Submitted:
02 May 2024
Posted:
06 May 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
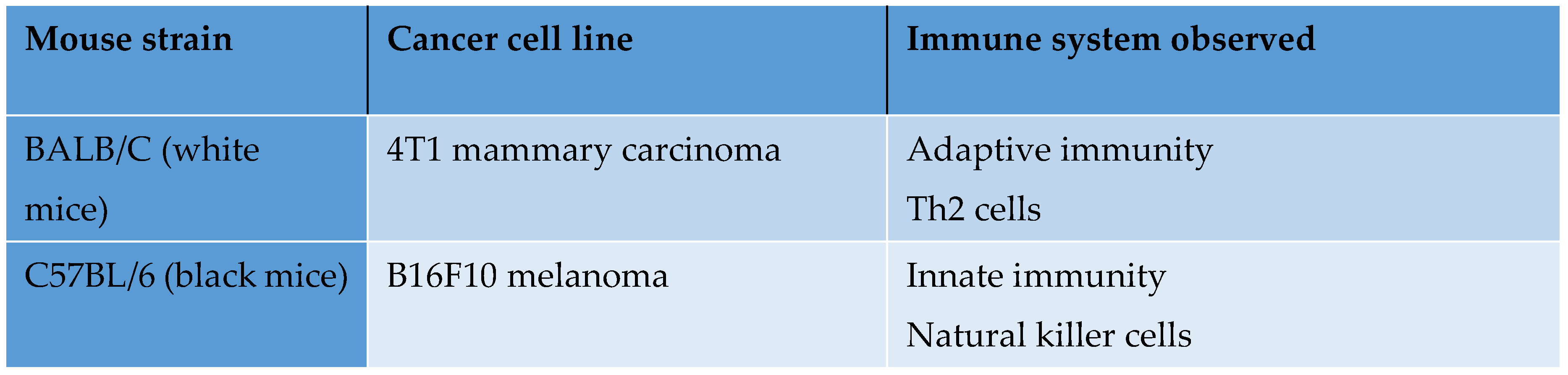
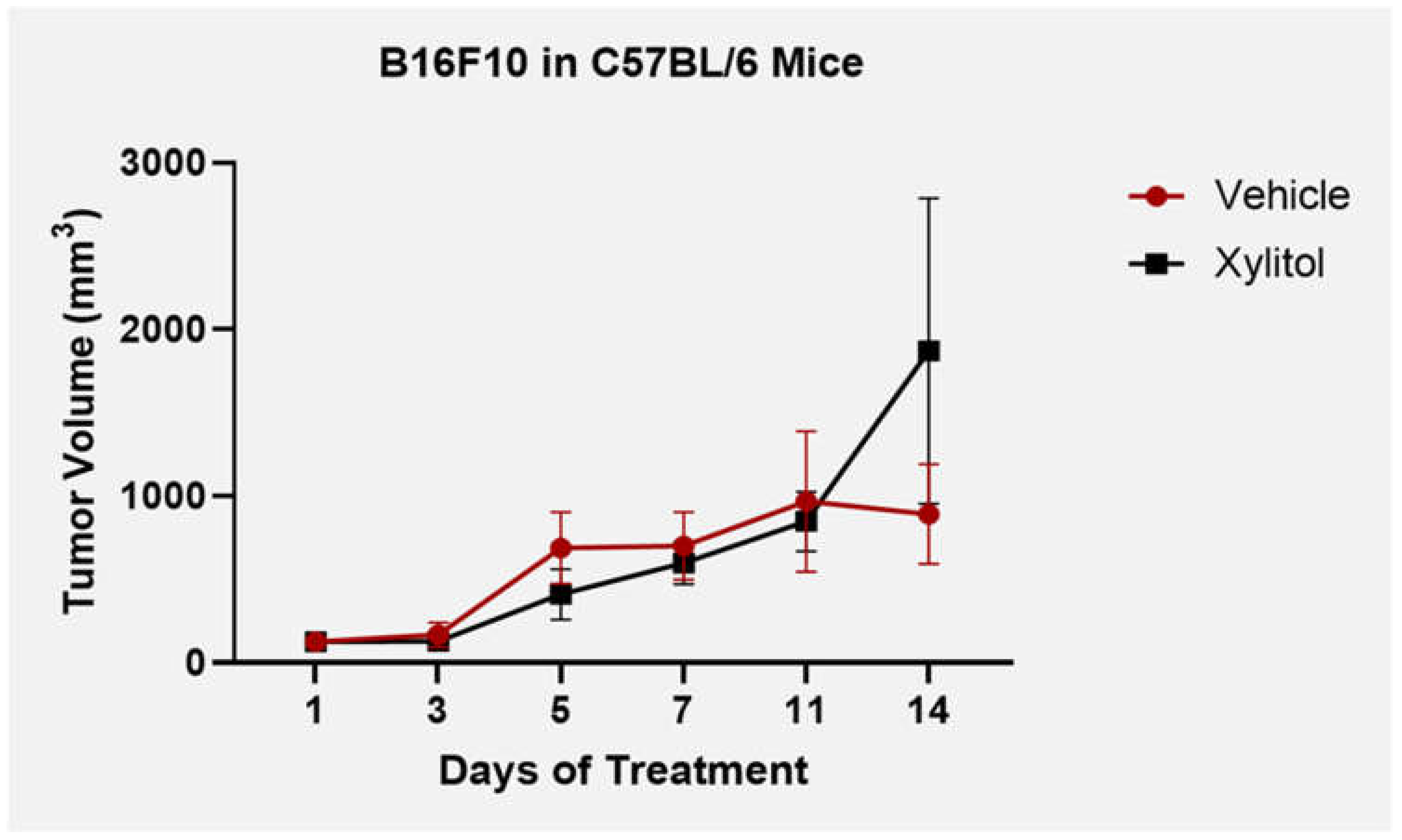
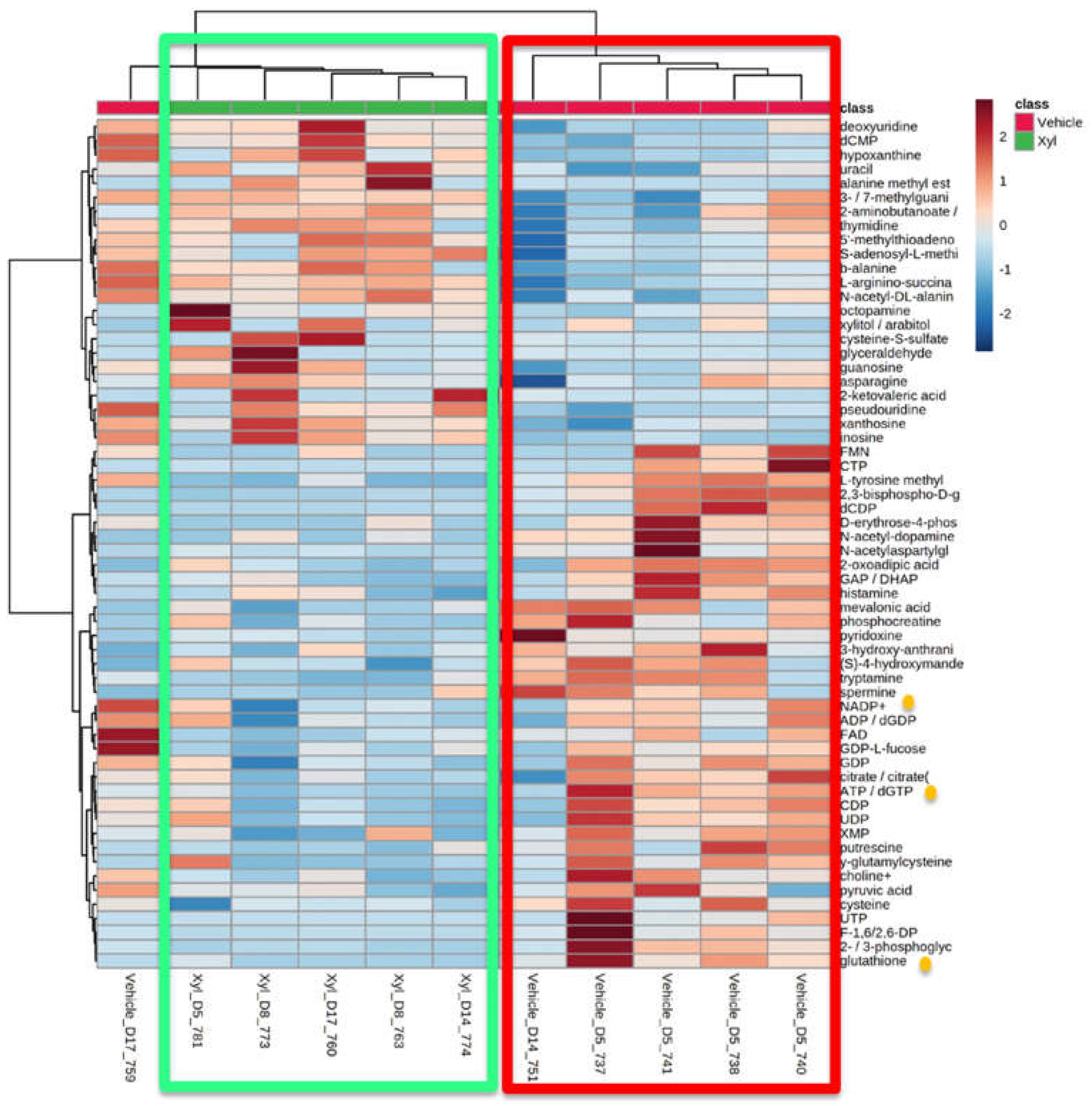
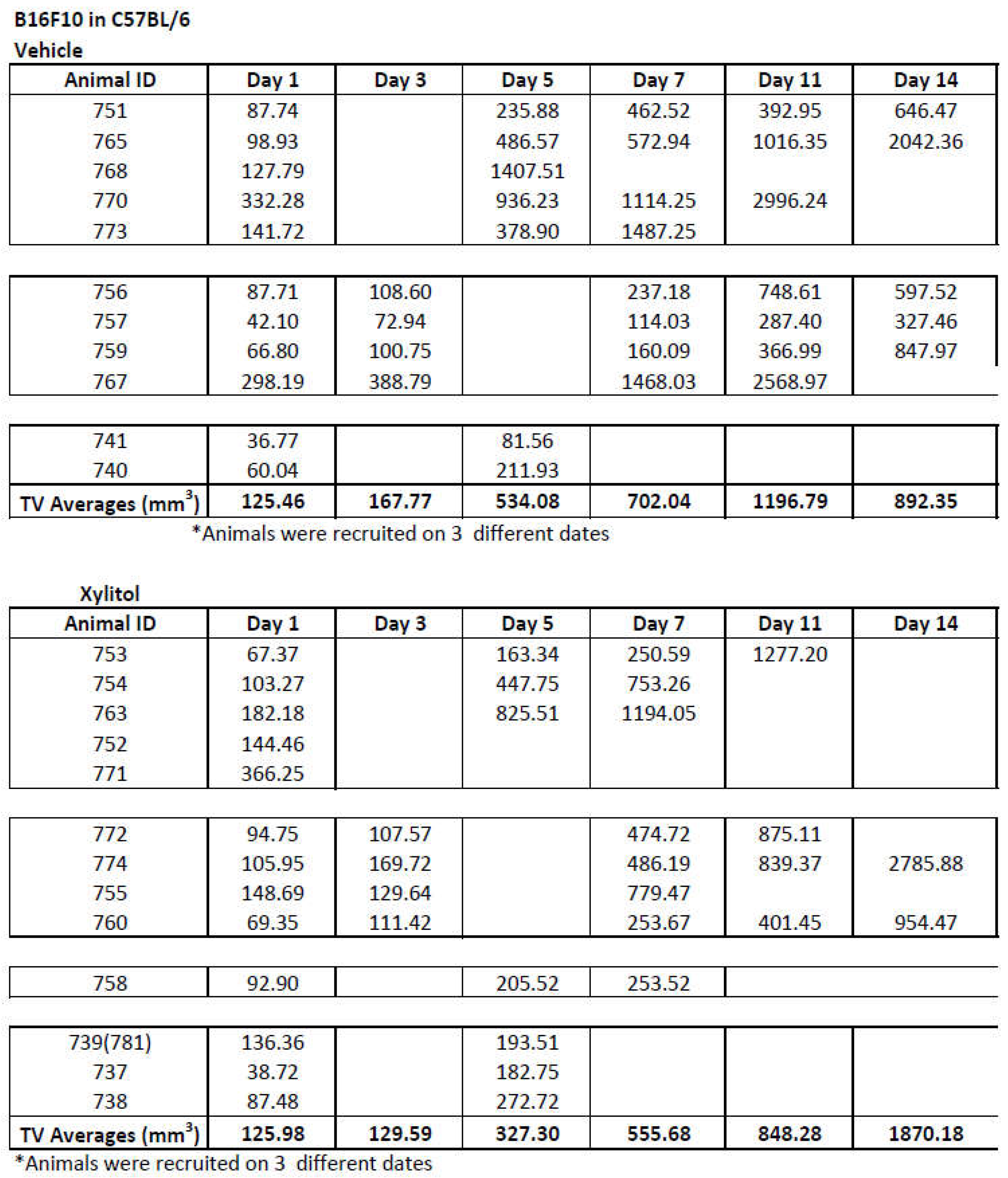
3. Results
4. Discussion
5. Conclusions
Data Availability
Ethics Statement
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nayak, P. A., Nayak, U. A., & Khandelwal, V. (2014). The effect of xylitol on dental caries and oral flora. Clinical, cosmetic and investigational dentistry, 6, 89–94. [CrossRef]
- Janakiram C., Deepan Kumar C. V., Joseph J., Xylitol in preventing dental caries: A systematic review and meta-analyses. J Nat Sci Biol Med. 2017 Jan-Jun; 8(1): 16–21.
- Kõljalg S, Smidt I, Chakrabarti A, Bosscher D, Mändar R. Exploration of singular and synergistic effect of xylitol and erythritol on causative agents of dental caries. Sci Rep. 2020 Apr 14;10(1):6297. [CrossRef] [PubMed] [PubMed Central]
- Mäkinen KK (2017) Sugar alcohols and prevention of oral diseases – comments and rectifications. Oral Health Care 2. [CrossRef]
- Salli, K., Lehtinen, M. J., Tiihonen, K., & Ouwehand, A. C. (2019). Xylitol's Health Benefits beyond Dental Health: A Comprehensive Review. Nutrients, 11(8), 1813. [CrossRef]
- de Cock P. Erythritol Functional Roles in Oral-Systemic Health. Adv Dent Res. 2018 Feb;29(1):104-109. doi: 10.1177/0022034517736499. PMID: 29355425.
- Bettina K. Wölnerhanssen, Anne Christin Meyer-Gerspach, Christoph Beglinger & Md. Shahidul Islam (2020) Metabolic effects of the natural sweeteners’ xylitol and erythritol: A comprehensive review, Critical Reviews in Food Science and Nutrition, 60:12, 1986-1998, DOI: 10.1080/10408398.2019.1623757.
- Cannon ML, Merchant M, Kabat W, Catherine L, White K, Unruh B, Ramones A. In Vitro Studies of Xylitol and Erythritol Inhibition of Streptococcus Mutans and Streptococcus Sobrinus Growth and Biofilm Production. J Clin Pediatr Dent. 2020 Sep 1;44(5):307-314. [CrossRef] [PubMed]
- Sánchez M.C., Romero-Lastra P., Ribeiro-Vidal H., et al. Comparative gene expression analysis of planktonic Porphyromonas gingivalis ATCC 33277 in the presence of a growing biofilm versus planktonic cells. BMC Microbiol. 2019;19(1):58. Published 2019 Mar 12. [CrossRef]
- Badet C., Furiga A., Thébaud N. Effect of xylitol on an in vitro model of oral biofilm. Oral Health Prev Dent. 2008;6(4):337-41.
- 11. Marleen Marga Janus, Catherine Minke Charlotte Volgenant, Bernd Willem Brandt, Mark Johannes Buijs, Bart Jan Frederik Keijser, Wim Crielaard, Egija Zaura & Bastiaan Philip Krom (2017) Effect of erythritol on microbial ecology of in vitro gingivitis biofilms, Journal of Oral Microbiology, 9:1. [CrossRef]
- Söderling E., Hietala, Lenkkeri A.M. (2010) Xylitol and erythritol decrease adherence of polysaccharide-producing oral streptococci. Curr Microbiol 60: 22-29.
- S. Ferreira, Aline; F. Silva-Paes-Leme, Annelisa; R.B. Raposo, Nadia; S. da Silva, Silvio. By Passing Microbial Resistance: Xylitol Controls Microorganisms Growth by Means of Its Anti-Adherence Property. Current Pharmaceutical Biotechnology, Volume 16, Number 1, January 2015, pp. 35-42(8).
- Bahador, A., Lesan, S., & Kashi, N. (2012). Effect of xylitol on cariogenic and beneficial oral streptococci: a randomized, double-blind crossover trial. Iranian journal of microbiology, 4(2), 75–81.
- Meng, C., Bai, C., Brown, T. D., Hood, L. E., & Tian, Q. (2018). Human Gut Microbiota and Gastrointestinal Cancer. Genomics, proteomics & bioinformatics, 16(1), 33–49. [CrossRef]
- Tsai, Y. L., Lin, T. L., Chang, C. J., Wu, T. R., Lai, W. F., Lu, C. C., & Lai, H. C. (2019). Probiotics, prebiotics and amelioration of diseases. Journal of biomedical science, 26(1), 3. [CrossRef]
- Yadav, M. K., Kumari, I., Singh, B., Sharma, K. K., & Tiwari, S. K. (2022). Probiotics, prebiotics and synbiotics: Safe options for next-generation therapeutics. Applied microbiology and biotechnology, 106(2), 505–521. [CrossRef]
- Zhou, C. B., Zhou, Y. L., & Fang, J. Y. (2021). Gut Microbiota in Cancer Immune Response and Immunotherapy. Trends in cancer, 7(7), 647–660. [CrossRef]
- Ting, N. L., Lau, H. C., & Yu, J. (2022). Cancer pharmacomicrobiomics: targeting microbiota to optimise cancer therapy outcomes. Gut, 71(7), 1412–1425. [CrossRef]
- Park, E., Park, M. H., Na, H. S., & Chung, J. (2015). Xylitol induces cell death in lung cancer A549 cells by autophagy. Biotechnology letters, 37, 983-990.
- Tomonobu, N., Komalasari, N. L. G. Y., Sumardika, I. W., Jiang, F., Chen, Y., Yamamoto, K. I., ... & Sakaguchi, M. (2020). Xylitol acts as an anticancer monosaccharide to induce selective cancer death via regulation of the glutathione level. Chemico-Biological Interactions, 324, 109085.
- Sahasakul, Y., Angkhasirisap, W., Lam-Ubol, A., Aursalung, A., Sano, D., Takada, K., & Trachootham, D. (2022). Partial Substitution of Glucose with Xylitol Prolongs Survival and Suppresses Cell Proliferation and Glycolysis of Mice Bearing Orthotopic Xenograft of Oral Cancer. Nutrients, 14(10), 2023. [CrossRef]
- Mehnert, H., Förster, H., & Dehmel, K. H. (1969). The effect of intravenous administration of xylitol solutions in normal persons and in patients with liver diseases and diabetes mellitus. In International Symposium on Metabolism, Physiology, and Clinical Use of Pentoses and Pentitols: Hakone, Japan, August 27th–29th, 1967 (pp. 293-302). Springer Berlin Heidelberg.
- Sato, J., Wang, Y. M., & van Eys, J. (1981). Metabolism of xylitol and glucose in rats bearing hepatocellular carcinomas. Cancer Research, 41(8), 3192-3199.
- Yi, E. Y., & Kim, Y. J. (2013). Xylitol inhibits in vitro and in vivo angiogenesis by suppressing the NF-κB and Akt signaling pathways. International Journal of Oncology, 43(1), 315-320.
- Ylikahri, R. H., & Leino, T. (1979). Metabolic interactions of xylitol and ethanol in healthy males. Metabolism: clinical and experimental, 28(1), 25–29. [CrossRef]
- Hutchenson, R. M., Reynolds, V. H., & Touster, O. (1956). The reduction of L-xylulose to xylitol by guinea pig liver mitochondria. The Journal of biological chemistry, 221(2), 697–709.
- Ahuja, V., Macho, M., Ewe, D., Singh, M., Saha, S., & Saurav, K. (2020). Biological and Pharmacological Potential of Xylitol: A Molecular Insight of Unique Metabolism. Foods (Basel, Switzerland), 9(11), 1592. [CrossRef]
- Islam M. S. (2011). Effects of xylitol as a sugar substitute on diabetes-related parameters in nondiabetic rats. Journal of medicinal food, 14(5), 505–511. [CrossRef]
- Wada, T., Sumardika, I. W., Saito, S., Ruma, I. M. W., Kondo, E., Shibukawa, M., & Sakaguchi, M. (2017). Identification of a novel component leading to anti-tumor activity besides the major ingredient cordycepin in Cordyceps militaris extract. Journal of chromatography. B, Analytical technologies in the biomedical and life sciences, 1061-1062, 209–219. [CrossRef]
- Park, E., Na, H. S., Kim, S. M., Wallet, S., Cha, S., & Chung, J. (2014). Xylitol, an anticaries agent, exhibits potent inhibition of inflammatory responses in human THP-1-derived macrophages infected with Porphyromonas gingivalis. Journal of periodontology, 85(6), e212–e223. [CrossRef]
- Trachootham, D., Chingsuwanrote, P., Yoosadiang, P., Mekkriangkrai, D., Ratchawong, T., Buraphacheep, N., ... & Tuntipopipat, S. (2017). Partial substitution of glucose with xylitol suppressed the glycolysis and selectively inhibited the proliferation of oral cancer cells. Nutrition and cancer, 69(6), 862-872.
- Qusa, M. H., Siddique, A. B., Nazzal, S., & El Sayed, K. A. (2019). Novel olive oil phenolic (−)-oleocanthal (+)-xylitol-based solid dispersion formulations with potent oral anti-breast cancer activities. International journal of pharmaceutics, 569, 118596.
- Ireson, C. R., Alavijeh, M. S., Palmer, A. M., Fowler, E. R., & Jones, H. J. (2019). The role of mouse tumour models in the discovery and development of anticancer drugs. British journal of cancer, 121(2), 101-108.
- Rinschen, M. M., Ivanisevic, J., Giera, M., & Siuzdak, G. (2019). Identification of bioactive metabolites using activity metabolomics. Nature reviews. Molecular cell biology, 20(6), 353–367. [CrossRef]
- Vander Heiden, M. G., & DeBerardinis, R. J. (2017). Understanding the Intersections between Metabolism and Cancer Biology. Cell, 168(4), 657–669. [CrossRef]
- Vernieri, C., Casola, S., Foiani, M., Pietrantonio, F., de Braud, F., & Longo, V. (2016). Targeting Cancer Metabolism: Dietary and Pharmacologic Interventions. Cancer discovery, 6(12), 1315–1333. [CrossRef]
- Schmidt, DR, Patel, R, Kirsch, DG, Lewis, CA, Vander Heiden, MG, Locasale, JW. Metabolomics in cancer research and emerging applications in clinical oncology. CA Cancer J Clin. 2021. [CrossRef]
- Ylikahri R. (1979). Metabolic and nutritional aspects of xylitol. Advances in food research, 25, 159–180. [CrossRef]
- DeBerardinis RJ, Chandel NS. We need to talk about the Warburg effect. Nat Metab. 2020 Feb;2(2):127-129. [CrossRef] [PubMed]
- Cortinovis, C., & Caloni, F. (2016). Household Food Items Toxic to Dogs and Cats. Frontiers in veterinary science, 3, 26. [CrossRef]
- Iannotti, L. L., Gyimah, E. A., Reid, M., Chapnick, M., Cartmill, M. K., Lutter, C. K., Hilton, C., Gildner, T. E., & Quinn, E. A. (2022). Child dietary patterns in Homo sapiens evolution: A systematic review. Evolution, medicine, and public health, 10(1), 371–390. [CrossRef]
- Marlowe, F. W., & Berbesque, J. C. (2009). Tubers as fallback foods and their impact on Hadza hunter-gatherers. American journal of physical anthropology, 140(4), 751–758. [CrossRef]
- Olm, M. R., Dahan, D., Carter, M. M., Merrill, B. D., Yu, F. B., Jain, S., Meng, X., Tripathi, S., Wastyk, H., Neff, N., Holmes, S., Sonnenburg, E. D., Jha, A. R., & Sonnenburg, J. L. (2022). Robust variation in infant gut microbiome assembly across a spectrum of lifestyles. Science (New York, N.Y.), 376(6598), 1220–1223. [CrossRef]
- De Filippo, C., Cavalieri, D., Di Paola, M., Ramazzotti, M., Poullet, J. B., Massart, S., Collini, S., Pieraccini, G., & Lionetti, P. (2010). Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proceedings of the National Academy of Sciences of the United States of America, 107(33), 14691–14696. [CrossRef]
- Marean C. W. (2010). When the sea saved humanity. Scientific American, 303(2), 54–61. [CrossRef]
- Trachootham, D., Alexandre, J., & Huang, P. (2009). Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach?. Nature reviews. Drug discovery, 8(7), 579–591. [CrossRef]
- Niu, B., Liao, K., Zhou, Y., Wen, T., Quan, G., Pan, X., & Wu, C. (2021). Application of glutathione depletion in cancer therapy: Enhanced ROS-based therapy, ferroptosis, and chemotherapy. Biomaterials, 277, 121110. [CrossRef]
- Wen, Y., Chen, H., Zhang, L., Wu, M., Zhang, F., Yang, D., Shen, J., & Chen, J. (2021). Glycyrrhetinic acid induces oxidative/nitrative stress and drives ferroptosis through activating NADPH oxidases and iNOS, and depriving glutathione in triple-negative breast cancer cells. Free radical biology & medicine, 173, 41–51. [CrossRef]
- Hellstrand, K., Brune, M., Naredi, P., Mellqvist, U. H., Hansson, M., Gehlsen, K. R., & Hermodsson, S. (2000). Histamine: a novel approach to cancer immunotherapy. Cancer investigation, 18(4), 347–355. [CrossRef]
- Stoyanov, E., Uddin, M., Mankuta, D., Dubinett, S. M., & Levi-Schaffer, F. (2012). Mast cells and histamine enhance the proliferation of non-small cell lung cancer cells. Lung cancer (Amsterdam, Netherlands), 75(1), 38–44. [CrossRef]
- Chakrabarty, R. P., & Chandel, N. S. (2021). Mitochondria as Signaling Organelles Control Mammalian Stem Cell Fate. Cell stem cell, 28(3), 394–408. [CrossRef]
- Simonsen, T. G., Gaustad, J. V., Leinaas, M. N., & Rofstad, E. K. (2012). High interstitial fluid pressure is associated with tumor-line specific vascular abnormalities in human melanoma xenografts. PloS one, 7(6), e40006. [CrossRef]
- Shin, G. R., Kim, H. E., Kim, J. H., Choi, S., & Kim, M. S. (2021). Advances in Injectable In Situ-Forming Hydrogels for Intratumoral Treatment. Pharmaceutics, 13(11), 1953. [CrossRef]
- Gould, H. J., 3rd, & Paul, D. (2022). Targeted Osmotic Lysis: A Novel Approach to Targeted Cancer Therapies. Biomedicines, 10(4), 838. [CrossRef]
- Liu, Y. X., Liu, W. J., Zhang, H. R., & Zhang, Z. W. (2018). Delivery of bevacizumab by intracranial injection: assessment in glioma model. OncoTargets and therapy, 11, 2673–2683. [CrossRef]
- Algrafi, A. S., Jamal, A. A., & Ismaeel, D. M. (2023). Microbiota as a New Target in Cancer Pathogenesis and Treatment. Cureus, 15(10), e47072. [CrossRef]
- Wu, H. J., & Wu, E. (2012). The role of gut microbiota in immune homeostasis and autoimmunity. Gut microbes, 3(1), 4–14. [CrossRef]
- Halley, A., Leonetti, A., Gregori, A., Tiseo, M., Deng, D. M., Giovannetti, E., & Peters, G. J. (2020). The Role of the Microbiome in Cancer and Therapy Efficacy: Focus on Lung Cancer. Anticancer research, 40(9), 4807–4818. [CrossRef]
- Zuo, Q. L., Cai, X., Zheng, X. Y., Chen, D. S., Li, M., Liu, Z. Q., Chen, K. Q., Han, F. F., & Zhu, X. (2021). Influences of Xylitol Consumption at Different Dosages on Intestinal Tissues and Gut Microbiota in Rats. Journal of agricultural and food chemistry, 69(40), 12002–12011. [CrossRef]
- Xiang, S., Ye, K., Li, M., Ying, J., Wang, H., Han, J., Shi, L., Xiao, J., Shen, Y., Feng, X., Bao, X., Zheng, Y., Ge, Y., Zhang, Y., Liu, C., Chen, J., Chen, Y., Tian, S., & Zhu, X. (2021). Xylitol enhances synthesis of propionate in the colon via cross-feeding of gut microbiota. Microbiome, 9(1), 62. [CrossRef]
- Koppel, N., Maini Rekdal, V., & Balskus, E. P. (2017). Chemical transformation of xenobiotics by the human gut microbiota. Science (New York, N.Y.), 356(6344), eaag2770. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




