1. Introduction
C-C chemokine receptor 5 (CCR5) is a G protein-coupled receptor (GPCR) which plays an important regulator for lymphocyte migration and activation.[
1] CCR5 is expressed on dendritic cells, macrophages, and natural killer cells, which are cells of the innate immune response, as well as on T and B cells of the adaptive immune response.[
2] CCR5 serves as a receptor for [macrophage-inflammatory protein-1α (MIP-1α); also known as C-C chemokine ligand 5 (CCL5)], MIP-1β [also known as C-C chemokine ligand 4 (CCL4)], and inflammatory β-chemokines, including [regulated on activation, normal T-expressed and secreted (RANTES); also known as C-C chemokine ligand 5, (CCL5)].[
2] CCR5 binds to these ligands and activates downstream signaling, which mediates the innate and adaptive immune response through the promotion of lymphocyte migration and the secretion of pro-inflammatory cytokines.[
3] These functions mediate the progression of tumors[
4,
5] and inflammatory diseases.[
3]
CCR5 is revealed as the principal co-receptor for the human immunodeficiency virus type 1 (HIV-1),[
6] and is involved in the disease progression in tumors[
4] and inflammatory diseases.[
7] CCR5 has been implicated in the inflammatory complications of coronavirus disease 2019 (COVID-19).[
8] Therefore, a CCR5 antagonist, maraviroc was first developed for acquired immunodeficiency syndrome (AIDS) therapy and is the only U.S. Food and Drug Administration (FDA)-approved drug for marketing.[
9] Furthermore, CCR5 antagonists, including monoclonal antibodies (mAbs) have been tested in various clinical trials.[
10] An anti-CCR5 mAb (PRO 140) has been developed and investigated for HIV, breast cancer, and COVID-19 in phase II or III trials.[
3,
11]
We have developed anti-mouse chemokine receptor mAbs against CCR1 (clone C
1Mab-6),[
12] CCR3 (clones C
3Mab-2, C
3Mab-3, and C
3Mab-4),[
13,
14,
15] CCR5 (clone C
5Mab-2),[
16] CCR8 (clones C
8Mab-1, C
8Mab-2, and C
8Mab-3),[
17,
18,
19] CXCR1 (clone Cx
1Mab-1),[
20] CXCR3 (clone Cx
3Mab-4),[
21] and CXCR4 (clone Cx
4Mab-1)[
22] using the Cell-Based Immunization and Screening (CBIS) method. Furthermore, we established anti-mouse chemokine receptor mAbs against CCR2 (clone C
2Mab-6),[
23] CCR3 (clones C
3Mab-6 and C
3Mab-7),[
24] CCR4 (clone C
4Mab-1),[
25] CCR9 (clone C
9Mab-24),[
26] CXCR6 (clone Cx
6Mab-1)[
27] using the N-terminal peptide immunization.
In this study, novel anti-mCCR5 mAbs were developed by the N-terminal peptide immunization.
2. Materials and Methods
2.1. Preparation of Cell Lines
L1210 (mouse lymphocytic leukemia) was obtained from the Cell Resource Center for Biomedical Research Institute of Development, Aging and Cancer Tohoku University (Miyagi, Japan). Chinese hamster ovary (CHO)-K1, LN229, and P3X63Ag8U.1 (P3U1) were obtained from the American Type Culture Collection (Manassas, VA).
The synthesized cDNA of mCCR5 (Accession No.: NM_009917, Eurofins Genomics KK, Tokyo, Japan) was subcloned into pCAGzeo and pCAGzeo-cPA vectors (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan). An anti-human podoplanin mAb (clone NZ-1) can detect the PA tag.[
28] The mCCR5 plasmids were transfected into CHO-K1 and LN229 cells, using a Neon transfection system (Thermo Fisher Scientific Inc., Waltham, MA).
2.2. Peptides
The N-terminal extracellular region of mCCR5 (1-MDFQGSVPTYSYDIDYGMS-19) plus C-terminal cysteine was synthesized by Eurofins Genomics KK. Subsequently, the keyhole limpet hemocyanin (KLH) was conjugated at the C-terminus of the peptide.
2.3 Production of Hybridomas
To develop mAbs against mCCR5, we intraperitoneally immunized one rat with 100 µg of the KLH-conjugated mCCR5 peptide (mCCR5-KLH) with Alhydrogel adjuvant 2% (InvivoGen). The hybridoma supernatants were screened using enzyme-linked immunosorbent assay (ELISA) with the mCCR5 peptide, followed by flow cytometry using CHO/mCCR5 and CHO-K1.
2.4. Antibodies
We previously developed an anti-mCCR5 mAb (C
5Mab-2) using the immunization of CHO/mCCR5 cells.[
16] Alexa Fluor 488-conjugated anti-rat IgG was purchased from Cell Signaling Technology, Inc. (Danvers, MA).
2.5. ELISA
The synthesized peptide (MDFQGSVPTYSYDIDYGMSC), was immobilized on Nunc Maxisorp 96 well immunoplates (Thermo Fisher Scientific Inc.). After blocking, plates were treated with supernatants of hybridomas, followed by peroxidase-conjugated anti-rat IgG (1:20,000 diluted; Sigma-Aldrich Corp., St. Louis, MO).
2.6. Flow Cytometric Analyses and Determination of Dissociation Constant (KD)
Cells were washed with 0.1% bovine serum albumin (BSA) in PBS (blocking buffer) and treated with primary mAbs for 30 min at 4°C. The cells were treated with Alexa Fluor 488-conjugated anti-rat IgG (1:2,000 or 1:200 for KD). The fluorescence data were collected using the SA3800 Cell Analyzer (Sony Corp.). The KD was subsequently calculated by GraphPad PRISM 6 (GraphPad Software, Inc., La Jolla, CA).
2.7. Western Blotting
Cell lysates from CHO-K1 and CHO/mCCR5-PA were treated with sodium dodecyl sulfate (SDS), separated on polyacrylamide gels and transferred onto polyvinylidene difluoride membranes (Merck KGaA). Membranes were incubated with 1 μg/mL of C
5Mab-4, C
5Mab-8, an anti-isocitrate dehydrogenase 1 (IDH1) (RcMab-1),[
29,
30] or 0.1 μg/mL of NZ-1. Membranes were then incubated with the peroxidase-conjugated anti-rat IgG.
3. Results
3.1. Development of Anti-mCCR5 mAbs Using N-Terminal Peptide Immunization
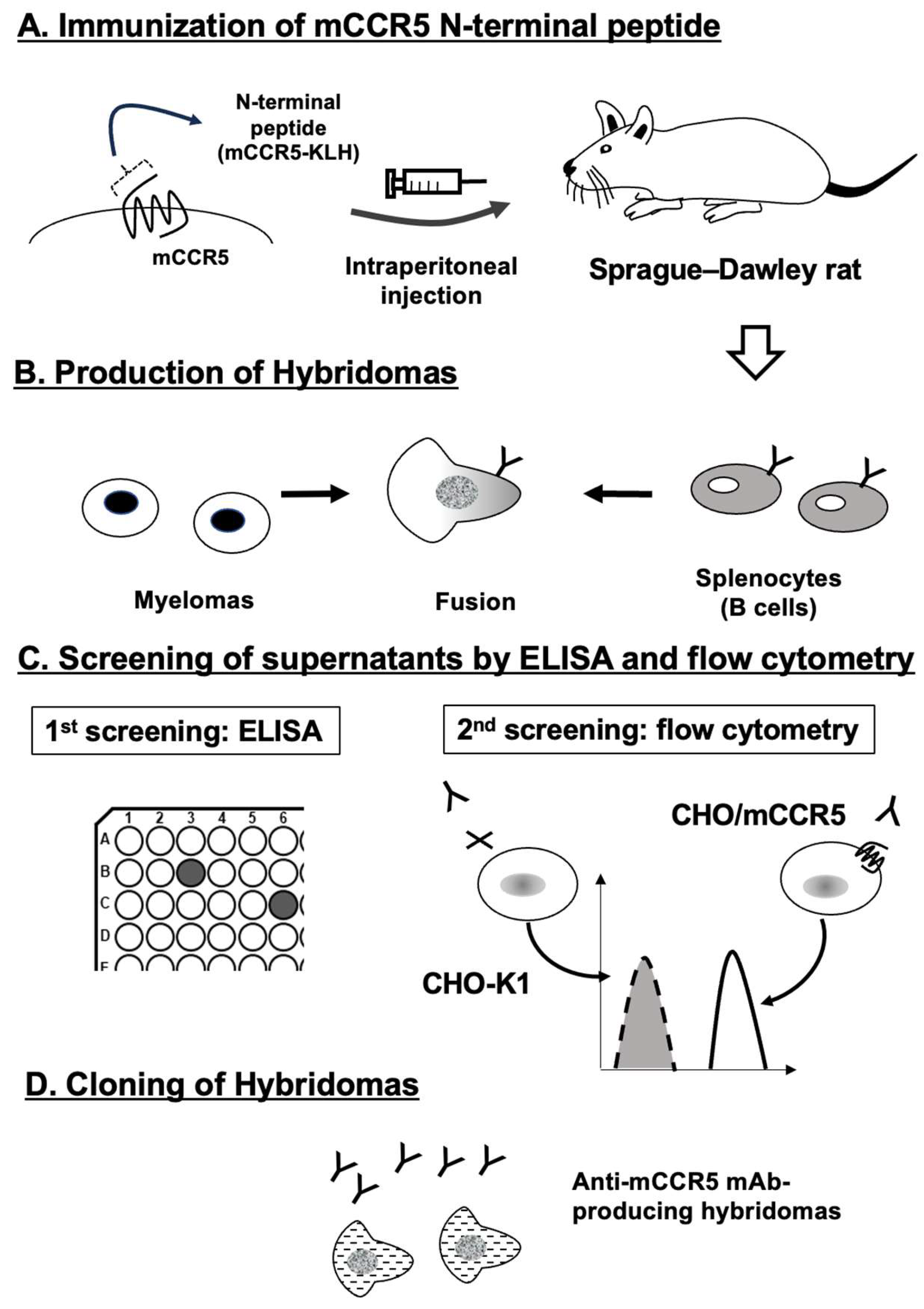
To develop mAbs against mCCR5, mCCR5-KLH was used as an immunogen (
Figure 1A). The rat splenocytes were fused with myeloma P3U1 cells (
Figure 1B). The naked mCCR5 peptide was used in ELISA to selected the hybridomas. The ELISA screening identified 68 out of 1438 wells (4.7%). Then, CHO/mCCR5-reactive and CHO-K1-non-reactive supernatants were further selected using flow cytometry (
Figure 1C). The flow cytometric screening identified 18 out of the 68 wells (26.5%), which exhibited strong signals to CHO/mCCR5 cells, but not to CHO-K1 cells. After the limiting dilution, C
5Mab-4 (rat IgG
2a, kappa) and C
5Mab-8 (rat IgG
1, kappa) were finally established (
Figure 1D).
3.2. Flow Cytometric Analysis Using C5Mab-4 and C5Mab-8
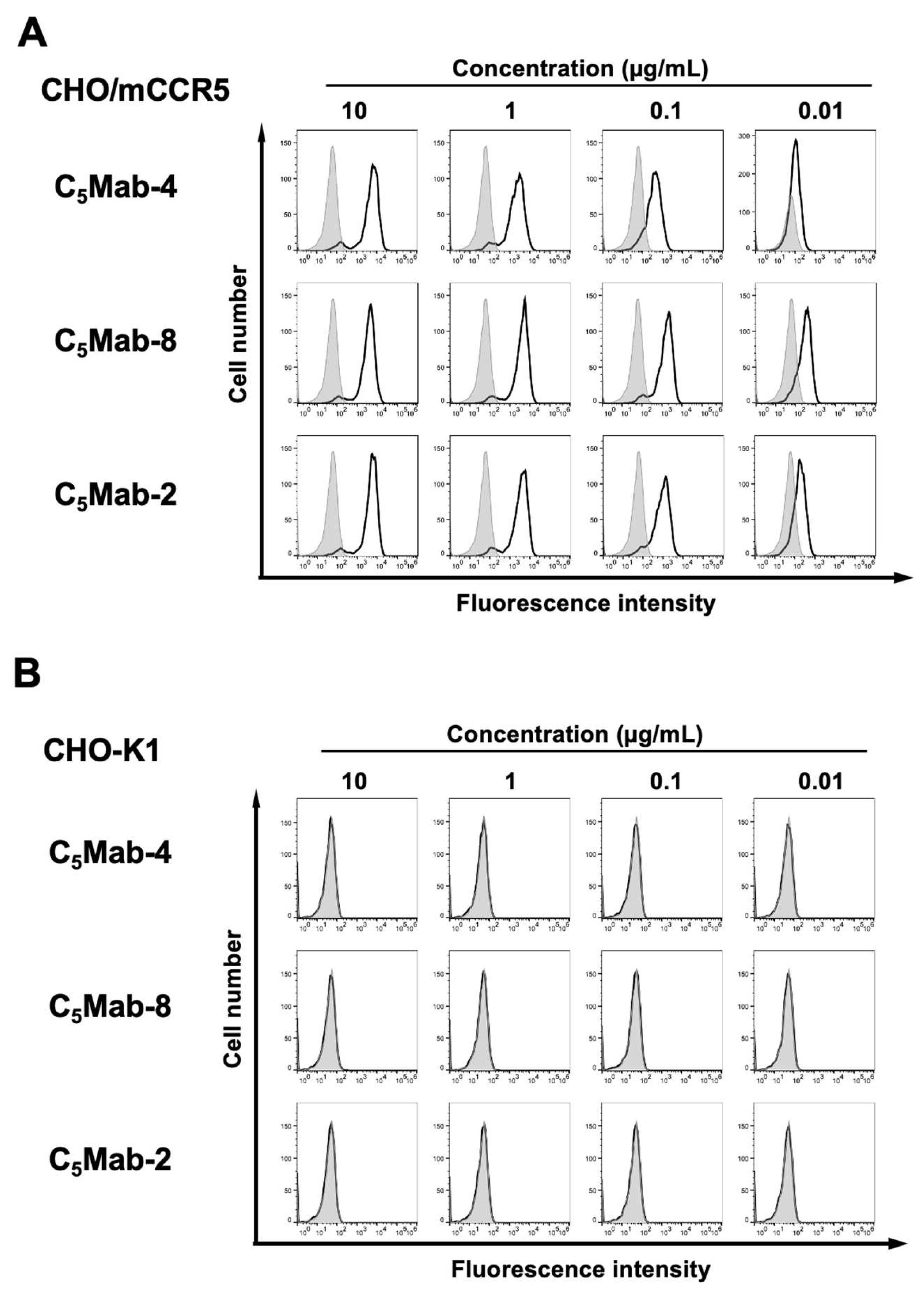
We conducted flow cytometry using three anti-mCCR5 mAbs: C
5Mab-2, C
5Mab-4, and C
5Mab-8 against CHO/mCCR5 and CHO-K1 cells. C
5Mab-2 was previously established by the CBIS method.[
16] Both C
5Mab-4 and C
5Mab-8 recognized CHO/mCCR5 cells dose-dependently at 10, 1, 0.1, and 0.01 μg/mL (
Figure 2A). C
5Mab-8 exhibited a superior reactivity against CHO/mCCR5 cells at 0.1 and 0.01 μg/mL compared to C
5Mab-2 and C
5Mab-4 (
Figure 2A). Parental CHO-K1 cells were not recognized by any mAbs even at 10 μg/mL (
Figure 2B). The superior reactivity of C
5Mab-8 was also observed in LN229/mCCR5 cells (supplementally
Figure 1).
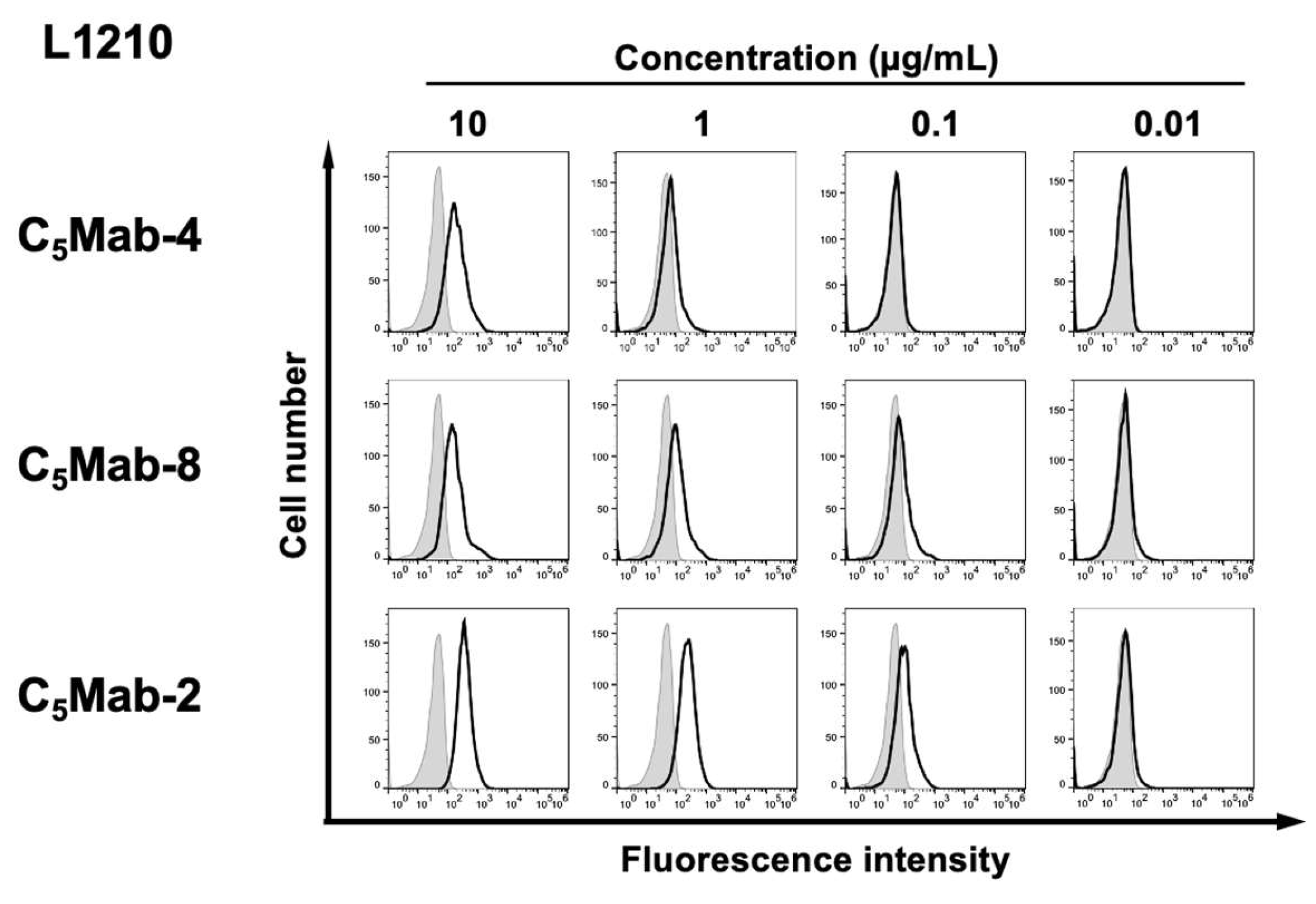
We next investigated the reactivity of C
5Mab-4 and C
5Mab-8 against an endogenously mCCR5-expressing cell line, L1210. C
5Mab-4 reacted with L1210 at more than 1 μg/mL (
Figure 3). C
5Mab-8 and C
5Mab-2 could react with L1210 at more than 0.1 μg/mL. These results suggested that C
5Mab-4 and C
5Mab-8 specifically recognize mCCR5, and are also useful for detecting endogenous mCCR5 by flow cytometry.
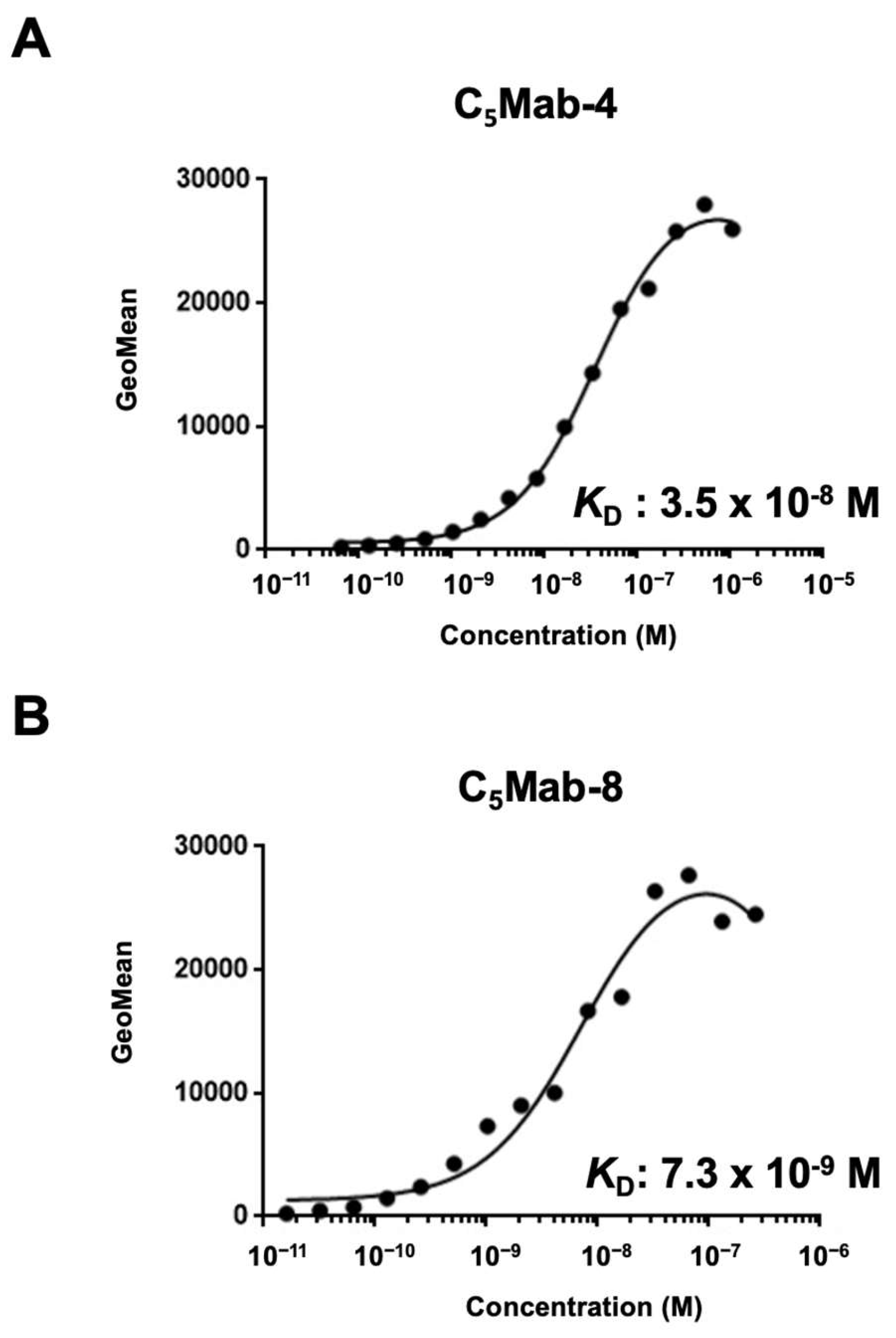
3.3. Determination of the Binding Affinity of C5Mab-4 and C5Mab-8 Using Flow Cytometry
To determine the
KD values of C
5Mab-4 and C
5Mab-8 against CHO/mCCR5, we conducted flow cytometry, and the geometric mean of the fluorescence intensity was plotted versus the concentration. The
KD values of C
5Mab-4 and C
5Mab-8 for CHO/mCCR5 were determined as 3.5 × 10
−8 M and 7.3 × 10
−9 M, respectively (
Figure 4). These results indicate that C
5Mab-4 and C
5Mab-8 possess the superior affinity to CHO/mCCR5 compared to C
5Mab-2 (4.3 × 10
−8).[
16]
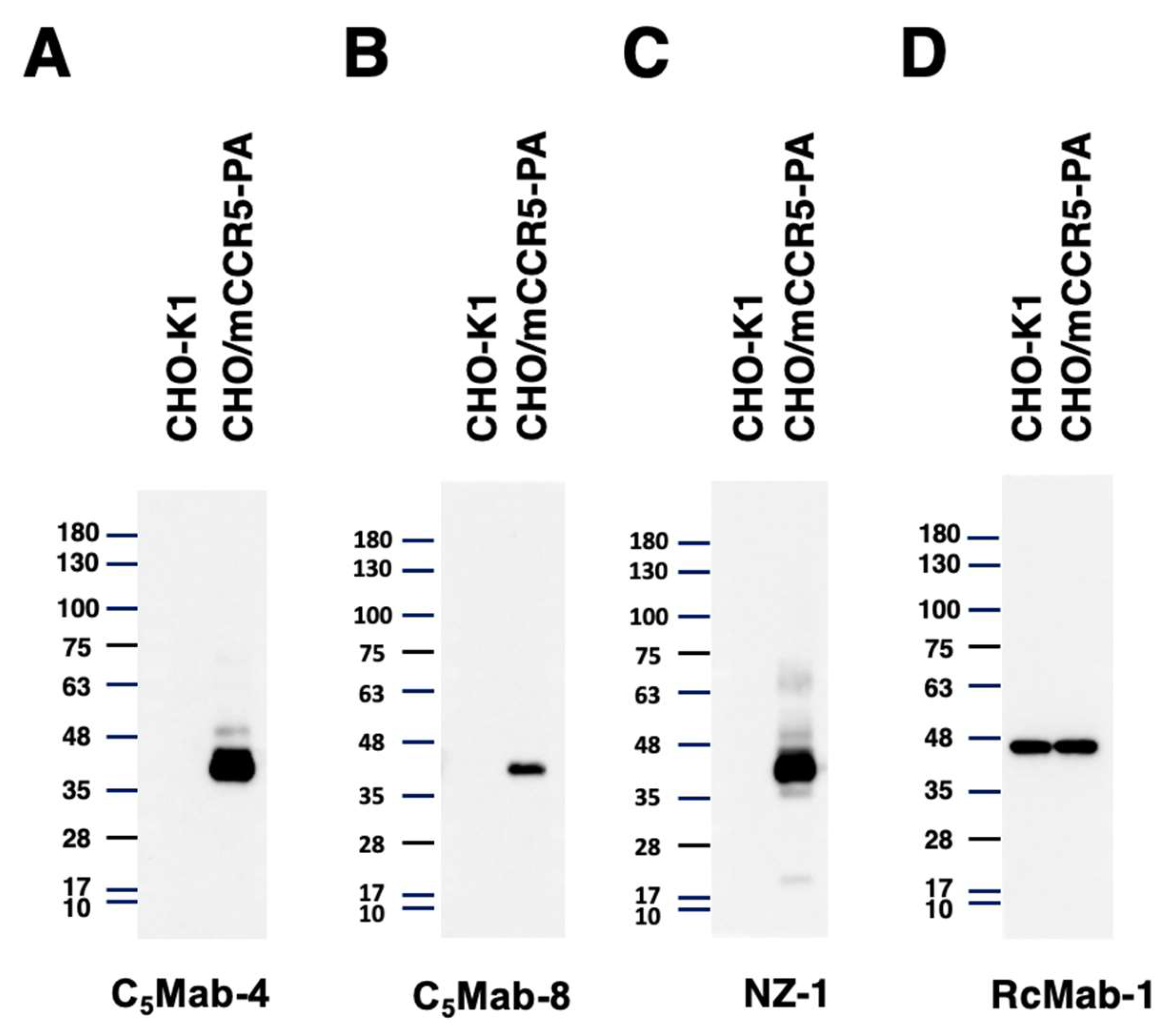
3.4. Western Blotting
Western blotting was performed to assess the reactivity of C
5Mab-4 and C
5Mab-8. Lysates of CHO-K1 and CHO/mCCR5-PA cells were probed. C
5Mab-4 (
Figure 5A) and C
5Mab-8 (
Figure 5B) detected mCCR5 as a ~40-kDa band. In contrast, C
5Mab-4 and C
5Mab-8 did not show any bands from the lysates of CHO-K1 cells. C
5Mab-4 exhibited a superior reactivity against CHO/mCCR5 cell lysate compared to C
5Mab-8 (
Figure 5A and B, the same exposure time). In contrast, C
5Mab-2 could not detect the CHO/mCCR5 cell lysate (data not shown). A similar molecular weight band was also detected by NZ-1 (
Figure 5C). These results suggest that C
5Mab-4 and C
5Mab-8 are useful for detecting mCCR5 by western blotting.
4. Discussion
In this study, we developed novel anti-mCCR5 mAbs (C
5Mab-4 and C
5Mab-8) using the N-terminal peptide immunization and showed the usefulness for flow cytometry (Figs. 2, 3, and 4) and western blotting (
Figure 5) to detect mCCR5. As we described in the result section, not all ELISA-positive supernatants recognized CHO/mCCR5 in flow cytometry. One of the possibilities is a modification of the CCR5 N-terminal region. The tyrosine sulfation of the human CCR5 N-terminal region enhances the binding affinity of the ligands and HIV envelope glycoprotein.[
31,
32] The human CCR5 N-terminal region possesses four potentially sulfated tyrosine residues, three of which are conserved in mCCR5 and included in the immunogen.[
33] Therefore, the epitope of C
5Mab-4 and C
5Mab-8 is thought to exclude the potentially sulfated tyrosine residues. We previously determined the epitope of Cx
6Mab-1 (an anti-mouse CXCR6 mAb) using 1× and 2× alanine scanning methods.[
34] We should determine the epitope C
5Mab-4 and C
5Mab-8 in future studies.
An understanding of the structural-based CCR activation is important for the development of therapeutic agents. Among the CCR family members, human CCR5 has been structurally solved in both inactive and active states.[
35,
36] Based on the nuclear magnetic resonance structure of CCL5 in complex with an N-terminal fragment (residues 1 to 27) of human CCR5 sulfated at residues Y10 and Y14, the sulfation at Y10 and Y14, which are conserved in mCCR5, was also revealed to be important for chemokine affinity.[
37] Furthermore, the structures of CCR8 in complex with either the antagonistic mAb or the endogenous ligand CCL1 were determined, which provides the specific activation mechanism by CCL1 and inhibition by mAb.[
38] We should determine the antagonistic activity of anti-mCCR5 mAbs, which could contribute to the understanding of the inactive state of mCCR5.
Unique functions of CCR5 in the neural systems have been reported using mouse models.[
39] By screening the 148 transgenic and knockout mutant mouse strains for contextual memory phenotypes, CCR5 was first identified to be involved in plasticity and memory. The CCR5-knockout mice exhibited a phenotype of memory enhancements for contextual conditioning after training.[
39] Furthermore, decreased CCR5 function enhances the long-term potentiation and hippocampus-dependent memory, while overexpression of CCR5 in neurons caused memory deficits in mice.[
39] These results indicated that CCR5 is a suppressor for cortical plasticity and hippocampus-dependent memory.
Additionally, an age-related increase in CCL5–CCR5 expression in neurons leads to impairments in memory linking in middle-aged mice that can be reversed by the FDA-approved CCR5 inhibitor, maraviroc.[
40] These results suggested that CCL5–CCR5 axis is a potential target for memory deficits. MAb therapies have been applied to neural disorders such as Alzheimer’s disease. The accumulation of soluble and insoluble aggregated amyloid-beta (Aβ) is thought to initiate or progress the pathologic processes.[
41] CCR5 plays an important role in modulating Aβ
1-42-induced learning and memory deficits in a mouse model, which suggests that CCR5 antagonists are a potential treatment to improve cognitive deficits associated with Alzheimer’s disease.[
42] Therefore, C
5Mab-4 and C
5Mab-8 could contribute to mouse pre-clinical studies to develop the treatment of memory deficits.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on
Preprints.org.
Institutional Review Board Statement
The animal study protocol was approved by the Animal Care and Use Committee of Tohoku University (Permit number: 2022MdA-001) for studies involving animals.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Proudfoot, A.E. Chemokine receptors: multifaceted therapeutic targets. Nat Rev Immunol 2002, 2, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, H.; Gurrola, T.; Berman, R.; Collins, M.; Sariyer, I.K.; Nonnemacher, M.R.; Wigdahl, B. Targeting CCR5 as a Component of an HIV-1 Therapeutic Strategy. Front Immunol 2021, 12, 816515. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Lan, T.; Wei, Y.; Wei, X. CCL5/CCR5 axis in human diseases and related treatments. Genes Dis 2022, 9, 12–27. [Google Scholar] [CrossRef] [PubMed]
- Hamid, R.; Alaziz, M.; Mahal, A.S.; Ashton, A.W.; Halama, N.; Jaeger, D.; Jiao, X.; Pestell, R.G. The Role and Therapeutic Targeting of CCR5 in Breast Cancer. Cells 2023, 12. [Google Scholar] [CrossRef]
- Aldinucci, D.; Casagrande, N. Inhibition of the CCL5/CCR5 Axis against the Progression of Gastric Cancer. Int J Mol Sci 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Alkhatib, G. The biology of CCR5 and CXCR4. Curr Opin HIV AIDS 2009, 4, 96–103. [Google Scholar] [CrossRef]
- Martin-Blondel, G.; Brassat, D.; Bauer, J.; Lassmann, H.; Liblau, R.S. CCR5 blockade for neuroinflammatory diseases--beyond control of HIV. Nat Rev Neurol 2016, 12, 95–105. [Google Scholar] [CrossRef]
- Chua, R.L.; Lukassen, S.; Trump, S.; Hennig, B.P.; Wendisch, D.; Pott, F.; Debnath, O.; Thürmann, L.; Kurth, F.; Völker, M.T.; et al. COVID-19 severity correlates with airway epithelium-immune cell interactions identified by single-cell analysis. Nat Biotechnol 2020, 38, 970–979. [Google Scholar] [CrossRef]
- Wood, A.; Armour, D. The discovery of the CCR5 receptor antagonist, UK-427,857, a new agent for the treatment of HIV infection and AIDS. Prog Med Chem 2005, 43, 239–271. [Google Scholar] [CrossRef]
- Scurci, I.; Martins, E.; Hartley, O. CCR5: Established paradigms and new frontiers for a 'celebrity' chemokine receptor. Cytokine 2018, 109, 81–93. [Google Scholar] [CrossRef]
- Qi, B.; Fang, Q.; Liu, S.; Hou, W.; Li, J.; Huang, Y.; Shi, J. Advances of CCR5 antagonists: From small molecules to macromolecules. Eur J Med Chem 2020, 208, 112819. [Google Scholar] [CrossRef] [PubMed]
- Ouchida, T.; Isoda, Y.; Nakamura, T.; Yanaka, M.; Tanaka, T.; Handa, S.; Kaneko, M.K.; Suzuki, H.; Kato, Y. Establishment of a Novel Anti-Mouse CCR1 Monoclonal Antibody C(1)Mab-6. Monoclon Antib Immunodiagn Immunother 2024. [Google Scholar] [CrossRef] [PubMed]
- Tateyama, N.; Asano, T.; Suzuki, H.; Li, G.; Yoshikawa, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Epitope Mapping of Anti-Mouse CCR3 Monoclonal Antibodies Using Flow Cytometry. Antibodies (Basel) 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Harigae, Y.; Li, G.; Asano, T.; Tanaka, T.; Suzuki, H.; Kaneko, M.K.; Kato, Y. C(3)Mab-2: An Anti-Mouse CCR3 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022, 41, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Asano, T.; Suzuki, H.; Tanaka, T.; Saito, M.; Li, G.; Goto, N.; Nanamiya, R.; Kaneko, M.K.; Kato, Y. C(3)Mab-3: A Monoclonal Antibody for Mouse CC Chemokine Receptor 3 for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2022, 41, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Tanaka, T.; Li, G.; Ouchida, T.; Kaneko, M.K.; Kato, Y. Development of a Sensitive Anti-Mouse CCR5 Monoclonal Antibody for Flow Cytometry Monoclon Antib Immunodiagn. Immunother 2024, in press. [Google Scholar]
- Suzuki, H.; Saito, M.; Asano, T.; Tanaka, T.; Kitamura, K.; Kudo, Y.; Kaneko, M.K.; Kato, Y. C(8)Mab-3: An Anti-Mouse CCR8 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022, 41, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Tanaka, T.; Asano, T.; Nakamura, T.; Yanaka, M.; Handa, S.; Komatsu, Y.; Harigae, Y.; Tateyama, N.; Nanamiya, R.; et al. C(8)Mab-2: An Anti-Mouse C-C Motif Chemokine Receptor 8 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022, 41, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Suzuki, H.; Tanaka, T.; Asano, T.; Kaneko, M.K.; Kato, Y. Development of an Anti-Mouse CCR8 Monoclonal Antibody (C(8)Mab-1) for Flow Cytometry and Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022, 41, 333–338. [Google Scholar] [CrossRef]
- Li, G.; Tanaka, T.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Cx1Mab-1: A Novel Anti-mouse CXCR1 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024, in press. [Google Scholar] [CrossRef]
- Ouchida, T.; Isoda, Y.; Tanaka, T.; Kaneko, M.K.; Suzuki, H.; Kato, Y. Cx(3)Mab-4: A Novel Anti-Mouse CXCR3 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024. [Google Scholar] [CrossRef] [PubMed]
- Ouchida, T.; Suzuki, H.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Cx(4)Mab-1: A Novel Anti-Mouse CXCR4 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2023. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Li, G.; Asano, T.; Saito, M.; Kaneko, M.K.; Suzuki, H.; Kato, Y. Development of a Novel Anti-Mouse CCR2 Monoclonal Antibody (C(2)Mab-6) by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022, 41, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Asano, T.; Suzuki, H.; Goto, N.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Establishment of Novel Anti-Mouse CCR3 Monoclonal Antibodies (C(3)Mab-6 and C(3)Mab-7) by N-terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022, 41, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Takei, J.; Suzuki, H.; Asano, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Development of a Novel Anti-Mouse CCR4 Monoclonal Antibody (C(4)Mab-1) by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022, 41, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Asano, T.; Suzuki, H.; Tanaka, T.; Yoshikawa, T.; Kaneko, M.K.; Kato, Y. Establishment of a Sensitive Monoclonal Antibody Against Mouse CCR9 (C(9)Mab-24) for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2023, 42, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, K.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Cx(6)Mab-1: A Novel Anti-Mouse CXCR6 Monoclonal Antibody Established by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022, 41, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Kaneko, M.K.; Kuno, A.; Uchiyama, N.; Amano, K.; Chiba, Y.; Hasegawa, Y.; Hirabayashi, J.; Narimatsu, H.; Mishima, K.; et al. Inhibition of tumor cell-induced platelet aggregation using a novel anti-podoplanin antibody reacting with its platelet-aggregation-stimulating domain. Biochem Biophys Res Commun 2006, 349, 1301–1307. [Google Scholar] [CrossRef]
- Kato, Y. Specific monoclonal antibodies against IDH1/2 mutations as diagnostic tools for gliomas. Brain Tumor Pathol 2015, 32, 3–11. [Google Scholar] [CrossRef]
- Ikota, H.; Nobusawa, S.; Arai, H.; Kato, Y.; Ishizawa, K.; Hirose, T.; Yokoo, H. Evaluation of IDH1 status in diffusely infiltrating gliomas by immunohistochemistry using anti-mutant and wild type IDH1 antibodies. Brain Tumor Pathol 2015, 32, 237–244. [Google Scholar] [CrossRef]
- Tsui, C.K.; Gupta, A.; Bassik, M.C. Finding host targets for HIV therapy. Nat Genet 2017, 49, 175–176. [Google Scholar] [CrossRef]
- Farzan, M.; Mirzabekov, T.; Kolchinsky, P.; Wyatt, R.; Cayabyab, M.; Gerard, N.P.; Gerard, C.; Sodroski, J.; Choe, H. Tyrosine sulfation of the amino terminus of CCR5 facilitates HIV-1 entry. Cell 1999, 96, 667–676. [Google Scholar] [CrossRef]
- Ludeman, J.P.; Stone, M.J. The structural role of receptor tyrosine sulfation in chemokine recognition. Br J Pharmacol 2014, 171, 1167–1179. [Google Scholar] [CrossRef]
- Isoda, Y.; Tanaka, T.; Suzuki, H.; Asano, T.; Nakamura, T.; Yanaka, M.; Handa, S.; Komatsu, Y.; Okuno, S.; Takahashi, N.; et al. Epitope Mapping of an Anti-Mouse CXCR6 Monoclonal Antibody (Cx(6)Mab-1) Using the 2 × Alanine Scanning Method. Monoclon Antib Immunodiagn Immunother 2022, 41, 275–278. [Google Scholar] [CrossRef]
- Isaikina, P.; Tsai, C.J.; Dietz, N.; Pamula, F.; Grahl, A.; Goldie, K.N.; Guixà-González, R.; Branco, C.; Paolini-Bertrand, M.; Calo, N.; et al. Structural basis of the activation of the CC chemokine receptor 5 by a chemokine agonist. Sci Adv 2021, 7. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.; Zhu, Y.; Li, J.; Chen, Z.; Han, G.W.; Kufareva, I.; Li, T.; Ma, L.; Fenalti, G.; Li, J.; et al. Structure of the CCR5 chemokine receptor-HIV entry inhibitor maraviroc complex. Science 2013, 341, 1387–1390. [Google Scholar] [CrossRef] [PubMed]
- Abayev, M.; Rodrigues, J.; Srivastava, G.; Arshava, B.; Jaremko, Ł.; Jaremko, M.; Naider, F.; Levitt, M.; Anglister, J. The solution structure of monomeric CCL5 in complex with a doubly sulfated N-terminal segment of CCR5. Febs j 2018, 285, 1988–2003. [Google Scholar] [CrossRef]
- Sun, D.; Sun, Y.; Janezic, E.; Zhou, T.; Johnson, M.; Azumaya, C.; Noreng, S.; Chiu, C.; Seki, A.; Arenzana, T.L.; et al. Structural basis of antibody inhibition and chemokine activation of the human CC chemokine receptor 8. Nat Commun 2023, 14, 7940. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Greenhill, S.; Huang, S.; Silva, T.K.; Sano, Y.; Wu, S.; Cai, Y.; Nagaoka, Y.; Sehgal, M.; Cai, D.J.; et al. CCR5 is a suppressor for cortical plasticity and hippocampal learning and memory. Elife 2016, 5. [Google Scholar] [CrossRef]
- Shen, Y.; Zhou, M.; Cai, D.; Filho, D.A.; Fernandes, G.; Cai, Y.; de Sousa, A.F.; Tian, M.; Kim, N.; Lee, J.; et al. CCR5 closes the temporal window for memory linking. Nature 2022, 606, 146–152. [Google Scholar] [CrossRef]
- Heidebrink, J.L.; Paulson, H.L. Lessons Learned from Approval of Aducanumab for Alzheimer's Disease. Annu Rev Med 2024, 75, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Salinas, S.; Cornell, J.; Udoh, I.B.; Shen, Y.; Zhou, M. CCR5 regulates Aβ(1-42)-induced learning and memory deficits in mice. Neurobiol Learn Mem 2024, 208, 107890. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).