1. Introduction
Diabetic retinopathy is a major source of visual disability [
1]. Diabetic macular ischemia is one of the vision - threatening complications of diabetic retinopathy. In macular ischemia, the structure of the foveal capillary network is damaged. Such damage includes enlargement and irregularity of the foveal avascular zone (FAZ) and macular non - perfusion, leading to macular dysfunction. Fluorescein angiography (FA) is a well - established modality to assess perfusion of the retina and the level of ischemia in the macula and the peripheral retina.
The sequelae for visual function of diabetic macular ischemia severity are well characterized since the Early Treatment Diabetic Retinopathy Study (ETDRS) in the 1980s [
2] . Diabetic macular ischemia is traditionally defined based on the evidence of an enlarged and irregular FAZ area of at least 0.5 mm
2 or parafoveal capillary dropout in at least one parafoveal quadrant if the FAZ area is less than 0.5 mm
2 on FA [
2,
3]. It is also understood that the magnitude of ischemic changes in the retina is more pronounced in the proliferative stage of diabetic retinopathy [
4].
OCT – angiography is a new imaging modality that has addressed several drawbacks of FA, including limited image resolution, invasiveness, lack of depth information, and two - dimensional imaging [
5,
6]. OCT angiography provides depth encoded imaging with improved resolution. It is now possible to visualize not only the superficial (SCP), but also the deep (DCP) capillary plexus of the retina, to quantify non – invasively several FAZ parameters and to assess separately the integrity of each capillary plexus [
7,
8,
9]. The OCT angiography images offer both qualitative as well as quantitative information. However, the association between the microvascular changes that can be detected in OCT angiography in diabetic macular ischemia and visual impairment is complex and largely unknown, while recent studies are trying to better characterize this relationship [
3,
10]. FAZ area size has been one of the biomarkers that has been associated with varying levels of retinopathy severity [
11]. Moreover, larger baseline deep capillary plexus fovea avascular zone (DCP FAZ) area has been reported to be predictive of worsening visual outcomes [
12].
Intravitreal injections of anti- vascular endothelial growth factor (VEGF) are the preferred initial treatment approach for diabetic macular edema (DME) due to their proven safety and effectiveness [
13]. These drugs selectively block VEGF, which is regarded as a key mediator in diabetic retinopathy. Notably, as the VEGF counteracts the blockage of the foveal capillary network which causes diabetic macular ischemia, administering anti-VEGF injections should be advantageous for diabetic macular ischemia. Furthermore, current research is actively attempting to assess the biomarkers from OCT and OCT angiography imaging that play a crucial role in predicting the visual outcome following intravitreal anti - VEGF injections [
14]. Larger DCP FAZ area has emerged as one of these factors [
15].
OCT angiography additionally offers a novel instrument for evaluating the impact of treatment [
16]. The objective of this pilot study was to investigate the alteration in the area of the FAZ area following the administration of intravitreal injections of anti-VEGF drugs. Our objective was to examine if there is a variation in the impact of the treatment effect between ranibizumab and aflibercept, as well as investigate the relationship between macular ischemia and visual acuity during the treatment process.
2. Materials and Methods
Treatment naive patients diagnosed with center - involved diabetic macular edema were consecutively enrolled in the Retina clinics at the University Hospital of Larissa, Greece between June 2017 and June 2022. This was a single - center prospective study. The allocation of patients to receive either ranibizumab or aflibercept was determined by the treating physician. The study protocol adhered to the Declaration of Helsinki and was approved by the University Hospital of Larissa Review Board. Before enrolling, eligible participants were required to sign an informed consent form and were provided with a thorough explanation of the study protocol.
The inclusion criteria were as follows: age 18 years or older, presence of type 1 or 2 diabetes, treated with insulin or oral anti- hyperglycemia agents, presence of center - involved DME with central subfield thickness measuring between 280 and 600 microns and best - corrected visual acuity (BCVA) Early Treatment Diabetic Retinopathy Study (ETDRS) score between 73 and 24 letters (equivalent to 20/40 - 20/320 decimal visual acuity) in the study eye. Patients were recruited in a consecutive if eligible fashion.
Patients who met any of the following criteria were not included in the study : poor glycemic control (hemoglobin HbA1c > 8 in the past 6 months), uncontrolled arterial hypertension, active proliferative diabetic retinopathy (PDR) or history of vitreous hemorrhage, prior panretinal photocoagulation, concomitant ocular conditions (other than diabetic retinopathy) that could affect visual acuity or imaging (e.g., retinal vein or artery occlusion, cornea scarring, dense cataract, uveitis or other ocular inflammatory disease, glaucoma, vitreomacular traction, epitretinal membrane etc.), previous vitrectomy and prior ocular surgeries (other than uneventful cataract extraction, performed at least three months before enrollment). Only treatment naive patients were included in the study, therefore patients who had previously received intravitreal anti -VEGF or corticosteroid injections or macula laser photocoagulation were excluded. Patients that would develop vitreous hemorrhage or necessitate rescue panretinal photocoagulation would exit the study. All patients were offered treatment with monthly injections up to 6 months. All injections were administered in time intervals of 30 ± 3 days. Each visit included BCVA measurement assessed with an ETDRS chart, slit lamp examination and Goldmann applanation tonometry, followed by OCT and OCT angiography imaging. At the end of the baseline visit, the first intravitreal injection of ranibizumab or aflibercept was given. Then, on visit 1, after the clinical examination and imaging sessions, the second injection was delivered. Likewise, visit 2 ended with the 3 rd intravitreal injection, visit 3 with the 4 th injection, visit 4 with the 5 th injection, visit 5 with the 6 th injection and visit 6 was scheduled at 1 month after the 6 th injection. OCT and OCT angiography imaging was performed using the Triton Plus swept source OCT (Topcon Inc, Japan). Scanning protocols included a 6 * 6 mm 3D OCT angiography scan with a resolution of 320 * 320 pixels and 3D OCT 7 * 7 mm scan with a resolution of 512 * 256 pixels. OCT central retina thickness (CRT) was quantified with the instrument’s built-in software. The automatic segmentation algorithms of the OCT angiography device were used. The SCP slab spanned the area between the inner limiting membrane (ILM) + 2.6 μm and an outer boundary at the inner plexiform layer (IPL) / inner nuclear layer (INL) + 15.6 μm. The DCP slab was segmented between IPL / INL + 15.6 μm and IPL / INL + 70.2 μm.
En face OCT angiography images of the SCP and DCP were analyzed using the instrument’s built in tool to quantify the area of the SCP and DCP FAZ area by two graders (DM and AD). The mean of the two measurements was recorded for each image. All scans were acquired by one grader (DM). Images were visually inspected for artifacts, motion artifacts, and low signal strength. Images with low signal strength (less than 70), artifacts and segmentation errors were rejected and scan was repeated.
All statistical testing was performed using the SPSS 23 statistical package (IBM Inc). Continuous variables were tested for normality and data were presented as mean and standard deviation or median and interquartile range accordingly. Repeated measurements of the outcome variables in specific timepoints over time were evaluated with repeated measures ANOVA and the Wilks’ Lambda test. Estimates of effect size and observed power were also calculated. Post hoc tests were ran with corresponding p - values to evaluate differences between individual visits. The amount of change in SCP FAZ area as well as DCP FAZ area was also calculated and compared between consecutive visits with repeated measures ANOVA. Independent samples t-test was used to compare between groups and chi square test was used to compare categorical variables. Furthermore, Pearson correlation tests were performed. The level of significance was set at 0.05.
3. Results
Fifty eyes with full sets of data were analyzed. Mean age was 67.0 ± 10.7 years. There were 25 men and 25 women. Twenty - five patients received monthly aflibercept and the other 25 received monthly ranibizumab for 6 consecutive months. There was no difference in age (68.7 ± 7.9 versus 64.4 ± 12.0, independent samples t-test, p = 0.067) or gender (11 men / 14 women vs 14 men / 11 women, chi-square p = 0.572) between the two groups. None of the patients developed vitreous hemorrhage or required laser photocoagulation during the study. None of the patients was lost to follow up.
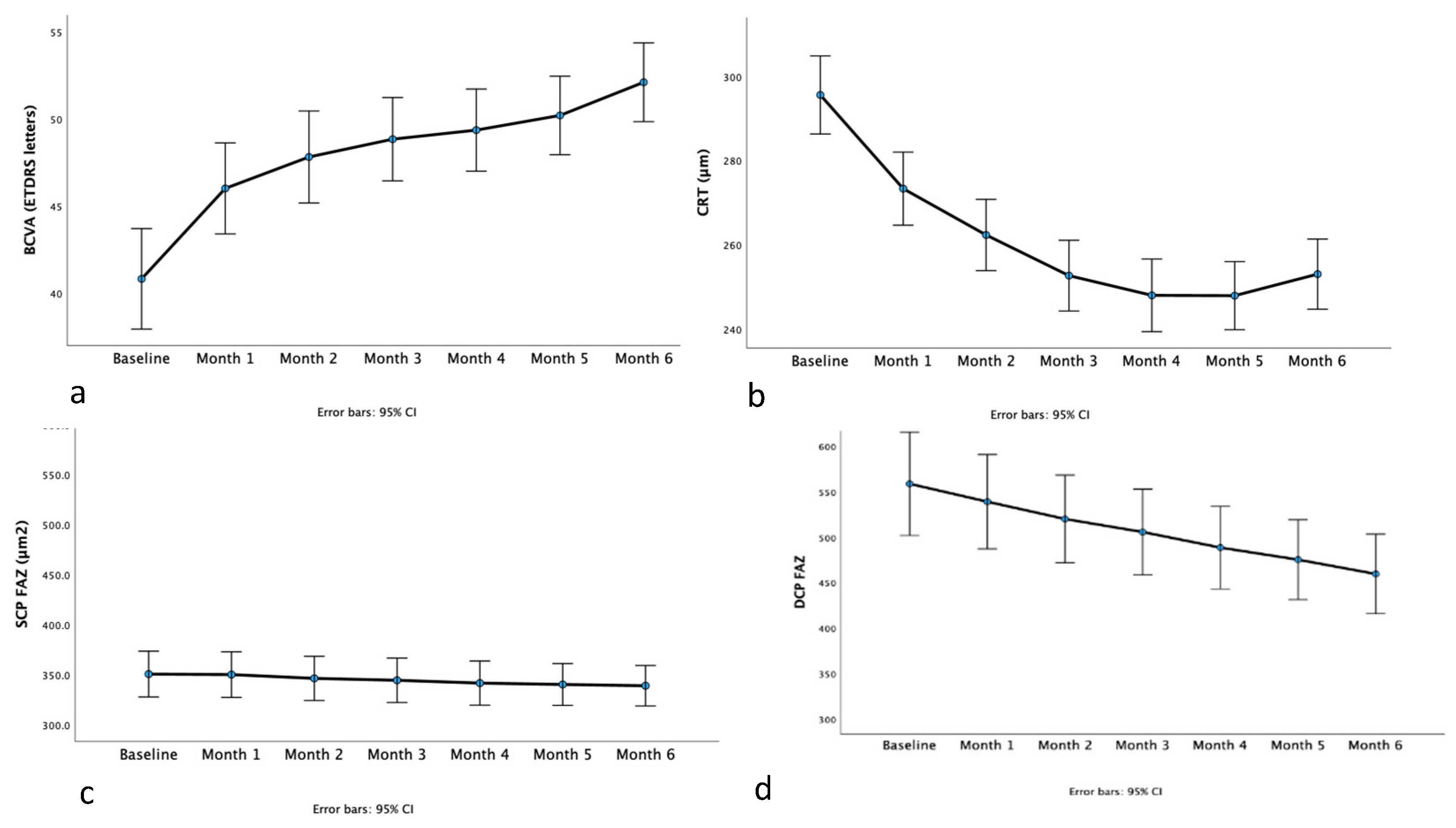
BCVA was 40.8 ± 10.0 ETDRS letters and increased to 52.1 ± 7.9 ETDRS letters at the last visit (
Table 1,
Figure 1a). There was significant improvement in BCVA in the course of the study (p < 0.001). Post hoc pairwise comparisons showed a significant increase in BCVA in all study visits compared to baseline (all p < 0.001). No effect of the choice of drug being either ranibizumab or aflibercept on BCVA results was found (p = 0.428).
CRT was 295.6 ± 34.0 at baseline and 247.9 ± 29.7 at the last study visit (
Table 1,
Figure 1b). CRT decreased with treatment (p < 0.001). Post hoc pairwise comparisons showed that CRT measurements at each subsequent study visit were lower compared to baseline (all p < 0.001). There was no effect of the type of treatment (ranibizumab or aflibercept) on the amount of CRT change (p = 0.261).
SCP FAZ area was 350.6 ± 79.5 μm
2 at baseline and 339.0 ± 71.3 μm
2 after 6 monthly injections (
Table 1,
Figure 1c). In visit 1, SCP FAZ area was 350.1 ± 79.2 μm
2, in visit 2 it was 346.3 ± 77.0 μm
2 , in visit 3 it was 344.4 ± 77.6 μm
2 , in visit 4 it was 341.6 ± 77.6 μm
2 and in visit 5, it measured 340.2 ± 73.3 μm
2 .There was no significant change in SCP FAZ area when comparing the SCP FAZ area at the 7 study timepoints (p = 0.132) or between the two drugs (p = 0.092).
DCP FAZ area was 558.6 ± 199.0 μm
2 at baseline and 459.5 ± 156.1 μm
2 after 6 monthly injections (p < 0.001,
Table 1,
Figure 1d). In visit 1, DCP FAZ area was 539.0 ± 182.0 μm
2, in visit 2 it was 520.0 ± 169.4 μm
2 , in visit 3 it was 505.6 ± 165.8 μm
2 , in visit 4 it was 488.5 ± 160.1 μm
2 and in visit 5, it measured 475.2 ± 156.1 μm
2 . There was no statistically significant effect of treatment with ranibizumab or aflibercept on the DCP FAZ area change (between - subjects effects p = 0.277).
Post hoc pairwise comparisons revealed that at each subsequent study visit, DCP FAZ area was smaller compared to baseline (all p < 0.001). In addition, DCP FAZ area at the last study timepoint was significantly smaller compared to any of the study visits timepoints (post hoc pairwise comparisons, all p <0.001).
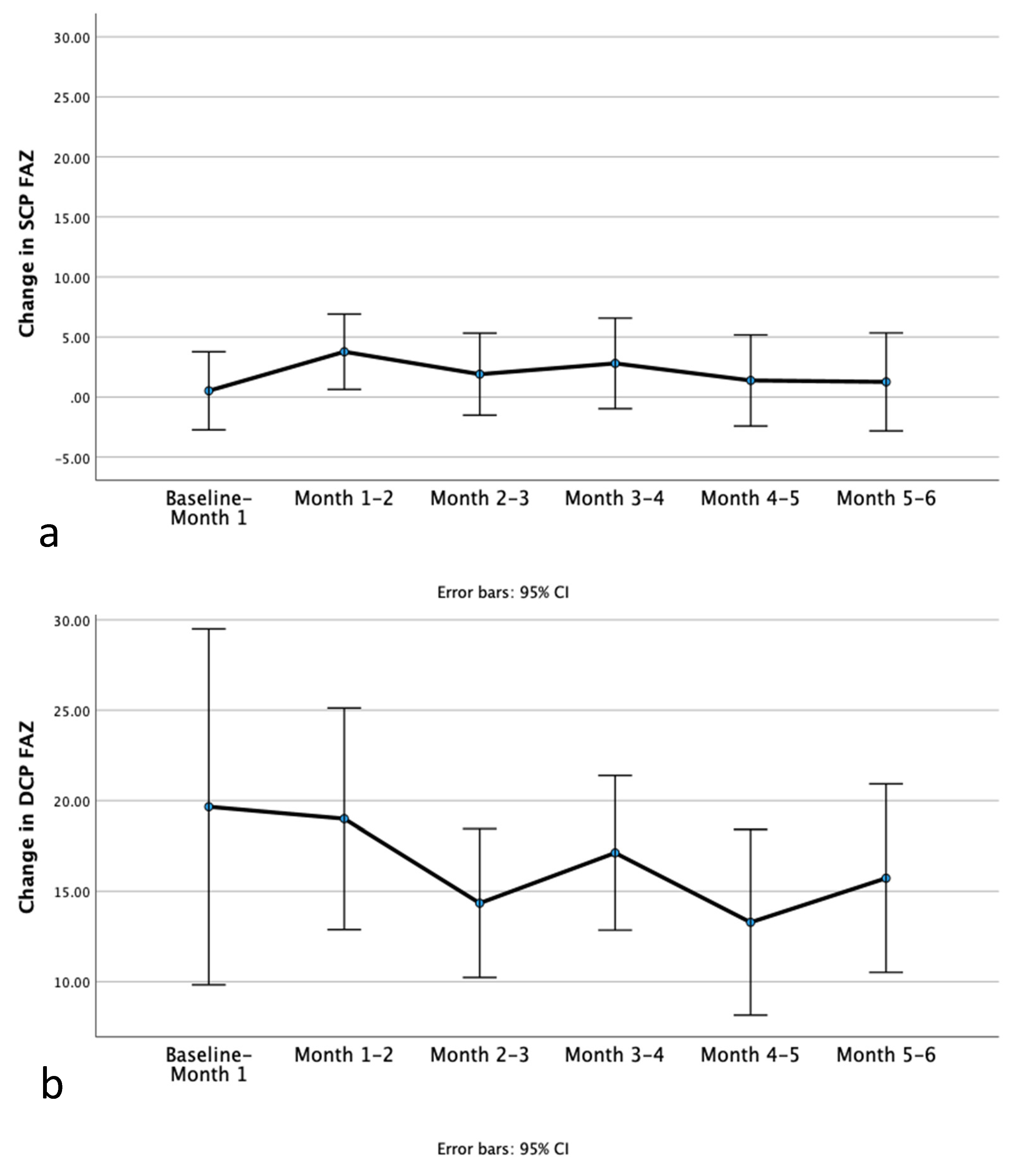
Then, the difference between FAZ area measurements in consecutive visits was calculated. As shown graphically in
Figure 2a and b, there was variability in the amount of change in SCP FAZ area and DCP FAZ area occurring every month.
There was no significant difference in the change in SCP FAZ area between consecutive monthly visits (repeated measures ANOVA p = 0.791). The change in SCP FAZ area was measured at 0.5 ± 11.5 μm2 from the baseline visit to visit 1, 3.8 ± 11.0 μm2 from visit 1 to visit 2, 1.9 ± 12.0 μm2 from visit 2 to visit 3, 2.8 ± 13.3 μm2 from visit 3 to visit 4, 1.4 ± 13.3 μm2 from visit 4 to visit 5, 1.3 ± 14.4 μm2 from visit 5 to visit 6.
Furthermore, there was no significant difference in the change in DCP FAZ area between consecutive monthly visits (repeated measures ANOVA p = 0.368). The change in DCP FAZ area was measured at 19.7 ± 34.6 μm2 from the baseline visit to visit 1, 19.0 ± 21.5 μm2 from visit 1 to visit 2, 14.3 ± 14.5 μm2 from visit 2 to visit 3, 17.1 ± 15.0 μm2 from visit 3 to visit 4, 13.3 ± 18.1 μm2 from visit 4 to visit 5, 15.6 ± 18.3 μm2 from visit 5 to visit 6.
Then, correlations between BCVA, CRT and FAZ area measurements were estimated. At baseline, there was no correlation between SCP FAZ area or DCP FAZ area or CRT and BCVA (p = 0.108, p = 0.337 and p = 0.184, respectively). A weak correlation existed between CRT and SCP FAZ area at baseline (r = 0.311, p = 0.028).
Interestingly, there was no correlation between SCP FAZ area and DCP FAZ area (p = 0.235) at baseline. In the final visit, after 6 intravitreal anti-VEGF injections, the only significant association was a weak correlation between SCP FAZ area and DCP FAZ area (r = 0.315, p = 0.026). SCP FAZ area was larger than DCP FAZ area at all timepoints (p < 0.001).
Finally, we tested whether the size of SCP FAZ area and DCP FAZ area at baseline correlates to the amount of BCVA gain with intravitreal treatment. There was no correlation between either SCP FAZ area (p = 0.598) or DCP FAZ area (p = 0.768) and BCVA gain.
4. Discussion
In the present study, OCT angiography imaging was used to quantify the effects of anti - VEGF intravitreal injections on SCP and DCP FAZ area and explore the changes that occur over time with a fixed treatment regimen. This was studied in all participants with monthly measurements and monthly injections for a period of 6 months. Interestingly, our results suggest that DCP FAZ area decreases with intravitreal injections of ranibizumab or aflibercept in treatment naïve DME. In fact, DCP FAZ area continued to decrease up to the 6 months visit. There was no effect of the choice of either ranibizumab or aflibercept detected in our cohort. There was also no significant change in SCP FAZ area, while BCVA increased and CRT decreased with treatment, as expected.
The role of anti-VEGF drugs in the evolution of ischemia has been previously addressed with the use of FA. Indeed it was speculated that treatment with anti-VEGF agents may in some cases even aggravate macula nonperfusion and ischemia [
17]. The hypothesis that anti - VEGF drugs could lead to increasing areas of ischemia was extensively studied analyzing data from the RISE and RIDE studies [
18] and also the RESTORE study [
19]. Indeed, Campochiaro et al. suggested that ranibizumab slows the progression of ischemia [
18]. Analysis of FA from the RESTORE study also concluded that repeated ranibizumab treatment was not associated with impaired macular perfusion. In fact, the authors urged physicians to offer therapy to patients with severe microangiopathy and advanced capillary dropout, based on their findings [
19]. In their study, Michaelides et al. also reported no change in FAZ area in a group of patients treated with bevacizumab for DME [
20]. Therefore, based on analysis of FA images, anti - VEGF treatment may in fact improve macular ischemia and change the natural history of the disease.
Previous studies in the literature with OCT angiography explored changes in FAZ with anti - VEGF treatment and reported either a decrease in FAZ area or no change. In their study, Falavarjani et al. measured FAZ area and vessel density before and after a single intravitreal anti - VEGF injection and no difference was detected [
21]. Our group previously reported that SCP FAZ remains similar, whereas DCP FAZ area and DCP ischemia decreases with 3 aflibercept injections [
22]. Superficial FAZ area showed improvement after 3 ranibizumab injections in the study by Hsieh et al. using Matlab to analyze OCT angiography images [
23]. SCP FAZ remained unchanged in another study in DME patients receiving anti - VEGF injections, in line to our study [
24]. In another recent report, Busch et al. quantified the FAZ area and retinal vascular area before and after aflibercept intravitreal injections in 23 eyes with DME [
25] and in their patients, FAZ area remained unchanged after treatment. Finally, Sorour et al. reported that macular vessel density remained unchanged in their cohort of patients with diabetic macular edema and proliferative diabetic retinopathy [
26]. These differences may be attributable to the segmentation algorithms and the differences in each measuring algorithms utilized by each imaging platform, the selection of Swept Source or Spectral Domain OCT and the evolution that each of these platforms has undergone in recent years, and possibly the inherent ocular characteristics of each study subject.
Moreover, the amount of difference in DCP FAZ area occurring every month with treatment did not significantly change over time. Interestingly, DCP FAZ area continued to decrease and therefore at least some patients continue to benefit with ongoing treatment. Analysis of our data did not point to a specific timepoint where this change occurs primarily in all patients. There is variability as shown also in the 95 % confidence intervals when looking at the DCP FAZ area change data.
Our study also found that SCP FAZ area is smaller than DCP FAZ area, which is in line with previous reports. Notably, the primary variation observed over time was in the DCP FAZ area, while the SCP FAZ area remained relatively constant throughout the duration of the study. Hence, the resolution of edema primarily impacts the structure surrounding the deep capillary network. The administration of ranibizumab or aflibercept resulted in comparable alterations in the DCP FAZ area. Furthermore, we observed no correlation between FAZ area measurements and BCVA gain or CRT improvement in our patients. This is not surprising, as there is a significant amount of variability in all of these parameters and the sample size is relatively small to accurately identify any possible associations. Larger studies with more patients can provide more power to detect such differences. Unlike OCT, it is generally accepted that there are likely no baseline OCT angiography biomarkers that directly correspond to the degree of improvement in anatomy [
23]. Moreover, employing new methods to analyze the data on FAZ area contained within the OCT angiography images could potentially reduce the extend of individual variability [
27].
There are specific constrains to our study. An important factor to consider is that the retinal vasculature exhibits a three - dimensional configuration. Our approach involves analyzing the changes that occur with treatment by looking only at two slabs from OCT angiography imaging. Therefore, we choose to analyze only a fraction of the information that is contained in the OCT angiography scans. This naturally is superior to looking at fluorescein angiography images, but examining the volume of the capillary plexi would be a more effective approach and would in fact provide a more accurate answer to the question of whether areas are trully re - perfused with treatment [
28].
In addition, the presence of edema impacts the OCT angiography software algorithm’s capacity to segment the tissue. It is more common for OCT angiography segmentation algorithms to fail to recognize correctly the anatomic boundaries of each slab in the presence of cystic spaces in the retina, compared to the healthy macula. For this reason, we also looked at specific images to check for segmentation errors. Remarkably, the built-in algorithm exhibited excellent performance and we did not observe any substantial deviations. A further limitation arises from the manual nature of the measurements conducted for SCP and DCP FAZ area sizes. This is counterbalanced by the fact that the mean of two measurements was documented for each timepoint, and also by the fact that the two graders showed remarkable agreement and there was very satisfactory intraindividual repeatability. The FAZ area is inherently variable, particularly when there is presence of intraretinal fluid. Nevertheless, previous studies have reported that the repeatability and reproducibility were excellent, in both normal eyes and in diabetics. However, it should be noted that there was greater variability in DCP FAZ area compared to SCP FAZ area [
29,
30].
In conclusion, our study measured the change over time in FAZ area in both the superficial and the deep capillary plexus of the retina. While no statistically significant difference was detected in the superficial capillary plexus FAZ area, deep layer ischemia improved and DCP FAZ area decreased with each injection. This effect was seen with both anti – VEGF drugs used; aflibercept and ranibizumab. Monthly measurements were conducted to more accurately describe the time series of changes and the impact in deep vasculature remained consistent. OCT angiography imaging can offer clinical insights for assessing and tracking the severity of deep macular ischemia and its evolution with anti - VEGF treatment.
Author Contributions
us “Conceptualization, D.M. and S.A.; methodology, D.M., E.T., A.B., S.A..; formal analysis, D.M. and A.D..; investigation, D.M.; resources, M.K. and A.K.; data curation, D.M.; writing—original draft preparation, D.M. and A.D.; writing—review and editing, S.A.; supervision, S.A... All authors have read and agreed to the published version of the manuscript
Funding
This research received no external funding
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of University Hospital of Larissa, Greece (protocol code 3355, 22.07.2016).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data are available on reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Bourne, R.R.A.; Jonas, J.B.; Bron, A.M.; Cicinelli, M.V.; Das, A.; Flaxman, S.R.; et al. Prevalence and causes of vision loss in high-income countries and in Eastern and Central Europe in 2015: magnitude, temporal trends and projections. Br J Ophthalmol. 2018, 102, 575–85. [Google Scholar] [CrossRef] [PubMed]
- Classification of Diabetic Retinopathy from Fluorescein Angiograms. Ophthalmology. 1991, 98, 807–22. [CrossRef]
- Abdelshafy, M.; Abdelshafy, A. Correlations Between Optical Coherence Tomography Angiography Parameters and the Visual Acuity in Patients with Diabetic Retinopathy. Clin Ophthalmol. 2020, 14, 1107–15. [Google Scholar] [CrossRef] [PubMed]
- Sim, D.A.; Keane, P.A.; Zarranz-Ventura, J.; Fung, S.; Powner, M.B.; Platteau, E.; et al. The Effects of Macular Ischemia on Visual Acuity in Diabetic Retinopathy. Investig Opthalmology Vis Sci. 2013, 54, 2353. [Google Scholar] [CrossRef] [PubMed]
- Cennamo, G.; Romano, M.R.; Nicoletti, G.; Velotti, N.; De Crecchio, G. Optical coherence tomography angiography versus fluorescein angiography in the diagnosis of ischaemic diabetic maculopathy. Acta Ophthalmol (Copenh) [Internet]. 2017 Feb [cited 2024 Mar 17];95(1). Available from: https://onlinelibrary.wiley.com/doi/10.1111/aos.13159.
- Garcia, J.M.B.D.B.; Lima, T.T.; Louzada, R.N.; Rassi, A.T.; Isaac, D.L.C.; Avila, M. Diabetic Macular Ischemia Diagnosis: Comparison between Optical Coherence Tomography Angiography and Fluorescein Angiography. J Ophthalmol. 2016, 2016, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Conrath, J.; Giorgi, R.; Raccah, D.; Ridings, B. Foveal avascular zone in diabetic retinopathy: quantitative vs qualitative assessment. Eye. 2005, 19, 322–6. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Kader, A.A.; Ramsey, D.J.; Yussuf, W.A.; Mohalhal, A.A.; Eldaly, M.A.; Elnahry, A.G. Diabetic microaneurysms detected by fluorescein angiography spatially correlate with regions of macular ischemia delineated by optical coherence tomography angiography. Indian J Ophthalmol. 2023, 71, 3085–90. [Google Scholar] [CrossRef]
- Waheed, N.K.; Rosen, R.B.; Jia, Y.; Munk, M.R.; Huang, D.; Fawzi, A.; et al. Optical coherence tomography angiography in diabetic retinopathy. Prog Retin Eye Res. 2023, 97, 101206. [Google Scholar] [CrossRef]
- Tsai, W.S.; Thottarath, S.; Gurudas, S.; Sen, P.; Pearce, E.; Giani, A.; et al. Correlation of Optical Coherence Tomography Angiography Characteristics with Visual Function to Define Vision-Threatening Diabetic Macular Ischemia. Diagnostics. 2022, 12, 1050. [Google Scholar] [CrossRef]
- Bhanushali, D.; Anegondi, N.; Gadde, S.G.K.; Srinivasan, P.; Chidambara, L.; Yadav, N.K.; et al. Linkin Retinal Microvasculature Features With Severity of Diabetic Retinopathy Using Optical Coherence Tomography Angiography. Investig Opthalmology Vis Sci. 2016, 57, OCT519. [Google Scholar] [CrossRef]
- Tsai, A.S.H.; Jordan-Yu, J.M.; Gan, A.T.L.; Teo, K.Y.C.; Tan, G.S.W.; Lee, S.Y.; et al. Diabetic Macular Ischemia: Influence of Optical Coherence Tomography Angiography Parameters on Changes in Functional Outcomes Over One Year. Investig Opthalmology Vis Sci. 2021, 62, 9. [Google Scholar] [CrossRef]
- Virgili, G.; Parravano, M.; Evans, J.R.; Gordon, I.; Lucenteforte, E. Anti-vascular endothelial growth factor for diabetic macular oedema: a network meta-analysis. Cochrane Eyes and Vision Group, editor. Cochrane Database Syst Rev [Internet]. 2018 Oct 16 [cited 2024 Mar 18];2018(10). Available from: http://doi.wiley.com/10.1002/14651858.CD007419.pub6.
- Chang, Y.C.; Huang, Y.T.; Hsu, A.Y.; Meng, P.P.; Lin, C.J.; Lai, C.T.; et al. Optical Coherence Tomography Biomarkers in Predicting Treatment Outcomes of Diabetic Macular Edema after Ranibizumab Injections. Medicina (Mex). 2023, 59, 629. [Google Scholar] [CrossRef]
- Rana, V.; Agarwal, A.; Arora, A.; Bansal, R.; Dogra, M.; Bhadada, S.K.; et al. Predicting visual outcomes following anti-vascular endothelial growth factor treatment for diabetic macular edema. Indian J Ophthalmol. 2024, 72 (Suppl 1), S16–21. [Google Scholar] [CrossRef] [PubMed]
- Ishibazawa, A.; Nagaoka, T.; Takahashi, A.; Omae, T.; Tani, T.; Sogawa, K.; et al. Optical Coherence Tomography Angiography in Diabetic Retinopathy: A Prospective Pilot Study. Am J Ophthalmol. 2015, 160, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, D.N.; Mendrinos, E.; Mangioris, G.; Donati, G.; Pournaras, C.J. Intravitreal Ranibizumab May Induce Retinal Arteriolar Vasoconstriction in Patients with Neovascular Age-related Macular Degeneration. Ophthalmology. 2009, 116, 1755–61. [Google Scholar] [CrossRef] [PubMed]
- Campochiaro, P.A.; Wykoff, C.C.; Shapiro, H.; Rubio, R.G.; Ehrlich, J.S. Neutralization of Vascular Endothelial Growth Factor Slows Progression of Retinal Nonperfusion in Patients with Diabetic Macular Edema. Ophthalmology. 2014, 121, 1783–9. [Google Scholar] [CrossRef]
- Karst, S.G.; Deak, G.G.; Gerendas, B.S.; Waldstein, S.M.; Lammer, J.; Simader, C.; et al. Association of Changes in Macular Perfusion With Ranibizumab Treatment for Diabetic Macular Edema: A Subanalysis of the RESTORE (Extension) Study. JAMA Ophthalmol. 2018, 136, 315. [Google Scholar] [CrossRef] [PubMed]
- Michaelides, M.; Fraser-Bell, S.; Hamilton, R.; Kaines, A.; Egan, C.; Bunce, C.; et al. MACULAR PERFUSION DETERMINED BY FUNDUS FLUORESCEIN ANGIOGRAPHY AT THE 4-MONTH TIME POINT IN A PROSPECTIVE RANDOMIZED TRIAL OF INTRAVITREAL BEVACIZUMAB OR LASER THERAPY IN THE MANAGEMENT OF DIABETIC MACULAR EDEMA (BOLT STUDY): Report 1. Retina. 2010, 30, 781–6. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi Falavarjani, K.; Iafe, N.A.; Hubschman, J.P.; Tsui, I.; Sadda, S.R.; Sarraf, D. Optical Coherence Tomography Angiography Analysis of the Foveal Avascular Zone and Macular Vessel Density After Anti-VEGF Therapy in Eyes With Diabetic Macular Edema and Retinal Vein Occlusion. Investig Opthalmology Vis Sci. 2017, 58, 30. [Google Scholar] [CrossRef]
- Dastiridou, A.; Karathanou, K.; Riga, P.; Anagnostopoulou, S.; Balasubramanian, S.; Mataftsi, A.; et al. OCT Angiography Study of the Macula in Patients with Diabetic Macular Edema Treated with Intravitreal Aflibercept. Ocul Immunol Inflamm. 2021, 29, 926–31. [Google Scholar] [CrossRef]
- Hsieh, Y.T.; Alam, M.N.; Le, D.; Hsiao, C.C.; Yang, C.H.; Chao, D.L.; et al. OCT Angiography Biomarkers for Predicting Visual Outcomes after Ranibizumab Treatment for Diabetic Macular Edema. Ophthalmol Retina. 2019, 3, 826–34. [Google Scholar] [CrossRef] [PubMed]
- Conti, F.F.; Song, W.; Rodrigues, E.B.; Singh, R.P. Changes in retinal and choriocapillaris density in diabetic patients receiving anti-vascular endothelial growth factor treatment using optical coherence tomography angiography. Int J Retina Vitr. 2019, 5, 41. [Google Scholar] [CrossRef] [PubMed]
- Busch, C.; Wakabayashi, T.; Sato, T.; Fukushima, Y.; Hara, C.; Shiraki, N.; et al. Retinal Microvasculature and Visual Acuity after Intravitreal Aflibercept in Diabetic Macular Edema: An Optical Coherence Tomography Angiography Study. Sci Rep. 2019, 9, 1561. [Google Scholar] [CrossRef]
- Sorour, O.A.; Sabrosa, A.S.; Yasin Alibhai, A.; Arya, M.; Ishibazawa, A.; Witkin, A.J.; et al. Optical coherence tomography angiography analysis of macular vessel density before and after anti-VEGF therapy in eyes with diabetic retinopathy. Int Ophthalmol. 2019, 39, 2361–71. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; He, Y.; Kong, L.; Yang, D.; Lu, N.; Yu, Y.; et al. Study of Foveal Avascular Zone Growth in Individuals With Mild Diabetic Retinopathy by Optical Coherence Tomography. Investig Opthalmology Vis Sci. 2023, 64, 21. [Google Scholar] [CrossRef]
- Spaide, R.F. Volume-Rendered Optical Coherence Tomography of Diabetic Retinopathy Pilot Study. Am J Ophthalmol. 2015, 160, 1200–10. [Google Scholar] [CrossRef]
- Carpineto, P.; Mastropasqua, R.; Marchini, G.; Toto, L.; Di Nicola, M.; Di Antonio, L. Reproducibility and repeatability of foveal avascular zone measurements in healthy subjects by optical coherence tomography angiography. Br J Ophthalmol. 2016, 100, 671–6. [Google Scholar] [CrossRef]
- Czakó, C.; Sándor, G.; Ecsedy, M.; Récsán, Z.; Horváth, H.; Szepessy, Z.; et al. Intrasession and Between-Visit Variability of Retinal Vessel Density Values Measured with OCT Angiography in Diabetic Patients. Sci Rep. 2018, 8, 10598. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).