Submitted:
19 April 2024
Posted:
22 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Biochemical Characterization of ASNases
2.1.1. ASNases Stability on Human Serum
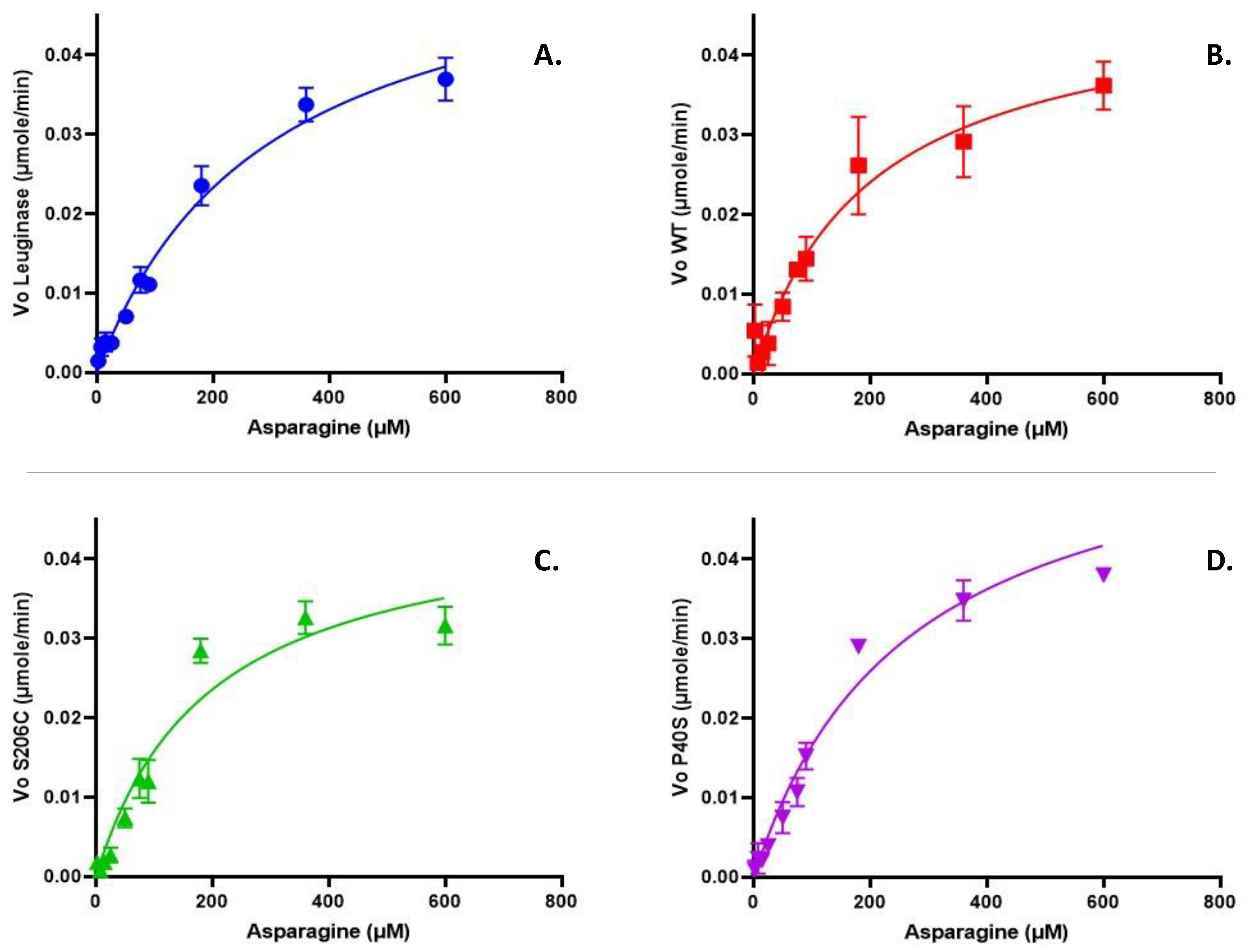
2.2. Kinetic Parameters of ASNases
2.3. Nonclinical Studies of ASNases
2.3.1. In Vitro Assays
2.3.1.1. Blood Cancer Cell Lines Cytotoxicity
2.3.1.2. Solid Tumour Cell Lines Cytotoxicity
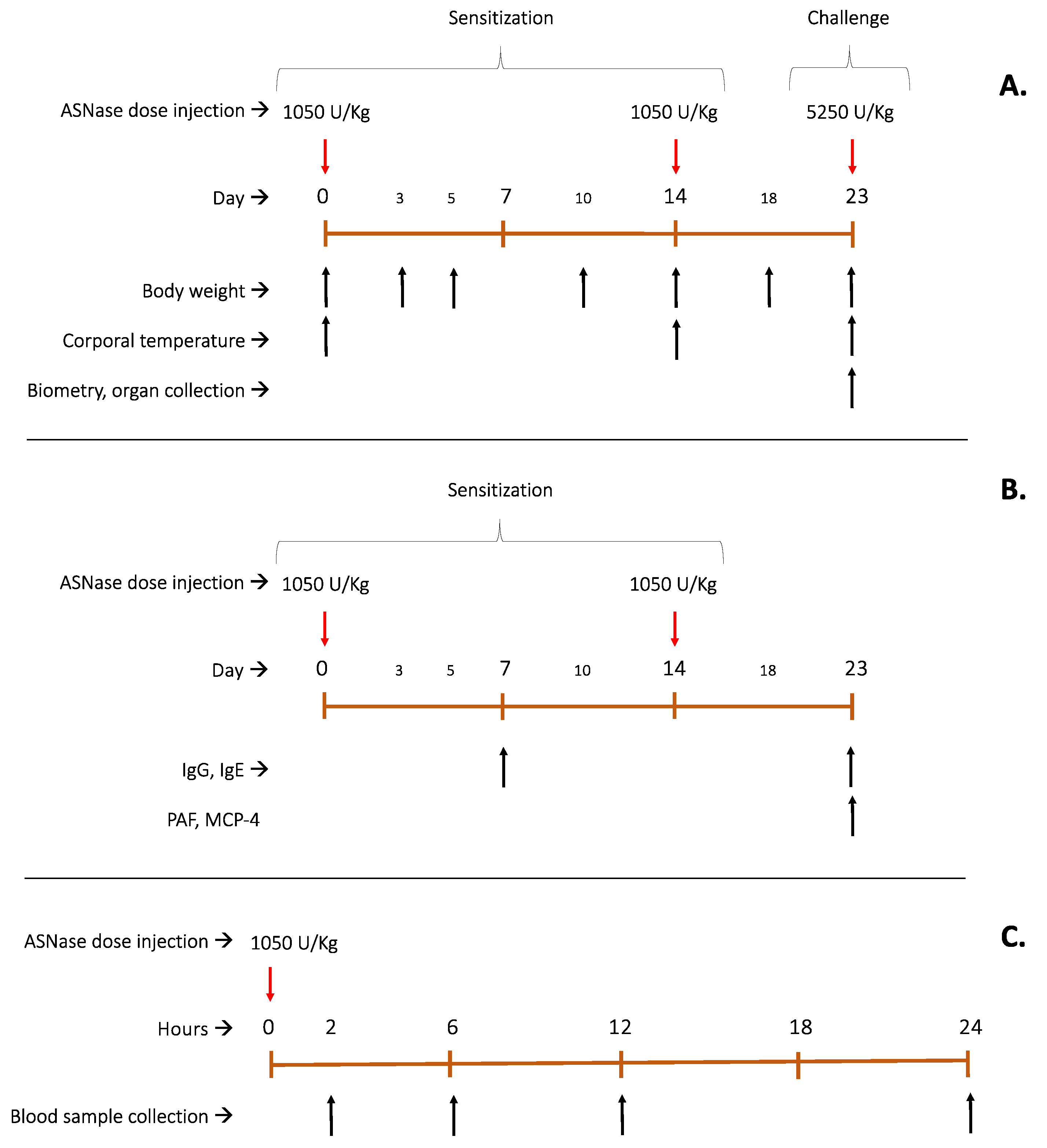
2.3.2. In Vivo Assays
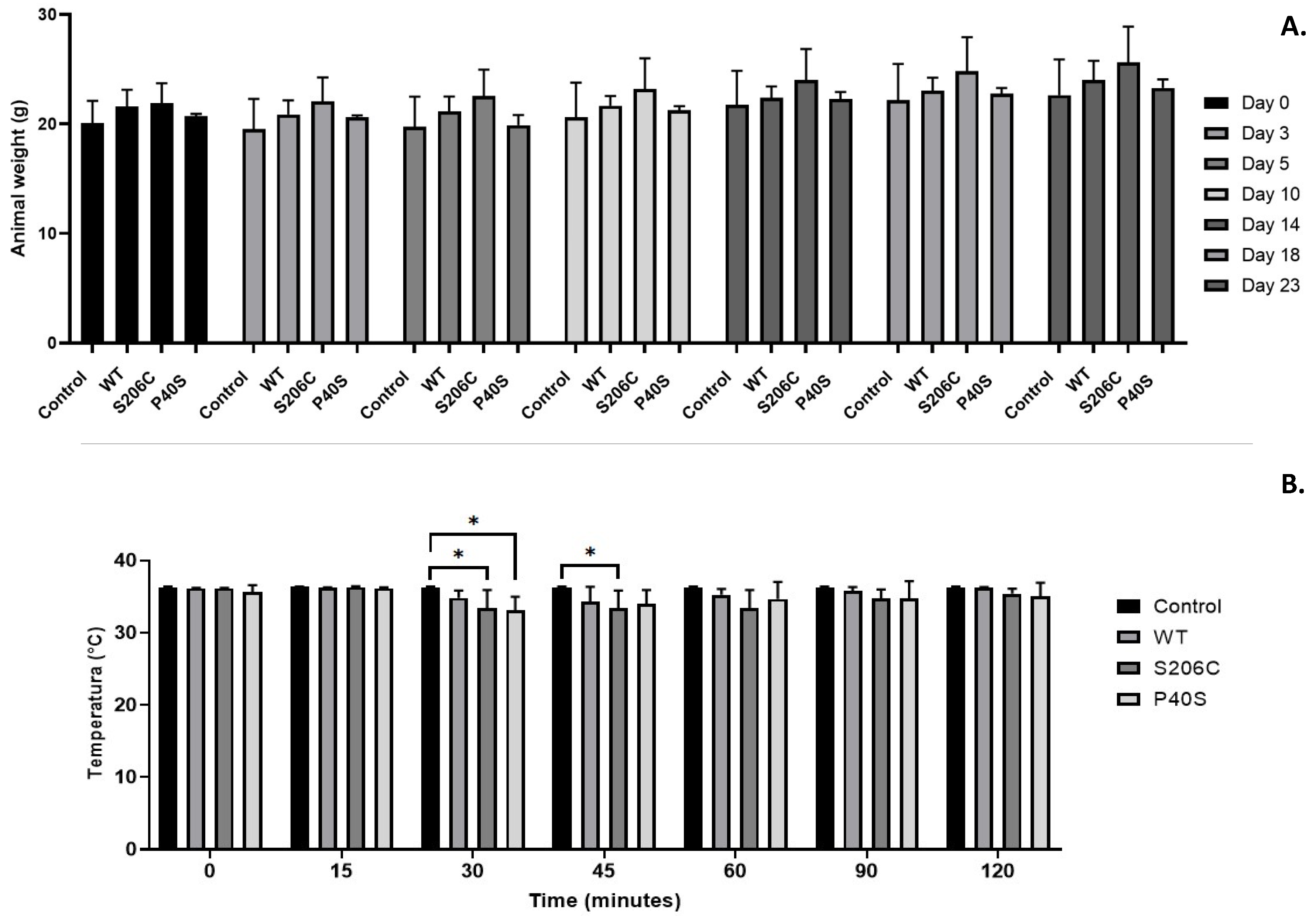
2.3.2.1. Organ Toxicity Analysis after ASNase Exposure
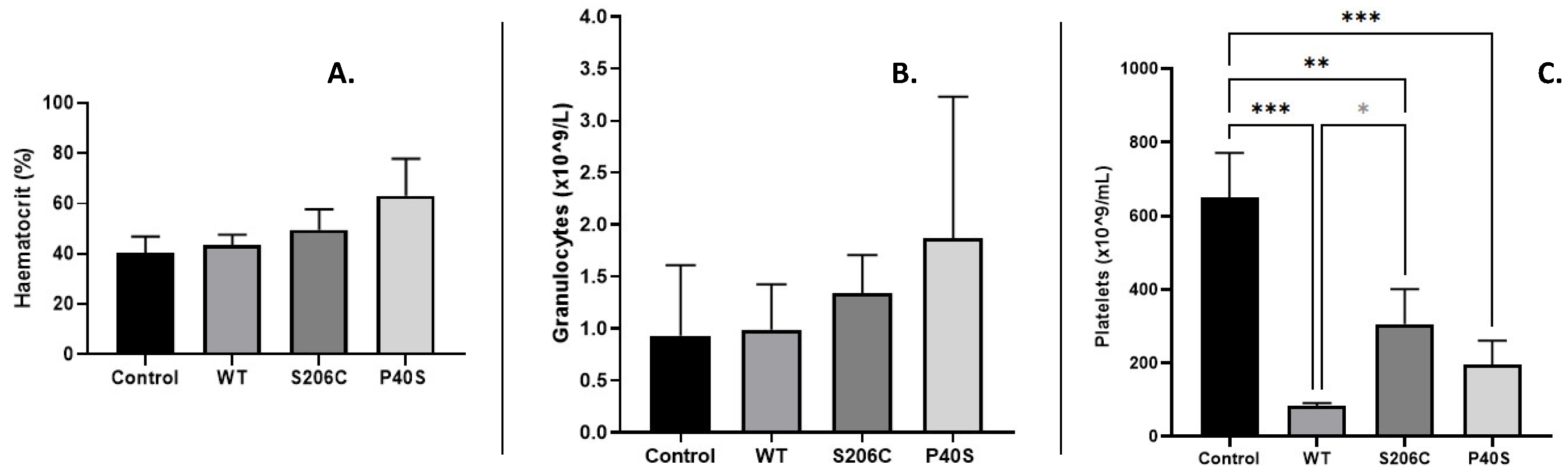
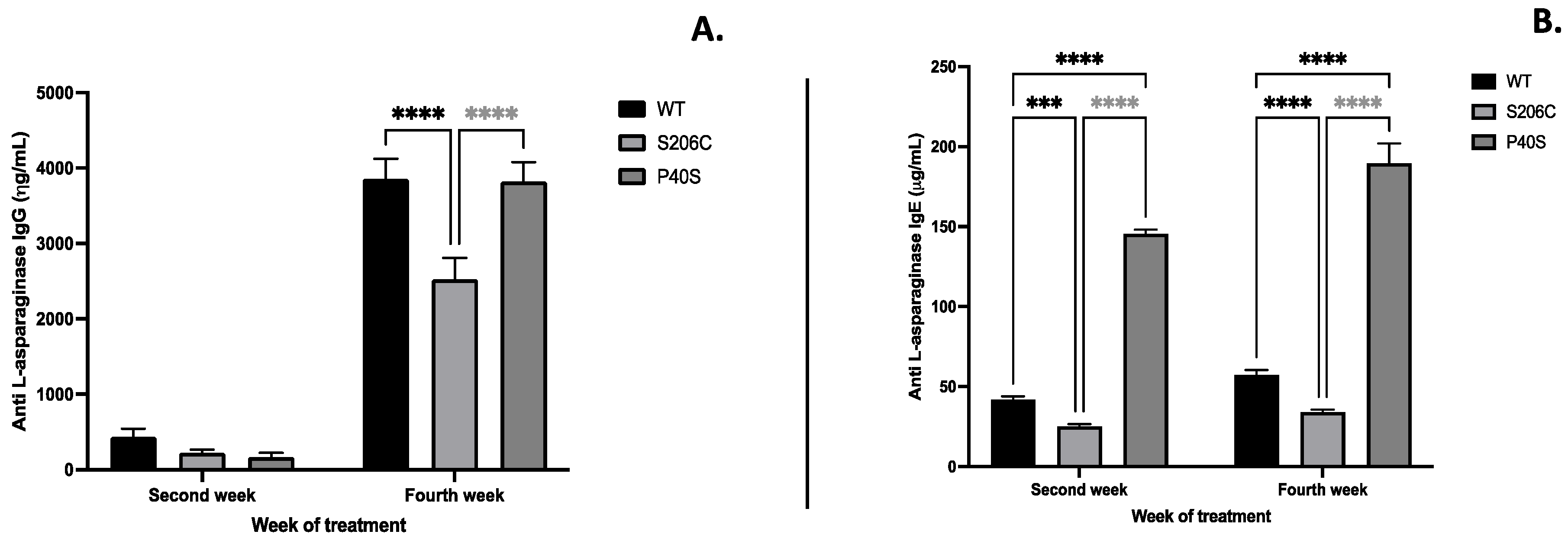
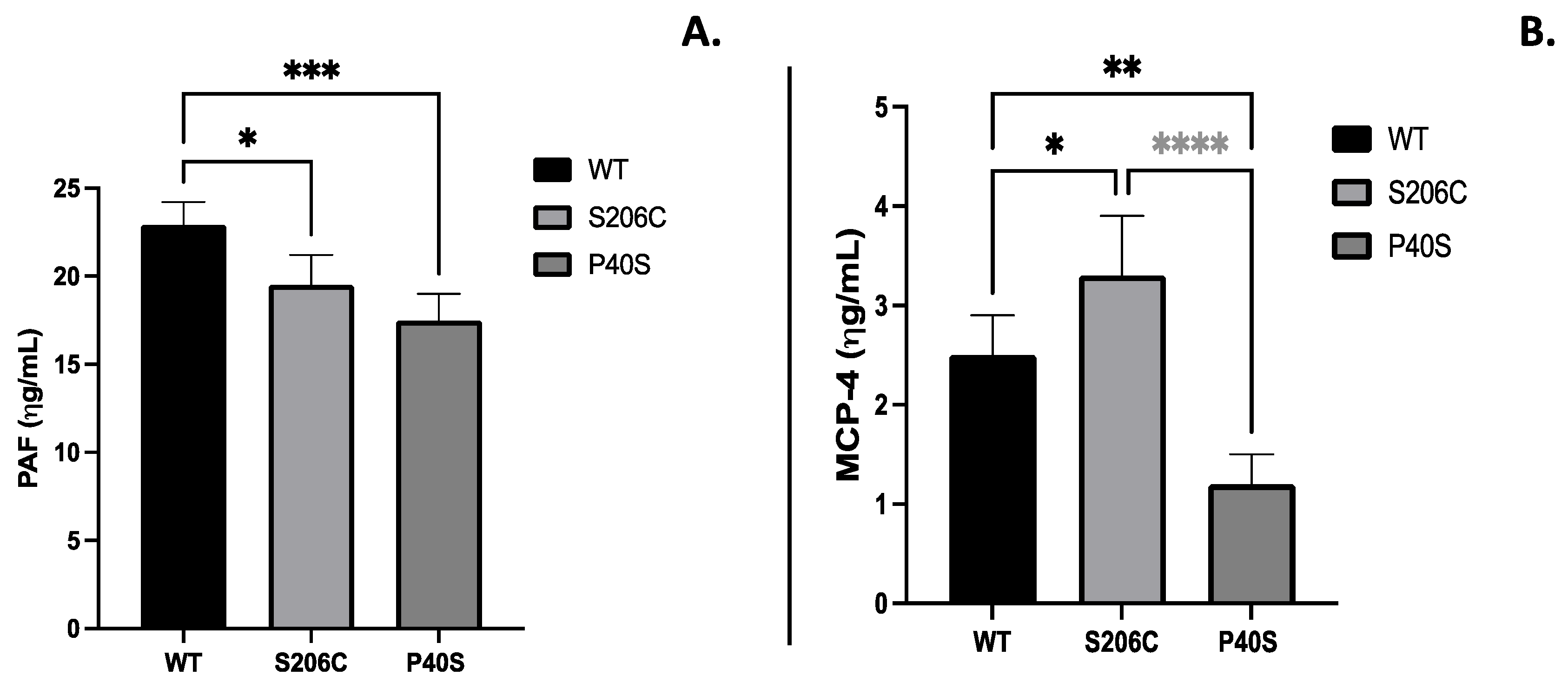
2.3.2.2. Immunogenic and Inflammatory Potential of ASNases
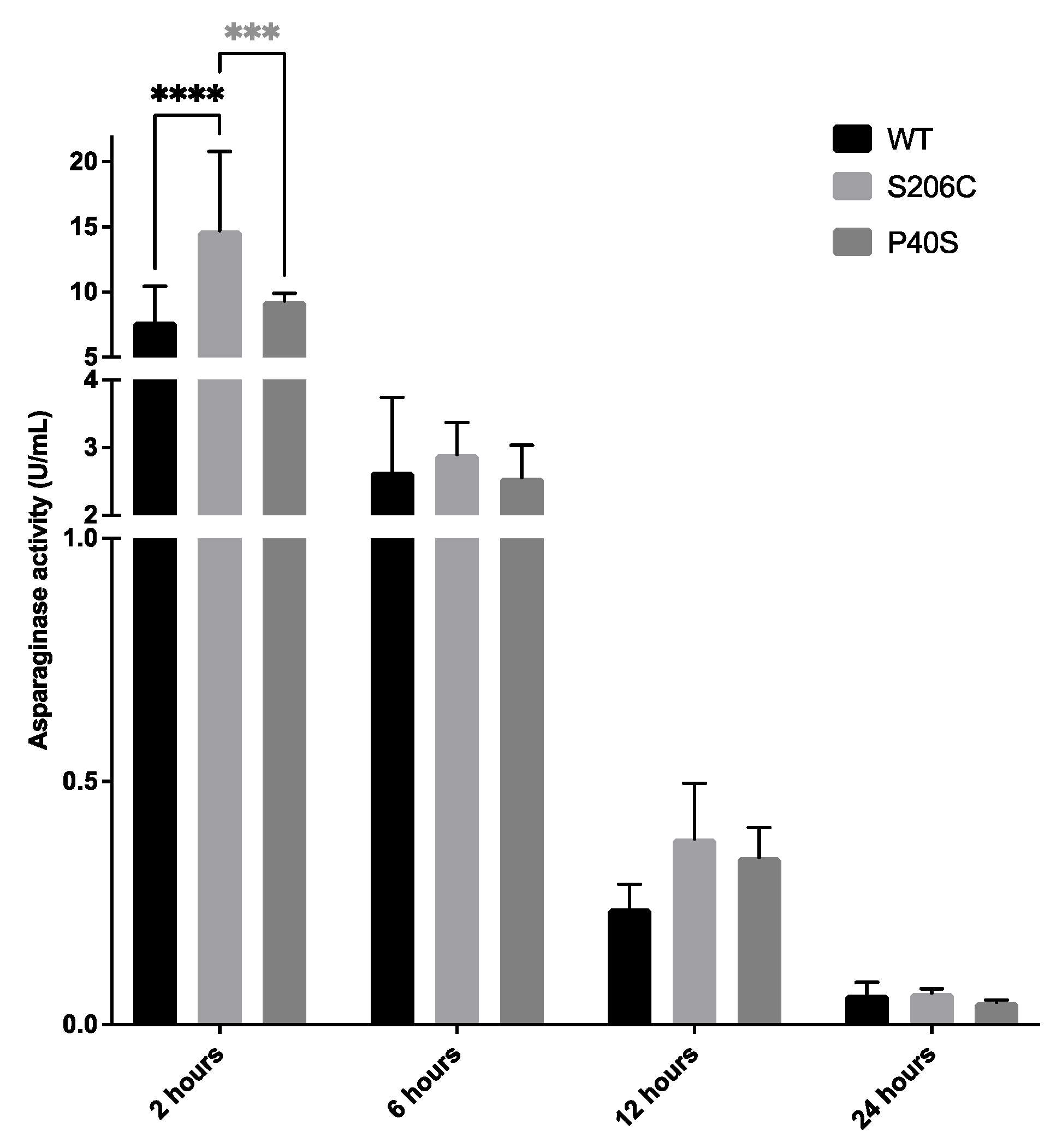
2.3.2.3. Pharmacokinetics of ASNases
3. Discussion
4. Materials and Methods
4.1. Enzyme Production and Purification
4.1.1. Gene and Vector Information
4.1.2. Bacterial Culture
4.1.3. Protein Purification
4.2. Enzymes’ Biochemical Characterization
4.2.1. Enzymes’ Stability on Human Serum
4.3. Kinetics Parameters Determination for ASNases Studied
4.4. Nonclinical Evaluation of ASNases
4.4.1. In Vitro Assays
4.4.1.1. Blood Cancer Cell Lines Cytotoxic Effect of ASNases
4.4.1.2. Solid Tumour Cell Lines Cytotoxic Effect of ASNases
4.4.2. In Vivo Assay
4.4.2.1. Organ Toxicity Analysis
4.4.2.2. Immunogenic and Inflammatory Potential
4.4.2.3. Pharmacokinetics Profile of ASNases
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Medicines Agency. ICH S6 (R1) Preclinical safety evaluation of biotechnology-derived pharmaceuticals - Scientific guideline | European Medicines Agency 1997. https://www.ema.europa.eu/en/ich-s6-r1-preclinical-safety-evaluation-biotechnology-derived-pharmaceuticals-scientific-guideline (accessed April 14, 2024).
- European Medicines Agency. ICH S9 Non-clinical evaluation for anticancer pharmaceuticals - Scientific guideline | European Medicines Agency 2010. https://www.ema.europa.eu/en/ich-s9-non-clinical-evaluation-anticancer-pharmaceuticals-scientific-guideline (accessed April 14, 2024).
- Costa-Silva TA, Costa IM, Biasoto HP, Lima GM, Silva C, Pessoa A, et al. Critical overview of the main features and techniques used for the evaluation of the clinical applicability of L-asparaginase as a biopharmaceutical to treat blood cancer. Blood Rev 2020;43. [CrossRef]
- Sengupta S, Biswas M, Gandhi K, Gota V, Sonawane A. Pre-Clinical Evaluation of Novel L-Asparaginase Mutants for the Treatment of Acute Lymphoblastic Leukemia. Blood 2021;138:4919–4919. [CrossRef]
- Fernandez CA, Smith C, Karol SE, Ramsey LB, Liu C, Pui CH, et al. Effect of premedications in a murine model of asparaginase hypersensitivity. J Pharmacol Exp Ther 2015;352:541–51. [CrossRef]
- Suresh SA, Ethiraj S, Rajnish KN. Toxicity Analysis of Recombinant L-asparaginase I and II in Zebrafish. Indian J Microbiol 2020;60:535. [CrossRef]
- Horvath TD, Chan WK, Pontikos MA, Martin LA, Du D, Tan L, et al. Assessment of l-Asparaginase Pharmacodynamics in Mouse Models of Cancer. Metabolites 2019;9. [CrossRef]
- Sengupta S, Biswas M, Gandhi KA, Gupta SK, Gera PB, Gota V, et al. Preclinical evaluation of engineered L-asparaginase variants to improve the treatment of Acute Lymphoblastic Leukemia. Transl Oncol 2024;43. [CrossRef]
- Rodrigues MAD, Pimenta M V., Costa IM, Zenatti PP, Migita NA, Yunes JA, et al. Influence of lysosomal protease sensitivity in the immunogenicity of the antitumor biopharmaceutical asparaginase. Biochem Pharmacol 2020;182:114230. [CrossRef]
- Ardalan N, Sepahi AA, Khavari-Nejad RA. Development of Escherichia coli asparaginase II for the Treatment of Acute Lymphocytic Leukemia: In Silico Reduction of asparaginase II Side Effects by a Novel Mutant (V27F). Asian Pac J Cancer Prev 2021;22:1137. [CrossRef]
- Munhoz Costa I, Schultz L, De Araujo B, Pedra B, Silva M, Leite M, et al. Recombinant L-asparaginase 1 from Saccharomyces cerevisiae: an allosteric enzyme with antineoplastic activity OPEN 2016. [CrossRef]
- Husain I, Sharma A, Kumar S, Malik F. Purification and Characterization of Glutaminase Free Asparaginase from Enterobacter cloacae: In-Vitro Evaluation of Cytotoxic Potential against Human Myeloid Leukemia HL-60 Cells. PLoS One 2016;11. [CrossRef]
- Brumano LP, da Silva FVS, Costa-Silva TA, Apolinário AC, Santos JHPM, Kleingesinds EK, et al. Development of L-asparaginase biobetters: Current research status and review of the desirable quality profiles. Front Bioeng Biotechnol 2019;6. [CrossRef]
- Saeed H, Hemida A, El-Nikhely N, Abdel-Fattah M, Shalaby M, Hussein A, et al. Highly efficient Pyrococcus furiosus recombinant L-asparaginase with no glutaminase activity: Expression, purification, functional characterization, and cytotoxicity on THP-1, A549 and Caco-2 cell lines. Int J Biol Macromol 2020;156:812–28. [CrossRef]
- Breland UM, Michelsen AE, Skjelland M, Folkersen L, Krohg-Sørensen K, Russell D, et al. Raised MCP-4 levels in symptomatic carotid atherosclerosis: An inflammatory link between platelet and monocyte activation. Cardiovasc Res 2010;86:265–73. [CrossRef]
- Horkheimer I, Schuster DP. THE ROLE OF PLATELET ACTIVATING FACTOR IN SEPSIS: A BENCH-TO-BEDSIDE REVIEW ADVANCES. ADVANCES IN SEPSIS 2002;2.
- Jacob S, Nair AB, Morsy MA. Dose Conversion Between Animals and Humans: A Practical Solution. Indian Journal of Pharmaceutical Education and Research 2022;56:600–7. [CrossRef]
- Wang S, Lai X, Deng Y, Song Y. Correlation between mouse age and human age in anti-tumor research: Significance and method establishment. Life Sci 2020;242:117242. [CrossRef]
- Nair A, Jacob S. A simple practice guide for dose conversion between animals and human. J Basic Clin Pharm 2016;7:27. [CrossRef]
- Silvânia M. P. Neves, Flavia de Moura Prates Ong, Lívia Duarte Rodrigues, Renata Alves dos Santos, Renata Spalutto Fontes, Roseni de Oliveira Santana. Manual de cuidados e procedimentos com animais de laboratório do Biotério de Produção e Experimentação da FCF-IQ/USP. São Paulo: 2013. [CrossRef]
- Rathod S, Ramsey M, Relling M V., Finkelman FD, Fernandez CA. Hypersensitivity reactions to asparaginase in mice are mediated by anti-asparaginase IgE and IgG and the immunoglobulin receptors FceRI and FcγRIII. Haematologica 2019;104:319–29. [CrossRef]
- Talbot SR, Biernot S, Bleich A, van Dijk RM, Ernst L, Häger C, et al. Defining body-weight reduction as a humane endpoint: a critical appraisal. Lab Anim 2020;54:99–110. [CrossRef]
- Morton DB, Griffiths PH. Guidelines on the recognition of pain, distress and discomfort in experimental animals and an hypothesis for assessment. Vet Rec 1985;116:431–6. [CrossRef]
- Gavin HE, Satchell KJF. Surface hypothermia predicts murine mortality in the intragastric Vibrio vulnificus infection model. BMC Microbiol 2017. [CrossRef]
- Vaquero J, Bélanger M, James L, Herrero R, Desjardins P, Côté J, et al. Mild Hypothermia Attenuates Liver Injury and Improves Survival in Mice With Acetaminophen Toxicity 2007. [CrossRef]
- Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, Gutteridge G, et al. Treatment of Comatose Survivors of Out-of-Hospital Cardiac Arrest with Induced Hypothermia. New England Journal of Medicine 2002;346:557–63. [CrossRef]
- Holzer M. Mild Therapeutic Hypothermia to Improve the Neurologic Outcome after Cardiac Arrest. New England Journal of Medicine 2002;346:549–56. [CrossRef]
- Eicher DJ, Wagner CL, Katikaneni LP, Hulsey TC, Bass WT, Kaufman DA, et al. Moderate hypothermia in neonatal encephalopathy: Efficacy outcomes. Pediatr Neurol 2005;32:11–7. [CrossRef]
- Shankaran S, Laptook AR, Ehrenkranz RA, Tyson JE, McDonald SA, Donovan EF, et al. Whole-Body Hypothermia for Neonates with Hypoxic–Ischemic Encephalopathy. New England Journal of Medicine 2005;353:1574–84. [CrossRef]
- Stewart CR, Landseadel JP, Gurka MJ, Fairchild KD. Hypothermia increases interleukin-6 and interleukin-10 in juvenile endotoxemic mice $watermark-text $watermark-text $watermark-text. Pediatr Crit Care Med 2010;11:109–16. [CrossRef]
- Laptook AR, Corbett RJT, Sterett R, Garcia D, Tollefsbol G. Quantitative relationship between brain temperature and energy utilization rate measured in Vivo using 31P and 1H magnetic resonance spectroscopy. Pediatr Res 1995;38:919–25. [CrossRef]
- Globus MY -T, Alonso O, Dietrich WD, Busto R, Ginsberg MD. Glutamate Release and Free Radical Production Following Brain Injury: Effects of Posttraumatic Hypothermia. J Neurochem 1995;65:1704–11. [CrossRef]
- Gunn AJ, Gunn TR, De Haan HH, Williams CE, Gluckman PD. Dramatic neuronal rescue with prolonged selective head cooling after ischemia in fetal lambs. Journal of Clinical Investigation 1997;99:248–56. [CrossRef]
- Ginsberg MD, Belayev L. Biological and molecular mechanisms of hypothermic neuroprotection. Therapeutic Hypothermia, CRC Press; 2004, p. 85–141. [CrossRef]
- Wei H, Yin M, Lu Y, Yang Y, Li B, Liao X-X, et al. Mild hypothermia improves neurological outcome in mice after cardiopulmonary resuscitation through Silent Information Regulator 1-actviated autophagy. Cell Death Discov 2019;5:129. [CrossRef]
- Morowski M, Vögtle T, Kraft P, Kleinschnitz C, Stoll G, Nieswandt B. Only severe thrombocytopenia results in bleeding and defective thrombus formation in mice. Blood 2013;121:4938–47. [CrossRef]
- Michael B, Yano B, Sellers RS, Perry R, Morton D, Roome N, et al. Evaluation of Organ Weights for Rodent and Non-Rodent Toxicity Studies: A Review of Regulatory Guidelines and a Survey of Current Practices. Http://DxDoiOrg/101080/01926230701595292 2017;35:742–50. [CrossRef]
- Koo SH. Nonalcoholic fatty liver disease: molecular mechanisms for the hepatic steatosis. Clin Mol Hepatol 2013;19:210–5. [CrossRef]
- Alex S, Boss A, Heerschap A, Kersten S. Exercise training improves liver steatosis in mice. Nutr Metab (Lond) 2015;12:1–11. [CrossRef]
- Robinson MW, Harmon C, O’Farrelly C. Liver immunology and its role in inflammation and homeostasis. Cellular & Molecular Immunology 2016 13:3 2016;13:267–76. [CrossRef]
- Van Der Meer LT, Terry SYA, Van Ingen Schenau DS, Andree KC, Franssen GM, Roeleveld DM, et al. In vivo imaging of antileukemic drug asparaginase reveals a rapid macrophage-mediated clearance from the bone marrow. Journal of Nuclear Medicine 2017;58:214–20. [CrossRef]
- Céspedes N, Donnelly EL, Lowder C, Hansten G, Wagers D, Briggs AM, et al. Mast Cell Chymase/Mcpt4 Suppresses the Host Immune Response to Plasmodium yoelii, Limits Malaria-Associated Disruption of Intestinal Barrier Integrity and Reduces Parasite Transmission to Anopheles stephensi. Front Immunol 2022;13. [CrossRef]
- Pejler G, Rönnberg E, Waern I, Wernersson S. Mast cell proteases: multifaceted regulators of inflammatory disease. Blood 2010;115:4981–90. [CrossRef]
- Madjene LC, Danelli L, Dahdah A, Vibhushan S, Bex-Coudrat J, Pacreau E, et al. Mast cell chymase protects against acute ischemic kidney injury by limiting neutrophil hyperactivation and recruitment. Kidney Int 2020;97:516–27. [CrossRef]
- Holland M, Castro F V., Alexander S, Smith D, Liu J, Walker M, et al. RAC2, AEP, and ICAM1 expression are associated with CNS disease in a mouse model of pre-B childhood acute lymphoblastic leukemia. Blood 2011;118:638–49. [CrossRef]
- Patel N, Krishnan S, Offman MN, Krol M, Moss CX, Leighton C, et al. A dyad of lymphoblastic lysosomal cysteine proteases degrades the antileukemic drug L-asparaginase. Journal of Clinical Investigation 2009;119:1964–73. [CrossRef]
- Qi Q, Obianyo O, Du Y, Fu H, Li S, Ye K. Blockade of Asparagine Endopeptidase Inhibits Cancer Metastasis. J Med Chem 2017;60:7244–55. [CrossRef]
- Riccardi R, Holcenberg S, Glaubiger D, Wood J, Poplack D. L-asparaginase pharmacokinetics and asparagine levels in cerebrospinal fluid of rhesus monkeys and humans . Cancer Res 1981;11:4554–8.
- Asselin B, Rizzari C. Asparaginase pharmacokinetics and implications of therapeutic drug monitoring. Leuk Lymphoma 2015;56:2273. [CrossRef]
- Purwaha P, Lorenzi PL, Silva LP, Hawke DH, Weinstein JN. Targeted metabolomic analysis of amino acid response to L-asparaginase in adherent cells. Metabolomics 2014;10:909–19. [CrossRef]
- Saeed H, Hemida A, Abdel-Fattah M, Eldoksh A, Shalaby M, Nematalla H, et al. Pseudomonas aeruginosa recombinant L-asparaginase: Large scale production, purification, and cytotoxicity on THP-1, MDA-MB-231, A549, Caco2 and HCT-116 cell lines. Protein Expr Purif 2021;181:105820. [CrossRef]
- Blachier J, Cleret A, Guerin N, Gil C, Fanjat JM, Tavernier F, et al. L-asparaginase anti-tumor activity in pancreatic cancer is dependent on its glutaminase activity and resistance is mediated by glutamine synthetase. Exp Cell Res 2023;426:113568. [CrossRef]
- Karpel-Massler G, Ramani D, Shu C, Halatsch ME, Westhoff MA, Bruce JN, et al. Metabolic reprogramming of glioblastoma cells by L-asparaginase sensitizes for apoptosis in vitro and in vivo. Oncotarget 2016;7:33512. [CrossRef]
- Schalk AM, Lavie A. Structural and Kinetic Characterization of Guinea Pig l-Asparaginase Type III. Biochemistry 2014;53:2318. [CrossRef]
- Munhoz Costa I, Custódio Moura D, Meira Lima G, Pessoa A, Oresco dos Santos C, de Oliveira MA, et al. Engineered asparaginase from Erwinia chrysanthemi enhances asparagine hydrolase activity and diminishes enzyme immunoreactivity - a new promise to treat acute lymphoblastic leukemia. Journal of Chemical Technology and Biotechnology 2022;97:228–39. [CrossRef]
- Cecconello DK, Rechenmacher C, Werlang I, Zenatti PP, Yunes JA, Alegretti AP, et al. Implementation of the asparaginase activity assessment technique for clinical use: experience of a Brazilian Center. Sci Rep 2020;10:21481. [CrossRef]

| Enzyme | ASNase activity (U/mg) | GLNase activity (U/mg) |
Relative GLNase activity % |
|---|---|---|---|
| WT | 76.22 ±0.82 | 1.79 ±0.09 | 2.35 |
| S206C | 74.86 ±0.32 | 1.73 ±0.04 | 2.31 |
| P40S | 60.82 ±0.72***### | 1.27 ±0.02**## | 2.09 |
| P40S/S206C | 52.88 ±1.12****####++ | 1.76 ±0.05++ | 3.33 |
| Enzyme |
ASNase activity (0 hours) |
ASNase activity (96 hours) |
Residual activity |
|---|---|---|---|
| WT-PBS 1x | 88.1 ±19.8 | 51.4 ±10.1** | 58.3% |
| WT-HS | 87.6 ±14.8 | 61.7 ±13.2 | 70.4% |
| S206C-PBS 1x | 74.7 ±5.0 | 43.9 ±14.8* | 58.8% |
| S206C-HS | 78.0 ±3.3 | 64.8 ±13.2 | 83.2% |
| P40S-PBS 1x | 69.4 ±2.5 | 28.0 ±10.8** | 40.3% |
| P40S-HS | 69.5 ±2.0 | 53.1 ±15.1 | 76.4% |
| P40S/S206C-PBS 1x | 61.1 ±1.6 | 12.1 ±5.6*** | 19.7% |
| P40S/S206C-HS | 62.8 ±1.7 | 27.6 ±9.4* | 44.0% |
| Leuginase | WT | S206C | P40S | ||
|---|---|---|---|---|---|
| Kinetic parameters | Vmax (µmole/min) |
0.05728 ±0.0063 | 0.04787 ±0.0073 | 0.04647 ±0.0065 | 0.06001 ±0.0055 |
| KM (µM) |
291.3 ±23.6 | 197.6 ±17.9* | 194.0 ±12.5* | 262.7.2 ±9.8# | |
| Goodness of fit | R2 | 0.9744 | 0.9203 | 0.9362 | 0.9389 |
| Cell line | ASNase type | ||
|---|---|---|---|
| WT | S206C | P40S | |
| MOLT-4 | 0.112 ±0.020 | 0.076 ±0.012 | 0.045 ±0.013* |
| REH | 0.114 ±0.019 | 0.149 ±0.031 | 0.114 ±0.002 |
| CELL LINE | ASNASE TYPE | ||
|---|---|---|---|
| WT | S206C | P40S | |
| MDA-MB-231 | 5.9 ±0.075 | 6.3 ±0.434 | 7.5 ±0.584**# |
| Caco-2 | 9.2 ±0.522 | 7.1±0.249* | 9.0 ±1.074# |
| SK-OV-3 | 8.5 ±1.344 | 11.9 ±0.43* | 9.4 ±1.013 |
| PANC-1 | 3.3 ±0.166 | 2.7 ±0.075** | 2.9 ±0.201* |
| U-87 MG | 1.5 ±0.107 | 1.2 ±0.099* | 0.8 ±0.098***## |
| Organ | Weight (mg) | |||
|---|---|---|---|---|
| Control | WT | S206C | P40S | |
| Heart | 147.1 ±7.3 | 162.8 ±4.0 | 151.9 ±3.7 | 153.8 ±11.5 |
| Spleen | 99.1 ±18.0 | 100.2 ±2.3 | 94.3 ±10.3 | 91.7 ±3.2 |
| Liver | 1529.2 ±38.2 | 1846.7* ±50.1 | 1686.7 ±163.7 | 1827.0* ±34.7 |
| Kidney | 317.5 ±27.0 | 291.9 ±1.1 | 264.8 ±7.2 | 251.5 ±12.5 |
| Pancreas | 940.0 ±2.9 | 774.8* ±21.3 | 732.2* ±30.2 | 713.3* ±38.1 |
| Thymus | 85.6 ±13.9 | 99.4 ±11.7 | 57.6 ±2.0 | 84.7 ±7.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




