Submitted:
17 May 2024
Posted:
23 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
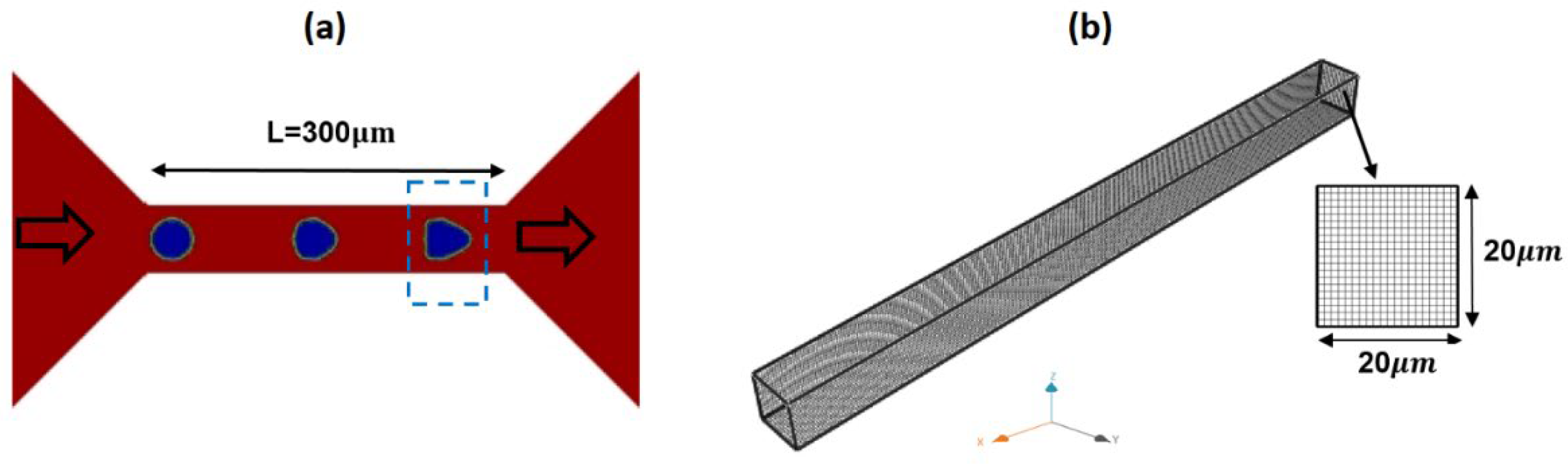
2. Numerical Method
3. Results and Discussions
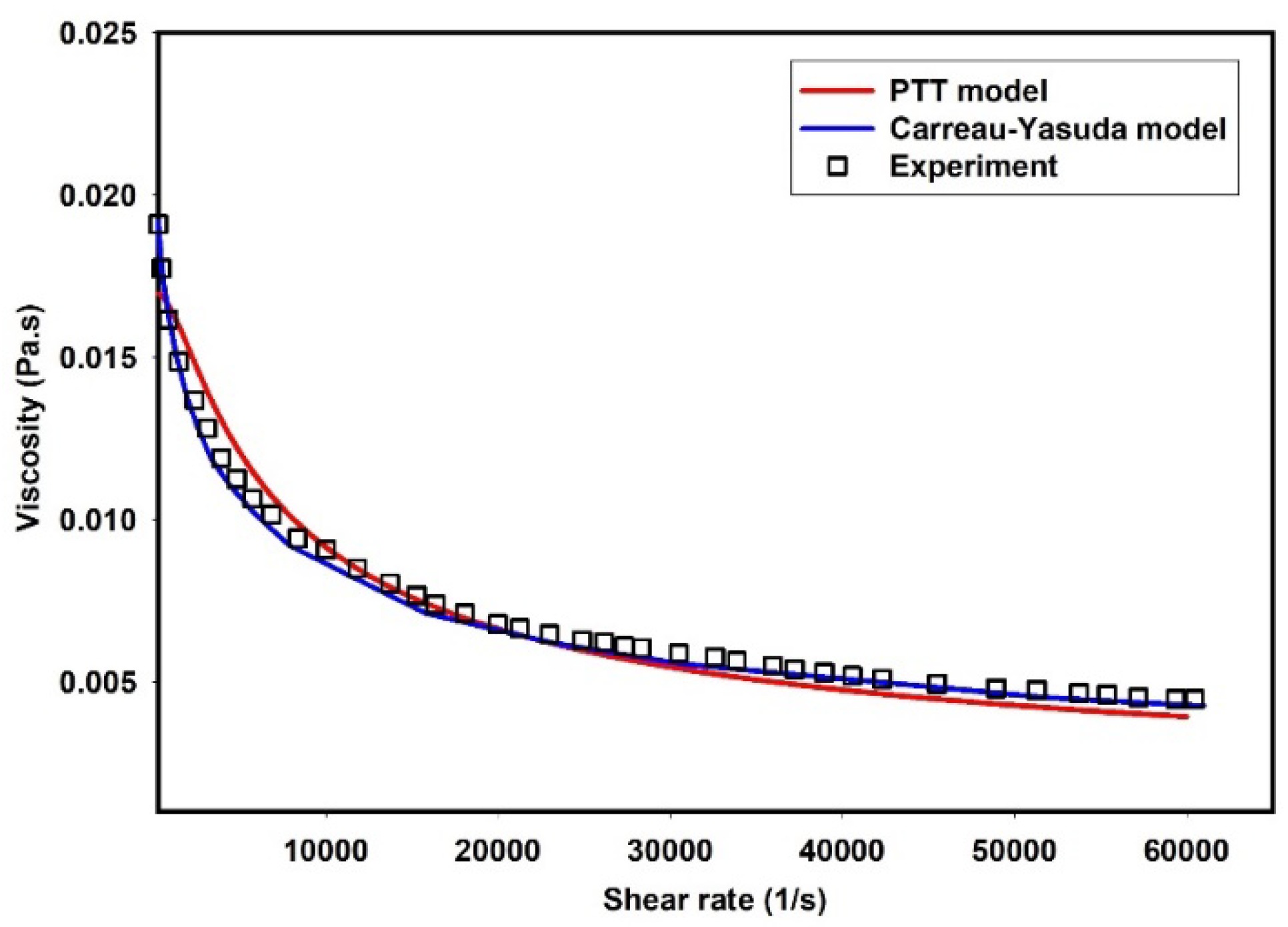
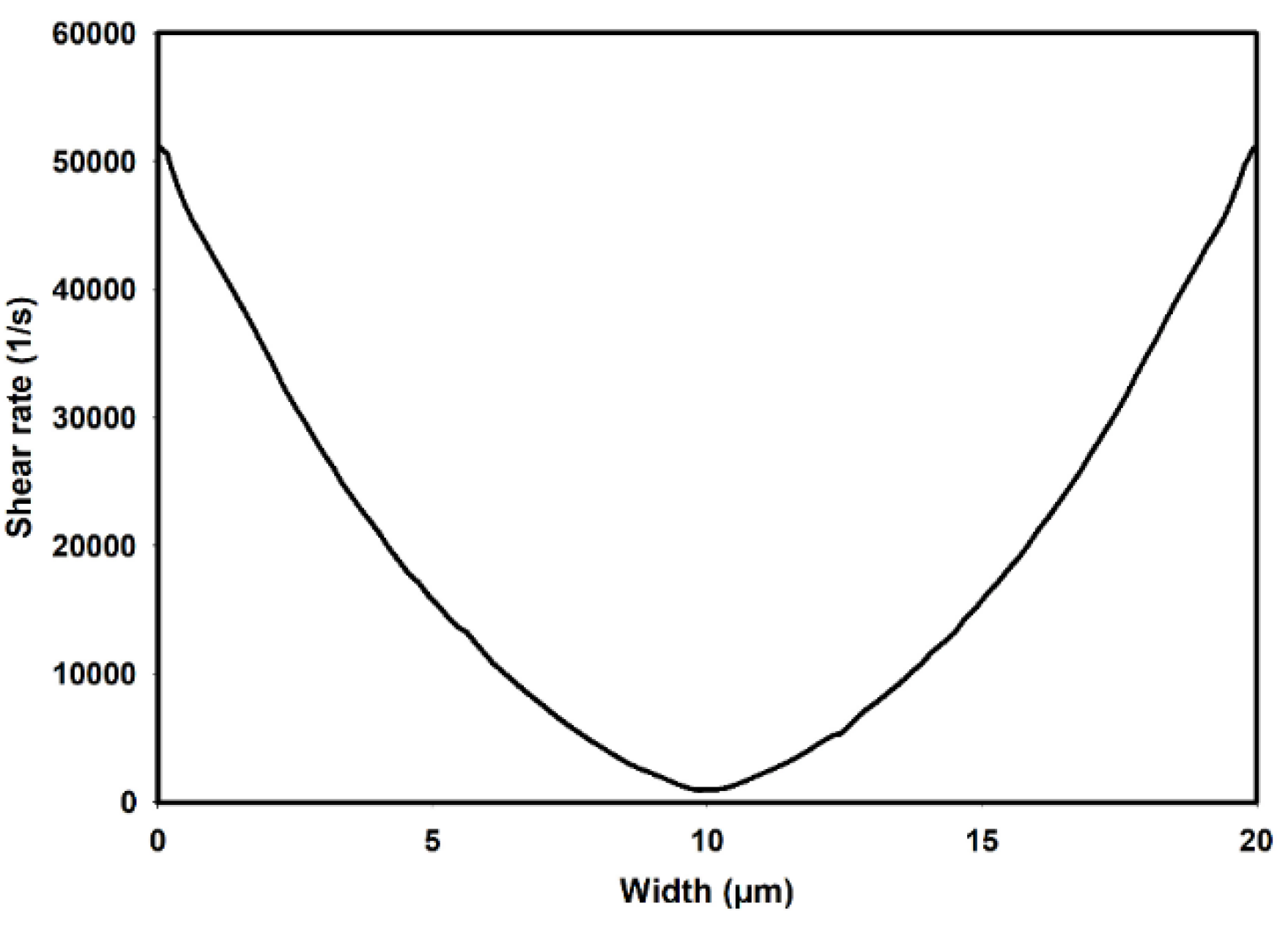
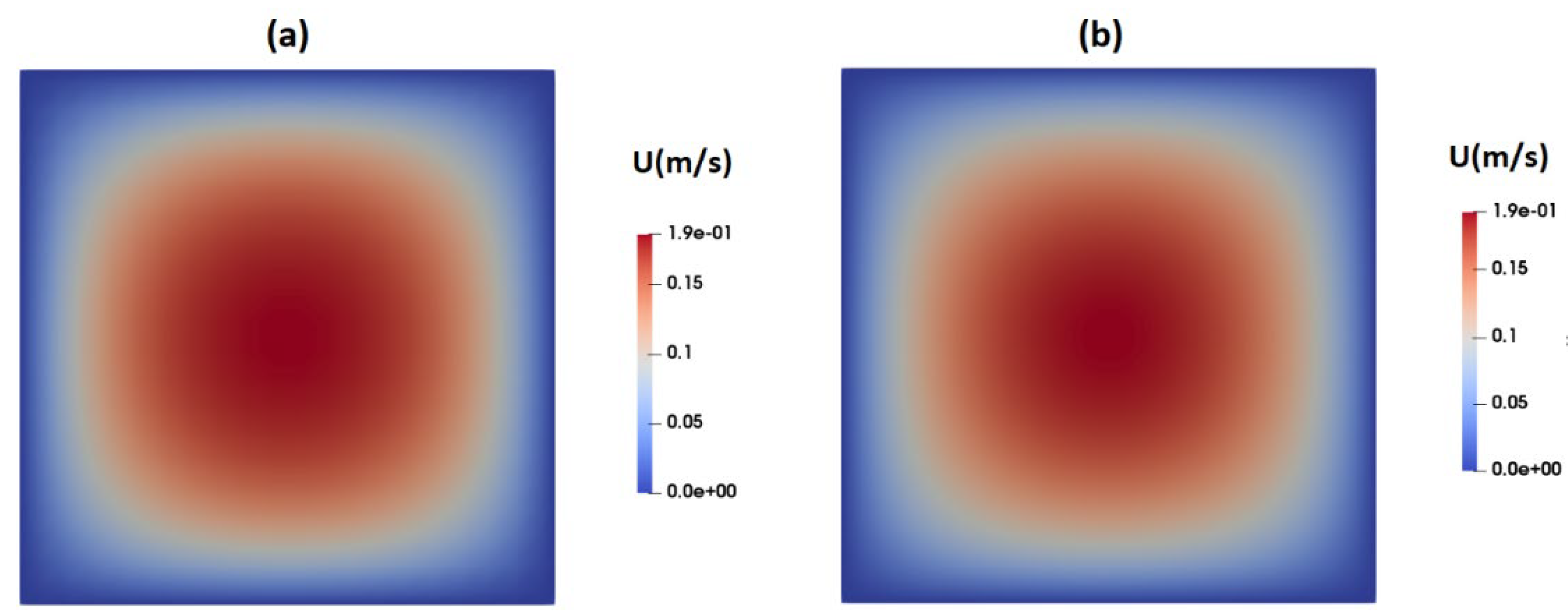
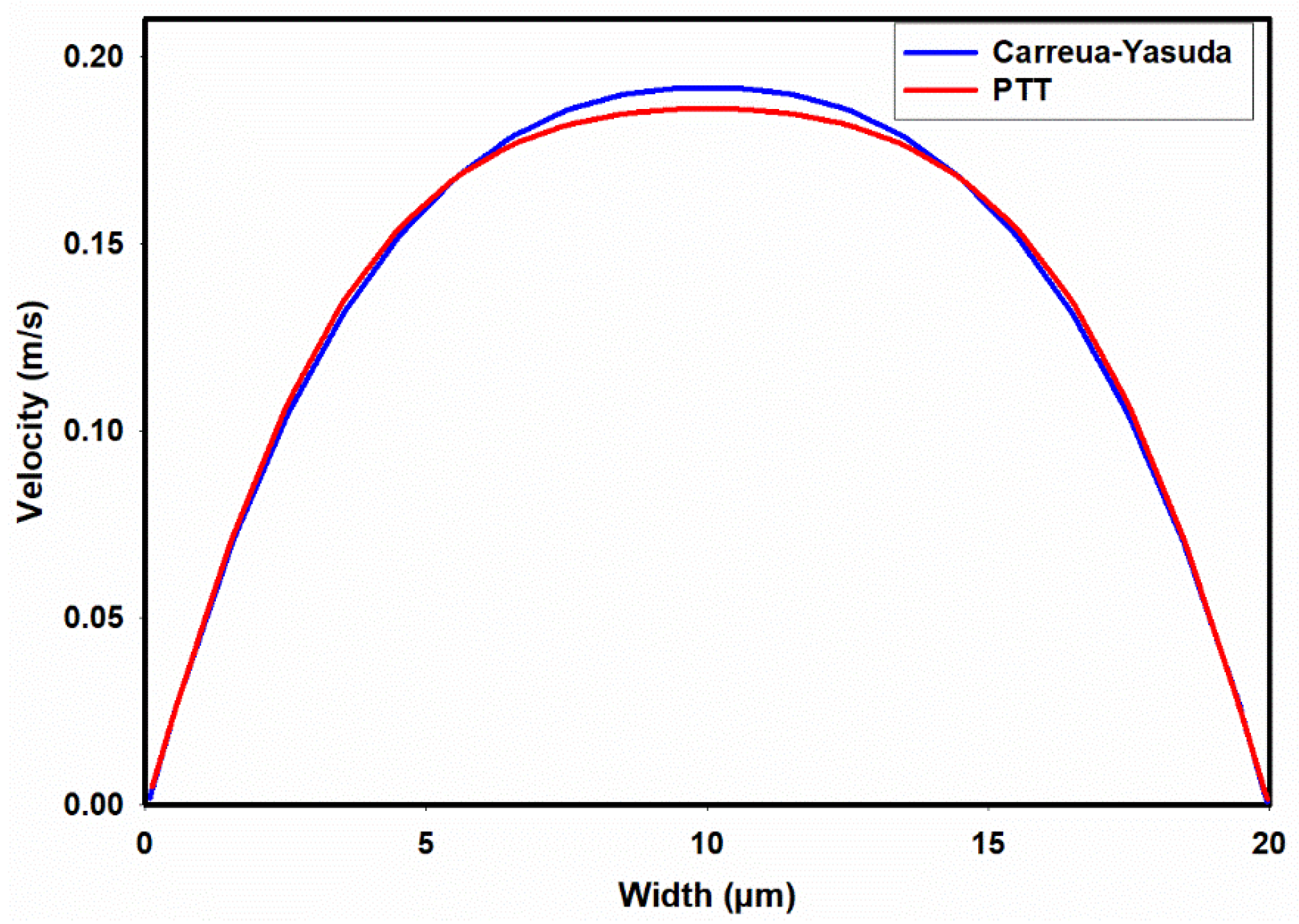
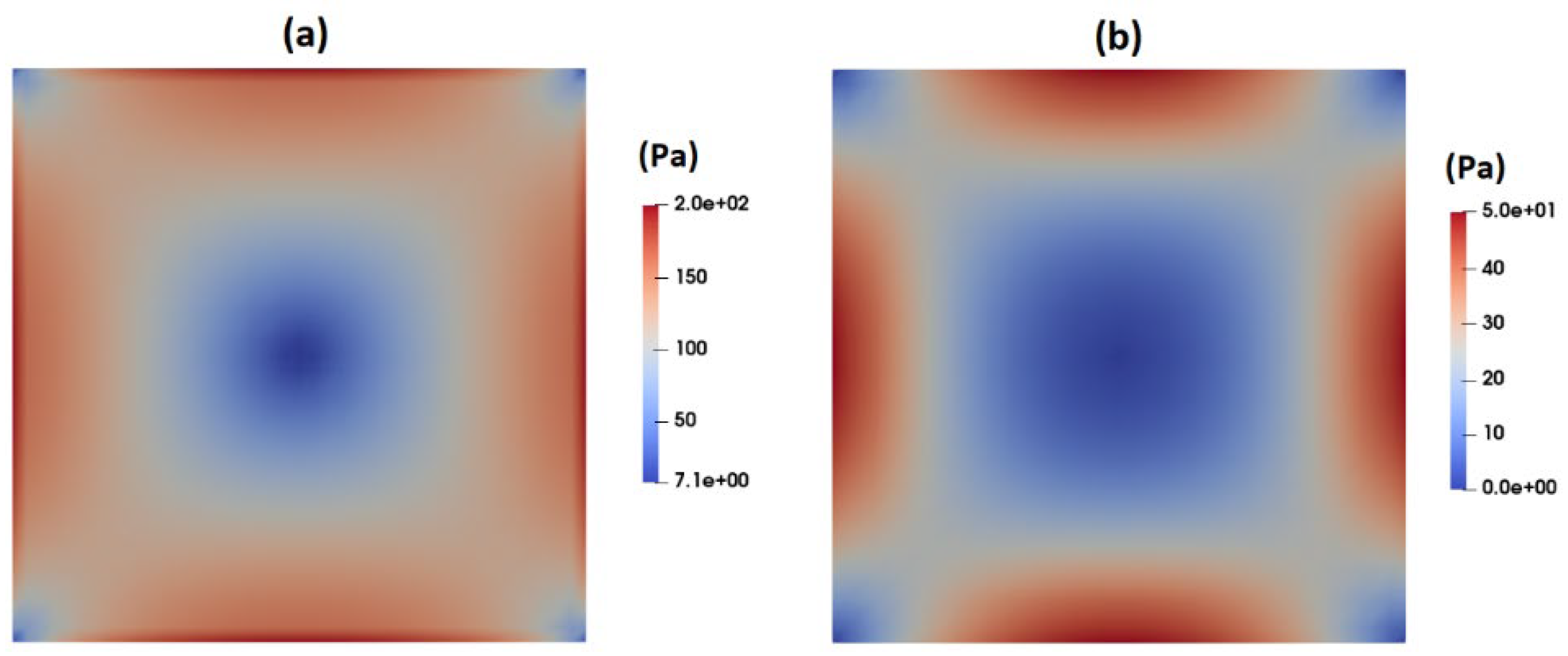
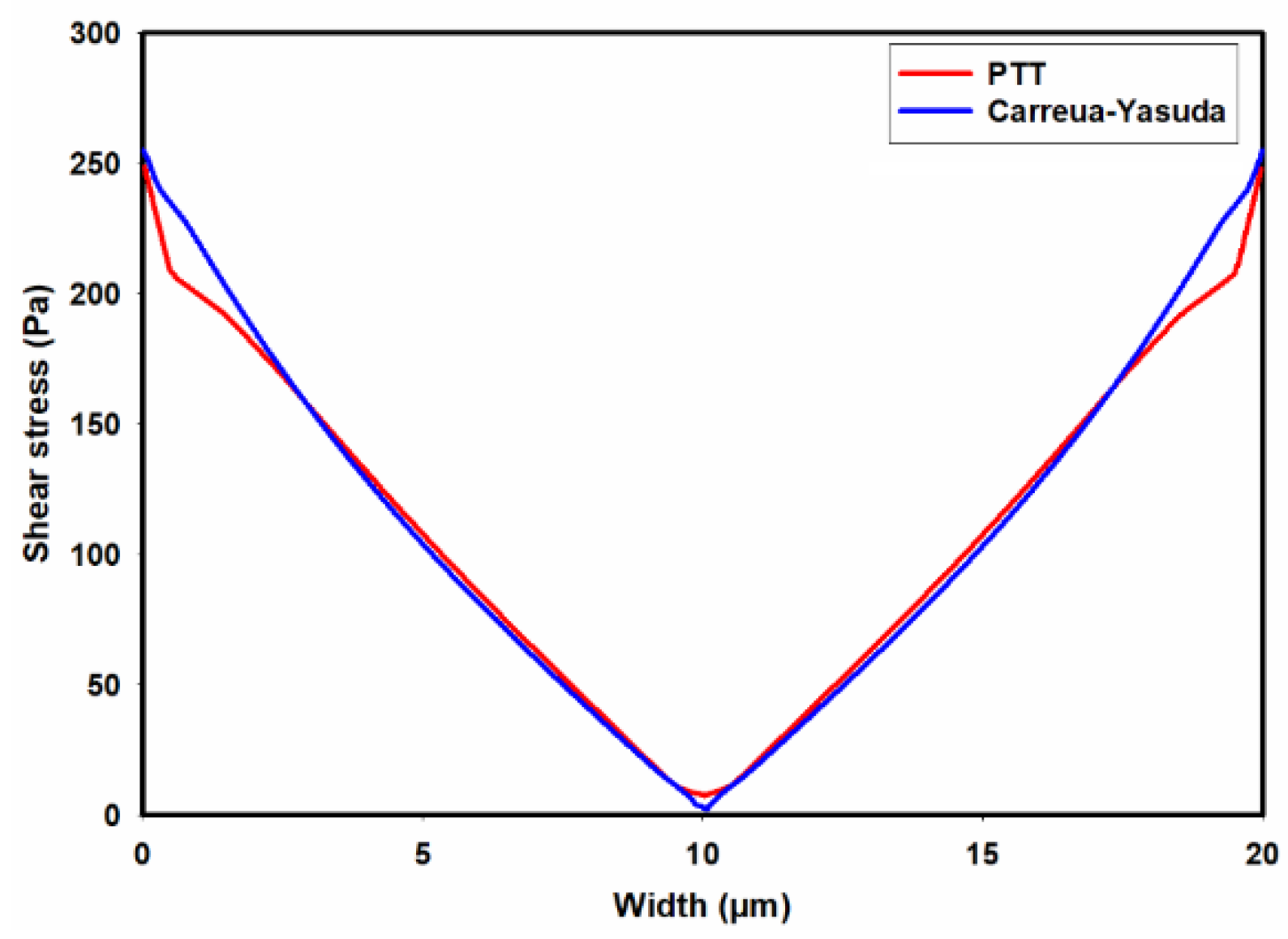
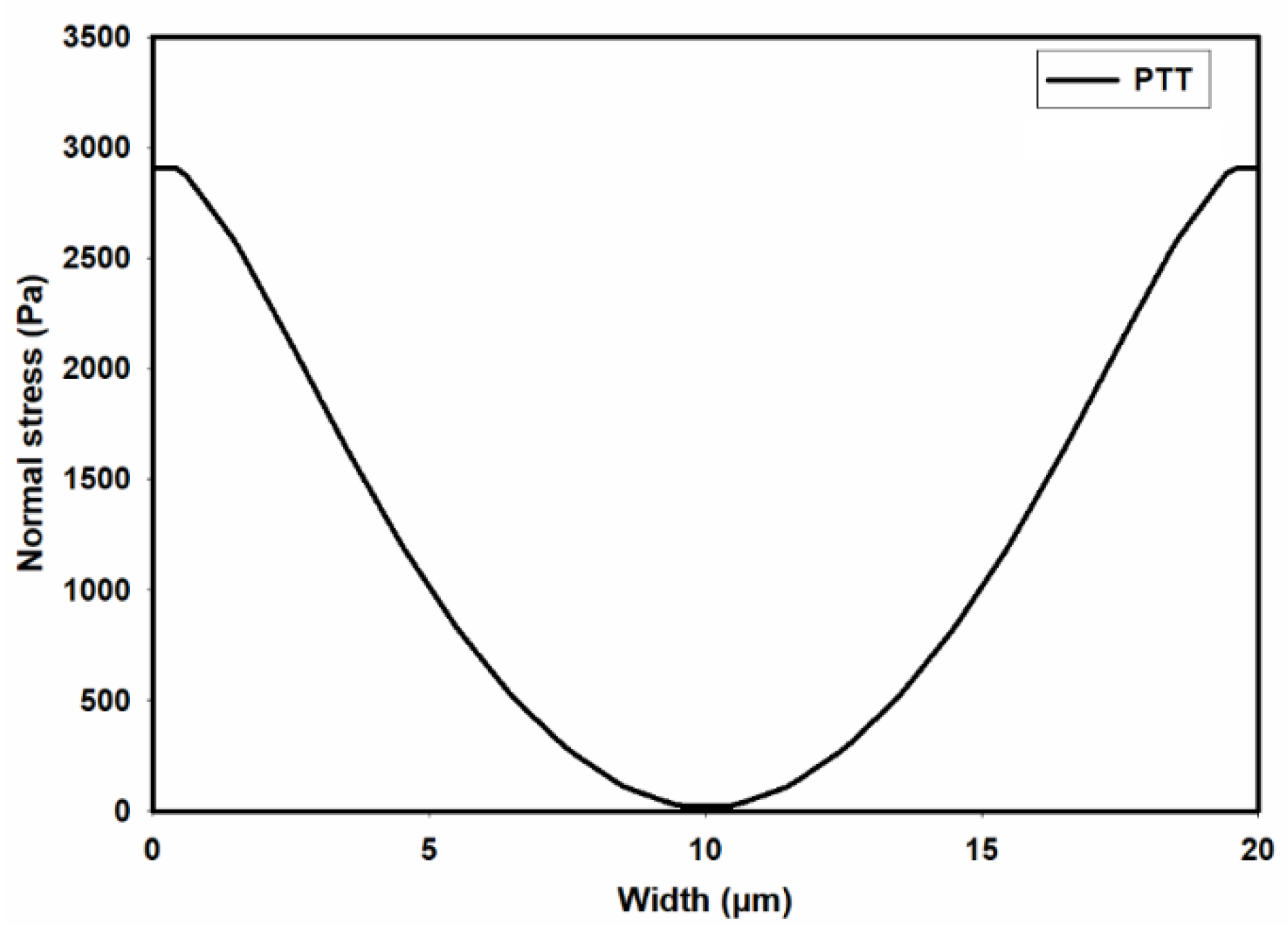
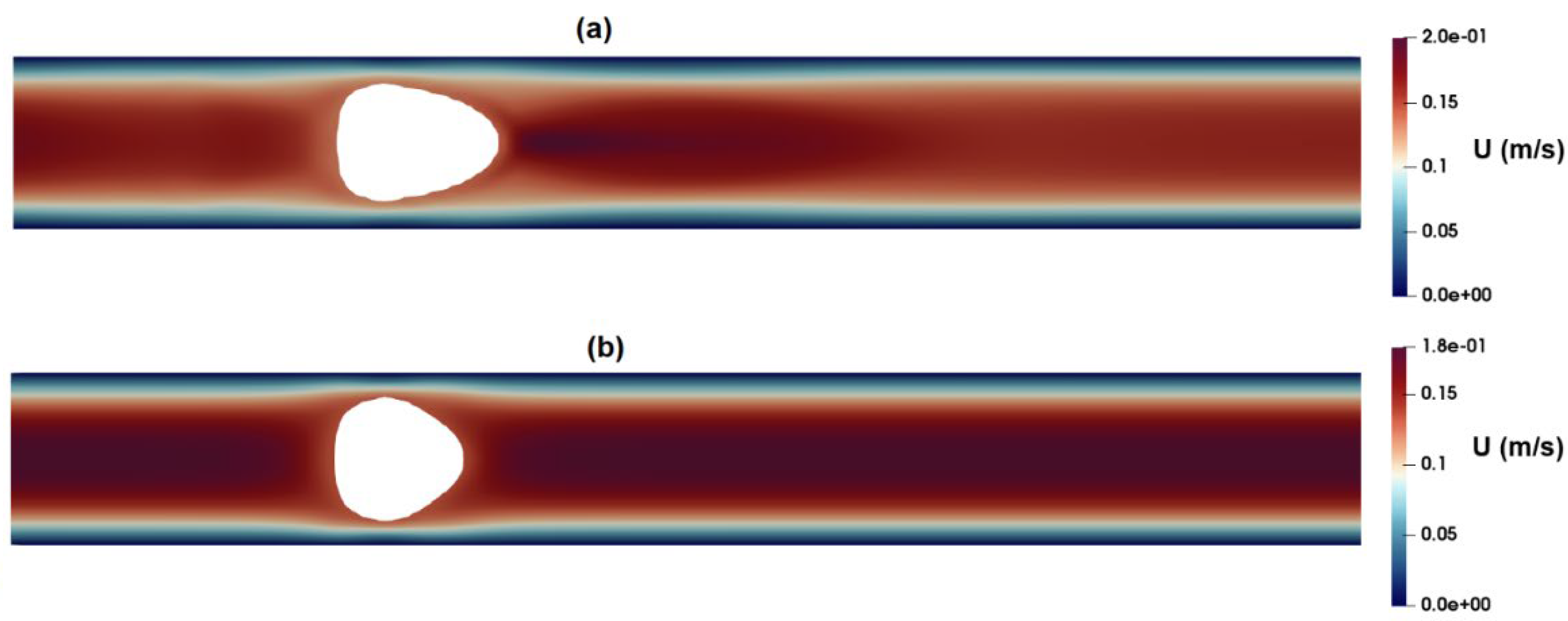
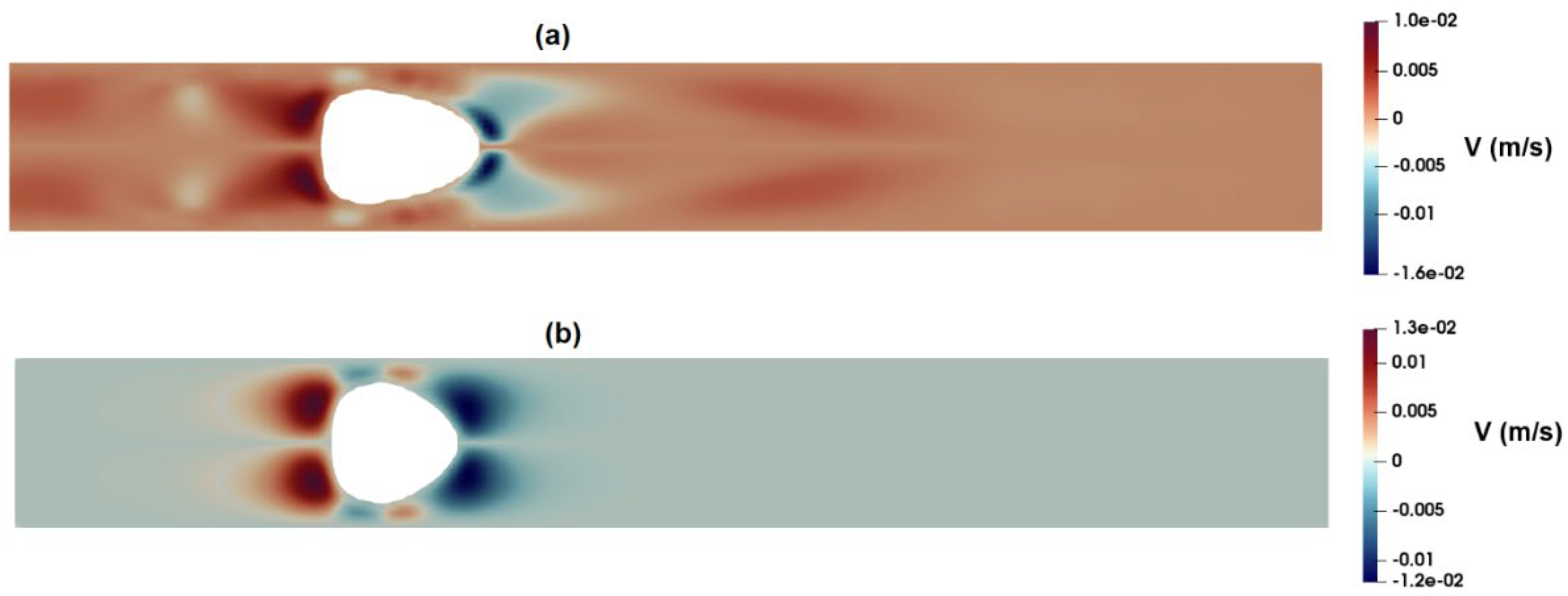
3.1. Flow-Field in the Microchannel Conveying Single-Phase MC-PBS Solution
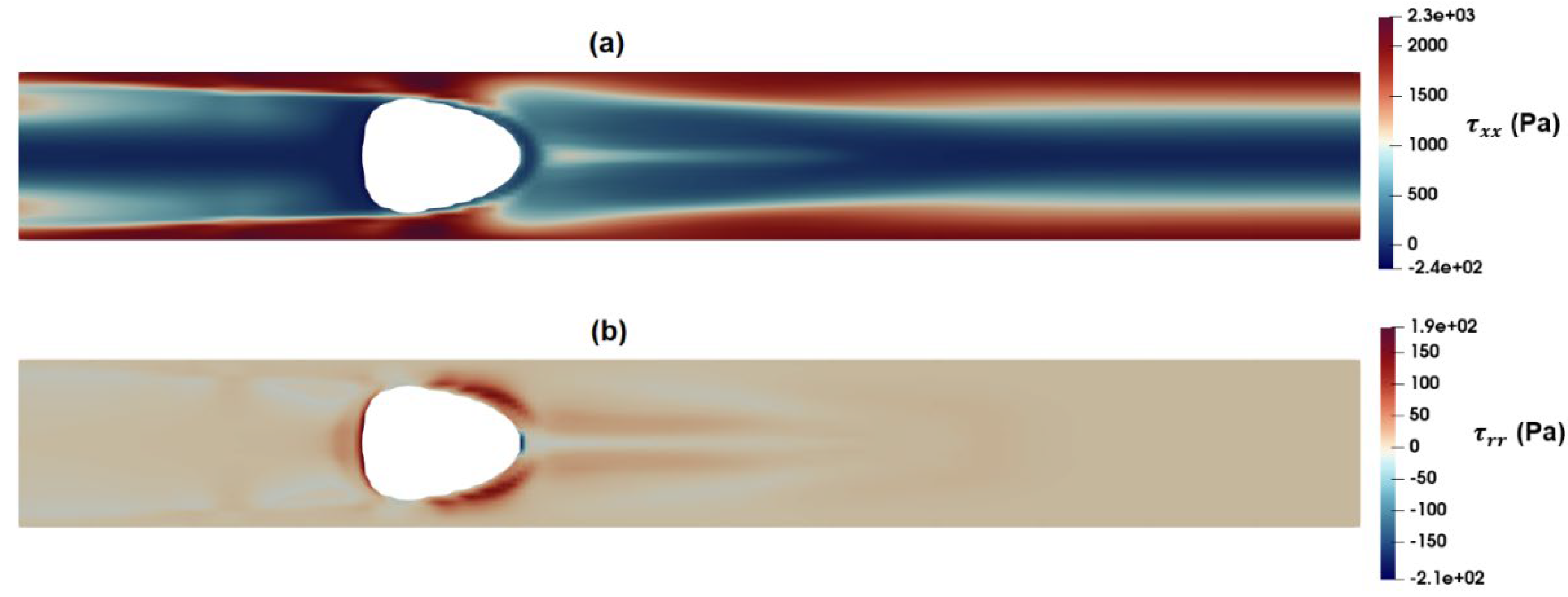
3.2. Numerical Modeling of Cell Deformation in a Hydrodynamic Constriction Channel
4. Conclusions
Conflicts of Interest
References
- Nematbakhsh, Y., & Lim, C. T. (2015). Cell biomechanics and its applications in human disease diagnosis. Acta Mechanica Sinica, 31, 268-273. [CrossRef]
- Darling, E. M., & Di Carlo, D. (2015). High-throughput assessment of cellular mechanical properties. Annual review of biomedical engineering, 17, 35-62.
- Otto, O., Rosendahl, P., Mietke, A., Golfier, S., Herold, C., Klaue, D., ... & Guck, J. (2015). Real-time deformability cytometry: on-the-fly cell mechanical phenotyping. Nature methods, 12(3), 199-202. [CrossRef]
- Guck, J., Schinkinger, S., Lincoln, B., Wottawah, F., Ebert, S., Romeyke, M., ... & Bilby, C. (2005). Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophysical journal, 88(5), 3689-3698.
- Swaminathan, V., Mythreye, K., O’Brien, E. T., Berchuck, A., Blobe, G. C., & Superfine, R. (2011). Mechanical stiffness grades metastatic potential in patient tumor cells and in cancer cell lines. Cancer research, 71(15), 5075-5080. [CrossRef]
- Tse, H. T., Gossett, D. R., Moon, Y. S., Masaeli, M., Sohsman, M., Ying, Y., ... & Di Carlo, D. (2013). Quantitative diagnosis of malignant pleural effusions by single-cell mechanophenotyping. Science translational medicine, 5(212), 212ra163-212ra163. [CrossRef]
- Bufi, N., Saitakis, M., Dogniaux, S., Buschinger, O., Bohineust, A., Richert, A., ... & Asnacios, A. (2015). Human primary immune cells exhibit distinct mechanical properties that are modified by inflammation. Biophysical journal, 108(9), 2181-2190. [CrossRef]
- Bashant, K. R., Vassallo, A., Herold, C., Berner, R., Menschner, L., Subburayalu, J., ... & Toepfner, N. (2019). Real-time deformability cytometry reveals sequential contraction and expansion during neutrophil priming. Journal of leukocyte biology, 105(6), 1143-1153.
- Ekpenyong, A. E., Whyte, G., Chalut, K., Pagliara, S., Lautenschläger, F., Fiddler, C., ... & Guck, J. (2012). Viscoelastic properties of differentiating blood cells are fate-and function-dependent.
- Lin, J., Kim, D., Tse, H. T., Tseng, P., Peng, L., Dhar, M., ... & Di Carlo, D. (2017). High-throughput physical phenotyping of cell differentiation. Microsystems & nanoengineering, 3(1), 1-7. [CrossRef]
- Guck, J., Ananthakrishnan, R., Mahmood, H., Moon, T. J., Cunningham, C. C., & Käs, J. (2001). The optical stretcher: a novel laser tool to micromanipulate cells. Biophysical journal, 81(2), 767-784. [CrossRef]
- Radmacher, M. (2007). Studying the mechanics of cellular processes by atomic force microscopy. Methods in cell biology, 83, 347-372.
- Hochmuth, R. M. (2000). Micropipette aspiration of living cells. Journal of biomechanics, 33(1), 15-22. [CrossRef]
- Thoumine, O., Ott, A., Cardoso, O., & Meister, J. J. (1999). Microplates: a new tool for manipulation and mechanical perturbation of individual cells. Journal of biochemical and biophysical methods, 39(1-2), 47-62. [CrossRef]
- Lange, J. R., Steinwachs, J., Kolb, T., Lautscham, L. A., Harder, I., Whyte, G., & Fabry, B. (2015). Microconstriction arrays for high-throughput quantitative measurements of cell mechanical properties. Biophysical journal, 109(1), 26-34. [CrossRef]
- Nyberg, K. D., Hu, K. H., Kleinman, S. H., Khismatullin, D. B., Butte, M. J., & Rowat, A. C. (2017). Quantitative deformability cytometry: rapid, calibrated measurements of cell mechanical properties. Biophysical journal, 113(7), 1574-1584. [CrossRef]
- Gossett, D. R., Tse, H. T., Lee, S. A., Ying, Y., Lindgren, A. G., Yang, O. O., ... & Di Carlo, D. (2012). Hydrodynamic stretching of single cells for large population mechanical phenotyping. Proceedings of the National Academy of Sciences, 109(20), 7630-7635. [CrossRef]
- Urbanska, M., Muñoz, H. E., Shaw Bagnall, J., Otto, O., Manalis, S. R., Di Carlo, D., & Guck, J. (2020). A comparison of microfluidic methods for high-throughput cell deformability measurements. Nature methods, 17(6), 587-593. [CrossRef]
- Guillou, L., Dahl, J. B., Lin, J. M. G., Barakat, A. I., Husson, J., Muller, S. J., & Kumar, S. (2016). Measuring cell viscoelastic properties using a microfluidic extensional flow device. Biophysical journal, 111(9), 2039-2050. [CrossRef]
- Armistead, F. J., De Pablo, J. G., Gadêlha, H., Peyman, S. A., & Evans, S. D. (2019). Cells under stress: an inertial-shear microfluidic determination of cell behavior. Biophysical Journal, 116(6), 1127-1135. [CrossRef]
- Mietke, A., Otto, O., Girardo, S., Rosendahl, P., Taubenberger, A., Golfier, S., ... & Fischer-Friedrich, E. (2015). Extracting cell stiffness from real-time deformability cytometry: theory and experiment. Biophysical journal, 109(10), 2023-2036. [CrossRef]
- Mokbel, M., Mokbel, D., Mietke, A., Traber, N., Girardo, S., Otto, O., ... & Aland, S. (2017). Numerical simulation of real-time deformability cytometry to extract cell mechanical properties. ACS Biomaterials Science & Engineering, 3(11), 2962-2973. [CrossRef]
- Wang, Z., Lu, R., Wang, W., Tian, F. B., Feng, J. J., & Sui, Y. (2023). A computational model for the transit of a cancer cell through a constricted microchannel. Biomechanics and Modeling in Mechanobiology, 1-15. [CrossRef]
- Büyükurgancı, B., Basu, S. K., Neuner, M., Guck, J., Wierschem, A., & Reichel, F. (2023). Shear rheology of methyl cellulose based solutions for cell mechanical measurements at high shear rates. Soft Matter, 19(9), 1739-1748. [CrossRef]
- Urbanska, M. (2022). Single-cell mechanical phenotyping across timescales and cell state transitions.
- Wu, P. H., Aroush, D. R. B., Asnacios, A., Chen, W. C., Dokukin, M. E., Doss, B. L., ... & Wirtz, D. (2018). A comparison of methods to assess cell mechanical properties. Nature methods, 15(7), 491-498. [CrossRef]
- Thien, N. P., & Tanner, R. I. (1977). A new constitutive equation derived from network theory. Journal of Non-Newtonian Fluid Mechanics, 2(4), 353-365. [CrossRef]
- Alves, M. A., Oliveira, P. J., & Pinho, F. T. (2021). Numerical methods for viscoelastic fluid flows. Annual Review of Fluid Mechanics, 53, 509-541. [CrossRef]
- Cruz, D. O. A., Pinho, F., & Oliveira, P. J. (2005). Analytical solutions for fully developed laminar flow of some viscoelastic liquids with a Newtonian solvent contribution. Journal of non-newtonian fluid mechanics, 132(1-3), 28-35. [CrossRef]
- Azaiez, J., Guénette, R., & Ait-Kadi, A. (1996). Numerical simulation of viscoelastic flows through a planar contraction. Journal of non-newtonian fluid mechanics, 62(2-3), 253-277. [CrossRef]
- Lizarraga-Garcia, E., Buongiorno, J., Al-Safran, E., & Lakehal, D. (2017). A broadly-applicable unified closure relation for Taylor bubble rise velocity in pipes with stagnant liquid. International journal of multiphase flow, 89, 345-358. [CrossRef]
- Caviezel, D., Narayanan, C., & Lakehal, D. (2008). Adherence and bouncing of liquid droplets impacting on dry surfaces. Microfluidics and Nanofluidics, 5, 469-478. [CrossRef]
- Lakehal, D., Larrignon, G., & Narayanan, C. (2008). Computational heat transfer and two-phase flow topology in miniature tubes. Microfluidics and nanofluidics, 4(4), 261-271. [CrossRef]
- Lakehal, D., Meier, M., & Fulgosi, M. (2002). Interface tracking towards the direct simulation of heat and mass transfer in multiphase flows. International Journal of Heat and Fluid Flow, 23(3), 242-257. [CrossRef]
- Suzuki, T., Kalyan, S., Berlinicke, C., Yoseph, S., Zack, D. J., & Hur, S. C. (2023). Deciphering viscoelastic cell manipulation in rectangular microchannels. Physics of Fluids, 35(10). [CrossRef]
- Pouraria, H., Foudazi, R., & Houston, J. P. (2023). Exploitation of elasto-inertial fluid flow for the separation of nano-sized particles: simulating the isolation of extracellular vesicles. Cytometry Part A. [CrossRef]
- Poryles, R., & Zenit, R. (2018). Encapsulation of droplets using cusp formation behind a drop rising in a non-Newtonian fluid. Fluids, 3(3), 54. [CrossRef]
- Ohta, M., Kobayashi, N., Shigekane, Y., Yoshida, Y., & Iwata, S. (2015). The dynamic motion of single bubbles with unique shapes rising freely in hydrophobically modified alkali-soluble emulsion polymer solutions. Journal of Rheology, 59(2), 303-316. [CrossRef]
- Esposito, G., Dimakopoulos, Y., & Tsamopoulos, J. (2023). Buoyancy driven flow of a viscous drop in viscoelastic materials. Journal of Non-Newtonian Fluid Mechanics, 321, 105124. [CrossRef]
- Poole, R. J. (2023). Inelastic and flow-type parameter models for non-Newtonian fluids. Journal of Non-Newtonian Fluid Mechanics, 105106. [CrossRef]
- Mirzaaghaian, A., Ramiar, A., Ranjbar, A. A., & Warkiani, M. E. (2020). Application of level-set method in simulation of normal and cancer cells deformability within a microfluidic device. Journal of biomechanics, 112, 110066. [CrossRef]
- Nooranidoost, M., & Kumar, R. (2020). Improving viability of leukemia cells by tailoring shell fluid rheology in constricted microcapillary. Scientific Reports, 10(1), 11570. [CrossRef]
- Hochmuth, R. M. (2000). Micropipette aspiration of living cells. Journal of biomechanics, 33(1), 15-22. [CrossRef]
- Tsai, M. A., Waugh, R. E., & Keng, P. C. (1996). Cell cycle-dependence of HL-60 cell deformability. Biophysical journal, 70(4), 2023-2029. [CrossRef]
- Rosenbluth, M. J., Lam, W. A., & Fletcher, D. A. (2006). Force microscopy of nonadherent cells: a comparison of leukemia cell deformability. Biophysical journal, 90(8), 2994-3003. [CrossRef]


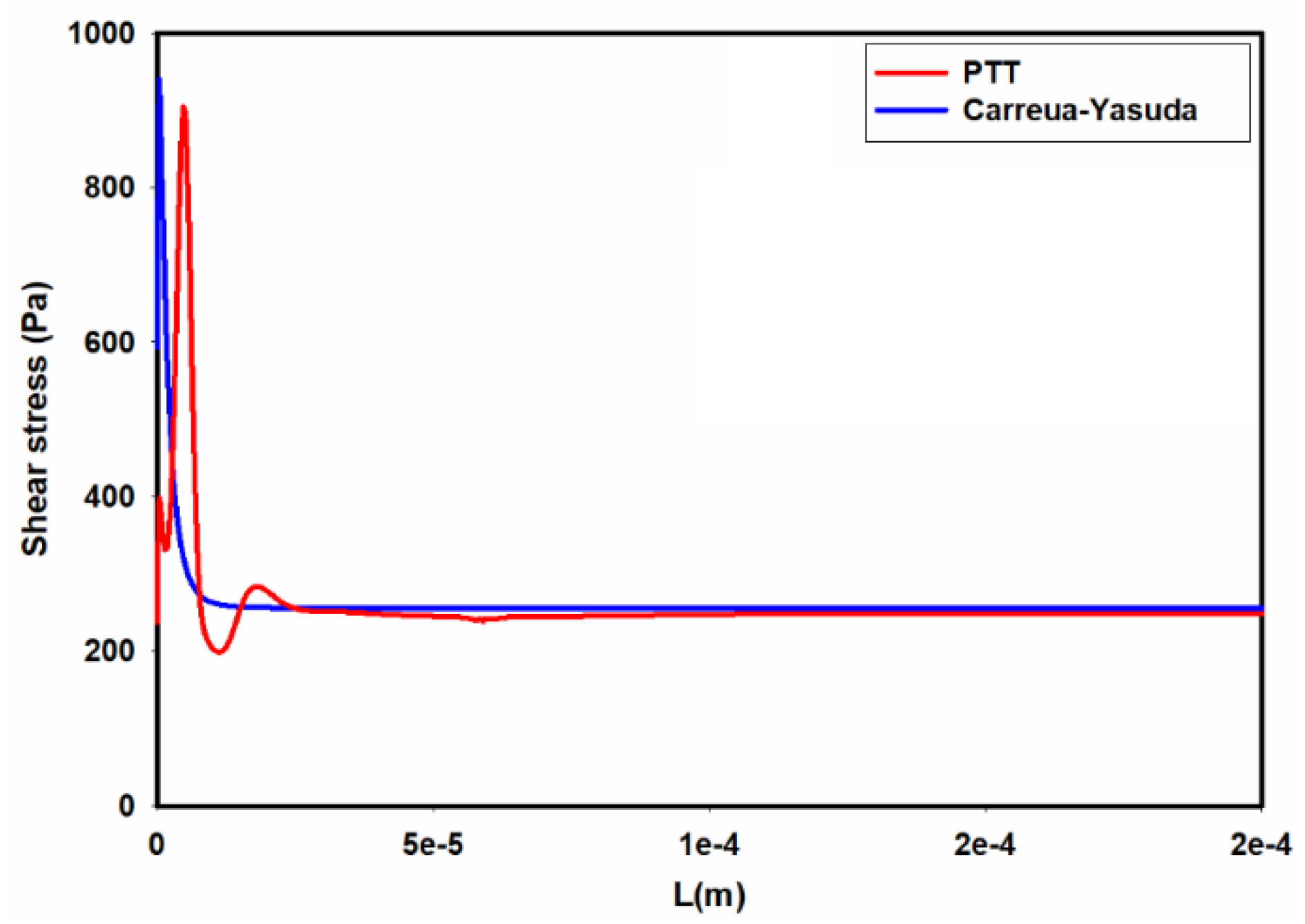
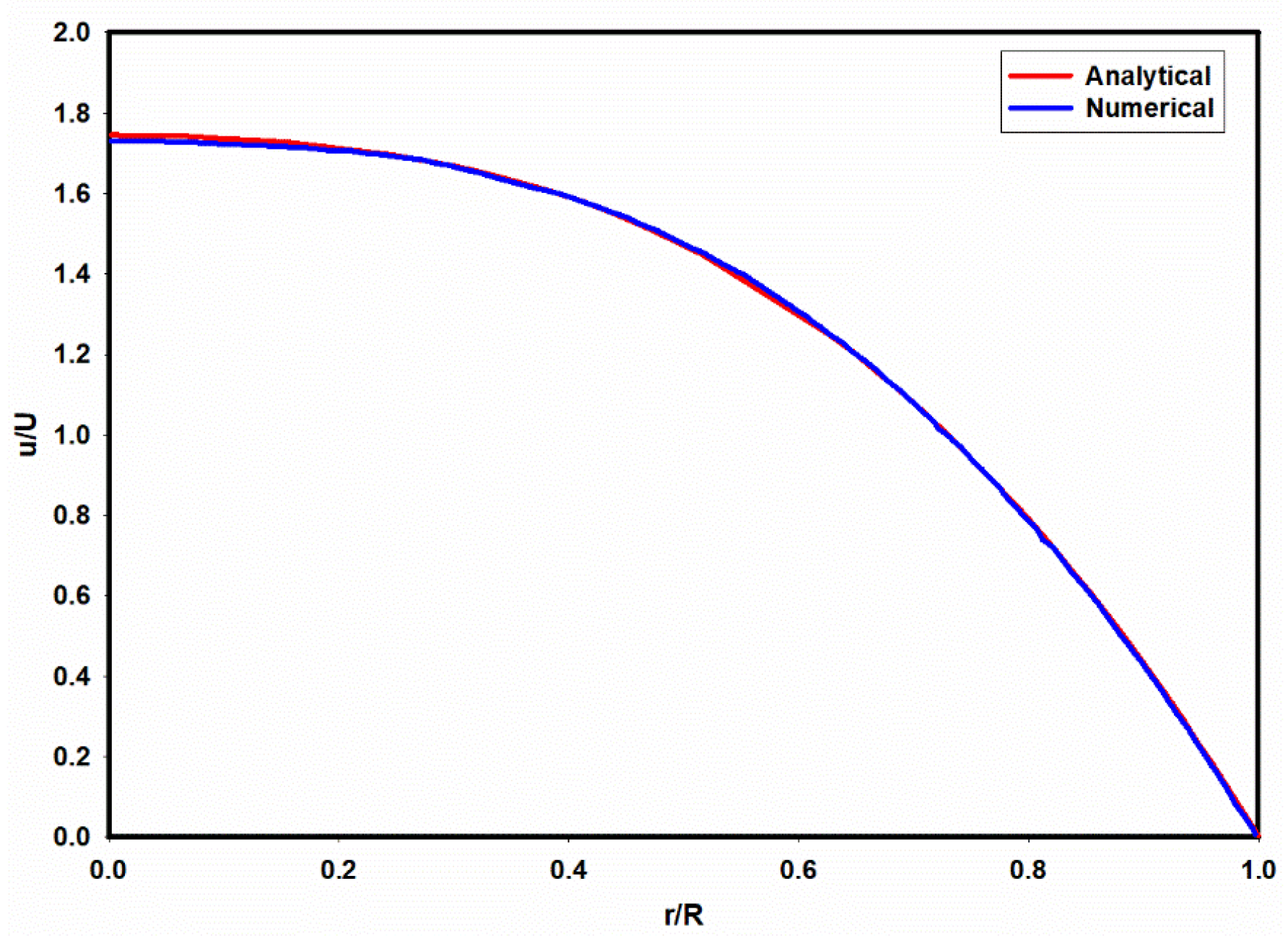
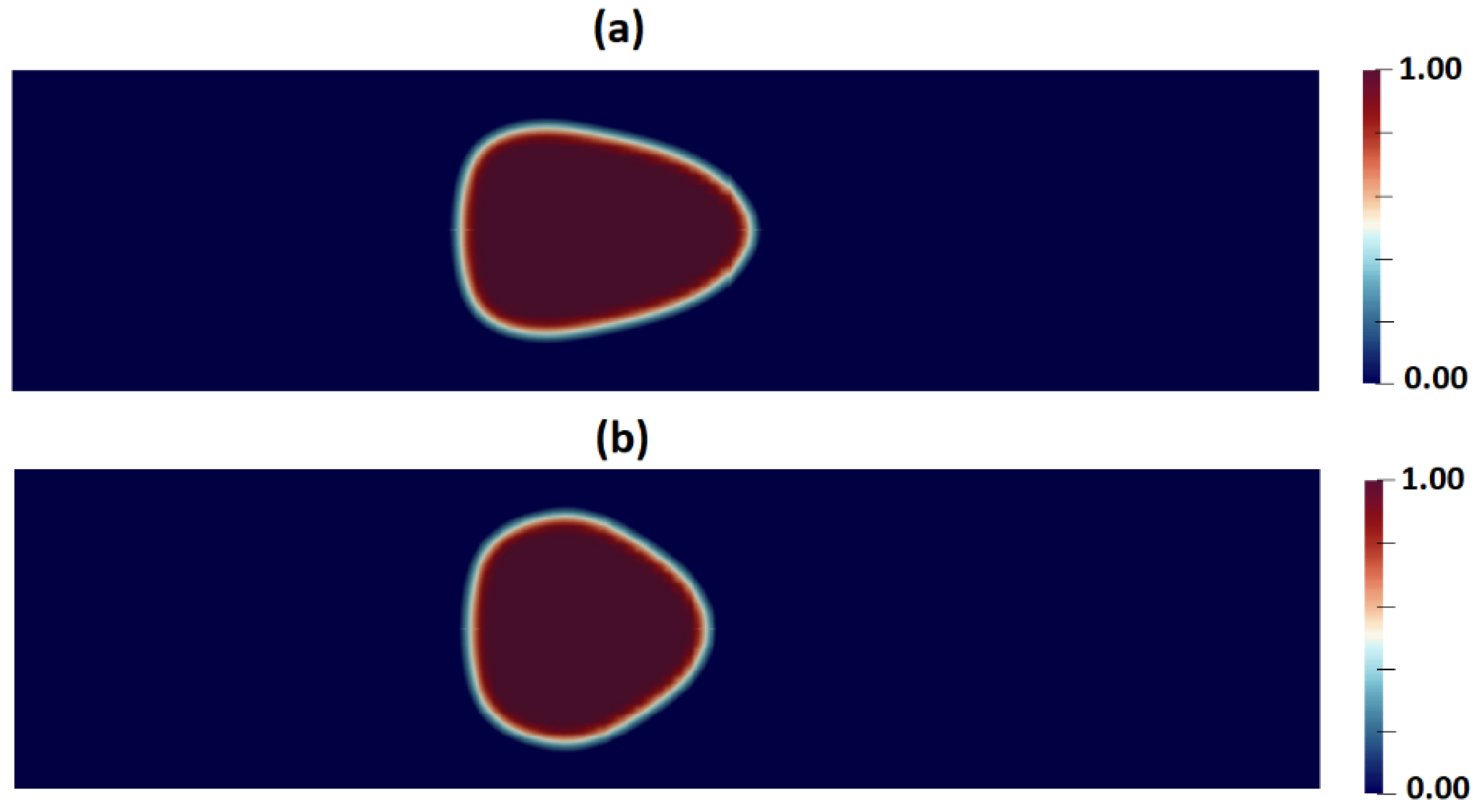
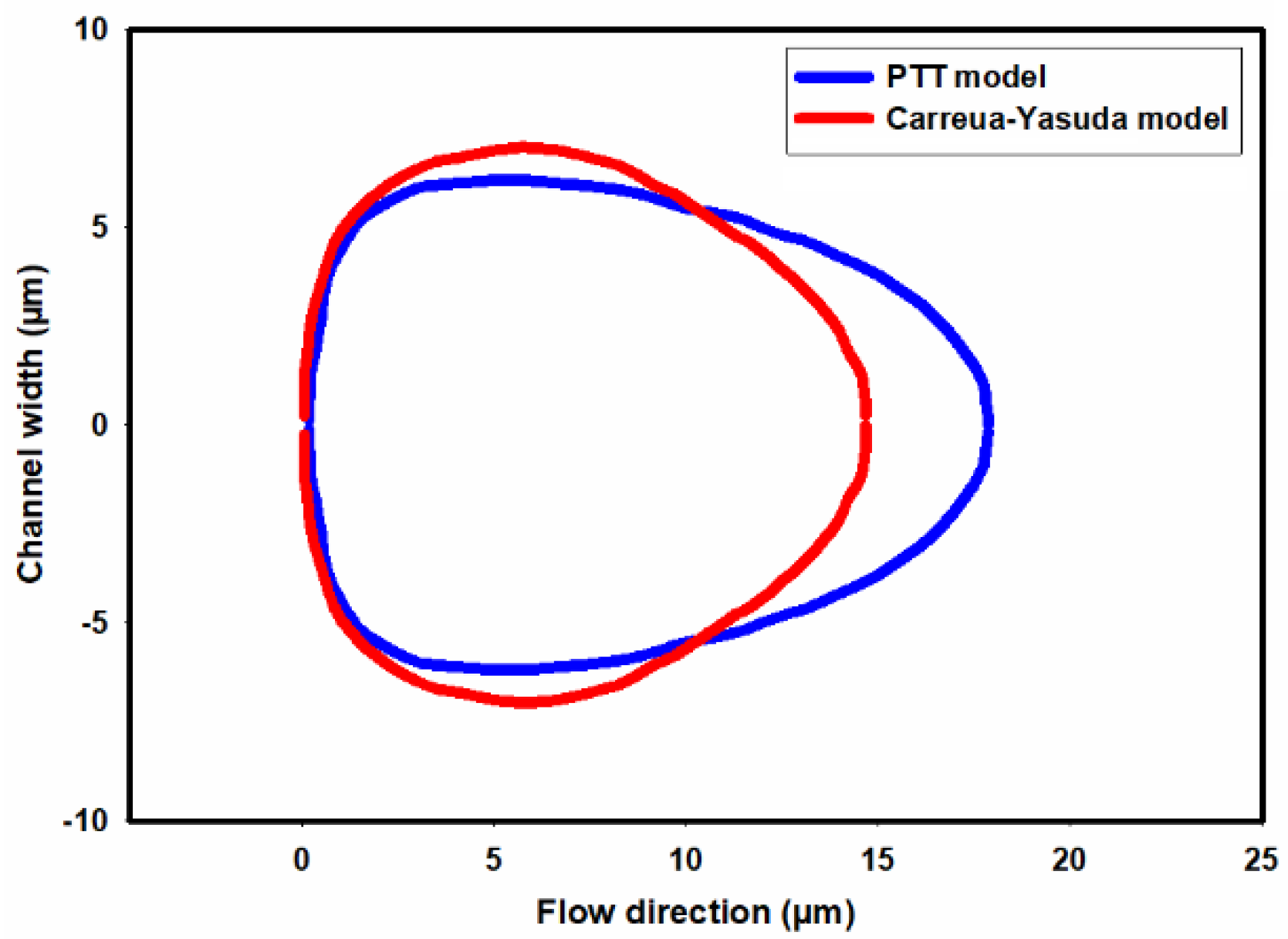

| Fluid | v | |||||
| 0.5% MC-PBS | 20mPa.s | 1mPa.s | 0.0012 | 0.65 | 1.02 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




