Figure 1.
Models mentioned in the text. a, Columnar model of Herrick (1910), postulating an unbent length axis ending in the telencephalon. Note the notochord lies only under the brainstem (Mes, Rhomb), but the postulated longitudinal columns also extend into the forebrain (Dienc, Tel). The rostral ends of the floor and roof domains were not indicated. b,c,d, Cross-sections through Tel, Dienc and Rhomb in a, illustrating the postulated four columns at each level. e, Modification of Herrick’s columnar model proposed by Swanson (2004, 2012). Essentially, a general division into basal (cream) and alar (blue) plates is proposed, again without topographic correlation with the notochord. Roof and floor are not mapped precisely, but the postulated floor possibly reaches the anterior commissure (ac), implying a prechordal part. f, Neuromeric model of Bergquist and Källén (1954), encompassing also His’s (1893, 1904) longitudinal zones (names at caudal end) and alar-basal boundary (red dashes). Note Tel forms a unit with the hypothalamus, the secondary prosencephalon (green). The diencephalon lies caudal to that and consists of three transverse neuromeres, thought to represent more faithfully the domains with identical color codes in a,c. All neuromeres extend from roof to floor, whose rostral ends are not represented. The intersection of longitudinal and transversal limits creates a number of quadrangular ‘migration areas’. M indicates mamillary body. g, Neuromeric (prosomeric) model of Puelles et al. (2012; updated significantly relative to earlier versions). There are three parallel axial references: 1) the floor (blue) ending rostrally at mamillary body, 2) the alar-basal boundary (red dash-line; determined molecularly in the forebrain by Shh and Nkx2.2 markers), and 3) the roof plate (yellow), fate-mapped in several vertebrate species to end at the anterior commissure (ac). The concept of unitary secondary prosencephalon from f is maintained, subdivided into two hypothalamo-telencephalic prosomeres (hp1, hp2), whose floor is retromamillary (RM) or mamillary (M). The respective parts of hypothalamus are named ‘peduncular hypothalamus’ (PHy) and ‘terminal hypothalamus (THy). Hp2 ends rostrally at the acroterminal rostromedian domain (green), stretched between rostral roof and rostral floor, which displays unique formations such as the alar preoptic lamina terminalis and the optic chiasma, and the basal infundibular tuberal region with the neurohypophysis. The dienc is trineuromeric, like in f. The midbrain has two neuromeres (m1, m2) and ends at the isthmo-mesencephalic boundary (thick black dashes). The hindbrain shows 12 rhombomeres, some of which are cryptic (detected only molecularly and experimentally). The hindbrain has four proneuromeres (prepontine: pp, pontine: pons, retropontine: rpon, and medulla: med; limits as intermediate dashes). The spinal cord (beyond the rhombospinal boundary; thick black dashes) has myelomeres.
Figure 1.
Models mentioned in the text. a, Columnar model of Herrick (1910), postulating an unbent length axis ending in the telencephalon. Note the notochord lies only under the brainstem (Mes, Rhomb), but the postulated longitudinal columns also extend into the forebrain (Dienc, Tel). The rostral ends of the floor and roof domains were not indicated. b,c,d, Cross-sections through Tel, Dienc and Rhomb in a, illustrating the postulated four columns at each level. e, Modification of Herrick’s columnar model proposed by Swanson (2004, 2012). Essentially, a general division into basal (cream) and alar (blue) plates is proposed, again without topographic correlation with the notochord. Roof and floor are not mapped precisely, but the postulated floor possibly reaches the anterior commissure (ac), implying a prechordal part. f, Neuromeric model of Bergquist and Källén (1954), encompassing also His’s (1893, 1904) longitudinal zones (names at caudal end) and alar-basal boundary (red dashes). Note Tel forms a unit with the hypothalamus, the secondary prosencephalon (green). The diencephalon lies caudal to that and consists of three transverse neuromeres, thought to represent more faithfully the domains with identical color codes in a,c. All neuromeres extend from roof to floor, whose rostral ends are not represented. The intersection of longitudinal and transversal limits creates a number of quadrangular ‘migration areas’. M indicates mamillary body. g, Neuromeric (prosomeric) model of Puelles et al. (2012; updated significantly relative to earlier versions). There are three parallel axial references: 1) the floor (blue) ending rostrally at mamillary body, 2) the alar-basal boundary (red dash-line; determined molecularly in the forebrain by Shh and Nkx2.2 markers), and 3) the roof plate (yellow), fate-mapped in several vertebrate species to end at the anterior commissure (ac). The concept of unitary secondary prosencephalon from f is maintained, subdivided into two hypothalamo-telencephalic prosomeres (hp1, hp2), whose floor is retromamillary (RM) or mamillary (M). The respective parts of hypothalamus are named ‘peduncular hypothalamus’ (PHy) and ‘terminal hypothalamus (THy). Hp2 ends rostrally at the acroterminal rostromedian domain (green), stretched between rostral roof and rostral floor, which displays unique formations such as the alar preoptic lamina terminalis and the optic chiasma, and the basal infundibular tuberal region with the neurohypophysis. The dienc is trineuromeric, like in f. The midbrain has two neuromeres (m1, m2) and ends at the isthmo-mesencephalic boundary (thick black dashes). The hindbrain shows 12 rhombomeres, some of which are cryptic (detected only molecularly and experimentally). The hindbrain has four proneuromeres (prepontine: pp, pontine: pons, retropontine: rpon, and medulla: med; limits as intermediate dashes). The spinal cord (beyond the rhombospinal boundary; thick black dashes) has myelomeres.
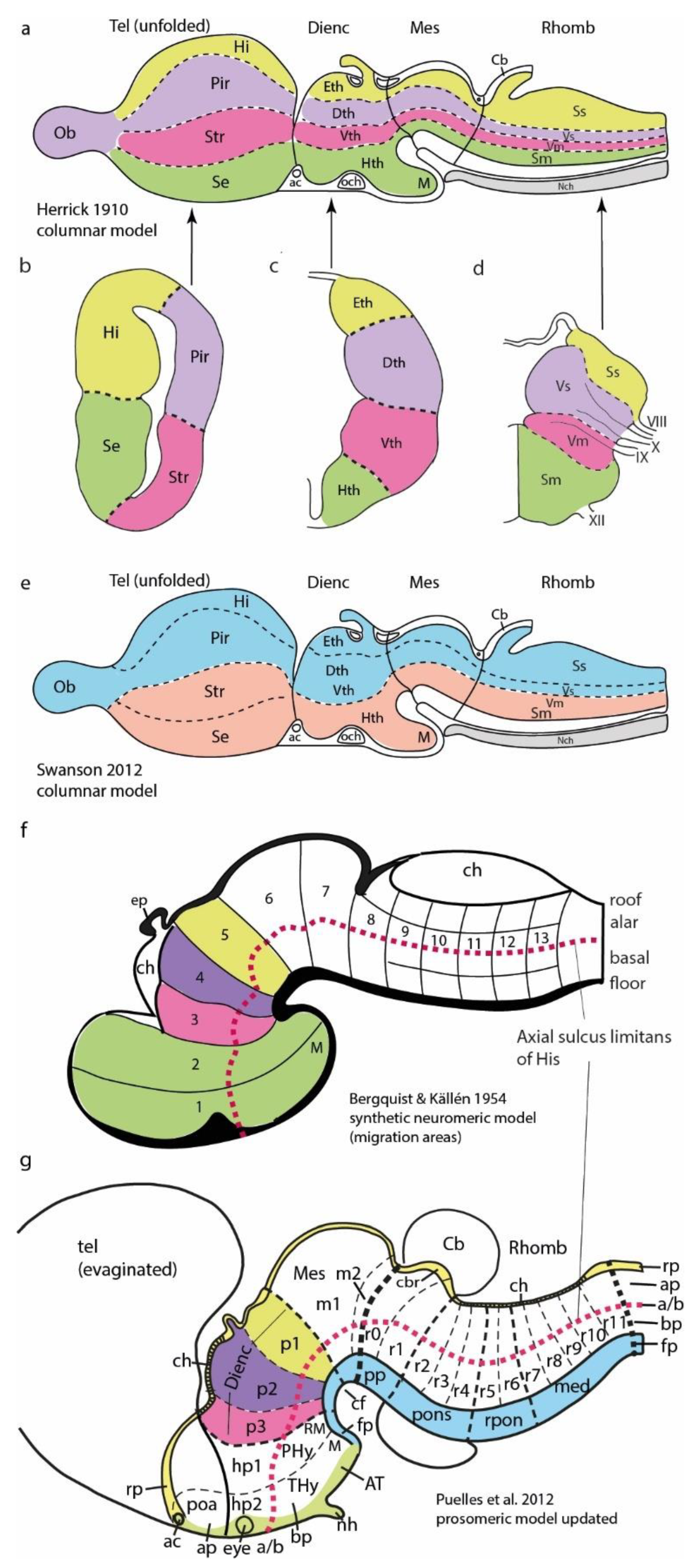
Figure 2.
Krieg’s (1932) nuclear structure of the rat hypothalamus. Krieg produced this relatively precise 3D view of hypothalamic nuclear variety in his Figure 21 (note the hypothalamus was one of Herrick’s diencephalic columns). There is no apparent order in the nuclear arrangement. The columnar caudorostral axis runs from right to left, so that the bottom of the reconstruction (Inf) was regarded as the floor of the diencephalon. We see the optic nerve (ON) and the anterior commissure (AC) at the rostral end. AC anterior commissure, Ant anterior nucleus, Arc arcuate nucleus, DMD dorsal part, dorsomedial nucleus, DMV ventral part, dorsomedial nucleus, Inf infundibulum, ML lateral mamillary nucleus, MM medial mamillary nucleus, MML mediomedial mamillary nucleus, MPI posterior intermediate mamillary nucleus, MP posterior mamillary nucleus, PA preoptic area, FilM filiform (paraventricular) nucleus, PD dorsal premamillary nucleus, Post posterior hypothalamic nucleus, Pre preoptic nucleus, PV ventral premamillary nucleus, OC optic chiasma, ON optic nerve, OT optic tract, Sch suprachiasmatic nucleus, Sum supramamillary nucleus, Sup supraoptic nucleus, VM ventromedial nucleus.
Figure 2.
Krieg’s (1932) nuclear structure of the rat hypothalamus. Krieg produced this relatively precise 3D view of hypothalamic nuclear variety in his Figure 21 (note the hypothalamus was one of Herrick’s diencephalic columns). There is no apparent order in the nuclear arrangement. The columnar caudorostral axis runs from right to left, so that the bottom of the reconstruction (Inf) was regarded as the floor of the diencephalon. We see the optic nerve (ON) and the anterior commissure (AC) at the rostral end. AC anterior commissure, Ant anterior nucleus, Arc arcuate nucleus, DMD dorsal part, dorsomedial nucleus, DMV ventral part, dorsomedial nucleus, Inf infundibulum, ML lateral mamillary nucleus, MM medial mamillary nucleus, MML mediomedial mamillary nucleus, MPI posterior intermediate mamillary nucleus, MP posterior mamillary nucleus, PA preoptic area, FilM filiform (paraventricular) nucleus, PD dorsal premamillary nucleus, Post posterior hypothalamic nucleus, Pre preoptic nucleus, PV ventral premamillary nucleus, OC optic chiasma, ON optic nerve, OT optic tract, Sch suprachiasmatic nucleus, Sum supramamillary nucleus, Sup supraoptic nucleus, VM ventromedial nucleus.
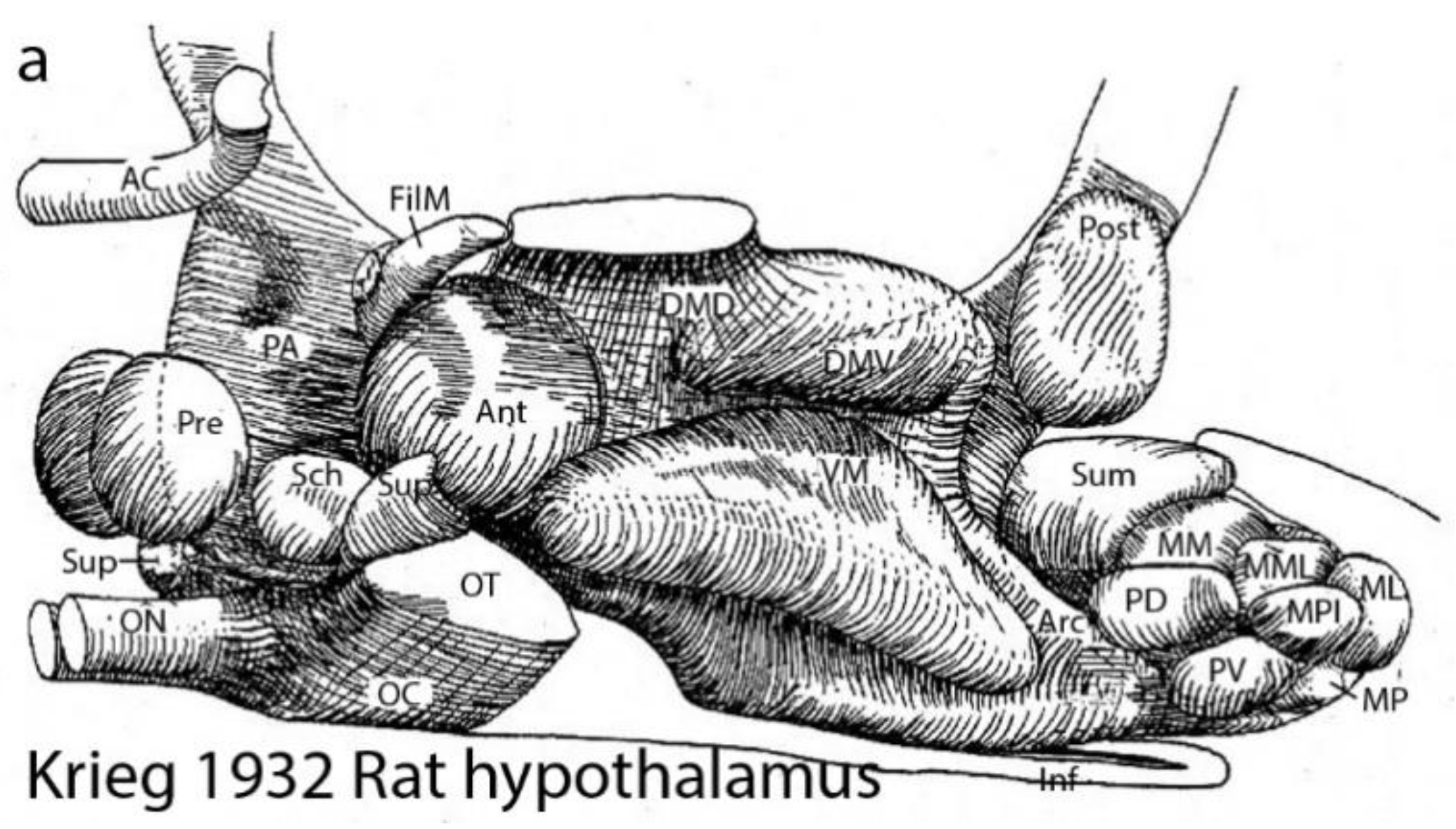
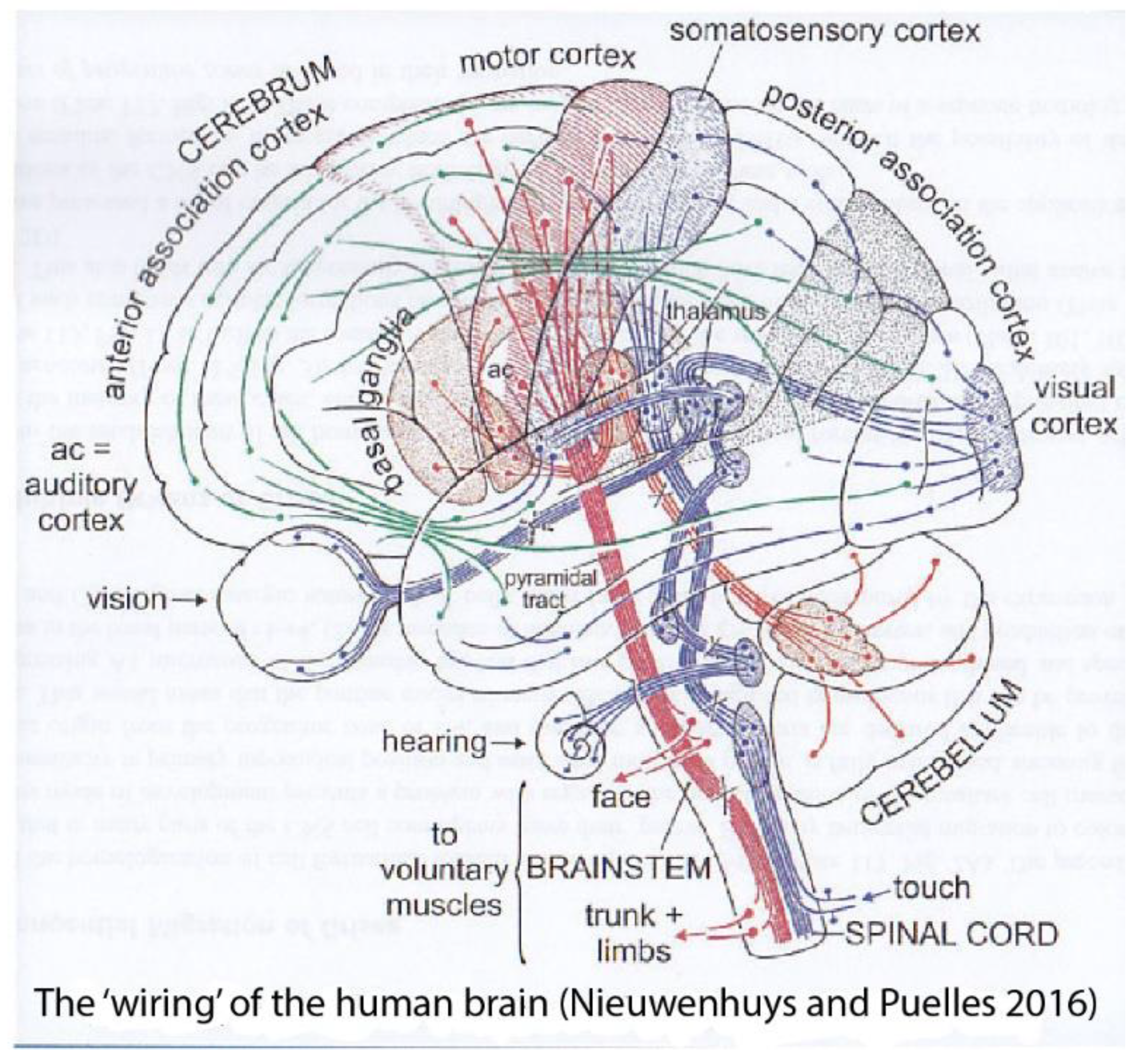
Figure 3.
The wiring of the human brain (Figure used in a 1992 talk, reproduced from Nieuwenhuys and Puelles 2016). Schema displaying sensory and motor pathways jointly with some interconnections.
Figure 3.
The wiring of the human brain (Figure used in a 1992 talk, reproduced from Nieuwenhuys and Puelles 2016). Schema displaying sensory and motor pathways jointly with some interconnections.
Figure 4.
The model of His (1893) and its DV elements, in modern perspective.
a, Schema of His (1893) with his precise DV subdivision into floor, basal, alar, and roof longitudinal zones (including the limiting and appropriately bent alar-basal boundary sulcus ending at the optic stalk). His also entered relatively imprecise AP partitions (Roman/Arabic tags) now substituted by neuromeres. He was the first to postulate the isthmic domain (tagged III1/III2) as a separate brain segment (see
Figure 5c).
b, Mouse E10.5 brain showing the alar-basal band of Nkx2.2 expression (downstream of notochordal SHH signal) which approximates the sulcus of His in
a. The neuromeric alar ventricular concavities of the three diencephalic segments are visible (p1, p2, p3). The zona limitans interthalamica (zl) is a transverse singularity of the p2/p3 boundary, caused by a separate Shh enhancer, secondarily affecting Nkx2.2.
c,
d, Floor plate labeling at E11.5 and E13.5 with Ntn1 (from Puelles et al. 2012; Allen Institute data). The floor stops rostrally over the tip of the notochord at the mamillary pouch (mam). Note also slight Ntn1 signal at E13.5 at the locus of the anterior commissure (fate-mapped rostralmost roof plate).
e, Three-day old chick embryo brain reacted wholemount for AChE (from Amat et al. 2022). This marker identifies postmitotic neurons and is negative generally in progenitor cells (labeling at the zl -p2/p3 interneuromeric limit as in
b- contrarily marks radial glia or progenitors). The neurogenetically precocious basal zone is massively full of neurons, while the retarded alar zone is relatively unpopulated at this stage. Note some transverse neuromeric borders, orthogonal to the alar-basal limit. The telencephalon shows a fully alar pattern, falsating columnar assumptions.
f,g, Selective expression of Dlk1 at the mouse acroterminal domain at E13.5 and E15.5 (data from the Allen Developmental Mouse Brain Atlas).
Figure 4.
The model of His (1893) and its DV elements, in modern perspective.
a, Schema of His (1893) with his precise DV subdivision into floor, basal, alar, and roof longitudinal zones (including the limiting and appropriately bent alar-basal boundary sulcus ending at the optic stalk). His also entered relatively imprecise AP partitions (Roman/Arabic tags) now substituted by neuromeres. He was the first to postulate the isthmic domain (tagged III1/III2) as a separate brain segment (see
Figure 5c).
b, Mouse E10.5 brain showing the alar-basal band of Nkx2.2 expression (downstream of notochordal SHH signal) which approximates the sulcus of His in
a. The neuromeric alar ventricular concavities of the three diencephalic segments are visible (p1, p2, p3). The zona limitans interthalamica (zl) is a transverse singularity of the p2/p3 boundary, caused by a separate Shh enhancer, secondarily affecting Nkx2.2.
c,
d, Floor plate labeling at E11.5 and E13.5 with Ntn1 (from Puelles et al. 2012; Allen Institute data). The floor stops rostrally over the tip of the notochord at the mamillary pouch (mam). Note also slight Ntn1 signal at E13.5 at the locus of the anterior commissure (fate-mapped rostralmost roof plate).
e, Three-day old chick embryo brain reacted wholemount for AChE (from Amat et al. 2022). This marker identifies postmitotic neurons and is negative generally in progenitor cells (labeling at the zl -p2/p3 interneuromeric limit as in
b- contrarily marks radial glia or progenitors). The neurogenetically precocious basal zone is massively full of neurons, while the retarded alar zone is relatively unpopulated at this stage. Note some transverse neuromeric borders, orthogonal to the alar-basal limit. The telencephalon shows a fully alar pattern, falsating columnar assumptions.
f,g, Selective expression of Dlk1 at the mouse acroterminal domain at E13.5 and E15.5 (data from the Allen Developmental Mouse Brain Atlas).
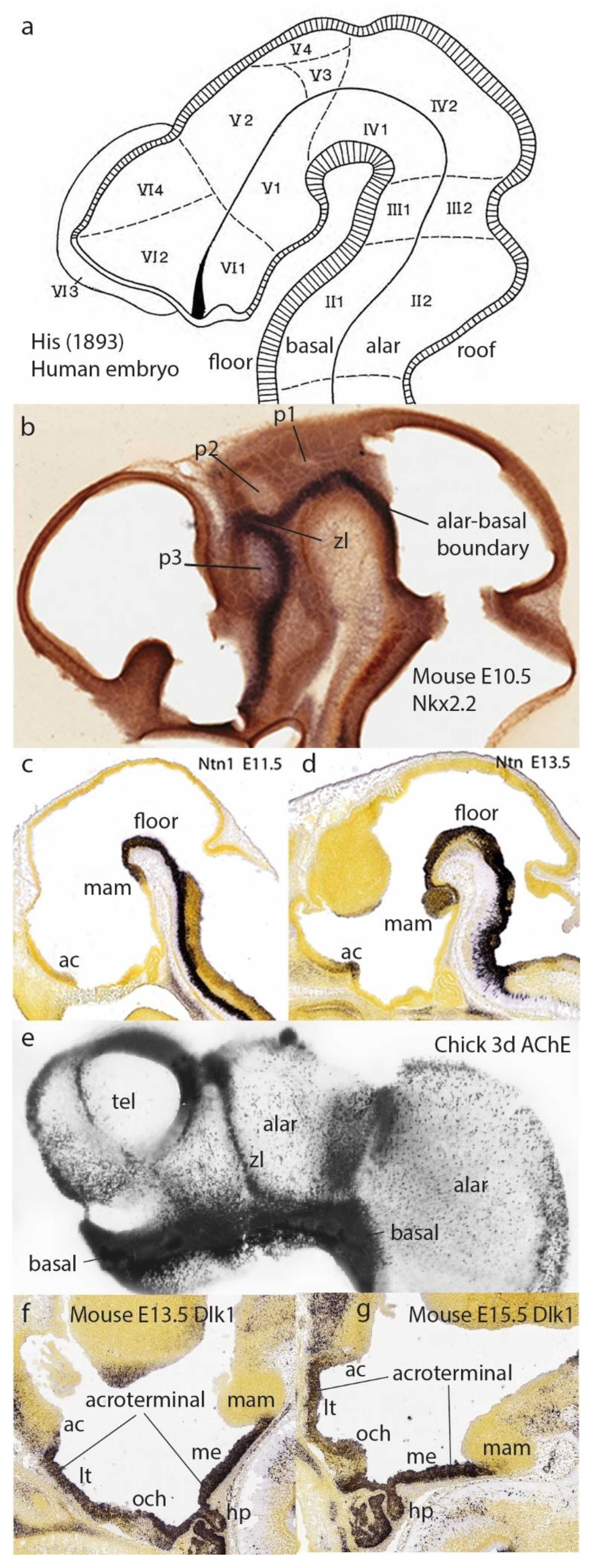
Figure 5.
Neuromeres, their DV subdivisions, plus underlying migratory and clonal aspects. a, Forebrain and hindbrain neuromeric bulges in the shark Squalus acanthias, drawn by Locy (1895). The hindbrain shows 5 preotic rhombomeres comprising prepontine and pontine units (now called r0-r4) and 3 postotic units (r5-r7) corresponding to retropontine (r5,r6) and the first medullary unit (r7). b, Graphic reconstruction of forebrain prosomeres in an E11.5 rat brain, created out of a sagittal section series. The postulated neuromeric cavities of hp1/hp2 (hypothalamus), p1-p3 (diencephalon) and m1/m2 (midbrain) were captured (Puelles et al. 2015); see the original for individual sections and correlated description of wholemount AChE-stained postmitotic neurons. c,d, Coronal and sagittal sections through transgenic adult mouse brains carrying a Fgf8-LacZ construct that labels all progeny derived from progenitors that expressed early on the Fgf8 gene (a marker typical of the embryonic isthmic rhombomere r0). The observed blue LacZ reaction demonstrates that the neuromere-derived adult brain part continues being transversal, limits with the caudal midbrain, participates in the vermal cerebellum, and is complete from ventricle to pia; i.e., the neuromere is still there with conserved boundaries, but has transformed into its adult counterpart (some blue-labeled cell populations seem to have migrated out of the isthmus into neighboring areas). Analogous material exists for other neuromeres. e,f, Schemata of fine microzonal regionalization subdividing the basal and alar domains, observed with molecular markers and experimentation at the spinal cord (e) and hindbrain neuromeres (f). Usually there are 5 basal microzones and 6-7 alar ones. Different neuronal populations are produced at each locus. The spinal DL domain (e) forms the layered dorsal horn, mixing derivatives from several microzones. In the basal plate, the MN microzone forms the motoneurons, whereas the others form different sorts of interneurons. In the hindbrain (f) the alar microzones form the sensory columns (color-coded) and the rhombic lip, dorsally, from where most neurons migrate subpially ventralwards, forming e.g. the pontine nuclei. Violet colored motoneurons form three separate columns, two of them migrated into the VL alar plate (these were classified classically as visceromotor, ignoring that all motoneurons -different subtypes- come from the same MN microzone). g, Radial migration in the cerebellar cortex of a transgenically LacZ-labeled clone composed of different neuron types (Purkinje: P, Golgi: G, Molecular: M), derived from a single very rare LacZ-reconstitutive event in a progenitor cell labeled by a non-functional LaacZ construct (Mathis et al. 1997). h, Pax7-LacZ progeny derives exclusively from the alar plate, so that one expects in the spinal cord only massive blue derivatives in the dorsal horn; however, a number of blue alar neurons apparently migrate into the ventral horn (basal plate), probably representing interneurons.
Figure 5.
Neuromeres, their DV subdivisions, plus underlying migratory and clonal aspects. a, Forebrain and hindbrain neuromeric bulges in the shark Squalus acanthias, drawn by Locy (1895). The hindbrain shows 5 preotic rhombomeres comprising prepontine and pontine units (now called r0-r4) and 3 postotic units (r5-r7) corresponding to retropontine (r5,r6) and the first medullary unit (r7). b, Graphic reconstruction of forebrain prosomeres in an E11.5 rat brain, created out of a sagittal section series. The postulated neuromeric cavities of hp1/hp2 (hypothalamus), p1-p3 (diencephalon) and m1/m2 (midbrain) were captured (Puelles et al. 2015); see the original for individual sections and correlated description of wholemount AChE-stained postmitotic neurons. c,d, Coronal and sagittal sections through transgenic adult mouse brains carrying a Fgf8-LacZ construct that labels all progeny derived from progenitors that expressed early on the Fgf8 gene (a marker typical of the embryonic isthmic rhombomere r0). The observed blue LacZ reaction demonstrates that the neuromere-derived adult brain part continues being transversal, limits with the caudal midbrain, participates in the vermal cerebellum, and is complete from ventricle to pia; i.e., the neuromere is still there with conserved boundaries, but has transformed into its adult counterpart (some blue-labeled cell populations seem to have migrated out of the isthmus into neighboring areas). Analogous material exists for other neuromeres. e,f, Schemata of fine microzonal regionalization subdividing the basal and alar domains, observed with molecular markers and experimentation at the spinal cord (e) and hindbrain neuromeres (f). Usually there are 5 basal microzones and 6-7 alar ones. Different neuronal populations are produced at each locus. The spinal DL domain (e) forms the layered dorsal horn, mixing derivatives from several microzones. In the basal plate, the MN microzone forms the motoneurons, whereas the others form different sorts of interneurons. In the hindbrain (f) the alar microzones form the sensory columns (color-coded) and the rhombic lip, dorsally, from where most neurons migrate subpially ventralwards, forming e.g. the pontine nuclei. Violet colored motoneurons form three separate columns, two of them migrated into the VL alar plate (these were classified classically as visceromotor, ignoring that all motoneurons -different subtypes- come from the same MN microzone). g, Radial migration in the cerebellar cortex of a transgenically LacZ-labeled clone composed of different neuron types (Purkinje: P, Golgi: G, Molecular: M), derived from a single very rare LacZ-reconstitutive event in a progenitor cell labeled by a non-functional LaacZ construct (Mathis et al. 1997). h, Pax7-LacZ progeny derives exclusively from the alar plate, so that one expects in the spinal cord only massive blue derivatives in the dorsal horn; however, a number of blue alar neurons apparently migrate into the ventral horn (basal plate), probably representing interneurons.
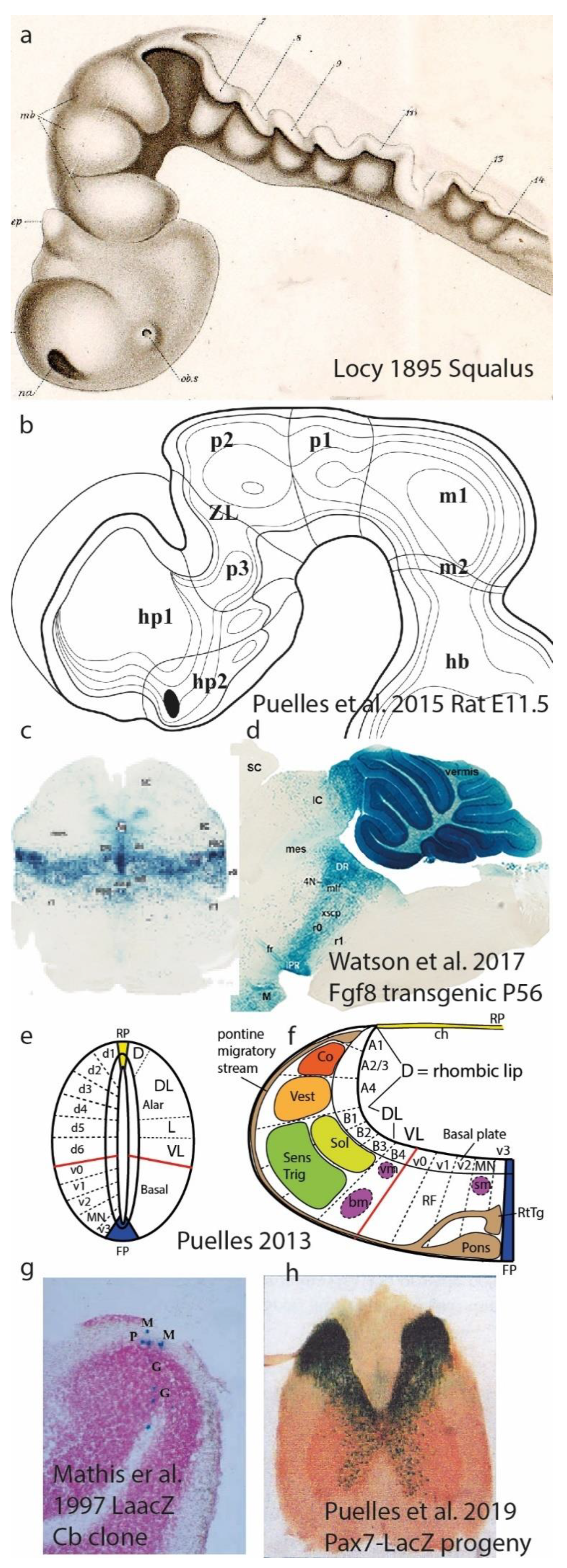
Figure 6.
Secondary (advanced) DV and AP microzonal regionalization of neuromeres. a, Prosomeric map of advanced DV patterning reported at different places of the neural tube, collected from literature and personal results by Nieuwenhuys and Puelles (2016). The mapping was already incomplete at forebrain levels at that date, due to hypothalamic data of Puelles et al. 2012 and unrepresented pretectal data from Ferran et al. 2009. However, it allows to appreciate the commonality of the pattern observed in spinal cord and hindbrain. b,c, These panels show partial molecular mapping data in the hypothalamus that suggest a systematic alar and basal DV subdivision into roughly parallel (longitudinal) molecularly diverse microzonal compartments, both at the lateral hypothalamus (b; Diaz et al. 2022) and in the general bineuromeric distribution of peptidergic neurons of several subtypes (c; Puelles et al. 2012). Several single cell transcriptomic studies corroborate multiple cell types of various sorts in these areas. Our studies correlated this distribution with our molecular characterization of the early progenitor domains. Note the optic chiasma roughly marks the alar-basal boundary. d-f, Illustration of advanced (secondary) AP microzonal tripartite pattern in the pretectal alar plate (p1 prosomere: precommissural PcP, juxtacommissural JcP, and commissural CoP AP compartments; Ferran et al. 2009). d shows an early stage previous to DV microzonation, while e displays the added DV subdivision. f presents a horizontal section in a 9d chick embryo (plane indicated in e by a blue bar), where the pretectum (alar p1; boundaries as dashed black lines) is seen lying intercalated between the thalamus (in alar p2) and the midbrain tectum (in alar m1). Foxp2 signal characterizes selectively intermediate and superficial strata of the rostral PcP subdomain, while PAX7 immunoreaction marks likewise diverse stratified CoP derivatives; the intermediate JcP subdomain is negative for these two markers, but stains specifically, e.g., for Six3. This result simultaneously shows the radially complete AP partition and reveals that each microzone forms different neuronal subtypes that occupy different layers of the microzonal mantle. Each of these derivatives is fated to produce different nuclei or cell layers, with differential connective properties. The m1 midbrain tectum also shows in this image a more thinly layered rostral microzonal component found next to the CoP, and a more thickly layered caudal component (the optic tectum). The signal-free boundary area between thalamus and PcP may represent the missing third thalamic subdivision (consistent with all neuromeres having this pattern).
Figure 6.
Secondary (advanced) DV and AP microzonal regionalization of neuromeres. a, Prosomeric map of advanced DV patterning reported at different places of the neural tube, collected from literature and personal results by Nieuwenhuys and Puelles (2016). The mapping was already incomplete at forebrain levels at that date, due to hypothalamic data of Puelles et al. 2012 and unrepresented pretectal data from Ferran et al. 2009. However, it allows to appreciate the commonality of the pattern observed in spinal cord and hindbrain. b,c, These panels show partial molecular mapping data in the hypothalamus that suggest a systematic alar and basal DV subdivision into roughly parallel (longitudinal) molecularly diverse microzonal compartments, both at the lateral hypothalamus (b; Diaz et al. 2022) and in the general bineuromeric distribution of peptidergic neurons of several subtypes (c; Puelles et al. 2012). Several single cell transcriptomic studies corroborate multiple cell types of various sorts in these areas. Our studies correlated this distribution with our molecular characterization of the early progenitor domains. Note the optic chiasma roughly marks the alar-basal boundary. d-f, Illustration of advanced (secondary) AP microzonal tripartite pattern in the pretectal alar plate (p1 prosomere: precommissural PcP, juxtacommissural JcP, and commissural CoP AP compartments; Ferran et al. 2009). d shows an early stage previous to DV microzonation, while e displays the added DV subdivision. f presents a horizontal section in a 9d chick embryo (plane indicated in e by a blue bar), where the pretectum (alar p1; boundaries as dashed black lines) is seen lying intercalated between the thalamus (in alar p2) and the midbrain tectum (in alar m1). Foxp2 signal characterizes selectively intermediate and superficial strata of the rostral PcP subdomain, while PAX7 immunoreaction marks likewise diverse stratified CoP derivatives; the intermediate JcP subdomain is negative for these two markers, but stains specifically, e.g., for Six3. This result simultaneously shows the radially complete AP partition and reveals that each microzone forms different neuronal subtypes that occupy different layers of the microzonal mantle. Each of these derivatives is fated to produce different nuclei or cell layers, with differential connective properties. The m1 midbrain tectum also shows in this image a more thinly layered rostral microzonal component found next to the CoP, and a more thickly layered caudal component (the optic tectum). The signal-free boundary area between thalamus and PcP may represent the missing third thalamic subdivision (consistent with all neuromeres having this pattern).
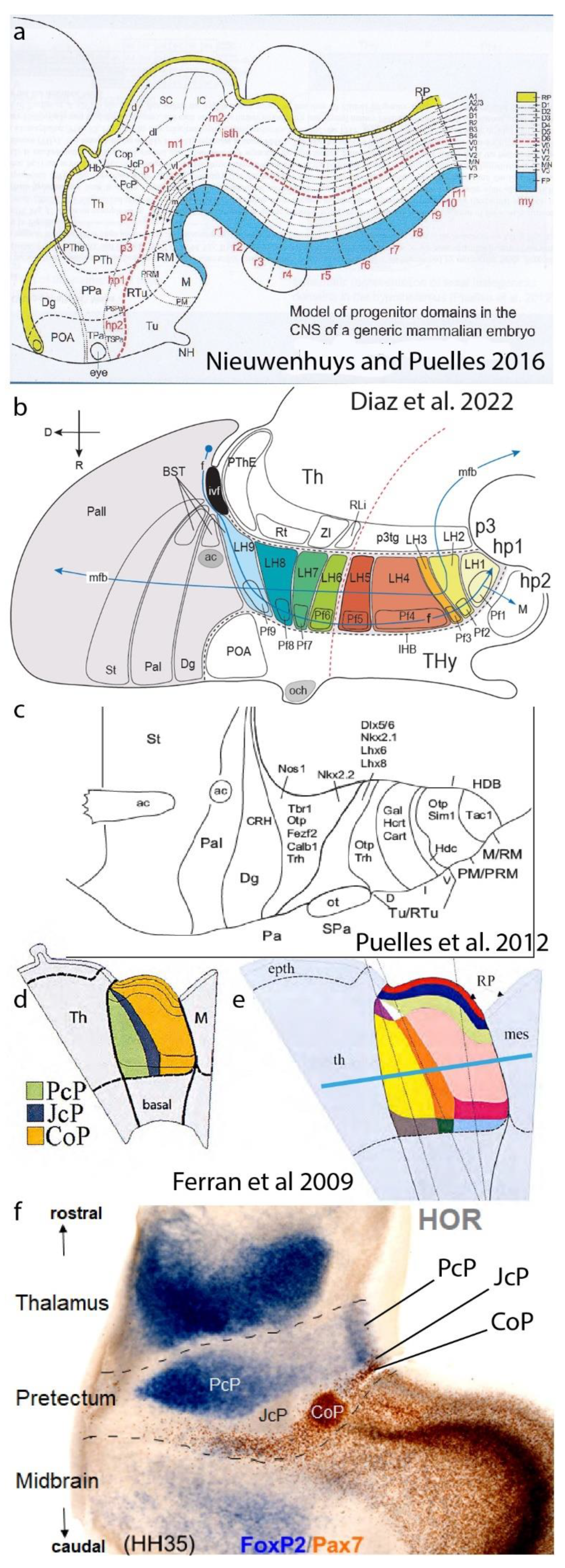
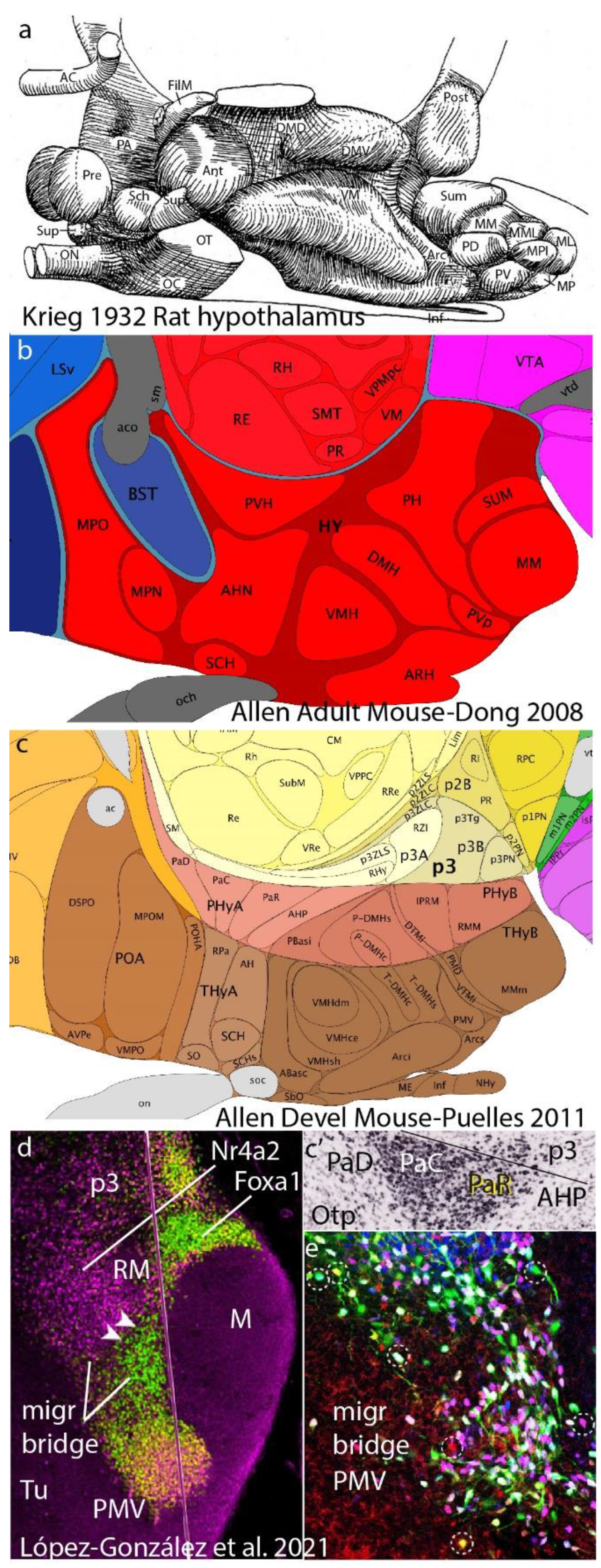
Figure 7.
Recapitulation of our argument using the hypothalamus as example. a, b. These panels show two examples of columnar order-less description of nuclei in the hypothalamus, by Krieg (1932) in a, and Dong (2008) in b, representing examples of what we have called ‘potato-sack morphology’, that is, description without any sort of classificatory or explaining concepts. c, Here LP, working in collaboration with the Allen Institute on the Ontology and Reference Atlases of the Developing Mouse Brain Atlas (2008-2011), proposed AP partition of the P56 hypothalamus into two hypothalamic neuromeres, defining their respective floor, basal, alar and roof domains plus DV microzonal divisions. The known adult nuclei fell naturally into the resulting intersectional pigeon-holes, and were consistent with gene patterns analyzed. This effort using some 4,000 genes at the Allen Institute helped evolving the more complete, updated prosomeric model of Puelles et al. 2012a. d,e, Jumping now to phenomena in a single microzone from López-González et al. 2021, we show a sagittal image of the retromamillary area (RM; a basal plate area of hp1 next to the floor, understood within columnar theory as ‘supramamillary area’) where we see distinct ventral and dorsal subareas labeled differentially for Foxa1 (green) and Nr4a2 (fuchsia). Both populations generate cells that enter a rostralward migration stream that courses above the mamillary body into hp2, and enters the suprajacent ventral tuberal area. As the cells approach rostrally the acroterminal area (which may have attracted them) they stop migrating, forming an oval-shaped nucleus, the classically known ventral premamillary nucleus. There is very little cell mixing with cells derived from hp2, whose cells often express quite different gene markers. In contrast, the PMV migration is composed of intermixed green- and fuchsia-labeled RM cells, which subdivide into several intermixed subgroups according to which of the two markers they express. The final PMV nucleus has a molecularly distinguishable core and shell structure with multiple cell types.
Figure 7.
Recapitulation of our argument using the hypothalamus as example. a, b. These panels show two examples of columnar order-less description of nuclei in the hypothalamus, by Krieg (1932) in a, and Dong (2008) in b, representing examples of what we have called ‘potato-sack morphology’, that is, description without any sort of classificatory or explaining concepts. c, Here LP, working in collaboration with the Allen Institute on the Ontology and Reference Atlases of the Developing Mouse Brain Atlas (2008-2011), proposed AP partition of the P56 hypothalamus into two hypothalamic neuromeres, defining their respective floor, basal, alar and roof domains plus DV microzonal divisions. The known adult nuclei fell naturally into the resulting intersectional pigeon-holes, and were consistent with gene patterns analyzed. This effort using some 4,000 genes at the Allen Institute helped evolving the more complete, updated prosomeric model of Puelles et al. 2012a. d,e, Jumping now to phenomena in a single microzone from López-González et al. 2021, we show a sagittal image of the retromamillary area (RM; a basal plate area of hp1 next to the floor, understood within columnar theory as ‘supramamillary area’) where we see distinct ventral and dorsal subareas labeled differentially for Foxa1 (green) and Nr4a2 (fuchsia). Both populations generate cells that enter a rostralward migration stream that courses above the mamillary body into hp2, and enters the suprajacent ventral tuberal area. As the cells approach rostrally the acroterminal area (which may have attracted them) they stop migrating, forming an oval-shaped nucleus, the classically known ventral premamillary nucleus. There is very little cell mixing with cells derived from hp2, whose cells often express quite different gene markers. In contrast, the PMV migration is composed of intermixed green- and fuchsia-labeled RM cells, which subdivide into several intermixed subgroups according to which of the two markers they express. The final PMV nucleus has a molecularly distinguishable core and shell structure with multiple cell types.