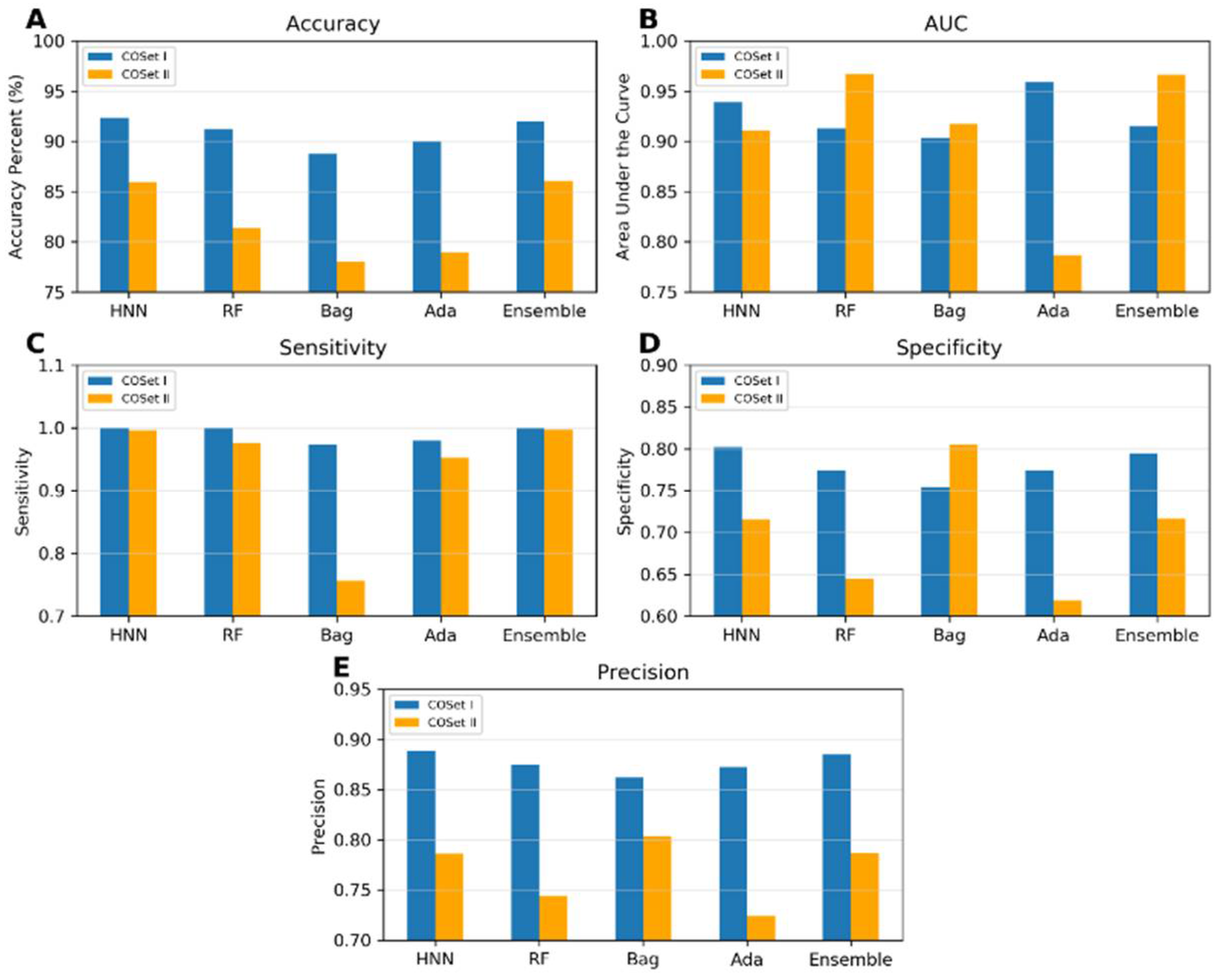
I.5a. Evaluation of Machine Learning Model Performance using Data Derived from Combinations of Experimental Mixtures and Drug Combination Datasets (Datasets I to III)
I.5b. Toxicity Prediction using Binary Classification with Virtual Mixtures and Drug Combination Datasets (Datasets IV to VI)
I.6. Compound Out Method
- I.
- AI-CPTM: the integration of the HNN Machine Learning Method with the CPTM Pathophysiology Method for the Assessment of Dose-Dependent Toxicity of Chemical Mixtures.
AI-CPTM Score Computations
II.1. Single chemical Toxicity – Binary Classification
II.1.1. Accuracy based on Experimental Toxicity
CPTM Performance without HNN Predictions Added
CPTM Performance with HNN Added (AI-CPTM)
II.1.2. Accuracy Based on HNN Predicted Toxicity
CPTM Performance without HNN Added
CPTM Performance with HNN Added
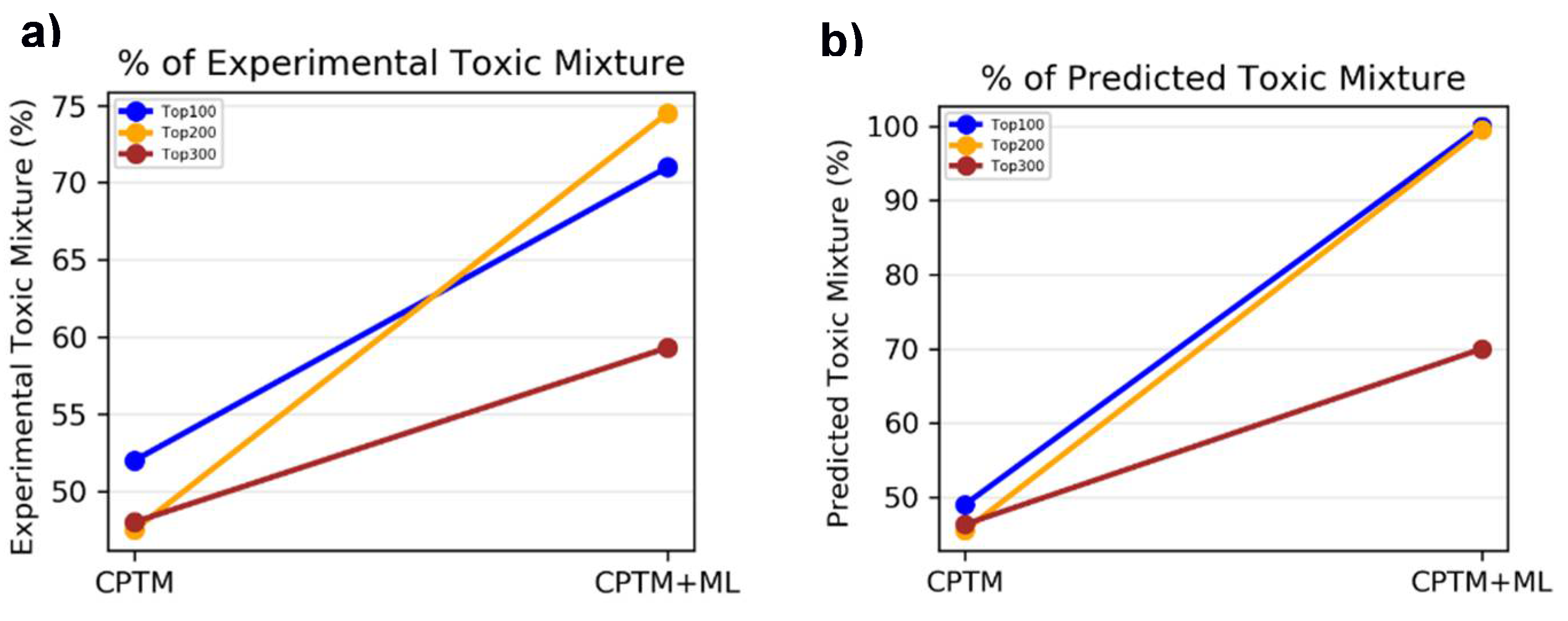
II.2. Chemical Mixture Toxicity – Binary Classification
II.2.1. Accuracy Based on Experimental Toxicity
CPTM Performance without HNN Predictions
CPTM Performance with HNN Added (AI-CPTM)
II.2.2. Accuracy Based on HNN Predicted Toxicity
CPTM Performance without HNN Predictions
CPTM Performance with HNN Added (AI-CPTM)
II.3. Experimental Validation
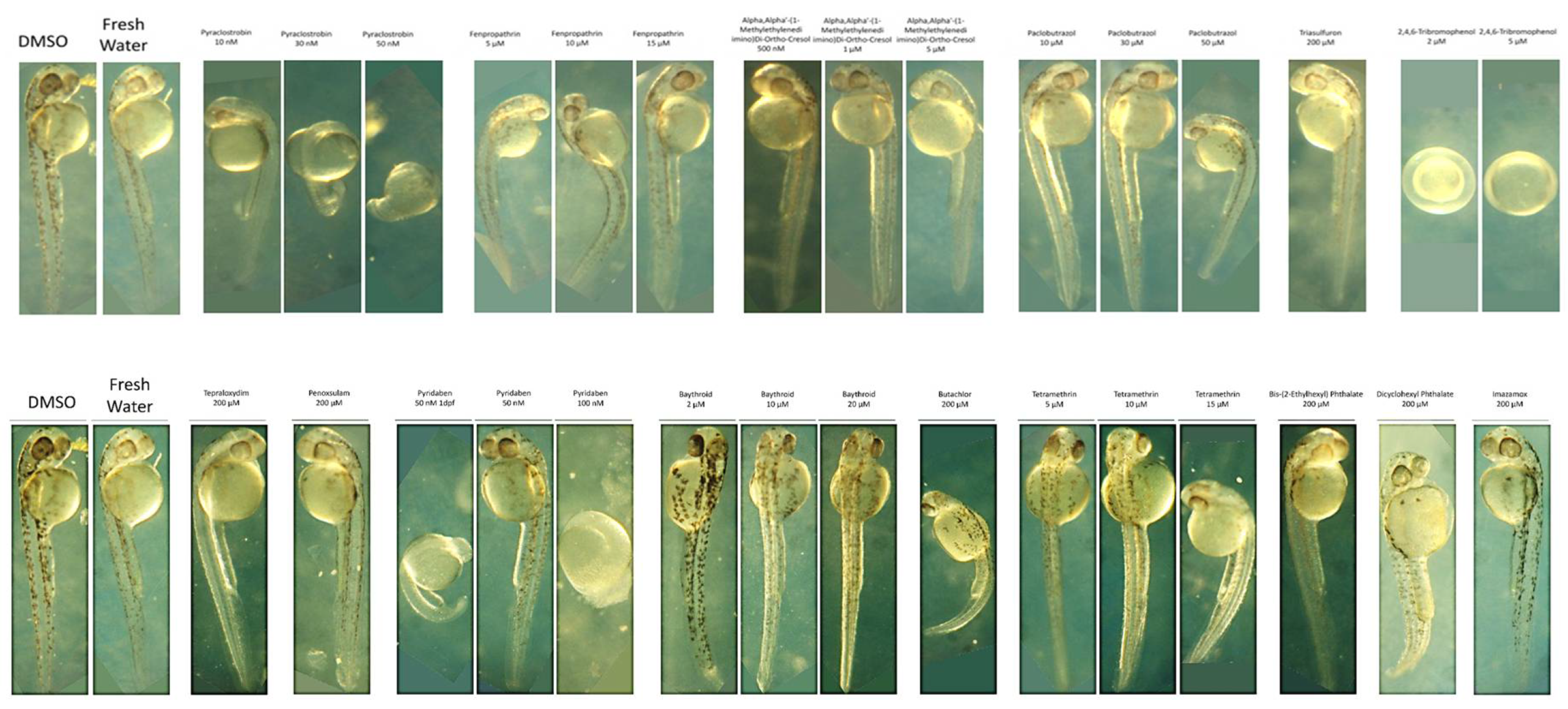
II.3.1. Zebrafish embryo toxicity studies of single chemicals
II.3.2. Zebrafish Embryo Toxicity of Chemical Mixtures and Chemical Interactions Analysis
II.3.2.A. Measurement of Mixture Toxicity in Zebrafish Models
Measurement of Developmental Toxicity
III.3.3. Comparison of zebrafish toxicity outcomes with the results from machine learning models for chemical mixtures
Determination of EC50 and Measurement of Mixed Chemical Interactions
Comparison Predicted vs Experimental Mixed Chemical Interactions
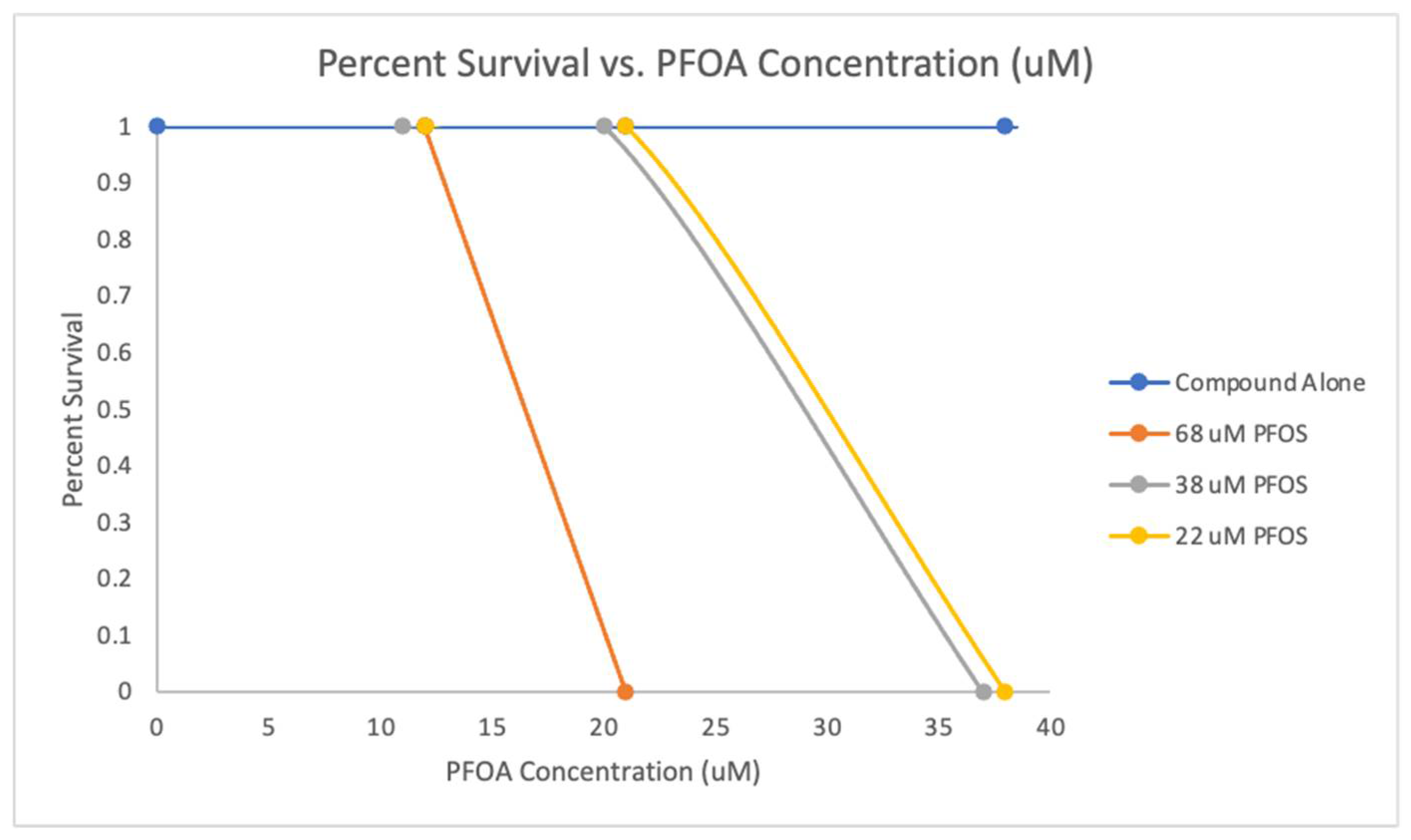
III.3.4. Toxicity Studies of Perfluorooctane Sulfonate (PFOS) and Perfluorooctanoic Acid (PFOA) chemical and their mixtures on Zebrafish Embryos
Zebrafish Survival Upon Exposure to PFOA and PFOS Mixtures
PFOS and PFOA Mixture Zebrafish Survival Assay
IV. Limitations
V. Future Studies
VI. Conclusions
Supplementary Materials
Acknowledgments
References
- Hayes, A. W. et al. New approaches to risk assessment of chemical mixtures. Toxicol. Res. Appl. 3, 2397847318820768 (2019).
- Chatterjee, M. & Roy, K. Recent Advances on Modelling the Toxicity of Environmental Pollutants for Risk Assessment: from Single Pollutants to Mixtures. Curr. Pollut. Rep. 8, 81–97 (2022).
- Cherkasov, A. et al. QSAR Modeling: Where Have You Been? Where Are You Going To? J. Med. Chem. 57, 4977–5010 (2014).
- Ankley, G. T. et al. Adverse outcome pathways: A conceptual framework to support ecotoxicology research and risk assessment. Environ. Toxicol. Chem. 29, 730–741 (2010).
- Nigsch, F., Macaluso, N. M., Mitchell, J. B. & Zmuidinavicius, D. Computational toxicology: an overview of the sources of data and of modelling methods. Expert Opin. Drug Metab. Toxicol. 5, 1–14 (2009).
- Altenburger, R. et al. Predictability of the toxicity of multiple chemical mixtures to Vibrio fischeri: Mixtures composed of similarly acting chemicals. Environ. Toxicol. Chem. 19, 2341–2347 (2000).
- Luan F, Xu X, Liu H, Cordeiro MNDS. 2013. Prediction of the baseline toxicity of non-polar narcotic chemical mixtures by QSAR approach. Chemosphere 90: 1980-1986.
- Qin, L.-T., Wu, J., Mo, L.-Y., Zeng, H.-H. & Liang, Y.-P. Linear regression model for predicting interactive mixture toxicity of pesticide and ionic liquid. Environ. Sci. Pollut. Res. 22, 12759–12768 (2015).
- Toropova, A. P. et al. CORAL: Models of toxicity of binary mixtures. Chemom. Intell. Lab. Syst. 119, 39–43 (2012).
- Netzeva, T. I. et al. Current Status of Methods for Defining the Applicability Domain of (Quantitative) Structure-Activity Relationships. Altern. Lab. Anim. 33, 155–173 (2005).
- Cash, G. G. Predicting Chemical Toxicity and Fate Edited by Mark T. D. Cronin and David J. Livingstone. CRC Press, Boca Raton, FL. 2004. xviii + 445 pp. 18 × 26 cm. ISBN 0-415-27180-0. $98.95. J. Med. Chem. 48, 4487–4488 (2005).
- Eriksson, L. et al. Methods for reliability and uncertainty assessment and for applicability evaluations of classification- and regression-based QSARs. Environ. Heal. Perspect. 111, 1361–1375 (2003).
- Duan, Q. et al. Machine learning for mixture toxicity analysis based on high-throughput printing technology. Talanta 207, 120299 (2020).
- Cipullo, S., Snapir, B., Prpich, G., Campo, P. & Coulon, F. Prediction of bioavailability and toxicity of complex chemical mixtures through machine learning models. Chemosphere 215, 388–395 (2019).
- Heys, K. A., Shore, R. F., Pereira, M. G., Jones, K. C. & Martin, F. L. Risk assessment of environmental mixture effects. RSC Adv. 6, 47844–47857 (2016).
- Rice, G. et al. An approach for assessing human exposures to chemical mixtures in the environment. Toxicol. Appl. Pharmacol. 233, 126–36 (2008).
- Khan, E. A. et al. Application of quantitative transcriptomics in evaluating the ex vivo effects of per- and polyfluoroalkyl substances on Atlantic cod (Gadus morhua) ovarian physiology. Sci. Total Environ. 755, 142904 (2021).
- Khan, E. A. et al. Application of quantitative transcriptomics in evaluating the ex vivo effects of per- and polyfluoroalkyl substances on Atlantic cod (Gadus morhua) ovarian physiology. Sci. Total Environ. 755, 142904 (2021).
- Taghizadeh, S. F. et al. Assessment of combined exposures to multiple chemicals: pesticides, metals, and polycyclic aromatic hydrocarbons levels in fig fruits. Int. J. Environ. Anal. Chem. 104, 827–846 (2024).
- Rider, C. V. & Simmons, J. E. Chemical Mixtures and Combined Chemical and Nonchemical Stressors. 1–12 (2018). [CrossRef]
- Bopp, S. K. et al. Current EU research activities on combined exposure to multiple chemicals. Environ. Int. 120, 544–562 (2018).
- Becker, R. A. et al. How well can carcinogenicity be predicted by high throughput “characteristics of carcinogens” mechanistic data? Regul. Toxicol. Pharmacol. 90, 185–196 (2017).
- Price, P. Interindividual Variation in Source-Specific Doses is a Determinant of Health Impacts of Combined Chemical Exposures. Risk Anal. 40, 2572–2583 (2020).
- Beronius, A. et al. Methodology for health risk assessment of combined exposures to multiple chemicals. Food Chem. Toxicol. 143, 111520 (2020).
- Rotter, S. et al. Overview on legislation and scientific approaches for risk assessment of combined exposure to multiple chemicals: the potential EuroMix contribution. Crit. Rev. Toxicol. 48, 796–814 (2018).
- Smalling, K. L. et al. Per- and polyfluoroalkyl substances (PFAS) in United States tapwater: Comparison of underserved private-well and public-supply exposures and associated health implications. Environ. Int. 178, 108033 (2023).
- Ding, N., Harlow, S. D., Randolph, J. F., Loch-Caruso, R. & Park, S. K. Perfluoroalkyl and polyfluoroalkyl substances (PFAS) and their effects on the ovary. Hum. Reprod. Updat. 26, dmaa018- (2020).
- Vieira, V. M. et al. Perfluorooctanoic Acid Exposure and Cancer Outcomes in a Contaminated Community: A Geographic Analysis. Environ. Heal. Perspect. 121, 318–323 (2013).
- Meek, M. E. B. et al. Risk assessment of combined exposure to multiple chemicals: A WHO/IPCS framework. Regul. Toxicol. Pharmacol. 60, S1–S14 (2011).
- Thayer, K. A., Heindel, J. J., Bucher, J. R. & Gallo, M. A. Role of Environmental Chemicals in Diabetes and Obesity: A National Toxicology Program Workshop Review. Environ. Heal. Perspect. 120, 779–789 (2012).
- Kumar, V., Boobis, A. R. & Moretto, A. Test and Risk Assessment Strategies for combined exposure to multiple chemicals. Food Chem. Toxicol. 144, 111607 (2020).
- Bopp, S., Kienzler, A. & Worth, A. The challenge of combined exposure to multiple chemicals: scientific and regulatory approaches to protect human health. Eur. J. Public Heal. 30, ckaa165.146 (2020).
- BLISS, C. I. THE TOXICITY OF POISONS APPLIED JOINTLY1. Ann. Appl. Biol. 26, 585–615 (1939).
- Hadrup, N. et al. Concentration Addition, Independent Action and Generalized Concentration Addition Models for Mixture Effect Prediction of Sex Hormone Synthesis In Vitro. PLoS ONE 8, e70490 (2013).
- Tanaka, Y. & Tada, M. Generalized concentration addition approach for predicting mixture toxicity. Environ. Toxicol. Chem. 36, 265–275 (2017).
- Løkke, H., Ragas, A. M. J. & Holmstrup, M. Tools and perspectives for assessing chemical mixtures and multiple stressors. Toxicology 313, 73–82 (2013).
- Sarigiannis, D. A. & Hansen, U. Considering the cumulative risk of mixtures of chemicals – A challenge for policy makers. Environ. Heal. 11, S18–S18 (2012).
- Qin, L.-T. et al. QSAR prediction of additive and non-additive mixture toxicities of antibiotics and pesticide. Chemosphere 198, 122–129 (2018).
- Wang D, Wu X, Lin Z, Ding Y. 2018. A comparative study on the binary and ternary mixture toxicity of anitbiotics towards three bacteria based on QSAR investigation. Environ Res 162: 127-134.
- Xu, S. & Nirmalakhandan, N. Use of QSAR models in predicting joint effects in multi-component mixtures of organic chemicals. Water Res. 32, 2391–2399 (1998).
- Rider, C. V. et al. Using the Key Characteristics of Carcinogens to Develop Research on Chemical Mixtures and Cancer. Environ. Heal. Perspect. 129, 035003 (2021).
- Arcos JC, Woo YT, Lai DY. 1988. Database on binary combination effects of chemical carcinogens. Environ Carcinog Rev 6:R5–R150.
- Rider, C. V., Dinse, G. E., Umbach, D. M., Simmons, J. E. & Hertzberg, R. C. Chemical Mixtures and Combined Chemical and Nonchemical Stressors. 235–270 (2018). [CrossRef]
- Conley, J. M. et al. Mixed “Antiandrogenic” Chemicals at Low Individual Doses Produce Reproductive Tract Malformations in the Male Rat. Toxicol. Sci. 164, 166–178 (2018).
- Limbu, S. & Dakshanamurthy, S. Predicting Dose-Dependent Carcinogenicity of Chemical Mixtures Using a Novel Hybrid Neural Network Framework and Mathematical Approach. Toxics 11, 605 (2023).
- Limbu, S. & Dakshanamurthy, S. A New Hybrid Neural Network Deep Learning Method for Protein–Ligand Binding Affinity Prediction and De Novo Drug Design. Int. J. Mol. Sci. 23, 13912 (2022).
- Limbu, S. & Dakshanamurthy, S. Predicting Chemical Carcinogens Using a Hybrid Neural Network Deep Learning Method. Sensors 22, 8185 (2022).
- Limbu, S., Zakka, C. & Dakshanamurthy, S. Predicting Dose-Range Chemical Toxicity using Novel Hybrid Deep Machine-Learning Method. Toxics 10, 706 (2022).
- Issa, N. T. et al. A novel chemo-phenotypic method identifies mixtures of salpn, vitamin D3, and pesticides involved in the development of colorectal and pancreatic cancer. Ecotoxicol. Environ. Saf. 233, 113330–113330 (2022).
- Schrödinger Release 2024-2: QikProp, Schrödinger, LLC, New York, NY, 2024.
- Moriwaki, H., Tian, Y.-S., Kawashita, N. & Takagi, T. Mordred: a molecular descriptor calculator. J. Cheminformatics 10, 4 (2018).
- Howard, G. J. & Webster, T. F. Generalized concentration addition: A method for examining mixtures containing partial agonists. J. Theor. Biol. 2009, 259, 469–477.
- Gaudin, T., Rotureau, P. & Fayet, G. Mixture Descriptors toward the Development of Quantitative Structure–Property Relationship Models for the Flash Points of Organic Mixtures. Ind. Eng. Chem. Res. 54, 6596–6604 (2015).
- Available online: https://www.epa.gov/pesticide-science-and-assessing-pesticide-risks/technical-overview-ecological-risk-assessment-0.

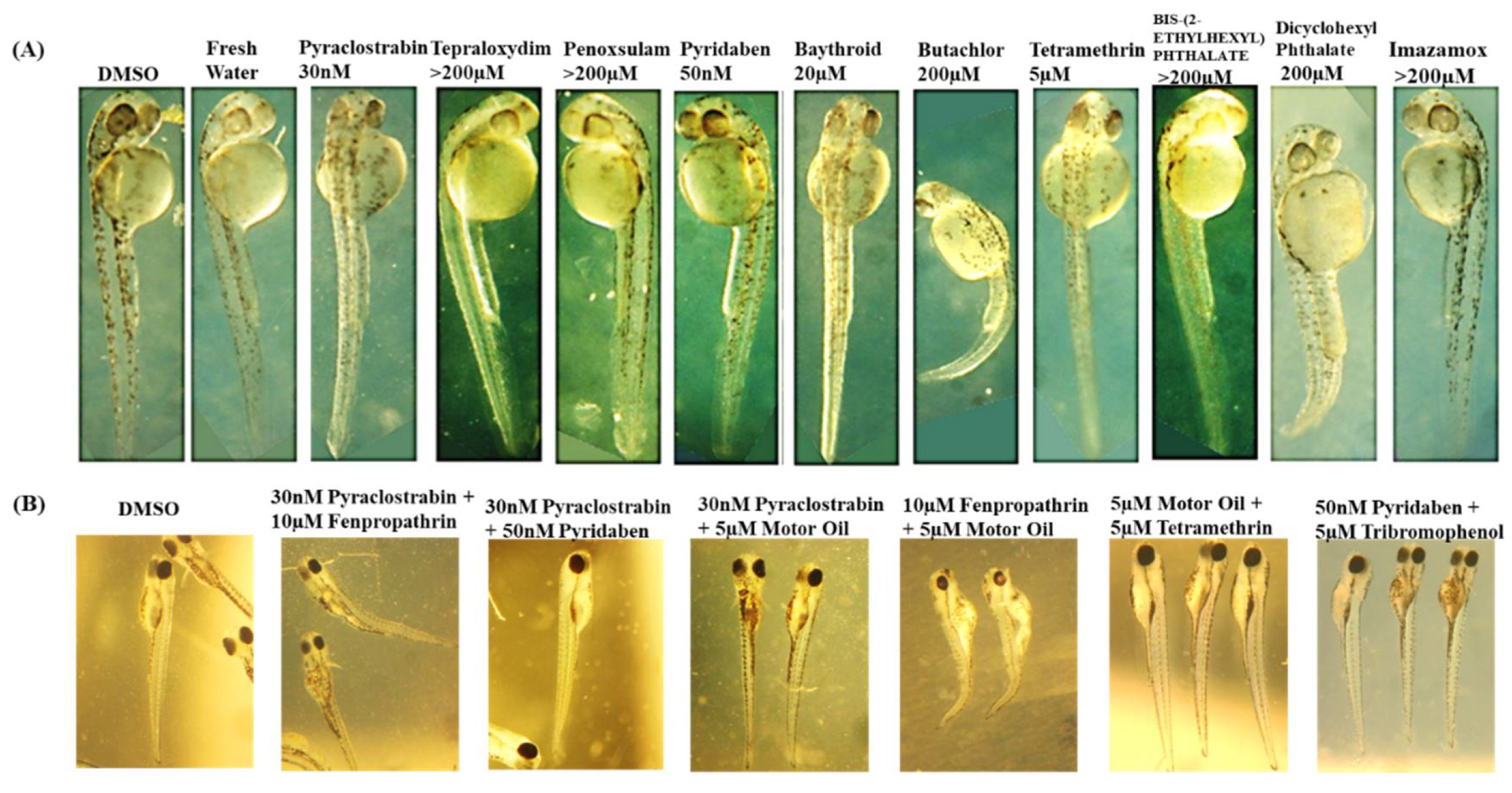
| Chemicals | CASRN |
|---|---|
| Pyraclostrobin | 175013-18-0 |
| Fenpropathrin | 39515-41-8 |
| Alpha, alpha’-(1-methylethylenediimino)di-ortho-cresol | 94-91-7 |
| Paclobutrazol | 76738-62-0 |
| 2,4,6 -Tribromophenol | 118-79-6 |
| Pyridaben | 96489-71-3 |
| Butachlor | 23184-66-9 |
| Tetramethrin | 7696-12-0 |
| Dicyclohexyl Phthalate (DCHP) | 84-61-7 |
| Pentadecafluorooctanoic acid (PFOA) | 335-67-1 |
| Prefluorooctane sulfonic acid (PFOS) | 1763-23-1 |
| Chemical 1 | Concentration LC50 (µM) | Chemical 2 | Concentration LC50 (µM) | AI-CPTM | AI-HNN | RF | Bagging | Adaboost | Experiment (zebrafish) | Mixture chemical interaction |
|---|---|---|---|---|---|---|---|---|---|---|
| Pyraclostrobin | 0.01 | Fenpropathrin | 5 | 0 | 0 | 1 | 1 | 1 | 0 | no interaction |
| Alpha, alpha’-(1-methylethylenediimino)di-ortho-cresol (motor fuel oil) | 2.5 | 0 | 0 | 1 | 1 | 1 | 0 | no interaction | ||
| Paclobutrazol | 50 | 1 | 1 | 1 | 1 | 1 | 0 | inconclusive | ||
| 2,4,6 -Tribromophenol | 2 | 1 | 1 | 1 | 1 | 1 | 1 | inconclusive | ||
| Pyridaben | 0.05 | 0 | 1 | 1 | 1 | 1 | 0 | inconclusive | ||
| Butachlor | 100 | 1 | 1 | 0 | 0 | 0 | 1 | no interaction | ||
| Tetramethrin | 5 | 1 | 0 | 0 | 1 | 1 | 1 | inconclusive | ||
| Dicyclohexyl Phthalate (DCHP) | 100 | 1 | 0 | 1 | 1 | 0 | 1 | inconclusive | ||
| Fenpropathrin | 5 | Alpha, alpha’-(1-methylethylenediimino)di-ortho-cresol | 2.5 | 1 | 1 | 1 | 0 | 1 | 1 | additive |
| Paclobutrazol | 50 | 1 | 1 | 1 | 1 | 1 | 1 | additive | ||
| 2,4,6 -Tribromophenol | 2 | 1 | 1 | 1 | 1 | 1 | 1 | additive | ||
| Pyridaben Pestanal | 0.05 | 1 | 1 | 1 | 1 | 1 | 0 | no interaction | ||
| Butachlor | 100 | 1 | 1 | 0 | 0 | 0 | 1 | additive | ||
| Tetramethrin | 5 | 1 | 1 | 1 | 1 | 1 | 1 | synergistic | ||
| Dicyclohexyl Phthalate (DCHP) | 100 | 1 | 1 | 1 | 1 | 0 | 1 | synergistic | ||
| Alpha, alpha’-(1-methylethylenediimino)di-ortho-cresol (motor oil) | 2.5 | Paclobutrazol | 50 | 1 | 1 | 1 | 1 | 1 | 1 | additive |
| 2,4,6 -Tribromophenol | 2 | 1 | 1 | 1 | 1 | 1 | 1 | additive | ||
| Pyridaben Pestanal | 0.05 | 0 | 0 | 0 | 1 | 1 | 0 | inconclusive | ||
| Butachlor | 100 | 0 | 0 | 1 | 0 | 0 | 1 | additive | ||
| Tetramethrin | 5 | 0 | 0 | 0 | 0 | 1 | 0 | no interaction | ||
| Dicyclohexyl Phthalate (DCHP) | 100 | 0 | 0 | 0 | 0 | 0 | 0 | no interaction | ||
| Paclobutrazol | 50 | 2,4,6 -Tribromophenol | 2 | 0 | 0 | 1 | 1 | 1 | 0 | no interaction |
| Pyridaben | 0.05 | 1 | 1 | 1 | 1 | 1 | 1 | additive | ||
| Butachlor | 100 | 1 | 1 | 1 | 1 | 1 | 1 | synergistic | ||
| Tetramethrin | 5 | 0 | 0 | 1 | 0 | 1 | 0 | additive | ||
| Dicyclohexyl Phthalate (DCHP) | 100 | 1 | 1 | 1 | 0 | 1 | 0 | additive | ||
| 2,4,6 -Tribromophenol | 2 | Pyridaben Pestanal | 0.05 | 1 | 1 | 1 | 1 | 1 | 0 | no interaction |
| Butachlor | 100 | 1 | 1 | 0 | 0 | 0 | 1 | synergistic | ||
| Tetramethrin | 5 | 1 | 1 | 1 | 1 | 1 | 1 | additive | ||
| Dicyclohexyl Phthalate (DCHP) | 100 | 1 | 1 | 1 | 0 | 0 | 1 | no interaction | ||
| Pyridaben | 0.05 | Butachlor | 100 | 1 | 1 | 1 | 1 | 1 | 1 | synergistic |
| Tetramethrin | 5 | 1 | 1 | 1 | 1 | 0 | 0 | inconclusive | ||
| Dicyclohexyl Phthalate (DCHP) | 100 | 1 | 1 | 1 | 1 | 0 | 0 | inconclusive | ||
| Butachlor | 100 | Tetramethrin | 5 | 1 | 1 | 1 | 0 | 1 | 1 | synergistic |
| Dicyclohexyl Phthalate (DCHP) | 100 | 1 | 1 | 1 | 1 | 1 | 1 | synergistic | ||
| Tetramethrin | 5 | Dicyclohexyl Phthalate (DCHP) | 100 | 1 | 1 | 1 | 1 | 1 | 1 | inconclusive |
| Prefluorooctane sulfonic acid (PFOS) | 53 | Pentadecafluorooctanoic acid (PFOA) | 187.5 | 1 | 1 | 1 | 1 | 1 | 1 | synergistic |
| Compound | LD50 | EC50 |
|---|---|---|
| PFOS | 53 µM | 11 µM |
| PFOA | 187.5 µM | 29.5 µM |
| Concentration of PFOS Present (µM) | 68 µM PFOS | 38 µM PFOS | 22 µM PFOS |
| LD50 (added PFOA concentration (µM) | 16.5 µM PFOA | 29 µM PFOA | 29.5 µM PFOA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




