Submitted:
15 May 2024
Posted:
16 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials & Methods
2.1. Study Design and Data Acquisition
2.2. Standard Statistical Analysis
2.3. Machine Learning Analysis
2.4. Data Sharing
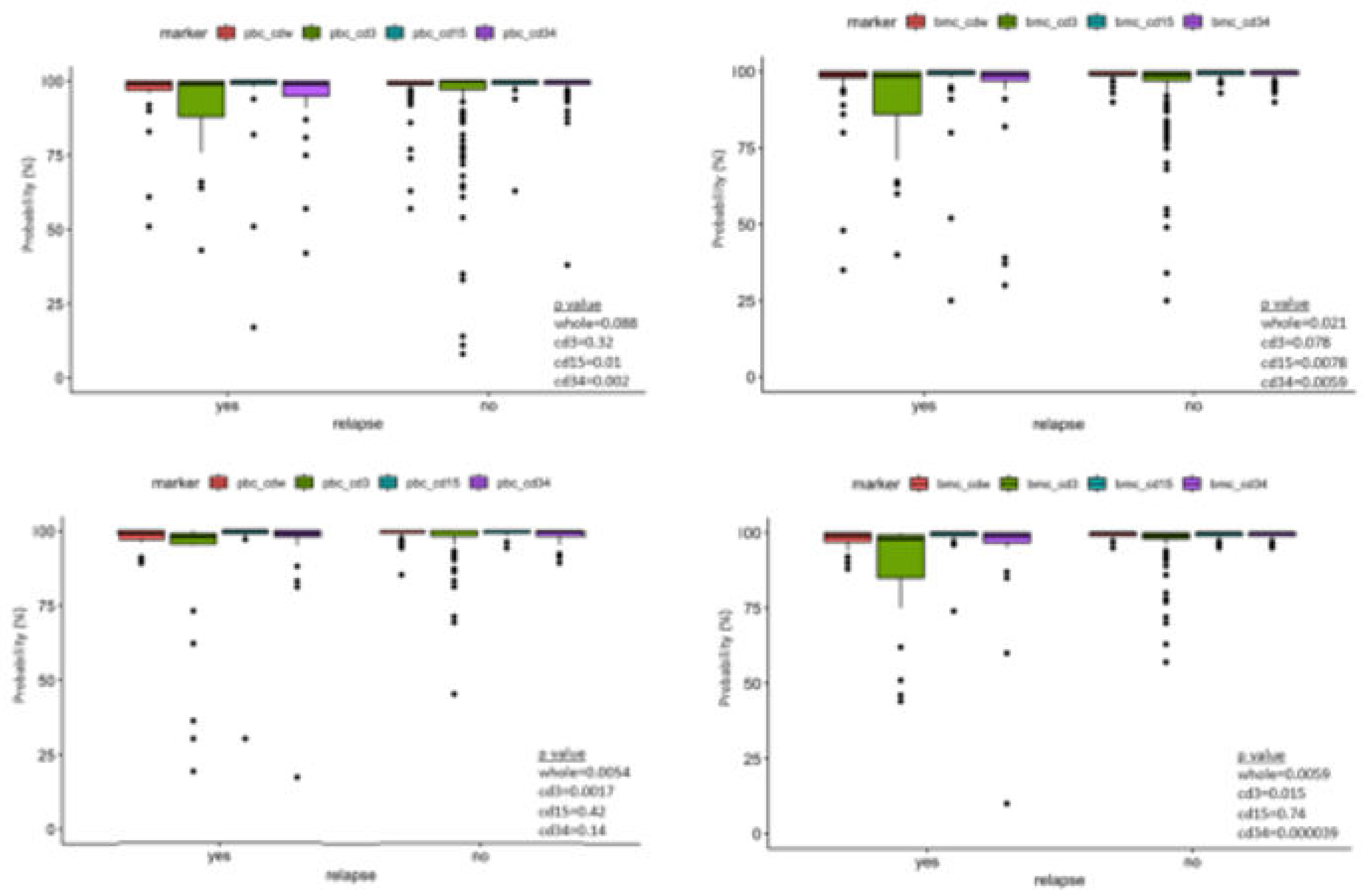
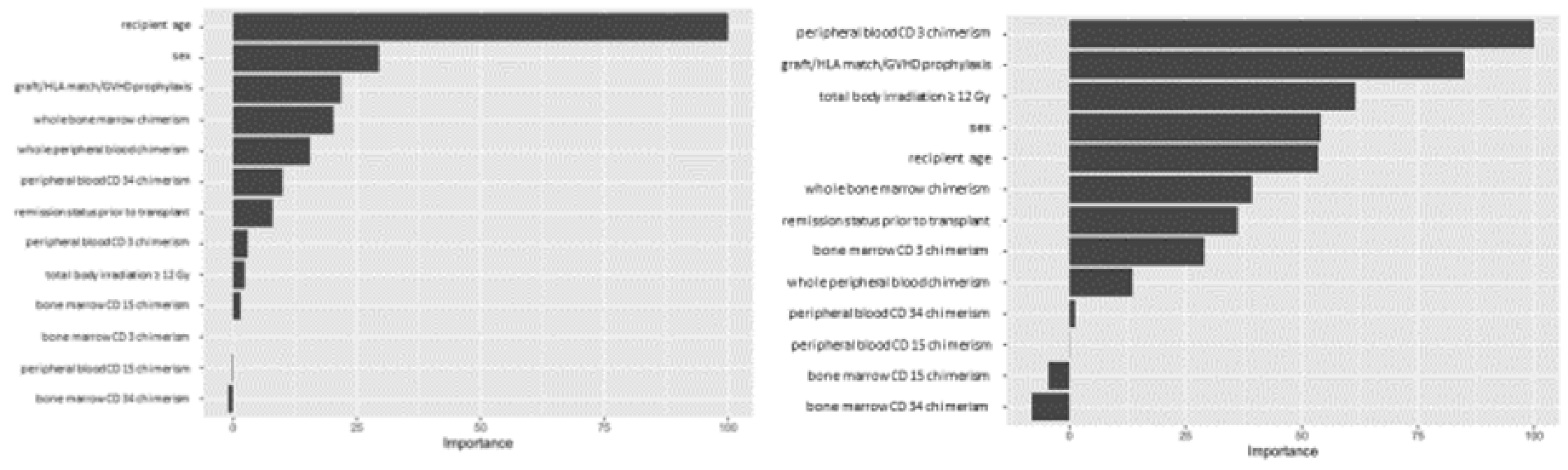
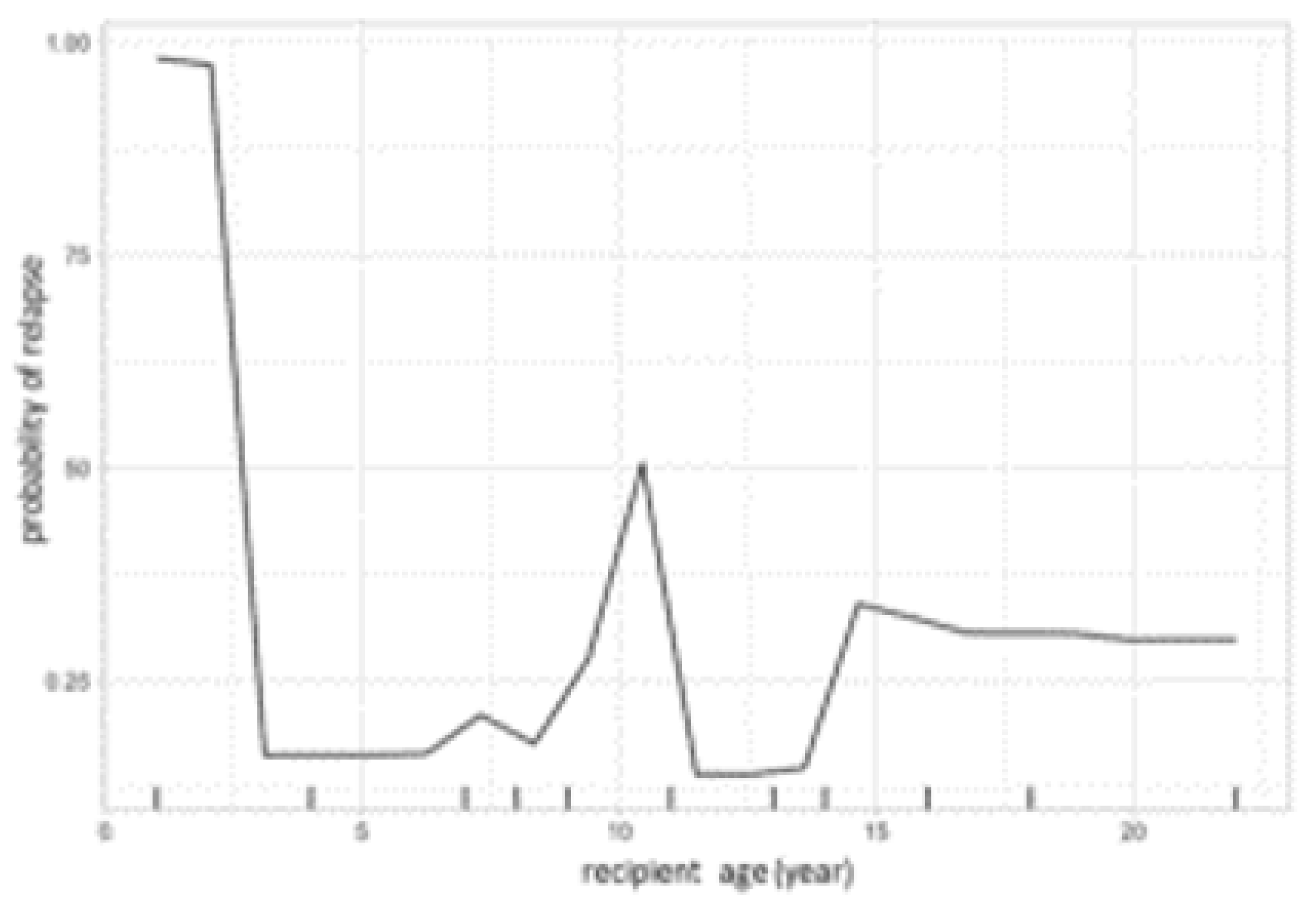
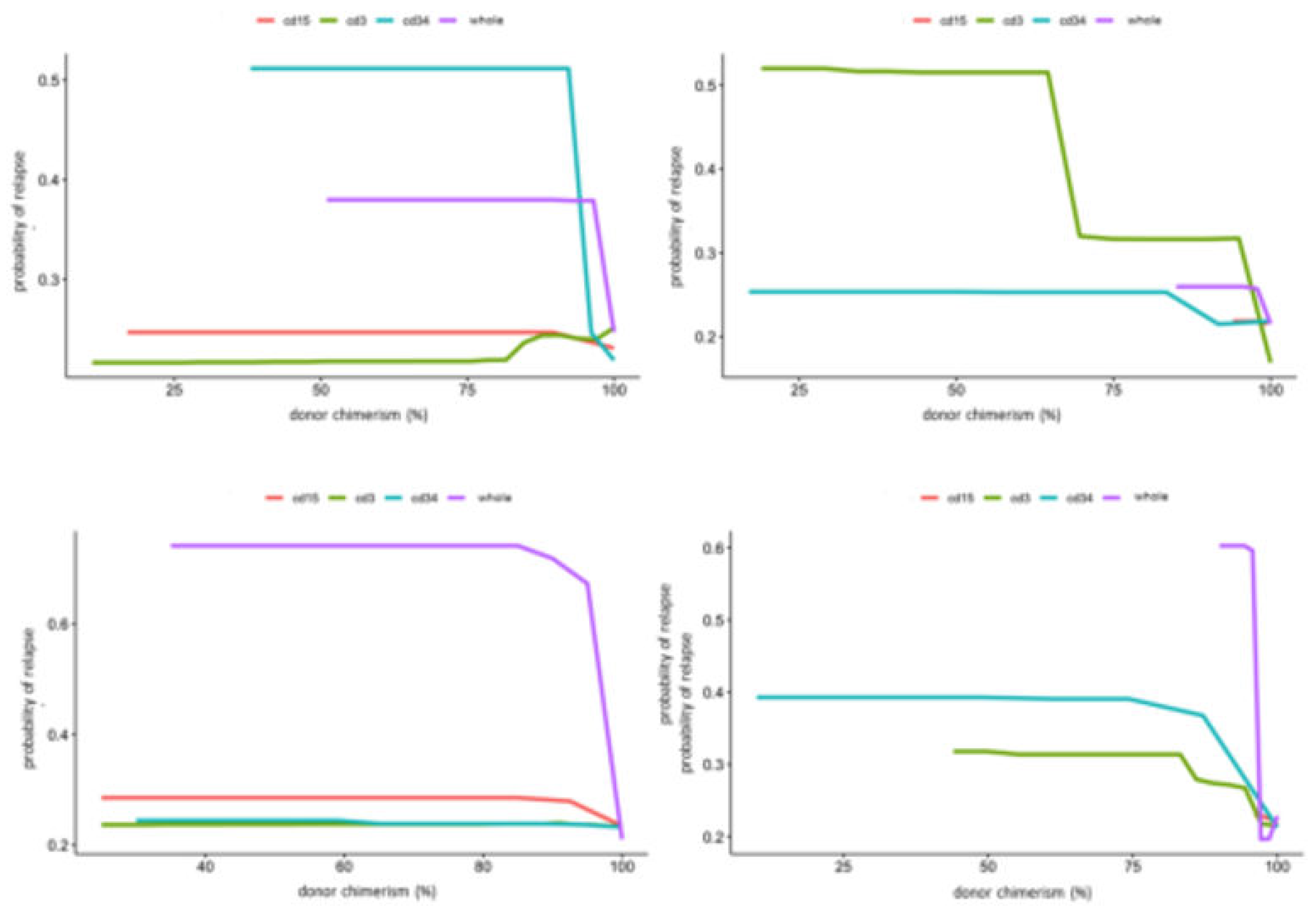
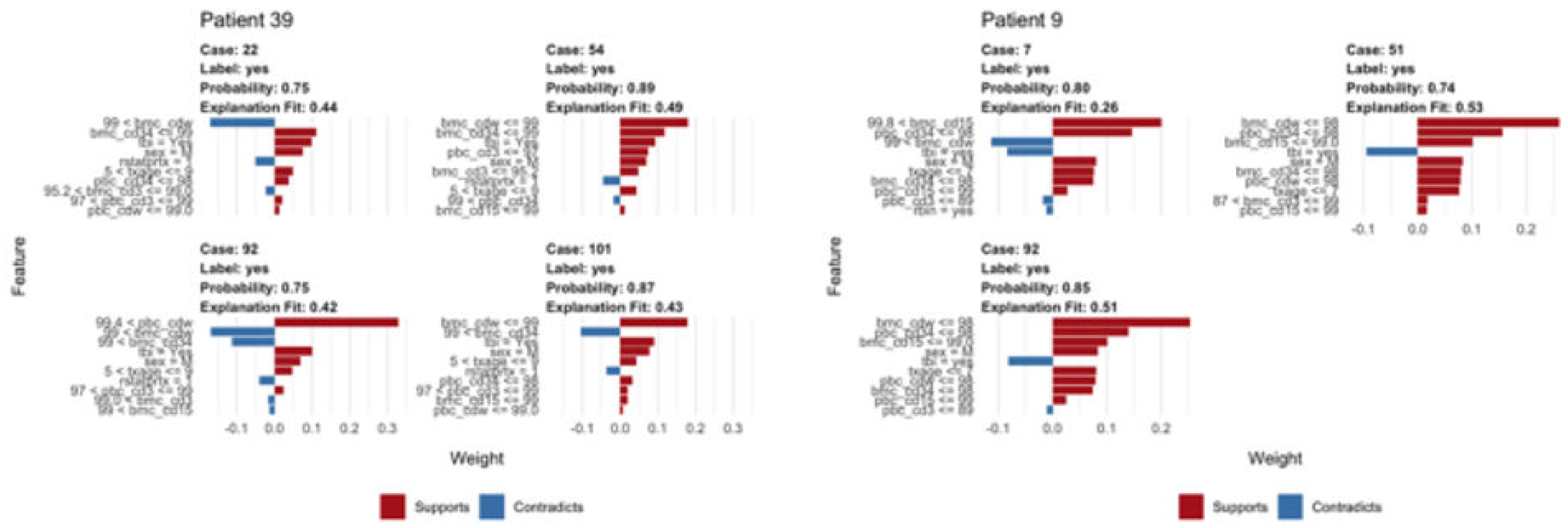
3. Results
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Styczynski J, Tridello G, Koster L, et al. Death after hematopoietic stem cell transplantation: changes over calendar year time, infections and associated factors. Bone Marrow Transplant. 2020;55(1):126-136. [CrossRef]
- Styczyński J, Tridello G, Koster L, et al. Death after hematopoietic stem cell transplantation: changes over calendar year time, infections and associated factors. Bone Marrow Transplant. 2020;55(1):126-136. [CrossRef]
- Broglie L, Helenowski I, Jennings LJ, et al. Early mixed T-cell chimerism is predictive of pediatric AML or MDS relapse after hematopoietic stem cell transplant. Pediatr Blood Cancer. 2017;64(9). [CrossRef]
- Kinsella FAM, Inman CF, Gudger A, et al. Very early lineage-specific chimerism after reduced intensity stem cell transplantation is highly predictive of clinical outcome for patients with myeloid disease. Leuk Res. 2019;83:106173. [CrossRef]
- Lee HC, Saliba RM, Rondon G, et al. Mixed T Lymphocyte Chimerism after Allogeneic Hematopoietic Transplantation Is Predictive for Relapse of Acute Myeloid Leukemia and Myelodysplastic Syndromes. Biol Blood Marrow Transplant. 2015;21(11):1948-1954. [CrossRef]
- Preuner S, Peters C, Potschger U, et al. Risk assessment of relapse by lineage-specific monitoring of chimerism in children undergoing allogeneic stem cell transplantation for acute lymphoblastic leukemia. Haematologica. 2016;101(6):741-746. [CrossRef]
- Thompson PA, Stingo F, Keating MJ, et al. Long-term follow-up of patients receiving allogeneic stem cell transplant for chronic lymphocytic leukaemia: mixed T-cell chimerism is associated with high relapse risk and inferior survival. Br J Haematol. 2017;177(4):567-577. [CrossRef]
- Lee CJ, Savani BN, Mohty M, et al. Post-remission strategies for the prevention of relapse following allogeneic hematopoietic cell transplantation for high-risk acute myeloid leukemia: expert review from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Bone Marrow Transplant. 2019;54(4):519-530. [CrossRef]
- DeFilipp Z, Langston AA, Chen Z, et al. Does Post-Transplant Maintenance Therapy With Tyrosine Kinase Inhibitors Improve Outcomes of Patients With High-Risk Philadelphia Chromosome-Positive Leukemia? Clin Lymphoma Myeloma Leuk. 2016;16(8):466-471 e461. [CrossRef]
- Yan CH, Liu QF, Wu DP, et al. Prophylactic Donor Lymphocyte Infusion (DLI) Followed by Minimal Residual Disease and Graft-versus-Host Disease-Guided Multiple DLIs Could Improve Outcomes after Allogeneic Hematopoietic Stem Cell Transplantation in Patients with Refractory/Relapsed Acute Leukemia. Biol Blood Marrow Transplant. 2017;23(8):1311-1319. [CrossRef]
- Lankester AC, Locatelli F, Bader P, et al. Will post-transplantation cell therapies for pediatric patients become standard of care? Biol Blood Marrow Transplant. 2015;21(3):402-411. [CrossRef]
- Hastie T, Tibshirani R, Friedman J. Boosting and Additive Trees. In: Hastie T, Tibshirani R, Friedman J, eds. The Elements of Statistical Learning: Data Mining, Inference, and Prediction. New York, NY: Springer New York; 2009:337-387.
- Shouval R, Fein JA, Savani B, Mohty M, Nagler A. Machine learning and artificial intelligence in haematology. Br J Haematol. 2021;192(2):239-250. [CrossRef]
- Muhsen IN, Shyr D, Sung AD, Hashmi SK. Machine Learning Applications in the Diagnosis of Benign and Malignant Hematological Diseases. Clin Hematol Int. 2021;3(1):13-20. [CrossRef]
- Zhou Y, Smith J, Keerthi D, et al. Longitudinal clinical data improve survival prediction after hematopoietic cell transplantation using machine learning. Blood Adv. 2024;8(3):686-698. [CrossRef]
- Powers S, Qian J, Jung K, et al. Some methods for heterogeneous treatment effect estimation in high dimensions. Stat Med. 2018;37(11):1767-1787. [CrossRef]
- Fuse K, Uemura S, Tamura S, et al. Patient-based prediction algorithm of relapse after allo-HSCT for acute Leukemia and its usefulness in the decision-making process using a machine learning approach. Cancer Med. 2019;8(11):5058-5067. [CrossRef]
- Shouval R, Labopin M, Unger R, et al. Prediction of Hematopoietic Stem Cell Transplantation Related Mortality- Lessons Learned from the In-Silico Approach: A European Society for Blood and Marrow Transplantation Acute Leukemia Working Party Data Mining Study. PLoS One. 2016;11(3):e0150637. [CrossRef]
- Zhang Y, Ling C. A strategy to apply machine learning to small datasets in materials science. npj Computational Materials. 2018;4(1):25.
- Caiafa CF, Sun Z, Tanaka T, Marti-Puig P, Solé-Casals J. Machine Learning Methods with Noisy, Incomplete or Small Datasets. Applied Sciences. 2021;11(9):4132. [CrossRef]
- Koppe G, Meyer-Lindenberg A, Durstewitz D. Deep learning for small and big data in psychiatry. Neuropsychopharmacology. 2021;46(1):176-190. [CrossRef]
- Shaikhina T, Lowe D, Daga S, Briggs D, Higgins R, Khovanova N. Machine Learning for Predictive Modelling based on Small Data in Biomedical Engineering. IFAC-PapersOnLine. 2015;48(20):469-474. [CrossRef]
- Caiafa CF, Sun Z, Tanaka T, Marti-Puig P, Solé-Casals J. Machine Learning Methods with Noisy, Incomplete or Small Datasets. Applied Sciences. 2021;11(9). [CrossRef]
- Murdoch WJ, Singh C, Kumbier K, Abbasi-Asl R, Yu B. Definitions, methods, and applications in interpretable machine learning. Proc Natl Acad Sci U S A. 2019;116(44):22071-22080. [CrossRef]
- Rudin C. Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nature Machine Intelligence. 2019;1(5):206-215. [CrossRef]
- Kumarakulasinghe NB, Blomberg T, Liu J, Leao AS, Papapetrou P. Evaluating Local Interpretable Model-Agnostic Explanations on Clinical Machine Learning Classification Models. Paper presented at: 2020 IEEE 33rd International Symposium on Computer-Based Medical Systems (CBMS); 28-30 July 2020, 2020. [CrossRef]
- RCoreTeam. R:A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria Web site. https://www.R-project.org. Published 2020. Accessed.
- Esplin IND, Berg JA, Sharma R, et al. Genome Sequences of 19 Novel Erwinia amylovora Bacteriophages. Genome Announc. 2017;5(46). [CrossRef]
- Kuhn M. Building Predictive Models in R Using the caret Package. 2008. 2008;28(5):26. [CrossRef]
- Cabitza F, Campagner A. The need to separate the wheat from the chaff in medical informatics: Introducing a comprehensive checklist for the (self)-assessment of medical AI studies. International Journal of Medical Informatics. 2021;153:104510. [CrossRef]
- Friedman JH, Popescu BE. Predictive Learning via Rule Ensembles. The Annals of Applied Statistics. 2008;2(3):916-954. [CrossRef]
- Bzdok D, Altman N, Krzywinski M. Statistics versus machine learning. Nat Methods. 2018;15(4):233-234. [CrossRef]

| ALL (n=63) | AML (n=46) | |
|---|---|---|
| Age (year) | ||
| Range | 1 to 22 | 1 to 23 |
| Median | 12 | 11 |
| Mean | 11 | 10 |
| Sex (n) | ||
| Male | 37 (59%) | 29 (63%) |
| Female | 26 (41%) | 17 (27%) |
| Pretransplant Remission Status (n) | ||
| Complete Remission 1 | 21 (33%) | 32 (70%) |
| Complete Remission 2 | 36 (57%) | 10 (22%) |
| Complete Remission 3 | 6 (10%) | 0 |
| Relapse | 0 | 3 (7%) |
| Unknown | 0 | 1 (2%) |
| Graft Source (n) | ||
| Bone Marrow | 37 (59%) | 22 (48%) |
| Cord Blood | 8 (13%) | 9 (20%) |
| Peripheral Blood Stem Cell | 18 (28%) | 15 (32%) |
| Total Body Irradiation (n) | ||
| Yes | 57 (90%) | 25 (54%) |
| No | 6 (10%) | 21 (46%) |
| Acute Graft versus Host Disease any grade (n) | ||
| Yes | 38 (60%) | 22 (48%) |
| No | 25 (40%) | 24 (52%) |
| Relapsed | ||
| Yes (n) | 14 (26%) | 13 (28%) |
| Range (days) | 53 to 620 | 54 to 621 |
| Mean (days) | 244 | 210 |
| Median (days) | 188 | 174 |
| Days from last peripheral blood chimerism | ||
| Range (days) | 7 to 531 | 15 to 467 |
| Mean (days) | 129 | 132 |
| Median (days) | 63 | 39 |
| Peripheral Blood | Bone Marrow | |||||
| ALL |
Number of tests (n, % data present) |
mean (post-TX days) |
Range (post-TX days) |
Number of tests (n, % data present) |
mean (post-TX days) |
Range (post-TX days) |
| Chimerism #1 | 55 (87%) | 27 | 12 to 40 | 63 (100%) | 21 | 24 to 62 |
| Chimerism #2 | 49 (78%) | 60 | 30 to 91 | 59 (94%) | 63 | 43 to 98 |
| Chimerism #3 | 47 (74%) | 100 | 42 to 186 | 55 (87%) | 96 | 77 to 186 |
| Chimerism #4 | 39 (62%) | 191 | 84 to 384 | 43 (68%) | 180 | 127 to 377 |
| Chimerism #5 | 23 (37%) | 332 | 139 to 532 | 14 (22%) | 321 | 173 to 449 |
| AML | ||||||
| Chimerism #1 | 36 (78%) | 28 | 12 to 37 | 42 (91%) | 32 | 21 to 43 |
| Chimerism #2 | 28 (61%) | 61 | 29 to 85 | 44 (95%) | 63 | 43 to 89 |
| Chimerism #3 | 26 (57%) | 97 | 62 to 145 | 36 (78%) | 93 | 69 to 119 |
| Chimerism #4 | 23 (50%) | 174 | 97 to 265 | 31 (67%) | 174 | 119 to 197 |
| Chimerism #5 | 17 (37%) | 345 | 243 to 518 | 19 (41%) | 335 | 182 to 557 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




