Submitted:
19 April 2024
Posted:
19 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patient Recruitment
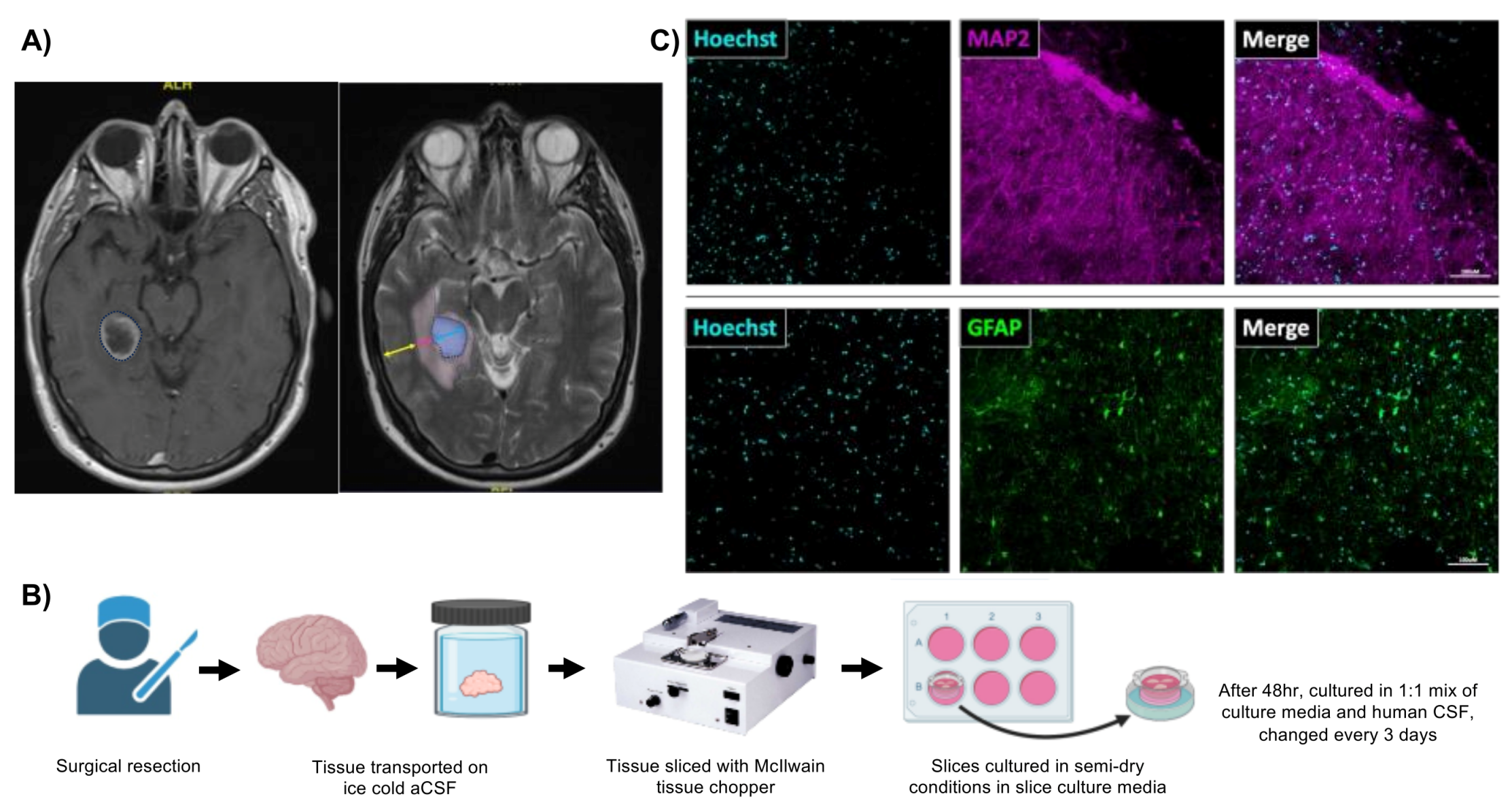
2.2. Collection of Brain Tissue, Blood, and Cerebrospinal Fluid
2.3. Human Brain Slice Culture
2.4. Tissue Clearing and Staining
2.5. Cell Culture
2.6. PBMC Isolation, CD4+ T-Cell Purification, and MDM Differentiation
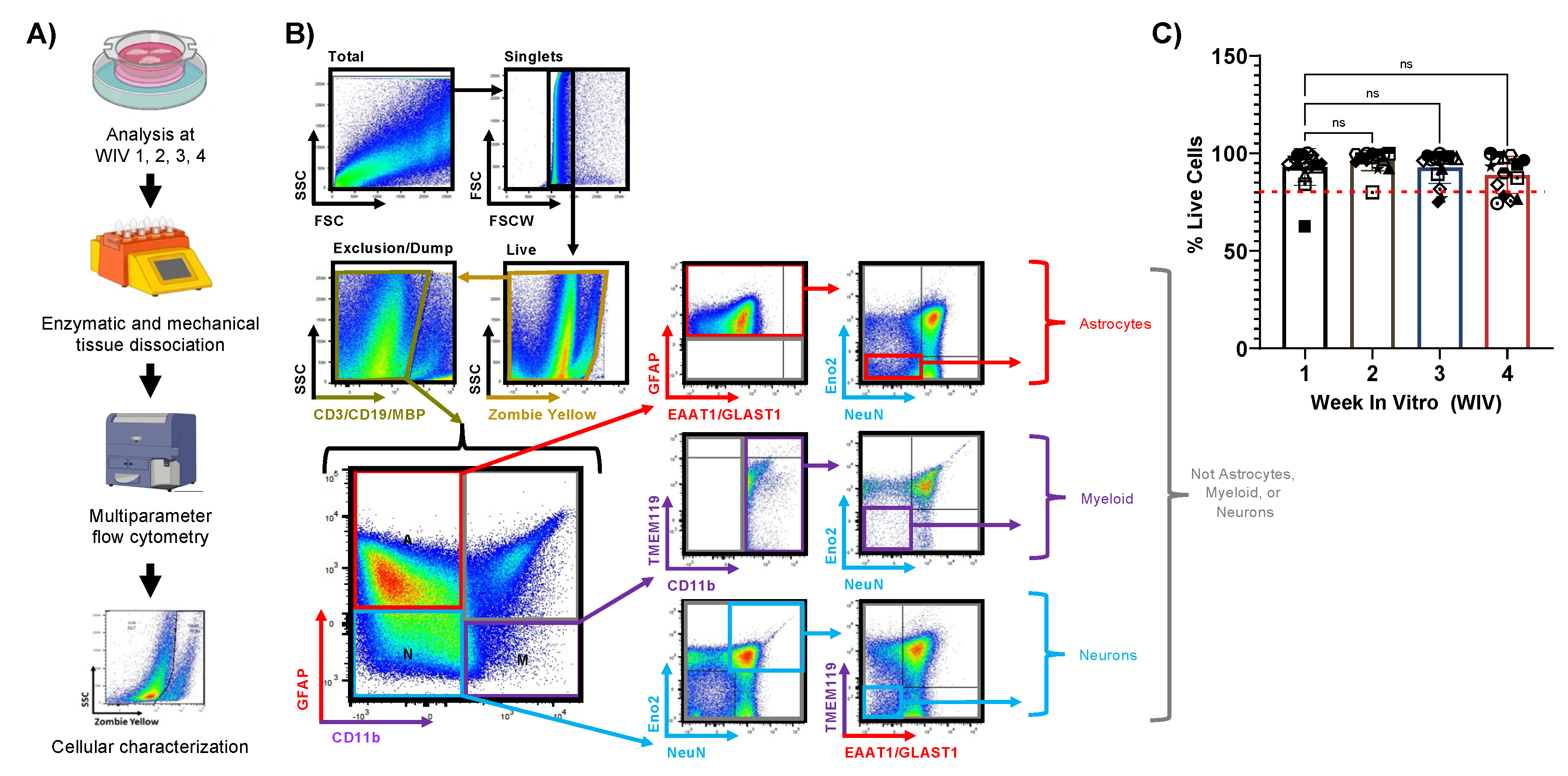
2.7. Mechanical and Enzymatic Dissociation of Tissue Slices
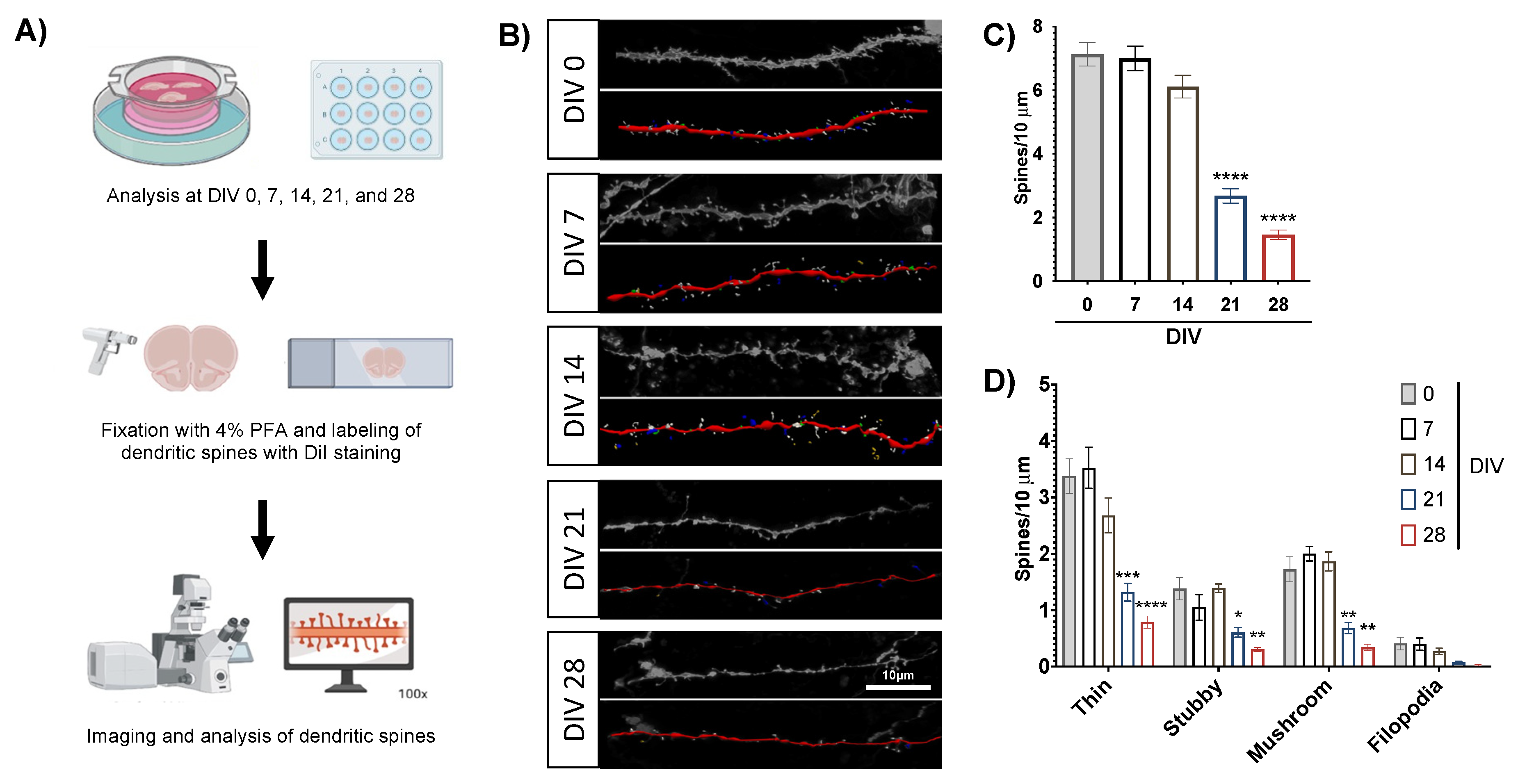
2.8. Dendritic Spine Staining and Analysis
2.9. Multielectrode Array (MEA) Electrophysiology
2.10. Cloning and Plasmids
2.11. Preparation of Virus Stocks
2.12. Infection of Producer Cells and Inoculation of Tissue Slices
2.13. Quantification of Spreading Infection by p24 alphaLISA
2.14. RNA Extraction, cDNA Synthesis, and qPCR from Tissue Slices
2.15. Flow Cytometry
2.16. Gating Strategy and Population Calculations
2.17. Experimental Design and Statistical Analysis
3. Results
3.1. Screening of Tissue Donors and Exclusion Criteria
| Age | N=17 |
|---|---|
| Mean (SD) | 57.5 (14.6) |
| Median | 58 |
| Range | 33, 82 |
| Sex, n (%) | |
| Female | 11 (64.7) |
| Male | 6 (35.3) |
| Tissue collected, n (%) | |
| Supratentorial neocortical parenchyma | 17 (100%) |
3.2. Preparing Human Brain Organotypic Slice Cultures
3.3. Adult Human Brain Tissue Slices are Viable Ex Vivo for at Least Four Weeks in Culture
3.4. Slice Cultures Maintain High Dendritic Spine Density with Mature Spine Morphologies
3.5. Multi-Electrode Array Recordings of Acute Brain Slices

3.6. Cell-Associated HIV-1 Infection of Ex Vivo Human Brain Slice Cultures
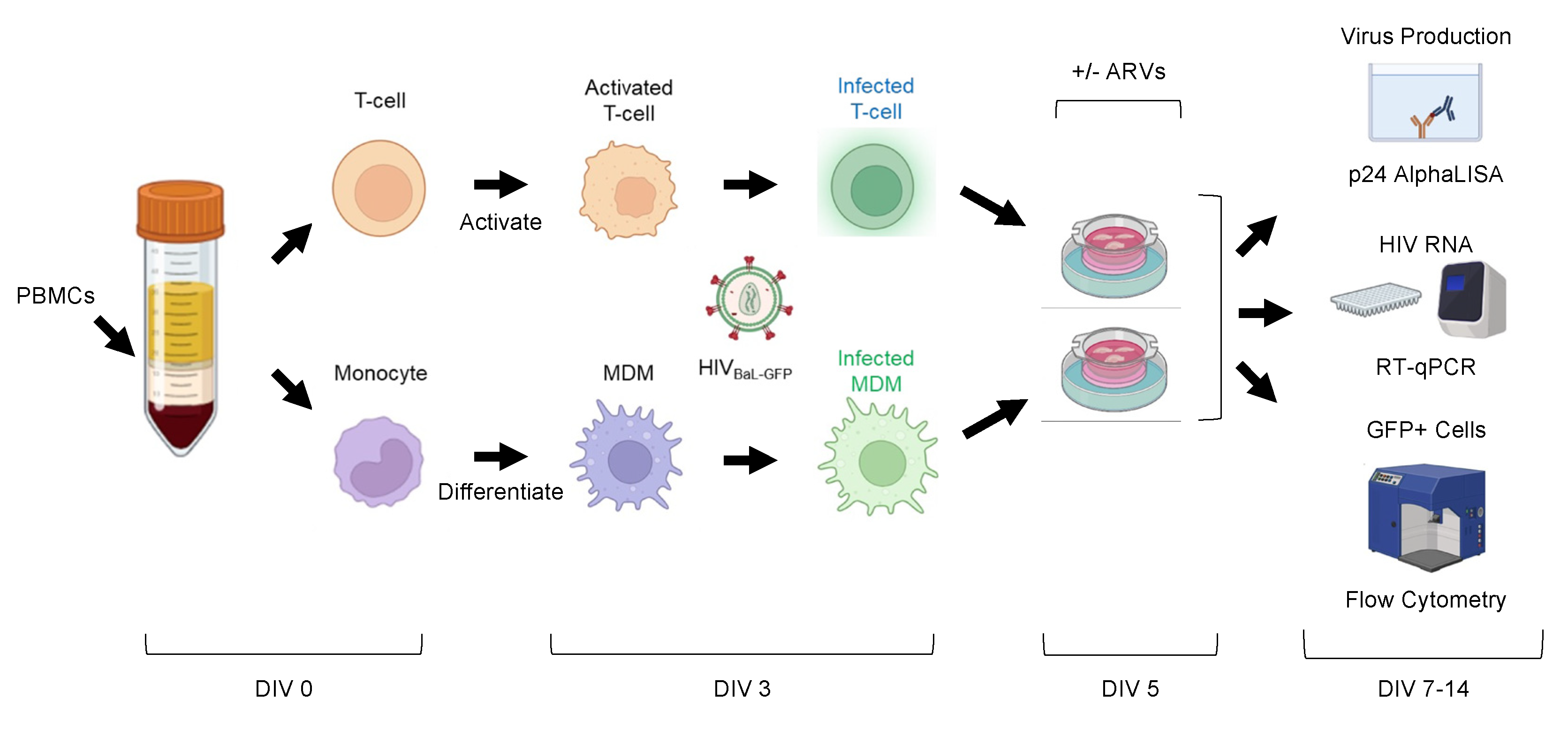
3.7. Human Brain Slices Inoculated with Infected MDMs and T-Cells can Support a HIV-1 Spreading Infection Ex Vivo
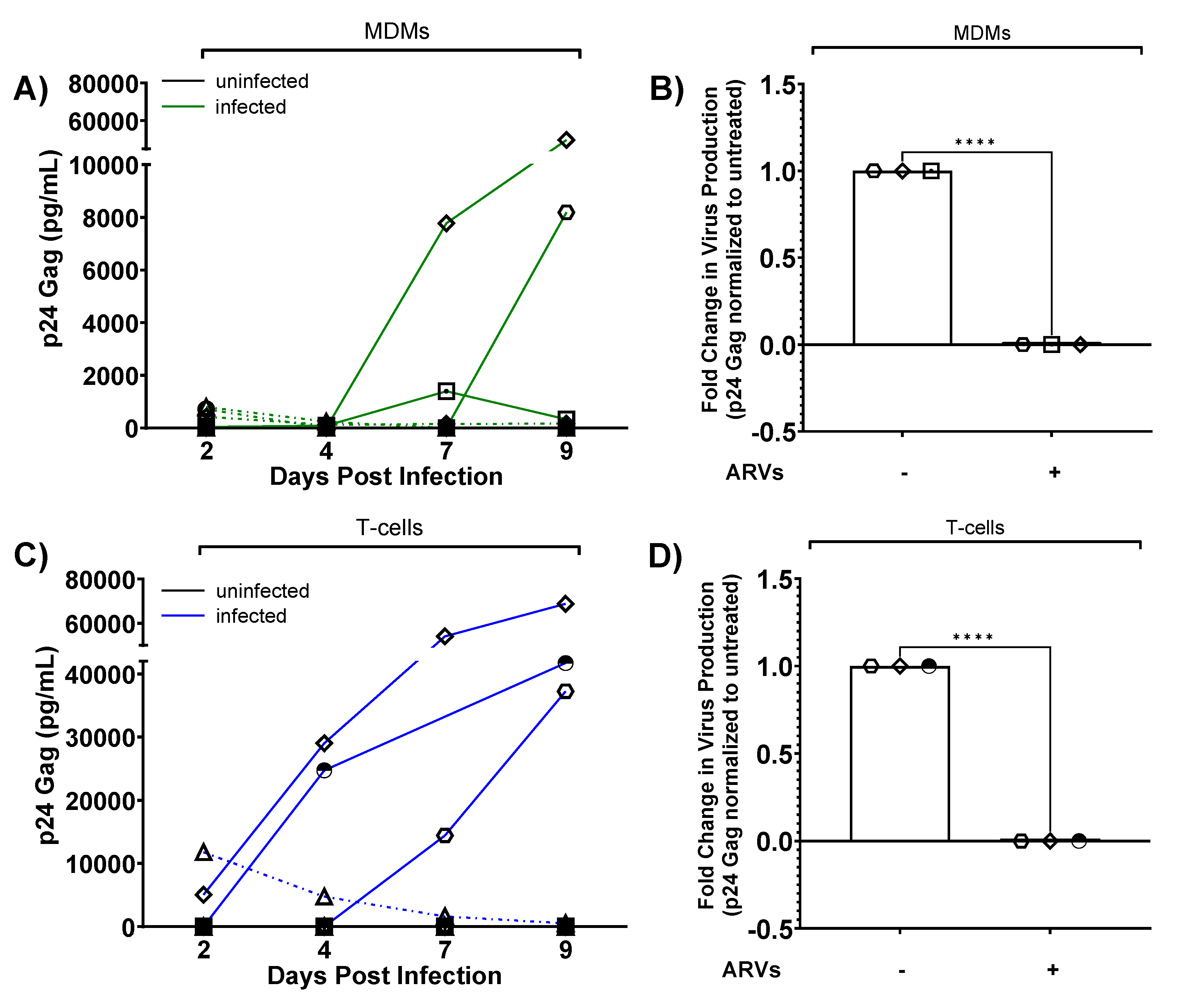
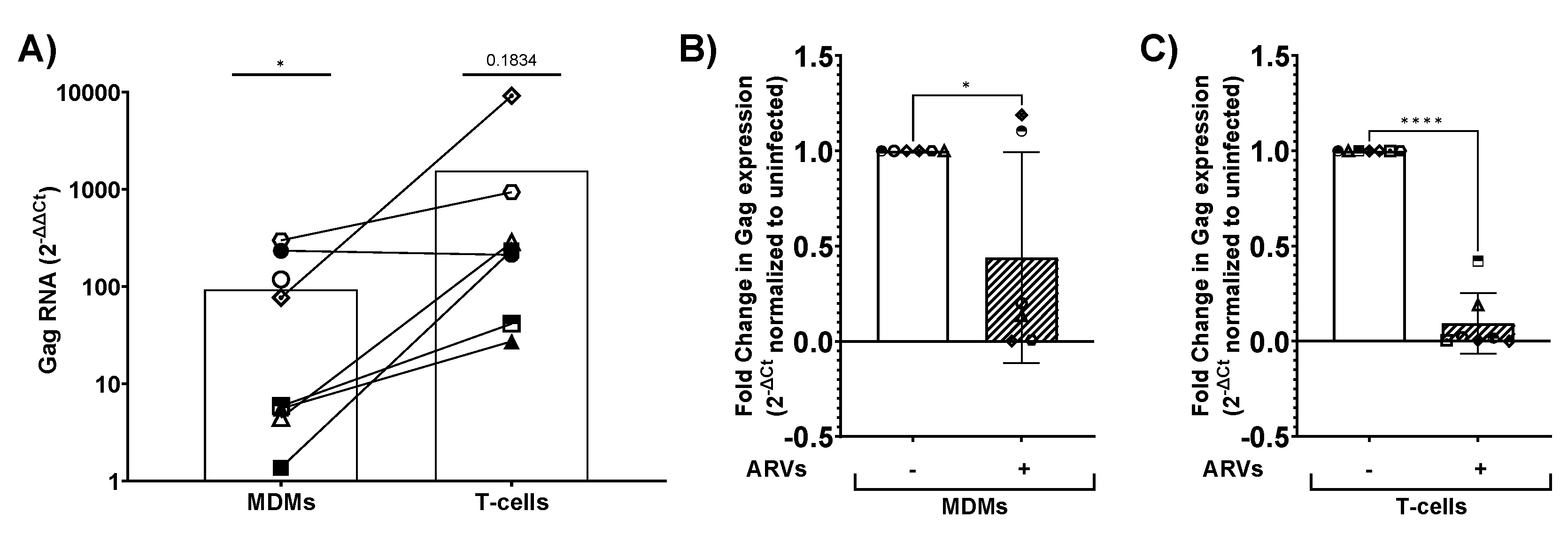
3.8. Infected Brain Slice Cultures Contain Viral RNA and can be Suppressed by ARVs
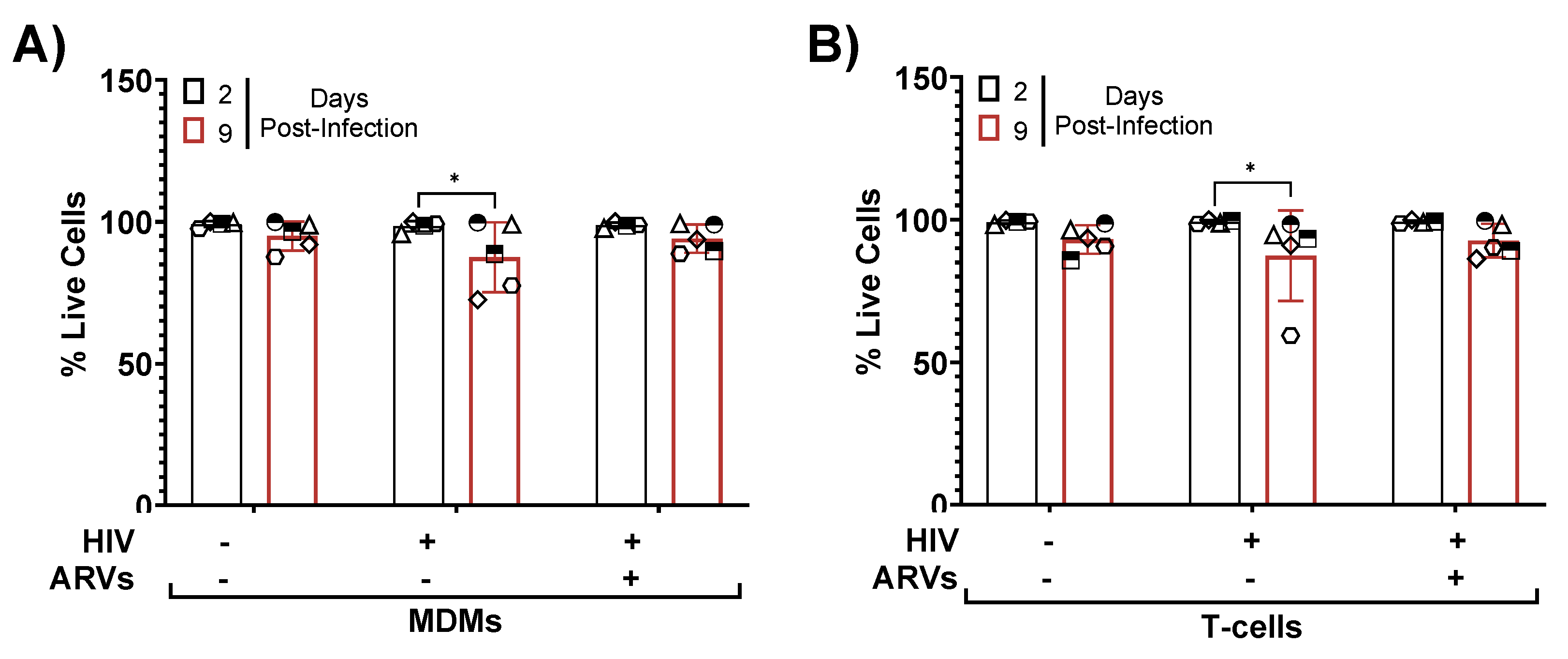
3.9. HIV-1 Infected Slices are Viable in Culture up to Nine Days Post-Infection
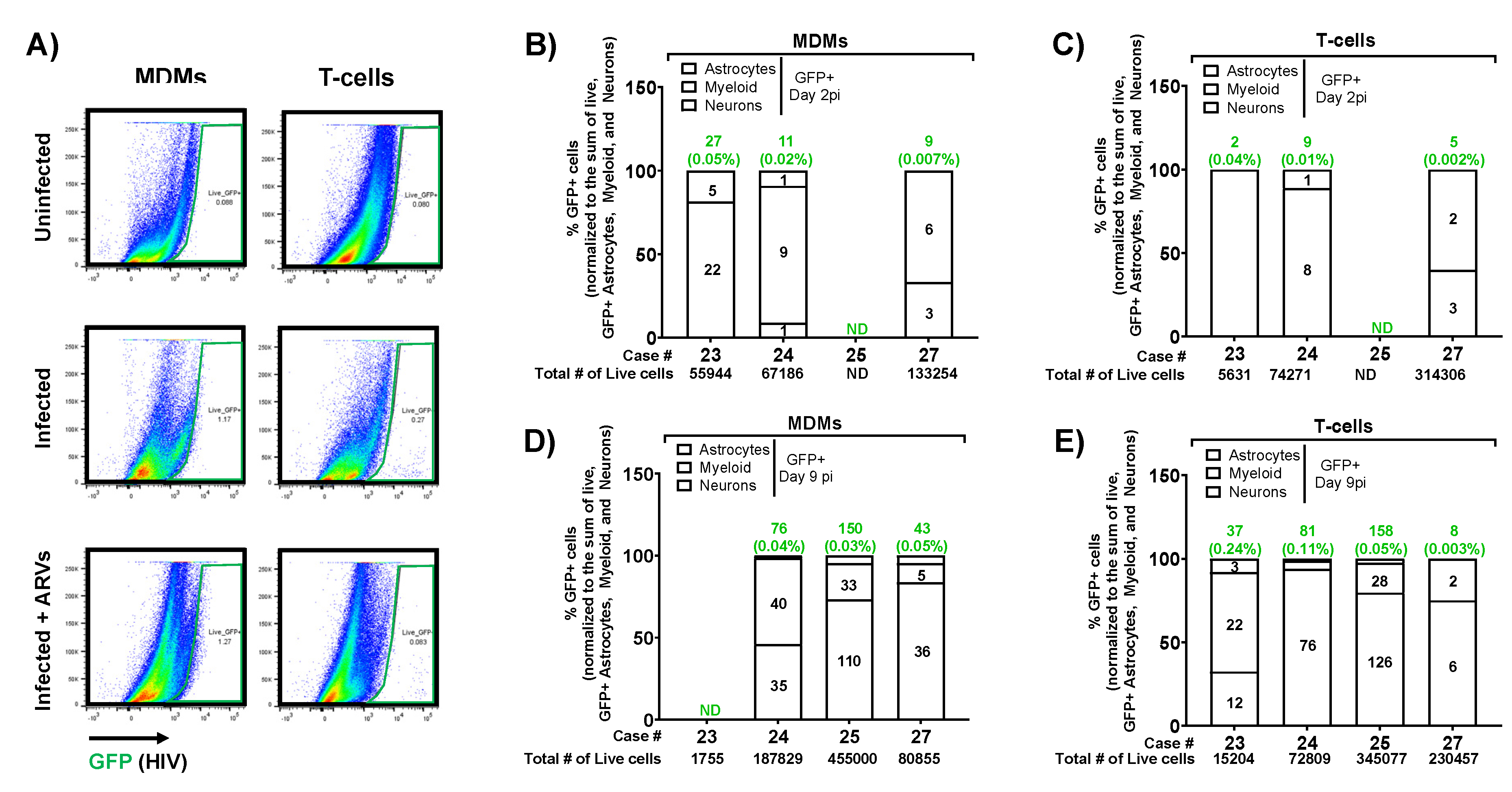
3.10. HIV-1 Infected, GFP+ Astrocytes and Myeloid Cells are Present in Human Brain Tissue Inoculated with Infected MDMs and T-Cells
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- van Schalkwyk, C.; Mahy, M.; Johnson, L.F.; Imai-Eaton, J.W. Updated Data and Methods for the 2023 UNAIDS HIV Estimates. J Acquir Immune Defic Syndr 2024, 95, e1-e4. [CrossRef]
- Ellis, R.J.; Marquine, M.J.; Kaul, M.; Fields, J.A.; Schlachetzki, J.C.M. Mechanisms underlying HIV-associated cognitive impairment and emerging therapies for its management. Nat Rev Neurol 2023, 19, 668-687. [CrossRef]
- Chan, P.; Brew, B.J. HIV associated neurocognitive disorders in the modern antiviral treatment era: Prevalence, characteristics, biomarkers, and effects of treatment. Curr HIV/AIDS Rep 2014, 11, 317-324. [CrossRef]
- Rosenthal, J.; Tyor, W. Aging, comorbidities, and the importance of finding biomarkers for HIV-associated neurocognitive disorders. J Neurovirol 2019, 25, 673-685. [CrossRef]
- Chan, P.; Valcour, V. Neurocognition and the Aging Brain in People With HIV: Implications for Screening. Top Antivir Med 2022, 29, 423-429.
- Nickoloff-Bybel, E.A.; Festa, L.; Meucci, O.; Gaskill, P.J. Co-receptor signaling in the pathogenesis of neuroHIV. Retrovirology 2021, 18, 24. [CrossRef]
- Sreeram, S.; Ye, F.; Garcia-Mesa, Y.; Nguyen, K.; El Sayed, A.; Leskov, K.; Karn, J. The potential role of HIV-1 latency in promoting neuroinflammation and HIV-1-associated neurocognitive disorder. Trends Immunol 2022, 43, 630-639. [CrossRef]
- Marino, J.; Wigdahl, B.; Nonnemacher, M.R. Extracellular HIV-1 Tat Mediates Increased Glutamate in the CNS Leading to Onset of Senescence and Progression of HAND. Front Aging Neurosci 2020, 12, 168. [CrossRef]
- Sviridov, D.; Bukrinsky, M. Neuro-HIV-New insights into pathogenesis and emerging therapeutic targets. FASEB J 2023, 37, e23301. [CrossRef]
- Gabuzda, D.; Yin, J.; Misra, V.; Chettimada, S.; Gelman, B.B. Intact Proviral DNA Analysis of the Brain Viral Reservoir and Relationship to Neuroinflammation in People with HIV on Suppressive Antiretroviral Therapy. Viruses 2023, 15. [CrossRef]
- Cochrane, C.R.; Angelovich, T.A.; Byrnes, S.J.; Waring, E.; Guanizo, A.C.; Trollope, G.S.; Zhou, J.; Vue, J.; Senior, L.; Wanicek, E.; et al. Intact HIV Proviruses Persist in the Brain Despite Viral Suppression with ART. Ann Neurol 2022, 92, 532-544. [CrossRef]
- Rudd, H.; Toborek, M. Pitfalls of Antiretroviral Therapy: Current Status and Long-Term CNS Toxicity. Biomolecules 2022, 12. [CrossRef]
- Yuan, N.Y.; Kaul, M. Beneficial and Adverse Effects of cART Affect Neurocognitive Function in HIV-1 Infection: Balancing Viral Suppression against Neuronal Stress and Injury. J Neuroimmune Pharmacol 2021, 16, 90-112. [CrossRef]
- Roth, L.M.; Zidane, B.; Festa, L.; Putatunda, R.; Romer, M.; Monnerie, H.; Jordan-Sciutto, K.L.; Grinspan, J.B. Differential effects of integrase strand transfer inhibitors, elvitegravir and raltegravir, on oligodendrocyte maturation: A role for the integrated stress response. Glia 2021, 69, 362-376. [CrossRef]
- Namba, M.D.; Xie, Q.; Barker, J.M. Advancing the preclinical study of comorbid neuroHIV and substance use disorders: Current perspectives and future directions. Brain Behav Immun 2023, 113, 453-475. [CrossRef]
- Murphy, A.; Barbaro, J.; Martinez-Aguado, P.; Chilunda, V.; Jaureguiberry-Bravo, M.; Berman, J.W. The Effects of Opioids on HIV Neuropathogenesis. Front Immunol 2019, 10, 2445. [CrossRef]
- Nash, B.; Festa, L.; Lin, C.; Meucci, O. Opioid and chemokine regulation of cortical synaptodendritic damage in HIV-associated neurocognitive disorders. Brain Res 2019, 1723, 146409. [CrossRef]
- Fitting, S.; McRae, M.; Hauser, K.F. Opioid and neuroHIV Comorbidity - Current and Future Perspectives. J Neuroimmune Pharmacol 2020, 15, 584-627. [CrossRef]
- Vines, L.; Sotelo, D.; Giddens, N.; Manza, P.; Volkow, N.D.; Wang, G.J. Neurological, Behavioral, and Pathophysiological Characterization of the Co-Occurrence of Substance Use and HIV: A Narrative Review. Brain Sci 2023, 13. [CrossRef]
- Soontornniyomkij, V.; Kesby, J.P.; Morgan, E.E.; Bischoff-Grethe, A.; Minassian, A.; Brown, G.G.; Grant, I.; Translational Methamphetamine, A.R.C.G. Effects of HIV and Methamphetamine on Brain and Behavior: Evidence from Human Studies and Animal Models. J Neuroimmune Pharmacol 2016, 11, 495-510. [CrossRef]
- Hartzler, B.; Dombrowski, J.C.; Crane, H.M.; Eron, J.J.; Geng, E.H.; Christopher Mathews, W.; Mayer, K.H.; Moore, R.D.; Mugavero, M.J.; Napravnik, S.; et al. Prevalence and Predictors of Substance Use Disorders Among HIV Care Enrollees in the United States. AIDS Behav 2017, 21, 1138-1148. [CrossRef]
- Garner, B.R.; Gotham, H.J.; Knudsen, H.K.; Zulkiewicz, B.A.; Tueller, S.J.; Berzofsky, M.; Donohoe, T.; Martin, E.G.; Brown, L.L.; Gordon, T. The Prevalence and Negative Impacts of Substance Use Disorders among People with HIV in the United States: A Real-Time Delphi Survey of Key Stakeholders. AIDS Behav 2022, 26, 1183-1196. [CrossRef]
- Irollo, E.; Luchetta, J.; Ho, C.; Nash, B.; Meucci, O. Mechanisms of neuronal dysfunction in HIV-associated neurocognitive disorders. Cell Mol Life Sci 2021, 78, 4283-4303. [CrossRef]
- Gelman, B.B.; Endsley, J.; Kolson, D. When do models of NeuroAIDS faithfully imitate “the real thing”? J Neurovirol 2018, 24, 146-155. [CrossRef]
- McGuire, J.L.; Barrett, J.S.; Vezina, H.E.; Spitsin, S.; Douglas, S.D. Adjuvant therapies for HIV-associated neurocognitive disorders. Ann Clin Transl Neurol 2014, 1, 938-952. [CrossRef]
- Omeragic, A.; Kayode, O.; Hoque, M.T.; Bendayan, R. Potential pharmacological approaches for the treatment of HIV-1 associated neurocognitive disorders. Fluids Barriers CNS 2020, 17, 42. [CrossRef]
- Gonzalez-Scarano, F.; Martin-Garcia, J. The neuropathogenesis of AIDS. Nat Rev Immunol 2005, 5, 69-81. [CrossRef]
- Peluso, R.; Haase, A.; Stowring, L.; Edwards, M.; Ventura, P. A Trojan Horse mechanism for the spread of visna virus in monocytes. Virology 1985, 147, 231-236. [CrossRef]
- Hsu, C.W.; Cerda, J., 3rd; Kirk, J.M.; Turner, W.D.; Rasmussen, T.L.; Flores Suarez, C.P.; Dickinson, M.E.; Wythe, J.D. EZ Clear for simple, rapid, and robust mouse whole organ clearing. Elife 2022, 11. [CrossRef]
- Seabold, G.K.; Daunais, J.B.; Rau, A.; Grant, K.A.; Alvarez, V.A. DiOLISTIC labeling of neurons from rodent and non-human primate brain slices. J Vis Exp 2010. [CrossRef]
- Rodriguez, A.; Ehlenberger, D.B.; Dickstein, D.L.; Hof, P.R.; Wearne, S.L. Automated three-dimensional detection and shape classification of dendritic spines from fluorescence microscopy images. PLoS ONE 2008, 3, e1997. [CrossRef]
- Schindler, M.; Munch, J.; Kirchhoff, F. Human immunodeficiency virus type 1 inhibits DNA damage-triggered apoptosis by a Nef-independent mechanism. J Virol 2005, 79, 5489-5498. [CrossRef]
- Schindler, M.; Wurfl, S.; Benaroch, P.; Greenough, T.C.; Daniels, R.; Easterbrook, P.; Brenner, M.; Munch, J.; Kirchhoff, F. Down-modulation of mature major histocompatibility complex class II and up-regulation of invariant chain cell surface expression are well-conserved functions of human and simian immunodeficiency virus nef alleles. J Virol 2003, 77, 10548-10556. [CrossRef]
- Chesebro, B.; Nishio, J.; Perryman, S.; Cann, A.; O’Brien, W.; Chen, I.S.; Wehrly, K. Identification of human immunodeficiency virus envelope gene sequences influencing viral entry into CD4-positive HeLa cells, T-leukemia cells, and macrophages. J Virol 1991, 65, 5782-5789. [CrossRef]
- Toohey, K.; Wehrly, K.; Nishio, J.; Perryman, S.; Chesebro, B. Human immunodeficiency virus envelope V1 and V2 regions influence replication efficiency in macrophages by affecting virus spread. Virology 1995, 213, 70-79. [CrossRef]
- Walter, B.L.; Wehrly, K.; Swanstrom, R.; Platt, E.; Kabat, D.; Chesebro, B. Role of low CD4 levels in the influence of human immunodeficiency virus type 1 envelope V1 and V2 regions on entry and spread in macrophages. J Virol 2005, 79, 4828-4837. [CrossRef]
- Chesebro, B.; Wehrly, K.; Nishio, J.; Perryman, S. Macrophage-tropic human immunodeficiency virus isolates from different patients exhibit unusual V3 envelope sequence homogeneity in comparison with T-cell-tropic isolates: Definition of critical amino acids involved in cell tropism. J Virol 1992, 66, 6547-6554. [CrossRef]
- Januszka, J.E.; Drwiega, E.N.; Badowski, M.E. Bictegravir/Emtricitabine/Tenofovir Alafenamide for HIV-1: What is the Hidden Potential of This Emerging Treatment? HIV AIDS (Auckl) 2023, 15, 705-711. [CrossRef]
- Landovitz, R.J.; Li, S.; Eron, J.J., Jr.; Grinsztejn, B.; Dawood, H.; Liu, A.Y.; Magnus, M.; Hosseinipour, M.C.; Panchia, R.; Cottle, L.; et al. Tail-phase safety, tolerability, and pharmacokinetics of long-acting injectable cabotegravir in HIV-uninfected adults: A secondary analysis of the HPTN 077 trial. Lancet HIV 2020, 7, e472-e481. [CrossRef]
- Letendre, S.L.; Mills, A.; Hagins, D.; Swindells, S.; Felizarta, F.; Devente, J.; Bettacchi, C.; Lou, Y.; Ford, S.; Sutton, K.; et al. Pharmacokinetics and antiviral activity of cabotegravir and rilpivirine in cerebrospinal fluid following long-acting injectable administration in HIV-infected adults. J Antimicrob Chemother 2020, 75, 648-655. [CrossRef]
- Schwarz, N.; Uysal, B.; Welzer, M.; Bahr, J.C.; Layer, N.; Loffler, H.; Stanaitis, K.; Pa, H.; Weber, Y.G.; Hedrich, U.B.; et al. Long-term adult human brain slice cultures as a model system to study human CNS circuitry and disease. Elife 2019, 8. [CrossRef]
- Bak, A.; Koch, H.; van Loo, K.M.J.; Schmied, K.; Gittel, B.; Weber, Y.; Ort, J.; Schwarz, N.; Tauber, S.C.; Wuttke, T.V.; et al. Human organotypic brain slice cultures: A detailed and improved protocol for preparation and long-term maintenance. J Neurosci Methods 2024, 404, 110055. [CrossRef]
- Martin, E.; El-Behi, M.; Fontaine, B.; Delarasse, C. Analysis of Microglia and Monocyte-derived Macrophages from the Central Nervous System by Flow Cytometry. J Vis Exp 2017. [CrossRef]
- Jurga, A.M.; Paleczna, M.; Kuter, K.Z. Overview of General and Discriminating Markers of Differential Microglia Phenotypes. Front Cell Neurosci 2020, 14, 198. [CrossRef]
- Runge, K.; Cardoso, C.; de Chevigny, A. Dendritic Spine Plasticity: Function and Mechanisms. Front Synaptic Neurosci 2020, 12, 36. [CrossRef]
- Everall, I.P.; Heaton, R.K.; Marcotte, T.D.; Ellis, R.J.; McCutchan, J.A.; Atkinson, J.H.; Grant, I.; Mallory, M.; Masliah, E. Cortical synaptic density is reduced in mild to moderate human immunodeficiency virus neurocognitive disorder. HNRC Group. HIV Neurobehavioral Research Center. Brain Pathol 1999, 9, 209-217. [CrossRef]
- Masliah, E.; Heaton, R.K.; Marcotte, T.D.; Ellis, R.J.; Wiley, C.A.; Mallory, M.; Achim, C.L.; McCutchan, J.A.; Nelson, J.A.; Atkinson, J.H.; et al. Dendritic injury is a pathological substrate for human immunodeficiency virus-related cognitive disorders. HNRC Group. The HIV Neurobehavioral Research Center. Ann Neurol 1997, 42, 963-972. [CrossRef]
- Schwarz, N.; Hedrich, U.B.S.; Schwarz, H.; P, A.H.; Dammeier, N.; Auffenberg, E.; Bedogni, F.; Honegger, J.B.; Lerche, H.; Wuttke, T.V.; et al. Human Cerebrospinal fluid promotes long-term neuronal viability and network function in human neocortical organotypic brain slice cultures. Sci Rep 2017, 7, 12249. [CrossRef]
- Akkina, R.; Barber, D.L.; Bility, M.T.; Bissig, K.D.; Burwitz, B.J.; Eichelberg, K.; Endsley, J.J.; Garcia, J.V.; Hafner, R.; Karakousis, P.C.; et al. Small Animal Models for Human Immunodeficiency Virus (HIV), Hepatitis B, and Tuberculosis: Proceedings of an NIAID Workshop. Curr HIV Res 2020, 18, 19-28. [CrossRef]
- Beck, S.E.; Queen, S.E.; Metcalf Pate, K.A.; Mangus, L.M.; Abreu, C.M.; Gama, L.; Witwer, K.W.; Adams, R.J.; Zink, M.C.; Clements, J.E.; et al. An SIV/macaque model targeted to study HIV-associated neurocognitive disorders. J Neurovirol 2018, 24, 204-212. [CrossRef]
- Mallard, J.; Williams, K. An SIV macaque model of SIV and HAND: The need for adjunctive therapies in HIV that target activated monocytes and macrophages. J Neurovirol 2018, 24, 213-219. [CrossRef]
- Vigorito, M.; Connaghan, K.P.; Chang, S.L. The HIV-1 transgenic rat model of neuroHIV. Brain Behav Immun 2015, 48, 336-349. [CrossRef]
- Dos Reis, R.S.; Sant, S.; Keeney, H.; Wagner, M.C.E.; Ayyavoo, V. Modeling HIV-1 neuropathogenesis using three-dimensional human brain organoids (hBORGs) with HIV-1 infected microglia. Sci Rep 2020, 10, 15209. [CrossRef]
- Gumbs, S.B.H.; Berdenis van Berlekom, A.; Kubler, R.; Schipper, P.J.; Gharu, L.; Boks, M.P.; Ormel, P.R.; Wensing, A.M.J.; de Witte, L.D.; Nijhuis, M. Characterization of HIV-1 Infection in Microglia-Containing Human Cerebral Organoids. Viruses 2022, 14. [CrossRef]
- Bodnar, B.; Zhang, Y.; Liu, J.; Lin, Y.; Wang, P.; Wei, Z.; Saribas, S.; Zhu, Y.; Li, F.; Wang, X.; et al. Novel Scalable and Simplified System to Generate Microglia-Containing Cerebral Organoids From Human Induced Pluripotent Stem Cells. Front Cell Neurosci 2021, 15, 682272. [CrossRef]
- Zhao, H.H.; Haddad, G. Brain organoid protocols and limitations. Front Cell Neurosci 2024, 18, 1351734. [CrossRef]
- Wei, Z.; Bodnar, B.; Zhao, R.T.; Xiao, Q.; Saribas, S.; Wang, X.; Ho, W.Z.; Hu, W. Human iPSC-derived brain organoids: A 3D mini-brain model for studying HIV infection. Exp Neurol 2023, 364, 114386. [CrossRef]
- Tang, Y.; Chaillon, A.; Gianella, S.; Wong, L.M.; Li, D.; Simermeyer, T.L.; Porrachia, M.; Ignacio, C.; Woodworth, B.; Zhong, D.; et al. Brain microglia serve as a persistent HIV reservoir despite durable antiretroviral therapy. J Clin Invest 2023, 133. [CrossRef]
- Williams, M.E.; Naude, P.J.W. The relationship between HIV-1 neuroinflammation, neurocognitive impairment and encephalitis pathology: A systematic review of studies investigating post-mortem brain tissue. Rev Med Virol 2024, 34, e2519. [CrossRef]
- Boerwinkle, A.H.; Meeker, K.L.; Luckett, P.; Ances, B.M. Neuroimaging the Neuropathogenesis of HIV. Curr HIV/AIDS Rep 2021, 18, 221-228. [CrossRef]
- Martinez, R.; Eraso, D.; Geffin, R.; McCarthy, M. A two-culture method for exposure of human brain organotypic slice cultures to replicating human immunodeficiency virus type 1. J Neurosci Methods 2011, 200, 74-79. [CrossRef]
- Almeida, G.M.; Souza, J.P.; Mendes, N.D.; Pontelli, M.C.; Pinheiro, N.R.; Nogueira, G.O.; Cardoso, R.S.; Paiva, I.M.; Ferrari, G.D.; Veras, F.P.; et al. Neural Infection by Oropouche Virus in Adult Human Brain Slices Induces an Inflammatory and Toxic Response. Front Neurosci 2021, 15, 674576. [CrossRef]
- Partiot, E.; Gorda, B.; Lutz, W.; Lebrun, S.; Khalfi, P.; Mora, S.; Charlot, B.; Majzoub, K.; Desagher, S.; Ganesh, G.; et al. Organotypic culture of human brain explants as a preclinical model for AI-driven antiviral studies. EMBO Mol Med 2024. [CrossRef]
- Kramvis, I.; Mansvelder, H.D.; Meredith, R.M. Neuronal life after death: Electrophysiologic recordings from neurons in adult human brain tissue obtained through surgical resection or postmortem. Handb Clin Neurol 2018, 150, 319-333. [CrossRef]
- Qi, X.R.; Verwer, R.W.H.; Bao, A.M.; Balesar, R.A.; Luchetti, S.; Zhou, J.N.; Swaab, D.F. Human Brain Slice Culture: A Useful Tool to Study Brain Disorders and Potential Therapeutic Compounds. Neurosci Bull 2019, 35, 244-252. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




