Submitted:
16 April 2024
Posted:
17 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Spinal Muscular Atrophy
1.2. SMA Orphan Drugs
1.3. Transcutaneous Spinal Cord Stimulation
1.4. Purpose and Hypothesis of the Study
2. Materials and Methods
2.1. Participants
2.2. Procedures
2.2.1. Treatment Goals
2.2.2. Physical Therapy
2.2.3. Transcutaneous Spinal Cord Stimulation
2.3. Outcome measures
2.3.1. Joint Goniometry
2.3.2. Revised Upper Limb Module
2.3.3. Hammersmith Function Motor Scale Expanded
2.3.4. Forced Vital Capacity
2.4. Statistics
3. Results
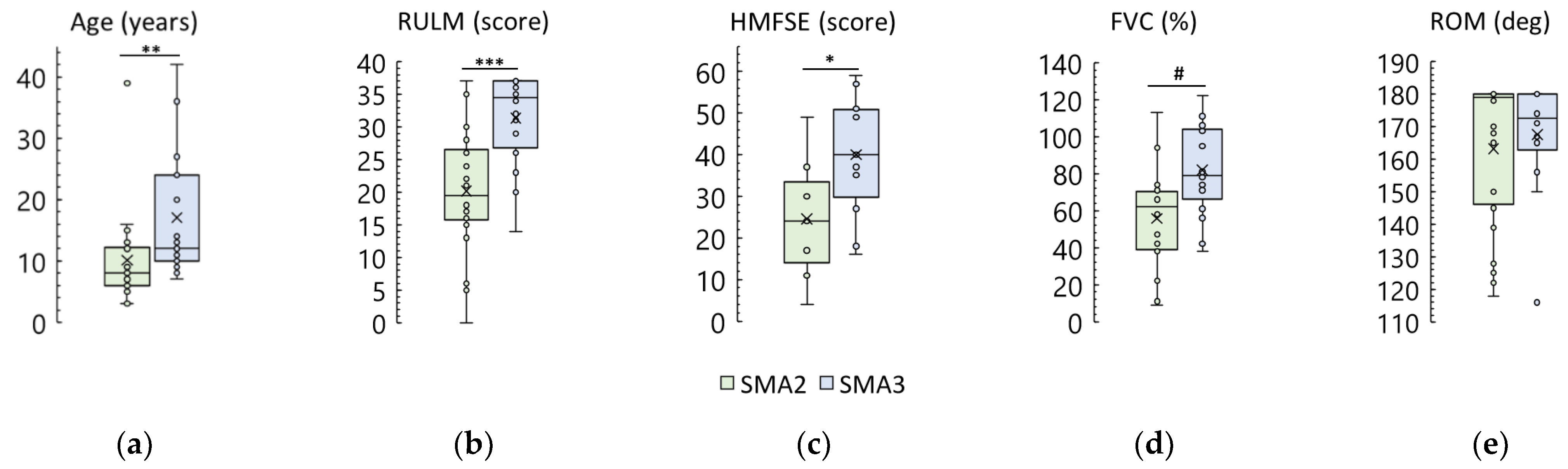
3.1. SMA type 2 and type 3 participants
3.2. Transcutaneous Spinal Cord Stimulation
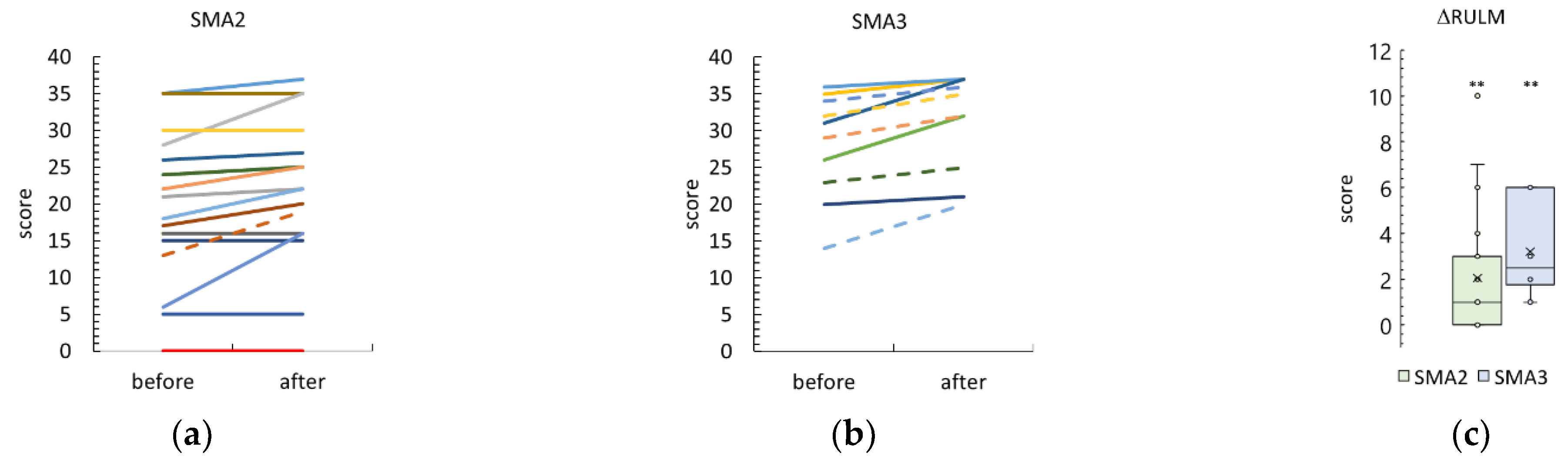
3.3. Revised Upper Limb Module
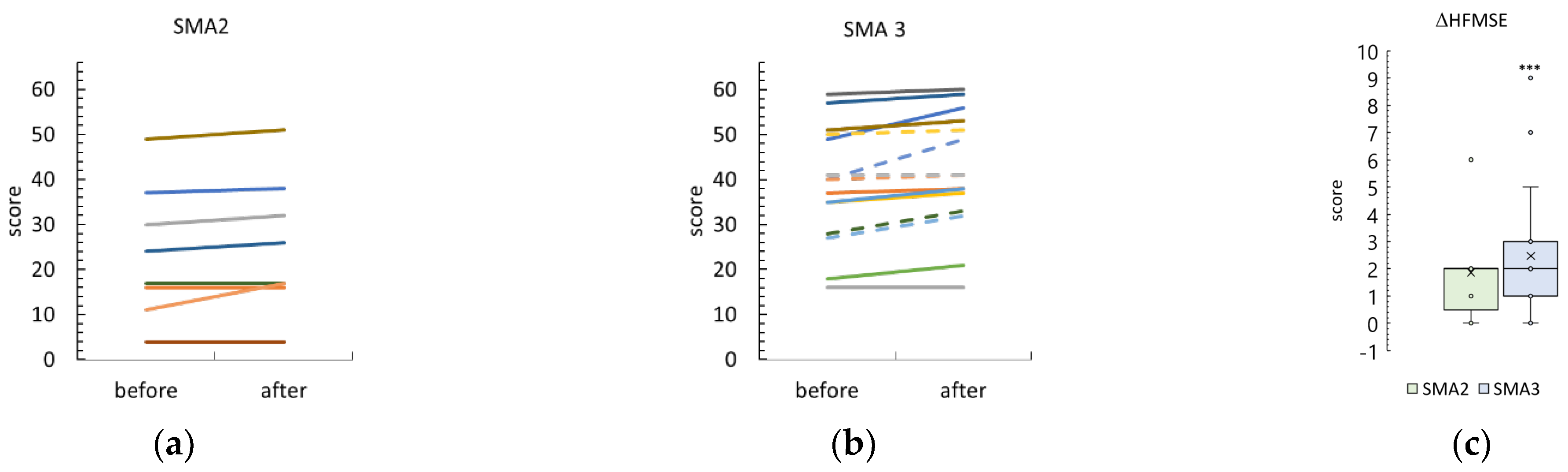
3.4. Hammersmith Function Motor Scale Expanded
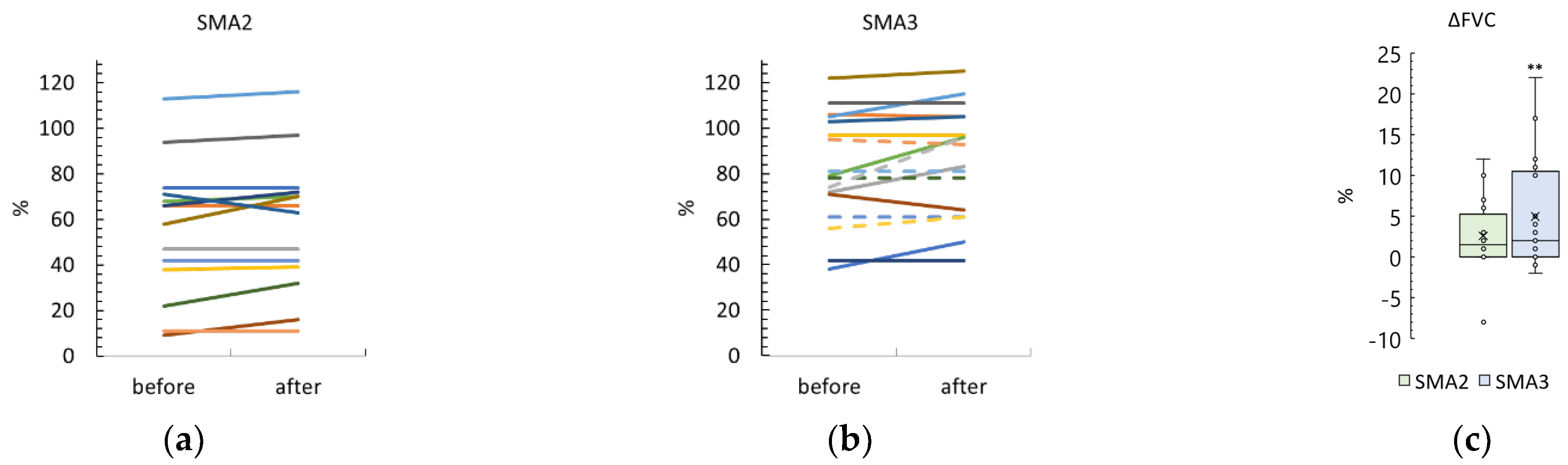
3.5. Forced Vital Capacity
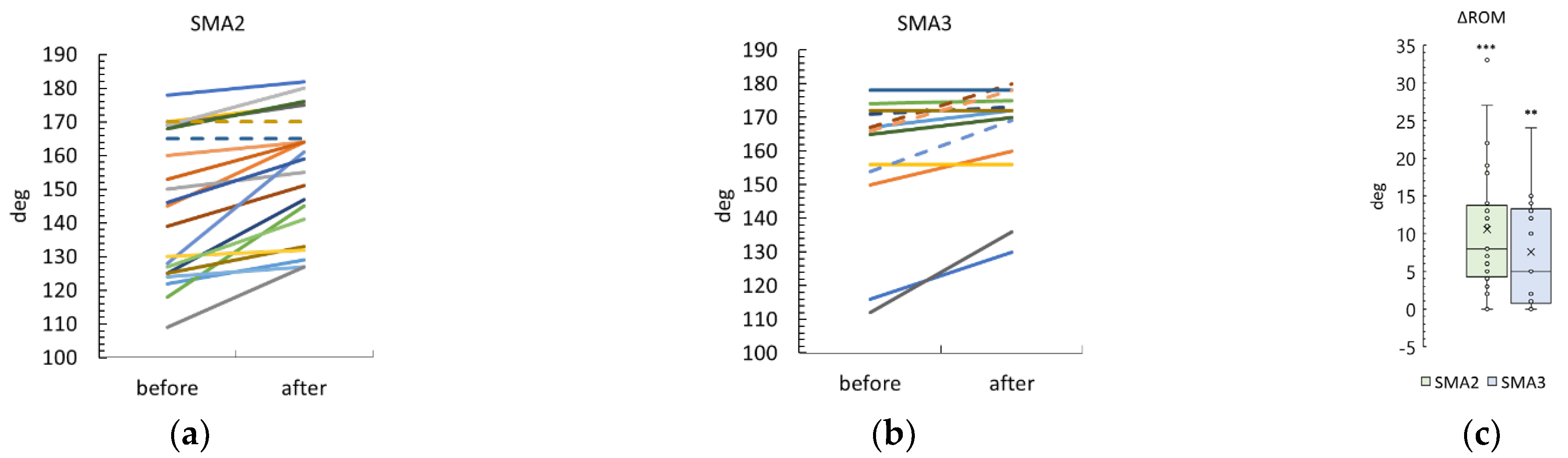
3.6. Knee Range of Motion
3.7 Motor skills
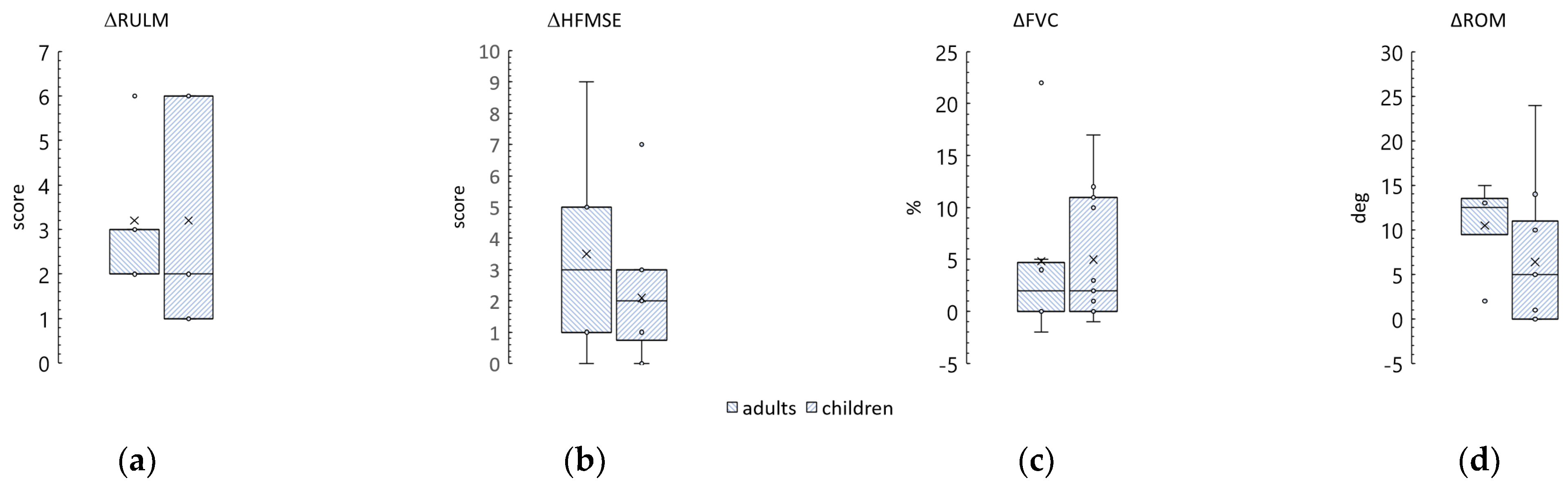
3.8. Age and Adults
4. Discussion
4.1. Minimal Clinically Important Difference and Stimulation Results
4.1.1. RULM and HMFSE Scales
4.1.2. ROM Results
4.1.3. FVC Results
4.1.4. Motor Skills Results
4.2. Age Influence
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Novikov, A.; Maldova, M.; Shandybina, N.; Shalmiev, I.; Shoshina, E.; Epoyan, N.; Moshonkina, T. First Use of Non-Invasive Spinal Cord Stimulation in Motor Rehabilitation of Children with Spinal Muscular Atrophy. Life 2023, 13, 449. [Google Scholar] [CrossRef] [PubMed]
- Verhaart, I.E.C.; Robertson, A.; Wilson, I.J.; Aartsma-Rus, A.; Cameron, S.; Jones, C.C.; Cook, S.F.; Lochmüller, H. Prevalence, Incidence and Carrier Frequency of 5q–Linked Spinal Muscular Atrophy – a Literature Review. Orphanet J Rare Dis 2017, 12, 124. [Google Scholar] [CrossRef] [PubMed]
- Mercuri, E.; Sumner, C.; Muntoni, F.; Darras, B.T.; Finkel, R.S. Spinal Muscular Atrophy. NATURE REvIEWS 2022. [Google Scholar] [CrossRef]
- Lefebvre, S.; Bürglen, L.; Reboullet, S.; Clermont, O.; Burlet, P.; Viollet, L.; Benichou, B.; Cruaud, C.; Millasseau, P.; Zeviani, M.; et al. Identification and Characterization of a Spinal Muscular Atrophy-Determining Gene. Cell 1995, 1, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Mercuri, E.; Finkel, R.S.; Muntoni, F.; Wirth, B.; Montes, J.; Main, M.; Mazzone, E.S.; Vitale, M.; Snyder, B.; Quijano-Roy, S.; et al. Diagnosis and Management of Spinal Muscular Atrophy: Part 1: Recommendations for Diagnosis, Rehabilitation, Orthopedic and Nutritional Care. Neuromuscular Disorders 2018, 28, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Nishio, H.; Niba, E.T.E.; Saito, T.; Okamoto, K.; Takeshima, Y.; Awano, H. Spinal Muscular Atrophy: The Past, Present, and Future of Diagnosis and Treatment. International Journal of Molecular Sciences 2023, 24, 11939. [Google Scholar] [CrossRef] [PubMed]
- Sirbu, C.A.; Ivan, R.; Samaha, F.J.; Ionita-Radu, F.; Jianu, D.C.; Vasiliu, O.; Constantin, C.; Tuță, S. Orphan Drugs in Neurology - A Narrative Review. [CrossRef]
- Dangouloff, T.; Botty, C.; Beaudart, C.; Servais, L.; Hiligsmann, M. Systematic Literature Review of the Economic Burden of Spinal Muscular Atrophy and Economic Evaluations of Treatments. Orphanet J Rare Dis 2021, 16, 47. [Google Scholar] [CrossRef] [PubMed]
- Finkel, R.S.; Mercuri, E.; Darras, B.T.; Connolly, N.L.; Kuntz, N.L.; Kirschner, C.A.; Chiriboga, C.A.; Saito, K.; Servais, L.; Tizzano, E. Nusinersen versus Sham Control in Infantile-Onset Spinal Muscular Atrophy. [CrossRef]
- Mercuri, E.; Darras, B.T.; Chiriboga, C.A.; Day, J.W.; Campbell, C.; Connolly, A.M.; Iannaccone, S.T.; Kirschner, J.; Kuntz, N.L.; Saito, K.; et al. Nusinersen versus Sham Control in Later-Onset Spinal Muscular Atrophy. N Engl J Med 2018, 378, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Dangouloff, T.; Servais, L. Clinical Evidence Supporting Early Treatment Of Patients With Spinal Muscular Atrophy: Current Perspectives. TCRM 2019, 15, 1153–1161. [Google Scholar] [CrossRef]
- Mendell, J.R.; Al-Zaidy, S.; Shell, R.; Arnold, W.D.; Rodino-Klapac, L.R.; Prior, T.; Lowes, L.P.; Alfano, L.; Berry, K.; Church, K. Single-Dose Gene-Replacement Therapy for Spinal Muscular Atrophy. [CrossRef]
- Ross, L.F.; Kwon, J.M. Spinal Muscular Atrophy: Past, Present, and Future. NeoReviews 2019, 20, e437–e451. [Google Scholar] [CrossRef]
- Gorodnichev, R.M.; Pivovarova, E.A.; Puhov, A.; Moiseev, S.A.; Gerasimenko, Y.P.; Savochin, A.A.; Moshonkina, T.R.; Chsherbakova, N.A.; Kilimnik, V.A.; Selionov, V.A.; et al. Transcutaneous Electrical Stimulation of the Spinal Cord: A Noninvasive Tool for the Activation of Stepping Pattern Generators in Humans. Human Physiology 2012, 38, 158–167. [Google Scholar] [CrossRef]
- Gerasimenko, Y.; Gorodnichev, R.; Moshonkina, T.; Sayenko, D.; Gad, P.; Reggie Edgerton, V. Transcutaneous Electrical Spinal-Cord Stimulation in Humans. Annals of Physical and Rehabilitation Medicine 2015, 58, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Moshonkina, T.; Grishin, A.; Bogacheva, I.; Gorodnichev, R.; Ovechkin, A.; Siu, R.; Edgerton, V.R.; Gerasimenko, Y. Novel Non-Invasive Strategy for Spinal Neuromodulation to Control Human Locomotion. Available online: https://www.frontiersin.org/articles/10.3389/fnhum.2020.622533/full (accessed on 28 July 2023).
- Shamantseva, N.; Timofeeva, O.; Gvozdeva, A.; Andreeva, I.; Moshonkina, T. Posture of Healthy Subjects Modulated by Transcutaneous Spinal Cord Stimulation. Life 2023, 13, 1909. [Google Scholar] [CrossRef] [PubMed]
- Gerasimenko, Y.P.; McKinney, Z.; Sayenko, D.G.; Gad, P.; Gorodnichev, R.M.; Grundfest, W.; Edgerton, V.R.; Kozlovskaya, I.B. Spinal and Sensory Neuromodulation of Spinal Neuronal Networks in Humans. Hum Physiol 2017, 43, 492–500. [Google Scholar] [CrossRef]
- Megía-García, Á.; Serrano-Muñoz, D.; Taylor, J.; Avendaño-Coy, J.; Comino-Suárez, N.; Gómez-Soriano, J. Transcutaneous Spinal Cord Stimulation Enhances Quadriceps Motor Evoked Potential in Healthy Participants: A Double-Blind Randomized Controlled Study. Journal of Clinical Medicine 2020, 9, 3275. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Lucas, K.; Keller, A.; Martin, R.; Behrman, A.; Vissarionov, S.; Gerasimenko, Y. Transcutaneous Spinal Stimulation From Adults to Children: A Review | Topics in Spinal Cord Injury Rehabilitation. [CrossRef]
- Moshonkina, T.R.; Zharova, E.N.; Ananev, S.S.; Shandybina, N.D.; Vershinina, E.A.; Lyakhovetskii, V.A.; Grishin, A.A.; Shlyakhto, E.V.; Gerasimenko, Y.P. A New Technology for Recovery of Locomotion in Patients after a Stroke | Doklady Biochemistry and Biophysics. Available online: https://link.springer.com/article/10.1134/S1607672922340087 (accessed on 3 February 2024).
- Palisano, R.; Rosenbaum, P.; Walter, S.; Russell, D.; Wood, E.; Galuppi, G. Development and Reliability of a System to Classify Gross Motor Function in Children with Cerebral Palsy - Palisano - 1997 - Developmental Medicine & Child Neurology - Wiley Online Library. Available online: https://doi.org/10.1111/j.1469-8749.1997.tb07414.x (accessed on 3 February 2024).
- Solopova, I.A.; Sukhotina, I.A.; Zhvansky, D.S.; Ikoeva, G.A.; Vissarionov, S.V.; Baindurashvili, A.G.; Edgerton, V.R.; Gerasimenko, Y.P.; Moshonkina, T.R. Effects of Spinal Cord Stimulation on Motor Functions in Children with Cerebral Palsy. Neuroscience Letters 2017, 639, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Gad, P.; Hastings, H.; Zhong, H.; Seth, G.; Kandhari, S.; Edgerton, V.R. Transcutaneous Spinal Neuromodulation Reorganizes Neural Networks in Patients with Cerebral Palsy | SpringerLink. Available online: https://link.springer.com/article/10.1007/s13311-021-01087-6 (accessed on 10 June 2023).
- Kariyawasam, D.; D’Silva, A.; Howells, J.; Herbert, K.; Geelan-Small, P.; Lin, C.S.-Y.; Farrar, M.A. Motor Unit Changes in Children with Symptomatic Spinal Muscular Atrophy Treated with Nusinersen. J Neurol Neurosurg Psychiatry 2021, 92, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Kessler, T.; Sam, G.; Wick, W.; Weiler, M. Evaluation of Risdiplam Efficacy in 5q Spinal Muscular Atrophy: A Systematic Comparison of Electrophysiologic with Clinical Outcome Measures. European Journal of Neurology 2024, 31, e16099. [Google Scholar] [CrossRef]
- Bovend’Eerdt, T.J.; Botell, R.E.; Wade, D.T. Writing SMART Rehabilitation Goals and Achieving Goal Attainment Scaling: A Practical Guide. Clin Rehabil 2009, 23, 352–361. [Google Scholar] [CrossRef]
- Solopova, I.A.; Selionov, V.A.; Zhvansky, D.S.; Gurfinkel, V.S.; Ivanenko, Y.P. Human Cervical Spinal Cord Circuitry Activated by Tonic Input Can Generate Rhythmic Arm Movements. Available online: https://journals.physiology.org/doi/epdf/10.1152/jn.00897.2015 (accessed on 22 March 2024).
- Baindurashvili, A.; Vissarionov, S.; Belianchikov, S.; Kartavenko, K.; Solokhina, I.; Kozyrev, A.; Pukhov, A.; Moshonkina, T.; Gerasimenko, Y. Comprehensive Treatment of a Patient with Complicated Thoracic Spine Injury Using Percutaneous Electrical Spinal Cord Stimulation (Case Report). Genij Ortopedii 2020, 26, 79–88. [Google Scholar] [CrossRef]
- Huang, R.; Nikooyan, A.A.; Moore, L.D.; Zdunowski, S.; Morikawa, E.; Sierro, T.; Sayenko, D.; Gad, P.; Homsey, T.; Le, T. Minimal Handgrip Force Is Needed for Transcutaneous Electrical Stimulation to Improve Hand Functions of Patients with Severe Spinal Cord Injury. Scientific Reports 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Megía García, A.; Serrano-Muñoz, D.; Taylor, J.; Avendaño-Coy, J.; Gómez-Soriano, J. Transcutaneous Spinal Cord Stimulation and Motor Rehabilitation in Spinal Cord Injury: A Systematic Review. Neurorehabil Neural Repair 2020, 34, 3–12. [Google Scholar] [CrossRef]
- Zhang, F.; Momeni, K.; Ramanujam, A.; Ravi, M.; Carnahan, J.; Kirshblum, S.; Forrest, G.F. Cervical Spinal Cord Transcutaneous Stimulation Improves Upper Extremity and Hand Function in People With Complete Tetraplegia: A Case Study. IEEE Transactions on Neural Systems and Rehabilitation Engineering 2020, 28, 3167–3174. [Google Scholar] [CrossRef]
- Iannaccone, S.T. Outcome Measures for Pediatric Spinal Muscular Atrophy. Arch Neurol 2002, 59, 1445. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.W.; Pepler, L.; Maturi, B.; Afonso, A.C.F.; Sarmiento, J.; Haldenby, R. Systematic Review of Motor Function Scales and Patient-Reported Outcomes in Spinal Muscular Atrophy. Am J Phys Med Rehabil 2022, 101, 590–608. [Google Scholar] [CrossRef] [PubMed]
- Salazar, R.; Montes, J.; Dunaway Young, S.; McDermott, M.P.; Martens, W.; Pasternak, A.; Quigley, J.; Mirek, E.; Glanzman, A.M.; Civitello, M.; et al. Quantitative Evaluation of Lower Extremity Joint Contractures in Spinal Muscular Atrophy: Implications for Motor Function. Pediatric Physical Therapy 2018, 30, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, E.S.; Mayhew, A.; Montes, J.; Ramsey, D.; Fanelli, L.; Young, S.D.; Salazar, R.; De Sanctis, R.; Pasternak, A.; Glanzman, A.; et al. Revised Upper Limb Module for Spinal Muscular Atrophy: Development of a New Module. Muscle Nerve 2017, 55, 869–874. [Google Scholar] [CrossRef]
- O’Hagen, J.M.; Glanzman, A.M.; McDermott, M.P.; Ryan, P.A.; Flickinger, J.; Quigley, J.; Riley, S.; Sanborn, E.; Irvine, C.; Martens, W.B.; et al. An Expanded Version of the Hammersmith Functional Motor Scale for SMA II and III Patients. Neuromuscular Disorders 2007, 17, 693–697. [Google Scholar] [CrossRef]
- Graham, B.L.; Steenbruggen, I.; Miller, M.R.; Barjaktarevic, I.Z.; Cooper, B.G.; Hall, G.L.; Hallstrand, T.S.; Kaminsky, D.A.; McCarthy, K.; McCormack, M.C.; et al. Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement. Am J Respir Crit Care Med 2019, 200, e70–e88. [Google Scholar] [CrossRef]
- Norkin, C.C.; White D., J. Measurement of Joint Motion: A Guide to Goniometry, Third edition; F.A. Davis Company: Philadelphia, 2004; ISBN 978-0-8036-4566-0. [Google Scholar]
- Iannaccone, S.T.; Hynan, L.S.; Morton, A.; Buchanan, R.; Limbers, C.A.; Varni, J.W. The PedsQLTM in Pediatric Patients with Spinal Muscular Atrophy: Feasibility, Reliability, and Validity of the Pediatric Quality of Life InventoryTM Generic Core Scales and Neuromuscular Module. Neuromuscular Disorders 2009, 19, 805–812. [Google Scholar] [CrossRef]
- Vázquez-Costa, J.F.; Povedano, M.; Nascimiento-Osorio, A.E.; Moreno Escribano, A.; Kapetanovic Garcia, S.; Dominguez, R.; Exposito, J.M.; González, L.; Marco, C.; Medina Castillo, J.; et al. Nusinersen in Adult Patients with 5q Spinal Muscular Atrophy: A Multicenter Observational Cohorts’ Study. European Journal of Neurology 2022, 29, 3337–3346. [Google Scholar] [CrossRef] [PubMed]
- Wijngaarde, C.A.; Veldhoen, E.S.; van Eijk, R.P.A.; Stam, M.; Otto, L.A.M.; Asselman, F.-L.; Wösten-van Asperen, R.M.; Hulzebos, E.H.J.; Verweij-van den Oudenrijn, L.P.; Bartels, B.; et al. Natural History of Lung Function in Spinal Muscular Atrophy. Orphanet J Rare Dis 2020, 15, 88. [Google Scholar] [CrossRef] [PubMed]
- Samaha, F.J.; Buncher, C.R.; Russman, B.S.; White, M.L.; Iannaccone, S.T.; Barker, L.; Burhans, K.; Smith, C.; Perkins, B.; Zimmerman, L. Pulmonary Function in Spinal Muscular Atrophy. J Child Neurol 1994, 9, 326–329. [Google Scholar] [CrossRef] [PubMed]
- Montes, J.; Garber, C.E.; Kramer, S.S.; Montgomery, M.J.; Dunaway, S.; Kamil-Rosenberg, S.; Carr, B.; Cruz, R.; Strauss, N.E.; Sproule, D.; et al. Single-Blind, Randomized, Controlled Clinical Trial of Exercise in Ambulatory Spinal Muscular Atrophy: Why Are the Results Negative? JND 2015, 2, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Beaton, D.; Shea, B.; Boers, M.; Simon, L.; Strand, V.; Brooks, P.; Tugwell, P. Minimal Clinically Important Differences: Review of Methods. The Journal of Rheumatology 2001. [Google Scholar]
- Stolte, B.; Bois, J.-M.; Bolz, S.; Kizina, K.; Totzeck, A.; Schlag, M.; Kleinschnitz, C.; Hagenacker, T. Minimal Clinically Important Differences in Functional Motor Scores in Adults with Spinal Muscular Atrophy. Euro J of Neurology 2020, 27, 2586–2594. [Google Scholar] [CrossRef] [PubMed]
- Pera, M.C.; Coratti, G.; Forcina, N.; Mazzone, E.S.; Scoto, M.; Montes, J.; Pasternak, A.; Mayhew, A.; Messina, S.; Sframeli, M.; et al. Content Validity and Clinical Meaningfulness of the HFMSE in Spinal Muscular Atrophy. BMC Neurology 2017, 17, 39. [Google Scholar] [CrossRef] [PubMed]
- Pera, M.C.; Coratti, G.; Mazzone, E.S.; Montes, J.; Scoto, M.; De Sanctis, R.; Main, M.; Mayhew, A.; Muni Lofra, R.; Dunaway Young, S.; et al. Revised Upper Limb Module for Spinal Muscular Atrophy: 12 Month Changes. Muscle & Nerve 2019, 59, 426–430. [Google Scholar] [CrossRef]
- Finkel, R.S.; Mercuri, E.; Meyer, O.H.; Simonds, A.K.; Schroth, M.K.; Graham, R.J.; Kirschner, J.; Iannaccone, S.T.; Crawford, T.O.; Woods, S.; et al. Diagnosis and Management of Spinal Muscular Atrophy: Part 2: Pulmonary and Acute Care; Medications, Supplements and Immunizations; Other Organ Systems; and Ethics. Neuromuscular Disorders 2018, 28, 197–207. [Google Scholar] [CrossRef]
- Chabanon, A.; Seferian, A.M.; Daron, A.; Péréon, Y.; Cances, C.; Vuillerot, C.; De Waele, L.; Cuisset, J.-M.; Laugel, V.; Schara, U.; et al. Prospective and Longitudinal Natural History Study of Patients with Type 2 and 3 Spinal Muscular Atrophy: Baseline Data NatHis-SMA Study. PLoS ONE 2018, 13, e0201004. [Google Scholar] [CrossRef]
- Elsheikh, B.; Severyn, S.; Zhao, S.; Kline, D.; Linsenmayer, M.; Kelly, K.; Tellez, M.; Bartlett, A.; Heintzman, S.; Reynolds, J.; et al. Safety, Tolerability, and Effect of Nusinersen in Non-Ambulatory Adults With Spinal Muscular Atrophy. Front. Neurol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Landfeldt, E.; Abner, S.; Pechmann, A.; Sejersen, T.; McMillan, H.J.; Lochmüller, H.; Kirschner, J. Caregiver Burden of Spinal Muscular Atrophy: A Systematic Review. PharmacoEconomics 2023, 41, 275–293. [Google Scholar] [CrossRef] [PubMed]
- Chiriboga, C.A. Pharmacotherapy for Spinal Muscular Atrophy in Babies and Children: A Review of Approved and Experimental Therapies. Pediatr Drugs 2022, 24, 585–602. [Google Scholar] [CrossRef] [PubMed]
| Group | N | Sex (m/f) |
Age (years) |
Orphan drug (n) | ||
|---|---|---|---|---|---|---|
| Nusinersen | Risdiplam | OA | ||||
| SMA2 | 20 | 11/9 | 8 [6; 13]1 | 13 | 6 | 1 |
| SMA3 | 17 | 10/7 | 12 [10; 24] | 17 | - | - |
| Group | Funct. status (n) | RULM | HFMSE | FVC | ||
|---|---|---|---|---|---|---|
| N-S | S | W | (score) | (%) | ||
| SMA2 | 12 | 8 | - | 20 ± 101 (20)2 | 24 [11; 37]3 (7) | 56 ± 30 (14) |
| SMA3 | 1 | 14 | 2 | 35 [28; 37] (17) | 40 ± 13 (16) | 82 ± 24 (17) |
| Group | ΔRULM | ΔHFMSE | ΔFVC | ||||||
|---|---|---|---|---|---|---|---|---|---|
| r | p | n | r | p | n | r | p | n | |
| SMA2 | 0.26 | 0.27 | 9 | 0.32 | 0.48 | 7 | 0.11 | 0.71 | 14 |
| SMA3 | 0.49 | 0.16 | 10 | 0.09 | 0.75 | 16 | -0.09 | 0.74 | 17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




