Submitted:
19 April 2024
Posted:
23 April 2024
You are already at the latest version
Abstract
Keywords:
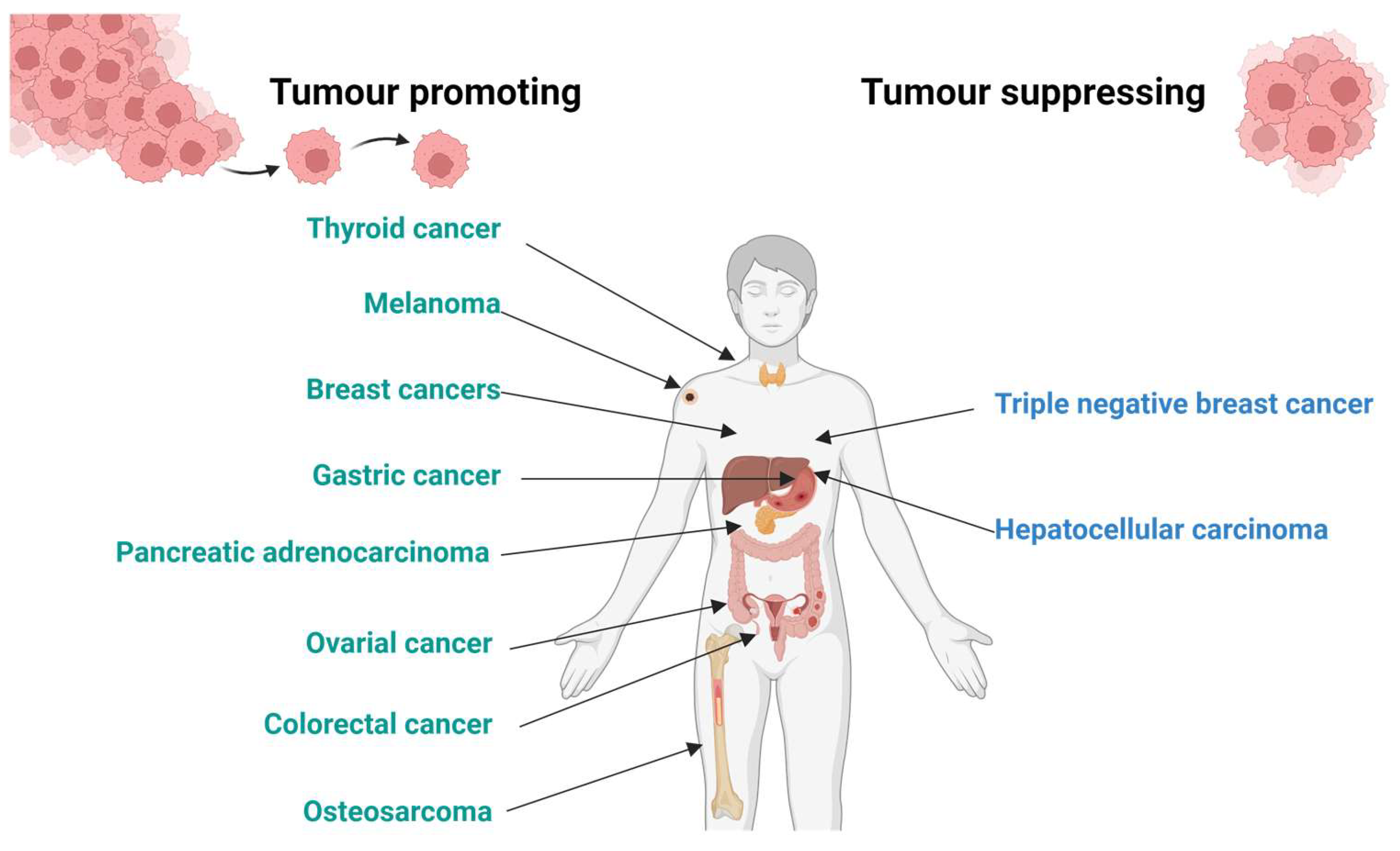
1. Introduction
2. Results
2.1. Inositol Phosphate Accumulation Assay
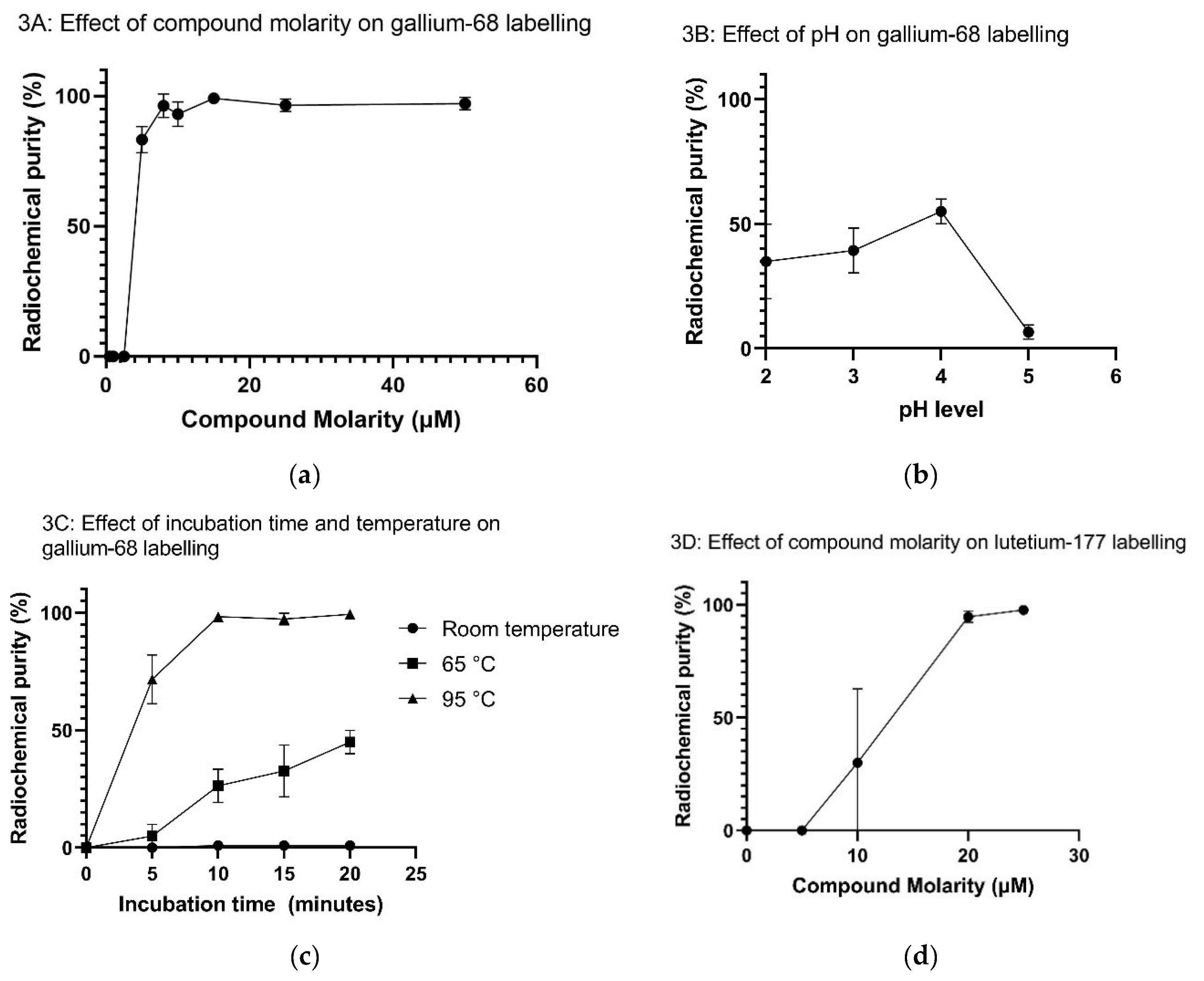
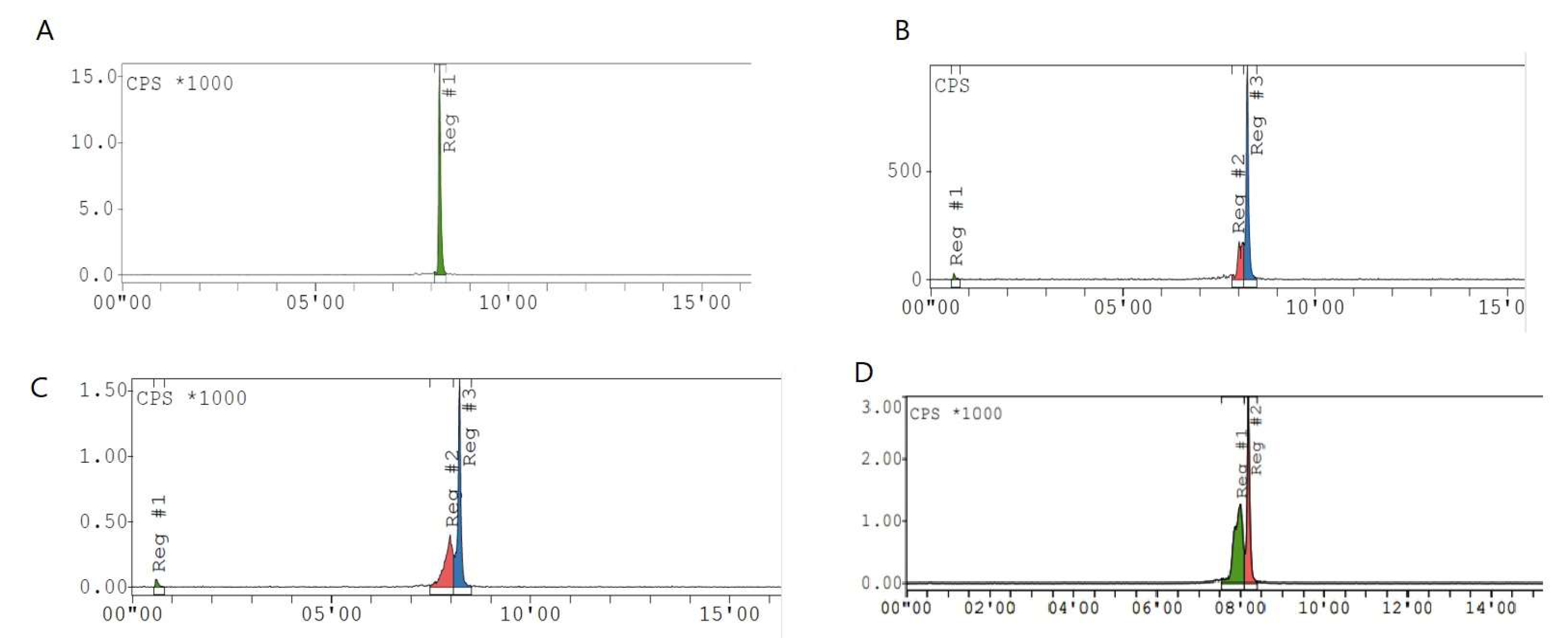
2.2. Radiolabeling Optimization and Radiochemical Purity
2.3. Log P Octanol/Phosphate Buffered Assay
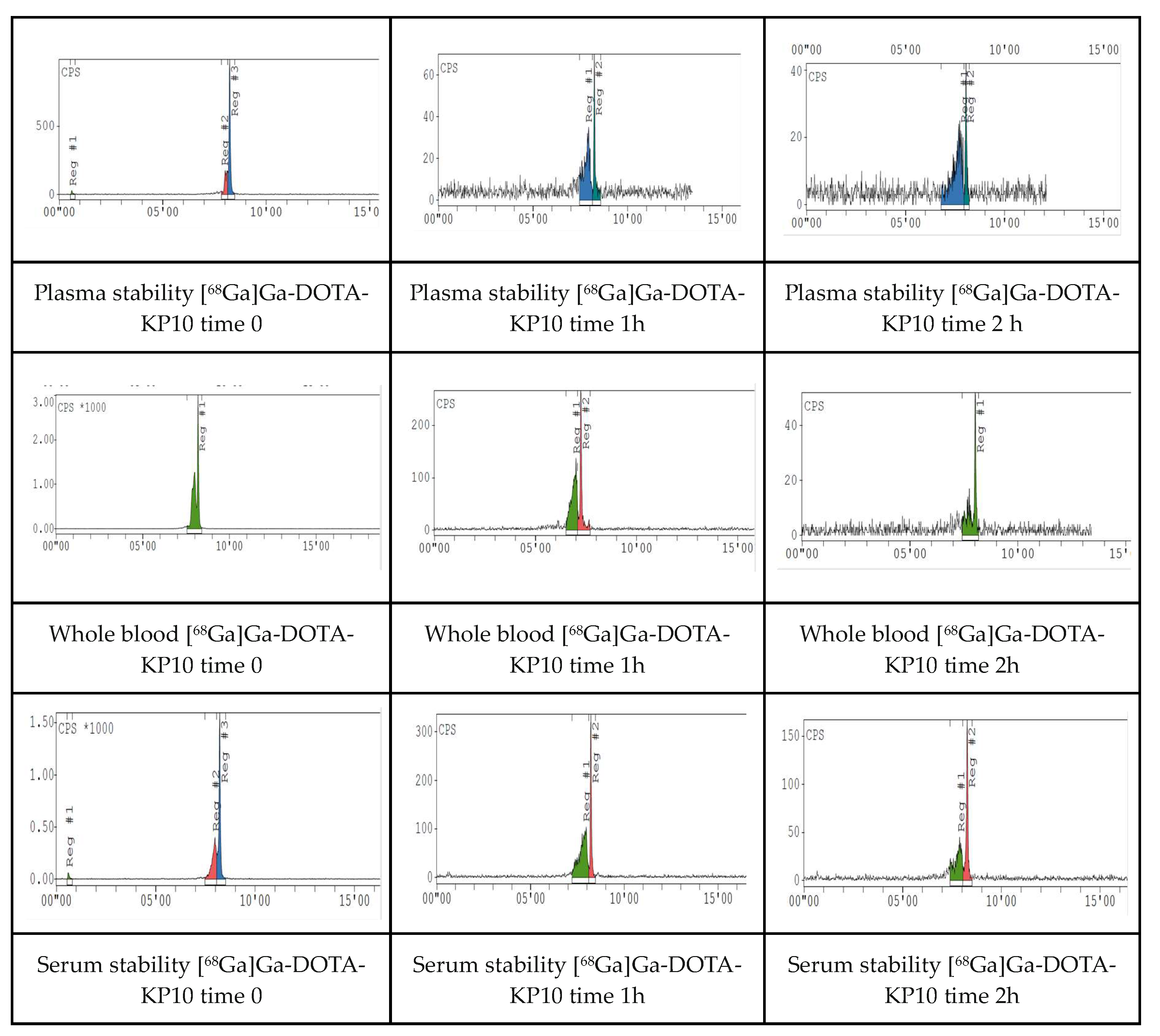
2.4. Ex Vivo Stability
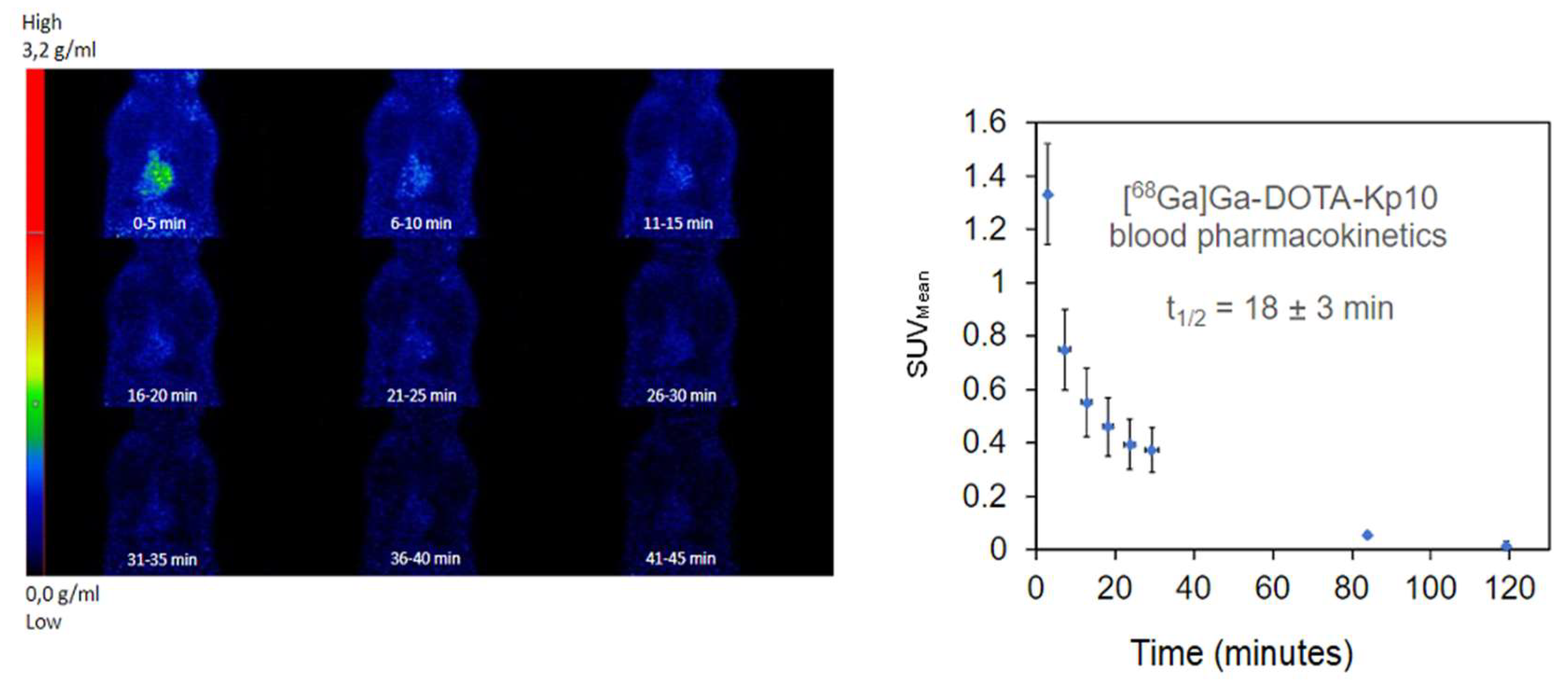
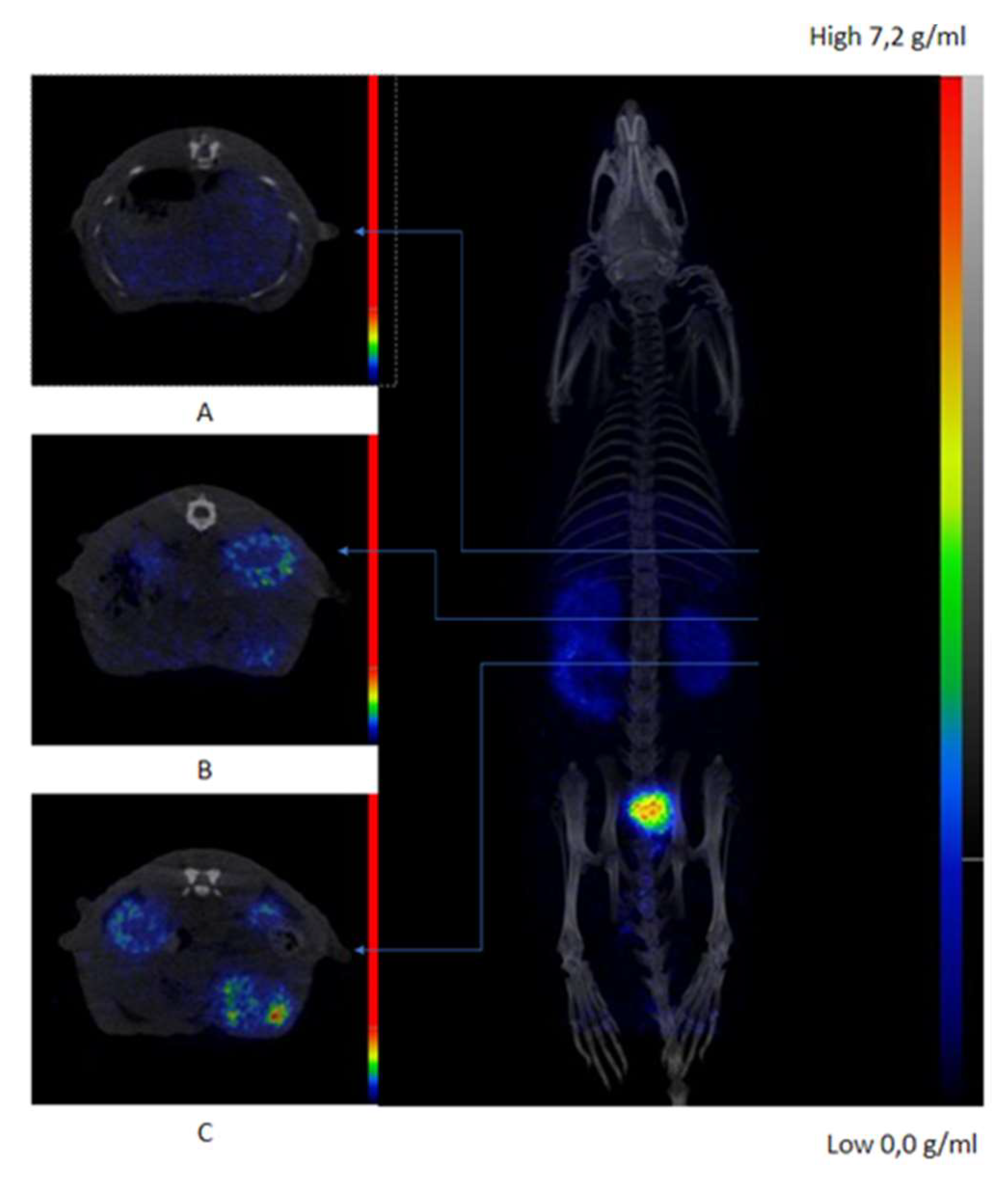
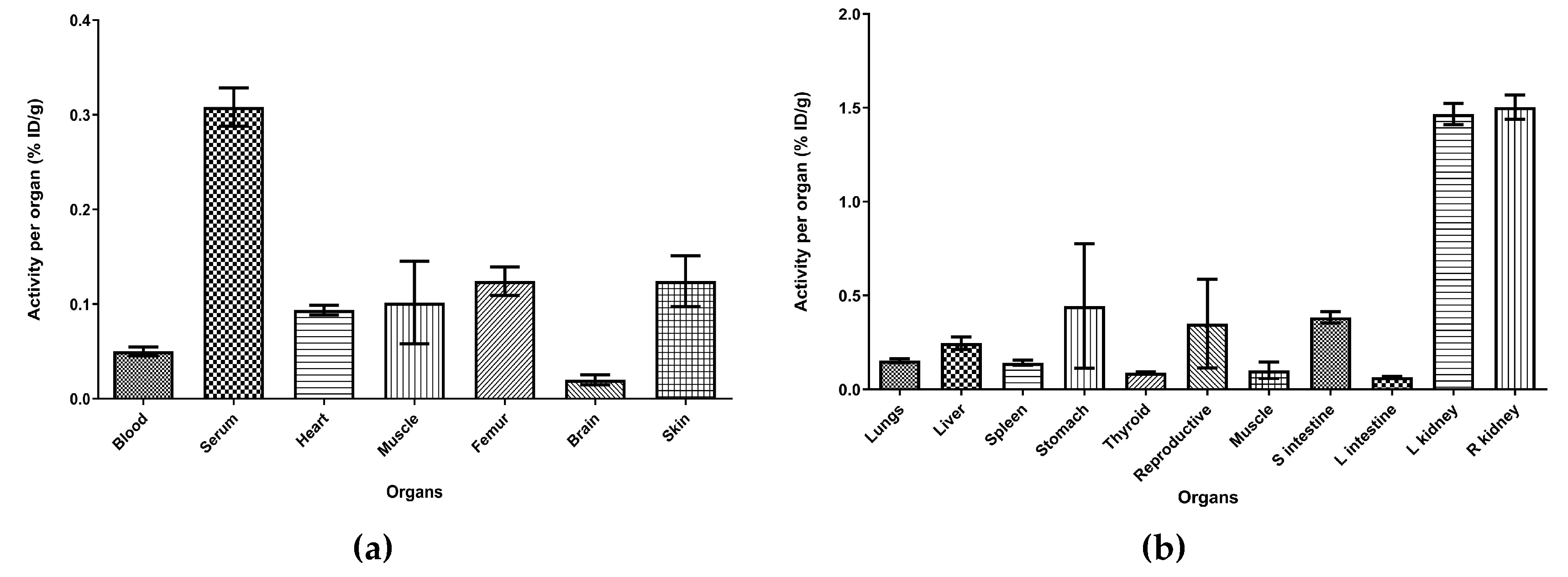
2.5. In Vivo Evaluation and Ex-Vivo Biodistribution
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. HEK Cell Culture Preparation and Inositol Phosphate Accumulation Assay
4.2.2. Radiolabeling Optimization
4.2.3. Product Purification for in Vivo Analysis
4.2.4. Radiochemical Purity Analysis
4.2.5. Log P Octanol/Phosphate Buffered Assay
4.2.6. Ex Vivo Stability
4.2.7. In Vivo Evaluation
4.2.8. Ex-Vivo Biodistribution
4.2.9. Statistical Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
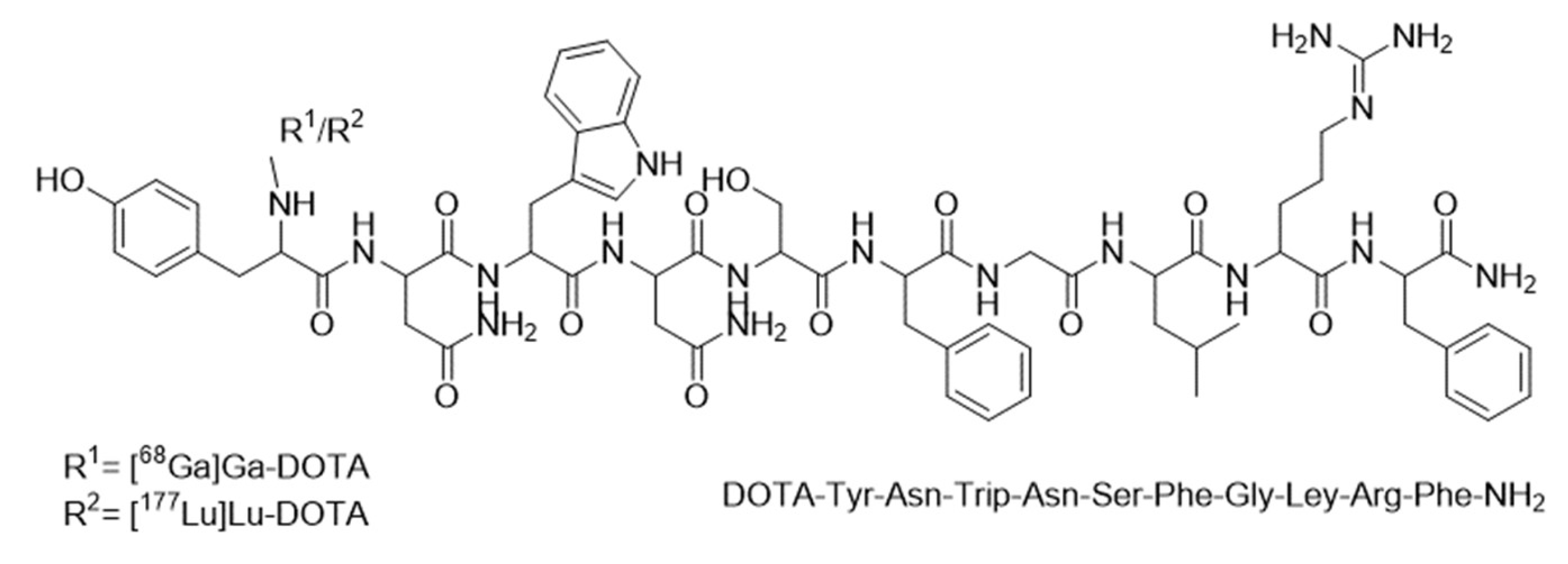
Appendix A. Structure of DOTA-KP10
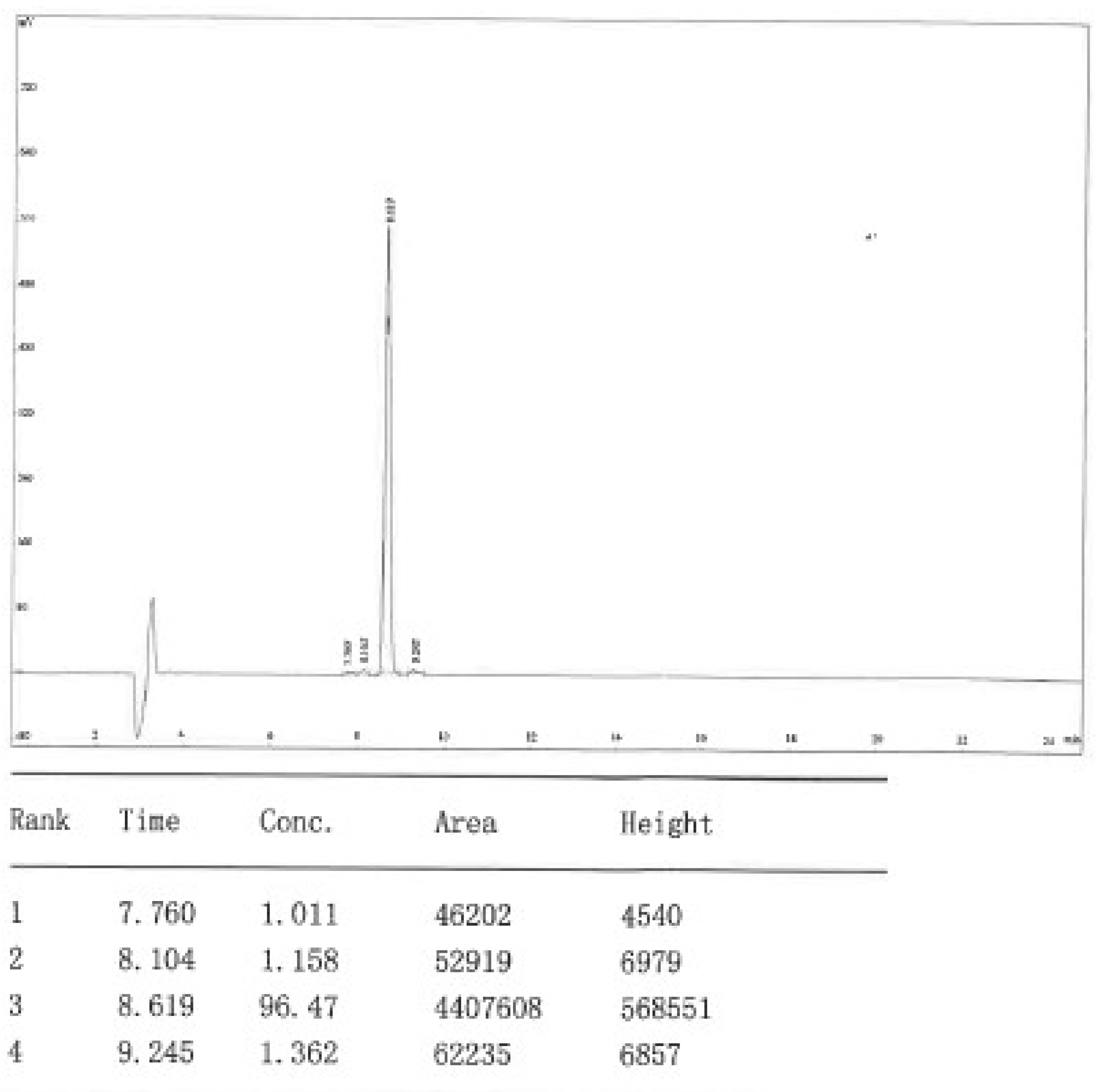
Appendix B. HPLC Dat of Raw Material DOTA-KP10
Appendix C
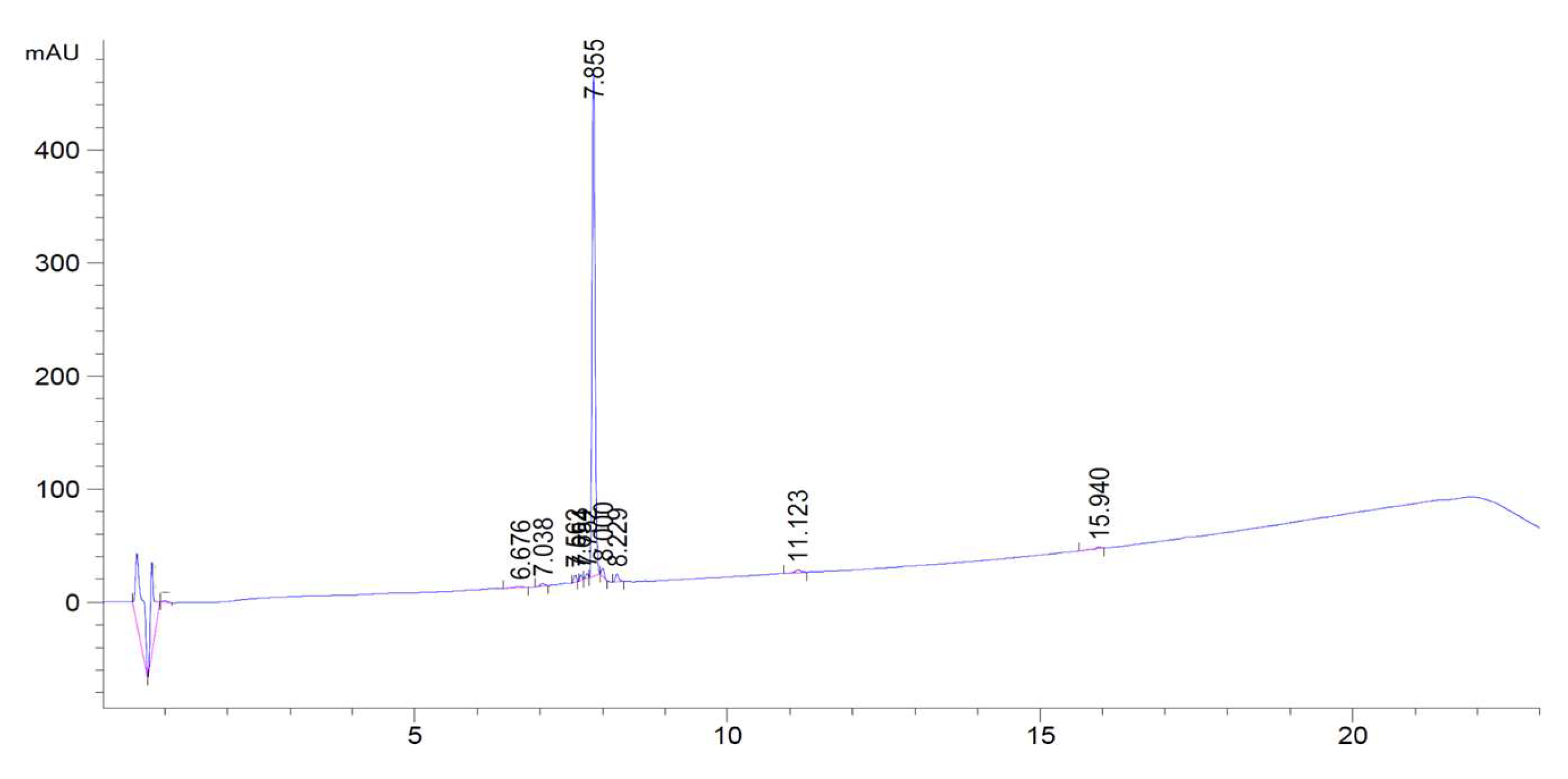
Appendix D. Chromatogram Examples
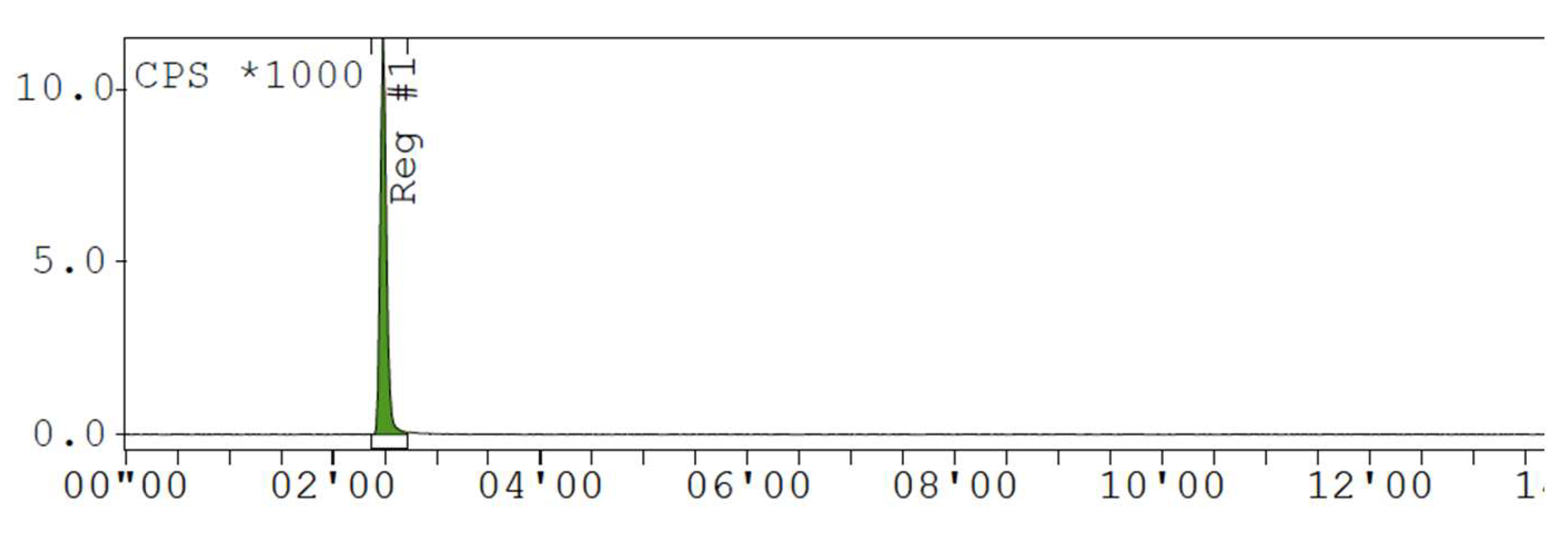
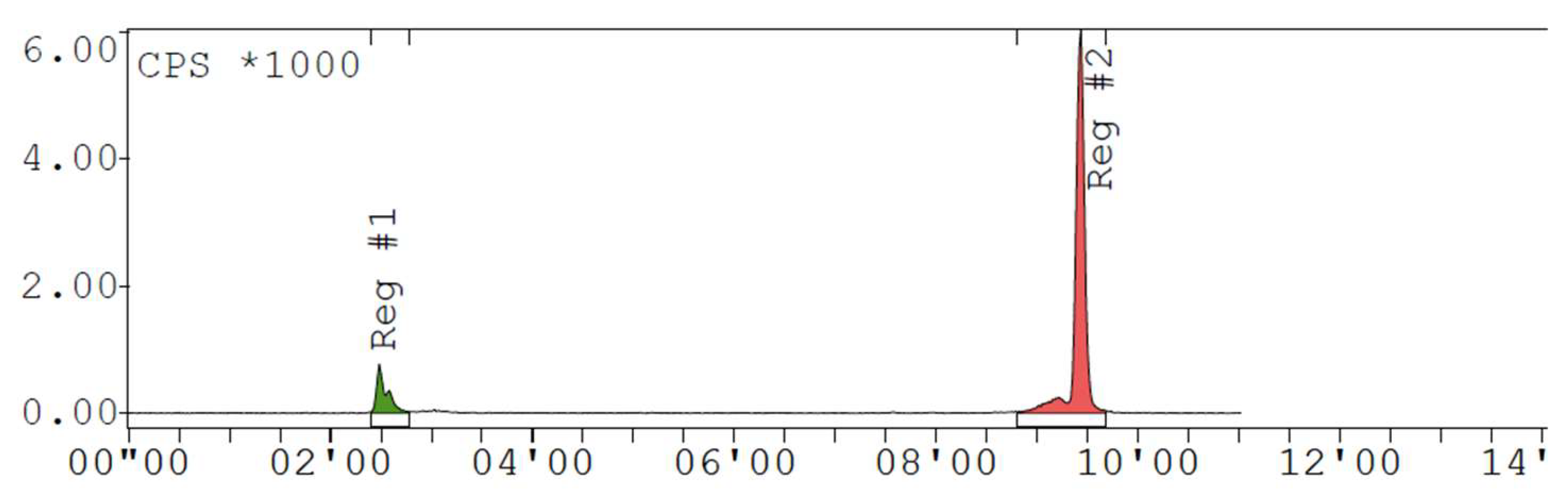
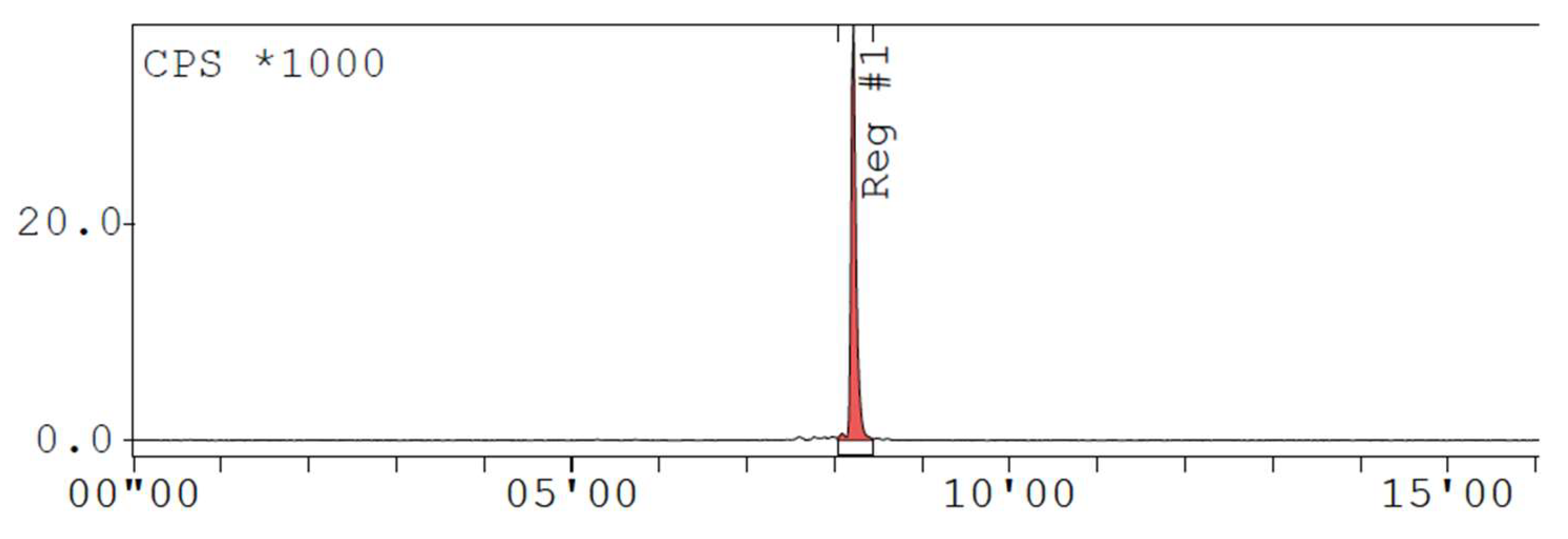
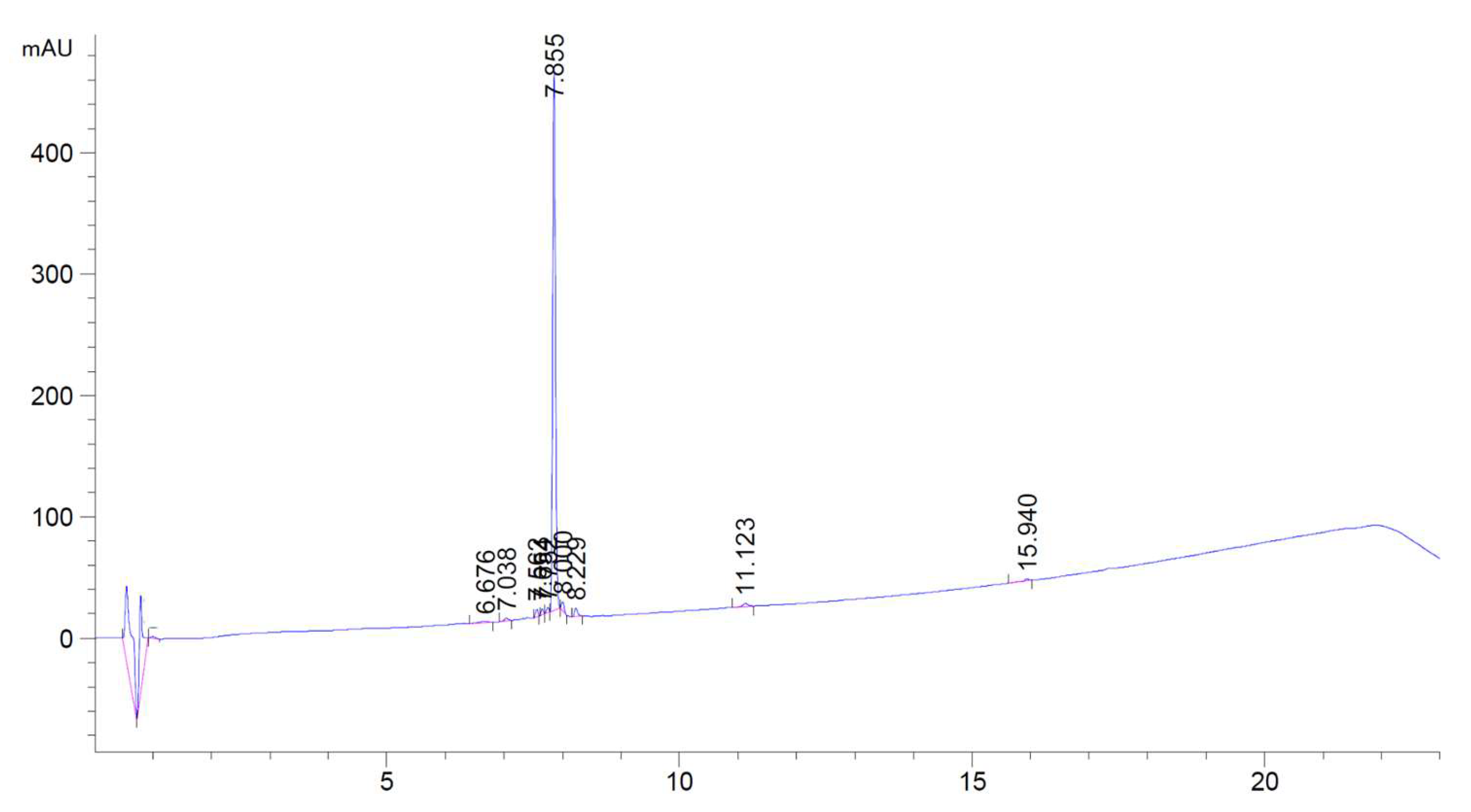
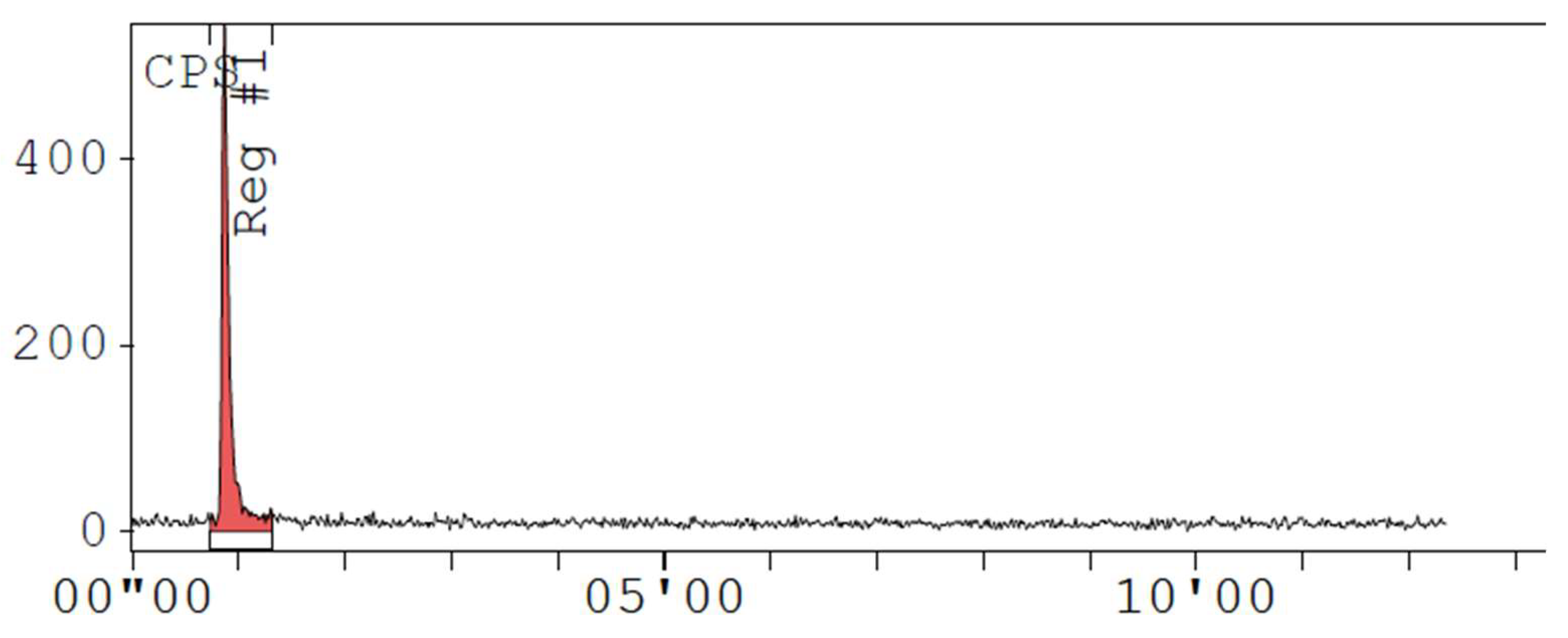
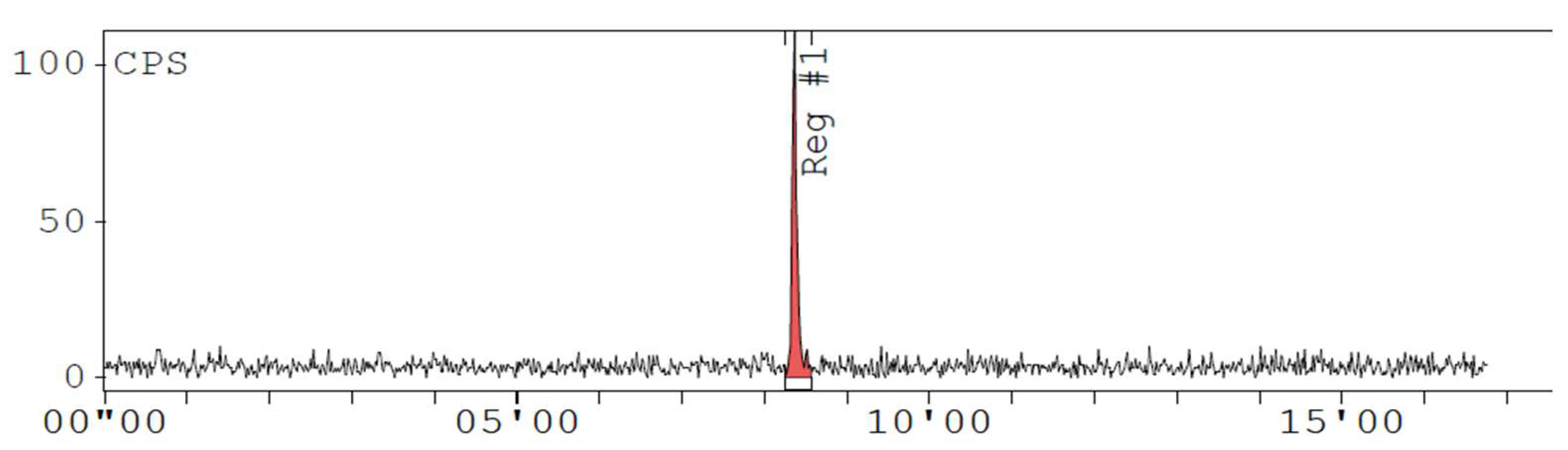
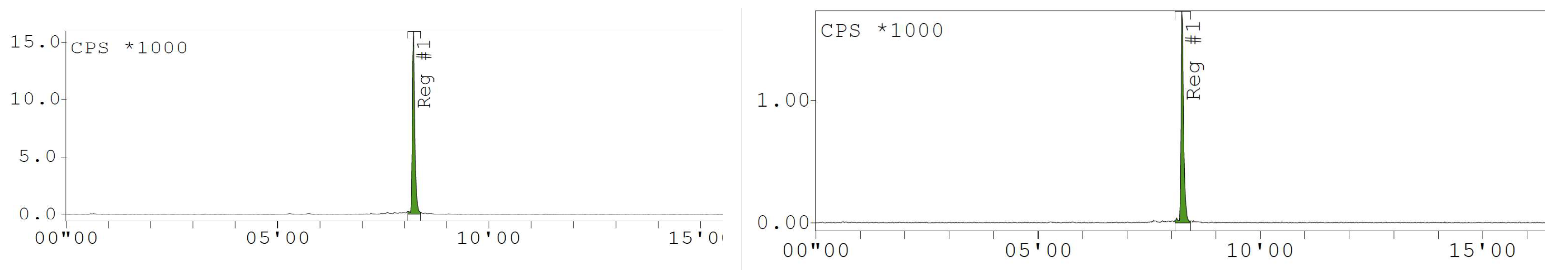
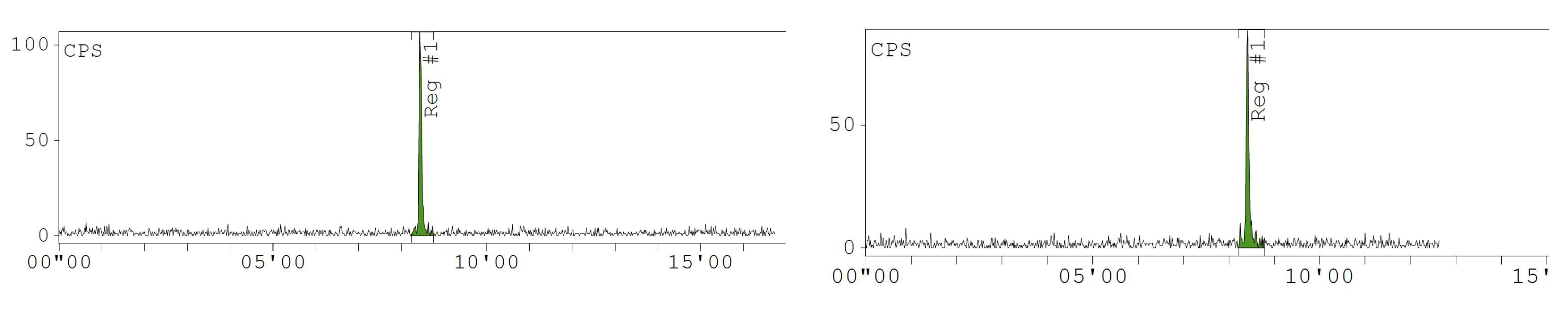
References
- Ciaramalla, V. , Della Corte, C.M., Ciardiello, F., Morgillo, F. Kisspeptin and Cancer: Molecular interaction, biological functions, and future perspectives. Front Endocrinol 2018, 9, 115. [CrossRef]
- Guzman, S. , Brackstone, M. , Radovick, S., Badwah, A.V., Bhattacharya, M.M. KISS1/KISS1R in cancer: friend or foe? Front Endocrinol 2018, 9, 437. [Google Scholar] [CrossRef]
- Blake, A. , Dragan, M., Tirona, R.G., Hardy, D.B., Brakstone, M., Truck, A.B., Badwah, A.V., Bhattacharya, M.G. G protein-coupled KISS1 Receptor is overexpressed in triple negative breast cancer and promotes drug resistance. Sci Rep 2017, 7, 46525. [CrossRef]
- Frantagelo, F. , Carroerop, M.V., Motti, M.L. Controversial role of kisspeptins/KiSS-1R signalling system in tumour development. Front Endocrinol 2018, 9, 192. [CrossRef]
- Dragan, M. , Nguyen, MU. , Guzman, S., Goertzen, B., Brackstone, M. G-protein coupled kisspeptin receptor induces metabolic reprogramming and tumorigenesis in oestrogen receptor-negative breast cancer. Cell Death Dis 2020, 11, 206. [Google Scholar] [CrossRef]
- Stathaki, M. , Stamatiou, M. E., Magioris, G., Simantaris, S., Syroigos, N. et al. The role of kisspeptin system in cancer biology. Crit Rev Oncol Hematol 2019, 142, 130–140. [Google Scholar] [CrossRef]
- Loosen, S.V. , Luedde, M. , Lurie, G., Spehlmann, M., Paffenholz, P., Ulmer T.F. et al. Serum levels of kisspeptin are elevated in patients with pancreatic cancer. Disease Markers 2019, 5603474. [Google Scholar] [CrossRef]
- Xoxakos, I. , Petraki, C., Msaouel, P., Armakolas, A., Grigorakis, A., Stefanakis, S., Koutsilieris, M. Expression of Kisspeptin (KISS1) and its receptor GPR54 (KISS1R) in prostate cancer. Anticancer Res 2020. 40(2), 709-718. [CrossRef]
- Banerjee Sr, Pamper, M.G. Clinical applications of gallium-68. Appl Radiat Isotopes, 2013, 76, 2-13. [CrossRef]
- Spang, P. , Herrmann, C., Roesch, F. Bifunctional gallium-68: past, present and future. Semin Nucl Med 2016, 46, 373-394. [CrossRef]
- Baranvai, Z. , Tircso, G. , Roesch, F. The use of the macrocylic chelator DOTA in radiochemical preparations. Eur J Inorg Chem 2020, 1, 36–56. [Google Scholar] [CrossRef]
- Rather, M.A. , Basha, S.H., Bhat, I.A., Sharma, N., Nadanpawar, P. Characterization, molecular docking, dynamics simulation and metadynamics of kisspeptin receptor with kisspeptin. In J Biol Macromol 2017, 101, 241-253. [CrossRef]
- Roseweir, A.K. , Millar, R. P. Kisspeptin antagonists. Adv Exp Med Biol 2013, 784, 159–186. [Google Scholar] [CrossRef]
- Paquier, J. , Kamech, et al. Molecular evolution of GPCRS: kisspeptin/kisspeptin receptors. J Mol Endorcinol 2014, 52, 3. [CrossRef]
- Jain, A. , Chakraborty, S., Sharma, H.D., Dash, A. A systematic comparative evaluation of [68Ga]Ga-labelled RGD peptides conjugated with different chelators. Nucl Med Mol Imaging 2018, 52, 152-134. [CrossRef]
- EDQM. European pharmacopoeia 8.0. Radiopharmaceutical preparation; gallium octreotide. European directorate for the quality Medicines, Strasbourg pp 1061-1064.
- Asami, T. , Nishizawa, N. , Ishibashi, Y., Nishibori, K., Nakayama, M. et al. Serum stability of selected decapeptide agonists of KISS1R using pseudopeptides. Bioorganic Med Chem Lett 2012, 22, 6391–6396. [Google Scholar] [CrossRef]
- Robinson, A.B. , Mckerrow, J.H., Cary, P. Controlled deamidation of peptides and proteins: an experimental hazard and a possible biological timer. PNAS 1970, 66, 753-757. [CrossRef]
- Robinson, N.E. , Robinson, Z.W., Robinson, B.R., Robinson, A.L., Robinson, J.A., Robinson, M.L., Robinson, A.B. Structure-dependant nonenzymatic deamidation of glutaminyl and asparaginyl pentapeptides. J Pept Res 2004, 63, 426-436. [CrossRef]
- Asami, T. , Nischizawa, N., Matsui, H., Nishibori, K., Ishibashi, Y., Horikoshi, Y., Nakayama, M., Mastumoto H et al. Design, synthesis and biological evaluation of novel investigational nonpeptide KISS1R agonists with testosterone-suppressive activity. J Med Chem 2013, 56, 8298-8307. [CrossRef]
- Asami, T. , Nischizawa, N. , Ishibashi, Y., Nishibori, K., Nakayama, M., Horikoshi, Y., Matsumoto, S.I., Yamaguchi, M et al. Serum stability of selected decapeptide agonists of KISS1R using pseudopeptides. Bioorg Med Chem Lett 2012, 22, 6391–6396. [Google Scholar] [CrossRef]
- Newton, C.L. , Anderson, R. C., Katz, A.A., Millar, R.P. Loss of function mutations in the human luteinizing hormone receptor predominantly cause intracellular retention. Endorinol 2016, 157, 4364–4377. [Google Scholar] [CrossRef]
- Hanyroup, S. , Anderson, R.C., Nataraja, S. Yu, H.N., Millar, R.P., Newton, C.L. Rescue of cell surface expression and signalling of mutant follicle-stimulating hormone receptors. Endocrinol 2021, 162,12. [CrossRef]
- Ebenhan, T. , Chadwick, N., Sathekge, M.M., Govender, P., Kruger, T., Marjanovic-Painter, B., Zeevaart, J.R., Peptide synthesis, characterization and 68Ga-radiolabelling of NOTA-conjugated ubiquicidin fragments for prospective infection imaging with PET/CT. Nucl Med Biol 2014, 41, 390-400. [CrossRef]
- Mdlophane, A.H. , Ebenhan, T., Marjanovic-Painter B, Govender, T., Sathekge, M.M., Zeevaart, J.R. Comaprison of DOTA and NODAGA as chelates for 68Ga-laebelled CDP1 as novel infection PET imaging agents. J Radioanal and Nucl Chem 2019, 322, 629-638. [CrossRef]
- Sinnes, J. , Nagel, J., Roesch, F. AAZTA5/AAZTA5-TOC: synthesis and radiochemical evaluation with 68Ga, 44Sc and 177Lu. EJNMMI Radiopharm & Radiochem 2019, 4, 18. [CrossRef]
- De Blois, E. , Sze, C.E., Naidoo, C., Prince, D., Krennig, E.P., Breeman, W.A.P. Characteristics of SnO3 based 68Ge/68Ga generator and aspects of radiolabeling DOTA-peptides. Appl Radiat Isot 2011, 69, 308-315. [CrossRef]
- Jain, A. , Kameswaran, M. , Pandey, U., Sarma, H.D., Dash, A. 68Ga-labelled erlotinib: a novel PET probe for imaging EGRF over-expressing tumours. Bioorgan Med Chem Lett 2017, 27, 4552–4557. [Google Scholar] [CrossRef]
- Shi, S. , Zhang, L., Wu, Z., Zhang, A., Hong, H., Choi, S.R., Zhu, L., Kung, H.F. [68Ga]Ga-HBED-CC-DiAsp: a new renal function imaging agent. Nucl Med Biol 2020, 82-83, 17-24. [CrossRef]
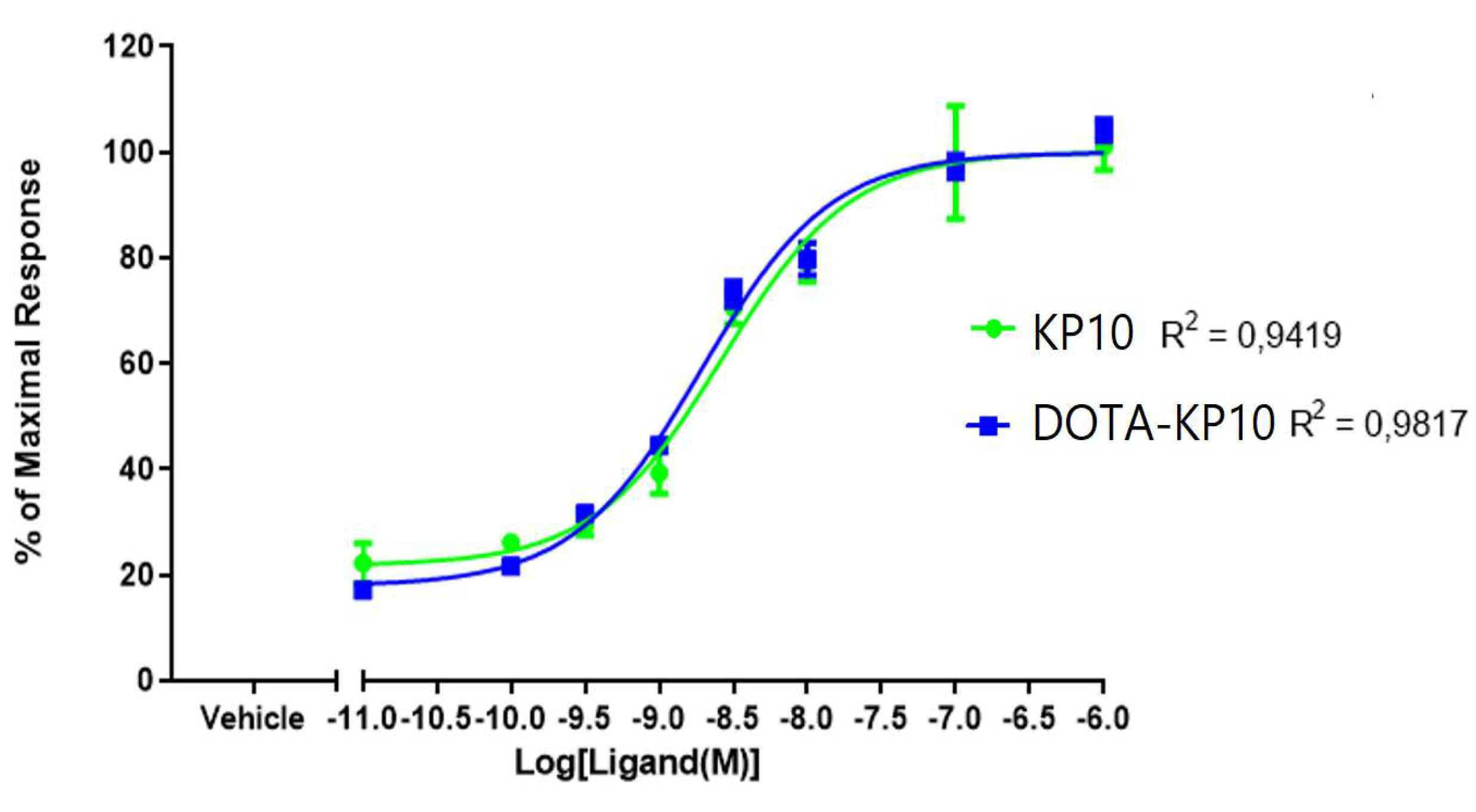
| Ligand | EC50 Mean nM ± SEM (N=4) |
|---|---|
| KP10 | 1.87 ± 0.095 |
| DOTA-KP-10 | 3.99 ± 0.075 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




