Submitted:
24 May 2024
Posted:
27 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Surgical Preparation and Injury Induction
2.3. Drug Preparation and Infusion Administration
2.4. Tracer Infusion
2.5. Tissue Processing
2.6. Quantification of Cathepsin B Activity
2.7. Western Blotting
2.8. Membrane Disruption and Total Cell Count Analysis
2.9. Cellular Cathepsin B Localization Analysis
2.10. Whisker Nuisance Task
2.11. Statistics
3. Results
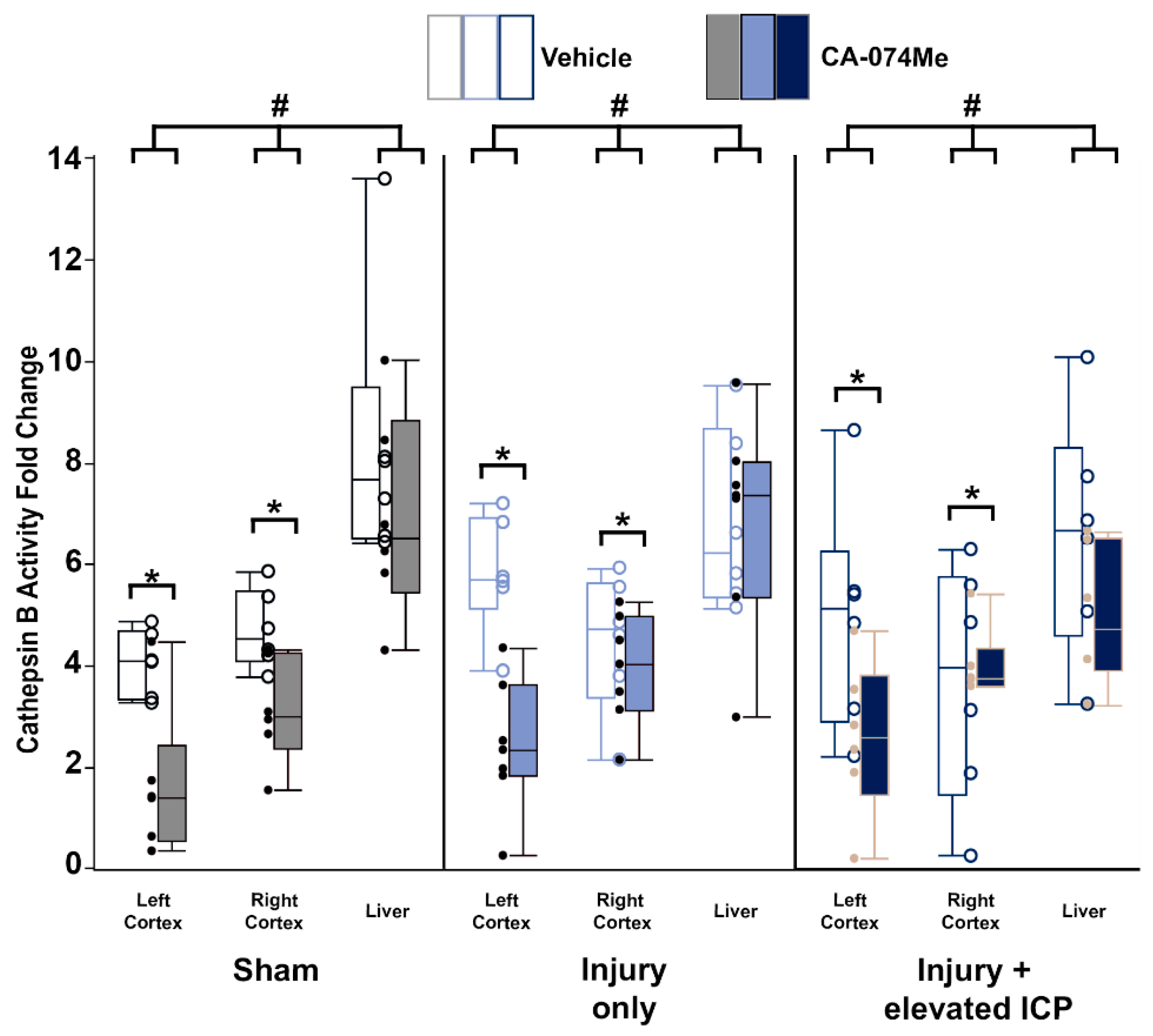
3.1. Cathepsin B activity Was Decreased in the Left and Right Cortex after 2w of Continuous CA-074Me infusion
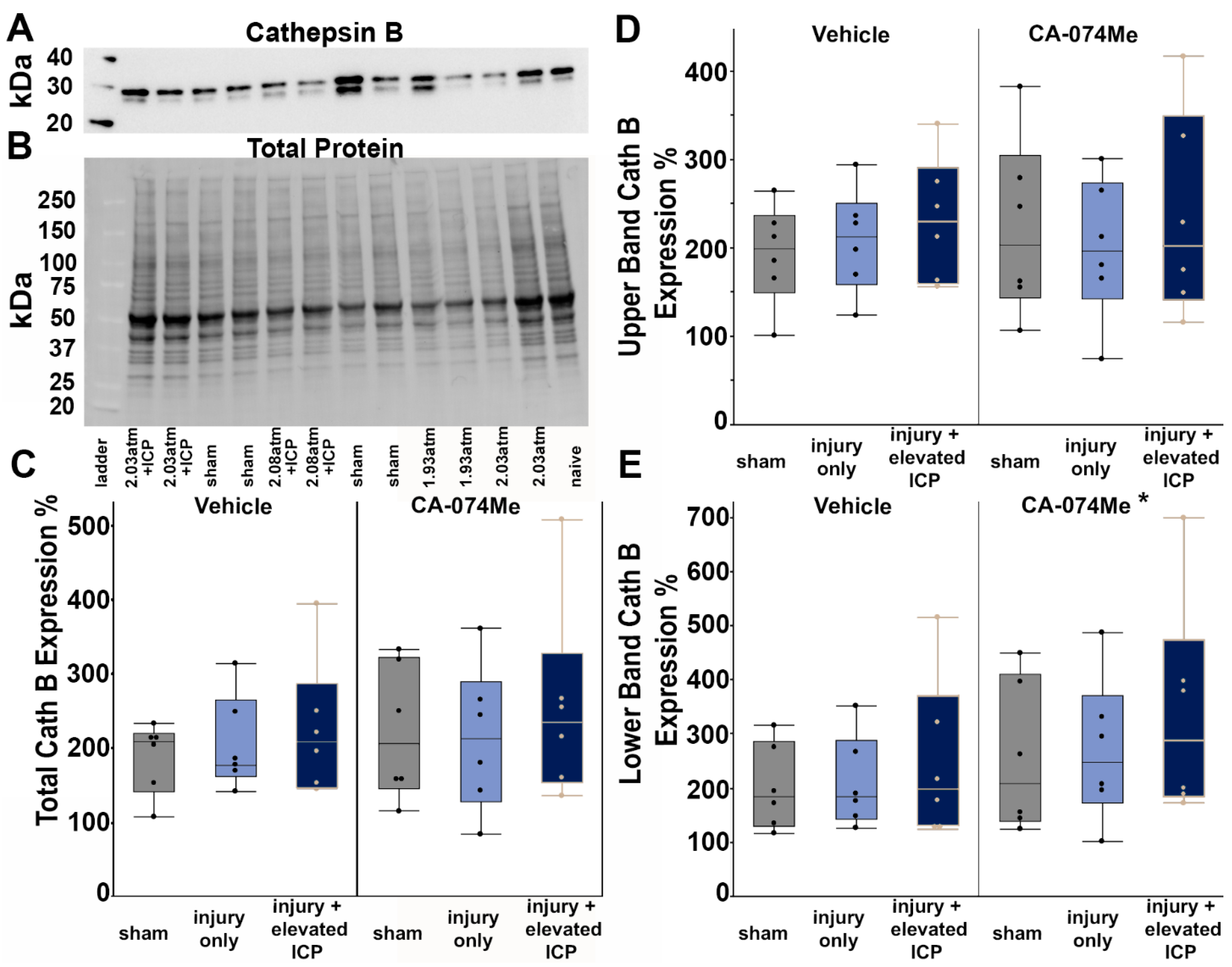
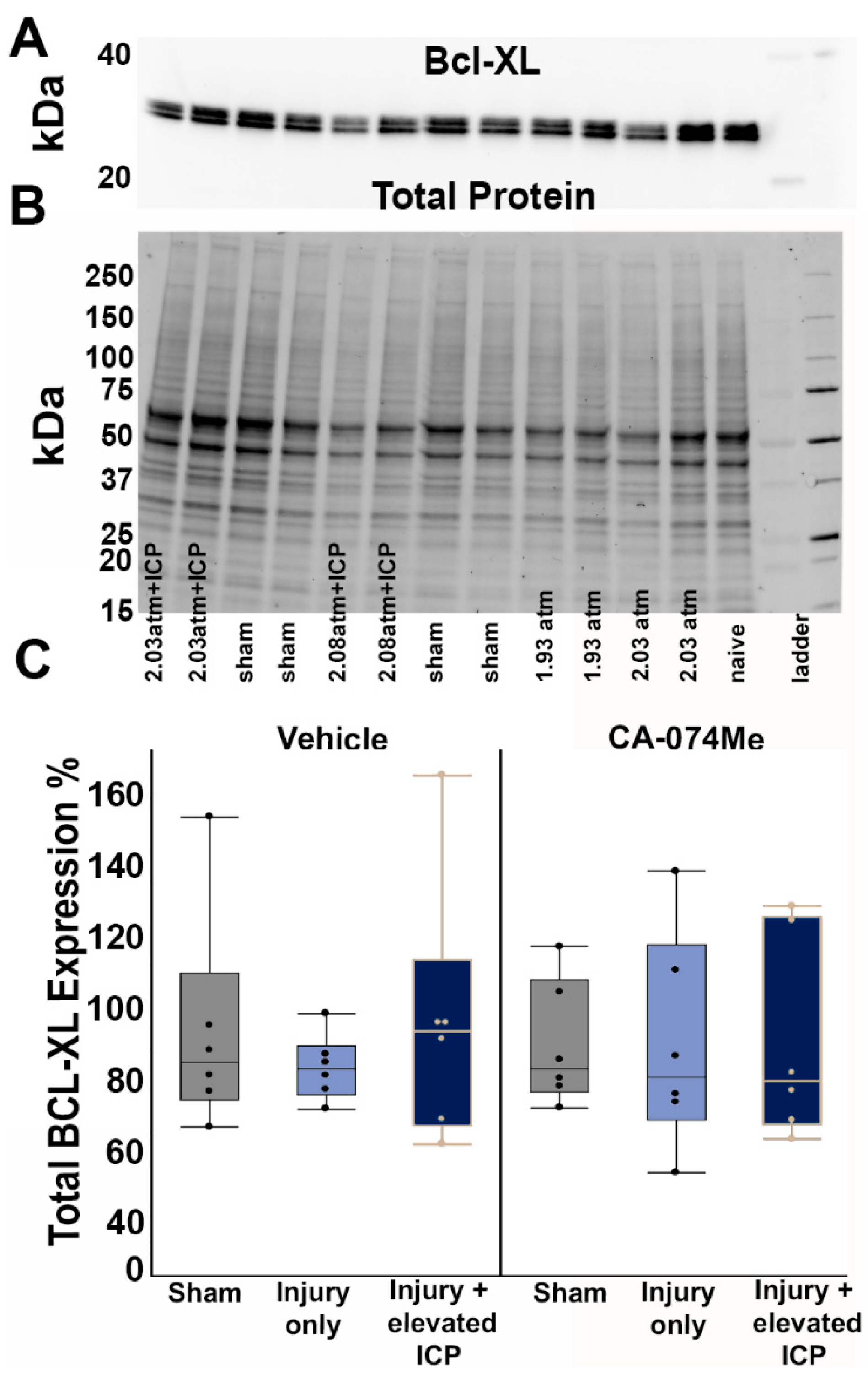
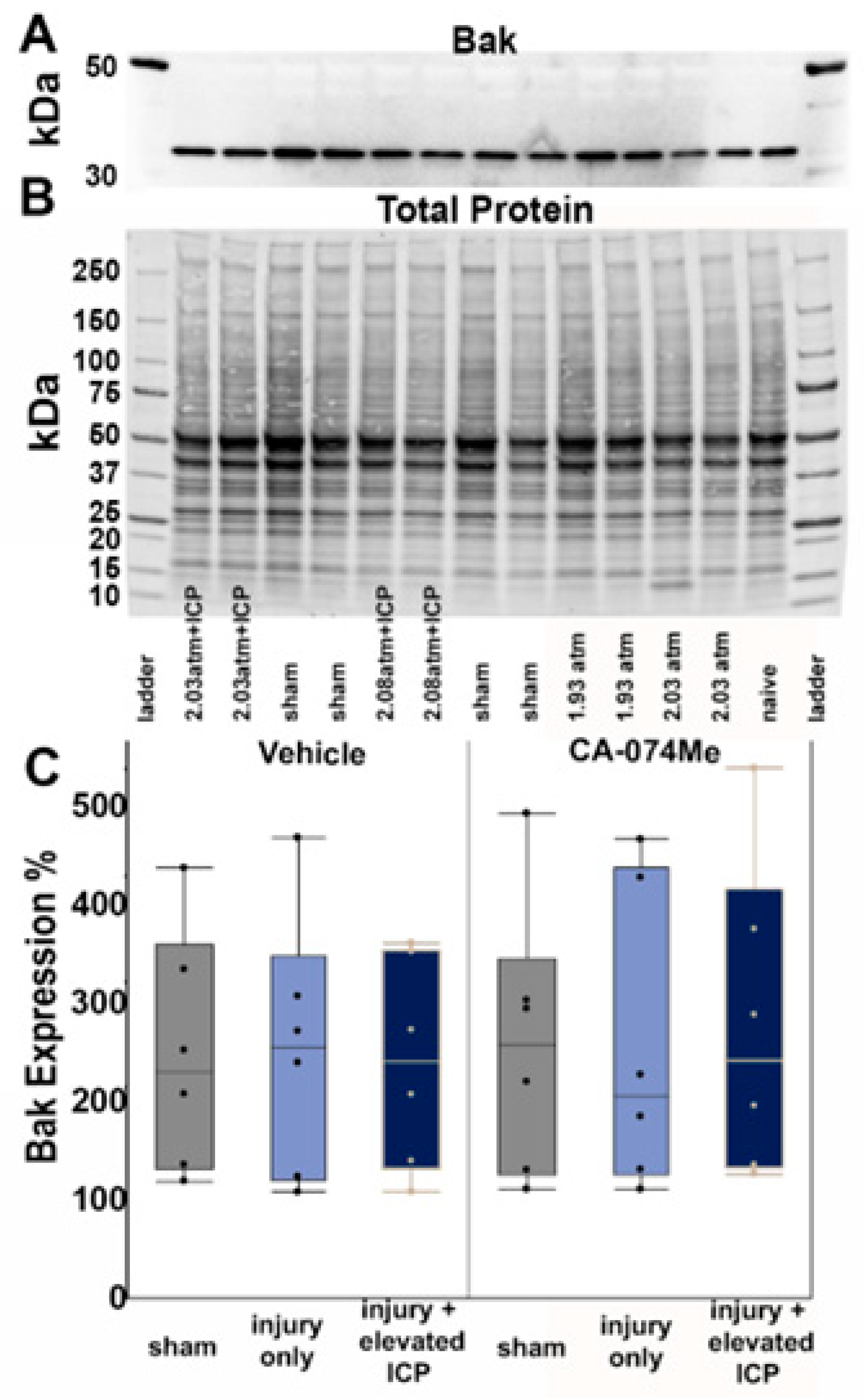
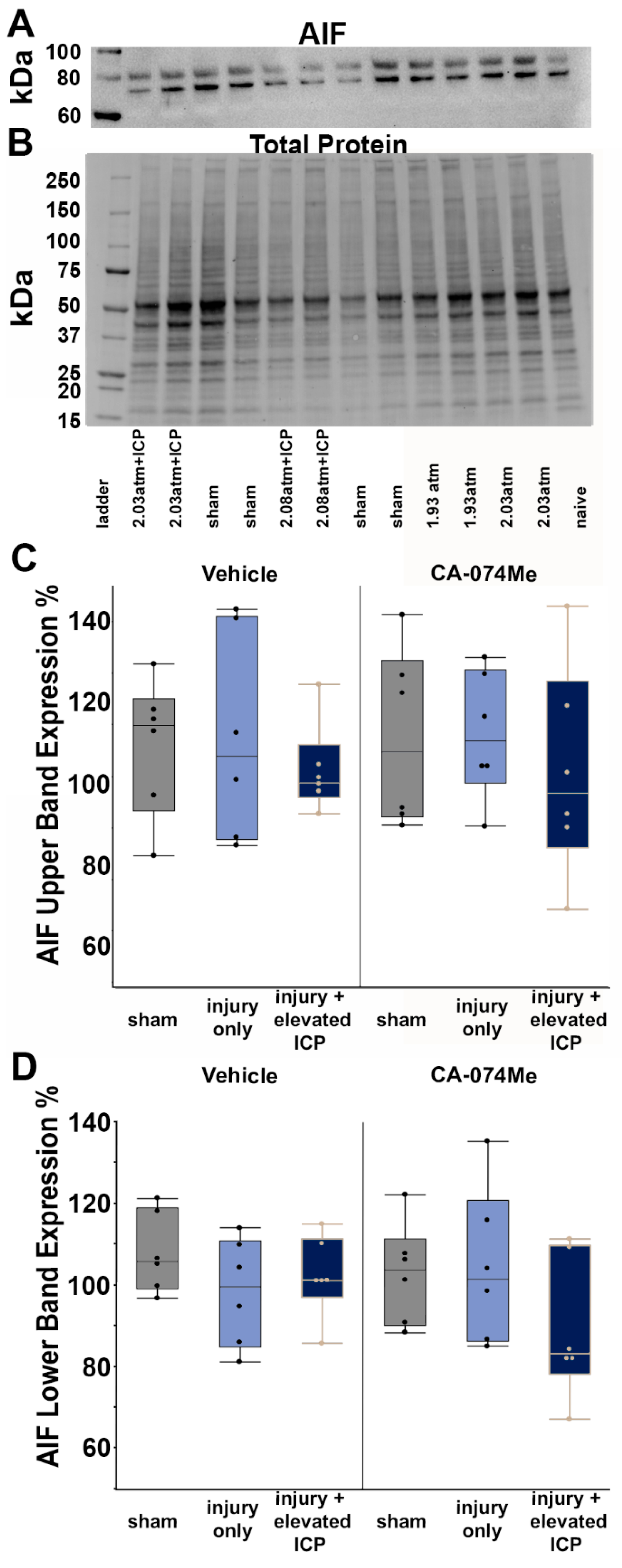
3.2. Protein Expression of Cathepsin B and Other Signaling Partners; Bcl-XL, Bak, and AIF
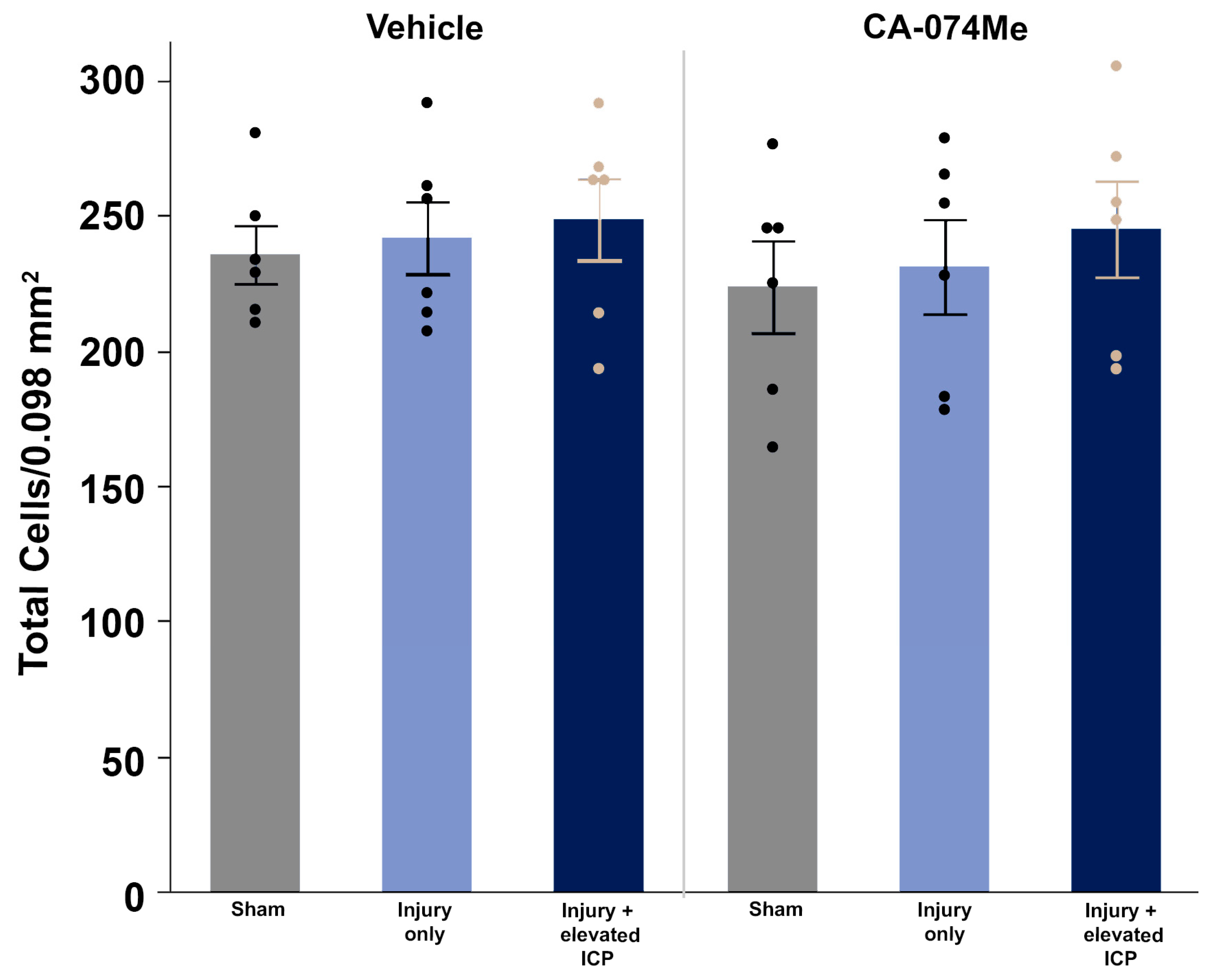
3.3. Elevation of ICP and/or Infusion of CA-074Me Did Not Impact the Number of Neurons in the Lateral Neocortex Following CFPI
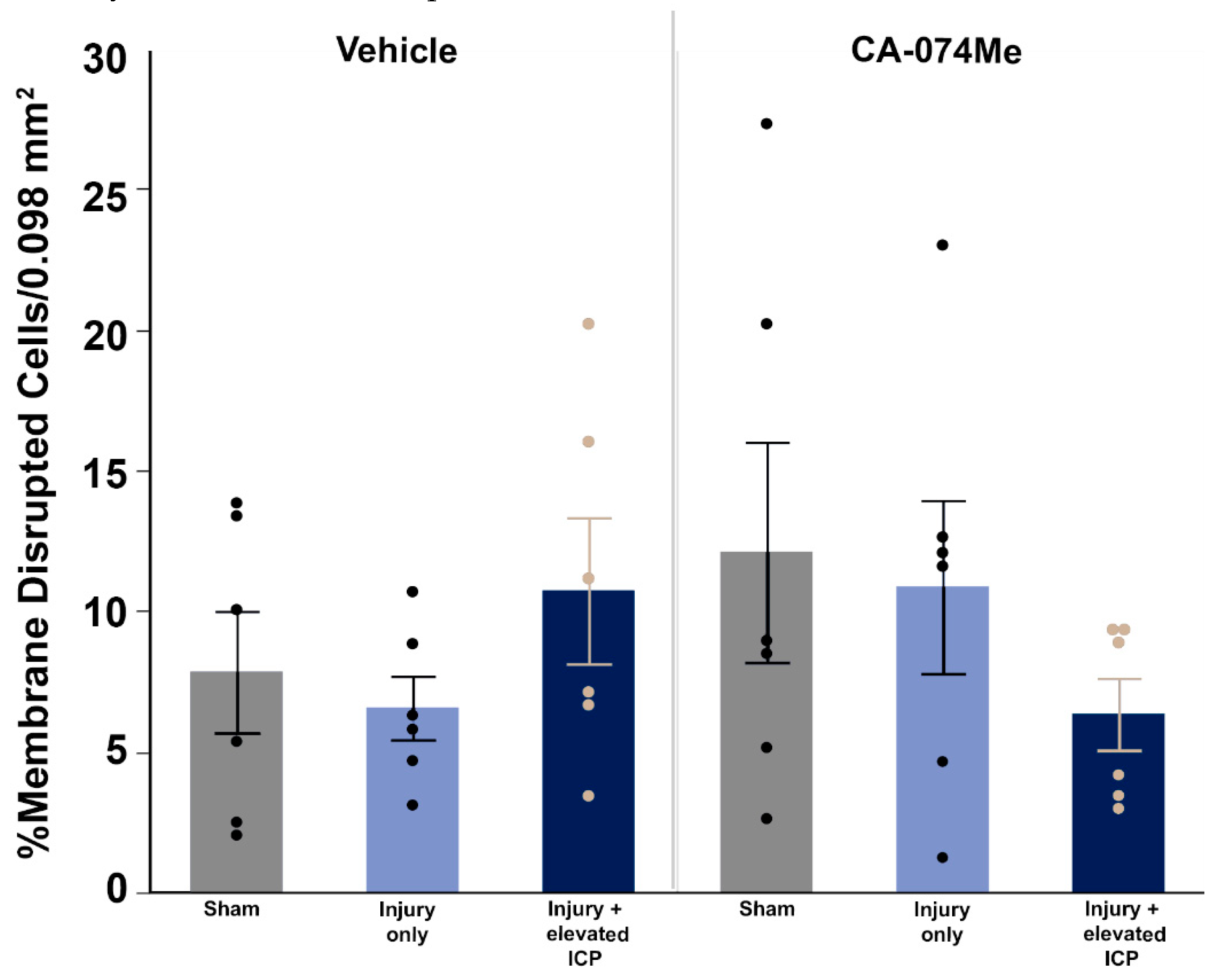
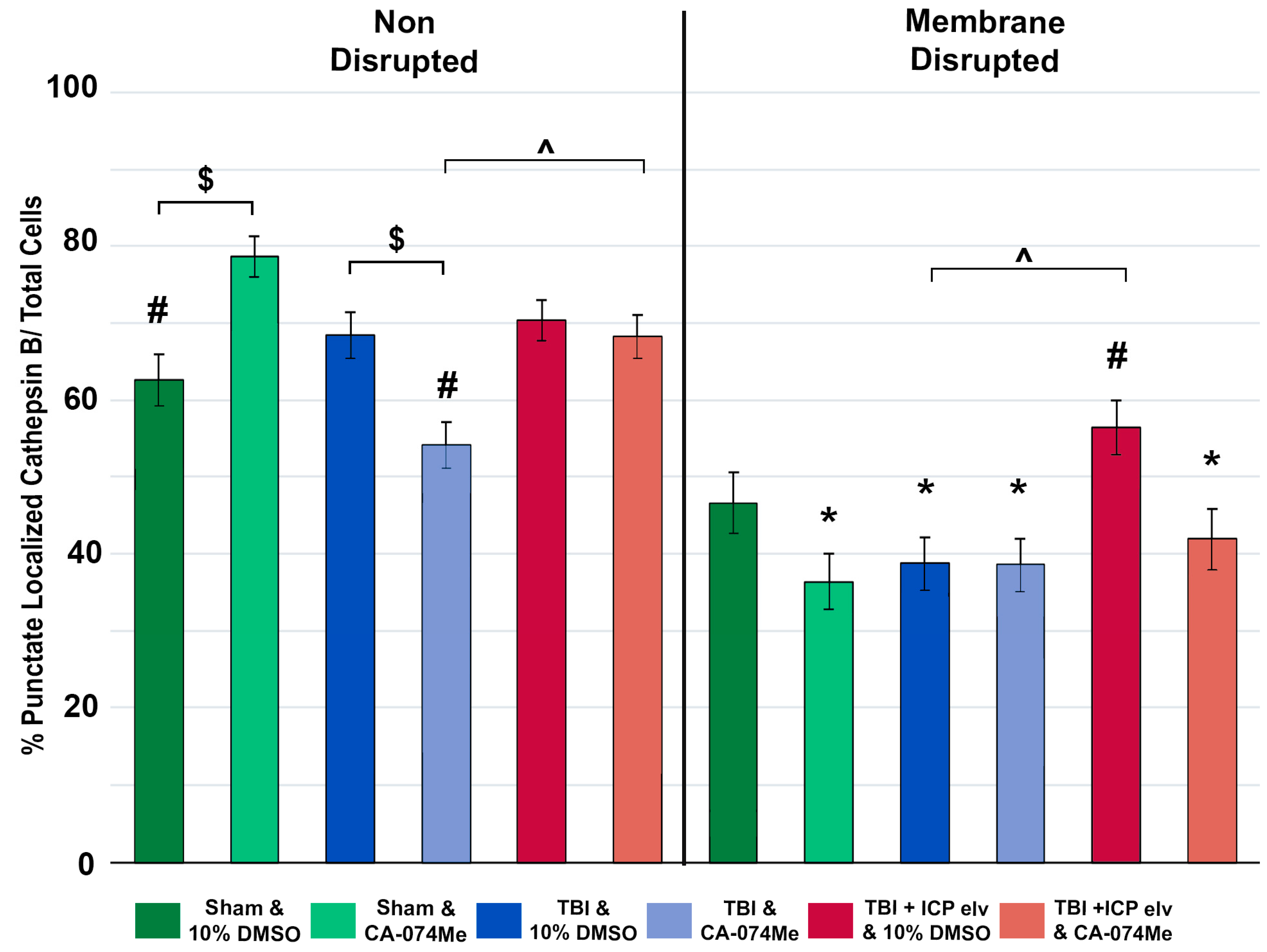
3.4. Neuronal Membrane Disruption Was Not Significantly Altered Following CFPI in the Presence of 10% DMSO or CA-074Me Regardless of ICP Elevation
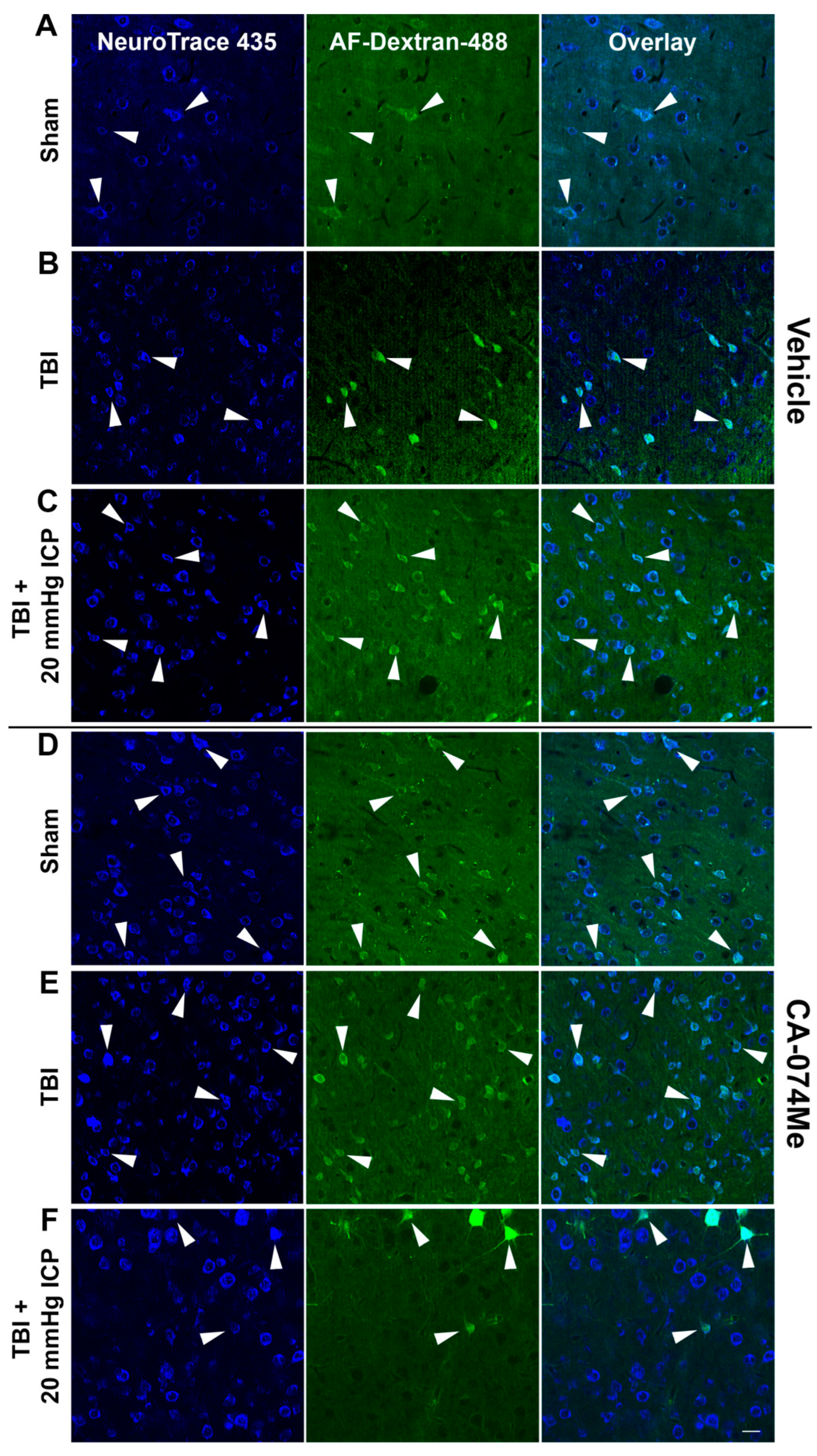
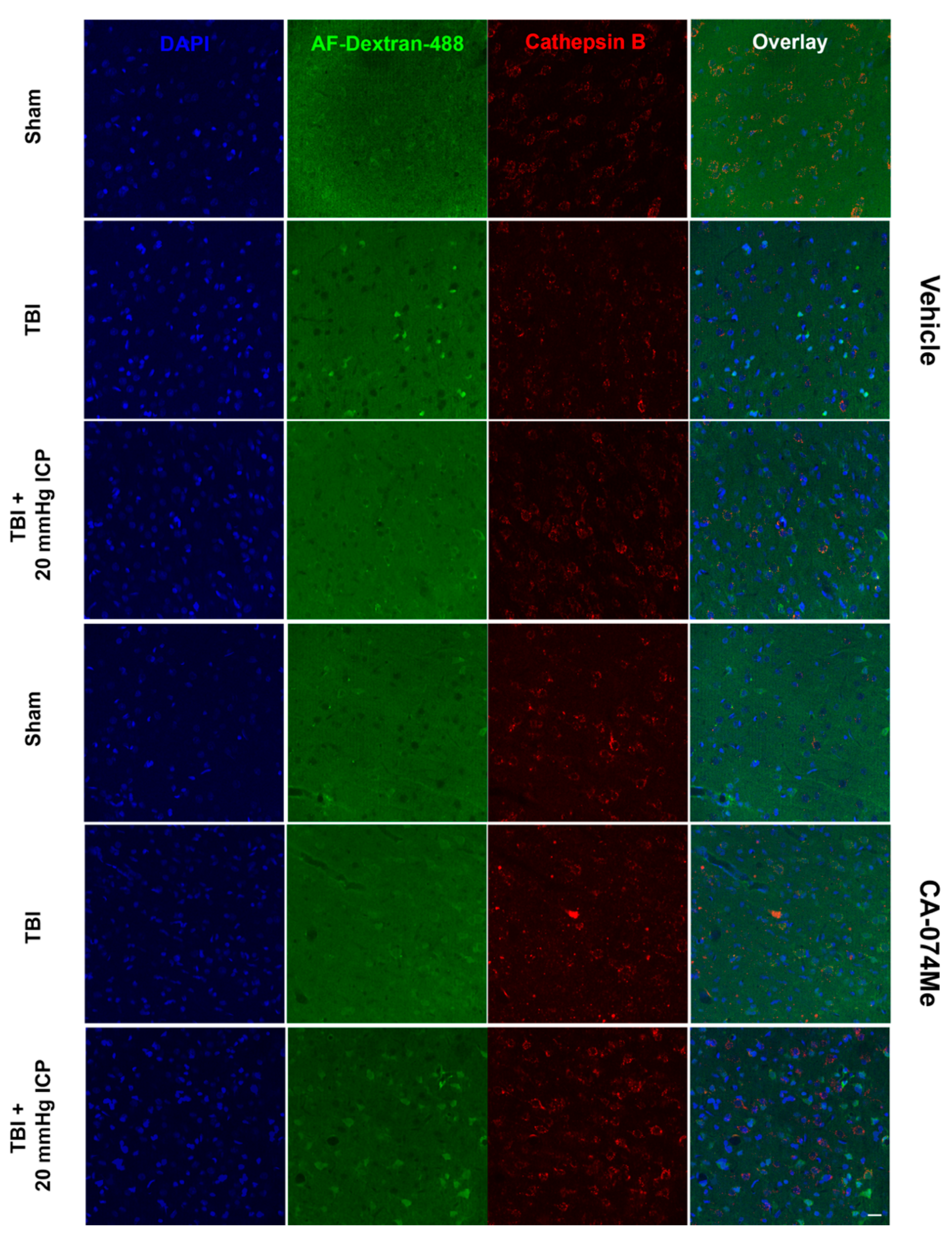
3.5. Cathepsin B Re-Localizes from Lysosomes to Cytosol in Disrupted Neurons at 2 Weeks Following Injury
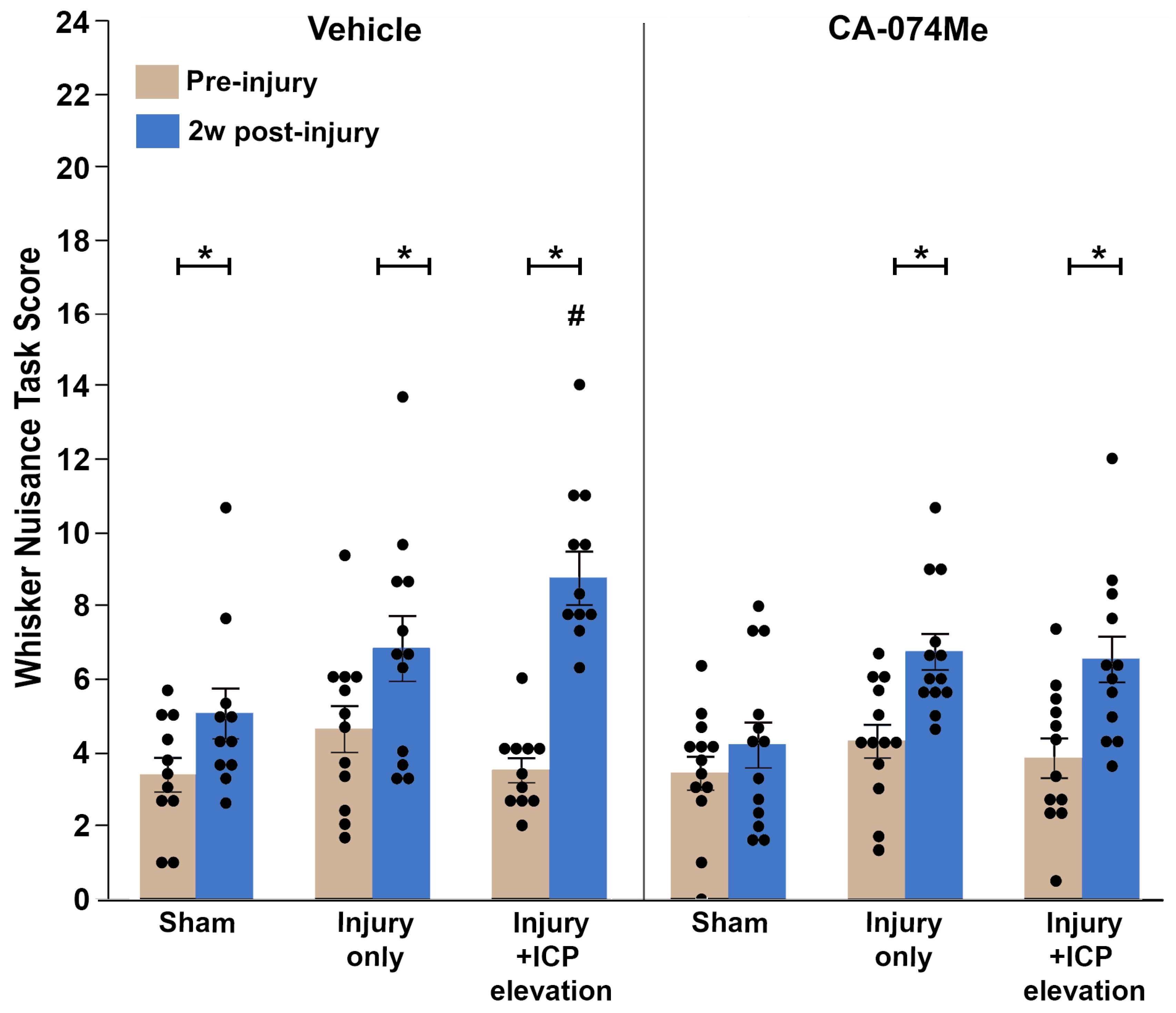
3.6. Secondary ICP Elevation Exacerbates Somatosensory Sensitivity at 2w Post-CFPI Which Is Ameliorated with CA-074Me Infusion
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maas, A.I.R.; Menon, D.K.; Manley, G.T.; Abrams, M.; Åkerlund, C.; Andelic, N.; Aries, M.; Bashford, T.; Bell, M.J.; Bodien, Y.G.; et al. Traumatic Brain Injury: Progress and Challenges in Prevention, Clinical Care, and Research. The Lancet Neurology 2022, 21, 1004–1060. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.A.; Bell, J.M.; Breiding, M.J.; Xu, L. Traumatic Brain Injury–Related Emergency Department Visits, Hospitalizations, and Deaths — United States, 2007 and 2013. MMWR. Surveillance Summaries 2017, 66, 1–16. [Google Scholar] [CrossRef] [PubMed]
- James, S.L.; Theadom, A.; Ellenbogen, R.G.; Bannick, M.S.; Montjoy-Venning, W.; Lucchesi, L.R.; Abbasi, N.; Abdulkader, R.; Abraha, H.N.; Adsuar, J.C.; et al. Global, Regional, and National Burden of Traumatic Brain Injury and Spinal Cord Injury, 1990–2016: A Systematic Analysis for the Global Burden of Disease Study 2016. The Lancet Neurology 2019, 18, 56–87. [Google Scholar] [CrossRef] [PubMed]
- Dams-O’Connor, K.; Juengst, S.B.; Bogner, J.; Chiaravalloti, N.D.; Corrigan, J.D.; Giacino, J.T.; Harrison-Felix, C.L.; Hoffman, J.M.; Ketchum, J.M.; Lequerica, A.H.; et al. Traumatic Brain Injury as a Chronic Disease: Insights from the United States Traumatic Brain Injury Model Systems Research Program. Lancet Neurol 2023, 22, 517–528. [Google Scholar] [CrossRef]
- Farahvar, A.; Gerber, L.M.; Chiu, Y.-L.; Carney, N.; Härtl, R.; Ghajar, J. Increased Mortality in Patients with Severe Traumatic Brain Injury Treated without Intracranial Pressure Monitoring. Journal of Neurosurgery 2012, 117, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.D.; Becker, D.P.; Ward, J.D.; Sullivan, H.G.; Adams, W.E.; Rosner, M.J. Significance of Intracranial Hypertension in Severe Head Injury. Journal of Neurosurgery 1977, 47, 503–516. [Google Scholar] [CrossRef]
- Lafrenaye, A.D.; Krahe, T.E.; Povlishock, J.T. Moderately Elevated Intracranial Pressure after Diffuse Traumatic Brain Injury Is Associated with Exacerbated Neuronal Pathology and Behavioral Morbidity in the Rat. Journal of Cerebral Blood Flow & Metabolism 2014, 34, 1628–1636. [Google Scholar] [CrossRef]
- Lafrenaye, A.D.; McGinn, M.J.; Povlishock, J.T. Increased Intracranial Pressure after Diffuse Traumatic Brain Injury Exacerbates Neuronal Somatic Membrane Poration but Not Axonal Injury: Evidence for Primary Intracranial Pressure-Induced Neuronal Perturbation. Journal of Cerebral Blood Flow & Metabolism 2012, 32, 1919–1932. [Google Scholar] [CrossRef] [PubMed]
- Cullen, D.K.; Vernekar, V.N.; LaPlaca, M.C. Trauma-Induced Plasmalemma Disruptions in Three-Dimensional Neural Cultures Are Dependent on Strain Modality and Rate. Journal of Neurotrauma 2011, 28, 2219–2233. [Google Scholar] [CrossRef]
- Farkas, O.; Lifshitz, J.; Povlishock, J.T. Mechanoporation Induced by Diffuse Traumatic Brain Injury: An Irreversible or Reversible Response to Injury? Journal of Neuroscience 2006, 26, 3130–3140. [Google Scholar] [CrossRef]
- Geddes, D.M.; Cargill, R.S.; LaPlaca, M.C. Mechanical Stretch to Neurons Results in a Strain Rate and Magnitude-Dependent Increase in Plasma Membrane Permeability. Journal of Neurotrauma 2003, 20, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- Geddes, D.M.; LaPlaca, M.C.; Cargill, R.S. Susceptibility of Hippocampal Neurons to Mechanically Induced Injury. Experimental Neurology 2003, 184, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.P.; Mietus, C.J.; Browne, K.D.; Wofford, K.L.; Keating, C.E.; Brown, D.P.; Johnson, B.N.; Wolf, J.A.; Smith, D.H.; Cohen, A.S.; et al. Neuronal Somatic Plasmalemmal Permeability and Dendritic Beading Caused by Head Rotational Traumatic Brain Injury in Pigs–An Exploratory Study. Frontiers in Cellular Neuroscience 2023, 17. [Google Scholar] [CrossRef] [PubMed]
- Keating, C.E.; Browne, K.D.; Duda, J.E.; Cullen, D.K. Neurons in Subcortical Oculomotor Regions Are Vulnerable to Plasma Membrane Damage after Repetitive Diffuse Traumatic Brain Injury in Swine. Journal of Neurotrauma 2020, 37, 1918–1932. [Google Scholar] [CrossRef] [PubMed]
- LaPlaca, M.C.; Lessing, M.C.; Prado, G.R.; Zhou, R.; Tate, C.C.; Geddes-Klein, D.; Meaney, D.F.; Zhang, L. Mechanoporation Is a Potential Indicator of Tissue Strain and Subsequent Degeneration Following Experimental Traumatic Brain Injury. Clinical Biomechanics 2019, 64, 2–13. [Google Scholar] [CrossRef] [PubMed]
- LaPlaca, M.C.; Thibault, L.E. Dynamic Mechanical Deformation of Neurons Triggers an Acute Calcium Response and Cell Injury Involving the N-Methyl-D-Aspartate Glutamate Receptor. Journal of Neuroscience Research 1998, 52, 220–229. [Google Scholar] [CrossRef]
- Prado, G.R.; LaPlaca, M.C. Neuronal Plasma Membrane Integrity Is Transiently Disturbed by Traumatic Loading. Neuroscience Insights 2020, 15, 263310552094609. [Google Scholar] [CrossRef]
- Singleton, R.H.; Povlishock, J.T. Identification and Characterization of Heterogeneous Neuronal Injury and Death in Regions of Diffuse Brain Injury: Evidence for Multiple Independent Injury Phenotypes. Journal of Neuroscience 2004, 24, 3543–3553. [Google Scholar] [CrossRef] [PubMed]
- Wofford, K.L.; Harris, J.P.; Browne, K.D.; Brown, D.P.; Grovola, M.R.; Mietus, C.J.; Wolf, J.A.; Duda, J.E.; Putt, M.E.; Spiller, K.L.; et al. Rapid Neuroinflammatory Response Localized to Injured Neurons after Diffuse Traumatic Brain Injury in Swine. Experimental Neurology 2017, 290, 85–94. [Google Scholar] [CrossRef]
- Hernandez, M.L.; Chatlos, T.; Gorse, K.M.; Lafrenaye, A.D. Neuronal Membrane Disruption Occurs Late Following Diffuse Brain Trauma in Rats and Involves a Subpopulation of NeuN Negative Cortical Neurons. Frontiers in Neurology 2019, 10. [Google Scholar] [CrossRef]
- Hernandez, M.L.; Marone, M.; Gorse, K.M.; Lafrenaye, A.D. Cathepsin B Relocalization in Late Membrane Disrupted Neurons Following Diffuse Brain Injury in Rats. ASN Neuro 2022, 14, 17590914221099112. [Google Scholar] [CrossRef] [PubMed]
- Chaitanya, G.V.; Babu, P.P. Multiple Apoptogenic Proteins Are Involved in the Nuclear Translocation of Apoptosis Inducing Factor during Transient Focal Cerebral Ischemia in Rat. Brain Research 2008, 1246, 178–190. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, K.D.; Sarkar, A.; Chatterjee, S.; Patra, D.; Sengupta, D.; Banerjee, S.; Chakraborty, P.; Sadhukhan, G.C. Cathepsin B Mediated Scramblase Activation Triggers Cytotoxicity and Cell Cycle Arrest by Andrographolide to Overcome Cellular Resistance in Cisplatin Resistant Human Hepatocellular Carcinoma HepG2 Cells. Environmental Toxicology and Pharmacology 2019, 68, 120–132. [Google Scholar] [CrossRef] [PubMed]
- de Castro, M.; Bunt, G.; Wouters, F. Cathepsin B Launches an Apoptotic Exit Effort upon Cell Death-Associated Disruption of Lysosomes. Cell Death Discovery 2016, 2, 16012. [Google Scholar] [CrossRef]
- Ellis, R.C.; O’Steen, W.A.; Hayes, R.L.; Nick, H.S.; Wang, K.K.W.; Anderson, D.K. Cellular Localization and Enzymatic Activity of Cathepsin B after Spinal Cord Injury in the Rat. Experimental Neurology 2005, 193, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Foghsgaard, L.; Wissing, D.; Mauch, D.; Lademann, U.; Bastholm, L.; Boes, M.; Elling, F.; Leist, M.; Jäättelä, M. Cathepsin B Acts as a Dominant Execution Protease in Tumor Cell Apoptosis Induced by Tumor Necrosis Factor. Journal of Cell Biology 2001, 153, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Guicciardi, M.E.; Deussing, J.; Miyoshi, H.; Bronk, S.F.; Svingen, P.A.; Peters, C.; Kaufmann, S.H.; Gores, G.J. Cathepsin B Contributes to TNF-α–Mediated Hepatocyte Apoptosis by Promoting Mitochondrial Release of Cytochrome c. Journal of Clinical Investigation 2000, 106, 1127–1137. [Google Scholar] [CrossRef] [PubMed]
- Mizunoe, Y.; Kobayashi, M.; Hoshino, S.; Tagawa, R.; Itagawa, R.; Hoshino, A.; Okita, N.; Sudo, Y.; Nakagawa, Y.; Shimano, H.; et al. Cathepsin B Overexpression Induces Degradation of Perilipin 1 to Cause Lipid Metabolism Dysfunction in Adipocytes. Scientific Reports 2020, 10. [Google Scholar] [CrossRef]
- Moles, A.; Tarrats, N.; Fernández-Checa, J.C.; Marí, M. Cathepsin B Overexpression Due to Acid Sphingomyelinase Ablation Promotes Liver Fibrosis in Niemann-Pick Disease. Journal of Biological Chemistry 2012, 287, 1178–1188. [Google Scholar] [CrossRef]
- Oberle, C.; Huai, J.; Reinheckel, T.; Tacke, M.; Rassner, M.; Ekert, P.G.; Buellesbach, J.; Borner, C. Lysosomal Membrane Permeabilization and Cathepsin Release Is a Bax/Bak-Dependent, Amplifying Event of Apoptosis in Fibroblasts and Monocytes. Cell Death and Differentiation 2010, 17, 1167–1178. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Zhang, X.; Xu, Y.; Chen, L.; Zhang, W.; Liu, E.; Xiao, C.; Kou, Q. Cathepsin B Aggravates Acute Pancreatitis by Activating the NLRP3 Inflammasome and Promoting the Caspase-1-Induced Pyroptosis. International Immunopharmacology 2021, 94, 107496. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.D.; Sheng, R.; Zhang, L.S.; Han, R.; Zhang, X.; Zhang, X.D.; Han, F.; Fukunaga, K.; Qin, Z.H. Neuronal Injury in Rat Model of Permanent Focal Cerebral Ischemia Is Associated with Activation of Autophagic and Lysosomal Pathways. Autophagy 2008, 4, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.-Q.; Xu, M.; Yuan, Y.; Li, F.-F.; Yang, Z.; Liu, Y.; Zhou, M.-Q.; Bian, Z.-Y.; Deng, W.; Gao, L.; et al. Cathepsin B Deficiency Attenuates Cardiac Remodeling in Response to Pressure Overload via TNF-α/ASK1/JNK Pathway. American Journal of Physiology-Heart and Circulatory Physiology 2015, 308, H1143–H1154. [Google Scholar] [CrossRef]
- Boutté, A.M.; Hook, V.; Thangavelu, B.; Sarkis, G.A.; Abbatiello, B.N.; Hook, G.; Jacobsen, J.S.; Robertson, C.S.; Gilsdorf, J.; Yang, Z.; et al. Penetrating Traumatic Brain Injury Triggers Dysregulation of Cathepsin B Protein Levels Independent of Cysteine Protease Activity in Brain and Cerebral Spinal Fluid. Journal of neurotrauma 2020, 37, 1574–1586. [Google Scholar] [CrossRef]
- Hook, G.R.; Yu, J.; Sipes, N.; Pierschbacher, M.D.; Hook, V.; Kindy, M.S. The Cysteine Protease Cathepsin B Is a Key Drug Target and Cysteine Protease Inhibitors Are Potential Therapeutics for Traumatic Brain Injury. Journal of Neurotrauma 2013, 31, 515–529. [Google Scholar] [CrossRef]
- Luo, C.-L.; Chen, X.-P.; Yang, R.; Sun, Y.-X.; Li, Q.-Q.; Bao, H.-J.; Cao, Q.-Q.; Ni, H.; Qin, Z.-H.; Tao, L.-Y. Cathepsin B Contributes to Traumatic Brain Injury-Induced Cell Death through a Mitochondria-Mediated Apoptotic Pathway. Journal of Neuroscience Research 2010, 88, 2847–2858. [Google Scholar] [CrossRef] [PubMed]
- Dixon, C.E.; Lyeth, B.G.; Povlishock, J.T.; Findling, R.L.; Hamm, R.J.; Marmarou, A.; Young, H.F.; Hayes, R.L. A Fluid Percussion Model of Experimental Brain Injury in the Rat. Journal of Neurosurgery 1987, 67, 110–119. [Google Scholar] [CrossRef]
- Yoon, M.C.; Solania, A.; Jiang, Z.; Christy, M.P.; Podvin, S.; Mosier, C.; Lietz, C.B.; Ito, G.; Gerwick, W.H.; Wolan, D.W.; et al. Selective Neutral pH Inhibitor of Cathepsin B Designed Based on Cleavage Preferences at Cytosolic and Lysosomal pH Conditions. ACS Chemical Biology 2021, 16, 1628–1643. [Google Scholar] [CrossRef] [PubMed]
- McNamara, K.C.S.S.; Lisembee, A.M.; Lifshitz, J. The Whisker Nuisance Task Identifies a Late-Onset, Persistent Sensory Sensitivity in Diffuse Brain-Injured Rats. Journal of Neurotrauma 2010, 27, 695–706. [Google Scholar] [CrossRef]
- Ryu, J.; Jeizan, P.; Ahmed, S.; Ehsan, S.; Jose, J.; Regan, S.; Gorse, K.; Kelliher, C.; Lafrenaye, A. Post-Injury Buprenorphine Administration Is Associated with Long-Term Region-Specific Glial Alterations in Rats. Pharmaceutics 2022, 14, 2068. [Google Scholar] [CrossRef]
- Boya, P.; Andreau, K.; Poncet, D.; Zamzami, N.; Perfettini, J.L.; Metivier, D.; Ojcius, D.M.; Jäättelä, M.; Kroemer, G. Lysosomal Membrane Permeabilization Induces Cell Death in a Mitochondrion-Dependent Fashion. Journal of Experimental Medicine 2003, 197, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Marshall, L.F.; Camp, P.E.; Bowers, S. Dimethyl Sulfoxide for the Treatment of Intracranial Hypertension: A Preliminary Trial. Neurosurgery 1984, 14, 659–663. [Google Scholar] [CrossRef] [PubMed]
- Waller, F.T.; Tanabe, C.T.; Paxton, H.D. Treatment of Elevated Intracranial Pressure with Dimethyl Sulfoxide. Annals of the New York Academy of Sciences 1983, 411, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Wolf, P.; Simon, M. Dimethyl Sulphoxide (DMSO) Induced Serum Hyperosmolality. Clinical Biochemistry 1983, 16, 261–262. [Google Scholar] [CrossRef] [PubMed]
- Di Giorgio, A.M.; Hou, Y.; Zhao, X.; Zhang, B.; Lyeth, B.G.; Russell, M.J. Dimethyl Sulfoxide Provides Neuroprotection in a Traumatic Brain Injury Model. Restorative Neurology and Neuroscience 2008, 26, 501–507. [Google Scholar] [PubMed]
- de la Torre, J.C.; Kawanaga, H.M.; Rowed, D.W.; Johnson, C.M.; Goode, D.J.; Kajihara, K.; Mullan, S. Dimethyl Sulfoxide in Central Nervous System Trauma. Annals of the New York Academy of Sciences 1975, 243, 362–389. [Google Scholar] [CrossRef] [PubMed]
- Tung, H.; James, H.E.; Laurin, R.; Marshall, L.F. Modification of the Effect of Dimethyl Sulfoxide on Intracranial Pressure, Brain Water, and Electrolyte Content by Indomethacin. Acta Neurochir (Wien) 1983, 68, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Karaca, M.; Bilgin, U.Y.; Akar, M.; de la Torre, J.C. Dimethly Sulphoxide Lowers ICP after Closed Head Trauma. Eur J Clin Pharmacol 1991, 40, 113–114. [Google Scholar] [CrossRef] [PubMed]
- Gurtovenko, A.A.; Anwar, J. Modulating the Structure and Properties of Cell Membranes: The Molecular Mechanism of Action of Dimethyl Sulfoxide. J. Phys. Chem. B 2007, 111, 10453–10460. [Google Scholar] [CrossRef]
- Notman, R.; Noro, M.; O’Malley, B.; Anwar, J. Molecular Basis for Dimethylsulfoxide (DMSO) Action on Lipid Membranes. J. Am. Chem. Soc. 2006, 128, 13982–13983. [Google Scholar] [CrossRef]
- Ménorval, M.-A. de; Mir, L.M.; Fernández, M.L.; Reigada, R. Effects of Dimethyl Sulfoxide in Cholesterol-Containing Lipid Membranes: A Comparative Study of Experiments In Silico and with Cells. PLOS ONE 2012, 7, e41733. [Google Scholar] [CrossRef] [PubMed]
- Shi, R.; Qiao, X.; Emerson, N.; Malcom, A. Dimethylsulfoxide Enhances CNS Neuronal Plasma Membrane Resealing after Injury in Low Temperature or Low Calcium. Journal of Neurocytology 2001, 30, 829–839. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



