Submitted:
10 April 2024
Posted:
11 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Illite and Kerogen Oil Phase Adsorption Experiment
2.1. Samples
2.2. Experimental Methods and Steps
3. Experimental Results
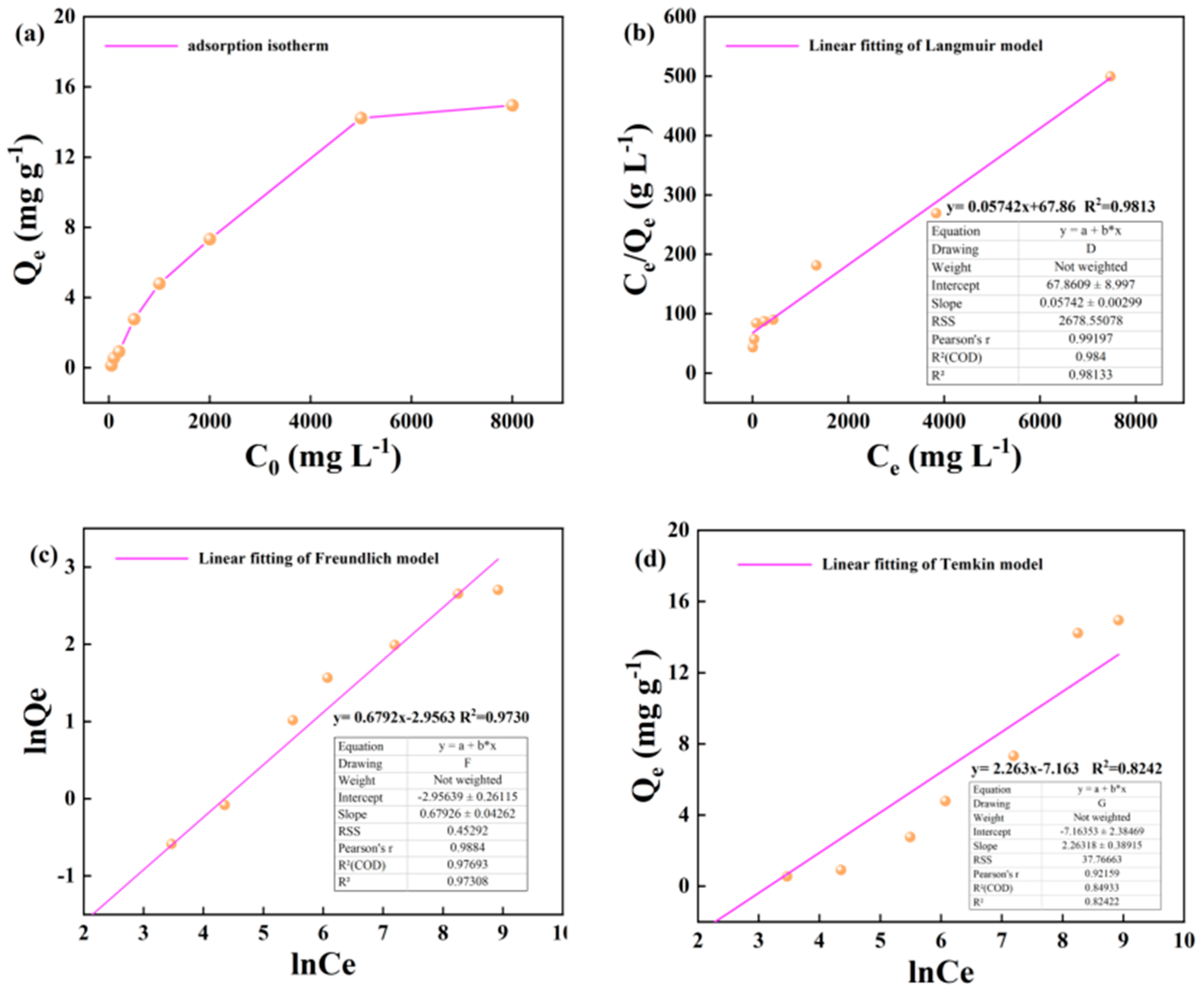
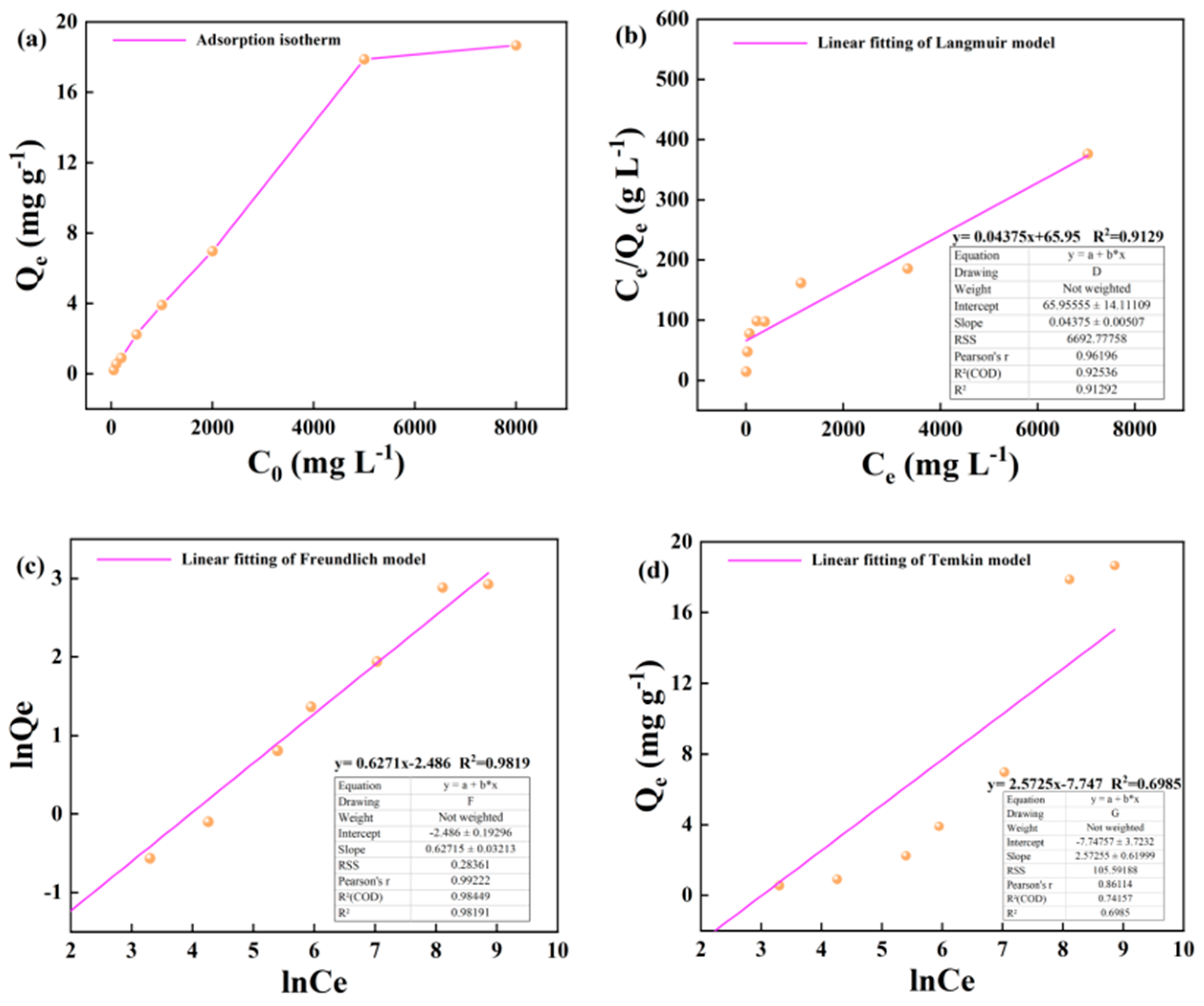
3.1. Illite Adsorption of Alkane Solution Isotherm Characteristics
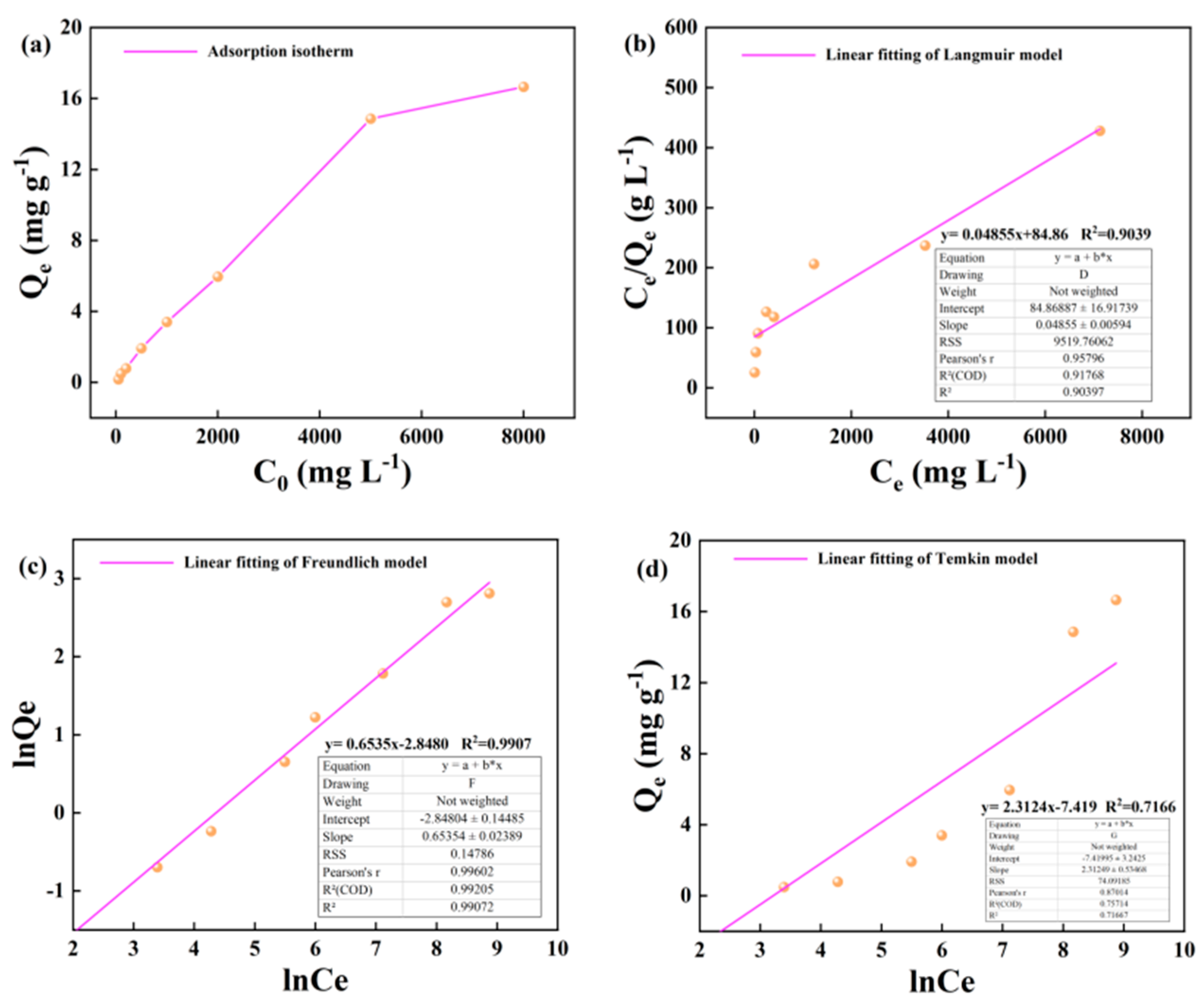
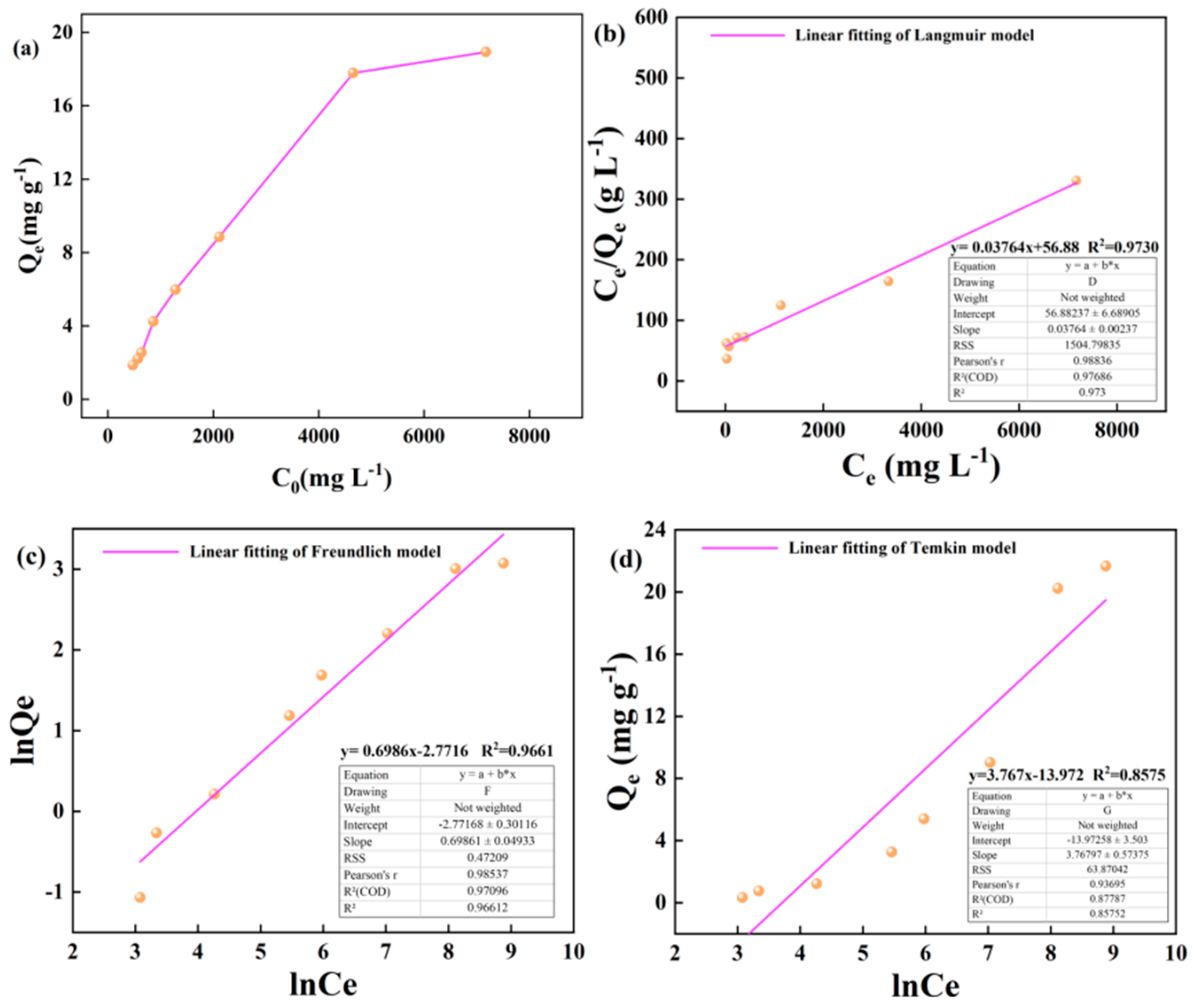
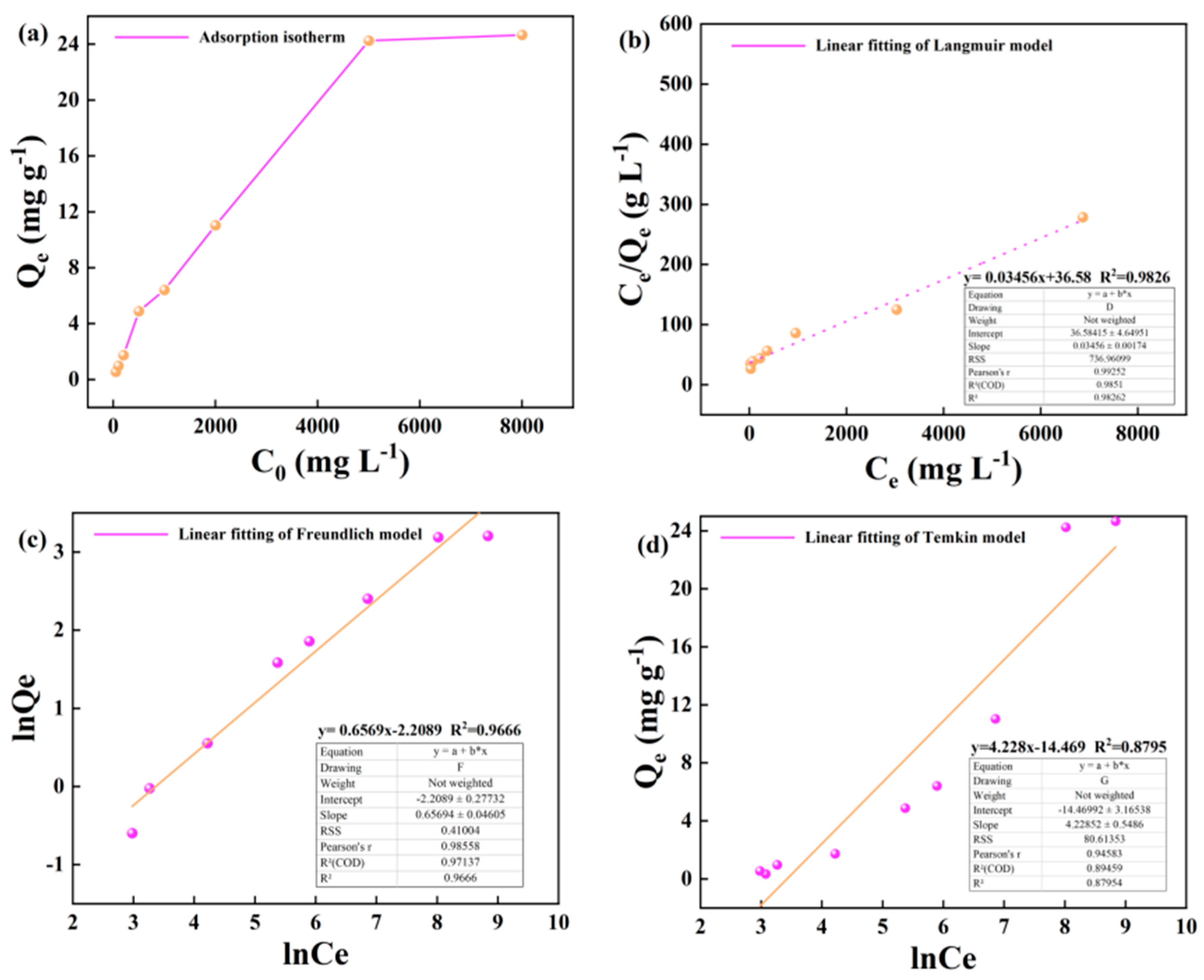
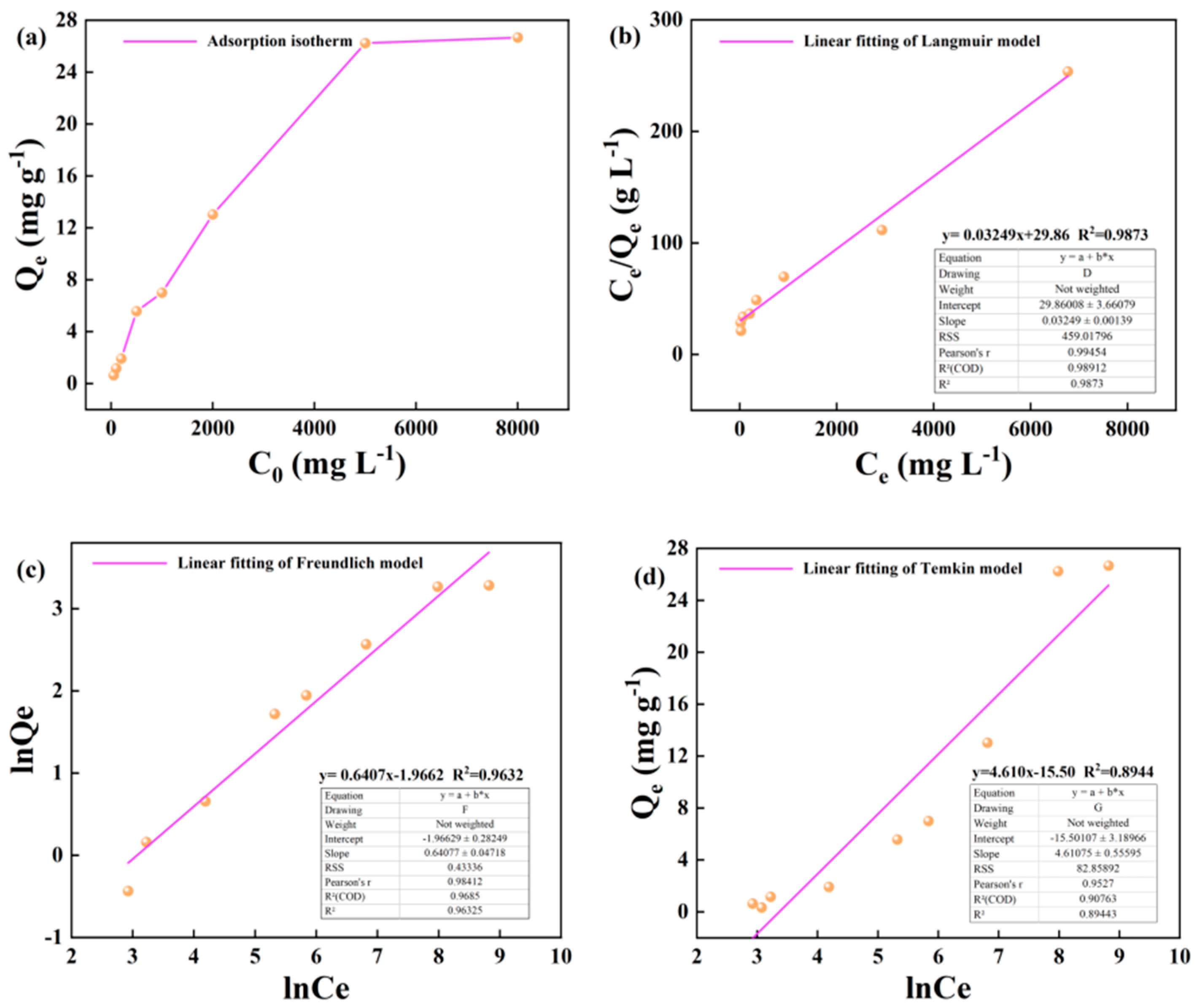
3.2. Kerogen Adsorption of Alkane Solution Isotherm Characteristics
4. Analysis and Discussion
4.1. Evaluation of Adsorption Isotherm Model

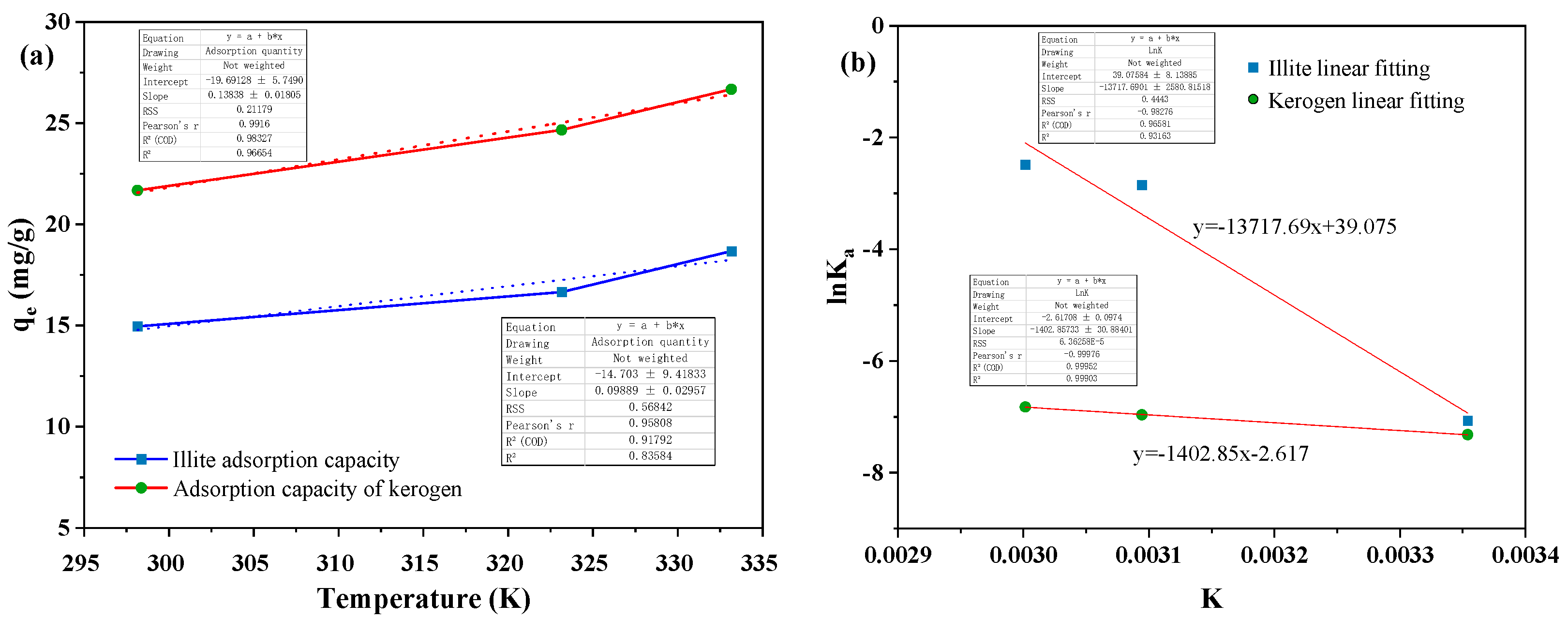
4.2. Thermodynamic Evaluation of Adsorption
| Sample | Temperature (K) | ΔHθ (kJ/mol) | ΔSθ (J/ (mol·K)) | ΔGθ (kJ/mol) |
|---|---|---|---|---|
| Illite | 298.15 | 114.049 | 324.869 | 18.145 |
| 323.15 | 7.654 | |||
| 333.15 | 6.887 | |||
| Kerogen | 298.15 | 11.663 | -21.757 | 18.146 |
| 323.15 | 18.711 | |||
| 333.15 | 18.899 |
5. Conclusion
- (1)
- The adsorption amount of illite and kerogen pores and surfaces increased with the increase of alkane solution concentration. At the initial adsorption stage of low concentration, the adsorption amount increased linearly with the increase of alkane concentration, and the adsorption rate slowed down with the continuous increase of alkane solution concentration. The reaction mechanism of temperature on the adsorption of heptadecane by illite and kerogen is basically the same, which almost does not affect the adsorption rate.
- (2)
- Based on the mathematical model of adsorption isotherm and the evaluation of fitting parameters, it was found that the adsorption of alkane solution by illite at 25 °C conformed to the Langmuir model, which was dominated by monolayer adsorption. At 50 °C and 60 °C, the adsorption heat and affinity do not need to be evenly distributed on the heterogeneous surface, and the adsorption process is more consistent with the Freundlich model, which is dominated by multi-layer adsorption. The Langmuir model is suitable for describing the adsorption process of kerogen to alkane solution. The adsorption process is mainly based on single molecule adsorption, forming a single molecule adsorption layer.
- (3)
- The adsorption process of heptadecane by illite and kerogen is an endothermic reaction. Temperature can improve the collision efficiency between adsorbate molecules and adsorbents, and accelerate the diffusion rate of heptadecane molecules into the micropores of matrix rocks, which is conducive to the adsorption reaction and enhances the adsorption capacity of matrix rocks.
- (4)
- The degree of chaos in the whole process of adsorption reaction of illite to heptadecane is increasing, while the degree of chaos in the whole process of adsorption reaction of kerogen to heptadecane is decreasing. Heptadecane shows the opposite phenomenon on the two substrates, which may be due to the different hydrophilic and lipophilic properties of the adsorption sites of illite and kerogen.
Acknowledgments
References
- MA Y, XIANG Q Y, DING K L. Development of oil shale at home and abroad[J]. World Petroleum Industry, 2024, 31 (1): 16 - 25.
- Zhijun J, Qian Z, Rukai Z, et al. Classification of lacustrine shale oil reservoirs in China and its significance[J]. Oil & Gas Geology, 2023,44 (4): 801-819. https: //doi.org/10.20944/preprints202404.0799.v1.
- Wenzhi Z, Congsheng B, Yongxin L, et al. Enrichment factors of movable hydrocarbons in lacustrine shale oil and exploration potential of shale oil in Gulong Sag, Songliao Basin, NE China[J]. Petroleum Exploration and Development, 2023, 50 (3): 455 - 467. https: //doi.org/10.1016/S1876-3804(23)60407-0.
- Ming L, Min L, Jinyou Z, et al. Evaluation of the compositions of lacustrine shale oil in China’s typical basins and its implications[J]. Oil & Gas Geology,2023,44 (6): 1479 - 1498.
- Caineng Z, Rukai Z, Dazhong D, Songtao W, et al. Scientific and technological progress,development strategy and policy suggestion regarding shale oil and gas[J]. Acta Petrolei Sinica, 2022, 43 (12): 1675 - 1686. https: //doi.org/10.7623/syxb202212001.
- Xiwu L, Xinyu W, Yuwei L, Jinqiang Z. Jiong L, Qian L. Current status and development direction of seismic prospecting technology for continental shale oil in China[J]. Acta Petrolei Sinica , 2023, 44 (12): 2270 - 2285. https: //doi.org/10.7623/syxb202312016.
- Caineng Z, Zhi Y, Rukai Z, et al. Progress in Chinas Unconventional Oil & Gas Exploration and Development and Theoretical Technologies[J]. ACTA GEOLOGICA SINICA, 2015, 89 (6): 979 - 1007. https: //doi.org/10.1111/1755-6724.12491.
- Qi C, Junqing C, Guiwu L, et al. Review on influencing factors and microscopic mechanism of shale adsorption capacity by molecular dynamics simulation[J]. Journal of Central South University (Science and Technology), 2022, 53 (09): 3474 - 3489.
- Laixing C, Tian Y, Jingchun T, et al. Advances in Studies of Development and Growth Mechanisms of Clay Minerals in Tight Sandstone Reservoirs[J]. ACTA SEDIMEN TOLOGICA SINICA, 2023, 41 (06): 1859 - 1889.
- Wei D, Jinchuan J, Fengqin W, et al. Thermodynamics and kinetics of water vapor adsorption onto shale: A case study of the Permian Shanxi Formation,Ordos Basin[J]. OIL & GAS GEOLOGY, 2021, 42 (01): 173 - 185.
- Hexin H , Rongxi L , Zhou L, et al. Comparative study of methane adsorption of Middle-Upper Ordovician marine shales in the western Ordos Basin, NW China: Insights into impacts of moisture on thermodynamics and kinetics of adsorption[J]. Chemical Engineering Journal, 2022, 446 (P4). https: //doi.org/10.1016/j.cej.2022.137411.
- TAO W. Mechanism of supercritical methane adsorption in Wufeng and Longmaxi shales from the Dongxi area, Sichuan Basin[D]. China University of Geosciences, 2023.
- FA A., Gauthier F , Amatalrhman R, et al. Marcellus shale characteristics and CO2 adsorption: equilibrium and kinetic modeling study[J]. IOP Conference Series: Earth and Environmental Science, 2022, 1003 (1). https: //doi.org/10.1088/1755-1315/1003/1/012027.
- Wei T. Study on the Adsorption Law of CH4, C2H6 and Their Binary Mixtures in Shale[D]. China University of Petroleum (Beijing), 2022.
- Zhan C, Younan L. Study on shale adsorption in Fuxian area of Ordos Basin[J]. 石油化工应用, 2023, 42 (10): 83 - 90.
- Lian P. Study on adsorption kinetics of nonionic surfactants in Bohai Oilfield. Unconventional Oil & Gas, 2023, 10 (06): 61 - 67.
- Shuling X , Manfei C , Hui Y , et al. Experimental investigation of thermodynamic and kinetic behaviors from water vapor adsorption on four typical clay minerals[J]. Gas Science and Engineering, 2023, 111. https: //doi.org/10.1016/j.jgsce.2023.204933.
- Xusong C. Study on the kinetics of adsorption of CH4 and CO2 on porous materials[D]. Chongqing University , 2022.
- Muath A , Qadeer A M S , Hamid R. A sorption-kinetics coupled dual-porosity poromechanical model for organic-rich shales[J]. Computers and Geotechnics, 2022, 147. https: //doi.org/10.1016/j.compgeo.2022.104755.
- Mingjie Z, Mingxin Y,Tianrang J, et al. Adsorption kinetic characteristics of anthracite in Longshan Mine[J]. Natural Gas Geoscience, 2022, 33 (02): 267 - 276.
- Pei X, Lixia Z, Quansheng L, et al. Isothermal adsorption properties of supercritical methane on shale[J]. Natural Gas Geoscience, 2020, 31 (09): 1261 - 1270.
- Zeyuan S. Productivity Difference of CO2 Rich Coalbed Methane Wells andAdsorption Thermodynamic Mechanism in the East of Haishiwan Coalfield[D]. HENAN POLYTECHNIC UNIVERSITY, 2022.
- Longjie M. Experimental Investigation into Simultaneous Adsorption Process of Water vapor and Methane onto Shales[D]. China University of Petroleum (Beijing), 2021.
- Wang M , Lun Z , Zhao C , et al. Influences of primary moisture on methane adsorption within Lower Silurian Longmaxi shales in the Sichuan Basin, China[J]. Energy Fuels, 2020, 34 (9): 10810 - 10824. https: //doi.org/10.1021/acs.energyfuels.0c01932.
- Shan H. The Characteristic and Influencing Factors of High-pressure Methane Adsorption on Dry Coals[D]. China University of Petroleum (Beijing), 2020.
- Zhenzhen Y , Kangle D , Chao H, et al. Adsorption of diesel oil onto natural mud shale: from experimental investigation to thermodynamic and kinetic modelling[J]. International Journal of Environmental Analytical Chemistry, 2023, 103 (15): 3468 - 3482. https: //doi.org/10.1080/03067319.2021.1910247.
- Yong D, Desheng H, Jitian Z, et al. Hydrocarbon accumulation regularities ,new fields and new types of exploration, and resource potentials in Beibuwan Basin[J]. ACTA PETROLEI SINICA, 2024, 45 (01): 202 - 225. https: //doi.org/10.7623/syxb202401012.
- Caihong Y, Donghui J, Xinghai Z, et al. Key Factors and Mode of Hydrocarbon Accumulation of Haizhong Sag in Beibu Gulf Basin[J]. OFFSHORE OIL, 2023, 43 (04): 41 - 45.
- Caineng Z, Rukai Z, Dazhong D, et al. Scientific and technological progress,development strategy and policy suggestion regarding shale oil and gas[J]. ACTA PETROLEI SINICA, 2022, 43 (12): 1675 - 1686. https: //doi.org/10.7623/syxb202212001.
- Jieqiong Z, Xiaohan L, Heng Y, et al. Shale characteristics and shale oil and gas resource potential of Liushagang Formation in Fushan sag,Beibu Gulf basin[J]. China Offshore Oil and Gas, 2022, 34 (6): 65 - 79.
- Rontani F J ,Smik L ,Divine D , et al. Gas chromatography-mass spectrometry selected ion monitoring and gas chromatography-tandem mass spectrometry selected reaction monitoring analyses of mono-, di- and tri-unsaturated Csub25/sub highly branched isoprenoid alkene biomarkers in sea ice and sediment samples: A comparative study.[J]. Rapid communications in mass spectrometry : RCM, 2024, 38 (6): e9704 - e9704. https: //doi.org/10.1002/rcm.9704.
- Naixin W, Zelong L, Jing Y, et, at. Research and Application on the Determination of Hydrocarbon Molecular Composition in Middle Distillate Oil by GC-MS[J/OL]. Acta Petrolei Sinica (Petroleum Processing Section), 1 - 13[ 2024 - 03 - 28].
- Shuling X ,Manfei C ,Hui Y , et al. Experimental investigation of thermodynamic and kinetic behaviors from water vapor adsorption on four typical clay minerals[J]. Gas Science and Engineering, 2023, 111. https: //doi.org/10.1016/j.jgsce.2023.204933.
- Yufeng C. Study on adsorption mechanism and thermodynamic model of shale gas in Micro-Nano pore [D]. CENTRAL SOUTH UNIVERSITY, 2022.
- Shangping L, Xijian L, Xing Y, et al. Optimal model of isothermal adsorption for shale gas under high temperature and high pressure[J]. CHINA MINING MAGAZINE, 2018, 27 (06): 160 - 166. https: //doi.org/10.12075/j.issn.1004-4051.2018.06.007.
- Qayyimah A A M , Farhah D M ,Belladonna M , et al. Supercritical methane adsorption measurement on shale using the isotherm modelling aspect.[J]. RSC advances, 2022, 12 (32): 20530 - 20543. https: //doi.org/10.1039/D2RA03367D.
- Wei T, Huiqin L, Qin W, et al. A New Method for Calculating the Isosteric Heat of Adsorption of CH4 on Shale Under High Pressure[J]. Contemporary Chemical Industry, 2022, 51 (08): 1916 - 1922 + 1926.
- Majd M M , Kordzadeh-Kermani V , Ghalandari V , et al. Adsorption isotherm models: A comprehensive and systematic review (2010 − 2020)[J]. Science of The Total Environment, 2022, 812: 151334. https: //doi.org/10.1016/j.scitotenv.2021.151334.
- Al-Ghouti M A , Da'ana D A . Guidelines for the use and interpretation of adsorption isotherm models: A review[J]. Journal of hazardous materials, 2020, 393: 122383. https: //doi.org/10.1016/j.jhazmat.2020.122383.
- Xiaohu D , Wenjing X ,Renjing L , et al. Insights into adsorption and diffusion behavior of shale oil in slit nanopores: A molecular dynamics simulation study[J]. Journal of Molecular Liquids, 2022, 359. https: //doi.org/10.1016/j.molliq.2022.119322.
- Yu L, Huiqin L, Yabin F, et al. Adsorption Behavior of Heavy Oil on Montmorillonite Surface by Typical Surfactant: Molecular Dynamics Simulation[J]. CHINESE JOURNAL OF COMPUTATIONAL PHYSICS, 2023, 40 (05): 583 - 596.
- Weitao W, Xiangli C, Baiqin Y. Calculation of Adsorption Thermodynamics Parameters for Adsorption on the Solid-Liquid Interface[J]. Univ. Chem, 2021, 36 (02): 233 - 240.
- Jiheng L, Liangqiong P, Lijun G, et al. Solid-liquid Adsorption Isotherm Model and Thermodynamic Parameter Calculation[J]. LEATHERSCIENCE ANDENGINEERING, 2023, 33 (06): 36 - 43.
- Jiaojiao H. Effect of temperature on adsorption efficiency of activated carbon[J]. Engineering Technology Magazine, 2021 (6): 1.
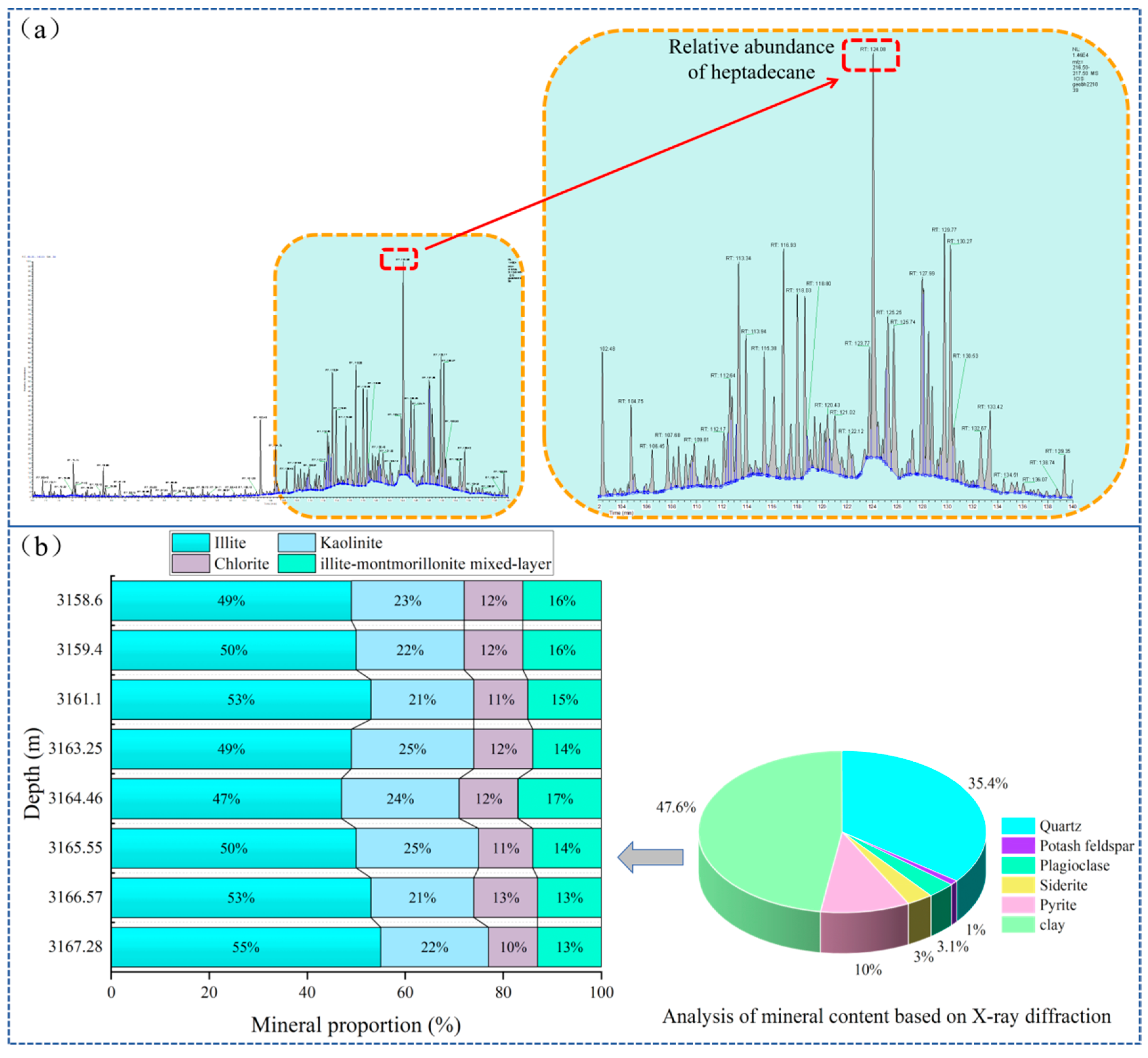
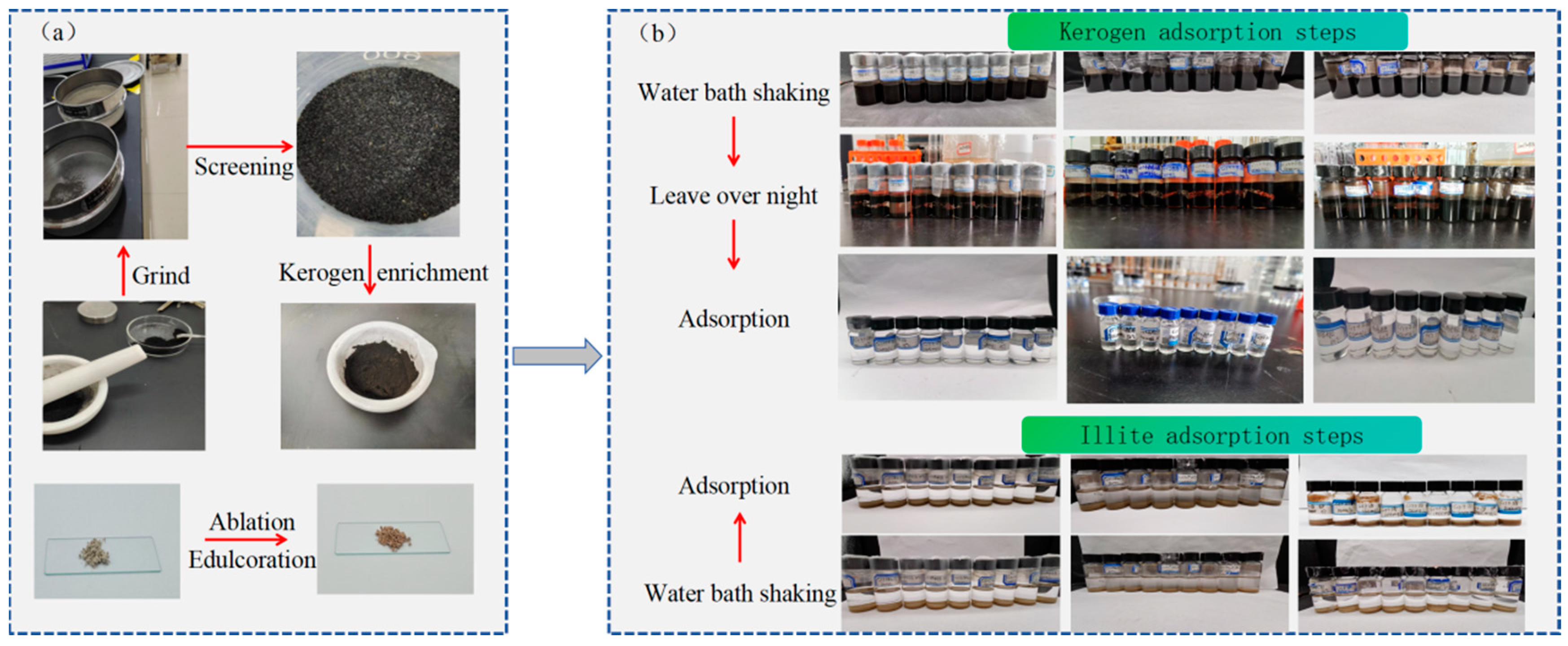
| Sample Kind | Purity/Specification | Sample Source | |
|---|---|---|---|
| Adsorbent | Illite | 75-100μm | Dehang Mineral Products Co., Ltd. |
| Kerogen | 90.51% | Shale core debris in Weixinan Sag, Beibuwan Basin | |
| Adsorbate | Heptadecane | AR | Chengdu Cologne Chemicals Co., Ltd. |
| Tetrachloroethylene | AR | Chengdu Cologne Chemicals Co., Ltd. | |
| Concentration (mg/L) | 20 | 40 | 60 | 80 | 100 |
| Volume (mL) | 10 | 10 | 10 | 10 | 10 |
| Concentration (mg/L) | 50 | 100 | 200 | 500 | 1000 | 2000 | 5000 | 8000 |
| Volume (mL) | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| Concentration (mg/L) | 5 | 10 | 20 | 50 | 100 |
| Volume (mL) | 10 | 10 | 10 | 10 | 10 |
| Concentration (mg/L) | 50 | 100 | 200 | 500 | 1000 | 2000 | 5000 | 10000 |
| Volume (mL) | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| Adsorbent | Temperature/℃ | Langmuir Model |
Freundlich Model |
Temkin model |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| KL | KF | n | A | |||||||
| Illite | 25 | y= 0.05742x+67.86 | y= 0.6792x-2.9563 | y= 2.263x-7.163 | ||||||
| 17.415 | 0.000849 | 0.981 | 0.0520 | 1.472 | 0.973 | 2.263 | -7.163 | 0.824 | ||
| 50 | y= 0.04855x+84.86 | y= 0.6535x-2.8480 | y= 2.3124x-7.419 | |||||||
| 20.597 | 0.00572 | 0.903 | 0.0579 | 1.530 | 0.990 | 2.312 | -7.419 | 0.716 | ||
| 60 | y= 0.04375x+65.95 | y= 0.6271x-2.486 | y= 2.5725x-7.747 | |||||||
| 22.857 | 0.000663 | 0.912 | 0.0832 | 1.594 | 0.981 | 2.572 | -7.747 | 0.698 | ||
| Kerogen | 25 | y= 0.03764x+56.88 | y= 0.6986x-2.7716 | y=3.767x-13.972 | ||||||
| 26.567 | 0.00066 | 0.973 | 0.062 | 1.431 | 0.966 | 3.767 | -13.972 | 0.857 | ||
| 50 | y= 0.03456x+36.58 | y=0.6569x-2.2089 | y=4.228x-14.469 | |||||||
| 28.935 | 0.00945 | 0.982 | 0.109 | 1.522 | 0.966 | 4.228 | -14.469 | 0.879 | ||
| 60 | y= 0.03249x+29.86 | y= 0.6407x-1.9662 | y=4.610x-15.50 | |||||||
| 30.778 | 0.00108 | 0.987 | 0.139 | 1.560 | 0.963 | 4.610 | -15.50 | 0.894 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




