1. Introduction
Cancer is a complex disease with multiple causes and outcomes. Thus, the treatment of cancer, whether curative or palliative, employs a broad spectrum of methodologies. During the past decades, the molecular understanding of cancer has increased tremendously. The discovery of the DNA structure as the genetic code [
1], started a revolution in biomedicine that sees Personalized Medicine at the “harvest time”, when the newly acquired knowledge of links between genes and diseases can be applied to diagnosis and treatment. In oncology, this paradigm shift also provided the rationale for mechanism-based interventions that may complement or replace non-discriminatory methods, such as cytostatic drugs, radiation and surgery, with procedures that are assumed to achieve better cure, cost efficacy and reduced toxicity [
2].
The molecular biology revolution has provided ample opportunities for discoveries resulting in new diagnostics and treatments [
3]. The increased capacity and quality of sequencing technologies, combined with a dramatic reduction of costs, has fostered the emergence of new research areas that combine advanced cell biology, informatics, computer science and medicine. At the same time, the extended biological knowledge made possible by scientific and technological advances has created the demand for new organisational structures, terminologies and cultures in the translational space between research and healthcare.
The past two decades has seen an exponential growth in the number of scientific articles including “personalized medicine” in their title, with a total of 115 464 articles published until March 2024 and more than 103 198 published after 2010 (PubMed, March 2024). However, the increased popularity of the name does not correspond to a common understanding of the concept. Many attempts have been made towards a comprehensive definition of Precision and Personalised Medicine [
4]. A common reference is the the paper by Schleidgen et al.[
5] that reviews 2457 articles containing the terms precision medicine. The authors argue that a common understanding of the definition of Precision Medicine is important for regulatory purposes. They posit that Precision Medicine is nothing new since medicine always has been individualized and tailored, and provide more technical understanding of precision medicine as the stratification of patients into treatment subgroups based on genetic or other omics related information on the patient and disease. In his article
Systems Biology and P4 Medicine: Past, Present, and Future [
6] - Hoods expands the personalized concept within a systems medicine approach that combines predictive, preventive, personalized, and participatory perspectives into a so called “P4 medicine”. This comprehensive approach aspires to include multiple measurable biological markers and diagnostic methods in order to detect disease early and tailor treatments based on computational analysis of the specific patient dataset. According to The European Union’s Horizon 2020 Advisory Group the concept of Precision Medicine can be described as "the characterization of individuals phenotypes and genotypes (e.g., molecular profiling, medical imaging and lifestyle data) for tailoring the right therapeutic strategy for the right person at the right time, and/or to determine the predisposition to disease and/or to deliver timely and targeted prevention" [
7].
A less comprehensive approach is implied in the concept of Precision Medicine that focuses on increased accuracy in diagnostic classification and treatment matching. Precision medicine strives to realise the medical principle “to provide the right treatment to the right patient at the right time” and has been particularly associated to the genomic stratification of patients into more exact disease subgroups. The concept has become a central focus of the expanding life science industry [
8].
With the increased technical development, medical science is increasingly transformed from an intimate private practice between doctors and patient to a complex system comprising multiple disciplines, roles, and organizational structures. The resource intense and difficult quest for explaining biological phenomena has fostered a scientific culture concentrated on specific discoveries [
9,
10]. In parallel with concepts of precision, personalization and individualisation oriented towards the diagnostic and pharmaceutical aspects, the concepts of systems and networks have also been popularised. Systems biology has emerged as a popular conceptualisation for the multiplicity of interrelated causal connections explaining macro phenomena such cancer cell phenotype. Networks together with systems medicine illustrate a form of medicine that can not depend on the individual judgement of a single medical specialist but requires the concerted involvement of multiple experts that can provide different layers of information to the same patient case [
11].
Despite multiple uses of the word “Personalized” and associated terms, an established convention refers to “Personalization” as the capacity to tailor treatments based on the identification of molecular biomarkers that drive disease. While the explosive development of genomic technologies substantiates the hope that this concept may be rapidly translated into healthcare practices, the introduction of personalized medicine on a global scale is still a distant goal. Nevertheless, many actors have made considerable progress, which has provided a deeper understanding of what genomic information can do to improve patient care. Their experience has given important insights into which forms of organization are better suited to manage a research and healthcare environment invested in personalized medicine [
12].
2. Methodology
In this review article involves exploration of published scientific literature, focusing on identifying key concepts, theories, evidence sources, and research gaps related to personalized Cancer Medicine. This review is informed by identified areas if interest from professionals of the field. This review is based on published articles collected from five different sources: PubMed, Google Scholar, Research Gate, Web of Science and Science direct. With the following inclusion criteria:
The following terms where searched for in whole articles, titles and abstracts: “Personalized” OR “Personalised” OR “Precision” AND “Medicine” AND “Cancer” OR “Oncology”; AND “Informatics” AND “Ethics” AND “Management” AND “Organisation” AND “Translation” AND “Implementation”.
Two hundred seventy six articles out of 4294 hits were selected for closer review of abstracts and conclusions. From this pool, 89 articles were singled out for detailed reading. Ulterior articles where identified from references in these articles based on relevance. Other articles where chosen based on authors prior knowledge. Articles where selected based on relevance to describe key organizational factors for implementation or based on significance in describing personalised or precision cancer medicine. Specific importance was given to sections of articles that disclose contextual information on the conditions leading to the successful or failed translation of a specific method, treatment or other technology associated with personalized medicine.
3. A Brief History of Personalized Cancer Medicine
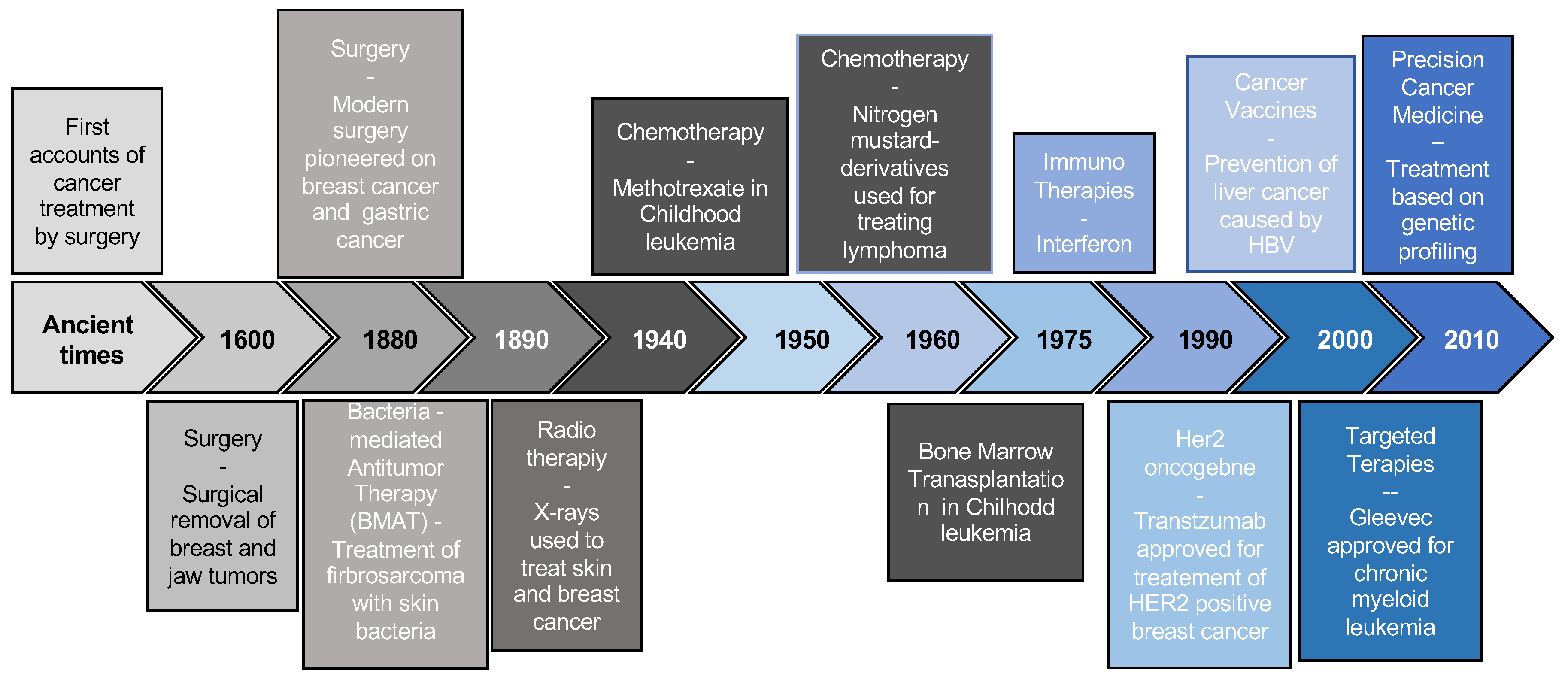
The concept of PCM can be traced to the mid 20th century when scientists first began to explore the relationship between genetics and cancer. [
Figure 1. The History of Cancer Therapies]. Of course the discovery of DNA as the genetic code in 1953 and subsequent developments to read the DNA base sequence in the 1980ies were instrumental starting points for what can be done in molecular diagnostics today. However, only in the 1990s did PCM begin to take shape thanks to the advent of new technologies, such as large-scale DNA sequencing. By the end of the 20th C. several complex causes of cancer could be descried [
13]. In the early 2000s, the completion of the Human Genome Project delivered a new research landscape allowing scientists to identify specific genetic mutations that contribute to the development and progression of the disease. Notable early examples of the successful use of this resource were the discovery that a subgroup of lung cancers carrying a mutation in EGFR responded to treatment with gefitinib therapy [
14]; the development of Her-2/Neu antibodies for the treatment of breast cancer [
15] and the development the small molecule inhibitor, Imitunib [
16], targeting the fusion gene BRC/ABL in Chronic Myelogenous Leukemia. However, in spite of the optimistic hope of quickly finding cures for all forms of cancer, the translation of rapidly increasing biological knowledge into efficient medicine has proven very challenging and time-consuming.
The US Food and Drug Administration (FDA) approved the use of Glivec, the first cancer drugs based on a patient’s genetic profile in the early 2000, marking a significant milestone in developing PCM [
17]. Almost two decades later, in 2017, the use of pembrolizumab was approved by the FDA based on the presence a genetic biomarker rather than the anatomical origin of the tumor (2017). Over the next decade, advances in genomic technologies and big data analysis have enabled the development of increasingly sophisticated methods for characterising cancer at the molecular level, leading to the approval of many new targeted therapies for cancer [Table 1, List of established targeted therapies in order of pioneering scientific publications].
Table 1.
Chronology of the development of current established targeted therapies in cancer based on date of pioneering articles they are cited.
Table 1.
Chronology of the development of current established targeted therapies in cancer based on date of pioneering articles they are cited.
| Targeted therapies
|
Pioneering articles |
Cancer/s |
| HER2-Target Therapies |
(Slamon et al)[18] |
Breast cancer |
| EGFR Inhibitors |
(Lynch et al., 2004)[19] |
Non-Small Cell Lung Cancer; Colorectal Cancer |
| MEK Inhibitors |
(Adjei et al., 2008)[20] |
Melanoma with BRAF mutation; Non-Small Cell Lung Cancer |
| FGFR Inhibitors |
(Fischer et al., 2008)[21] |
Urothelial bladder cancer with FGFR alterations; Cholangiocarcinoma with FGRFR alterations |
| PARP Inhibitors |
(Fong et al., 2010)[22] |
Ovarian cancer, Breast cancer and Pancreatic cancer with BRCA mutations |
| BRAF Inhibitors |
(Flaherty et al., 2010)[23] |
Melanoma with BRAF mutations; Colorectal cancer with BRAF mutations |
| ALK Inhibitors |
(Kwak et al., 2010)[24] |
Non-Small Cell Lung Cancer |
| BRD4 Inhibitors |
(Delmore et al., 2011)[25] |
No specific cancer |
| ROS1 Inhibitors |
(Shaw et al., 2014)[26] |
Non-Small Cell Lung Cancer |
| CDK4/6 Inhibitors |
(Finn et al., 2016)[27] |
HER2 negative breast cancer |
| RET Inhibitors |
(Drilon et al., 2020)[28] |
Medullary thyroid cancer with RET mutations; Lung cancer with RET rearrangements |
| TRK Inhibitors |
(Drilon et al., 2020)[29] |
Cancers with NTRK gene fusions, various cancer types, paediatric cancers and solid tumours |
| KRAS Inhibitors |
(Hong et al., 2020)[30] |
Colorectal cancer; Lung cancer |
Today, “personalization” has become a cornerstone of cancer care, and many healthcare providers have integrated personalised approaches into their routine clinical practice with the hope to improve patient outcomes and quality of life. Despite these advances, much work remains to realise the potential of PCM.
4. The Challenges of Personalized Cancer Medicine (PCM)
The effective treatment of cancer faces many challenges. While
early detectionis crucial for effective cancer treatment, many cancers are not detected until they have advanced to later stages [
31]. The development of effective treatment is complicated by
cancer heterogeneity, which refers to differences between individual cancers and even within different parts of the same tumour [
32]. Cancer specialists are also faced with the enormous adaptability of cancer cells and their capacity to become resistance to treatment.
Drug resistance is a common occurrence [
33], which motivates the development of several alternative lines of treatment. A fundamental challenge within cancer is
immune evasion [
34], which entails the capacity of cancer cells to evade the immune system, making it difficult for the body to fight cancer effectively. Current trends in cancer treatment strategies are increasingly oriented towards the reactivation of the immune system’s ability to identify cancer cells as a means for a cure. Several successful studies have shown the potential of immune therapies [
35], often in combination with conventional treatments. However, much still needs to be done regarding clinical effectiveness, especially in minimizing the risk of negative immune reactions with potentially lethal effects on patients. Finally, the ability to mitigate the
toxicity of treatments [
36] is still a major factor within all established cancer care plans. Many cancer treatments can cause significant side effects, making it difficult for patients to tolerate the treatments and thus limiting the treatment options due to patients’ performance status and other risk factors.
4.1. Achieving Precision within PCM
The effect of cancer heterogeneity on cancer treatments and personalised cancer medicine is significant because it can lead to treatment resistance, recurrence, and therapeutic failure. To address these challenges, personalised cancer medicine aims to identify specific genetic and molecular changes in each patient’s tumor to inform the development of targeted and effective treatments. The case with cancer heterogeneity shows a strong paradigm change and a continuous ongoing transformation in treatment concepts between older treatment paradigms that are primarily focused on the anatomic detection of cancer and the subsequent removal (surgery, radiotherapy) and killing of cancer cells (chemotherapy). The clinical actionability of cancer heterogeneity is still a developing field.
4.2. Enabling Tools
Genomics is at the forefront of precision medicine revolution and plays a fundamental role in today’s precision cancer medicine, offering comprehensive insights into the genomic alterations that drive tumor development, progression, and response to treatment. Through the utilization of high-throughput genomic technologies like next-generation sequencing (NGS), researchers and clinicians are now equipped to employ personalized approaches for cancer patients. Genomic profiling has revolutionized cancer diagnosis and classification by surpassing the limitations of traditional histopathology methods. By analyzing various genomic alterations, it enables a more accurate characterization of tumors. The Cancer Genome Atlas (TCGA) project has been instrumental in enhancing our understanding of cancer subtypes and refining diagnosis based on genomic profiles. Moreover, genomic profiling assists in distinguishing primary tumors from metastatic lesions, aiding in treatment decisions and identification of tumor origins. Genomic alterations also serve as valuable prognostic and predictive biomarkers, facilitating the development of personalized treatment strategies. By identifying these biomarkers through genomic profiling, clinicians can tailor treatment plans to individual patients. Targeted therapies that exploit cancer-associated genomic alterations have been made possible through genomic profiling, optimizing treatment selection while minimizing adverse effects. Liquid biopsies and genomic techniques for monitoring minimal residual disease offer non-invasive approaches to dynamically track tumor evolution and guide therapeutic interventions. Despite the rapid progress with new emerging omics technologies that will provide a more comprehensive spatial biological understanding of the tumour, genomics will continue to empower precision cancer medicine by providing a comprehensive understanding of tumor biology, aiding in diagnosis, treatment selection, and disease monitoring [
37].
4.2.1. Molecular Pathology
Molecular pathology of cancer refers to the use of molecular and genetic techniques providing insights in the biology of cancer and creates the conditions for informed personalised cancer treatment. It involves the analysis of DNA, RNA, and proteins from cancer tissues and biopsy samples to identify specific genetic alterations, such as mutations and chromosomal rearrangements, that are significant factors for the emergence and progression of cancer [
38,
39].
4.2.2. The Use of Imaging Modalities in Personalised Cancer Medicine
Imaging plays a crucial role in personalised cancer medicine by providing detailed visual information about a patient’s tumour and surrounding tissues. This information helps physicians determine the size, location, and extent of the cancer, as well as how it is affecting normal tissue. Computed tomography (CT), magnetic resonance imaging (MRI) and hybrid imaging using Positron emission tomography (PET) combined with CT (PET/CT) are the main imaging modalities used for diagnostic purposes in oncology [
40]. Advancement in precision oncology include the use of imaging for diagnosis, treatment planning, and monitoring response to therapy [
41]. Several parallel imaging techniques are adopted throughout the cancer healthcare pathway. This includes ultrasonography enabling real time imaging for obtaining biopsy specimen and for directing local treatments. Routine surveillance of systemic treatment is usually performed by computed tomography (CT) as the first line imaging modality. These modalities for cancer evaluation are increasingly complemented by molecular imaging techniques, that provides information specific to the tumour, that indicate improvements to the manouverability and assessment of potential cancer treatments [
42]. Furthermore the systematization of imaging data using computational models, AI and machine learning may further increase precision beyond subjective human assessment [
43].
4.2.3. Cellular Models for Drug Sensitivity Profiling
In recent years the technology of using 3D cell cultures that can replicate the function of human organs has become an efficient tool in actionable cancer biology and precision medicine [
44]. A live environment for cancer cells provides a more accurate model to study cancer cell behaviour such as growth factors, drug reactions and resistance. In personalized medicine retrieving cancer cells from patients to test drug response have shown many promising results for several cancer types [
45].
4.2.4. Biobanking
The introduction of personalised cancer medicine has led to a shift in the way biobanking and biopsy procedures are performed and analyzed, with a greater focus on obtaining precise and informative samples, and utilizing advanced technologies for analysis [
46]. Biobanking involves collecting, storing, and managing biological samples (such as blood, tissue, or saliva) from patients with cancer. These samples are used to gather information about the patient’s tumor, including its genetic makeup, which can then be used for personalized treatment decisions [
47]. The procedure is virtually the same with the introduction of personalized cancer medicine however it has had a significant impact on the way biopsies are performed and analyzed. In personalized cancer medicine, biopsies play a crucial role in providing information about the patient’s tumor and its genetic makeup, which is used for informed treatment decisions. Biopsies are now often performed with a greater level of precision, as they are guided by information gathered from genomic sequencing, imaging, and other tests. To maximize the amount of informative data, there is an increased interest in obtaining biopsy samples from specific parts of the tumor, such as areas with the highest levels of genetic abnormalities. In addition, the analysis of biopsy samples has become increasingly complemented by techniques such as next-generation sequencing (NGS) that allows comprehensive analysis of the patient’s tumor at the genetic level. With the introduction of fine needle biopsies or Fine Needle Aspiration (FNA) access to tumour samples has been improved with less complications for patients [
48]. This less invasive technique provides the opportunity for repeated biopsies providing material for sequential and more precise diagnostics by following tumor progression, treatment effects and possible resistance.
4.2.5. AI in Personalised Cancer Medicine
The field of personalised cancer medicine is rapidly being transformed by the emerging technologies based on Artificial intelligence (AI) that are enabling a more efficient and accurate analysis of large amounts of data, including genomic data, imaging data and other clinically applicable data sources with different modalities [
49]. AI algorithms can help identify novel therapeutic targets and predict treatment response, leading to more personalized treatment decisions for cancer patients [
50,
51,
52,
53].
4.3. PCM Today
This chapter will provide a brief review of examples where the concept of PCM has been implemented in clinical practice. Organizational insights from leading PCM studies.
Access to new treatments – a principal determining factor– The possibility to access new potentially more effective drugs and drug combinations is often described as a major obstacle in the implementation of precision medicine mentions organizational challenges as well as financial, legislative, and policy-related factors making it difficult to design clinical drug studies for individually tailored combination therapies. Proof-of-concept studies with single patients have demonstrated promising results and can inform the design of formal clinical trials [
54]. According to Malani et al.[
55] the use of a functional precision medicine tumour board (FPMTB) approach and the ex vivo drug response testing described in these studies can help establish international collaborations between public and private stakeholders to overcome current obstacles in the application of individually tailored precision medicine.
The advent of data-driven medicine, catalyzed by the introduction of systems medicine, has catalyzed a paradigm shift towards patient-centric cancer care. Despite the seeming tautology as all care and medicine is centred on benefiting the patient, patient involvement within the current complex translation scenario entails securing patient involvement on several parallel levels through different means. Patients now play an active role as agents in their treatment journey, contributing health data for early detection and intervention. Their involvement extends to clinical study design, enhancing research efficacy. However, challenges persist. Organizational hurdles impede seamless integration of patient-generated data, while privacy concerns loom. Moreover, while patient engagement holds potential, evidence gaps hinder informed implementation. The healthcare system must address these challenges to fully leverage personalized medicine’s benefits and empower patients effectively.
Table 2.
Organizational insights from leading PCM studies. The most prominent PCM characterized clinical trials bring both scientific and organisational insights. This table singles out some key observations based on the clinical trial design and organizational model selected for the study from a selection of published papers from the studies.
Table 2.
Organizational insights from leading PCM studies. The most prominent PCM characterized clinical trials bring both scientific and organisational insights. This table singles out some key observations based on the clinical trial design and organizational model selected for the study from a selection of published papers from the studies.
| PCM Study
|
Selected Organizational Insights |
| ComboMATCH [56](Meric-Bernstam et al., 2023) |
ComboMATCH, a precision medicine initiative, is developed collaboratively with NCI/CTEP and academia, fostering interdisciplinary engagement. It leverages the CTEP Investigational New Drug program, integrating resources like ETCTN and PDXNet. This patient-centric approach utilizes routine clinical genomic profiling for therapy selection, emphasizing teamwork and biomarker-directed therapy to enhance patient outcomes. |
| DRUP, 2023 [57](Geurts et al., 2023) |
Drug repurposing and biomarker-driven treatment selection which required collaboration with pharmaceutical companies to obtain access to the drugs and to ensure that they were provided to patients in a timely and consistent manner. |
| MASTER, 2021 [58](Cecchini et al., 2019) |
Algorithm-based treatment selection and comprehensive molecular profiling with multidisciplinary team approach. Showcasing potential of algorithm-based treatment selection and comprehensive molecular profiling. |
| Mi-ONCOSEQ, 2021 [59](King et al., 2021) |
Comprehensive genomic profiling using multidisciplinary team approach. Need for a coordinated effort between multiple centers to achieve a sufficient sample size. |
| WINTHER, 2019
[60](Rodon et al., 2019) |
The trial used a novel “window of opportunity” design. Coordinated effort between multiple centers to achieve a sufficient sample size. |
| MOSCATO, 2017 [61](Massard et al., 2017) |
Emphasis on collaboration, extensive coordination is required. Importance of data sharing and collaboration in precision medicine research. |
| SHIVA, 2015 [62](Le Tourneau et al., 2015) |
Importance of a coordinated, multidisciplinary approach to clinical research. |
5. Determining Factors in PCM
The majority of published scientific literature on personalised cancer medicine is focused on the revelation of significant positive results that provide insights into cancer biology and medical outcomes. Methodology sections and comment sections can include indirect comments on broader organizational, financial, legal or even cultural determinants that the authors find significant for the specific work or the field in general. These statements are important evidence of what the field itself perceives as significant determinants of the present conditions. In the article “Personalised Medicine barriers facilitators healthcare systems” Stefanicka-Wojtas and Kurpas explore the needs, barriers, and facilitators of implementing Personalized Medicine (PM) in European healthcare systems [
63]. The barriers for the implementation of precision medicine are categorized into seven areas of need and ten key stakeholders.
5.1. Health Policy Factors
The implementation of personalised medicine is resource intensive and requires substantial changes in research and healthcare policy regulations [
64]. Governmental support is vital for the success in paradigmatic change in health care. Health policies driven by governments impact the conditions for implementing personalized cancer medicine by ensuring that it is delivered in a responsible, ethical, and effective manner. Policies generally target patients access to healthcare, the conditions for production and structure of high quality healthcare and research data, patient rights, data privacy and the regulation of the life science, pharmaceutical and health care industry. Several government initiatives launched during the recent decades have promoted Personalized Cancer Medicine. In 2015 The US President Barack Obama made a State of the Union Address identifying personalised medicine as a prioritised area for healthcare
[1]. The German Cancer Research Initiative DKFZ
[2], and Cancer Research UK
[3] are other examples of large scale national investments towards the introduction of personalised medicine in different fields. Other significant national initiatives are the Japanese genetic profiling multicentre study SCRUM-Japan MONSTAR-SCREEN-2
[4] (2015)and the European initiative Cancer Core Europe
[5]. The US National Cancer Institute (NCI) has initiated several programs to promote personalised cancer medicine, including the Precision Medicine Initiative (PMI)
[6] and the Cancer Genome Atlas (TCGA)
[7]. In Europe, the Innovative Medicines Initiative (IMI) is working to advance personalised medicine through research and the development of new diagnostic and treatment strategies. The UKs National Health Service (NHS) is also a promoter of personalised cancer medicine through, for example, the NHS Cancer Plan and the 100,000 Genomes Project.
A primary role of public healthcare policies should be to ensure that all patients have access to personalized cancer medicine, regardless of their financial status or location. Health insurance policies should provide coverage for personalised cancer medicine. Moreover before treatment patients should be fully informed about the benefits and risks and give their informed consent. Access to healthcare also encompasses that patients privacy and security can be guaranteed. At the same time, policies also need to support and fund ongoing research into personalized cancer medicine to ensure that it continues to advance and improve. Regulatory frameworks should both facilitate translational efforts and ensure that personalized cancer medicine is based on high-quality evidence and that its efficacy and safety are regularly monitored.
5.2. Health Economics Factors
The successful implementation of personalized cancer medicine is conditioned by several economic factors [
65]. Developing a new medicine usually takes at least a decade and costs an average of
$2.6 billion from drug discovery to FDA approval. Less than 12% of the candidate medicines make it to Phase one clinical trials. Personalized cancer medicine (PCM) often requires multiple funding agents, with traditional basic research funds not always geared towards application-centered research. Additionally, PCM frequently requires state or public support for infrastructures such as sequencing, clinical trial offices, molecular pathology, and IT. These barriers to PCM implementation have been documented in studies such as that of Chalmers et al (2014)[
66].
The costs associated with personalized cancer medicine highlight the need for a careful balance between ensuring access to effective treatments and controlling healthcare costs. This may require a combination of public and private investment, as well as a rethinking of traditional healthcare financing models. The relation between public funding and industry investment is thus an important factor to understand the introduction of personalized cancer medicine. Both public funding and industry investments have significant roles in the development of personalized cancer medicine. Public funding, often provided by government agencies such as the US National Cancer Institute, supports basic and clinical research into the underlying biology of cancer and the development of new drugs. This form of public funding provides the foundation for much of the innovation in the field and helps to advance the development of personalized cancer medicine. Industry investments, on the other hand, provide the financial resources needed to bring new personalized cancer treatments to market. Pharmaceutical and biotech companies are often the primary drivers of the development and commercialization of personalized cancer medicines, and their investments in research and development, clinical trials, and regulatory approval processes are essential for bringing these treatments to patients.
PCM faces several cost-related barriers, including expensive treatments and reimbursement policies that vary across countries and health insurance plans, potentially limiting patient access. The development of new PCM technologies requires significant investment, and companies may seek to recoup these costs through high prices for treatments. The adoption of PCM may also impact overall healthcare costs, as more individualized treatments may be more expensive than traditional treatments. The implementation of PCM requires training of healthcare professionals, leading to additional costs, while the regulatory approval process can be lengthy and expensive, potentially impacting the availability of new treatments. Cost effectiveness in personalised cancer medicine refers to the cost benefit balance of using personalised therapies compared to more traditional approaches [
67]. This includes considering the cost of the diagnostic tests, the cost of the personalised treatment itself, and any potential cost savings from improved patient outcomes and reduced side effects. In order to be considered cost-effective, the benefits of personalized cancer medicine, such as improved efficacy and reduced toxicity, must outweigh the additional costs incurred. Furthermore, cost-effectiveness analyses take into account the long-term benefits of personalized treatments for patients. This includes assessing the potential for reduced need for additional medicine and improved quality of life. The major objective with cost-effectiveness analyses is to balance the value of the treatment in terms of improved health outcomes for patients to its impact in terms of resource expenditure by the healthcare system and general society. Personalized medicine is estimated to have a significant potential to provide cost benefits in the treatment of cancer by enabling the development of targeted and more effective treatments [
68]. This can be done for example by increasing the evidence that certain treatments, such as expensive pharmaceuticals, have the desired effect and thus provide value to the patient. Personalized medicine has been shown to improve patient outcomes by providing more effective treatments and reducing the risk of side effects associated with chemotherapy and other traditional cancer treatments. This can lead to reduced costs associated with hospital stays and other medical procedures.
5.3. Organizational Factors
The translation of Personalized medicine into clinical practice is dependent on several critical organisational factors. One critical factor is the need for a
new form of scientific leadership.This includes researchers who are well-versed in the latest developments in the field and have experience in conducting clinical trials. Without this leadership, there may be a lack of direction and coordination, making it difficult to move research findings into clinical practice. Despite the challenges with assessing the effect of scientific leadership for scientific output in empirical terms recent studies provide clear indication of it as an important determinant factor [
69]. Another important factor is creating a clear
communicative environment between all stakeholders involved in the translation process. Effective communication between researchers, clinicians, patients, and regulators is critical to ensure that everyone is working towards a common goal and that any challenges or obstacles are quickly addressed. Establishing conditions for
team science is also critical. Personalized cancer medicine is a multidisciplinary field that requires collaboration between researchers with different areas of expertise, such as geneticists, oncologists, and pharmacologists [
70]. A collaborative environment can foster innovation, allow for the exchange of ideas and expertise, and facilitate the development of more effective treatment options.
Educationis another critical factor in the translation of personalized cancer medicine. Physicians, researchers, and other healthcare professionals need to be well-educated in the latest developments in the field to ensure that they can effectively diagnose and treat patients. Additionally, educating patients about personalized cancer medicine and its potential benefits can help to increase patient engagement and support for research efforts.
Efforts to boost translation from public and private interests are essential. Collaboration between biotech and pharmaceutical companies, academic institutions, and government regulators is essential to accelerate the translation of personalized cancer medicine into clinical practice. Additionally, targeted investment in research infrastructure and the development of new technologies can help to speed up the translation process. Regulation and government policy play a significant role in the translation of personalized cancer medicine. Regulations related to clinical trials, drug development, and approval processes can impact the translation of research findings into clinical practice. Government policies related to funding and investment in research infrastructure can also influence the translation process.
6. Implementing PCM
The introduction of PCM should ensure that patients receive the best possible care while also taking into account the needs and interests of different stakeholders, including healthcare providers, policymakers, and industry. This requires careful planning and coordination to ensure that the approach is effective, equitable, and sustainable.
6.1. PCM Requires Scientific Leadership
Several sources emphasize that personalized cancer medicine requires strong scientific leadership to guide research, development, and implementation. One of the primary roles of scientific leadership is to set the research agenda. Scientific leaders must identify the most pressing questions and challenges in the field and prioritize the allocation of resources towards finding solutions. This requires a deep understanding of the latest advances in the field and the ability to identify areas where further research is needed. Scientific leaders are also required for creating the ideal conditions to facilitate development. They must work closely with clinicians, researchers, and technology developers to translate scientific discoveries into practical applications. This requires strong communication and collaboration skills, as well as an understanding of the complex regulatory and ethical issues involved in personalized medicine. Scientific leadership is crucial in setting up routines and procedures that can guarantee standards, such as a high quality, accessibility and reliability of the data that forms the ground of PCM.
Numerous studies that have explored the impact of scientific leadership on the successful introduction of personalized cancer medicine. For example, Luchini et al.[
71]found that strong scientific leadership was critical for the development of new AI-based technologies in cancer diagnosis and treatment. Abernethy et al. (2010)[
72] posits that political and scientific leadership working closely with clinicians, researchers, and technology developers ensure the rapid translation of scientific findings through "Rapid-learning systems" providing actionable and practical applications that can improve treatment outcomes.
6.2. PCM Depends on Team Science
The systemic character of precision medicine requires collaboration across a multitude of different professions and organizations. The establishment of interdisciplinary collaborations between biomedical researchers, clinical expertise, informatics and computational expertise has brought about new titles and medical professions that are complementing or in some case replacing old categories. The difference in scope, funding, regulation and organisational culture between science and healthcare provokes a series of challenges. Conventional scientific literature lacks or provides minimal insights into the organisational measures needed to transform organizations towards a personalised medicine capacity. Translational cancer research is one of the fields that depend on team science, a discipline that aims to solve complex scientific challenges and build the adequate organisations or infrastructures [
73]. The focus of team science is to bring together experts from different fields to work together and leverage their collective expertise to tackle issues that cannot be addressed by individual researchers alone. Team science recognizes that complex problems require interdisciplinary approaches and that diverse perspectives are essential to the development of new knowledge and innovative solutions. Effective team science involves clear communication, shared decision-making, and a supportive collaborative work environment that fosters creativity, productivity, and innovation.
6.3. Work Conditions Are a Key Implementation Factor
Work conditions play a significant role in the successful translation. Optimal work conditions are determined by access to adequate funding, technology, and a supportive organizational structure and culture. By addressing these factors, organizations can improve the quality of care and advance the field of personalized medicine in cancer. Clinical researchers and nurses have a fundamental role in the implementation of personalized cancer medicine. Work conditions for clinical researchers and nurses, such as job satisfaction, workload, and resources, can affect performance and ultimately the capacity for translation [
74]. The quality of their work and their possibility to operate under good conditions directly affects patient outcomes [
75]. A common issue is high workloads and stress levels over long periods of time. This can lead to burnout among clinical researchers and nurses, which can negatively impact their ability to provide quality care to patients. Inadequate funding or inadequate staffing can impact the ability of clinical researchers and nurses to carry out research activities to prioritize essential care activities stalling an effective implementation of personalized cancer medicine. Access to state-of-the-art equipment and technology is also essential for ensuring that researchers and clinicians have the tools they need to effectively diagnose and treat cancer.
Another aspect of work conditions that can impact the successful translation is the organizational structure and culture of the workplace [
76]. For example, a hierarchical structure can create barriers to communication and collaboration, while a culture that values innovation and interdisciplinary collaboration can support the development and implementation of personalized medicine. The culture of the workplace defined by a multitude of factors can also impact the successful translation of personalized medicine in cancer [
77].
6.4. Enabling Collaboration between Industry and Academia
The successful translation of personalized medicine in cancer is highly dependent on good relationships between industry and academia in order to close the gap between basic scientific research and concretely actionable care in the clinic. While academic researchers exploring new scientific concepts and innovative technologies, the industry focuses on the commercialization of these technologies and the development of new products. These collaborations can also help to overcome barriers of commercialization, such as the high cost entailed with research including adhering to complex regulatory environment. Moreover, industry-academia collaborations are essential for the development of new technologies, such as AI-based technologies, diagnostic methods or new drugs. Industry academia collaborations can bring together the expertise and resources of both sectors, leading to the development of new predictive models and the testing, transfer and implementation of scientific discoveries into clinical practice.
The relationship between industry and academia have been explored in several scientific articles studying personalized medicine in cancer, but there is no consensus on the optimal models for implementation. In France and the UK, academic hospitals provide genomic testing free of charge, whereas in countries like the US, biomarker companies offer these as paid services. It is yet to be defined what the optimal model for implementation should be, and how it should align with the overall model of the healthcare system [
78].
7. Addressing the Translational Divide—Enabling Technology and Innovation Transfer from Biomedical Research to Diagnosis and Treatment
7.1. Addressing Legal and Ethical Issues
Several legal and ethical issues related to the implementation of PCM are discussed in the literature. Personalized cancer medicine follows the general trend within advanced modern medicine of increased reliance on the collections, and analysis of sensitive personal and medical data. This essential activity raises concerns about data privacy and security for patients. To achieve the best possible care in the PCM context legal issues need careful consideration in order to ensure that patients receive optimal care while protecting their rights and interests. The legal costs associated with these challenges can vary widely, and may include fees for legal advice, patent applications, regulatory submissions, and litigating any disputes that arise. Personalized cancer medicine often involves complex molecular and genetic testing, which can lead to disputes over who owns the data and any resulting treatments. The development of personalized cancer treatments entails significant investment in research and development, leading to questions about ownership of the rights to the resulting diagnostic tests, treatments, and technologies. Personalized cancer medicine generates large amounts of sensitive personal information, which must be protected under privacy laws. Companies that develop and market personalized cancer medicines may face liability for any adverse events or lack of efficacy associated with the treatment. Personalized cancer medicine may involve the use of experimental treatments, which raises questions about liability and informed consent, particularly if the treatment is not effective or causes harm. The development and approval of personalized cancer treatments is subject to rigorous regulatory standards, which can impact the speed and availability of these treatments [
79]. Finally, personalized cancer medicine can be more expensive than traditional treatments, which raises questions about reimbursement by insurance companies and government healthcare programs.
The implementation of PCM brings to the forefront several ethical issues that must be considered [
80]. One of the primary ethical concerns is access to care [
81]. Since personalised cancer medicine is usually expensive, not all patients may have equal access to these treatments, raising questions of fairness and equity. Data privacy is another ethical issue. The collection and computation of large amounts of complex and heterogeneous patient data raise concerns about privacy and confidentiality. The risk of discrimination is another ethical concern. Unequal treatment based on factors such as race, gender, or socioeconomic status could result in discrimination. Informed consent is an essential component of ethical medical practice, and personalized cancer medicine is no exception. An important ethical issue is clinical validity. Since personalized cancer medicine is still in the early stages of development, the validity and reliability of these treatments may be questionable, leading to ethical concerns about the use of unproven or ineffective treatments [
82].
Informed consent is an essential component of ethical medical practice, and personalized cancer medicine is no exception. Patients need to be fully informed about the benefits and risks of personalized cancer medicine and must give their informed consent before treatment begins. Moreover, conflicts of interest are a common ethical dilemma in personalized cancer medicine. The financial interests of pharmaceutical companies and healthcare providers may conflict with the best interests of patients, leading to ethical dilemmas. Addressing these ethical concerns is critical to ensuring that personalized cancer medicine is implemented in a responsible and ethical manner. To achieve this goal, policymakers, researchers, and healthcare providers must work together to develop ethical guidelines for personalized cancer medicine that consider the perspectives of patients, healthcare providers, and society as a whole.
In recent years the EU is following a development in the North American context regarding stricter requirements on quality and standards for medical devices and wet lab tests used for patient care. In 2022 the Invitro Diagnostic Regulation (IVDR) replaced an older directive which will greatly impact the processes of introducing advanced and large scale molecular diagnostics in the clinical setting.
7.2. Achieving Equity for Personalized Cancer Medicine
Achieving equity in personalized cancer medicine can be a challenge, as access to these treatments is often limited by factors such as cost, availability, and geographic location [
83]. Several strategies can be employed to help ensure that everyone has access to personalized cancer treatments, regardless of their ability to pay or where they live [
84]. Achieving equity in personalized cancer treatments requires that everyone has access to essential health services, including through universal health coverage programs. Universal health coverage ensures that everyone has access to healthcare services. This is particularly important for individuals who are unable to pay for the cost of these treatments or have other barriers to access [
85]. Patient assistance programs, either through government programs or charitable organisations, can provide financial assistance to these individuals.
Community-based initiatives can also play a critical role in ensuring equity in personalised cancer treatments [
86]. Working with local communities to raise awareness of personalised cancer medicine can help to remove barriers to access and improve health outcomes. Community-based initiatives can also provide access to these treatments to those who might not otherwise have access. This may involve increasing the availability of diagnostic tests in low- and middle-income countries, where access to these tests may be unavailable.
To ensure that everyone has equal and fair access to personalised cancer treatments, productive collaborative conditions must exist between healthcare providers and patients. This involves providing patients with the relevant information and resources needed for qualified medical decisions. Access to information and resources can be improved through the development of productive collaborative conditions, which enable patients to work with healthcare providers to identify the most appropriate treatments for their individual needs. By addressing these key factors, we can work towards achieving equity in personalized cancer treatments.
In summary the available literature suggests that the following actions are required for successful introduction of personalized cancer medicine into clinical practice:
Engage with key stakeholders, including patients, healthcare providers, policymakers, and industry, to ensure that the introduction of personalized cancer medicine is informed by the needs and perspectives of all those who will be affected.
Conduct rigorous clinical validation studies to conclusively demonstrate how personalized cancer treatments are efficient and safe in conjunction with their impact on patient outcomes.
Work with regulators and payers to ensure that personalized cancer treatments are approved for use and covered by insurance (public or private), so that patients have access to these treatments when they need them.
Provide education and training for healthcare providers and patients to ensure that everyone has the information and resources they require to make well-informed decisions about personalized cancer medicine.
Develop and implement effective data management policies and procedures to ensure that patient data is handled appropriately and securely.
Address the issue of equity and access to ensure that everyone has access to personalized cancer treatments regardless of patients ability to pay, social status or residence.
Continuously evaluate and improve the introduction of personalized cancer medicine to ensure that patients receive the best possible care and that the approach remains effective over time.
7.3. Education and Dissemination
A often mentioned key factor for succeeding in implementing the paradigmatic shift into personalized medicine is the preparation of new generations of medical professionals. Part of the resistance to new methods can be addressed with a change management approach [
87], however a complete transformation might only be possible once the new generations take on leadership positions. More importantly given the rapid developments in the field there will be a need for efficient dissemination and further education of persons with key roles in the intertwined chain of translation.
7.4. Patient Involvement
Active patient involvement, participation, and agency are vital components of the concept of personalization, extending to related concepts such as P4 medicine, which emphasizes "participation." Among these, perhaps the most pivotal aspect is the patient’s consent to participate in clinical trials or, more fundamentally, to accept recommended medical treatment. However, in recent years, the involvement of patients in various stages of treatment has emerged as a field of extensive research and development. Patients should not be perceived merely as passive recipients of medicine or dehumanized sources of data for research purposes. While the primary goal of medicine is to enhance patients’ lives and health, this fundamental aim can sometimes get overshadowed by the intricate network of efforts spanning from research to healthcare delivery. Integrating patients’ expressed ideas, intuitions, and self-reported symptoms can provide a unique source of information to complement the multitude of biological markers informing diagnosis and treatment decisions. Furthermore, granting agency to patients leads to a genuinely personalized healthcare experience. Transforming the traditionally passive and subordinate role of patients into a more active and rewarding experience preserves patients’ integrity and dignity. Although patients should not feel obligated or pressured to delve into the complexities of their medical conditions and treatments, fostering an environment conducive to patient involvement is crucial for enhancing their relationship with healthcare. Ongoing studies demonstrate the value of structured patient-reported symptoms, offering critical insights to guide physicians during pivotal decision-making moments. Additionally, patient-reported symptoms and outcomes serve as valuable predictive markers, enhancing the precision of dosing and treatment protocols.
8. New Forms of Organization That Bridge the Divide
The impleamentation of PCM has fostered the building of new organizations capable of handling the complexity of a rapidly developing discipline.
8.1. Molecular Tumour Boards
The Molecular Tumor Boards (MTB) is an organizational innovation that has emerged form the introduction of personalized medicine approaches to cancer care [
88,
89,
90]. With the introduction of molecular markers, the increased complexity of tumour classification and treatment selection has urged oncologists to form specific meetings where actionable targets are discussed based on results from genomic analysis and other available evidence [
91]. MTBs are multidisciplinary teams of experts from different fields, including oncology, genetics, pathology, and bioinformatics, that are brought together regularly to analyse large amounts of genomic data and translate this information into treatment recommendations based on the molecular characteristics of patient’s tumours and decisions on potential inclusion to clinical trials [
92]. The introduction of these meetings has affected the translational and implementation processes for personalized cancer medicine [
93]. MTBs are expected to become perfect translational means by which to extend the molecular diagnostic tools including multi-omics with transcriptomics and proteomics [
94].
Several studies have demonstrated the impact of MTB on the implementation processes for personalised cancer medicine [
95]. MTBs were shown to improve the accuracy of genomic data interpretation and increased the proportion of patients who received targeted therapy [
96,
97,
98]. This also includes improvement of the capacity to communicate with patients by facilitating the presentation of genomic information [
99]. This has improved the adoption of precision medicine in cancer care and helped to accelerate the translation of genomic discoveries into clinical practice. MTBs also provide a framework for educating clinicians and guiding them through incorporating genomic data into clinical decision-making [
100]. The organizational phenomena of MTBs is becoming more institutionalized with the introduction of transnational cooperation’s and studies investigating the procedures and results of these meetings. Furthermore, tools such as decision support systems designed to improve the quality of decisions during MTBs by generating digestible data overviews based on large quantities of accumulated evidence have been proven effective [
101].
8.2. Comprehensive Cancer Centers
Comprehensive Cancer Centres (CCC) have been identified as main catalyzers to bridge the translational divide. CCCs are specialized institutions at the level of medical faculties and university hospitals or cancer centres that aim to provide a seamless integration of cancer research and patient care [
102,
103].
They are designed to bridge the divide between research and clinic by creating an environment where scientists and clinicians can work together to develop and implement new cancer treatments. In this environment a unique approach to cancer care is provided by bringing together medical professions, including oncologists, surgeons, radiologists, pathologists, and nurses, with biomedical researchers, to provide a comprehensive and personalized treatment plan for each patient following team science principles.
A key feature of CCCs is the emphasis on multidisciplinarity. By bringing together experts from different fields, CCCs can provide a more holistic approach to cancer care, taking into account not only the medical aspects but also the social, psychological, and emotional needs of patients. Tumor management groups are a common example of multidisciplinary teams, where different specialists collaborate to determine the most effective treatment plan for each patient.
The success of CCC’s is critically dependent on their organization. Program directors are typically scientists who are responsible for fostering interactions between different institutes and programs within the CCC. These interactions can be informal, such as regular meetings or collaborations, or more formal, such as sponsored symposia or workshops. By creating a collaborative environment, CCCs encourage the sharing of knowledge and expertise, leading to more efficient and effective cancer treatments. The functionality of relevant branches is essential to CCC’s operation. CCC’s must have the necessary infrastructure, equipment, and personnel to carry out cancer research and clinical trials. These resources are typically shared among different programs and divisions within the CCC, ensuring that all researchers and clinicians have access to the tools they need to advance their work.
The establishment of comprehensive care networks are another crucial aspect of CCC’s operation. These networks connect the CCC with other healthcare providers in the region, such as community hospitals and primary care physicians. By collaborating with these providers, CCCs can ensure seamless transitions between different levels of care, giving to the patients the best possible treatment at every stage of their cancer journey [
104].
Governance is another critical component of CCCs. These institutions are typically governed by a board of directors, which oversees the operations of the CCC and ensures that it is meeting its goals and objectives. The board is responsible for ensuring that the CCC has the necessary resources to carry out its mission and that it is adhering to ethical and legal standards.
8.3. Large Scale Infrastructure and Screening Consortia
In the realm of precision medicine in cancer, large-scale infrastructures play a vital role from an organizational perspective. These infrastructures encompass various interconnected elements such as data integration platforms, high-performance computing systems, advanced analytics tools, and collaborative networks. By establishing such infrastructures, organizations can effectively harness the vast amounts of genomic, clinical, and imaging data that are generated in cancer research and treatment. These integrated systems facilitate seamless data sharing, storage, and analysis, thereby empowering researchers and clinicians to gain deeper insights into the molecular underpinnings of cancer and develop personalized treatment approaches. A notable example is the work by Alarcon Garavito et al. (2023) [
105], which showcases the successful implementation of a large-scale infrastructure that integrates genomic data, electronic health records, and computational resources to bolster precision medicine endeavors in the field of oncology. This infrastructure not only expedites the identification of actionable genomic alterations but also provides real-time support for clinical decision-making, ultimately leading to improved patient outcomes.
In the European context several large scale national infrastructures are worth mentioning such as SciLifeLab and Genomics Medicine Sweden[
106] and Genomics Medicine England[
107].
Table 3.
National Cancer initiatives promoting precision cancer medicine.
Table 3.
National Cancer initiatives promoting precision cancer medicine.
| Consortia
|
Start Year |
Annual Budget |
| Genomics Medicine Sweden |
2019 |
81 million Euro |
| Cancer Research UK: International Cancer |
2008 |
210 million Euro |
| National Cancer Institute: Cancer Research |
2005 |
4.6 billion Euro |
| Genomics Medicine England |
2013 |
123 million Euro |
9. Conclusions
We here point to the need for a comprehensive and systematic qualitative study on the determining factors for the successful translation of personalized cancer medicine. Despite the methodological challenges associated with an exhaustive analysis (addressing social complexity, contextual difference and biases) of the conditions required for the successful implementation of translational approaches, a number of factors can still be identified as significant. PCM faces several challenges that need to be addressed, including funding, infrastructure support, and development of new regulatory approval processes. Overcoming these challenges will require multi-stakeholder collaboration, including governments, healthcare systems, research institutions, and the pharmaceutical industry, to ensure that patients can access the benefits of personalized cancer medicine. In addition, the translation of personalized cancer medicine into clinical practice requires the convergence of various environmental factors including strong scientific leadership, a clear communicative environment and the establishment of collaborative team science. Matching science and education with patients needs, and regulatory policies plays critical roles in the translation process. Effective data management is essential to ensure the success of personalized cancer medicine, and careful consideration and planning are required to achieve this aspect. In-house infrastructure needs are essential, as they can help to ensure that patient data is managed appropriately and securely. The capacity to build relationships with industry is important, as it can help to foster collaborations and facilitate the development and implementation of new technologies.
Beyond the fragmentation – fostering bridging connections – A recurring observations from multiple stakeholders is the fragmented nature of the relationship between research, healthcare, industry, government and patients [
108]. There are multiple obstructions, or lack of capacities, resulting in divides in the continuum from research to healthcare. Interviews with stakeholders have confirmed this fragmentation resulting in different obstructions in the translational efforts. Possible measures to bridge the divides include:
Pooling resources across stakeholders
Investing in time for clinical investigators to provide the best working conditions so they can bring the translational process forward
Provide the right conditions for a multitude of clinical trials of different designs, sponsorships and purposes.
Pooling common resources across stakeholders is essential for advancing translational research in medicine. Bringing together researchers, healthcare providers, and other stakeholders, can leverage the collective expertise and resources to accelerate the development of new treatments and therapies. However, it is not just about having access to resources, but also investing in time to ensure that clinical investigators have the best working conditions to bring the translational process forward. This includes providing the necessary infrastructure, support staff, and funding to carry out clinical trials of different designs, sponsorships, and purposes. By creating the conditions for a multitude of clinical trials, one can improve the efficiency and effectiveness of the translational process, leading to better outcomes for patients.
Author Contributions
Conceptualization, original draft preparation Michele Masucci; methodology, Michele Masucci and Ingemar Ernberg; writing, review and editing, Michele Masucci, Ingemar Ernberg; review, Claes Karlsson and Lennart Blomqvist; project administration, Ingemar Ernberg; funding acquisition, Ingemar Ernberg. All authors have read and agreed to the published version of the manuscript.
Funding
This research funded by the Cancer Research Funds of Radiumhemmet.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
Copies of searches are available upon request.
Acknowledgments
This research was made possible thanks to the generous support from the Cancer Research Funds of Radiumhemmet.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Crick, F.H.C.; Watson, J.D. The complementary structure of deoxyribonucleic acid. Proceedings of the Royal Society of London 1954, 223, 1152. [Google Scholar]
- Visvikis-Siest, S.; Theodoridou, D.; Kontoe, M. S.; Kumar, S.; Marschler, M. Milestones in Personalized Medicine: From the Ancient Time to Nowadays-the Provocation of COVID-19. Front Genet. 2020, 11, 569175. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, V.; Tarazona, N.; Cejalvo, J. M.; Lombardi, P.; Huerta, M.; Rosello, S.; Fleitas, T.; Roda, D.; Cervantes, A. Personalized Medicine: Recent Progress in Cancer Therapy Cancers (Basel). 2020, 12, 4. [Google Scholar]
- Fournier, V.; Prebet, T.; Dormal, A.; Brunel, M.; Cremer, R.; Schiaratura, L. Definition of Personalized Medicine and Targeted Therapies: Does Medical Familiarity Matter? J Pers Med. 2021, 11, 26. [Google Scholar] [CrossRef] [PubMed]
- Schleidgen, S.; Klingler, C.; Bertram, T.; Rogowski, W. H.; Marckmann, G. What is personalized medicine: sharpening a vague term based on a systematic literature review. BMC medical ethics. 2013, 14, 55. [Google Scholar] [CrossRef] [PubMed]
- Hood, L. Systems biology and p4 medicine: past, present, and future. Rambam Maimonides Med J. 2013, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Nimmesgern, E.; Benediktsson, I.; Norstedt, I. A. Precision Medicine: From Science To Value. Clin Transl Sc. 2017, 10, 61–63. [Google Scholar] [CrossRef]
- Ginsburg, G. S.; Phillips, K. A. Precision Medicine: From Science To Value. Health Aff (Mill-wood). 2018, 37, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, A.; Fuchs, E.; Mitchison, T.; Shaw, R. J.; St Johnston, D.; Strasser, A. et al. A decade of molecular cell biology: achievements and challenges. Nat Rev Mol Cell Biol. 2011, 12, 669–74. [Google Scholar] [CrossRef] [PubMed]
- Malani, D.; Kumar, A.; Bruck, O.; Kontro, M.; Yadav, B.; Hellesoy, M. Implementing a Functional Precision Medicine Tumor Board for Acute Myeloid Leukemia. Cancer Discov. 2022, 12, 388–401. [Google Scholar] [CrossRef] [PubMed]
- Bohr, A.; Memarzadeh, K. The rise of artificial intelligence in healthcare applications. HArtificial Intelligence in Healthcare. 2020, 37, 694–701. [Google Scholar] [CrossRef]
- Rapport, F.; Clay-Williams, R.; Churruca, K.; Shih, P.; Hogden, A.; Braithwaite, J. The struggle of trans-lating science into action: Foundational concepts of implementation science. Cell 2000, 24, 117–126. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R. A. The hallmarks of cancer. Cell 2000, 100, 57. [Google Scholar] [CrossRef]
- Paez, J. G.; Jänne, P. A.; Lee, J. C.; Tracy, S.; Greulich, H.; Gabriel, S. EGFR Mutations in Lung Cancer: Correlation with Clinical Response to Gefitinib Therapy. Science (American Association for the Advancement of Science). 2004, 304, 1497–1500. [Google Scholar] [CrossRef] [PubMed]
- Slamon, D. J. Use of the anti HER-2/neu antibody Herceptin in the treatment of human breast cancer: bi-ological rationale and clinical results. Breast cancer research : BCR. 2000, 2. [Google Scholar] [CrossRef]
- Ginsburg, G. S.; Phillips, K. A. Precision Medicine: From Science To Value. Health Aff (Mill-wood). 2018, 37, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M. H.; Johnson, J. R.; Pazdur, R. U.S. Food and Drug Administration Drug Approval Summary: Conversion of Imatinib Mesylate (STI571; Gleevec) Tablets from Accelerated Approval to Full Approv-al. Clinical cancer research. 2005, 11, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Slamon, D. J. Use of the anti HER-2/neu antibody Herceptin in the treatment of human breast cancer: bi-ological rationale and clinical results. Breast cancer research : BCR. 2000, 2. [Google Scholar] [CrossRef]
- Lynch, T. J.; Bell, D. W.; Sordella, R.; Gurubhagavatula, S.; Okimoto, R. A.; Brannigan, B. W. , et al. Ac-tivating Mutations in the Epidermal Growth Factor Receptor Underlying Responsiveness of Non–Small-Cell Lung Cancer to Gefitinib. The New England journal of medicine. 2004, 350, 2129–2139. [Google Scholar] [CrossRef] [PubMed]
- Adjei, A. A.; Cohen, R. B.; Franklin, W.; Morris, C.; Wilson, D.; Molina, J. R.; et al. Phase I pharmaco-kinetic and pharmacodynamic study of the oral, small-molecule mitogen-activated protein kinase kinase 1/2 inhibitor AZD6244 (ARRY-142886) in patients with advanced cancers. Health Aff (Millwood). 2008, 26, 2139–46. [Google Scholar] [CrossRef]
- Fischer, H.; Taylor, N.; Allerstorfer, S.; Grusch, M.; Sonvilla, G.; Holzmann, K. , et al. Fibroblast growth factor receptor-mediated signals contribute to the malignant phenotype of non-small cell lung cancer cells: therapeutic implications and synergism with epidermal growth factor receptor inhibition. Mol Cancer Ther. 2008, 37, 694–701. [Google Scholar] [CrossRef]
- Fong, P. C.; Yap, T. A.; Boss, D. S.; Carden, C. P.; Mergui-Roelvink, M.; Gourley, C. , et al. Poly(ADP)-ribose polymerase inhibition: frequent durable responses in BRCA carrier ovarian cancer cor-relating with platinum-free interval. J Clin Oncol. 2010, 28, 2512–9. [Google Scholar] [CrossRef] [PubMed]
- Flaherty, K. T.; Puzanov, I.; Kim, K. B.; Ribas, A.; McArthur, G. A. J.; Sosman, A. , et al. Inhibition of Mutated, Activated BRAF in Metastatic Melanoma. N Engl J Med. 2010, 363, 809–819. [Google Scholar] [CrossRef] [PubMed]
- Bang, Y.; Kwak, E. L.; Shaw, A. T.; Camidge, D. R.; Iafrate, A. J.R.; Maki, G. , et al. Clinical activity of the oral ALK inhibitor PF-02341066 in ALK-positive patients with non-small cell lung cancer (NSCLC). Journal of clinical oncology. 2010, 28. [Google Scholar] [CrossRef]
- Delmore, J. E.; Issa, G. C.; Lemieux, M. E. ; Rahl,P. B.; Shi, J.; Jacobs, H. M. et al. PBET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell. 2011, 146, 904–17. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A. T.; Ou, S. H.; Bang, Y. J.; Camidge, D. R.; Solomon, B. J.; Salgia, R. , et al. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N Engl J Med. 2014, 371, 1963–71. [Google Scholar] [CrossRef] [PubMed]
- Finn, R. S.; Martin, M.; Rugo, H. S.; Jones, S.; Im, S. A.; Gelmon, K. , et al. Palbociclib and Letrozole in Advanced Breast Cancer. Health Aff (Millwood). 2018, 37, 694–701. [Google Scholar] [CrossRef]
- Drilon, A.; Siena, S.; Dziadziuszko, R.; Barlesi, F.; Krebs, M. G. A.; Shaw, T. , et al. N Engl J Med. 2020, 21, 261–270. [Google Scholar] [CrossRef]
- Drilon, A.; Siena, S.; Dziadziuszko, R.; Barlesi, F.; Krebs, M. G. A.; Shaw, T. , et al. N Engl J Med. 2020, 21, 261–270. [Google Scholar] [CrossRef]
- Hong, D. S.; Fakih, M. G.; Strickler, J. H.; Desai, J.; Durm, G. A. G.; Shapiro, I. , et al. KRAS(G12C) In-hibition with Sotorasib in Advanced Solid Tumors. N Engl J Med. 2020, 383, 1207–1217. [Google Scholar] [CrossRef] [PubMed]
- Crosby, D.; Bhatia, S.; Brindle, K. M.; Coussens, L. M.; Dive, C.; Emberton, M. , et al. Early detection of cancer. Science. 20, 375. [Google Scholar] [CrossRef]
- De Sousa, E. M. F.; Vermeulen, L.; Fessler, E. ; Medema., J. P. Cancer heterogeneity: a multifaceted view. EMBO Rep. 2013, 14, 686–95. [Google Scholar] [CrossRef]
- Devilakshmi, S.; Madhumathi, J.; Verma, R. S. Immunotoxins, Resistance and Cancer Stem Cells: Future Perspective. Springer International Publishing. 2015. [Google Scholar] [CrossRef]
- Vinay, D. S.; Ryan, E. P.; Pawelec, G.; Talib, W. H.; Stagg, J.; Elkord, E. , et al. Immune evasion in cancer: Mechanistic basis and therapeutic strategies. Semin Cancer Biol. 2015, 35, S185–S198. [Google Scholar] [CrossRef] [PubMed]
- Esfahani, K.; Roudaia, L.; Buhlaiga, N.; Del Rincon, S. V.; Papneja, N.; Miller Jr., W. H. A review of cancer immunotherapy: from the past, to the present, to the future. Curr Oncol. 2020, 27, S87–S97. [Google Scholar] [CrossRef] [PubMed]
- Yazbeck, V.; Alesi, E.; Myers, J.; Hackney, M. H.; Cuttino, L.; Gewirtz, D. A. An overview of chemotox-icity and radiation toxicity in cancer therapy. Adv Cancer Res. 2022, 155, 1–27. [Google Scholar] [CrossRef]
- Tomczak, K.; Czerwinska, P.; Wiznerowicz, M. The Cancer Genome Atlas (TCGA): an immeasurable source of knowledge. Contemp Oncol (Pozn). 2015, 19, A68–77. [Google Scholar] [CrossRef] [PubMed]
- Denninghoff, V. C. Molecular Pathology in the New Age of Personalized Medicine. Pathology-From Classics to Innovations. IntechOpen. 2020. [Google Scholar] [CrossRef]
- Skipper, M. Cancer genomics: A panoramic view of cancer. Nat Rev Genet. 2013, 14, 750. [Google Scholar] [CrossRef]
- Kircher, M. F.; Hricak, H. ; Larson., S. M. Molecular imaging for personalized cancer care. Mol Oncol. 2012, 182–95. [Google Scholar] [CrossRef]
- Shaw, A.; Seban, R. D.; Besson, F. L.; Vila-Reyes, H.; Ammari, S.; Mokrane, F. Z. , et al. Breakthrough in Imaging-Guided Precision Medicine in Oncology. Front Oncol. 2022, 22. [Google Scholar] [CrossRef]
- Polici, M.; Laghi, A.; Caruso, D. Editorial for Special Issue on Imaging Biomarker in Oncology. Cancers (Basel). 15. [CrossRef] [PubMed]
- Rundo, L.; Rundo, L.; Militello, C.; Conti, V.; Zaccagna, F.; Han, C. Advanced Computational Methods for Oncological Image Analysis. Springer International Publishing. 2021. [Google Scholar] [CrossRef] [PubMed]
- Clevers, H. Modeling Development and Disease with Organoids. Springer International Publish-ing. 2016, 65, 1586–1597. [Google Scholar] [CrossRef] [PubMed]
- Murumagi, A.; Ungureanu, D.; Khan, S.; Arjama, M.; Valimaki, K.; Ianevski, A. , et al. Drug response profiles in patient-derived cancer cells across histological subtypes of ovarian cancer: real-time therapy tailoring for a patient with low-grade serous carcinoma. Br J Cancer. 2023, 128, 678–690. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, R. E. Biobanking: the foundation of personalized medicine. Current opinion in oncolo-gy. 2011, 23, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Annaratone, L.; De Palma, G.; Bonizzi, G.; Sapino, A.; Botti, G.; Berrino, E. , et al. Basic principles of biobanking: from biological samples to precision medicine for patients. Virchows Arch. 2021, 479, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Mann, R.; Behniwal, N. S.; Baadh, I.; Gulati, A. Su1322 Comparing diagnostic yield of EUS guided Fine Needle Biopsy (FNB) in the diagnostis of solid lesions: Retrospective Review Study in a Community Hos-pital. 2020, 91, AB331–AB331. [Google Scholar] [CrossRef]
- Luchini, C.; Pea, A.; Scarpa, A. Artificial intelligence in oncology: current applications and future per-spectives. Br J Cancer. 2022, 126, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Bhinder, B.; Gilvary, C.; Madhukar, N. S.; Elemento, O. Artificial Intelligence in Cancer Research and Precision Medicine. Cancer Discov. 2021, 11, 900–915. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, H.; Nakayama, K. I. Artificial intelligence in oncology. Cancer Sci. 2020, 111, 1452–1460. [Google Scholar] [CrossRef] [PubMed]
- Paez, J. G.; Jänne, P. A.; Lee, J. C.; Tracy, S.; Greulich, H.; Gabriel, S. , et al. EGFR Mutations in Lung Cancer: Correlation with Clinical Response to Gefitinib Therapy. Science (American Association for the Advancement of Science). 2004, 304, 1497–1500. [Google Scholar] [CrossRef] [PubMed]
- MacEachern, S. J.; Forkert, N. D. Machine learning for precision medicine. Genome. 2021, 64, 416–425. [Google Scholar] [CrossRef] [PubMed]
- Kurzrock, R.; Giles, F. J. Precision oncology for patients with advanced cancer: the challenges of malignant snowflakes. Cell Cycle. 2015, 14, 2219–21. [Google Scholar] [CrossRef] [PubMed]
- Malani, D.; Kumar, A.; Bruck, O.; Kontro, M.; Yadav, B.; Hellesoy, M. , et al. Implementing a Functional Precision Medicine Tumor Board for Acute Myeloid Leukemia. Cancer Discov. 2022, 12, 388–401. [Google Scholar] [CrossRef] [PubMed]
- Meric-Bernstam, F.; Ford, J. M.; O’Dwyer, P. J.; Shapiro, G. I.; McShane, L. M.; Freidlin, B. , et al. National Cancer Institute Combination Therapy Platform Trial with Molecular Analysis for Therapy Choice (Com-boMATCH). Clin Cancer Res. 2023, 29, 1412–1422. [Google Scholar] [CrossRef] [PubMed]
- Geurts, B.; Zeverijn, L. J.; Battaglia, T. W.; van de Haar, J.; van Berge Henegouwen, J. M.; Hoes, L. R. , et al. Efficacy and predictors of response of nivolumab in treatment-refractory MSI solid tumors: Results of a tumor-agnostic DRUP cohort. Journal of clinical oncology. 2023, 41, 2590–2590. [Google Scholar] [CrossRef]
- Cecchini, M.; Rubin, E. H.; Blumenthal, G. M.; Ayalew, K.; Burris, H. A.; Russell-Einhorn, M. , et al. Challenges with Novel Clinical Trial Designs: Master Protocols. Clin Cancer Res. 2019, 25, 2049–2057. [Google Scholar] [CrossRef] [PubMed]
- King, D.; Foucar, C. E.; Ma, V.; Benitez, L.; Perissinotti, A. J.; Marini, B. L. , et al. Identification of variant APL translocations PRKAR1A-RARalpha and ZBTB16-RARalpha (PLZF-RARalpha) through the MI-ONCOSEQ platform. Cancer Genet. 2021, 258-259, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Rodon, J.; Soria, J. C.; Berger, R.; Miller, W. H.; Rubin, E.; Kugel, A. , et al. Genomic and transcriptomic profiling expands precision cancer medicine: the WINTHER trial. Nat Med. 2019, 25, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Massard, C.; Michiels, S.; Ferte, C.; Le Deley, M. C.; Lacroix, L.; Hollebecque, A. , et al. High-Throughput Genomics and Clinical Outcome in Hard-to-Treat Advanced Cancers: Results of the MOSCATO 01 Tri-al. Cancer Discov. 2017, 7, 586–595. [Google Scholar] [CrossRef]
- Le Tourneau, C.; Delord, J. P.; Goncalves, A.; Gavoille, C.; Dubot, C.; Isambert, N. , et al. Molecularly targeted therapy based on tumour molecular profiling versus conventional therapy for advanced cancer (SHIVA): a multicentre, open-label, proof-of-concept, randomised, controlled phase 2 trial. Journal of clinical oncology. 2023, 41, 2590–2590. [Google Scholar] [CrossRef]
- Stefanicka-Wojtas, D.; Kurpas, D. Barriers and Facilitators to the Implementation of Personalised Medi-cine across Europe. J Pers Med. 2023, 13. [Google Scholar] [CrossRef] [PubMed]
- Tarkkala, H.; Helén, I.; Snell, K. From health to wealth: The future of personalized medicine in the mak-ing. Journal of clinical oncology. 2019, 109, 142–152. [Google Scholar] [CrossRef]
- Sarvas, H.; Carlisle, B.; Dolter, S.; Vinarov, E.; Kimmelman, J. Impact of Precision Medicine on Effi-ciencies of Novel Drug Development in Cancer. J Natl Cancer Inst. 2020, 112, 859–862. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, I.; Bracken, M. B.; Djulbegovic, B.; Garattini, S.; Grant, J.; Gulmezoglu, A. M. , et al. How to increase value and reduce waste when research priorities are set. Lancet. 2014, 383, 156–65. [Google Scholar] [CrossRef]
- Kasztura, M.; Richard, A.; Bempong, N. E.; Loncar, D.; Flahault, A. Cost-effectiveness of precision medicine: a scoping review. Int J Public Health. 2019, 64, 1261–1271. [Google Scholar] [CrossRef]
- Vellekoop, H.; Versteegh, M.; Huygens, S.; Corro Ramos, I.; Szilberhorn, L.; Zelei, T. , et al.The Net Benefit of Personalized Medicine: A Systematic Literature Review and Regression Analysis. Value Health. 2022, 25, 1428–1438. [Google Scholar] [CrossRef] [PubMed]
- Simoes, N.; Crespo, N. On the measurement of scientific leadership. Journal of information sci-ence. 2022, 48, 131–138. [Google Scholar] [CrossRef]
- Salgia, R.; Salgia, R.; Kulkarni, P. Integrating Clinical and Translational Research Networks—Building Team Medicine. MDPI. 2023. [Google Scholar] [CrossRef]
- Luchini, C.; Pea, A.; Scarpa, A. Artificial intelligence in oncology: current applications and future per-spectives. Br J Cancer. 2022, 126, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Abernethy, A. P.; Etheredge, L. M.; Ganz, P. A.; Wallace, P.; German, R. R.; Neti, C. , et al. Rapid-learning system for cancer care. 2010, 28:27 Pages 4268-74. 2010, 28, 4268–74. [Google Scholar] [CrossRef] [PubMed]
- Hall, K. L.; Feng, A. X.; Moser, R. P.; Stokols, D.; Taylor, B. K. Moving the science of team science for-ward: collaboration and creativity. Am J Prev Med. 2008, 35, S243–9. [Google Scholar] [CrossRef]
- Chen, Y. C.; Guo, Y. L.; Chin, W. S.; Cheng, N. Y.; Ho, J. J.; Shiao, J. S. Patient-Nurse Ratio is Related to Nurses’ Intention to Leave Their Job through Mediating Factors of Burnout and Job Dissatisfaction. Int J Environ Res Public Health. 2019, 16, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Parlangeli, O.; Guidi, S.; Marchigiani, E.; Bracci, M. ; Liston; P. M. Perceptions of Work-Related Stress and Ethical Misconduct Amongst Non-tenured Researchers in Italy. Sci Eng Ethics. 2020, 21, 159–181. [Google Scholar] [CrossRef] [PubMed]
- Weitzel, J. N.; Blazer, K. R.; MacDonald, D. J.; Culver, J. O.; Offit, K. Genetics, genomics, and cancer risk assessment: State of the Art and Future Directions in the Era of Personalized Medicine. CA Cancer J Clin. 2011, 61, 327–59. [Google Scholar] [CrossRef]
- Ginsburg, G. S.; Phillips, K. A. Precision Medicine: From Science To Value. Health Aff (Mill-wood). 2018, 37, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Arnedos, M.; Soria, J. C.; Andre, F.; Tursz, T. Personalized treatments of cancer patients: a reality in daily practice, a costly dream or a shared vision of the future from the oncology community? Cancer Treat Rev. 2014, 40, 1192–8. [Google Scholar] [CrossRef] [PubMed]
- Lander, B.; Atkinson-Grosjean, J. Translational science and the hidden research system in universities and academic hospitals: a case study. Soc Sci Med. 2011, 72, 537–44. [Google Scholar] [CrossRef]
- Nelson, B.; Wiles, A. A sharper focus on the bioethics of precision medicine: In working toward more personalized health solutions, researchers are grappling with the enduring biases and inequities that have excluded entire groups: In working toward more personalized health solutions, researchers are grappling with the enduring biases and inequities that have excluded entire groups. Cancer Cytopathol 2022, 130, 398–399. [Google Scholar] [CrossRef] [PubMed]
- Horne, L. L.; Oprea-Ilies, G. M.; Stanley, E. R.; Holloway, C. M.; Hooker, M. P.; Isom, A. , et al. Democ-ratizing precision cancer medicine and advancing health equity in the black belt. Cancer research (Chicago, Ill.). 2022, 82, LB173–LB173. [Google Scholar] [CrossRef]
- Winkler, E. C.; Knoppers, B. M. Ethical challenges of precision cancer medicine. Semin Cancer Biol. 2022, 84, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Williams, J. S.; Walker, R. J.; Egede, L. E. Achieving Equity in an Evolving Healthcare System: Oppor-tunities and Challenges. Am J Med Sci. 2016, 351, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Giri, V. N.; Shimada, A.; Leader, A. E. Predictors of Population Awareness of Cancer Genetic Tests: Im-plications for Enhancing Equity in Engaging in Cancer Prevention and Precision Medicine. JCO Precis Oncol. 2021, 5, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Leahy, D.; Donnelly, A.; Irwin, K.; D’Alton, P. Barriers and facilitators to accessing cancer care for people with significant mental health difficulties: A qualitative review and narrative synthesis. Psychoon-cology. 2021, 30, 2012–2022. [Google Scholar] [CrossRef] [PubMed]
- Neil-Sztramko, S. E.; Smith-Turchyn, J.; Fong, A.; Kauffeldt, K.; Tomasone, J. R. Community-Based Exercise Programs for Cancer Survivors: A Scoping Review of Program Characteristics Using the Consol-idated Framework for Implementation Research. Arch Phys Med Rehabil. 2022, 103, 542–558. [Google Scholar] [CrossRef] [PubMed]
- Hagl, C.; Kanitz, R.; Gonzalez, K.; Hoegl, M. Change management interventions: Taking stock and moving forward. Human Resource Management Review. 2024, 34. [Google Scholar] [CrossRef]
- Behel, V.; Noronha, V.; Choughule, A.; Shetty, O.; Chandrani, P.; Kapoor, A. , et al. Impact of Molecular Tumor Board on the Clinical Management of Patients With Cancer. JCO Glob Oncol. 2022, 8. [Google Scholar] [CrossRef]
- Peh, K. H.; Przybylski, D. J.; Fallon, M. J.; Bergsbaken, J. J.; Hutson, P. R.; Yu, M. , et al. Clinical utility of a regional precision medicine molecular tumor board and challenges to implementation. J Oncol Pharm Pract. 2022. [Google Scholar] [CrossRef]
- Kato, S.; Kim, K. H.; Lim, H. J.; Boichard, A.; Nikanjam, M.; Weihe, E. , et al. Real-world data from a molecular tumor board demonstrates improved outcomes with a precision N-of-One strategy. Nat Commun. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Crimini, E.; Repetto, M.; Tarantino, P.; Ascione, L.; Antonarelli, G.; Rocco, E. G. , et al. Challenges and Obstacles in Applying Therapeutical Indications Formulated in Molecular Tumor Boards. Cancers (Basel). 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Larson, K. L.; Huang, B.; Weiss, H. L.; Hull, P.; Westgate, P. M.; Miller, R. W. , et al. Clinical Outcomes of Molecular Tumor Boards: A Systematic Review. JCO Precis Oncol. 2021, 5. [Google Scholar] [CrossRef]
- Devaraj, S.; Sharma, S. K.; Fausto, D. J.; Viernes, S.; Kharrazi, H. Barriers and Facilitators to Clinical Decision Support Systems Adoption: A Systematic Review. Journal of Business Administration Research. 2014, 3. [Google Scholar] [CrossRef]
- Hunt, A. L.; Nutcharoen, A.; Randall, J.; Papazian, A.; Deeken, J.; Maxwell, G. L. , et al. Integration of Multi-omic Data in a Molecular Tumor Board Reveals EGFR-Associated ALK-Inhibitor Resistance in a Patient With Inflammatory Myofibroblastic Cancer. The oncologist (Dayton, Ohio). 2023, 28, 730–736. [Google Scholar] [CrossRef]
- Behel, V.; Noronha, V.; Choughule, A.; Shetty, O.; Chandrani, P.; Kapoor, A. , et al. Impact of Molecular Tumor Board on the Clinical Management of Patients With Cancer. JCO Glob Oncol. 2022, 8. [Google Scholar] [CrossRef]
- Slootbeek, P. H. J.; Kloots, I. S. H.; Smits, M.; van Oort, I. M.; Gerritsen, W. R.; Schalken, J. A. , et al. Impact of molecular tumour board discussion on targeted therapy allocation in advanced prostate can-cer. Br J Cancer. 2022, 126, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Bourret, P.; Cambrosio, A. Genomic expertise in action: molecular tumour boards and decision-making in precision oncology. Sociol Health Illn. 2019, 41, 1568–1584. [Google Scholar] [CrossRef] [PubMed]
- Irelli, A.; Chiatamone Ranieri, S.; Di Giacomo, D.; Malatesta, S.; Patruno, L. V.; Tessitore, A. , et al. Role of the Molecular Tumor Board for the Personalized Treatment of Patients with Metastatic Breast Cancer: A Focus on the State of the Art in Italy. Cancers (Basel). 2023, 15, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Giuse, N. B.; Kusnoor, S. V.; Koonce, T. Y.; Naylor, H. M.; Chen, S. C.; Blasingame, M. N. , et al. Guiding Oncology Patients Through the Maze of Precision Medicine. J Health Commun. 2016, 21, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Bourret, P.; Cambrosio, A. Genomic expertise in action: molecular tumour boards and decision-making in precision oncology. Cost-effectiveness of precision medicine: a scoping review. Sociol Health Illn. 2019, 41, 1568–1584. [Google Scholar] [CrossRef]
- Tamborero, D.; Dienstmann, R.; Rachid, M. H.; Boekel, J.; Lopez-Fernandez, A.; Jonsson, M. , et al. The Molecular Tumor Board Portal supports clinical decisions and automated reporting for precision oncolo-gy. Nat Cancer. 2022, 3, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Brandts, C. H. Innovating the outreach of comprehensive cancer centers. Molecular oncology. 2019, 13, 619–623. [Google Scholar] [CrossRef] [PubMed]
- Oberst, S. Bridging research and clinical care - the comprehensive cancer centre. Mol Oncol. 2019, 13, 614–618. [Google Scholar] [CrossRef]
- Brandts, C. H. Innovating the outreach of comprehensive cancer centers. Molecular oncology. 2019, 13, 619–623. [Google Scholar] [CrossRef] [PubMed]
- Alarcon Garavito, G. A.; Moniz, T.; Deom, N.; Redin, F.; Pichini, A.; Vindrola-Padros, C. The imple-mentation of large-scale genomic screening or diagnostic programmes: A rapid evidence review. Eur J Hum Genet. 2023, 31, 282–295. [Google Scholar] [CrossRef] [PubMed]
- Fioretos, T.; Wirta, V.; Cavelier, L.; Berglund, E.; Friedman, M.; Akhras, M. , et al. Implementing precision medicine in a regionally organized healthcare system in Sweden. Nat Med. 2022, 28, 1980–1982. [Google Scholar] [CrossRef]
- Friedrich, B.; Vindrola-Padros, C.; Lucassen, A. M.; Patch, C.; Clarke, A.; Lakhanpaul, M. , et al. A very big challenge": a qualitative study to explore the early barriers and enablers to implementing a national genomic medicine service in England. Front Genet. 2023, 14, 1282034. [Google Scholar] [CrossRef]
- Keeling, P.; Clark, J.; Finucane, S. Challenges in the clinical implementation of precision medicine com-panion diagnostics. Expert Rev Mol Diagn. 2020, 20, 593–599. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).