Submitted:
19 April 2024
Posted:
19 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Protein Concentration Assay
2.3. Western Blotting
2.4. Growth Studies
2.5. FACS Analysis
2.6. Metabolic Activity Using MTT Assay
2.7. Apoptosis Studies
2.8. Microscopy (Bright Field, Fluorescence and Live)
2.9. Statistical Analysis
3. Results
3.1. Cultured OPCs
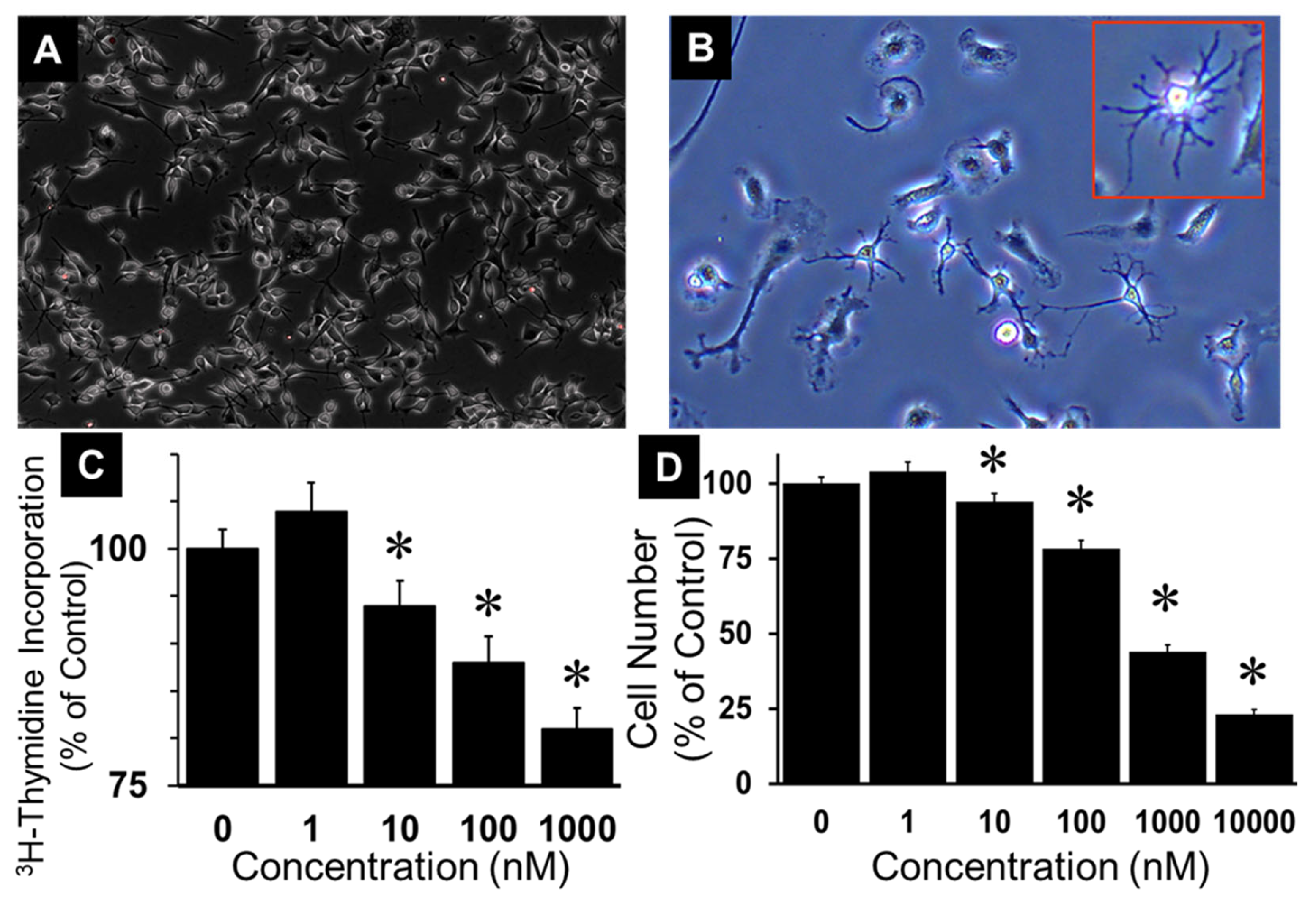
3.2. 2ME Inhibits Oli-Neu Cell Growth
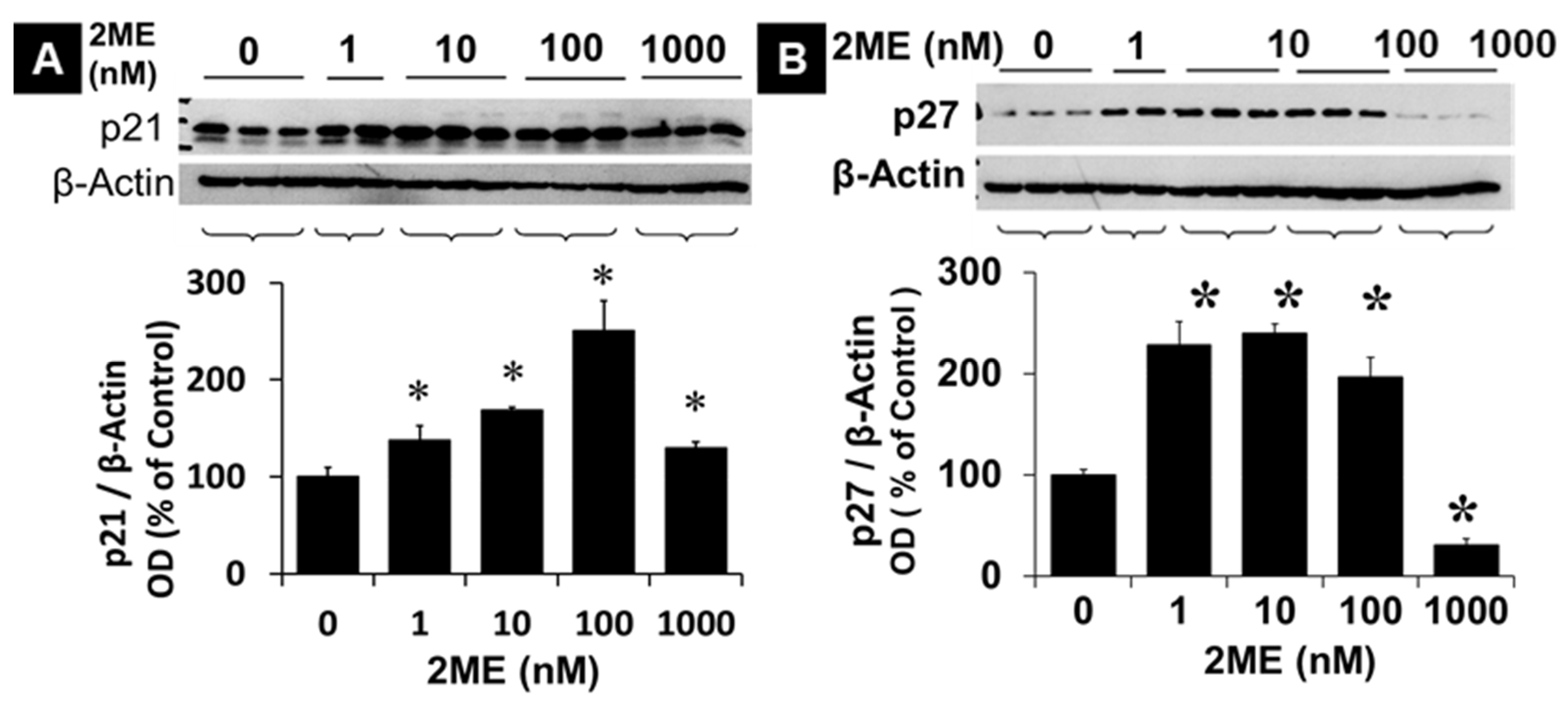
3.3. 2ME Modulates p21 and p27 Expression
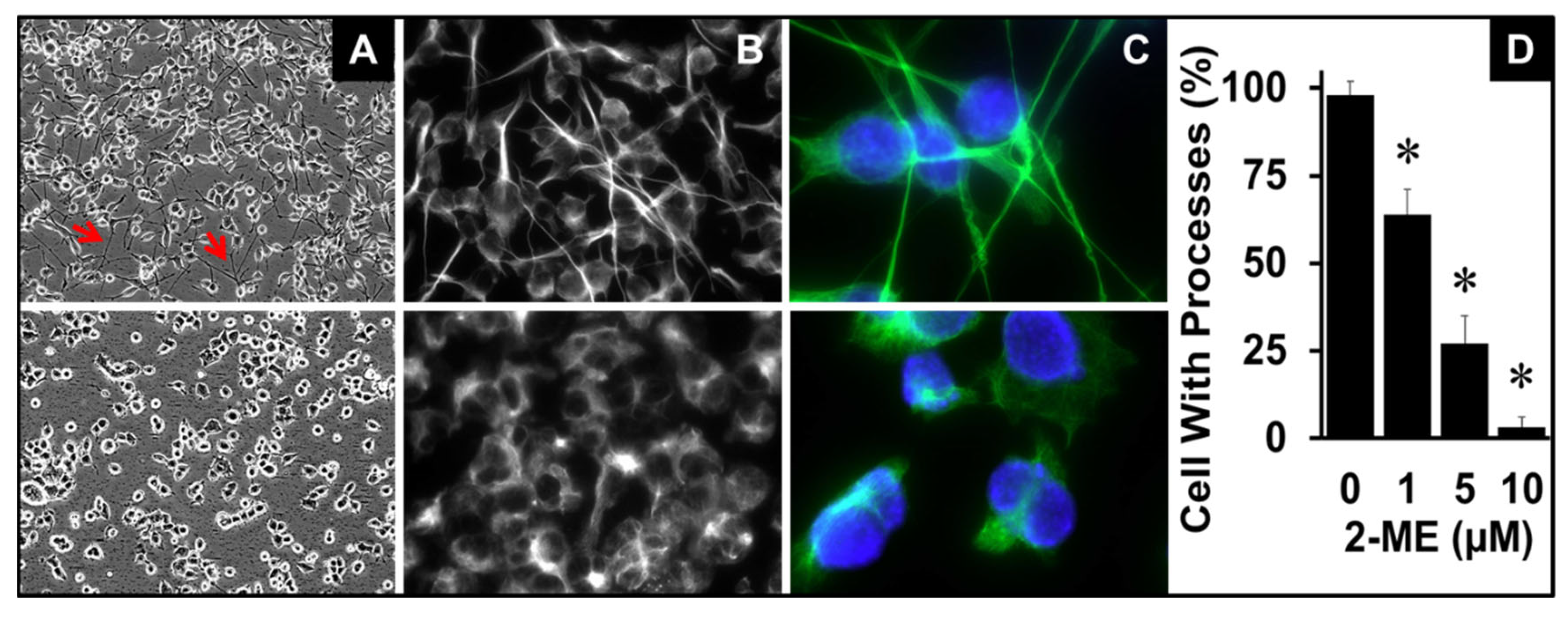
3.4. 2-ME Modulates OPC Morphology
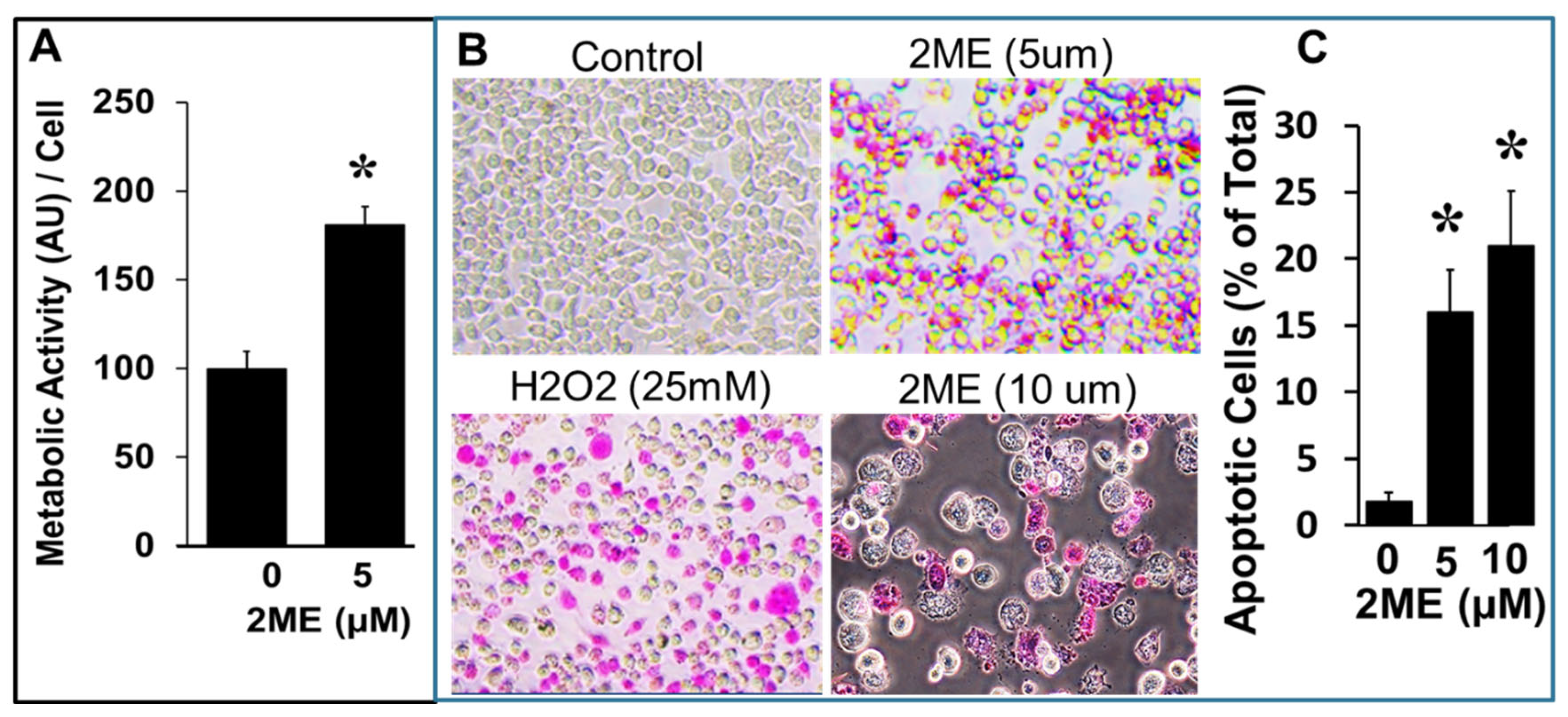
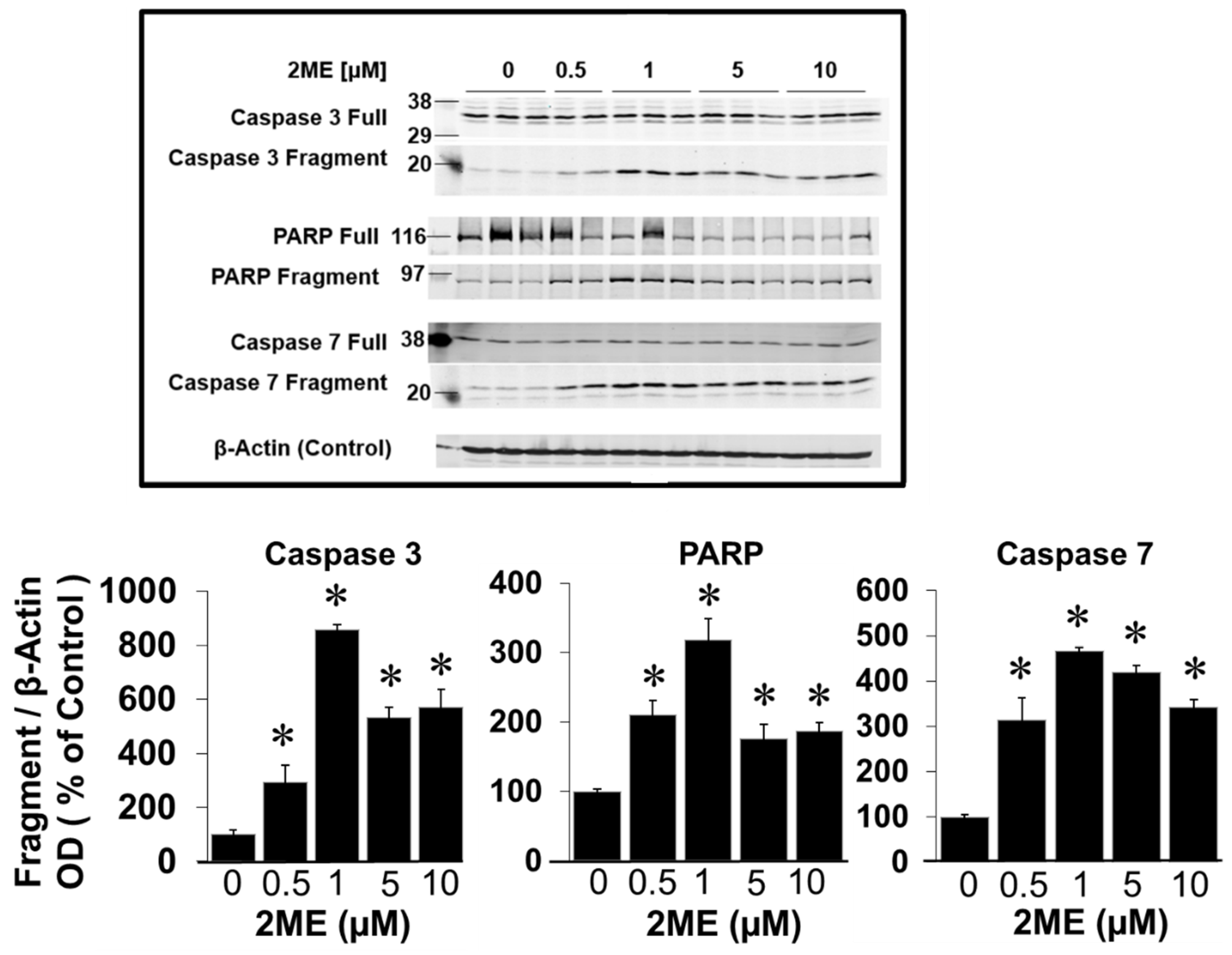
3.5. 2ME Treatment Induces Apoptosis
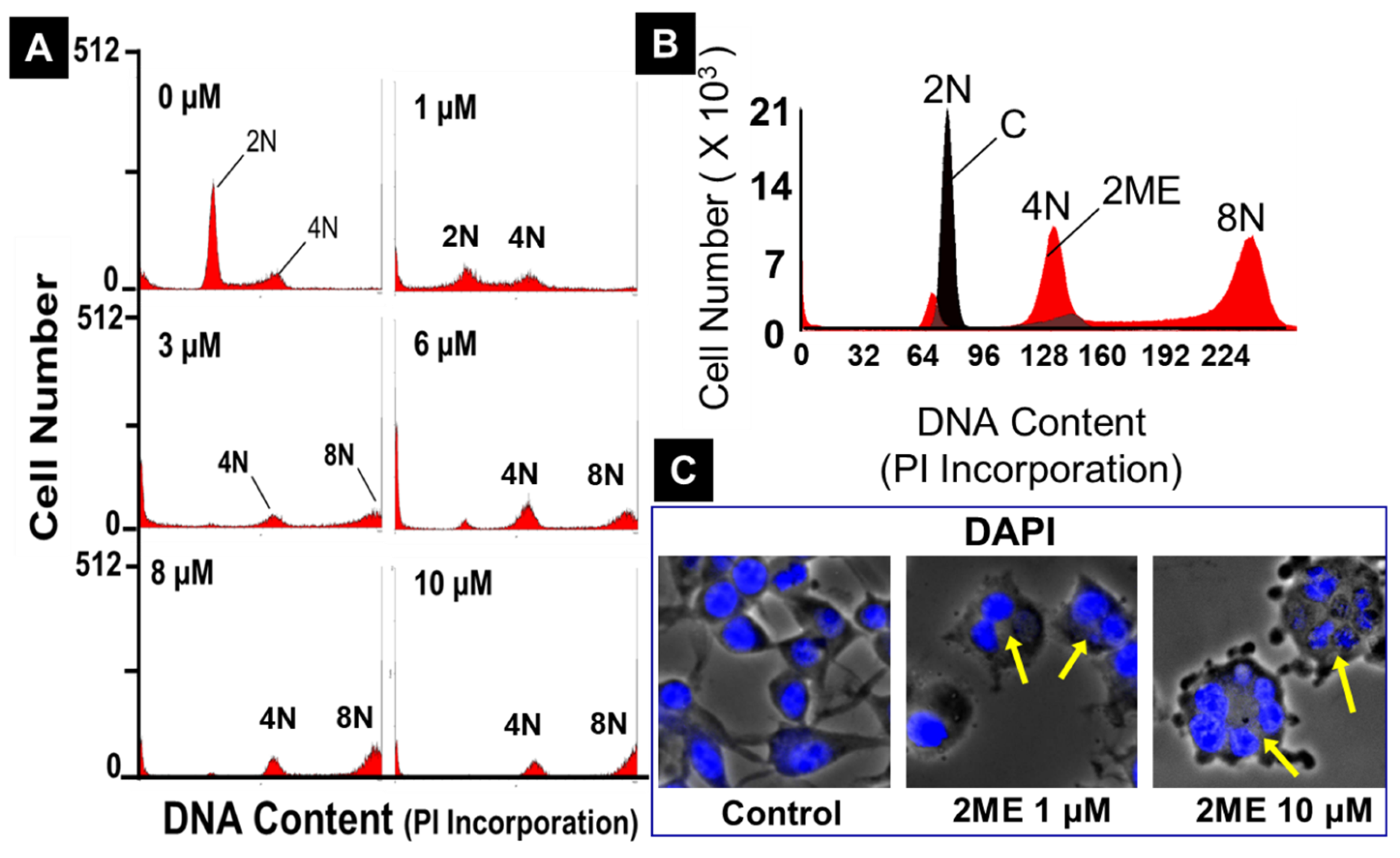
3.6. 2ME Treatment Induces Endoreduplication
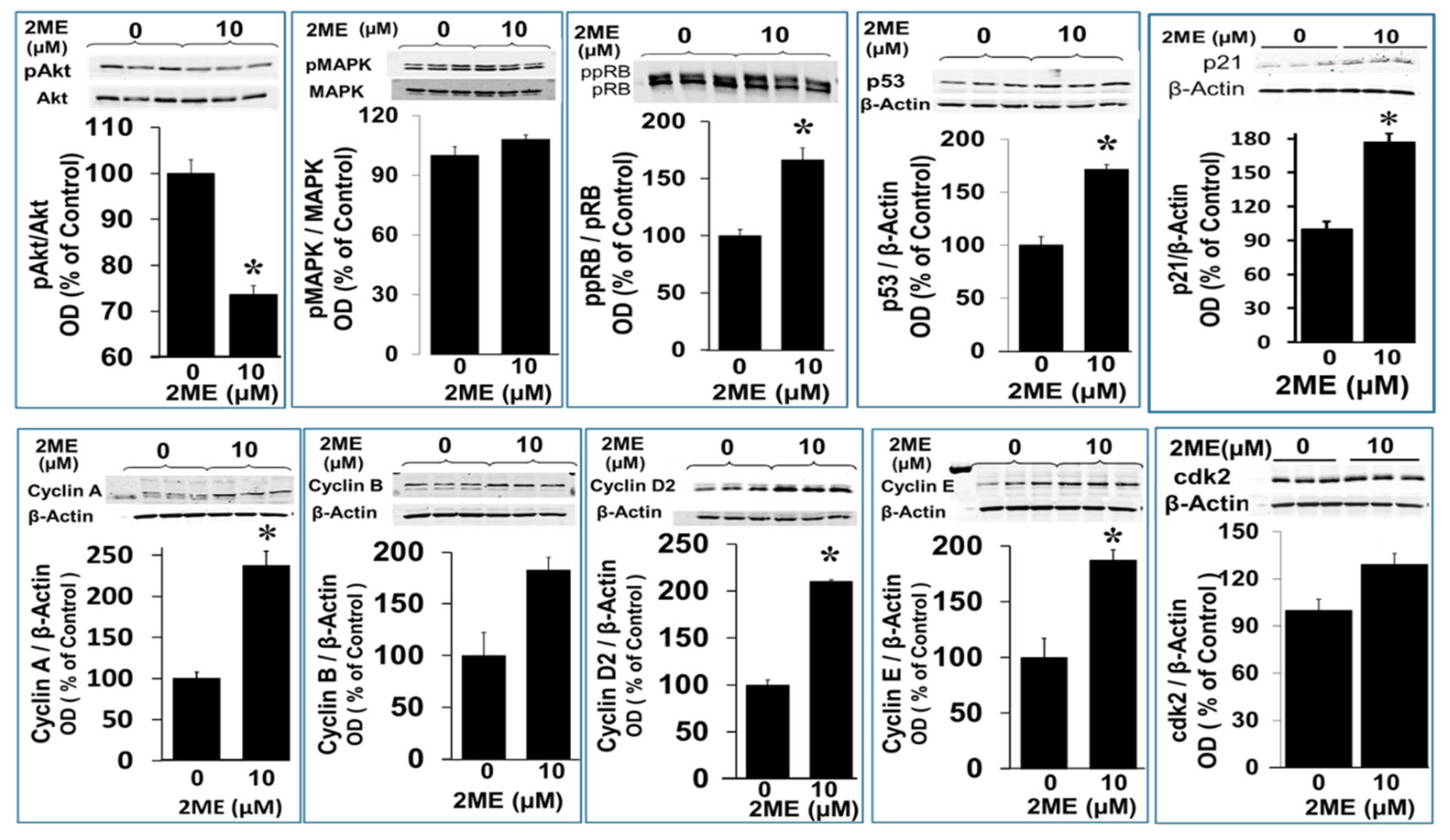
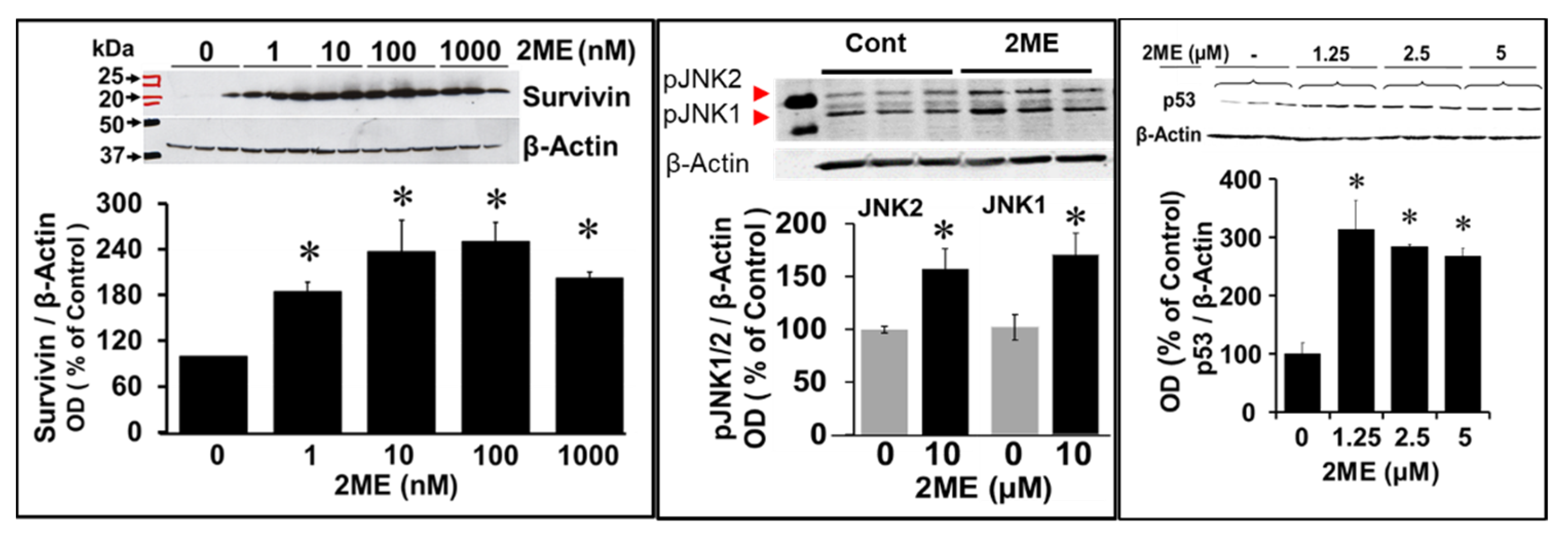
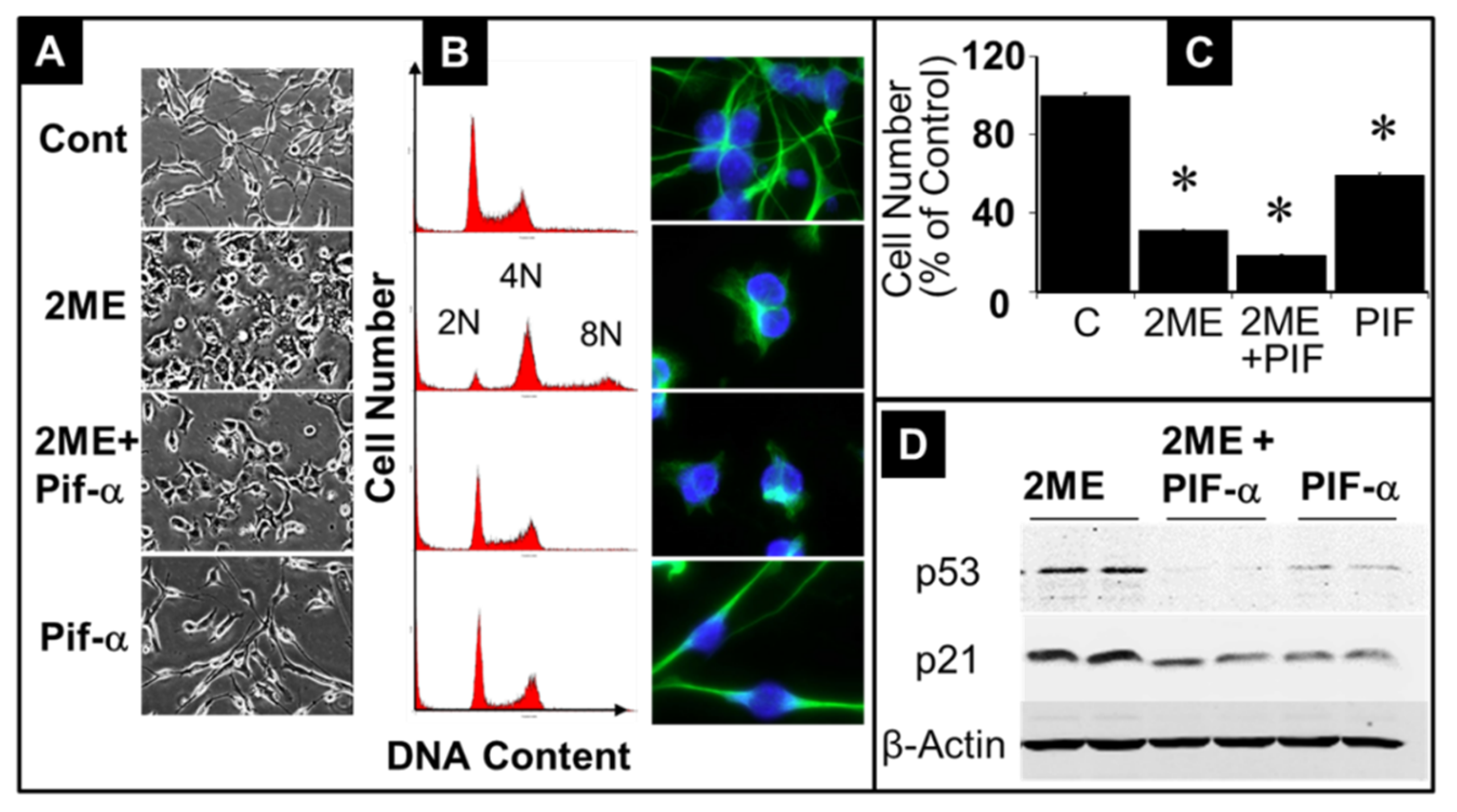
3.7. Mechanism Underlying 2ME-Induced Endoreduplication
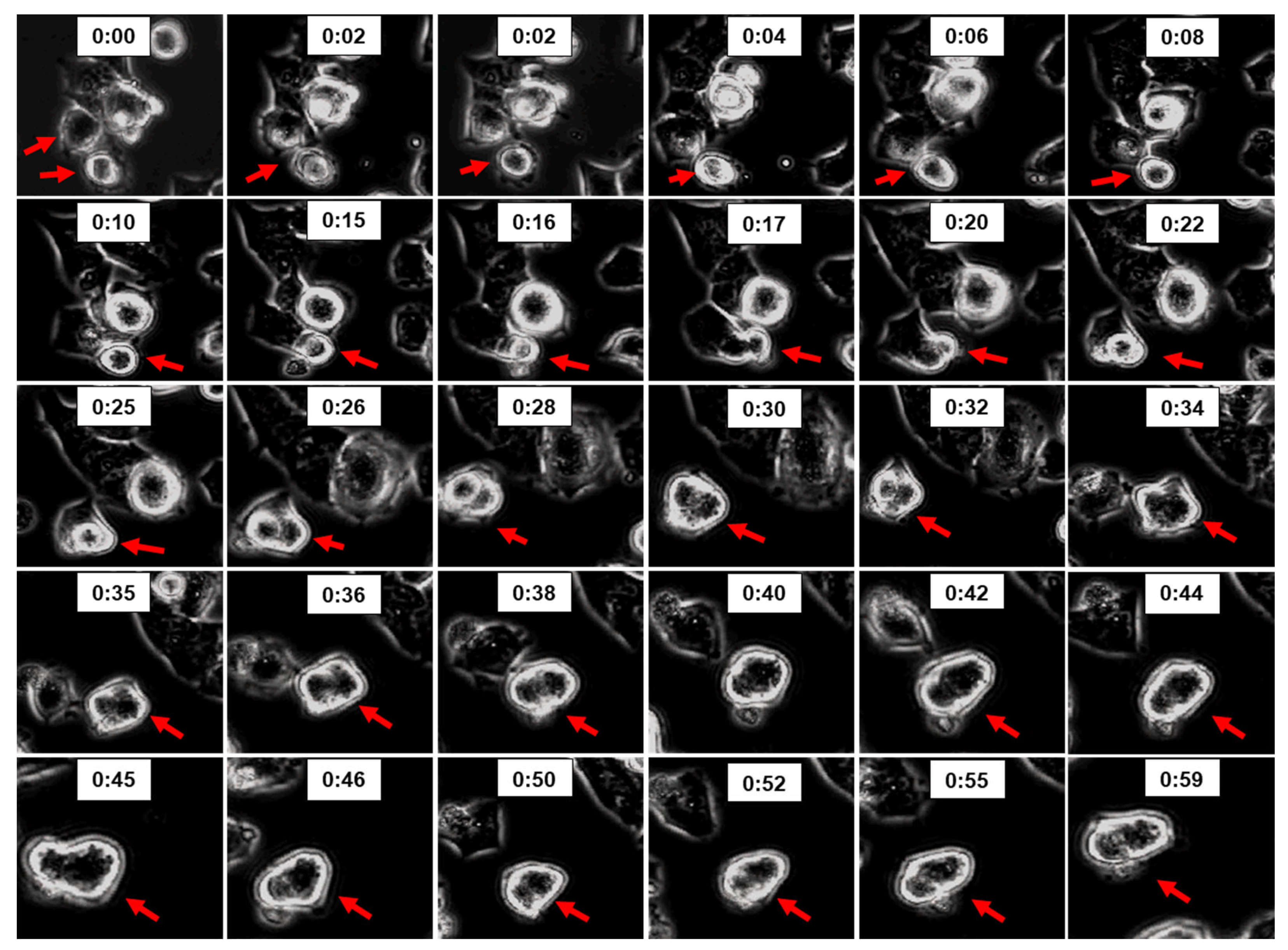
3.8. 2ME Trigger Cell Fusion in OPCs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kipp, M., Oligodendrocyte Physiology and Pathology Function. Cells, 2020. 9(9). [CrossRef]
- Bou Zerdan, M. and H.I. Assi, Oligodendroglioma: A Review of Management and Pathways. Front Mol Neurosci, 2021. 14: p. 722396. [CrossRef]
- Wei, J., K. Gabrusiewicz, and A. Heimberger, The controversial role of microglia in malignant gliomas. Clin Dev Immunol, 2013. 2013: p. 285246. [CrossRef]
- Geribaldi-Doldán, N., et al., The Role of Microglia in Glioblastoma. Front Oncol, 2020. 10: p. 603495. [CrossRef]
- Dumas, A.A., et al., Microglia promote glioblastoma via mTOR-mediated immunosuppression of the tumour microenvironment. Embo j, 2020. 39(15): p. e103790.
- Khan, F., et al., Macrophages and microglia in glioblastoma: heterogeneity, plasticity, and therapy. J Clin Invest, 2023. 133(1). [CrossRef]
- Zamler, D.B. and J. Hu, Primitive Oligodendrocyte Precursor Cells Are Highly Susceptible to Gliomagenic Transformation. Cancer Res, 2023. 83(6): p. 807-808. [CrossRef]
- Weng, Q., et al., Single-Cell Transcriptomics Uncovers Glial Progenitor Diversity and Cell Fate Determinants during Development and Gliomagenesis. Cell Stem Cell, 2019. 24(5): p. 707-723.e8. [CrossRef]
- Dufour, A., et al., Modeling the dynamics of oligodendrocyte precursor cells and the genesis of gliomas. PLoS Comput Biol, 2018. 14(3): p. e1005977. [CrossRef]
- Galvao, R.P., et al., Transformation of quiescent adult oligodendrocyte precursor cells into malignant glioma through a multistep reactivation process. Proc Natl Acad Sci U S A, 2014. 111(40): p. E4214-23. [CrossRef]
- Wordeman, L. and J.J. Vicente, Microtubule Targeting Agents in Disease: Classic Drugs, Novel Roles. Cancers (Basel), 2021. 13(22). [CrossRef]
- Chen, J.G., et al., Gene expression and mitotic exit induced by microtubule-stabilizing drugs. Cancer Res, 2003. 63(22): p. 7891-9.
- Čermák, V., et al., Microtubule-targeting agents and their impact on cancer treatment. Eur J Cell Biol, 2020. 99(4): p. 151075.
- Krause, W., Resistance to anti-tubulin agents: From vinca alkaloids to epothilones. Cancer Drug Resist, 2019. 2(1): p. 82-106. [CrossRef]
- Dubey, R.K., B. Imthurn, and E.K. Jackson, 2-Methoxyestradiol: a potential treatment for multiple proliferative disorders. Endocrinology, 2007. 148(9): p. 4125-7. [CrossRef]
- Dubey, R.K. and E.K. Jackson, Potential vascular actions of 2-methoxyestradiol. Trends Endocrinol Metab, 2009. 20(8): p. 374-9. [CrossRef]
- Fukui, M. and B.T. Zhu, Mechanism of 2-methoxyestradiol-induced apoptosis and growth arrest in human breast cancer cells. Mol Carcinog, 2009. 48(1): p. 66-78. [CrossRef]
- Ray, G., et al., Modulation of cell-cycle regulatory signaling network by 2-methoxyestradiol in prostate cancer cells is mediated through multiple signal transduction pathways. Biochemistry, 2006. 45(11): p. 3703-13. [CrossRef]
- Choi, H.J. and B.T. Zhu, Critical role of cyclin B1/Cdc2 up-regulation in the induction of mitotic prometaphase arrest in human breast cancer cells treated with 2-methoxyestradiol. Biochim Biophys Acta, 2012. 1823(8): p. 1306-15.
- Lambert, C., et al., 2-methoxyestradiol induces caspase-independent, mitochondria-centered apoptosis in DS-sarcoma cells. Int J Cancer, 2004. 108(4): p. 493-501. [CrossRef]
- Dahut, W.L., et al., Phase I clinical trial of oral 2-methoxyestradiol, an antiangiogenic and apoptotic agent, in patients with solid tumors. Cancer Biol Ther, 2006. 5(1): p. 22-7. [CrossRef]
- Kang, S.H., et al., Antitumor effect of 2-methoxyestradiol in a rat orthotopic brain tumor model. Cancer Res, 2006. 66(24): p. 11991-7.
- Jung, M., et al., Lines of murine oligodendroglial precursor cells immortalized by an activated neu tyrosine kinase show distinct degrees of interaction with axons in vitro and in vivo. Eur J Neurosci, 1995. 7(6): p. 1245-65. [CrossRef]
- Motwani, M., X. Li, and G.K. Schwartz, Flavopiridol, a cyclin-dependent kinase inhibitor, prevents spindle inhibitor-induced endoreduplication in human cancer cells. Clin Cancer Res, 2000. 6(3): p. 924-32.
- Rizzoni, M. and F. Palitti, Regulatory mechanism of cell division. I. Colchicine-induced endoreduplication. Exp Cell Res, 1973. 77(1): p. 450-8. [CrossRef]
- Cahill, D.P., et al., Genetic instability and darwinian selection in tumours. Trends Cell Biol, 1999. 9(12): p. M57-60. [CrossRef]
- Sandberg, A.A., Chromosome markers and progression in bladder cancer. Cancer Res, 1977. 37(8 Pt 2): p. 2950-6.
- Rabinovitch, P.S., et al., Progression to cancer in Barrett's esophagus is associated with genomic instability. Lab Invest, 1989. 60(1): p. 65-71.
- Li, R., et al., Aneuploidy vs. gene mutation hypothesis of cancer: recent study claims mutation but is found to support aneuploidy. Proc Natl Acad Sci U S A, 2000. 97(7): p. 3236-41.
- Schutte, B., et al., Retrospective analysis of the prognostic significance of DNA content and proliferative activity in large bowel carcinoma. Cancer Res, 1987. 47(20): p. 5494-6.
- Stephenson, R.A., et al., Flow cytometry of prostate cancer: relationship of DNA content to survival. Cancer Res, 1987. 47(9): p. 2504-7.
- Braeuninger, S., et al., Short incubation with 2-methoxyestradiol kills malignant glioma cells independent of death receptor 5 upregulation. Clin Neuropathol, 2005. 24(4): p. 175-83.
- Chamaon, K., et al., Micromolar concentrations of 2-methoxyestradiol kill glioma cells by an apoptotic mechanism, without destroying their microtubule cytoskeleton. J Neurooncol, 2005. 72(1): p. 11-6. [CrossRef]
- D'Amato, R.J., et al., 2-Methoxyestradiol, an endogenous mammalian metabolite, inhibits tubulin polymerization by interacting at the colchicine site. Proc Natl Acad Sci U S A, 1994. 91(9): p. 3964-8. [CrossRef]
- Buckner, J.C., et al., Radiation plus Procarbazine, CCNU, and Vincristine in Low-Grade Glioma. N Engl J Med, 2016. 374(14): p. 1344-55. [CrossRef]
- Gui, Y. and X.L. Zheng, 2-methoxyestradiol induces cell cycle arrest and mitotic cell apoptosis in human vascular smooth muscle cells. Hypertension, 2006. 47(2): p. 271-80. [CrossRef]
- Gui, Y., et al., Endoreduplication of human smooth muscle cells induced by 2-methoxyestradiol: a role for cyclin-dependent kinase 2. Am J Physiol Heart Circ Physiol, 2007. 292(3): p. H1313-20. [CrossRef]
- Ganguly, A. and F. Cabral, New insights into mechanisms of resistance to microtubule inhibitors. Biochim Biophys Acta, 2011. 1816(2): p. 164-71. [CrossRef]
- Khan, S.H. and G.M. Wahl, p53 and pRb prevent rereplication in response to microtubule inhibitors by mediating a reversible G1 arrest. Cancer Res, 1998. 58(3): p. 396-401.
- Lanni, J.S. and T. Jacks, Characterization of the p53-dependent postmitotic checkpoint following spindle disruption. Mol Cell Biol, 1998. 18(2): p. 1055-64. [CrossRef]
- Minn, A.J., L.H. Boise, and C.B. Thompson, Expression of Bcl-xL and loss of p53 can cooperate to overcome a cell cycle checkpoint induced by mitotic spindle damage. Genes Dev, 1996. 10(20): p. 2621-31.
- Stewart, Z.A., S.D. Leach, and J.A. Pietenpol, p21(Waf1/Cip1) inhibition of cyclin E/Cdk2 activity prevents endoreduplication after mitotic spindle disruption. Mol Cell Biol, 1999. 19(1): p. 205-15.
- Cross, S.M., et al., A p53-dependent mouse spindle checkpoint. Science, 1995. 267(5202): p. 1353-6. [CrossRef]
- Di Leonardo, A., et al., DNA rereplication in the presence of mitotic spindle inhibitors in human and mouse fibroblasts lacking either p53 or pRb function. Cancer Res, 1997. 57(6): p. 1013-9.
- Shamloo, B. and S. Usluer, p21 in Cancer Research. Cancers (Basel), 2019. 11(8). [CrossRef]
- Bansal, R., et al., S-phase entry of oligodendrocyte lineage cells is associated with increased levels of p21Cip1. J Neurosci Res, 2005. 80(3): p. 360-8. [CrossRef]
- Zeng, J., et al., Cyclin E-induced replicative stress drives p53-dependent whole-genome duplication. Cell, 2023. 186(3): p. 528-542.e14. [CrossRef]
- Rosa, J., et al., Survivin modulates microtubule dynamics and nucleation throughout the cell cycle. Mol Biol Cell, 2006. 17(3): p. 1483-93. [CrossRef]
- Beltrami, E., et al., Acute ablation of survivin uncovers p53-dependent mitotic checkpoint functions and control of mitochondrial apoptosis. J Biol Chem, 2004. 279(3): p. 2077-84. [CrossRef]
- Do, M., et al., Survivin protects fused cancer cells from cell death. BMB Rep, 2017. 50(7): p. 361-366. [CrossRef]
- Moulder, D.E., et al., The Roles of p53 in Mitochondrial Dynamics and Cancer Metabolism: The Pendulum between Survival and Death in Breast Cancer? Cancers (Basel), 2018. 10(6).
- Floriddia, E.M., et al., Distinct oligodendrocyte populations have spatial preference and different responses to spinal cord injury. Nat Commun, 2020. 11(1): p. 5860.
- Peters, J.M., The anaphase-promoting complex: proteolysis in mitosis and beyond. Mol Cell, 2002. 9(5): p. 931-43. [CrossRef]
- Niculescu, A.B., 3rd, et al., Effects of p21(Cip1/Waf1) at both the G1/S and the G2/M cell cycle transitions: pRb is a critical determinant in blocking DNA replication and in preventing endoreduplication. Mol Cell Biol, 1998. 18(1): p. 629-43.
- Pagano, M., et al., Cyclin A is required at two points in the human cell cycle. Embo j, 1992. 11(3): p. 961-71. [CrossRef]
- Li, Y., et al., Cell cycle expression and p53 regulation of the cyclin-dependent kinase inhibitor p21. Oncogene, 1994. 9(8): p. 2261-8.
- Eichenlaub-Ritter, U., et al., 2-methoxyestradiol induces spindle aberrations, chromosome congression failure, and nondisjunction in mouse oocytes. Biol Reprod, 2007. 76(5): p. 784-93. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



