Submitted:
24 April 2024
Posted:
25 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
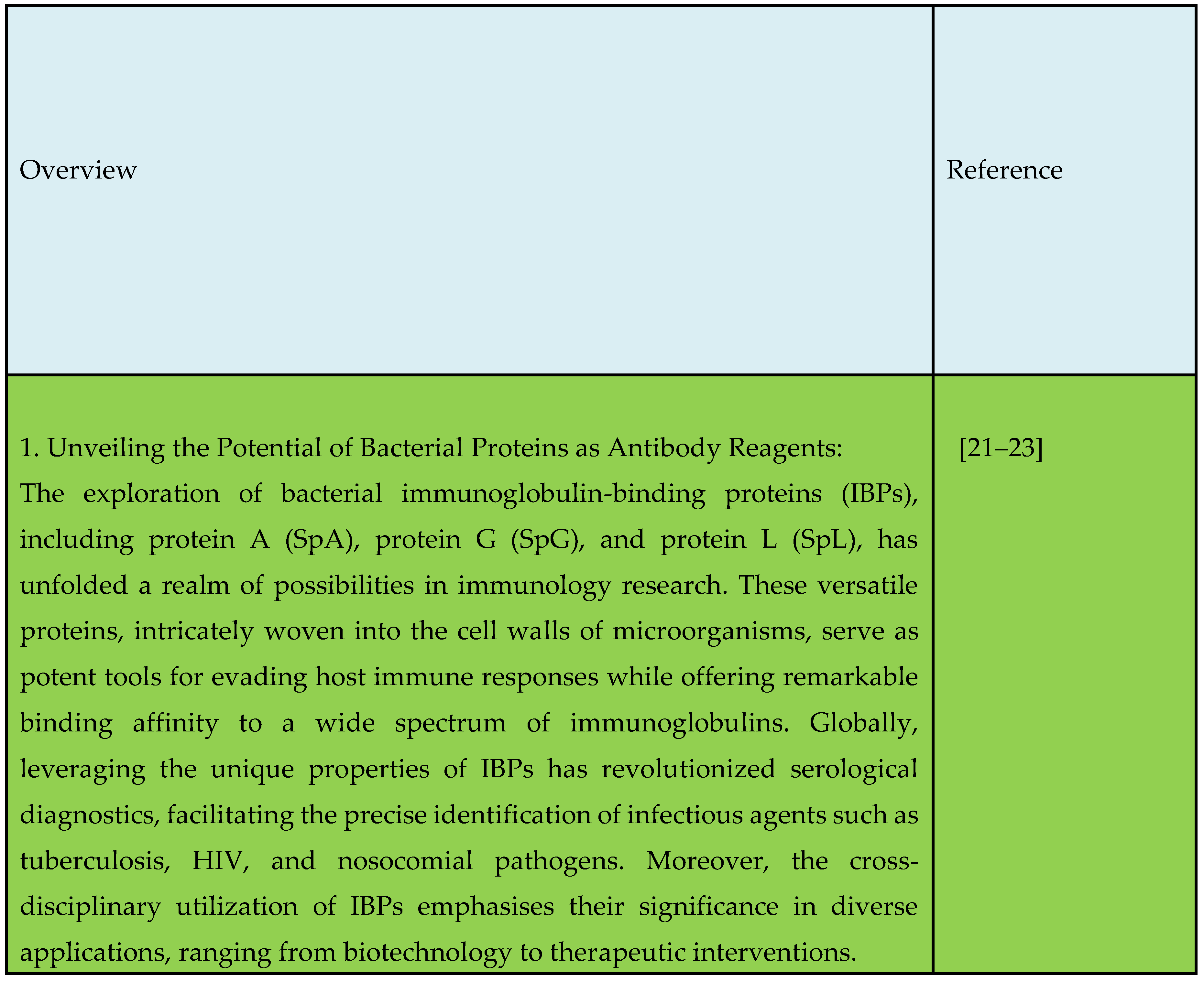
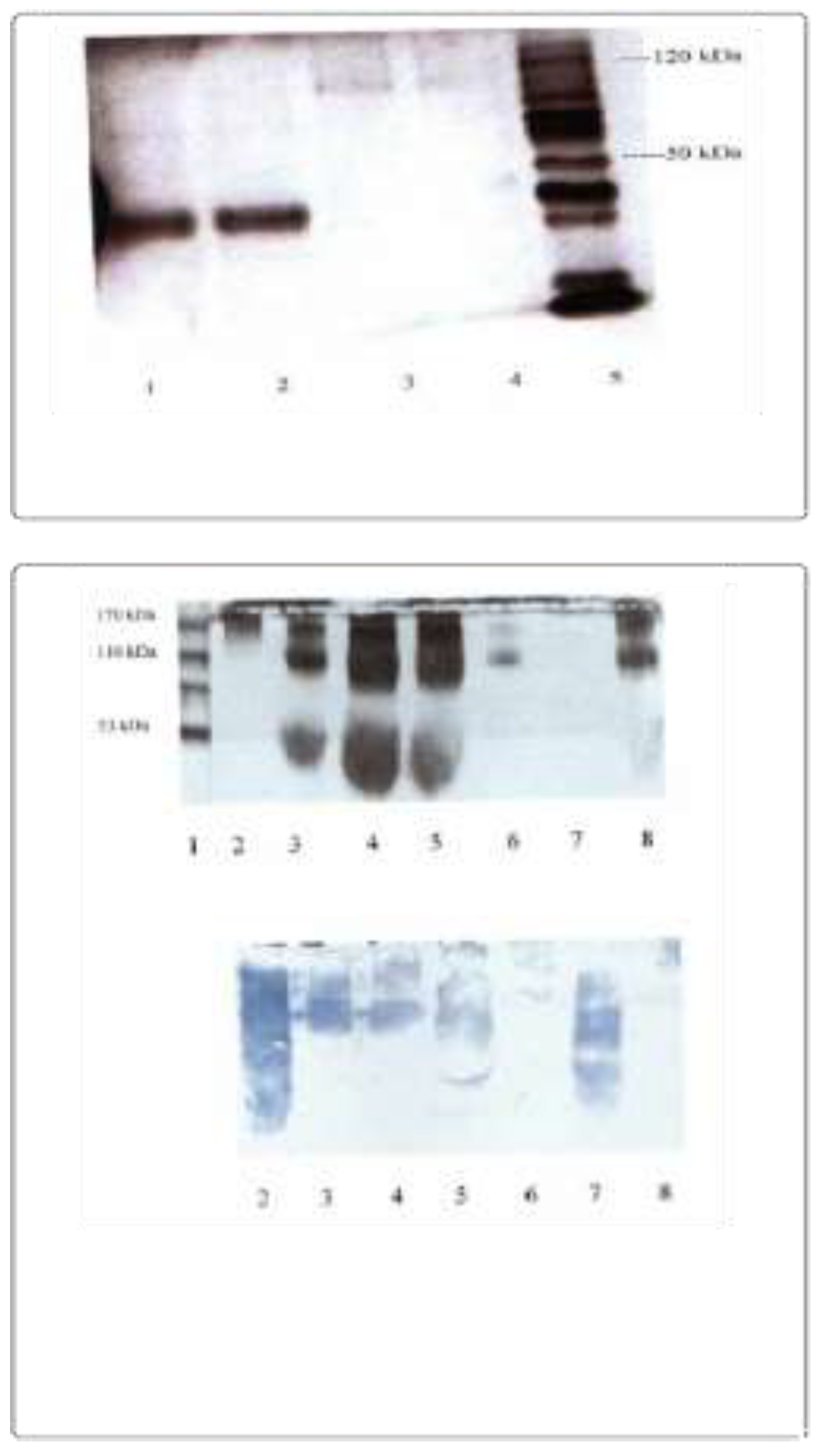
2. Unveiling the Potential of Bacterial Proteins as Antibody Reagents
3. Engineering Chimeric Proteins: Pioneering Immunodiagnosis
4. Illuminating Pathways in Vaccine Development and Clinical Studies
4.1. A Vaccine for Salmonellosis
4.2. An HIV Experimental Vaccines
5. Microbiological Insights and Antimicrobial Resistance Surveillance
5.1. Extended-Spectrum Beta-Lactamases (ESBLs) Present a Significant Public Health Challenge Globally
5.2. Nosocomial Infections In Trinidad and Tobago
6. Evolution of Immunological Techniques and Advancements in Blood Banking
8. Advancements in Cancer Research: Insights and Innovations
9. Egg Antibody Technology
10. Confronting Tuberculosis: A Global Perspectives
11. Conclusion




Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lynch, S.V.; Pedersen, O. The Human Intestinal Microbiome in Health and Disease. N. Engl. J. Med. 2016, 375, 2369–2379. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Thammavongsa, V.; Schneewind, O.; Missiakas, D. Recurrent Infections and Immune Evasion Strategies of Staphylococcus Aureus. Curr. Opin. Microbiol. 2012, 15, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Yazdani, R.; Fekrvand, S.; Shahkarami, S.; Azizi, G.; Moazzami, B.; Abolhassani, H.; Aghamohammadi, A. The Hyper IgM Syndromes: Epidemiology, Pathogenesis, Clinical Manifestations, Diagnosis and Management. Clin. Immunol. 2019, 198, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Drikic, M.; Olsen, S.; De Buck, J. Detecting Total Immunoglobulins in Diverse Animal Species with a Novel Split Enzymatic Assay. BMC Vet. Res. 2019, 15, 374. [Google Scholar] [CrossRef] [PubMed]
- de Lima Cavalcanti, T.Y.V.; Pereira, M.R.; de Paula, S.O.; Franca, R.F. de O. A Review on Chikungunya Virus Epidemiology, Pathogenesis and Current Vaccine Development. Viruses 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Milligan, R.; Paul, M.; Richardson, M.; Neuberger, A. Vaccines for Preventing Typhoid Fever. Cochrane Database Syst. Rev. 2018, 5, CD001261. [Google Scholar] [CrossRef] [PubMed]
- Kumi Smith, M.; Jewell, B.L.; Hallett, T.B.; Cohen, M.S. Treatment of HIV for the Prevention of Transmission in Discordant Couples and at the Population Level. Adv. Exp. Med. Biol. 2018, 1075, 125–162. [Google Scholar] [PubMed]
- Husna, A.; Rahman, M.M.; Badruzzaman, A.T.M.; Sikder, M.H.; Islam, M.R.; Rahman, M.T.; Alam, J.; Ashour, H.M. Extended-Spectrum β-Lactamases (ESBL): Challenges and Opportunities. Biomedicines 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Medina, E.; Pieper, D.H. Tackling Threats and Future Problems of Multidrug-Resistant Bacteria. Curr. Top. Microbiol. Immunol. 2016, 398, 3–33. [Google Scholar]
- Ferrer, E.; Lares, M.; Viettri, M.; Medina, M. [Comparison between immunological and molecular techniques for the diagnosis of Chagas disease]. Enferm. Infecc. Microbiol. Clin. 2013, 31, 277–282. [Google Scholar] [CrossRef]
- Storch, E.K.; Custer, B.S.; Jacobs, M.R.; Menitove, J.E.; Mintz, P.D. Review of Current Transfusion Therapy and Blood Banking Practices. Blood Rev. 2019, 38, 100593. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, A.S.; Każarnowicz, A.; Karaseva, N.; Sánchez, A.; De Boer, R.; Andric, Z.; Reck, M.; Atagi, S.; Lee, J.-S.; Garassino, M.; et al. Safety and Patient-Reported Outcomes of Atezolizumab, Carboplatin, and Etoposide in Extensive-Stage Small-Cell Lung Cancer (IMpower133): A Randomized Phase I/III Trial. Ann. Oncol. 2020, 31, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Gennery, A.R. Progress in Treating Chronic Granulomatous Disease. Br. J. Haematol. 2021, 192, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Emsen, A.; Uçaryılmaz, H.; Güler, T.; Artaç, H. Regulatory T and B Cells in Transient Hypogammaglobulinemia of Infancy. Turk. J. Pediatr. 2022, 64, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Nagafuchi, Y.; Fujio, K. Clinical and Immunological Biomarkers for Systemic Lupus Erythematosus. Biomolecules 2021, 11. [Google Scholar] [CrossRef]
- Chan, S.-W.B.; Zhong, Y.; Lim, S.C.J.; Poh, S.; Teh, K.L.; Soh, J.Y.; Chong, C.Y.; Thoon, K.C.; Seng, M.; Tan, E.S.; et al. Implementation of Universal Newborn Screening for Severe Combined Immunodeficiency in Singapore While Continuing Routine Bacille-Calmette-Guerin Vaccination Given at Birth. Front. Immunol. 2021, 12, 794221. [Google Scholar] [CrossRef]
- Iraqi, M.; Edri, A.; Greenshpan, Y.; Goldstein, O.; Ofir, N.; Bolel, P.; Abu Ahmad, M.; Zektser, M.; Campbell, K.S.; Rouvio, O.; et al. Blocking the PCNA/NKp44 Checkpoint to Stimulate NK Cell Responses to Multiple Myeloma. Int. J. Mol. Sci. 2022, 23. [Google Scholar] [CrossRef]
- Utsunomiya, A. Progress in Allogeneic Hematopoietic Cell Transplantation in Adult T-Cell Leukemia-Lymphoma. Front. Microbiol. 2019, 10, 2235. [Google Scholar] [CrossRef]
- Hamal, K.R.; Burgess, S.C.; Pevzner, I.Y.; Erf, G.F. Maternal Antibody Transfer from Dams to Their Egg Yolks, Egg Whites, and Chicks in Meat Lines of Chickens. Poult. Sci. 2006, 85, 1364–1372. [Google Scholar] [CrossRef]
- Bussi, C.; Gutierrez, M.G. Mycobacterium Tuberculosis Infection of Host Cells in Space and Time. FEMS Microbiol. Rev. 2019, 43, 341–361. [Google Scholar] [CrossRef]
- Forsgren, A.; Sjöquist, J. “Protein A” from S. Aureus. I. Pseudo-Immune Reaction with Human Gamma-Globulin. J. Immunol. 1966, 97, 822–827. [Google Scholar] [CrossRef] [PubMed]
- Kronvall, G. A Surface Component in Group A, C, and G Streptococci with Non-Immune Reactivity for Immunoglobulin G. J. Immunol. 1973, 111, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Björck, L. Protein L. A Novel Bacterial Cell Wall Protein with Affinity for Ig L Chains. J. Immunol. 1988, 140, 1194–1197. [Google Scholar] [CrossRef] [PubMed]
- Justiz-Vaillant, A.A.; Akpaka, P.E.; McFarlane-Anderson, N.; Smikle, M.F. Comparison of Techniques of Detecting Immunoglobulin-Binding Protein Reactivity to Immunoglobulin Produced by Different Avian and Mammalian Species. West Indian Med. J. 2013, 62, 12–20. [Google Scholar]
- De Château, M.; Nilson, B.H.; Erntell, M.; Myhre, E.; Magnusson, C.G.; Akerström, B.; Björck, L. On the Interaction between Protein L and Immunoglobulins of Various Mammalian Species. Scand. J. Immunol. 1993, 37, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Stöbel, K.; Schönberg, A.; Staak, C. A New Non-Species Dependent ELISA for Detection of Antibodies to Borrelia Burgdorferi S. L. in Zoo Animals. Int. J. Med. Microbiol. 2002, 291 Suppl 33, 88–99. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Wei, M.X.; Zhou, Z.Y.; Yu, J.Y.; Shi, X.Q. Prevalence of Antibodies to Toxoplasma Gondii in the Sera of Rare Wildlife in the Shanghai Zoological Garden, People’s Republic of China. Parasitol. Int. 2000, 49, 171–174. [Google Scholar] [CrossRef]
- Genovese, A.; Bouvet, J.P.; Florio, G.; Lamparter-Schummert, B.; Björck, L.; Marone, G. Bacterial Immunoglobulin Superantigen Proteins A and L Activate Human Heart Mast Cells by Interacting with Immunoglobulin E. Infect. Immun. 2000, 68, 5517–5524. [Google Scholar] [CrossRef] [PubMed]
- Kozlowski, L.M.; Soulika, A.M.; Silverman, G.J.; Lambris, J.D.; Levinson, A.I. Complement Activation by a B Cell Superantigen. J. Immunol. 1996, 157, 1200–1206. [Google Scholar] [CrossRef]
- A Justiz-Vaillant, A. Direct ELISA for Investigating the Binding of Chemically-Made Protein-LAG to Immunoglobulins. v1. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Direct ELISA for Investigating the Binding of Chemically-Made Protein-LAG-Anti-IgY-Peroxidase to Both Avian and Mammalian Immunoglobulins. v1. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Direct ELISA for Investigating the Binding of Peroxidase-Labeled Anti-Chicken IgY Conjugate with Avian Immunoglobulins v1. protocols.io 2020.
- A Justiz-Vaillant, A. Direct ELISA for Investigating the Binding of Recombinant or Chemically-Made Protein-AG to Immunoglobulins. v1. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Direct ELISA for Investigating the Binding of Recombinant or Chemically-Made Protein-LG to Immunoglobulins. v1. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; F. Smikle, M. Direct ELISA for Investigating the Binding of Recombinant Protein-LA to Immunoglobulins. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Universal Sandwich ELISA for Investigating the Binding of Avian and Mammalian Immunoglobulins to Streptococcal Protein-G (SpG) Using a Peroxidase-Labeled Protein LAG Conjugate (SpLAG-HRP). v1. protocols.io 2020.
- A Justiz-Vaillant, A. Universal Immunoblot Analysis for Investigating Protein-AG (SpAG)-Binding to Avian and Mammalian Immunoglobulins. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Universal Immunoblot Analysis for Investigating Protein-LAG (SpLAG)-Binding to Mammalian and Avian Immunoglobulins. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Immunoblot Analyses for Investigating SpLA Binding to Purified Mammalian and Avian Immunoglobulins. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Universal Sandwich ELISA for Investigating the Binding of Protein-LAG (SpLAG) to Avian Immunoglobulins Using Anti-IgY-Peroxidase as Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; F. Smikle, M. Sandwich ELISA for Investigating the Binding of Protein-LG (SpLG) to Avian Immunoglobulins Using Anti-IgY-Peroxidase as Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; F. Smikle, M. Universal Sandwich ELISA for Investigating the Binding of Protein-LA (SpLA) to Avian Immunoglobulins Using a Peroxidase-Labeled -Anti-IgY Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; F. Smikle, M. Universal Sandwich ELISA for Investigating the Binding of Protein-AG (SpAG) to Avian Immunoglobulins Using a Peroxidase-Labeled -Anti-IgY Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Universal Sandwich Enzyme Linked Immunosorbent Assay for Investigating Protein-LG (SpLG) Interactions with Immunoglobulins Using a SpA-HRP Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Universal Sandwich Enzyme Linked Immunosorbent Assay for Investigating Protein-LA (SpLA) Interactions with Immunoglobulins Using a SpG-HRP Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Universal Sandwich Enzyme Linked Immunosorbent Assay for Investigating Protein-AG (SpAG) Interactions with Immunoglobulins Using a SpL-HRP Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Enzyme Linked Immunosorbent Assay for Investigating the Binding of Protein-AG (SpAG) to Immunoglobulins. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Universal Sandwich Enzyme Linked Immunosorbent Assay for Investigating Staphylococcal Protein-A (SpA) Interactions with Immunoglobulins Using a SpLG-HRP Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Universal Sandwich Enzyme Linked Immunosorbent Assay for Investigating Streptococcal Protein-G (SpG) Interactions with Immunoglobulins Using a SpLA-HRP Conjugate. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Universal Sandwich Enzyme Linked Immunosorbent Assay for Investigating Immunoglobulin-Binding Protein (IBP) Interactions Using a Conjugate SpAG-HRP. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Enzyme Linked Immunosorbent Assay for Investigating the Binding of Chemically Prepared Protein-LAG-Anti-IgY (SpLAG-Anti-IgY) to Avian and Mammalian Immunoglobulins. v1. protocols.io 2020.
- (9) (PDF) Enzyme Linked Immunosorbent Assay for Investigating the Binding of Chemically Prepared Protein-LAG (SpLAG) to Immunoglobulins. v1 Available online: https://www.researchgate.net/publication/363669178_Enzyme_linked_immunosorbent_assay_for_investigating_the_binding_of_chemically_prepared_protein-LAG_SpLAG_to_immunoglobulins_v1/fulltext/636003f512cbac6a3e11828a/Enzyme-linked-immunosorbent-assay-for-investigating-the-binding-of-chemically-prepared-protein-LAG-SpLAG-to-immunoglobulins-v1.pdf (accessed on 14 April 2024).
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Enzyme Linked Immunosorbent Assay for Investigating the Binding of Protein-LG (SpLG) to Immunoglobulins. v1. protocols.io 2020.
- A Justiz-Vaillant, A.; McFarlane-Anderson, N. Enzyme Linked Immunosorbent Assay for Investigating the Binding of Protein-LA (SpLA) to Immunoglobulins. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Preparation of Horseradish Peroxidase (HRP) Conjugated to Chicken Anti-IgY v1. protocols.io 2020.
- A Justiz-Vaillant, A. Protocol of Preparation of a Protein-LAG Conjugated to Horseradish Peroxidase by the Periodate Method. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Preparation of a Protein-AG Conjugated to Horseradish Peroxidase by the Periodate Method. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Preparation of a Protein-LG Conjugated to Horseradish Peroxidase by the Periodate Method. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Preparation of a Protein-LA Conjugated to Horseradish Peroxidase by the Periodate Method. v1. protocols.io 2020.
- A Justiz-Vaillant, A. Preparation of Horseradish Peroxidase (HRP) Conjugated Streptococcal Protein-G by the Periodate Method. v1. protocols.io 2020.
- Justiz-Vaillant, A.A.; Ferrer-Cosme, B.; Curtello, S. Universal Enzyme-Linked Immunosorbent Assays (ELISA) and Utility in the Detection of Antibodies against Salmonella Spp. in Several Animal Species. bioRxiv, 3347. [Google Scholar] [CrossRef]
- Curtello, S.; Vaillant, A.A.J.; Asemota, H.; Smikle, M.P.; Akpaka, P.E. A DNA Vaccine versus Attenuated Vaccine to Protect against Salmonella Infection in Chickens. JAMMR 2014, 828–842. [Google Scholar] [CrossRef] [PubMed]
- Justiz Vaillant, A.A.; Grella, M.J. Vaccine (Vaccination); StatPearls Publishing, 2023;
- Vega, C.G.; Bok, M.; Vlasova, A.N.; Chattha, K.S.; Fernández, F.M.; Wigdorovitz, A.; Parreño, V.G.; Saif, L.J. IgY Antibodies Protect against Human Rotavirus Induced Diarrhea in the Neonatal Gnotobiotic Piglet Disease Model. PLoS One 2012, 7, e42788. [Google Scholar] [CrossRef] [PubMed]
- Justiz Vaillant, A.; Cosme, B.; Smikle, MF.; Pérez, O. Eggs from Hens Immunized with Specific KLH-Conjugated HIV Peptide Candidate Vaccines to Chicks Induces Specific Anti-HIV gp120 and gp41 Antibodies That Neutralize the Original HIV Antigens. Vaccine Res. 2020, 7, 92–96. [Google Scholar]
- Justiz Vaillant, A.A.; Gulick, P.G. HIV and AIDS Syndrome. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2022. [Google Scholar]
- Justiz Vaillant, A. A protocol and detailed methodological study on immunogenicity of various experimental vaccines. 2021, 7, 0–0.
- Justiz Vaillant, A.; Akpaka, P.E. Immunogenicity Studies of Various Experimental Vaccines in Chickens. bioRxiv 2020, 2020.08.31.276154. [CrossRef]
- Paterson David, L.; Bonomo Robert, A. Extended-Spectrum β-Lactamases: A Clinical Update. Clin. Microbiol. Rev. 2005, 18, 657–686. [Google Scholar] [CrossRef] [PubMed]
- Bush, K.; Courvalin, P.; Dantas, G.; Davies, J.; Eisenstein, B.; Huovinen, P.; Jacoby, G.A.; Kishony, R.; Kreiswirth, B.N.; Kutter, E.; et al. Tackling Antibiotic Resistance. Nat. Rev. Microbiol. 2011, 9, 894–896. [Google Scholar] [CrossRef]
- Paterson, D.L.; Bonomo, R.A. Extended-Spectrum Beta-Lactamases: A Clinical Update. Clin. Microbiol. Rev. 2005, 18, 657–686. [Google Scholar] [CrossRef]
- Pitout, J.D.D.; Laupland, K.B. Extended-Spectrum Beta-Lactamase-Producing Enterobacteriaceae: An Emerging Public-Health Concern. Lancet Infect. Dis. 2008, 8, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Baño, J.; Alcalá, J.C.; Cisneros, J.M.; Grill, F.; Oliver, A.; Horcajada, J.P.; Tórtola, T.; Mirelis, B.; Navarro, G.; Cuenca, M.; et al. Community Infections Caused by Extended-Spectrum Beta-Lactamase-Producing Escherichia Coli. Arch. Intern. Med. 2008, 168, 1897–1902. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, P.; Carrer, A. [Carbapenemases in enterobacteriaceae]. Arch. Pediatr. 2010, 17 Suppl 4, S154–S162. [Google Scholar] [CrossRef]
- World Health Organization Antimicrobial Resistance in the WHO African Region: Biannual Report 2020; 2020.
- Sulis, G.; Sayood, S.; Gandra, S. Antimicrobial Resistance in Low- and Middle-Income Countries: Current Status and Future Directions. Expert Rev. Anti. Infect. Ther. 2022, 20, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Rupp, M.E.; Fey, P.D. Extended Spectrum Beta-Lactamase (ESBL)-Producing Enterobacteriaceae: Considerations for Diagnosis, Prevention and Drug Treatment. Drugs 2003, 63, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Cassini, A.; Högberg, L.D.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.S.; Colomb-Cotinat, M.; Kretzschmar, M.E.; Devleesschauwer, B.; Cecchini, M.; et al. Attributable Deaths and Disability-Adjusted Life-Years Caused by Infections with Antibiotic-Resistant Bacteria in the EU and the European Economic Area in 2015: A Population-Level Modelling Analysis. Lancet Infect. Dis. 2019, 19, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Global Action Plan on Antimicrobial Resistance. Microbe Wash. DC 2015, 10, 354–355.
- Akpaka, P.E.; Vaillant, A.; Wilson, C.; Jayaratne, P. Extended Spectrum Beta-Lactamase (ESBL) Produced by Gram-Negative Bacteria in Trinidad and Tobago. Int. J. Microbiol. 2021, 2021, 5582755. [Google Scholar] [CrossRef] [PubMed]
- Elliott, C.; Justiz-Vaillant, A. Nosocomial Infections: A 360-Degree Review. International Biological and Biomedical Journal 2018, 4, 0–0. [Google Scholar]
- Justiz Vaillant, A.; Mcfarlane-Anderson, N. Detection of Red Blood Cell Antibodies Using Bacterial Antiglobulin: A New Assay. West Indian Med. J. 2001, 50 (Suppl. 5). P-16.
- Charles, K.S.; Chisholm, K.; Gabourel, K.; Philip, K.; Ramdath, S.; Abdul-Hakeem, H.; Vaillant, A.; Pooransingh, S.; Legall, G.; Chantry, A. A Follow-up Survey of Knowledge, Attitudes and Practices Surrounding Blood Donation in Trinidad and Tobago. ISBT Sci. Ser. 2017, 12, 349–356. [Google Scholar] [CrossRef]
- Justiz-Vaillant, A.A.; Gopaul, D.; Akpaka, P.E.; Soodeen, S.; Arozarena Fundora, R. Severe Combined Immunodeficiency-Classification, Microbiology Association and Treatment. Microorganisms 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- McCusker, C.; Upton, J.; Warrington, R. Primary Immunodeficiency. Allergy Asthma Clin. Immunol. 2018, 14, 61. [Google Scholar] [CrossRef]
- Basheer, F.; Lee, E.; Liongue, C.; Ward, A.C. Zebrafish Model of Severe Combined Immunodeficiency (SCID) Due to JAK3 Mutation. Biomolecules 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Kumrah, R.; Vignesh, P.; Patra, P.; Singh, A.; Anjani, G.; Saini, P.; Sharma, M.; Kaur, A.; Rawat, A. Genetics of Severe Combined Immunodeficiency. Genes Dis 2020, 7, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Justiz-Vaillant, A.A.; Hoyte, T.; Davis, N.; Deonarinesingh, C.; De Silva, A.; Dhanpaul, D.; Dookhoo, C.; Doorpat, J.; Dopson, A.; Durgapersad, J.; et al. A Systematic Review of the Clinical Diagnosis of Transient Hypogammaglobulinemia of Infancy. Children 2023, 10. [Google Scholar] [CrossRef]
- Justiz Vaillant, A.A.; Wilson, A.M. Transient Hypogammaglobulinemia of Infancy; StatPearls Publishing, 2023;
- Moschese, V.; Graziani, S.; Avanzini, M.A.; Carsetti, R.; Marconi, M.; La Rocca, M.; Chini, L.; Pignata, C.; Soresina, A.R.; Consolini, R.; et al. A Prospective Study on Children with Initial Diagnosis of Transient Hypogammaglobulinemia of Infancy: Results from the Italian Primary Immunodeficiency Network. Int. J. Immunopathol. Pharmacol. 2008, 21, 343–352. [Google Scholar] [CrossRef]
- Justiz-Vaillant, A.A.; Williams-Persad, A.F.-A.; Arozarena-Fundora, R.; Gopaul, D.; Soodeen, S.; Asin-Milan, O.; Thompson, R.; Unakal, C.; Akpaka, P.E. Chronic Granulomatous Disease (CGD): Commonly Associated Pathogens, Diagnosis and Treatment. Microorganisms 2023, 11. [Google Scholar] [CrossRef]
- Mortaz, E.; Azempour, E.; Mansouri, D.; Tabarsi, P.; Ghazi, M.; Koenderman, L.; Roos, D.; Adcock, I.M. Common Infections and Target Organs Associated with Chronic Granulomatous Disease in Iran. Int. Arch. Allergy Immunol. 2019, 179, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Roos, D. Chronic Granulomatous Disease. Br. Med. Bull. 2016, 118, 50–63. [Google Scholar] [CrossRef]
- Grammatikos, A.; Gennery, A.R. Inflammatory Complications in Chronic Granulomatous Disease. J. Clin. Med. Res. 2024, 13. [Google Scholar] [CrossRef]
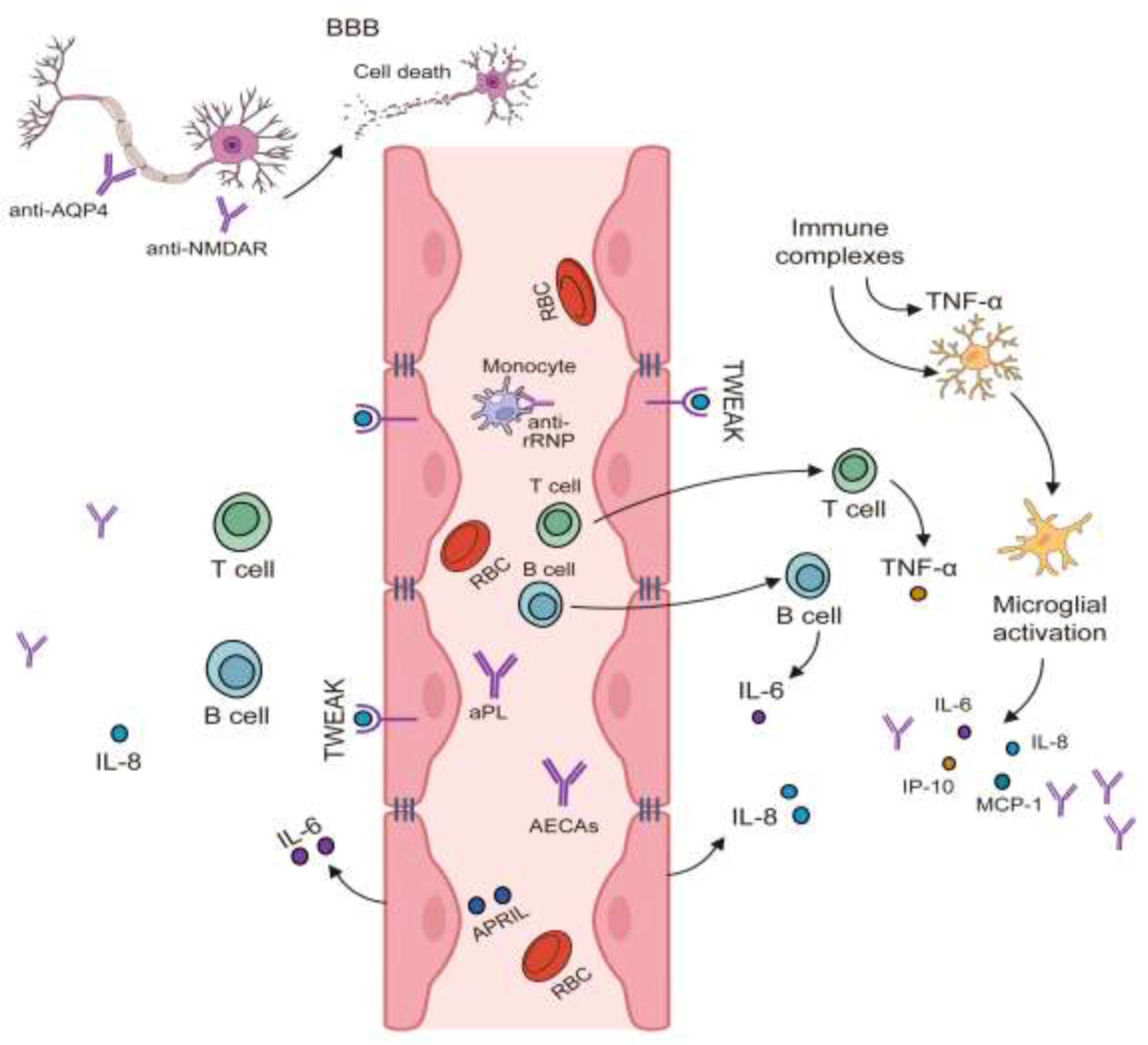
- Justiz-Vaillant, A.A.; Gopaul, D.; Soodeen, S.; Arozarena-Fundora, R.; Barbosa, O.A.; Unakal, C.; Thompson, R.; Pandit, B.; Umakanthan, S.; Akpaka, P.E. Neuropsychiatric Systemic Lupus Erythematosus: Molecules Involved in Its Imunopathogenesis, Clinical Features, and Treatment. Molecules 2024, 29. [Google Scholar] [CrossRef] [PubMed]
- Manca, E. Autoantibodies in Neuropsychiatric Systemic Lupus Erythematosus (NPSLE): Can They Be Used as Biomarkers for the Differential Diagnosis of This Disease? Clin. Rev. Allergy Immunol. 2022, 63, 194–209. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, N.; Stock, A.D.; Putterman, C. Neuropsychiatric Lupus: New Mechanistic Insights and Future Treatment Directions. Nat. Rev. Rheumatol. 2019, 15, 137–152. [Google Scholar] [CrossRef] [PubMed]
- Stock, A.D.; Wen, J.; Putterman, C. Neuropsychiatric Lupus, the Blood Brain Barrier, and the TWEAK/Fn14 Pathway. Front. Immunol. 2013, 4, 484. [Google Scholar] [CrossRef]
- Gasparotto, M.; Gatto, M.; Binda, V.; Doria, A.; Moroni, G. Lupus Nephritis: Clinical Presentations and Outcomes in the 21st Century. Rheumatology 2020, 59, v39–v51. [Google Scholar] [CrossRef]
- Shin, J.I.; Lee, K.H.; Park, S.; Yang, J.W.; Kim, H.J.; Song, K.; Lee, S.; Na, H.; Jang, Y.J.; Nam, J.Y.; et al. Systemic Lupus Erythematosus and Lung Involvement: A Comprehensive Review. J. Clin. Med. Res. 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wang, Z.; Zhang, S.; Wu, Y.; Zhang, L.; Zhao, J.; Wang, Q.; Tian, X.; Li, M.; Zeng, X. Progress in the Pathogenesis and Treatment of Neuropsychiatric Systemic Lupus Erythematosus. J. Clin. Med. Res. 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Justiz Vaillant, A.; Akpaka, P. Cytokines (IL-17, IL-23 and IL-33) in Systemic Lupus Erythematosus in Trinidad and Tobago. International Biological and Biomedical Journal 2021. [Google Scholar]
- Justiz Vaillant, A. Insights in the Management of Human T Cell-Lymphotropic Virus-1 Associated Adult-T Cell Leukaemia/lymphoma (ATL). International Biological and Biomedical Journal 2020, 6, 0–0. [Google Scholar]
- Justiz Vaillant, A. Multiple Myeloma Update. International Biological and Biomedical Journal 2018, 4, 136–141. [Google Scholar]
- Justiz-Vaillant, A.A.; Fundora, R.A.; Thompson, R.; Gopaul, D. Modifiable Cancer Risk Factors. IRJO 2023, 74–84. [Google Scholar]
- View of What Role, If Any, Should Economic Evaluation Play in Market Access Decisions of Pharmaceutical Treatments for Cancer Patients with Short Life Expectancy? Available online: https://ihrjournal.com/ihrj/article/view/421/1029 (accessed on 15 April 2024).
- Justiz-Vaillant, A.; Gardiner, L.; Mohammed, M.; Surajbally, M.; Maharaj, L.; Ramsingh, L.; Simon, M.; Seegobin, M.; Niles, M.; Vuma, S. Narrative Literature Review on Risk Factors Involved in Breast Cancer, Brain Cancer, Colon Rectal Cancer, Gynecological Malignancy, Lung Cancer, and Prostate Cancer. Preprints 2021.
- Justiz Vaillant, A. Risk factors for liver cancer worldwide. International Biological and Biomedical Journal 2021, 7, 0–0. [Google Scholar]
- Vaillant, A.J.; Ferrer-Cosme, B.; Vuma, S. Production of Antibodies in Egg Whites of Chickens. CJAST 2021, 17–22. [Google Scholar] [CrossRef]
- Justiz Vaillant, A.A.; Sabir, S.; Jan, A. Physiology, Immune Response. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2022. [Google Scholar]
- Mohammadi, M.; Zangooei, M.; Abbasi, E.; Ebrahimi Fana, S.; Aminian, M. Production of Anti-Tetanus Toxin IgY and Study of Its Protective Effects in a Mouse Model. J. Immunoassay Immunochem. 2023, 44, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Bentes, G.A.; Lanzarini, N.M.; Guimarães, J.R.; Heinemann, M.B.; Volotão, E. de M.; da Silva, A.D.S.; Heneine, L.G.D.; de Oliveira, J.M.; Pinto, M.A. Production and Evaluation of Chicken Egg Yolk Immunoglobulin (IgY) against Human and Simian Rotaviruses. Viruses 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Vega, C.; Bok, M.; Chacana, P.; Saif, L.; Fernandez, F.; Parreño, V. Egg Yolk IgY: Protection against Rotavirus Induced Diarrhea and Modulatory Effect on the Systemic and Mucosal Antibody Responses in Newborn Calves. Vet. Immunol. Immunopathol. 2011, 142, 156–169. [Google Scholar] [CrossRef] [PubMed]
- Diraviyam, T.; Zhao, B.; Wang, Y.; Schade, R.; Michael, A.; Zhang, X. Effect of Chicken Egg Yolk Antibodies (IgY) against Diarrhea in Domesticated Animals: A Systematic Review and Meta-Analysis. PLoS One 2014, 9, e97716. [Google Scholar] [CrossRef] [PubMed]
- Akpaka, P.E.; Tulloch-Reid, M.; Justiz-Vaillant, A.; Smikle, M.F. Prevalence of Human Immunodeficiency Virus Infection in Patients with Pulmonary Tuberculosis at the National Chest Hospital in Jamaica. Rev. Panam. Salud Publica 2006, 19, 38–43. [Google Scholar] [CrossRef]
- Jagielski, T.; Minias, A.; van Ingen, J.; Rastogi, N.; Brzostek, A.; Żaczek, A.; Dziadek, J. Methodological and Clinical Aspects of the Molecular Epidemiology of Mycobacterium Tuberculosis and Other Mycobacteria. Clin. Microbiol. Rev. 2016, 29, 239–290. [Google Scholar] [CrossRef]
- Chandra, P.; Grigsby, S.J.; Philips, J.A. Immune Evasion and Provocation by Mycobacterium Tuberculosis. Nat. Rev. Microbiol. 2022, 20, 750–766. [Google Scholar] [CrossRef] [PubMed]
- Parbhoo, T.; Mouton, J.M.; Sampson, S.L. Phenotypic Adaptation of Mycobacterium Tuberculosis to Host-Associated Stressors That Induce Persister Formation. Front. Cell. Infect. Microbiol. 2022, 12, 956607. [Google Scholar] [CrossRef]
- Yakin, M.; Kesav, N.; Cheng, S.K.; Caplash, S.; Gangaputra, S.; Sen, H.N. The Association between QuantiFERON-TB Gold Test and Clinical Manifestations of Uveitis in the United States. Am. J. Ophthalmol. 2021, 230, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Goossens, S.N.; Sampson, S.L.; Van Rie, A. Mechanisms of Drug-Induced Tolerance in Mycobacterium Tuberculosis. Clin. Microbiol. Rev. 2020, 34. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Wu, X. Differential Diagnosis of Latent Tuberculosis Infection and Active Tuberculosis: A Key to a Successful Tuberculosis Control Strategy. Front. Microbiol. 2021, 12, 745592. [Google Scholar] [CrossRef] [PubMed]
- Herrera, M.T.; Guzmán-Beltrán, S.; Bobadilla, K.; Santos-Mendoza, T.; Flores-Valdez, M.A.; Gutiérrez-González, L.H.; González, Y. Human Pulmonary Tuberculosis: Understanding the Immune Response in the Bronchoalveolar System. Biomolecules 2022, 12. [Google Scholar] [CrossRef]
- Justiz Vaillant, A.A.; Qurie, A. Interleukin; StatPearls Publishing, 2022.
- Baboolal, S.; Millet, J.; Akpaka, P.E.; Ramoutar, D.; Rastogi, N. First Insight into Mycobacterium Tuberculosis Epidemiology and Genetic Diversity in Trinidad and Tobago. J. Clin. Microbiol. 2009, 47, 1911–1914. [Google Scholar] [CrossRef] [PubMed]
- Millet, J.; Baboolal, S.; Akpaka, P.E.; Ramoutar, D.; Rastogi, N. Phylogeographical and Molecular Characterization of an Emerging Mycobacterium Tuberculosis Clone in Trinidad and Tobago. Infect. Genet. Evol. 2009, 9, 1336–1344. [Google Scholar] [CrossRef]
- Baboolal, S.; Ramoutar, D.; Akpaka, P.E. Comparison of the QuantiFERON®-TB Gold Assay and Tuberculin Skin Test to Detect Latent Tuberculosis Infection among Target Groups in Trinidad & Tobago. Rev. Panam. Salud Publica 2010, 28, 36–42. [Google Scholar]
- Montane Jaime, L.K.; Akpaka, P.E.; Vuma, S.; Justiz-Vaillant, A.A. A Healthy Patient with Positive Mantoux Test but Negative Quantiferon Gold Assay and No Evidence of Risk Factors - to Treat or Not to Treat? IDCases 2019, 18, e00658. [Google Scholar] [CrossRef]
- Millet, J.; Baboolal, S.; Streit, E.; Akpaka, P.E.; Rastogi, N. A First Assessment of Mycobacterium Tuberculosis Genetic Diversity and Drug-Resistance Patterns in Twelve Caribbean Territories. Biomed Res. Int. 2014, 2014, 718496. [Google Scholar] [CrossRef] [PubMed]
- Streit, E.; Baboolal, S.; Akpaka, P.E.; Millet, J.; Rastogi, N. Finer Characterization of Mycobacterium Tuberculosis Using Spoligotyping and 15-Loci MIRU-VNTRs Reveals Phylogeographical Specificities of Isolates Circulating in Guyana and Suriname. Infect. Genet. Evol. 2015, 30, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Soodeen, S.; Justiz-Vaillant, A.; Jalsa, N. It Is Possible Molecular Docking of Carbohydrates to a Mycobacterium Tuberculosis Molecule? Preprints 2023.
| Candidate Vaccines | XOD (SD), day 0 (Pre-immunized birds) |
XOD (SD), 45 days post-Immunized Birds |
P-value |
| HIV gp 41 (fragment 579-601) 3 birds | 0.170 (0.021) | 0.885 (0.044) |
<0.001 |
| HIV gp120 (fragment 308-331) 3 birds | 0.156 (0.015) | 0.910 (0.023) | <0.001 |
| HIV gp120 (fragment 421-438) 3 birds | 0.188 (0.01) |
0.865 (0.037) | <0.001 |
|
Candidate vaccines |
X%I (SD) of non-fed chicks with hyper-immune eggs and corn | X%I (SD) of fed chicks with hyper-immune eggs and corn (15- days post-immunization) |
P-value |
| HIV gp 41 (fragment 579-601) | 3.13 (0.37) | 18.05 (2.40) | 0.007 |
| HIV gp120 (fragment 308-331) | 2.41 (0.63) | 19.62 (3.13) | 0.009 |
| HIV gp120 (fragment 421-438) | 3.19 (0.51) | 13.92 (3.96) | 0.041 |
|
Candidate vaccines |
X%I (SD) of non-fed chicks with hyper-immune eggs (given corn and water) | X%I (SD) of fed chicks with hyper-immune eggs and corn (30- days post-immunization) |
p-value |
| HIV gp 41 (fragment 579-601) 3 birds | 3.13 (0.37) | 20.08 (3.23) | 0.011 |
| HIV gp120 (fragment 308-331) 3 birds | 2.41 (0.63) | 18.55 (2.47) | 0.005 |
| HIV gp120 (fragment 421-438) 3 birds | 3.19 (0.51) | 16.79 (3.85) | 0.024 |
| Microorganisms | SCID | CGD | THI |
| Bacteria |
Staphylococcus aureus, Pseudomonas spp. Mycobacterium bovis atypical mycobacteria, Klebsiella pneumoniae Pseudomonas aeruginosa, Burkholderia, and Chryseobacterium |
S. aureus. Nocardia spp. Burkholderia spp. Serratia spp. Salmonella spp. |
Streptococcus pneumoniae, Haemophilus influenzae type b, Pseudomonas aeruginosa, S. aureus |
| Viruses |
Epstein–Barr virus, Cytomegalovirus, Adenovirus, enterovirus, Herpes simplex virus, Respiratory syncytial virus, Rotavirus, and Parainfluenza virus |
Viral infections are not primary concern. | Respiratory Syncytial Virus, Enteroviruses, Rotavirus, |
| Fungi |
Pneumocystis jirovecii, Histoplasma capsulatum, Cryptococcus neoformans Candida albicans Aspergillus spp. Acremonium and Pichia |
Asperegillus spp. Candida spp |
Candida spp. |
| Parasites |
Giardia duodenalis Giradia intestinalis Cryptosporidium spp. Schistosoma spp, Blastocystis hominis, Fasciola spp, Trichostrongylus spp Cryptosporidium spp |
Parasitic infections are not a primary concern. |
Giardia lamblia |
| Reference |
[84] | [91] | [88] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




