Submitted:
27 May 2024
Posted:
27 May 2024
You are already at the latest version
Abstract
Keywords:
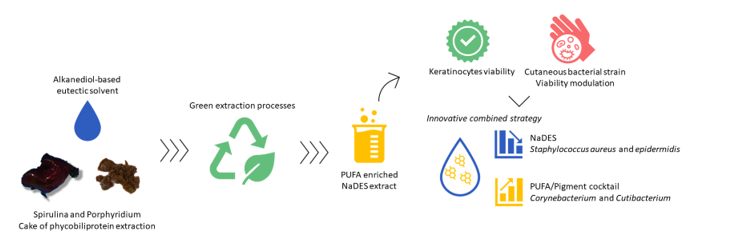
1. Introduction
2. Results and Discussion
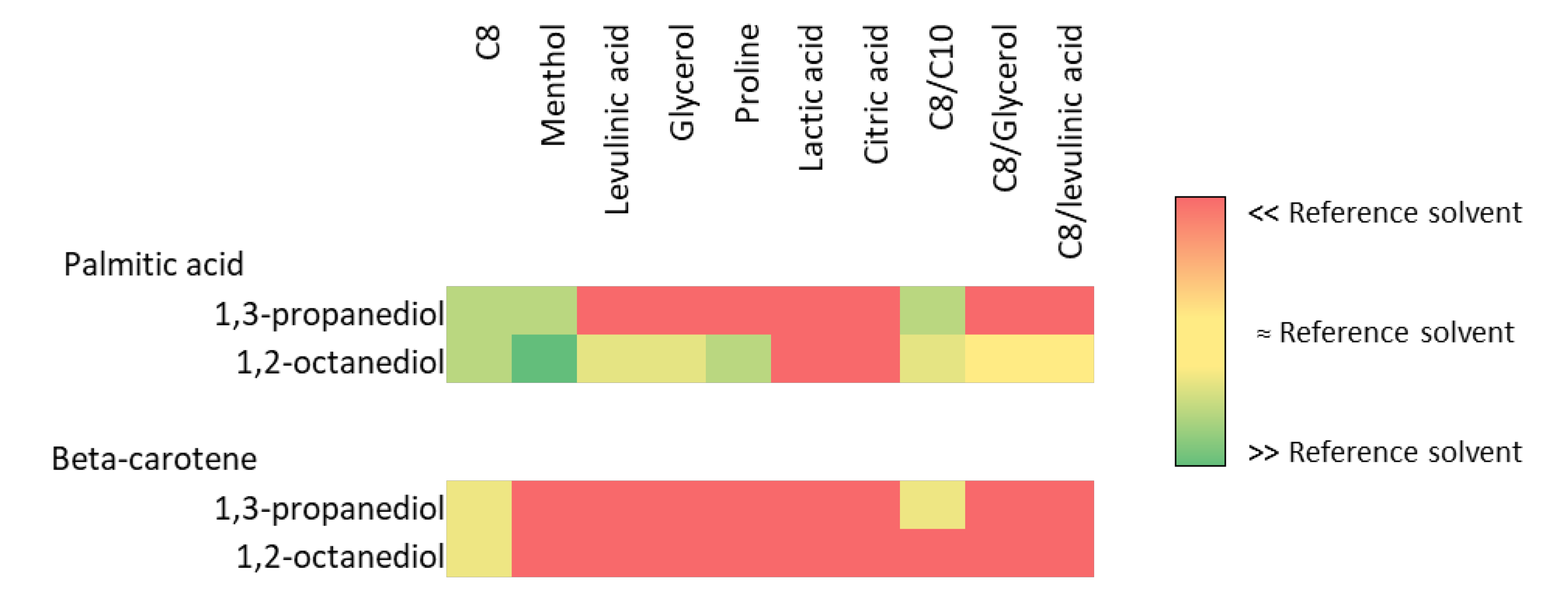
2.1. Alkanediol-Based NaDES Design and Characterization
2.2. FFA Extraction Using Hydrotrope-Based NaDES
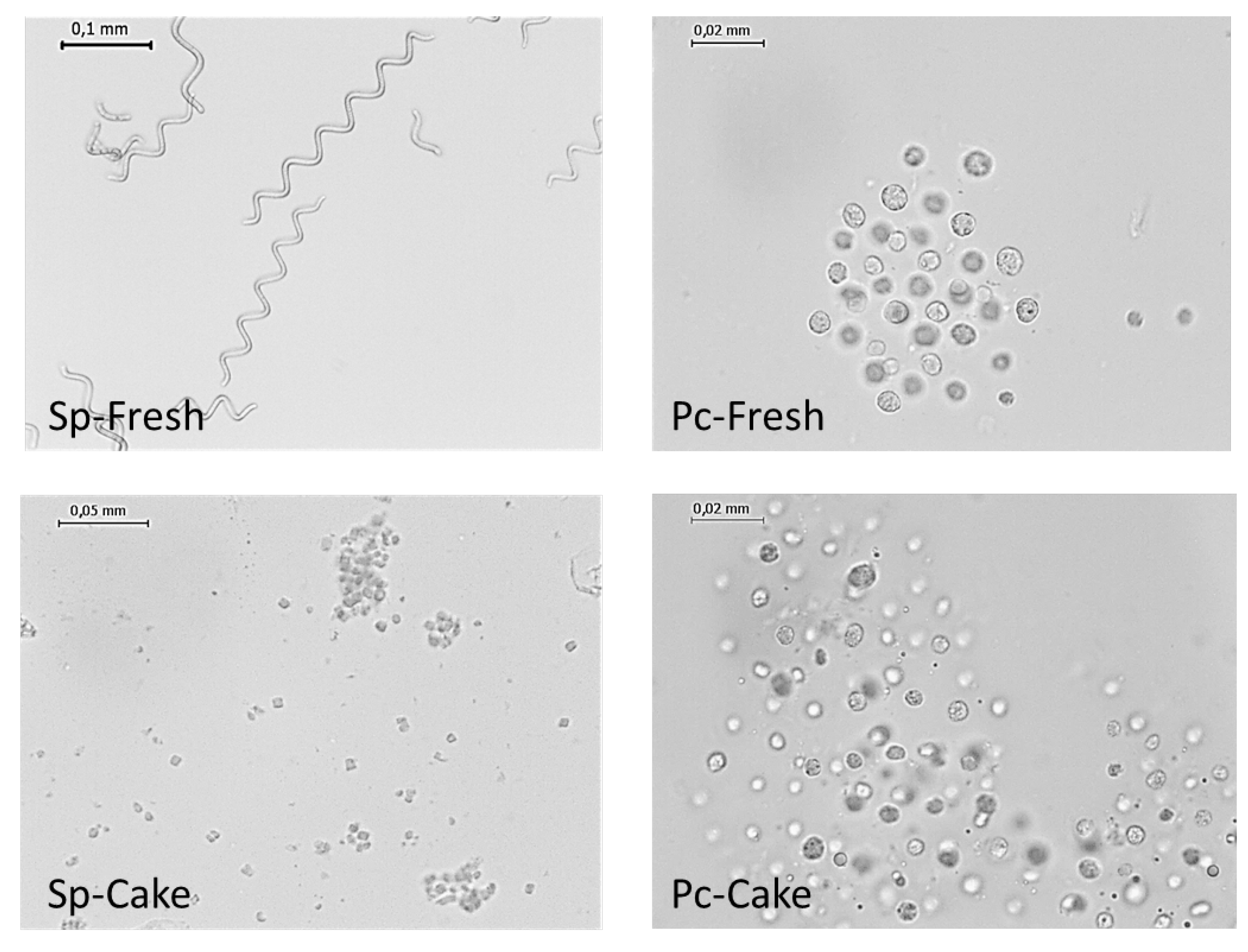
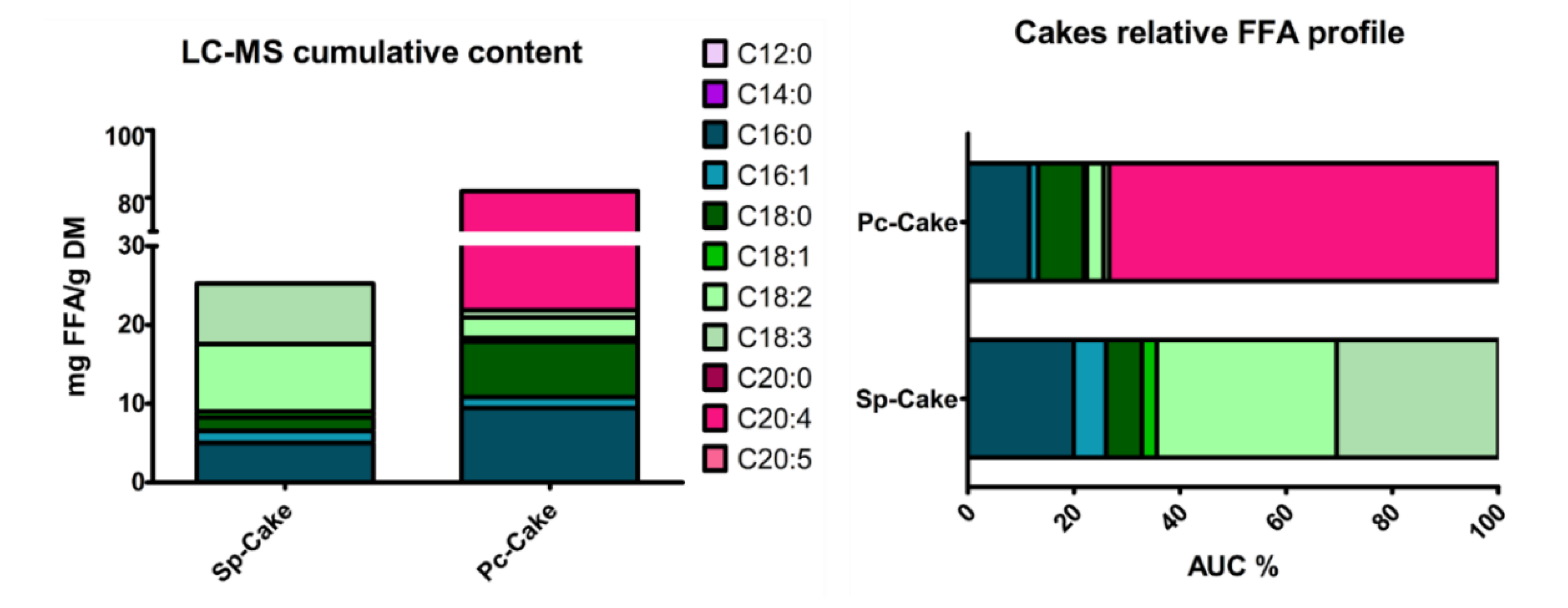
2.2.1. Cakes Characterization
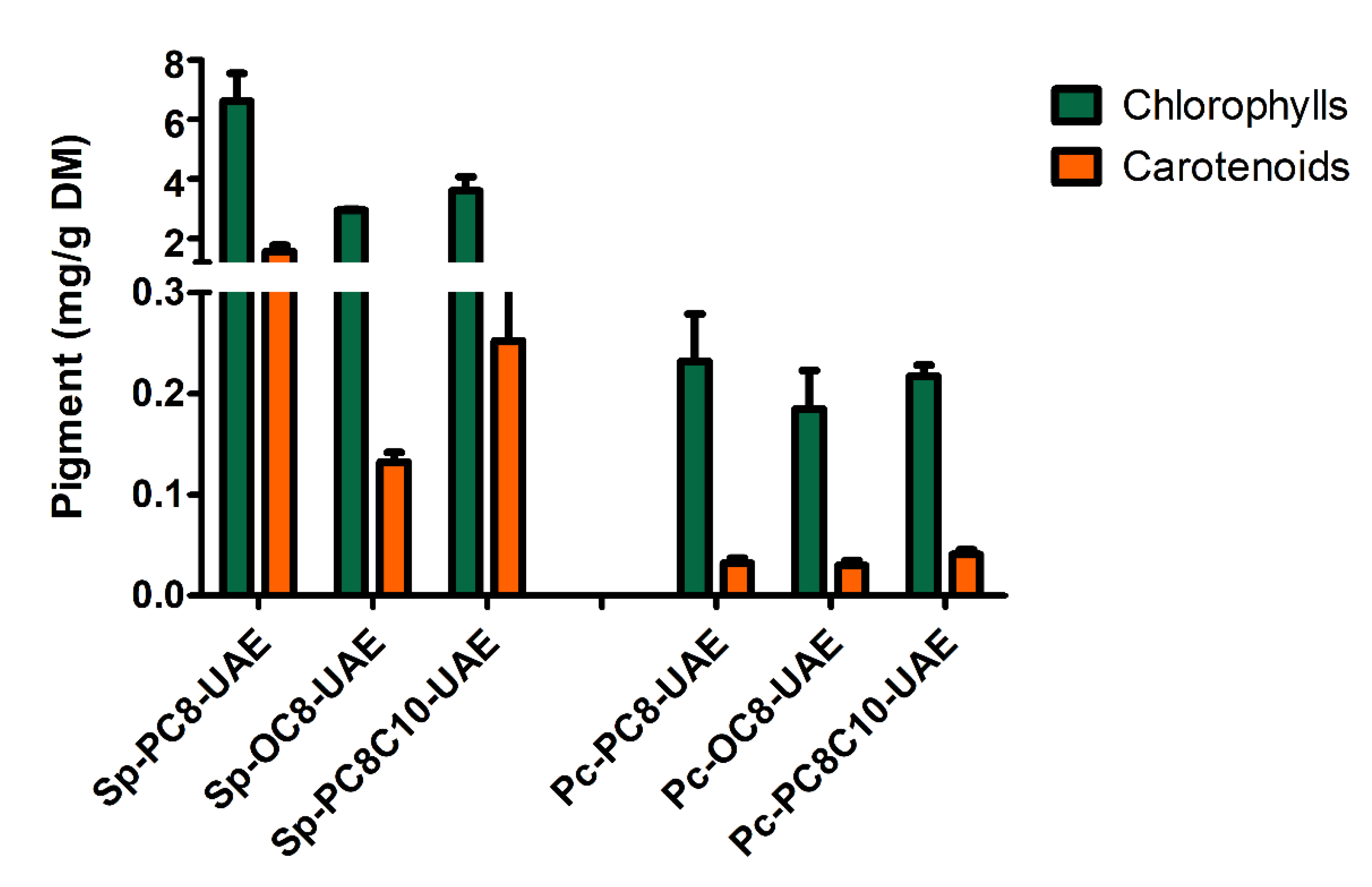
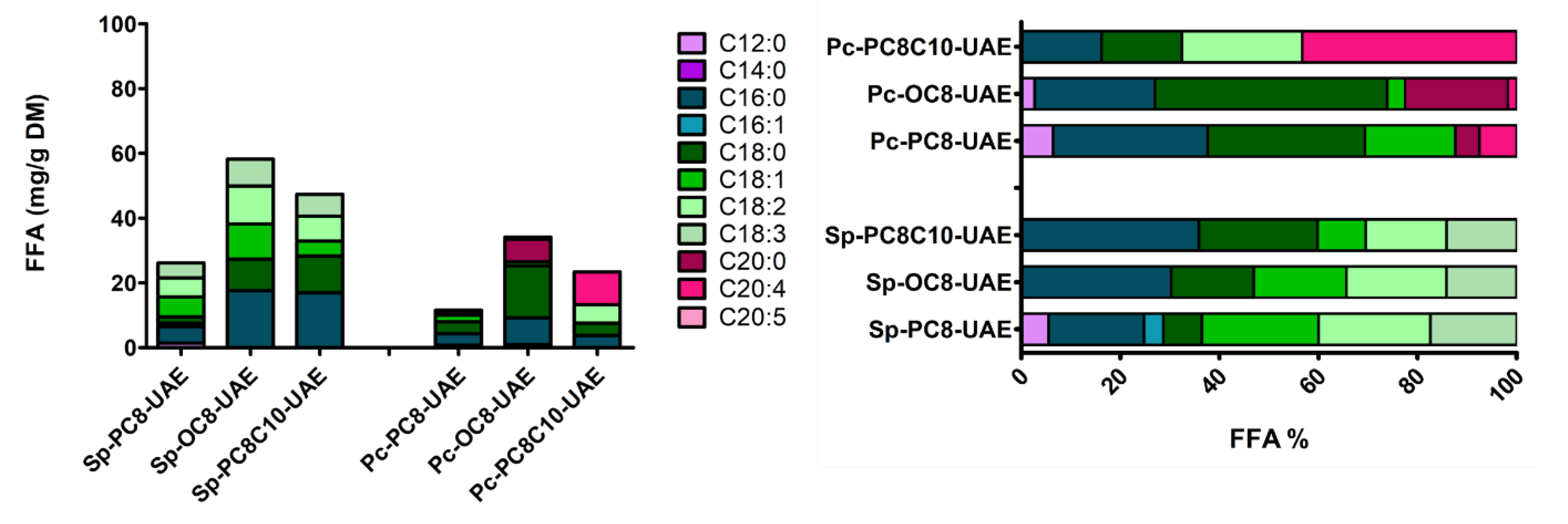
2.2.2. Ultrasound Assisted Extraction (UAE)
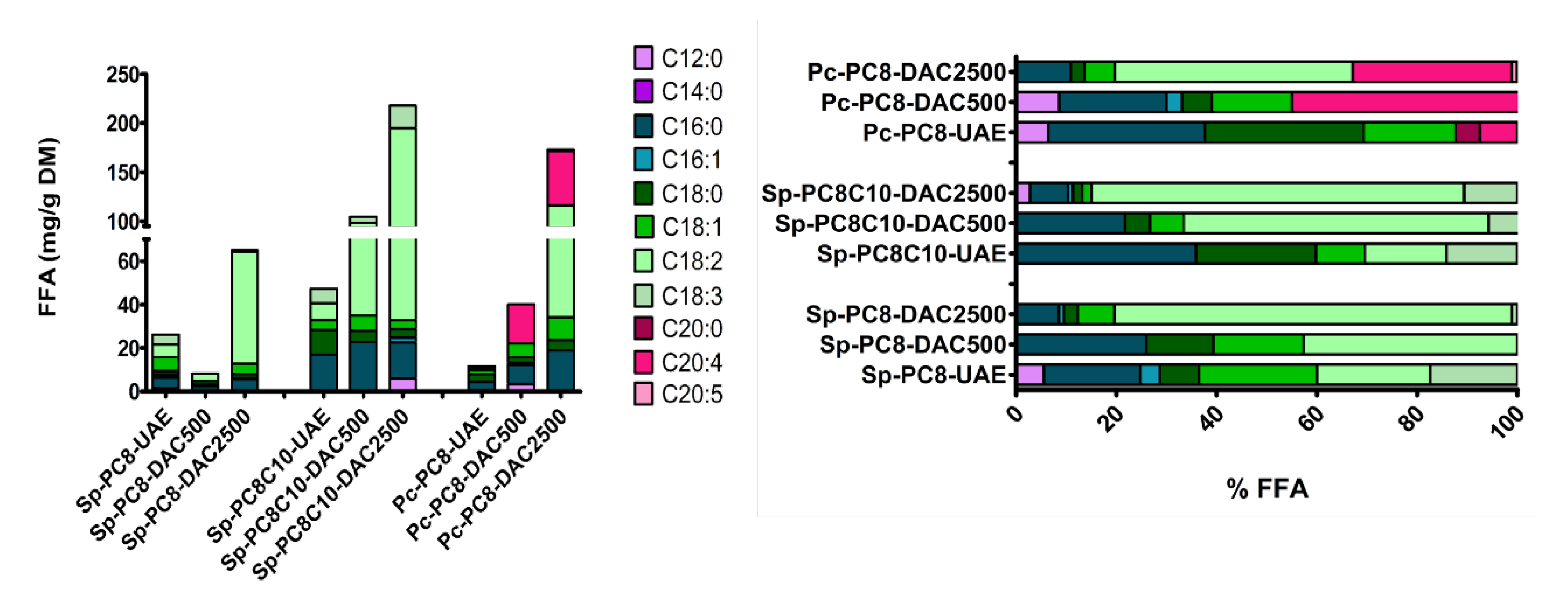
2.2.3. Dual Asymmetric Centrifugation (DAC) Extraction
2.3. Biological Evaluation for Cosmetic Use
2.3.1. Impact on Keratinocytes Viability
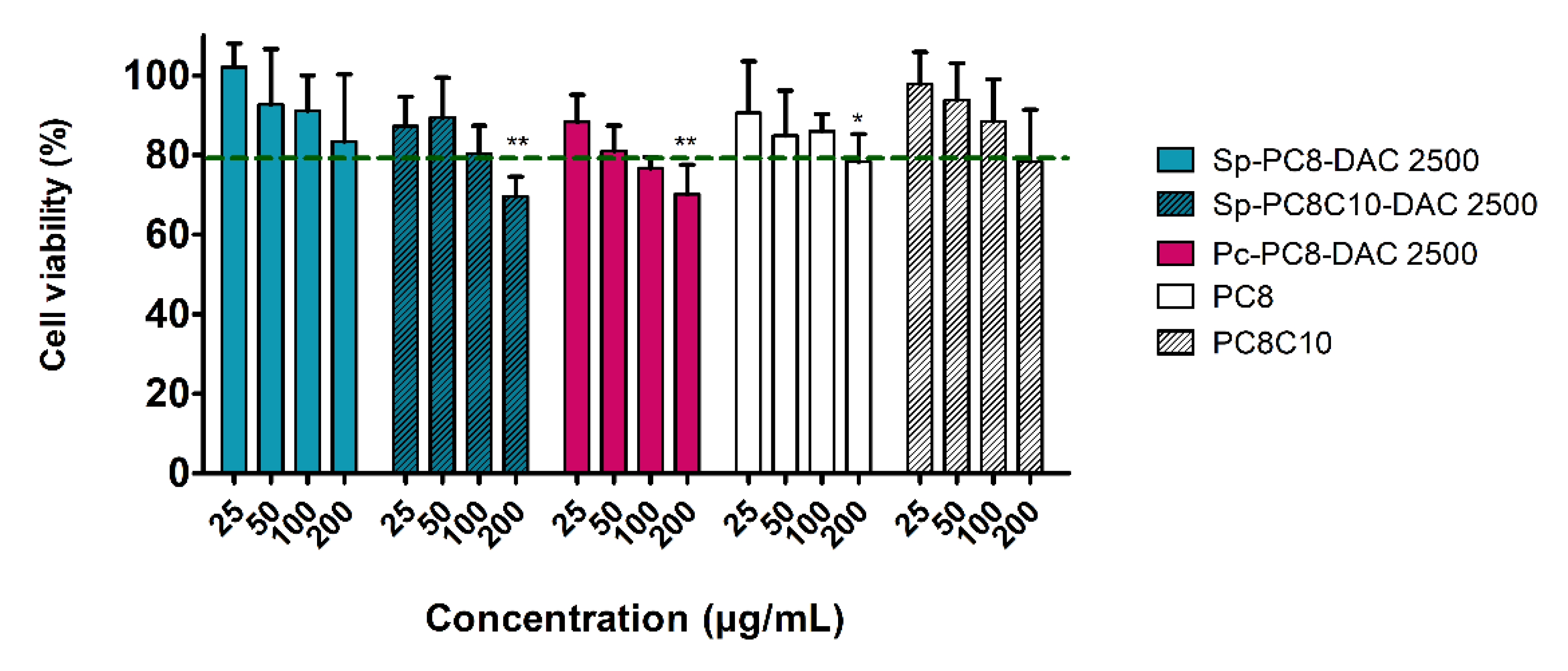
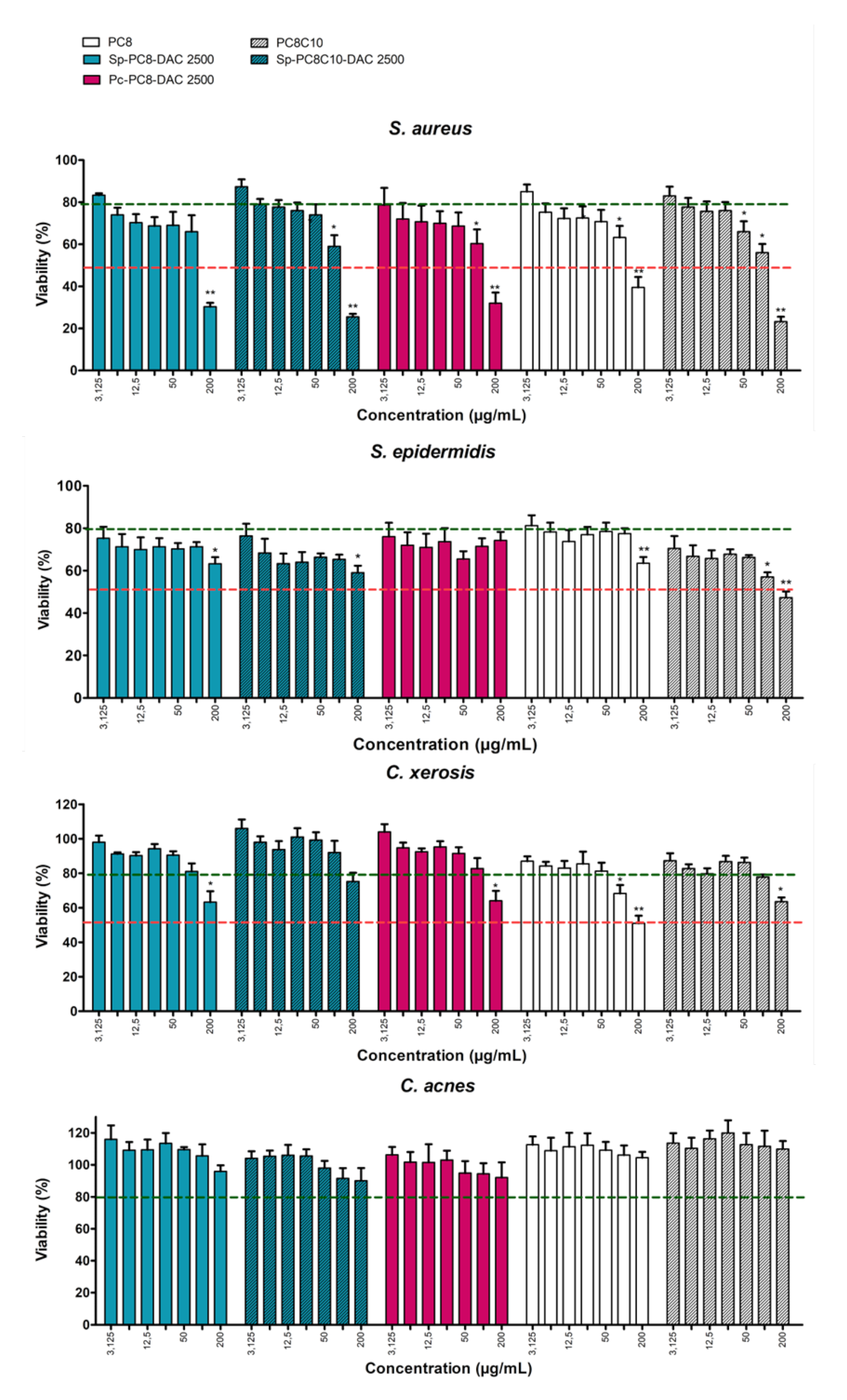
2.3.2. Impact on Skin Microbiota Bacterial Strains
3. Materials and Methods
3.1. Raw Material
3.1.1. Microscopy
3.1.2. Dry Matter Determination
3.2. Chemicals
3.3. NaDES Preparation and Characterization
3.3.1. NaDES Preparation
3.3.2. Density
3.3.3. Water Miscibility
3.3.4. Palmitic Acid and Beta-Carotene Solubilization Ability
3.4. Extraction Process
3.4.1. NaDES Screening and UAE
3.4.2. Dual Asymetric Centrifuge Extraction
3.5. Analytical Protocols
3.5.1. Pigment Content
3.5.2. Dye Profile
3.5.3. FFA Profile
3.6. Biological Evaluation
3.6.1. Sample Preparation
3.6.2. Determination of Keratinocyte Viability
3.6.3. Determination of Bacterial Viability
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Dolganyuk, V.; Belova, D.; Babich, O.; Prosekov, A.; Ivanova, S.; Katserov, D.; Patyukov, N.; Sukhikh, S. Microalgae: A Promising Source of Valuable Bioproducts. Biomolecules 2020, 10, 1153. [Google Scholar] [CrossRef] [PubMed]
- Jaime, L.; Mendiola, J.A.; Herrero, M.; Soler-Rivas, C.; Santoyo, S.; Señorans, F.J.; Cifuentes, A.; Ibáñez, E. Separation and Characterization of Antioxidants from Spirulina Platensis Microalga Combining Pressurized Liquid Extraction, TLC, and HPLC-DAD. J Sep Sci 2005, 28, 2111–2119. [Google Scholar] [CrossRef] [PubMed]
- Wils, L.; Yagmur, M.; Phelippe, M.; Montigny, B.; Clément-Larosière, B.; Jacquemin, J.; Boudesocque-Delaye, L. Alternative Solvents for the Biorefinery of Spirulina: Impact of Pretreatment on Free Fatty Acids with High Added Value. Mar Drugs 2022, 20, 600. [Google Scholar] [CrossRef] [PubMed]
- Pagels, F.; Guedes, A.C.; Amaro, H.M.; Kijjoa, A.; Vasconcelos, V. Phycobiliproteins from Cyanobacteria: Chemistry and Biotechnological Applications. Biotechnol Adv 2019, 37, 422–443. [Google Scholar] [CrossRef] [PubMed]
- Mühling, M.; Belay, A.; Whitton, B.A. Variation in Fatty Acid Composition of Arthrospira (Spirulina) Strains. J Appl Phycol 2005, 17, 137–146. [Google Scholar] [CrossRef]
- Rebolloso Fuentes, M. Biomass Nutrient Profiles of the Microalga Porphyridium Cruentum. Food Chem 2000, 70, 345–353. [Google Scholar] [CrossRef]
- Ruffell, S.E.; Müller, K.M.; McConkey, B.J. Comparative Assessment of Microalgal Fatty Acids as Topical Antibiotics. J Appl Phycol 2016, 28, 1695–1704. [Google Scholar] [CrossRef]
- Desbois, A.P.; Smith, V.J. Antibacterial Free Fatty Acids: Activities, Mechanisms of Action and Biotechnological Potential. Appl Microbiol Biotechnol 2010, 85, 1629–1642. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.Q.; Lin, L.; Lin, T.; Hao, F.; Zeng, F.Q.; Bi, Z.G.; Yi, D.; Zhao, B. Skin Colonization by Staphylococcus Aureus in Patients with Eczema and Atopic Dermatitis and Relevant Combined Topical Therapy: A Double-Blind Multicentre Randomized Controlled Trial. British Journal of Dermatology 2006, 155, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Alexander, H.; Paller, A.S.; Traidl-Hoffmann, C.; Beck, L.A.; De Benedetto, A.; Dhar, S.; Girolomoni, G.; Irvine, A.D.; Spuls, P.; Su, J.; et al. The Role of Bacterial Skin Infections in Atopic Dermatitis: Expert Statement and Review from the International Eczema Council Skin Infection Group. British Journal of Dermatology 2020, 182, 1331–1342. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, S.; Karim, M.R.; Iqbal, S.; Lee, J.H.; Mathiyalagan, R.; Kim, Y.J.; Yang, D.U.; Yang, D.C. Atopic Dermatitis: Pathophysiology, Microbiota, and Metabolome – A Comprehensive Review. Microbiol Res 2024, 281, 127595. [Google Scholar] [CrossRef] [PubMed]
- Desbois, A.; Lawlor, K. Antibacterial Activity of Long-Chain Polyunsaturated Fatty Acids against Propionibacterium Acnes and Staphylococcus Aureus. Mar Drugs 2013, 11, 4544–4557. [Google Scholar] [CrossRef] [PubMed]
- Hilali, S.; Van Gheluwe, L.; Yagmur, M.; Wils, L.; Phelippe, M.; Clément-Larosière, B.; Montigny, B.; Jacquemin, J.; Thiery, E.; Boudesocque-Delaye, L. NaDES-Based Biorefinery of Spirulina (Arthrospira Platensis): A New Path for Sustainable High Value-Added Metabolites. Sep Purif Technol 2024, 329, 125123. [Google Scholar] [CrossRef]
- Wils, L.; Leman-Loubière, C.; Bellin, N.; Clément-Larosière, B.; Pinault, M.; Chevalier, S.; Enguehard-Gueiffier, C.; Bodet, C.; Boudesocque-Delaye, L. Natural Deep Eutectic Solvent Formulations for Spirulina: Preparation, Intensification, and Skin Impact. Algal Res 2021, 56, 102317. [Google Scholar] [CrossRef]
- Afonso, J.; Mezzetta, A.; Marrucho, I.M.; Guazzelli, L. History Repeats Itself Again: Will the Mistakes of the Past for ILs Be Repeated for DESs? From Being Considered Ionic Liquids to Becoming Their Alternative: The Unbalanced Turn of Deep Eutectic Solvents. Green Chemistry 2023, 25, 59–105. [Google Scholar] [CrossRef]
- Abranches, D.O.; Coutinho, J.A.P. Everything You Wanted to Know about Deep Eutectic Solvents but Were Afraid to Be Told. Annu Rev Chem Biomol Eng 2023, 14, 141–163. [Google Scholar] [CrossRef] [PubMed]
- Florindo, C.; Branco, L.C.; Marrucho, I.M. Quest for Green-Solvent Design: From Hydrophilic to Hydrophobic (Deep) Eutectic Solvents. ChemSusChem 2019, 12, 1549–1559. [Google Scholar] [CrossRef] [PubMed]
- van Osch, D.J.G.P.; Dietz, C.H.J.T.; van Spronsen, J.; Kroon, M.C.; Gallucci, F.; van Sint Annaland, M.; Tuinier, R. A Search for Natural Hydrophobic Deep Eutectic Solvents Based on Natural Components. ACS Sustain Chem Eng 2019, 7, 2933–2942. [Google Scholar] [CrossRef]
- Sanati, A.; Malayeri, M.R.; Busse, O.; Weigand, J.J. Inhibition of Asphaltene Precipitation Using Hydrophobic Deep Eutectic Solvents and Ionic Liquid. J Mol Liq 2021, 334, 116100. [Google Scholar] [CrossRef]
- Van Osch, D.J.G.P.; Dietz, C.H.J.T.; Warrag, S.E.E.; Kroon, M.C. The Curious Case of Hydrophobic Deep Eutectic Solvents: A Story on the Discovery, Design, and Applications. ACS Sustain Chem Eng 2020, 8, 10591–10612. [Google Scholar] [CrossRef]
- Silva, Y.P.A.; Ferreira, T.A.P.C.; Jiao, G.; Brooks, M.S. Sustainable Approach for Lycopene Extraction from Tomato Processing By-Product Using Hydrophobic Eutectic Solvents. J Food Sci Technol 2019, 56, 1649–1654. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.; Banerjee, T. Liquid-Liquid Extraction of Lower Alcohols Using Menthol-Based Hydrophobic Deep Eutectic Solvent: Experiments and COSMO-SAC Predictions. Ind Eng Chem Res 2018, 57, 3371–3381. [Google Scholar] [CrossRef]
- Muhammad, G.; Xu, J.; Li, Z.; Zhao, L.; Zhang, X. Current Progress and Future Perspective of Microalgae Biomass Pretreatment Using Deep Eutectic Solvents. Science of The Total Environment 2024, 924, 171547. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Krishna Paidi, M.; Kulshrestha, A.; Bharmoria, P.; Kumar Mandal, S.; Kumar, A. Deep Eutectic Solvents Based Biorefining of Value-Added Chemicals from the Diatom Thalassiosira Andamanica at Room Temperature. Sep Purif Technol 2022, 298, 121636. [Google Scholar] [CrossRef]
- Xu, D.; Chow, J.; Weber, C.C.; Packer, M.A.; Baroutian, S.; Shahbaz, K. Evaluation of Deep Eutectic Solvents for the Extraction of Fucoxanthin from the Alga Tisochrysis Lutea – COSMO-RS Screening and Experimental Validation. J Environ Chem Eng 2022, 10, 108370. [Google Scholar] [CrossRef]
- Pitacco, W.; Samorì, C.; Pezzolesi, L.; Gori, V.; Grillo, A.; Tiecco, M.; Vagnoni, M.; Galletti, P. Extraction of Astaxanthin from Haematococcus Pluvialis with Hydrophobic Deep Eutectic Solvents Based on Oleic Acid. Food Chem 2022, 379, 132156. [Google Scholar] [CrossRef]
- Sousa, S.C.; Freitas, A.C.; Gomes, A.M.; Carvalho, A.P.; Sousa, S.C.; Freitas, A.C.; Gomes, A.M.; Carvalho, A.P.; Pt, A. Citation: Extraction of Nannochloropsis Fatty Acids Using Different Green Technologies: The Current Path. 2023. [Google Scholar] [CrossRef]
- Silva, J.M.; Silva, E.; Reis, R.L.; Duarte, A.R.C. A Closer Look in the Antimicrobial Properties of Deep Eutectic Solvents Based on Fatty Acids. Sustain Chem Pharm 2019, 14, 100192. [Google Scholar] [CrossRef]
- Jeong, K.M.; Ko, J.; Zhao, J.; Jin, Y.; Yoo, D.E.; Han, S.Y.; Lee, J. Multi-Functioning Deep Eutectic Solvents as Extraction and Storage Media for Bioactive Natural Products That Are Readily Applicable to Cosmetic Products. J Clean Prod 2017, 151, 87–95. [Google Scholar] [CrossRef]
- Vieira, V.; Calhelha, R.C.; Barros, L.; Coutinho, J.A.P.; C. F. R. Ferreira, I.; Ferreira, O. Insights on the Extraction Performance of Alkanediols and Glycerol: Using Juglans Regia L. Leaves as a Source of Bioactive Compounds. Molecules 2020, 25, 2497. [Google Scholar] [CrossRef] [PubMed]
- Aravena, P.; Cea-Klapp, E.; Gajardo-Parra, N.F.; Held, C.; Garrido, J.M.; Canales, R.I. Effect of Water and Hydrogen Bond Acceptor on the Density and Viscosity of Glycol-Based Eutectic Solvents. J Mol Liq 2023, 389, 122856. [Google Scholar] [CrossRef]
- Yue, J.; Zhu, Z.; Yi, J.; Li, H.; Chen, B.; Rao, J. One-Step Extraction of Oat Protein by Choline Chloride-Alcohol Deep Eutectic Solvents: Role of Chain Length of Dihydric Alcohol. Food Chem 2022, 376, 131943. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, B.; Gonzalez-Miquel, M. Alkanediol-Based Deep Eutectic Solvents for Isolation of Terpenoids from Citrus Essential Oil: Experimental Evaluation and COSMO-RS Studies. Sep Purif Technol 2019, 227, 115707. [Google Scholar] [CrossRef]
- Yu, W.; Bo, Y.; Luo, Y.; Huang, X.; Zhang, R.; Zhang, J. Enhancing Effect of Choline Chloride-Based Deep Eutectic Solvents with Polyols on the Aqueous Solubility of Curcumin–Insight from Experiment and Theoretical Calculation. Chin J Chem Eng 2023, 59, 160–168. [Google Scholar] [CrossRef]
- Boudesocque-Delaye, L.; Ardeza, I.M.; Verger, A.; Grard, R.; Théry-Koné, I.; Perse, X.; Munnier, E. Natural Deep Eutectic Solvents as a Novel Bio-Based Matrix for Ready-to-Use Natural Antioxidants-Enriched Ingredients: Extraction and Formulation Optimization. Cosmetics 2024, 11, 17. [Google Scholar] [CrossRef]
- Benoit, C.; Virginie, C.; Boris, V. The Use of NADES to Support Innovation in the Cosmetic Industry. In; 2021; pp. 309–332.
- Silva, J.M.; Pereira, C. V.; Mano, F.; Silva, E.; Castro, V.I.B.; Sá-Nogueira, I.; Reis, R.L.; Paiva, A.; Matias, A.A.; Duarte, A.R.C. Therapeutic Role of Deep Eutectic Solvents Based on Menthol and Saturated Fatty Acids on Wound Healing. ACS Appl Bio Mater 2019, 2, 4346–4355. [Google Scholar] [CrossRef] [PubMed]
- Kodicek, E.; Worden, A.N. The Effect of Unsaturated Fatty Acids on Lactobacillus Helveticus and Other Gram-Positive Micro-Organisms. Biochemical Journal 1945, 39, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Radošević, K.; Čanak, I.; Panić, M.; Markov, K.; Bubalo, M.C.; Frece, J.; Srček, V.G.; Redovniković, I.R. Antimicrobial, Cytotoxic and Antioxidative Evaluation of Natural Deep Eutectic Solvents. Environmental Science and Pollution Research 2018, 25, 14188–14196. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.; Bogorad, L. COMPLEMENTARY CHROMATIC ADAPTATION IN A FILAMENTOUS BLUE-GREEN ALGA. J Cell Biol 1973, 58, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Boutin, R.; Munnier, E.; Renaudeau, N.; Girardot, M.; Pinault, M.; Chevalier, S.; Chourpa, I.; Clément-Larosière, B.; Imbert, C.; Boudesocque-Delaye, L. Spirulina Platensis Sustainable Lipid Extracts in Alginate-Based Nanocarriers: An Algal Approach against Biofilms. Algal Res 2019. [Google Scholar] [CrossRef]

| NaDES code | Component 1 | Component 2 | Component 3 | Molar Ratio (water%) | Heating* (°C) | Density (g/mL) | Water miscibility |
|---|---|---|---|---|---|---|---|
| PC8 | 1,3-propanediol | Octanoic acid | - | 1:5 | 50 | 0.914 | - |
| PM | L-Menthol | - | 1:1 | 60 | 0.946 | - | |
| PG | Glycerol | - | 1:1 | 60 | 1.135 | + | |
| PCit | Citric acid | - | 1:1 | 80 | 1.272 | + | |
| PLac | Lactic acid | - | 1:1 | 50 | 1.109 | + | |
| PLev | Levulinic acid | - | 1:1 | 50 | 1.095 | + | |
| PP | Proline | - | 8:1 | 60 | 1.072 | + | |
| PGC8 | Glycerol | Octanoic acid | 2:1:1 | 50 | 1.028 | - | |
| PLevC8 | Levulinic acid | Octanoic acid | 2:1:1 | 50 | 1.018 | - | |
| PC8C10 | Octanoic acid | Decanoic acid | 1:3:1 | 20 | 0.912 | - | |
| OC8 | 1,2-octanediol | Octanoic acid | - | 1:5 | 50 | 0.901 | - |
| OM | L-Menthol | - | 1:1 | 60 | 0.886 | - | |
| OG | Glycerol | - | 1:1 | 60 | 1.010 | - | |
| OCit | Citric acid | water | 1:1 (20%) | 80 | 1.161 | + | |
| OLac | Lactic acid | - | 1:1 | 60 | 0.975 | + | |
| OLev | Levulinic acid | - | 1:1 | 60 | 0.980 | + | |
| OP | Proline | - | 8:1 | 60 | 0.941 | - | |
| OGC8 | Glycerol | Octanoic acid | 2:1:1 | 50 | 0.967 | - | |
| OLevC8 | Levulinic acid | Octanoic acid | 2:1:1 | 50 | 0.956 | - | |
| OC8C10 | Octanoic acid | Decanoic acid | 1:1:2 | 50 | 0.900 | - |
| Microalgae | Code | DM (%) | Phycobiliprotein ; (mg/g DM) ; n=3 |
Chlorophylls ; (mg/g DM) n=3 |
Carotenoids ; (mg/g DM) ; n=3 |
|---|---|---|---|---|---|
| A. platensis | Sp-Cake | 17.2 | 84.8 ± 0.4 | 4.4 ± 0,1 | 1.6 ± 0,1 |
| P. cruentum | Pc-Cake | 9.0 | 12.9 ± 0.2 | 1.9 ± 0.1 | 0.8 ± 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




