Submitted:
22 April 2024
Posted:
24 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Site Description and Experimental Design
| Common garden | Latitude (°N) | Longitude (°E) | Mean annual temperature (℃) | Mean annual precipitation (mm) | Soil nitrogen concentration (g·kg-1) | Soil phosphorus concentration (g·kg-1) |
| Maoershan (MES) | 45.40 | 127.50 | 3.1 | 629.0 | 10.94a | 2.31a |
| Jiagedaqi (JGDQ) | 50.42 | 124.07 | −1.2 | 525.4 | 6.37b | 1.98b |
| Provenance | Code | Latitude (°N) | Longitude (°E) | Mean annual temperature (℃) | Mean annual precipitation (mm) | Mean annual evaporation (mm) | Average temperature in January (℃) | Average temperature in January July (℃) |
| Hebei (HB) | 1 | 47°33′ | 130°25′ | 1.6 | 575 | 1235 | -22.0 | 20.3 |
| Wuyiling (WYL) | 2 | 48°40′ | 129°25′ | -0.96 | 650.54 | 1038 | -24.28 | 19.06 |
| Sanzhan (SZ) | 3 | 49°37′ | 126°48′ | -1.3 | 527.2 | 1076 | -18.0 | 25.0 |
| Zhongyangzhan (ZYZ) | 4 | 50.7 | 126.72 | -2.20 | 484.4 | 1387 | −26.80 | 17.6 |
| Tahe (TH) | 5 | 52°19′ | 124°43′ | -2.74 | 487.9 | 1028 | -25.48 | 16.69 |
| Xinlin (XL) | 6 | 51°42′ | 124°20′ | -3.35 | 507.3 | 912 | -26.32 | 17.61 |
| Chuoer (CE) | 7 | 48°10′ | 121°15′ | -3.4 | 462.3 | 1122 | -24.95 | 16.19 |
| Kuduer (KDE) | 8 | 49°47′ | 121°53′ | -4.0 | 500.0 | 1280 | -27.5 | 16.2 |
| Moerdaoga (MEDG) | 9 | 51°15′ | 120°35′ | -4.5 | 471.0 | 999 | -30.0 | 17.0 |
| Genhe (GH) | 10 | 50°41′ | 121°57′ | -5.0 | 436.3 | 991 | -25.7 | 17.8 |
| Mangui (MG) | 11 | 52°03′ | 122°11′ | -5.83 | 466.0 | 1100 | -31.3 | 16.13 |
2.2. Measurements of Leaf Gas Exchange
2.3. Measurements of Leaf and Soil Traits
2.4. Data Analyses
3. Results
3.1. Comparison of Photosynthetic Characteristics
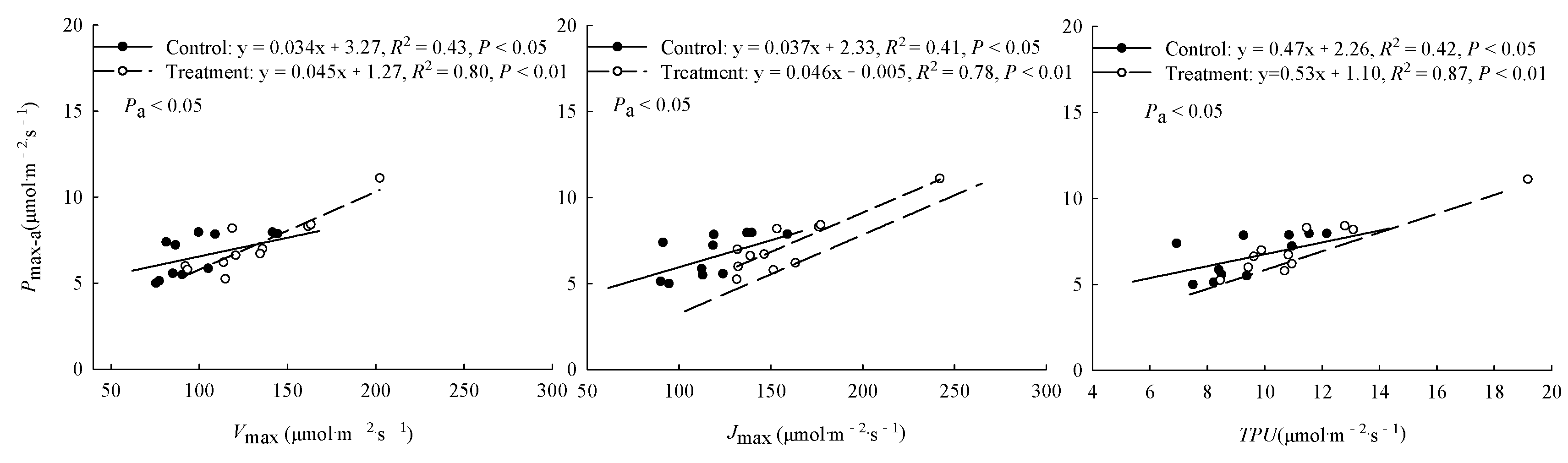
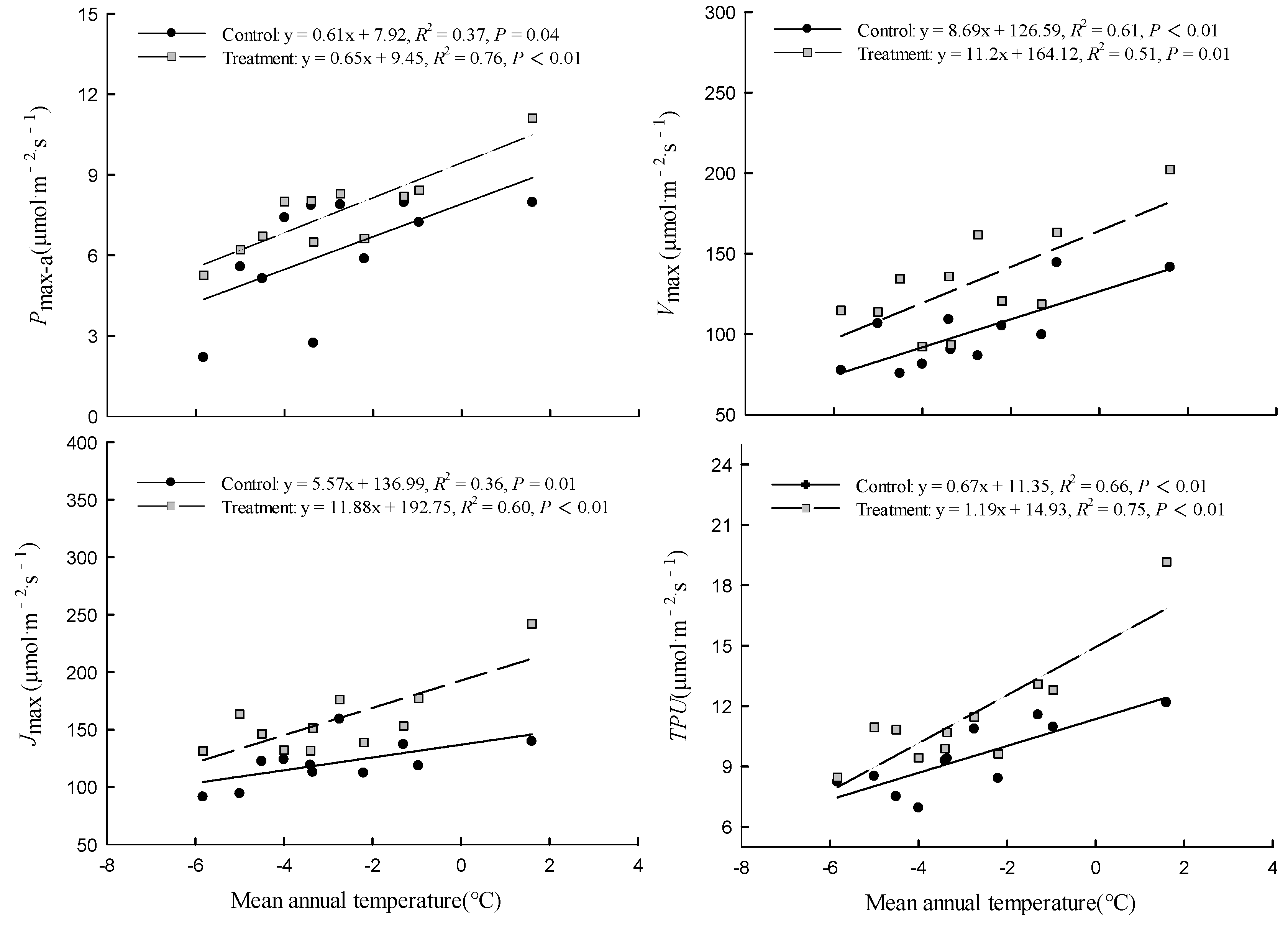
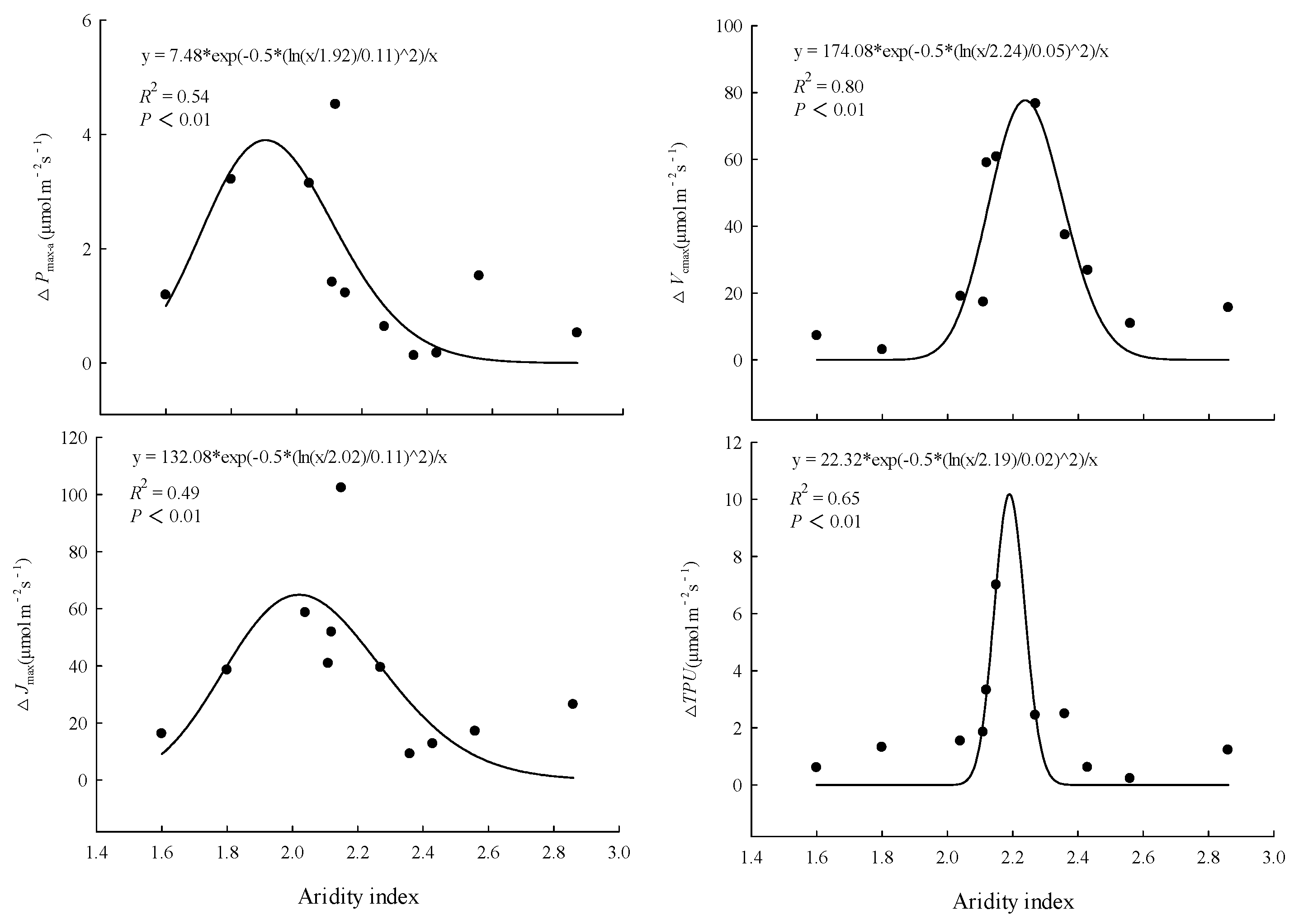
3.2. Comparison of Factors Related to Photosynthetic Capacity
4. Discussion
4.1. Effect of Warming on Photosynthetic Capacity
4.2. Differences among Provenances in the Response of Photosynthetic Capacity to Warming
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, J.Y.; Marotzke, J.; Bala, G.; Cao, L.; Corti, S.; Dunne, J.P.; Engelbrecht, F.; Fischer, E.; Fyfe, J.C.; Jones, C.; Maycock, A.; Mutemi, J.; Ndiaye, O.; Panickal, S.; Zhou, T.J. Future Global Climate: Scenario-Based Projections and Near-Term Information. In Climate Change 2021: The Physical Science Basis.; Masson-Delmotte, V., Zhai, P., Pirani, A., Connors, S.L., Péan, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M.I., Huang, M., Leitzell, K., Lonnoy, E., Matthews, J.B.R., Maycock, T.K., Waterfield, T., Yelekçi, O., Yu, R., Zhou, B.Q., Eds.; Cambridge University Press: Cambridge, United Kingdom and New York, NY, USA, 2021; pp. 553–672. [Google Scholar]
- Reich, P.B.; Sendall, K.M.; Rice, K.; Rich, R.L.; Stefanski, A.; Hobbie, S.E.; Montgomery, R.A. Geographic range predicts photosynthetic and growth response to warming in co-occurring tree species. Nat. Clim. Change 2015, 5, 148–152. [Google Scholar] [CrossRef]
- Sendall, K.M.; Reich, P.B.; Zhao, C.M.; Hou, J.H.; Wei, X.R.; Stefanski, A.; Rice, K.; Rich, R.L.; Montgomery, R.A. Acclimation of photosynthetic temperature optima of temperate and boreal tree species in response to experimental forest warming. Global Change Biol. 2015, 21, 1342–1357. [Google Scholar] [CrossRef]
- Reich, P.B.; Sendall, K.M.; Stefanski, A.; Wei, X.; Rich, R.L.; Montgomery, R.A. Boreal and temperate trees show strong acclimation of respiration to warming. Nature 2016, 531, 633–636. [Google Scholar] [CrossRef] [PubMed]
- Scafaro, A.P.; Xiang, S.; Long, B.M.; Bahar, N.H.A.; Weerasinghe, L.K.; Creek, D.; Evans, J.R.; Reich, P.B.; Atkin, O.K. Strong thermal acclimation of photosynthesis in tropical and temperate wet-forest tree species: The importance of altered Rubisco content. Global Change Biol. 2017, 23, 2783–2800. [Google Scholar] [CrossRef]
- Varhammar, A.; Wallin, G.; McLean, C.M.; Dusenge, M.E.; Medlyn, B.E.; Hasper, T.B.; Nsabimana, D.; Uddling, J. Photosynthetic temperature responses of tree species in Rwanda: Evidence of pronounced negative effects of high temperature in montane rainforest climax species. New Phytol. 2015, 206, 1000–1012. [Google Scholar] [CrossRef]
- Streit, K.; Siegwolf, R.T.W.; Hagedorn, F.; Schaub, M.; Buchmann, N. Lack of photosynthetic or stomatal regulation after 9 years of elevated CO2 and 4 years of soil warming in two conifer species at the alpine treeline. Plant Cell Environ. 2014, 37, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Way, D.A.; Sage, R.F. Elevated growth temperatures reduce the carbon gain of black spruce [Picea mariana (Mill. ) BSP]. Global Change Biol. 2008, 14, 624–636. [Google Scholar] [CrossRef]
- Aspinwall, M.J.; Drake, J.E.; Campany, C.; Vårhammar, A.; Ghannoum, O.; Tissue, D.T.; Reich, P.B.; Tjoelker, M.G. Convergent acclimation of leaf photosynthesis and respiration to prevailing ambient temperatures under current and warmer climates in Eucalyptus tereticornis. New Phytol. 2016, 212, 354–367. [Google Scholar] [CrossRef]
- Chang, H.; An, J.; Roh, Y.; Son, Y. Experimental warming and drought treatments reduce physiological activities and increase mortality of Pinus koraiensis seedlings. Plant Ecol. 2020, 221, 515–527. [Google Scholar] [CrossRef]
- Dusenge, M.E.; Madhavji, S.; Way, D.A. Contrasting acclimation responses to elevated CO2 and warming between an evergreen and a deciduous boreal conifer. Global Change Biol. 2020, 26, 3639–3657. [Google Scholar] [CrossRef]
- Togashi, H.F.; Prentice, I.C.; Atkin, O.K.; Macfarlane, C.; Prober, S.M.; Bloomfield, K.J.; Evans, B.J. Thermal acclimation of leaf photosynthetic traits in an evergreen woodland, consistent with the coordination hypothesis. Biogeosciences 2018, 15, 3461–3474. [Google Scholar] [CrossRef]
- Sage, R.F.; Kubien, D.S. The temperature response of C3 and C4 photosynthesis. Plant Cell Environ. 2007, 30, 1086–1106. [Google Scholar] [CrossRef] [PubMed]
- Gunderson, C.A.; O’Hara, K.H.; Campion, C.M.; Walker, A.V.; Edwards, N.T. Thermal plasticity of photosynthesis: The role of acclimation in forest responses to a warming climate. Global Change Biol. 2010, 16, 2272–2286. [Google Scholar] [CrossRef]
- Davis, M.B.; Shaw, R.G. Range Shifts and Adaptive Responses to Quaternary Climate Change. Science 2001, 292, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Bresson, C.C.; Vitasse, Y.; Kremer, A.; Delzon, S. To what extent is altitudinal variation of functional traits driven by genetic adaptation in European oak and beech? Tree Physiol. 2011, 31, 1164–1174. [Google Scholar] [CrossRef] [PubMed]
- Drake, J.E.; Vårhammar, A.; Kumarathunge, D.; Medlyn, B.E.; Pfautsch, S.; Reich, P.B.; Tissue, D.T.; Ghannoum, O.; Tjoelker, M.G. A common thermal niche among geographically diverse populations of the widely distributed tree species Eucalyptus tereticornis: No evidence for adaptation to climate-of-origin. Global Change Biol. 2017, 23, 5069–5082. [Google Scholar] [CrossRef]
- Benomar, L.; Lamhamedi, M.S.; Pepin, S.; Rainville, A.; lambert, M.-C.; Margolis, H.A.; Bousquet, J.; Beaulieu, J. Thermal acclimation of photosynthesis and respiration of southern and northern white spruce seed sources tested along a regional climatic gradient indicates limited potential to cope with temperature warming. Ann. Bot-London 2018, 121, 443–457. [Google Scholar] [CrossRef] [PubMed]
- Robson, T.M.; Sanchez-Gomez, D.; Javier-Cano, F.; Aranda, I. Variation in functional leaf traits among beech provenances during a Spanish summer reflects the differences in their origin. Tree Genet. Genomes 2012, 8, 1111–1121. [Google Scholar] [CrossRef]
- Aspinwall, M.J.; Vårhammar, A.; Blackman, C.J.; Tjoelker, M.G.; Ahrens, C.; Byrne, M.; Tissue, D.T.; Rymer, P.D. Adaptation and acclimation both influence photosynthetic and respiratory temperature responses in Corymbia calophylla. Tree Physiol. 2017, 37, 1095–1112. [Google Scholar] [CrossRef]
- Quan, X.K.; Wang, C.K. Acclimation and adaptation of leaf photosynthesis, respiration and phenology to climate change: A 30-year Larix gmelinii common-garden experiment. Forest Ecol. Manag. 2018, 411, 166–175. [Google Scholar]
- Tenkanen, A.; Suprun, S.; Oksanen, E.; Keinanen, M.; Keski-Saari, S.; Kontunen-Soppela, S. Strategy by latitude? Higher photosynthetic capacity and root mass fraction in northern than southern silver birch (Betula pendula Roth) in uniform growing conditions. Tree Physiology 2021, 41, 974–991. [Google Scholar] [CrossRef]
- Crous, K.Y.; Drake, J.E.; Aspinwall, M.J.; Sharwood, R.E.; Tjoelker, M.G.; Ghannoum, O. Photosynthetic capacity and leaf nitrogen decline along a controlled climate gradient in provenances of two widely distributed Eucalyptus species. Global Change Biol. 2018, 24, 4626–4644. [Google Scholar] [CrossRef]
- Ju, Y.L.; Wang, C.K.; Wang, N.; Quan, X.K. Transplanting larch trees into warmer areas increases the photosynthesis and its temperature sensitivity. Tree Physiol. 2022, 42, 2521–2533. [Google Scholar] [CrossRef]
- Sharkey, T.D.; Bernacchi, C.J.; Farquhar, G.D.; Singsaas, E.L. Fitting photosynthetic carbon dioxide response curves for C3 leaves. Plant Cell Environ. 2007, 30, 1035–1040. [Google Scholar] [CrossRef]
- Hikosaka, K. Mechanisms underlying interspecific variation in photosynthetic capacity across wild plant species. Plant Biotechnol. 2010, 27, 223–229. [Google Scholar] [CrossRef]
- Warren, C.R.; Dreyer, E.; Adams, M.A. Photosynthesis-Rubisco relationships in foliage of Pinus sylvestris in response to nitrogen supply and the proposed role of Rubisco and amino acids as nitrogen stores. Trees-Struct. Funct. 2003, 17, 359–366. [Google Scholar] [CrossRef]
- Grassi, G.; Magnani, F. Stomatal, mesophyll conductance and biochemical limitations to photosynthesis as affected by drought and leaf ontogeny in ash and oak trees. Plant Cell Environ. 2005, 28, 834–849. [Google Scholar] [CrossRef]
- Dong, N.; Prentice, I.C.; Wright, I.J.; Evans, B.J.; Togashi, H.F.; Caddy-Retalic, S.; McInerney, F.A.; Sparrow, B.; Leitch, E.; Lowe, A.J. Components of leaf-trait variation along environmental gradients. New Phytol. 2020, 228, 82–94. [Google Scholar] [CrossRef]
- Liang, X.Y.; Zhang, T.; Lu, X.K.; Ellsworth, D.S.; BassiriRad, H.; You, C.M.; Wang, D.; He, P.C.; Deng, Q.; Liu, H.; Mo, J.M.; Ye, Q. Global response patterns of plant photosynthesis to nitrogen addition: A meta-analysis. Global Change Biol. 2020, 26, 3585–3600. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.S.; Shang, B.; Feng, Z.Z.; Tarvainen, L. Effect of elevated ozone, nitrogen availability and mesophyll conductance on the temperature responses of leaf photosynthetic parameters in poplar. Tree Physiol. 2020, 40, 484–497. [Google Scholar] [CrossRef]
- Lei, Z.Y.; Liu, F.; Wright, I.J.; Carriquí, M.; Niinemets, Ü.; Han, J.M.; Jia, M.M.; Atwell, B.J.; Cai, X.Y.; Zhang, W.F.; Zhou, Z.L.; Zhang, Y.L. Comparisons of photosynthetic and anatomical traits between wild and domesticated cotton. J. Exp. Bot. 2022, 73, 873–885. [Google Scholar] [CrossRef]
- Flexas, J.; Barbour, M.M.; Brendel, O.; Cabrera, H.M.; Carriquí, M.; Díaz-Espejo, A.; Douthe, C.; Dreyerc, E.; Ferrio, J.P.; Gago, J.; Gallé, A.; Galmés, J.; Kodama, N.; Medrano, H.; Niinemets, Ü.; Peguero-Pina, J.J.; Poua, A.; Ribas-Carbó, M.; Tomás, M.; Tosens, T.; Warren, C.R. Mesophyll diffusion conductance to CO2: An unappreciated central player in photosynthesis. Plant Sci. 2012, 196, 31. [Google Scholar] [CrossRef]
- von Caemmerer, S.; Evans, J.R. Temperature responses of mesophyll conductance differ greatly between species. Plant Cell Environ. 2015, 38, 629–637. [Google Scholar] [CrossRef]
- Li, S.L.; Tan, T.T.; Fan, Y.F.; Raza, M.A.; Wang, Z.L.; Wang, B.B.; Zhang, J.W.; Tan, X.M.; Ping, C.; Shafiq, I.; Yang, W.Y.; Feng, Y. Responses of leaf stomatal and mesophyll conductance to abiotic stress factors. J. Integr. Agr. 2022, 21, 2787–2804. [Google Scholar] [CrossRef]
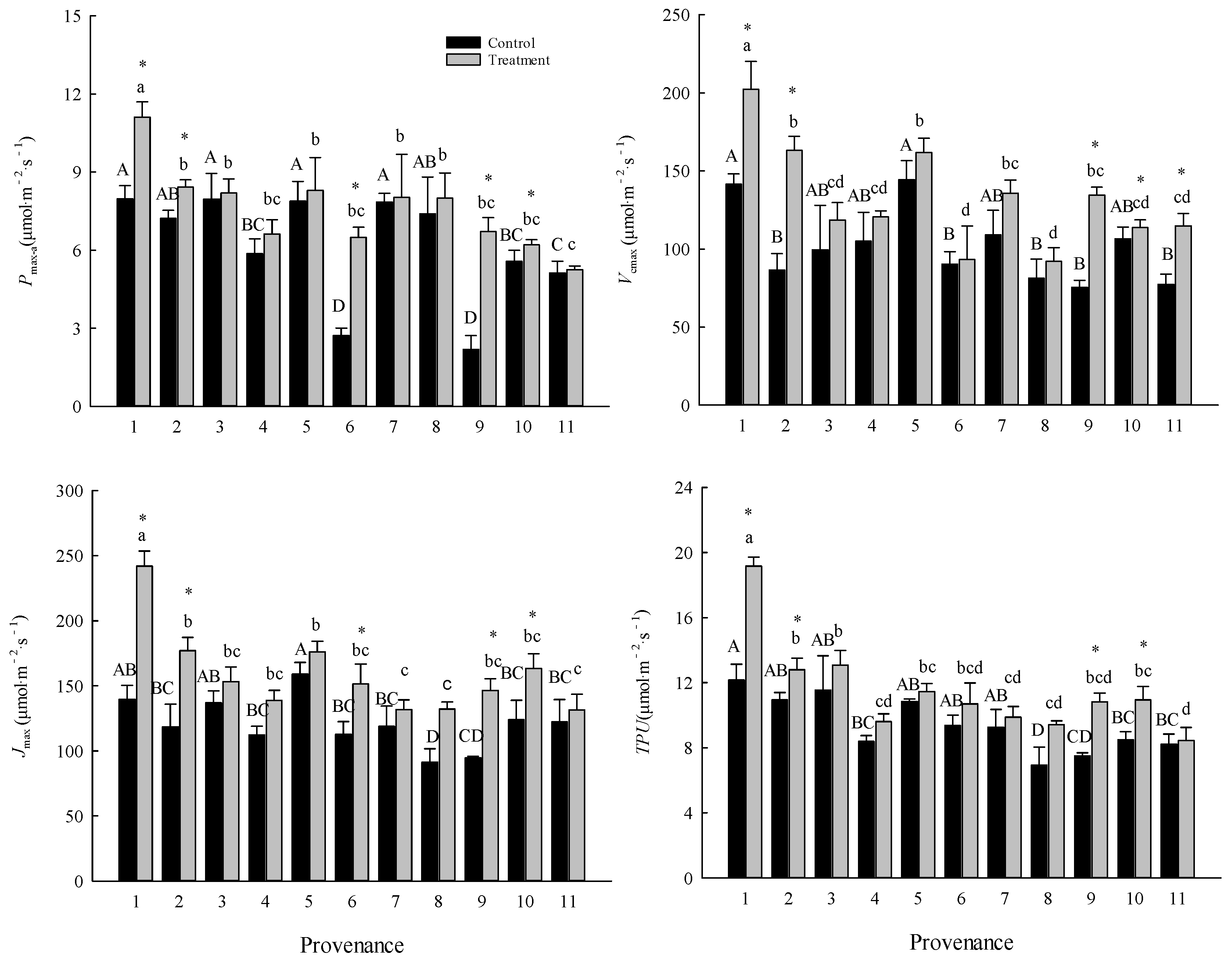
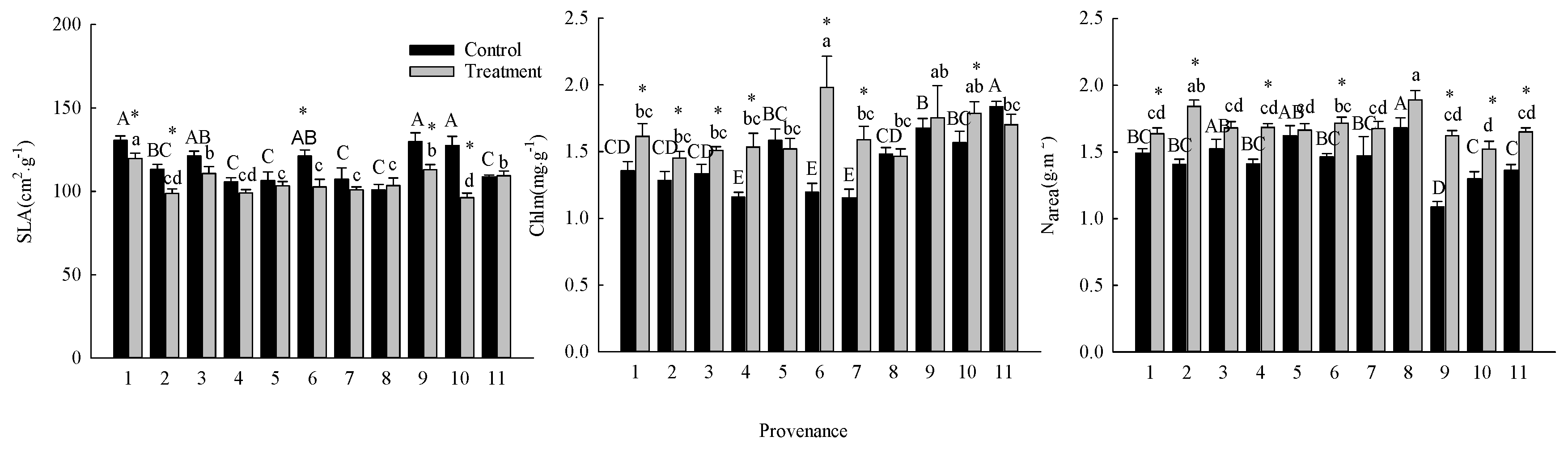
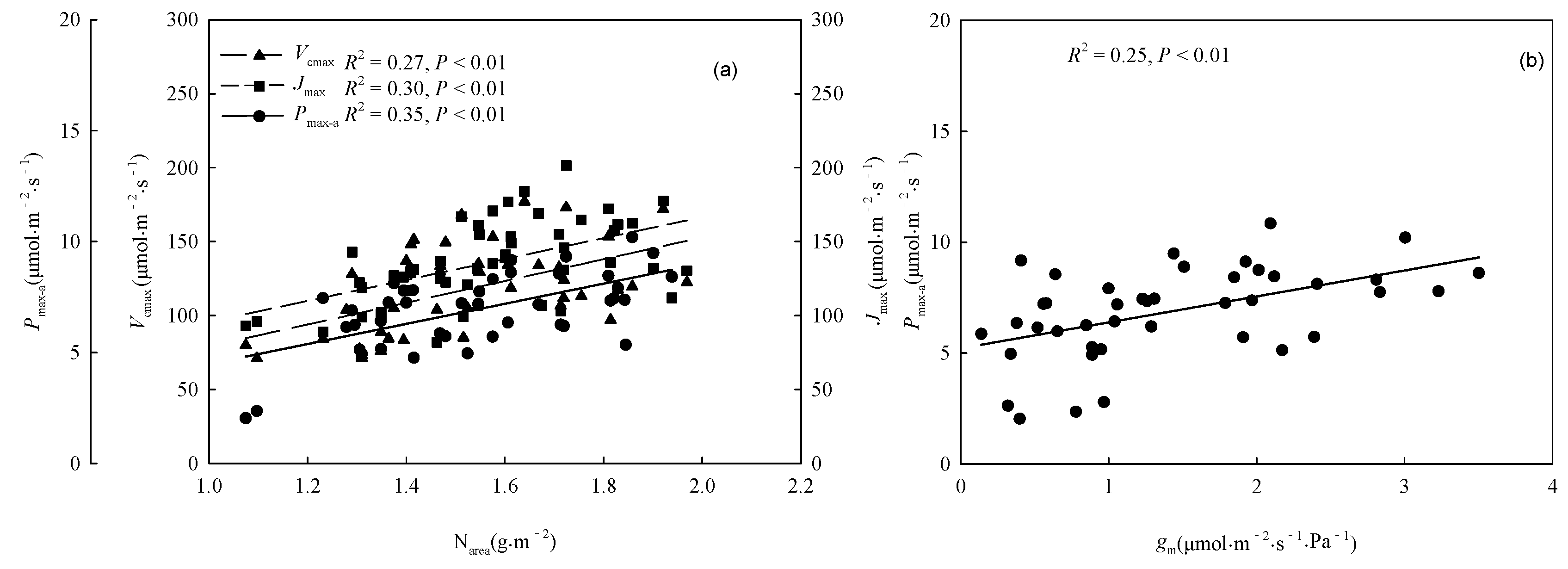
| Variable | Unit | Provenance | Treatment | Provenance × Treatment | |||
| F | P | F | P | F | P | ||
| Pmax-a | μmol m-2 s-1 | 11.76 | < 0.01 | 21.83 | < 0.01 | 2.81 | < 0.01 |
| PNUE | μmol g-1 s-1 | 12.86 | < 0.01 | 26.78 | < 0.01 | 5.00 | < 0.01 |
| Vcmax | μmol m-2 s-1 | 9.38 | < 0.01 | 35.67 | < 0.01 | 2.07 | < 0.05 |
| Jmax | μmol m-2 s-1 | 6.34 | < 0.01 | 46.15 | < 0.01 | 2.20 | < 0.05 |
| TPU | μmol m-2 s-1 | 11.36 | < 0.01 | 25.80 | < 0.01 | 2.17 | < 0.05 |
| SLA | cm2 g-1 | 7.15 | < 0.01 | 16.37 | < 0.01 | 4.59 | < 0.01 |
| Narea | g m-2 | 5.31 | < 0.01 | 71.24 | < 0.01 | 2.93 | < 0.01 |
| gm | μmol m-2 s-1 Pa-1 | 1.01 | 0.45 | 8.73 | < 0.01 | 1.61 | 0.13 |
| Chlm | mg g-1 | 4.95 | < 0.01 | 6.47 | < 0.05 | 3.51 | < 0.01 |
| Leaf traits | DG | PP |
| Pmax-a | 43.22 | 25.41 |
| PNUE | 35.54 | 27.71 |
| Vcmax | 48.08 | 42.46 |
| Jmax | 32.63 | 48.17 |
| TPU | 51.51 | 34.73 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




