Submitted:
28 May 2024
Posted:
28 May 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Cell Culture
2.2. Regents and Antibodies
2.3. Western Blot (WB)
2.4. Construction of Stable MDH2 Knockdown Cell Line
2.5. Transient Transfection of Plasmid
2.6. Cell Proliferation Assay
2.7. Colony Formation Assay
2.8. Quantitative Real-Time PCR (RT-qPCR)
2.9. IP and Immunoblotting Analysis
2.10. Ubiquitination Assay
2.11. Cell Death Analysis
2.12. Cell Viability Assay
2.13. Intracellular ROS, Fe2+, and Lipid ROS Levels Detection
2.14. Statistical Analysis
3. Results
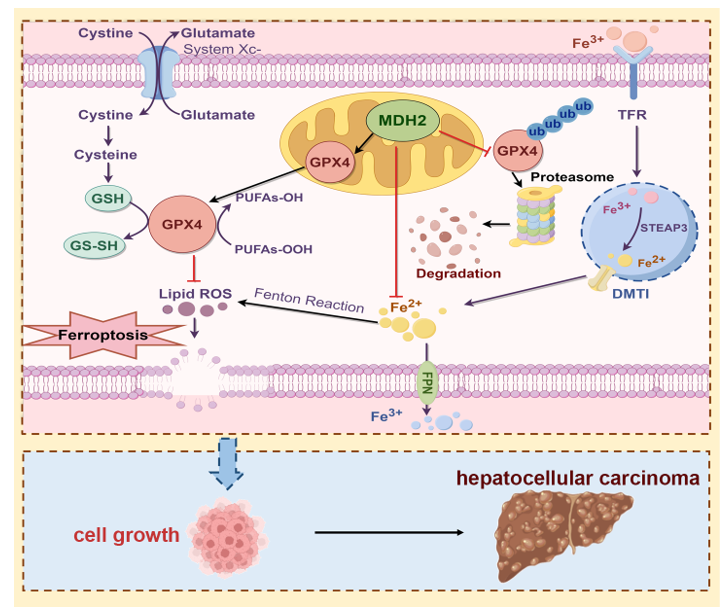
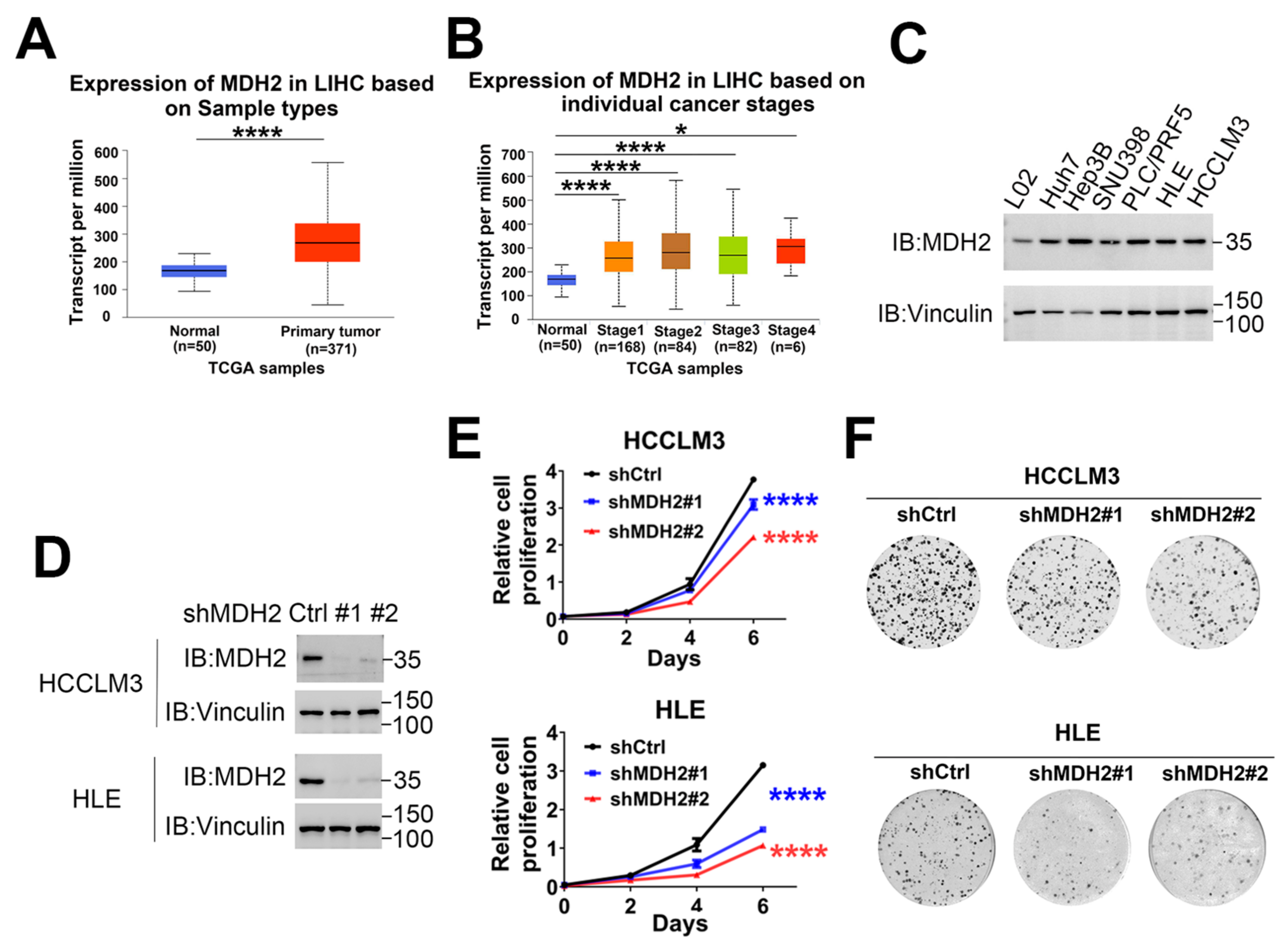
3.1. MDH2 Is Highly Expressed in HCC and Promotes HCC Growth
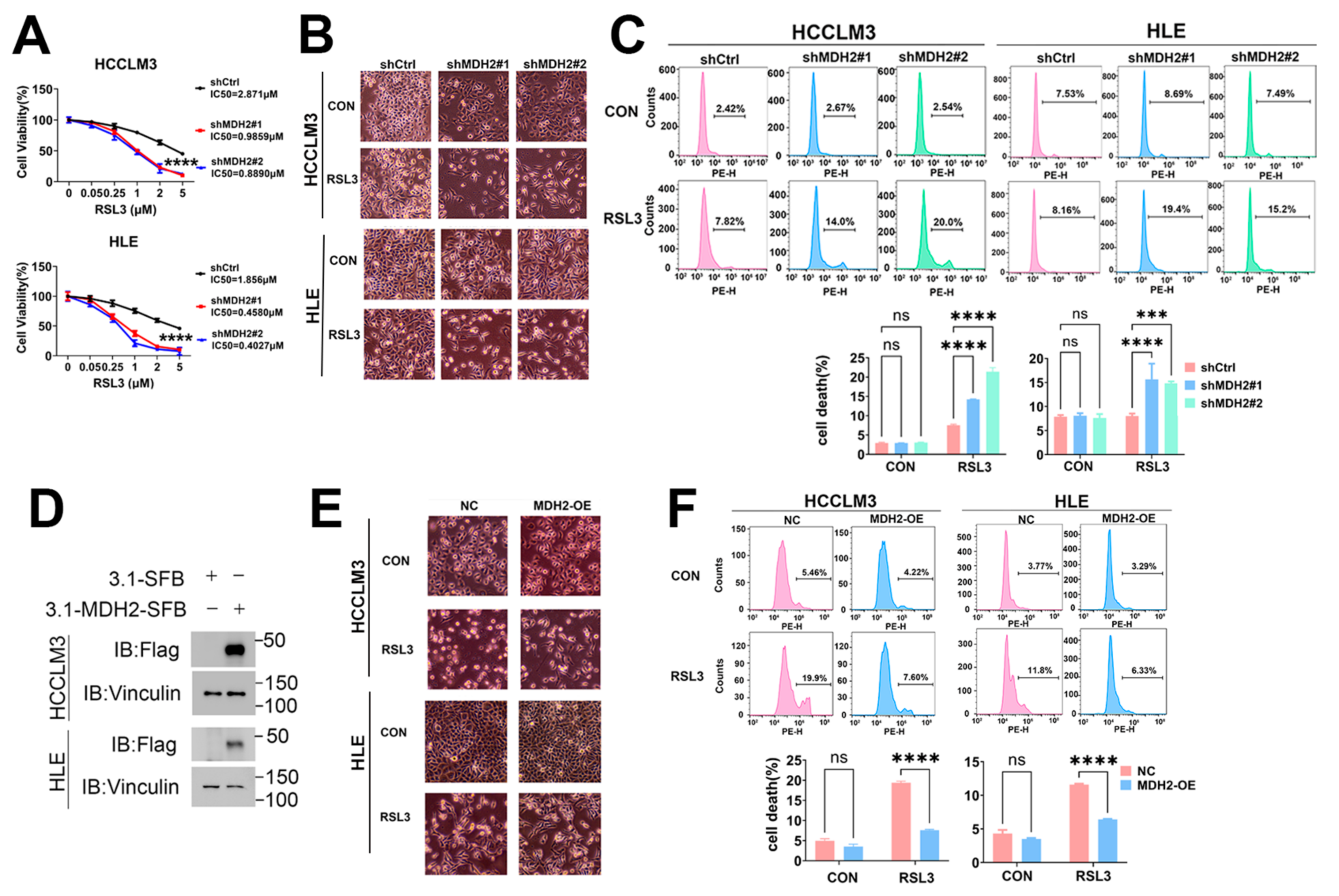
3.2. Knockdown of MDH2 Increases RSL3-Induced HCC Cell Death
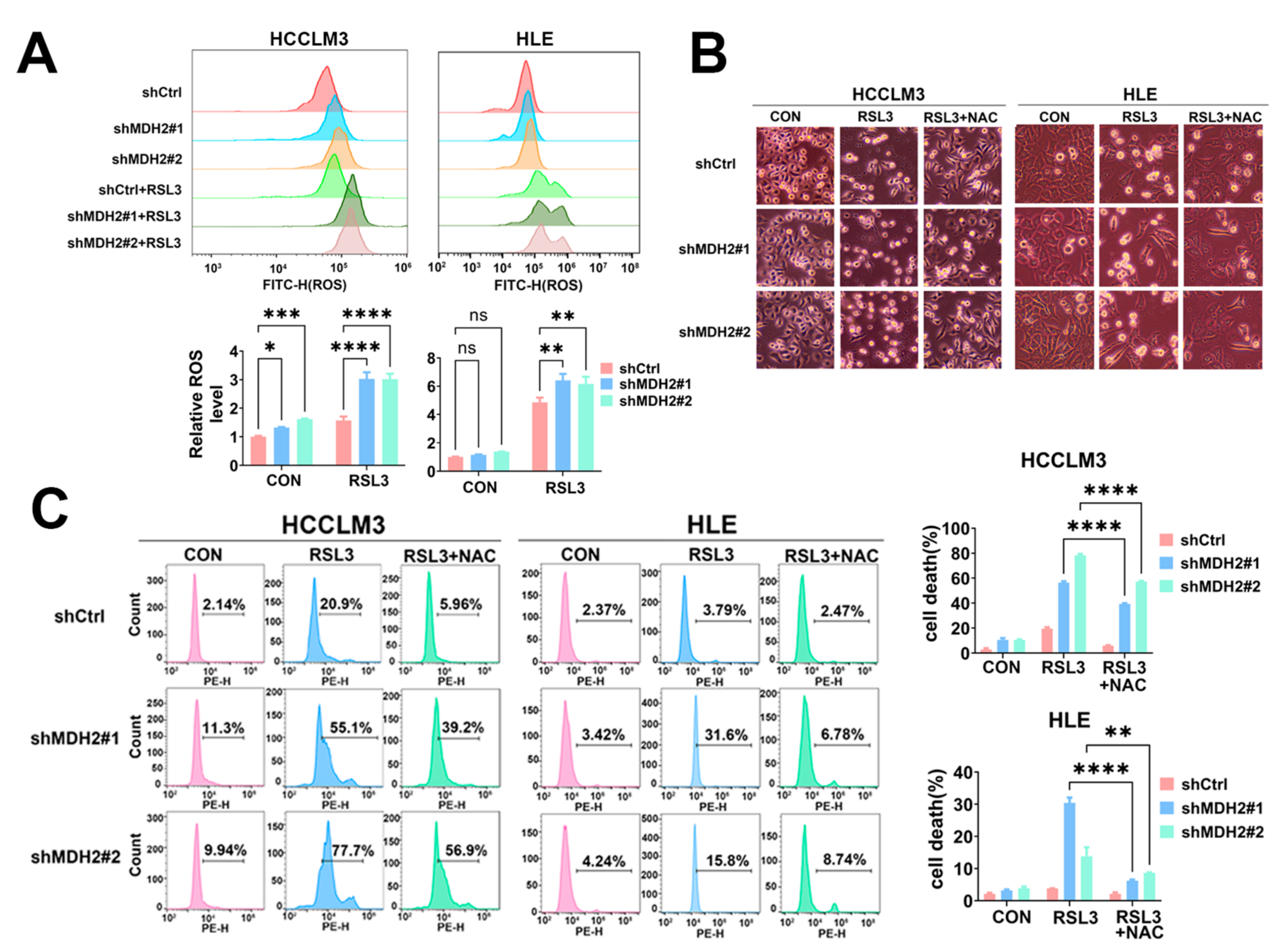
3.3. MDH2 Knockdown Increases RSL3-Induced HCC Cell Death via Upregulating Reactive Oxygen Species
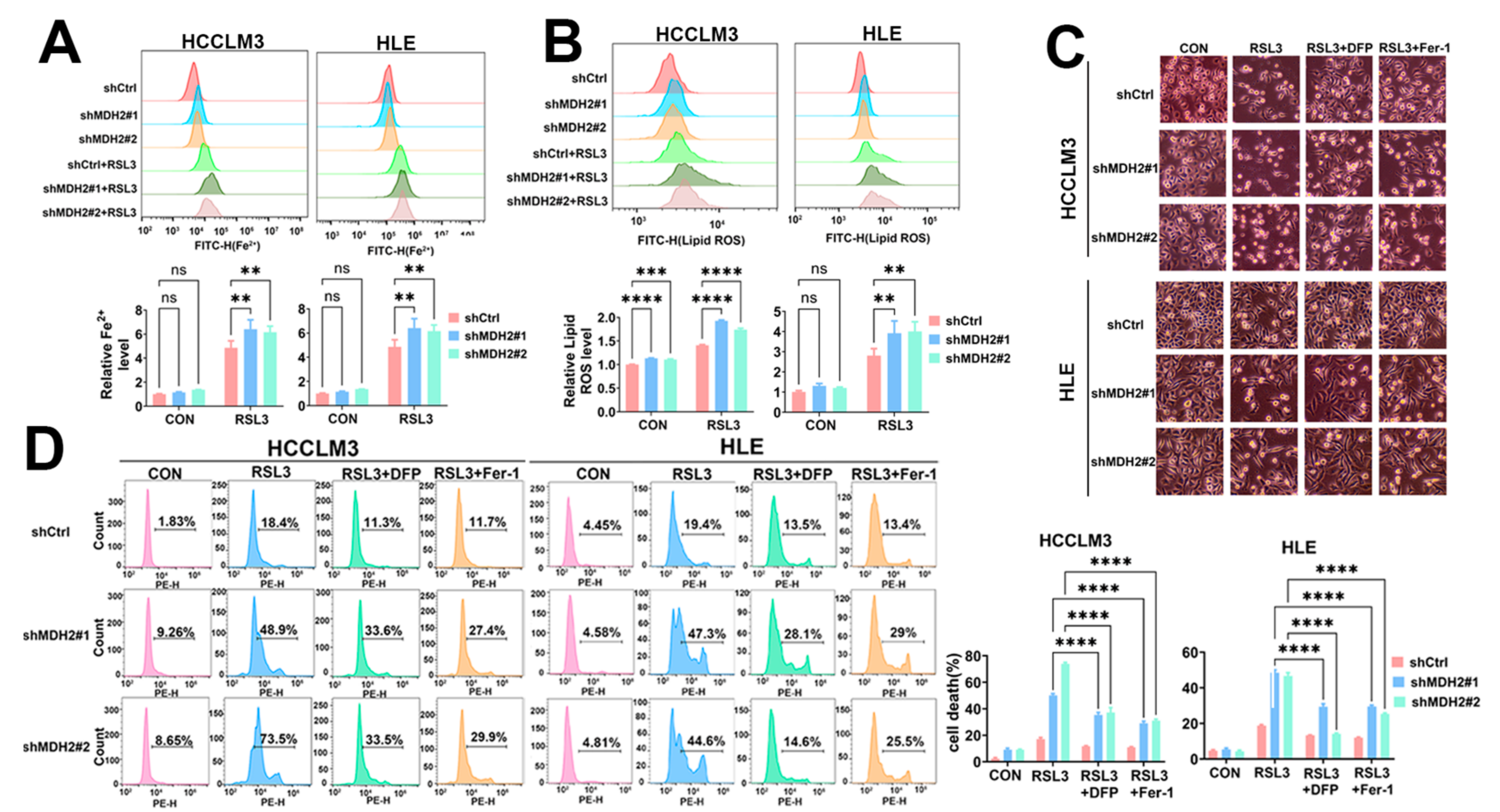
3.4. MDH2 Knockdown Sensitizes HCC to RSL3-Induced Ferroptosis
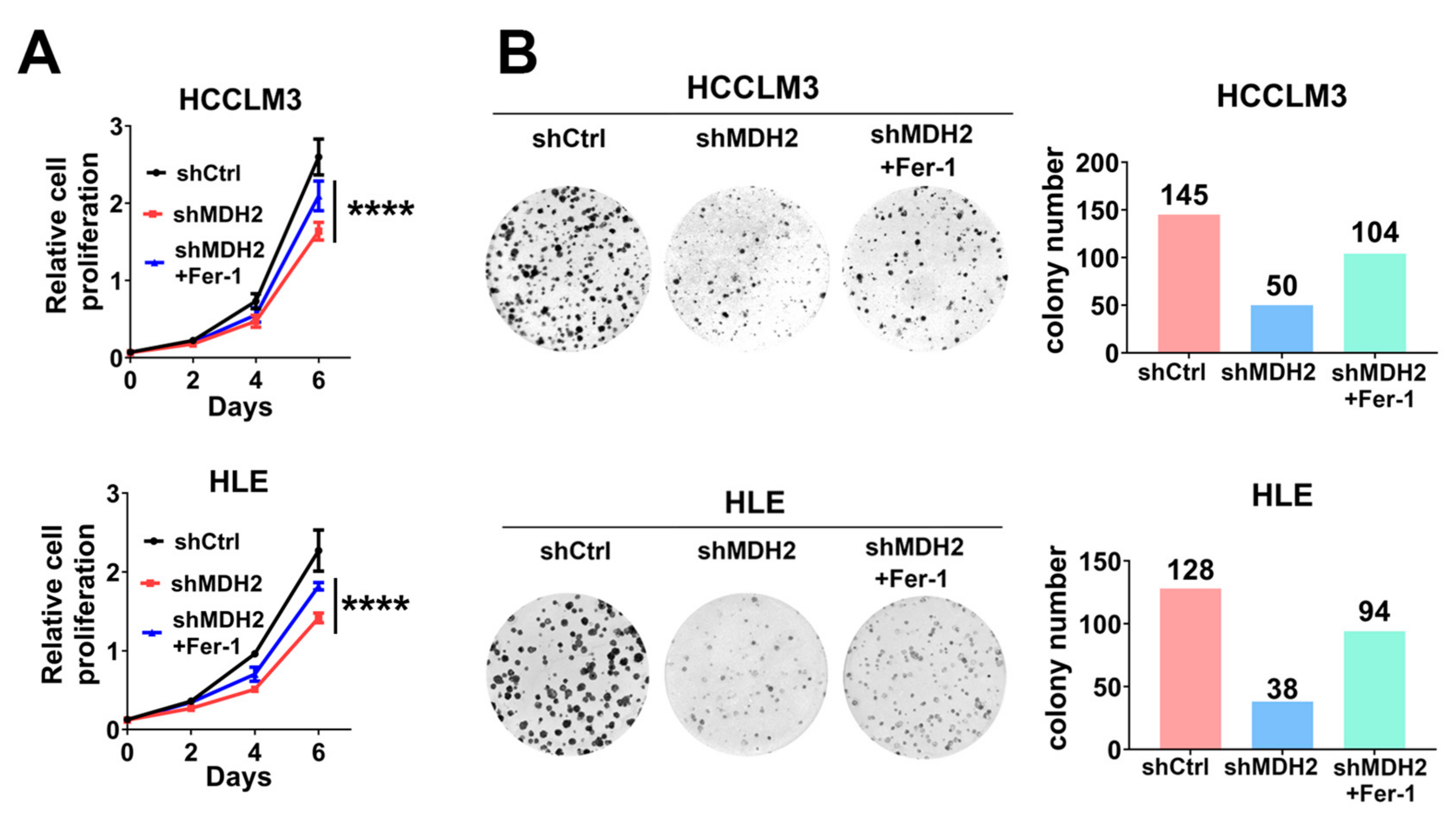
3.5. Knockdown of MDH2 Inhibits HCC Cell Growth Partially Dependent on Ferroptosis
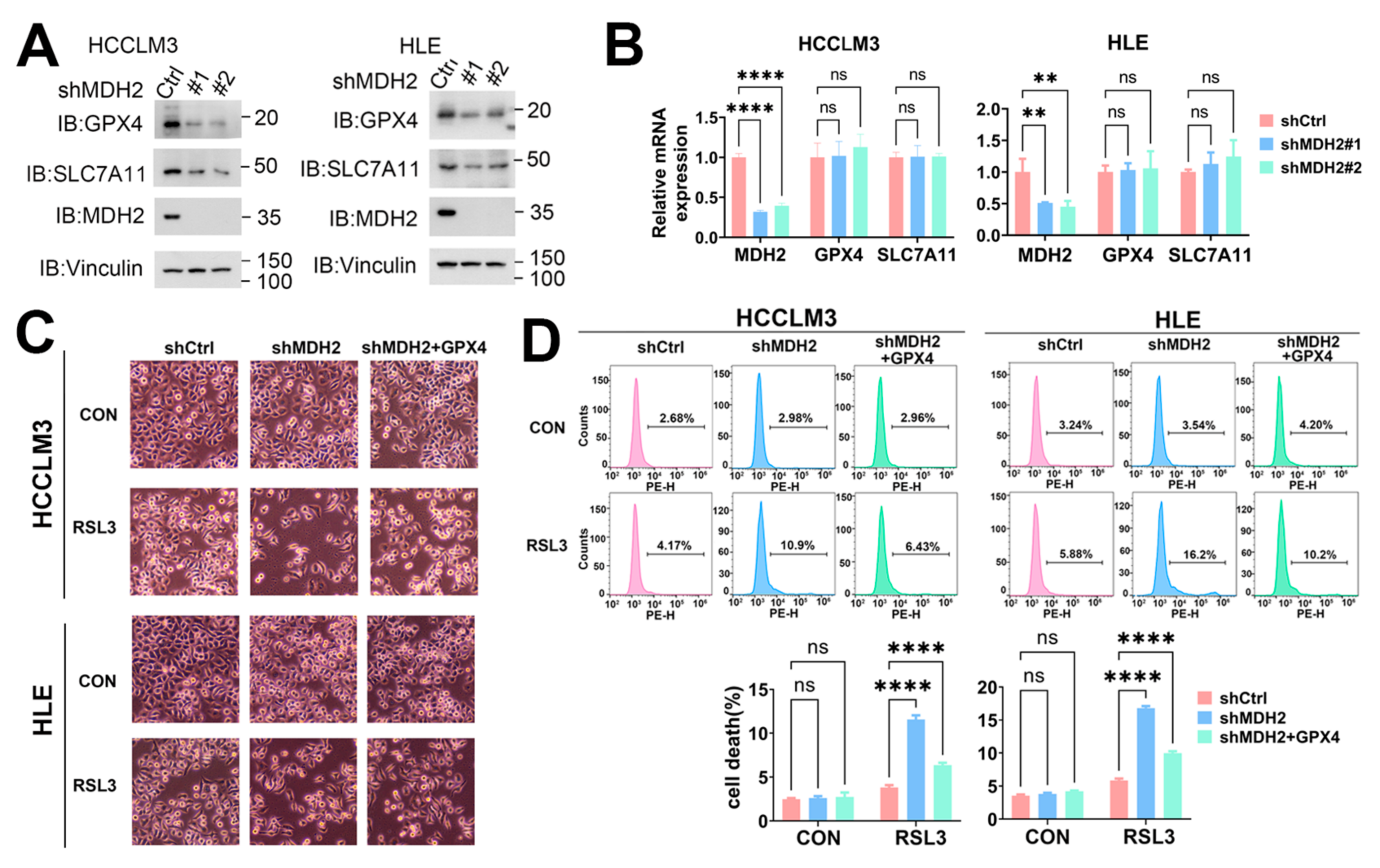
3.6. MDH2 Knockdown Sensitizes HCC to Ferroptotic Cell Death via GPX4
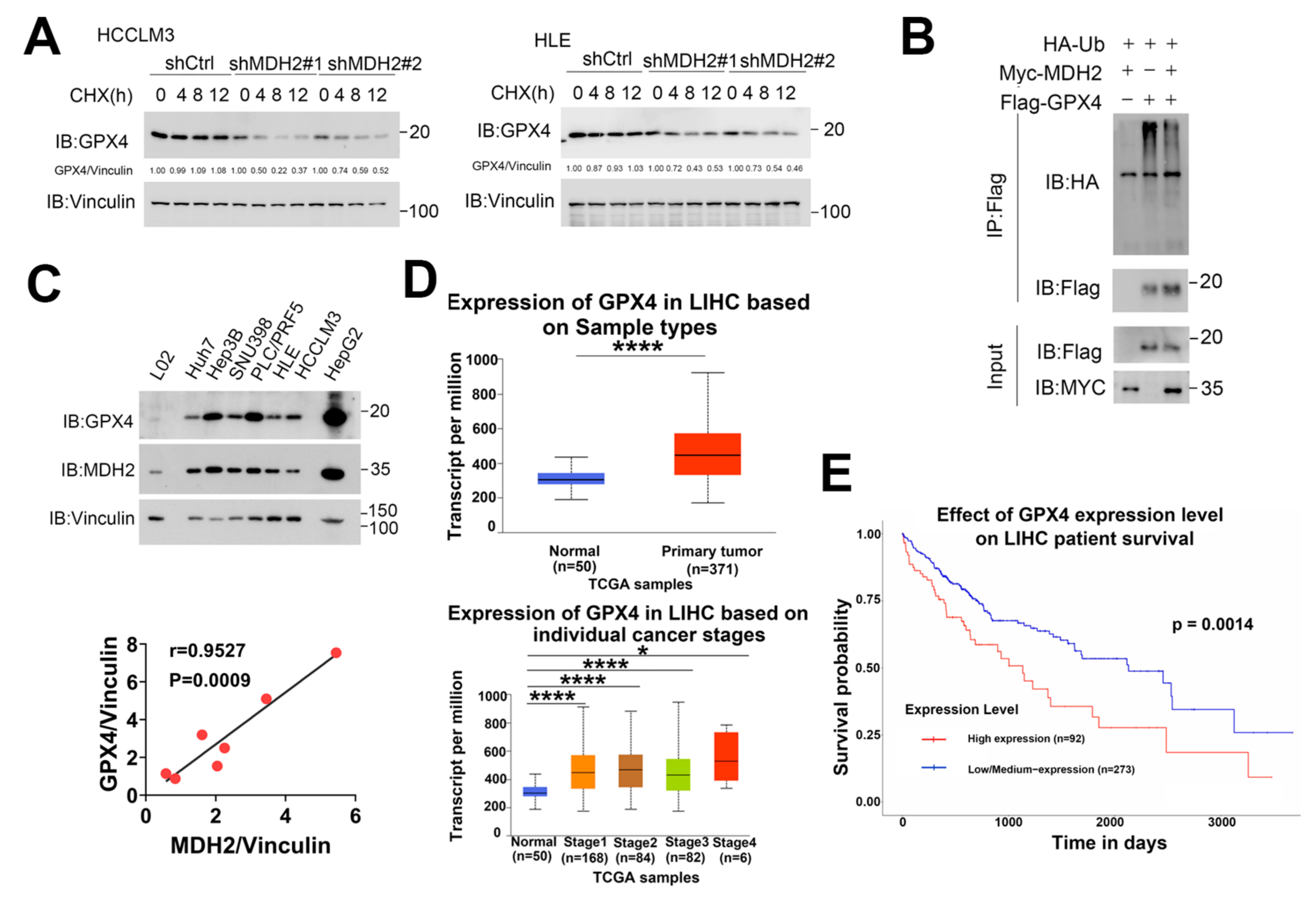
3.7. MDH2 Stabilizes GPX4
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Forner, M. Reig, J. Bruix, Hepatocellular carcinoma, Lancet, 391 (2018) 1301-1314. [CrossRef]
- S.J. Dixon, K.M. Lemberg, M.R. Lamprecht, R. Skouta, E.M. Zaitsev, C.E. Gleason, D.N. Patel, A.J. Bauer, A.M. Cantley, W.S. Yang, B. Morrison, 3rd, B.R. Stockwell, Ferroptosis: An iron-dependent form of nonapoptotic cell death, Cell, 149 (2012) 1060-1072. [CrossRef]
- K. Hadian, B.R. Stockwell, SnapShot: Ferroptosis, Cell, 181 (2020) 1188-1188.e1181. [CrossRef]
- Hassannia, P. Vandenabeele, T. Vanden Berghe, Targeting Ferroptosis to Iron Out Cancer, Cancer Cell, 35 (2019) 830-849. [CrossRef]
- F. Wang, H. Lv, B. Zhao, L. Zhou, S. Wang, J. Luo, J. Liu, P. Shang, Iron and leukemia: New insights for future treatments, J Exp Clin Cancer Res, 38 (2019) 406. [CrossRef]
- S. Doll, M. Conrad, Iron and ferroptosis: A still ill-defined liaison, IUBMB Life, 69 (2017) 423-434. [CrossRef]
- J. Nie, B. Lin, M. Zhou, L. Wu, T. Zheng, Role of ferroptosis in hepatocellular carcinoma, J Cancer Res Clin Oncol, 144 (2018) 2329-2337. [CrossRef]
- Dupourque, E. Kun, Malate dehydrogenases of ox kidney. 2. Two substrate kinetic and inhibition analyses, Eur J Biochem, 7 (1969) 247-252. [CrossRef]
- Jochmanová, Z. Zhuang, K. Pacak, Pheochromocytoma: Gasping for Air, Horm Cancer, 6 (2015) 191-205. [CrossRef]
- J. Gao, Q. Zhou, D. Wu, L. Chen, Mitochondrial iron metabolism and its role in diseases, Clin Chim Acta, 513 (2021) 6-12. [CrossRef]
- Y. Chen, Y. Zhang, S. Zhang, H. Ren, Molecular insights into sarcopenia: Ferroptosis-related genes as diagnostic and therapeutic targets, J Biomol Struct Dyn, (2024) 1-19. [CrossRef]
- P.P. Hou, L.J. Luo, H.Z. Chen, Q.T. Chen, X.L. Bian, S.F. Wu, J.X. Zhou, W.X. Zhao, J.M. Liu, X.M. Wang, Z.Y. Zhang, L.M. Yao, Q. Chen, D. Zhou, Q. Wu, Ectosomal PKM2 Promotes HCC by Inducing Macrophage Differentiation and Remodeling the Tumor Microenvironment, Mol Cell, 78 (2020) 1192-1206.e1110. [CrossRef]
- X.L. Bian, H.Z. Chen, P.B. Yang, Y.P. Li, F.N. Zhang, J.Y. Zhang, W.J. Wang, W.X. Zhao, S. Zhang, Q.T. Chen, Y. Zheng, X.Y. Sun, X.M. Wang, K.Y. Chien, Q. Wu, Nur77 suppresses hepatocellular carcinoma via switching glucose metabolism toward gluconeogenesis through attenuating phosphoenolpyruvate carboxykinase sumoylation, Nat Commun, 8 (2017) 14420. [CrossRef]
- Xu, F. Shao, X. Bian, Y. Meng, T. Liang, Z. Lu, The Evolving Landscape of Noncanonical Functions of Metabolic Enzymes in Cancer and Other Pathologies, Cell Metab, 33 (2021) 33-50. [CrossRef]
- X. Jiang, B.R. Stockwell, M. Conrad, Ferroptosis: Mechanisms, biology and role in disease, Nat Rev Mol Cell Biol, 22 (2021) 266-282. [CrossRef]
- M.A. Badgley, D.M. Kremer, H.C. Maurer, K.E. DelGiorno, H.J. Lee, V. Purohit, I.R. Sagalovskiy, A. Ma, J. Kapilian, C.E.M. Firl, A.R. Decker, S.A. Sastra, C.F. Palermo, L.R. Andrade, P. Sajjakulnukit, L. Zhang, Z.P. Tolstyka, T. Hirschhorn, C. Lamb, T. Liu, W. Gu, E.S. Seeley, E. Stone, G. Georgiou, U. Manor, A. Iuga, G.M. Wahl, B.R. Stockwell, C.A. Lyssiotis, K.P. Olive, Cysteine depletion induces pancreatic tumor ferroptosis in mice, Science, 368 (2020) 85-89. [CrossRef]
- P. Koppula, Y. Zhang, L. Zhuang, B. Gan, Amino acid transporter SLC7A11/xCT at the crossroads of regulating redox homeostasis and nutrient dependency of cancer, Cancer Commun (Lond), 38 (2018) 12. [CrossRef]
- Tang, G. Kroemer, Ferroptosis, Curr Biol, 30 (2020) R1292-r1297. [CrossRef]
- G.C. Forcina, S.J. Dixon, GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis, Proteomics, 19 (2019) e1800311. [CrossRef]
- A.S. Reisch, O. Elpeleg, Biochemical assays for mitochondrial activity: Assays of TCA cycle enzymes and PDHc, Methods Cell Biol, 80 (2007) 199-222. [CrossRef]
- D.L. Abeyawardhane, H.R. Lucas, Iron Redox Chemistry and Implications in the Parkinson's Disease Brain, Oxid Med Cell Longev, 2019 (2019) 4609702. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



