Submitted:
09 April 2024
Posted:
10 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Plant Material and Fixed Oil Extraction
2.2. Cell Lines
2.3. Animals
2.4. Gas Chromatography-Mass Spectrometry Analysis (GC-MS)
2.5. Experimental Design
- Control-group: animals served as control and received equivalent volume of H2O + NaCl (1ml) every day (5/7),
- DMBA-group: animals were treated with one dose per week of DMBA (20 mg/kg b.w./week) for 28 days,
- DMBA+PL-group: animals received (20 mg/kg b.w.) once a week and daily dose (5/7) of PL FOt (100 mg/kg b.w.) for 28 days,
- PL-group: animals received daily dose (5/7) of PL FOt (100 mg/kg b.w.) for 28 days,
2.6. Anthropometric Parameters
2.7. Plasma Biochemical Parameters
2.8. Preparation of Homogenate and Estimation of Protein
2.9. Oxidative Stress Biomarkers
2.9.1. Lipid Peroxidation (TBARS) Assay
2.9.2. Sulfhydryl Groups (-SH) Determination
2.9.3. Hydrogen Peroxide (H2O2) Determination
2.10. Enzymes Antioxidant Capacity
2.11. Histopathological Analysis
2.12. In Vitro Anticancer Effect
2.13. Statistical Analysis
3. Results
3.1. Gas Chromatography-Mass Spectrometry Analysis (GC-MS)
| Peaks N° |
Retention time (min) | Library identified compound name/ID | Area (%) | Chemical class | Molecular formula |
|---|---|---|---|---|---|
| 1 | 18.018 | Bicyclo(2.2.1)heptan-2-one, 1 | 05.2 | Cyclic monoterpene | C7H10O |
| 2 | 19.078 | Benzeneacetic acid | 13.7 | Monocarboxylic acid | C8H8O2 |
| 3 | 34.896 | 1H-Indole-3-acetic acid, methyl ester | 53.8 | Methyl ester | C11H11NO2 |
| 4 | 36.922 | Hexadecanoic acid, | 09.9 | Fatty acidethyl ester | C16H32O2 |
| 5 | 40.103 | Methy (l1Z,14Z,17Z)-eicosatrienoate | 11.9 | Acid methyl ester | C21H36O2 |
| 6 | 40.211 | 13-octodecenoic acid, methyl | 05.5 | Monocarboxylic acid | C28H50O6 |
| Monoterpene | 05.2 | ||||
| Fatty acid | 19.2 | ||||
| Ester | 75.6 | ||||
| Total identified | 100% | ||||
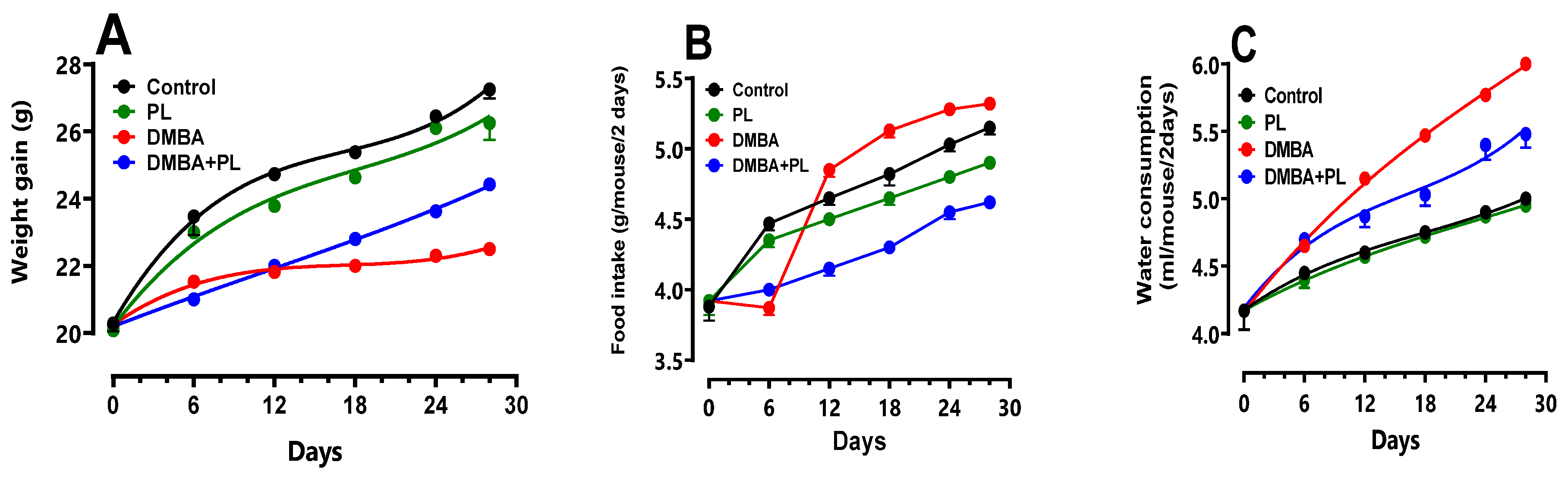
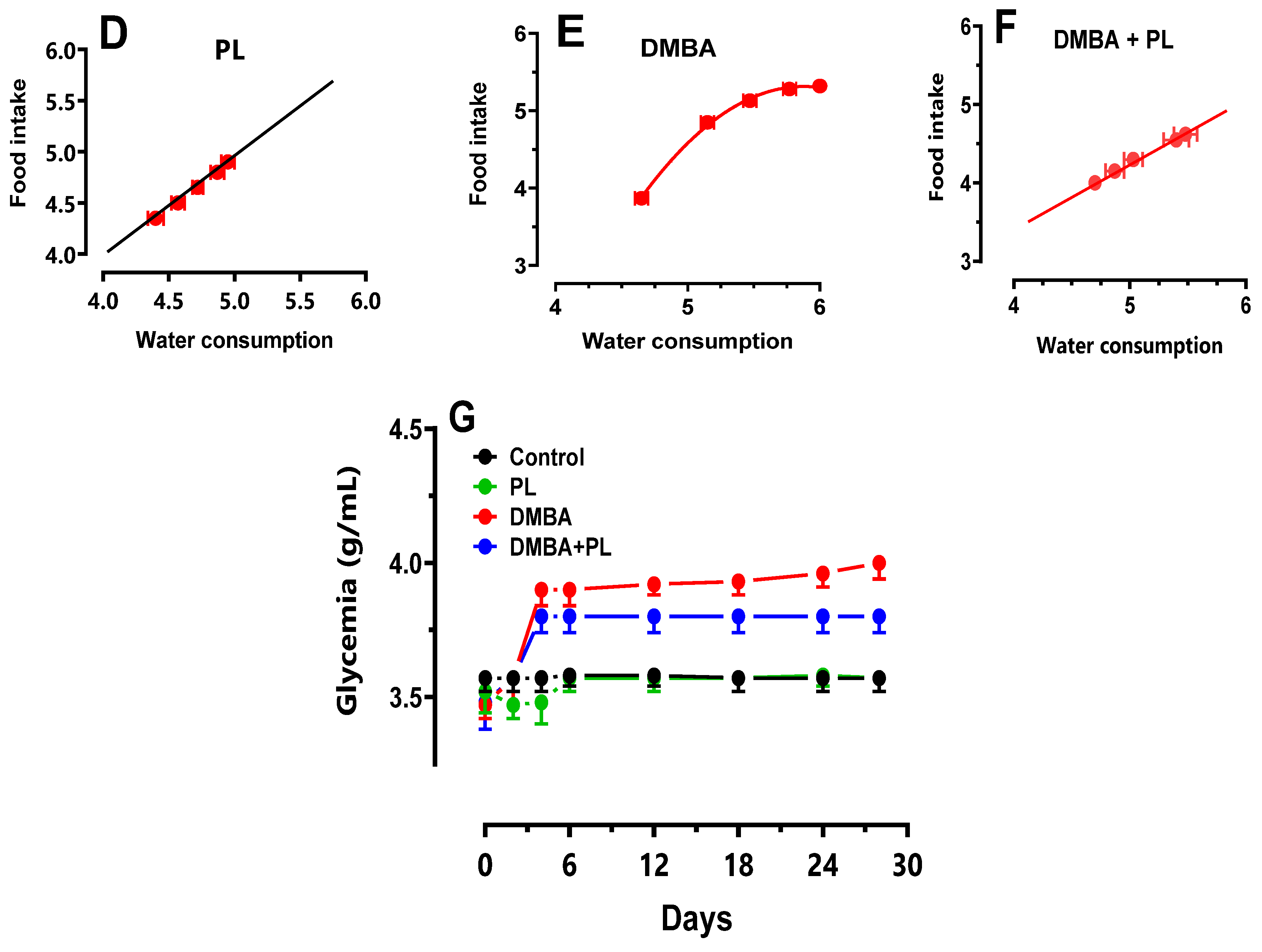
3.2. Anthropometric Parameters
3.3. Plasma Biochemical Parameters
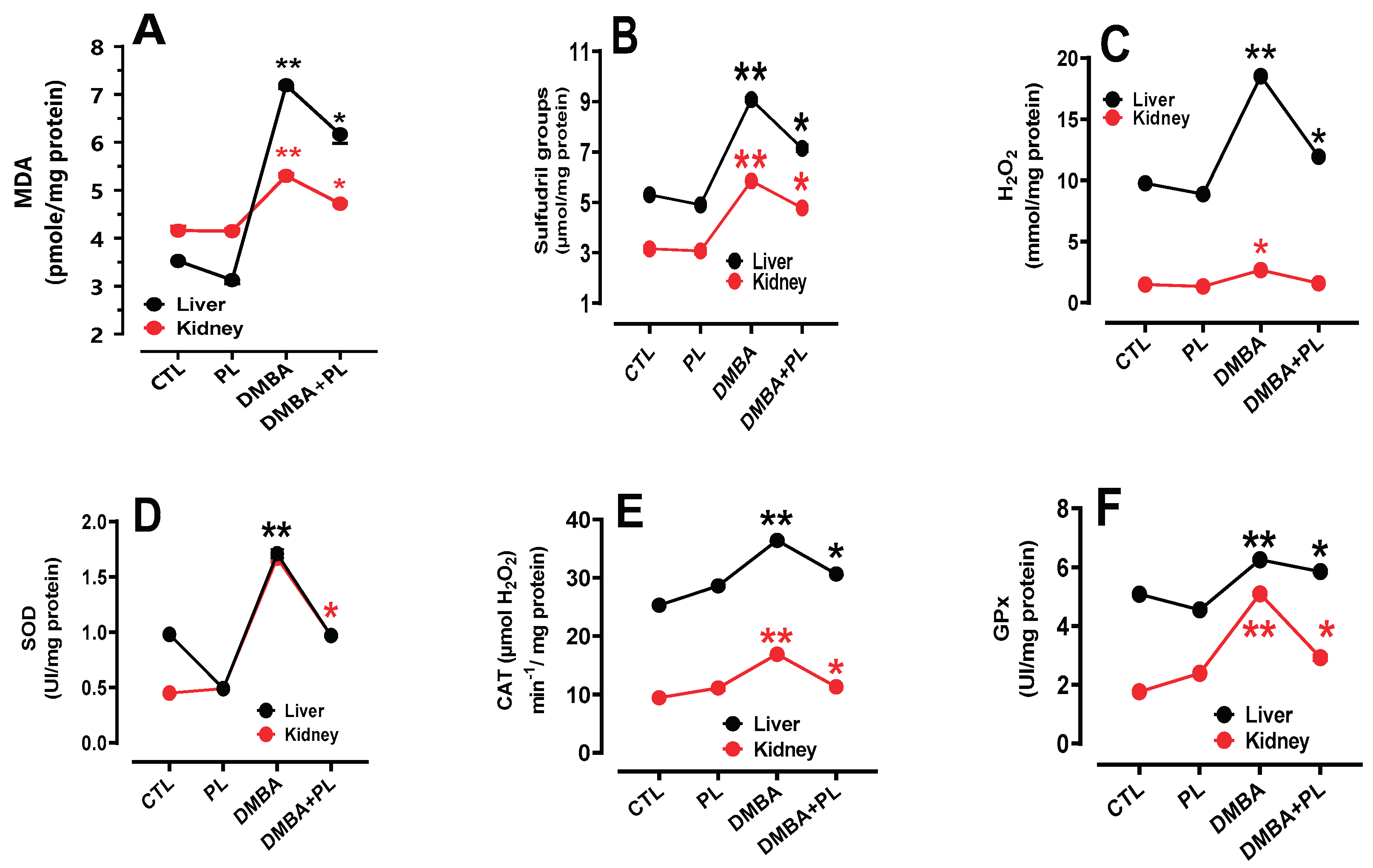
3.4. Oxidative Stress Biomarkers
3.4.1. In Liver Tissues
3.4.2. In Kidney Tissues
3.5. Enzymes Antioxidant Capacity
3.5.1. In Liver Tissues
3.5.2. In Kidney Tissues
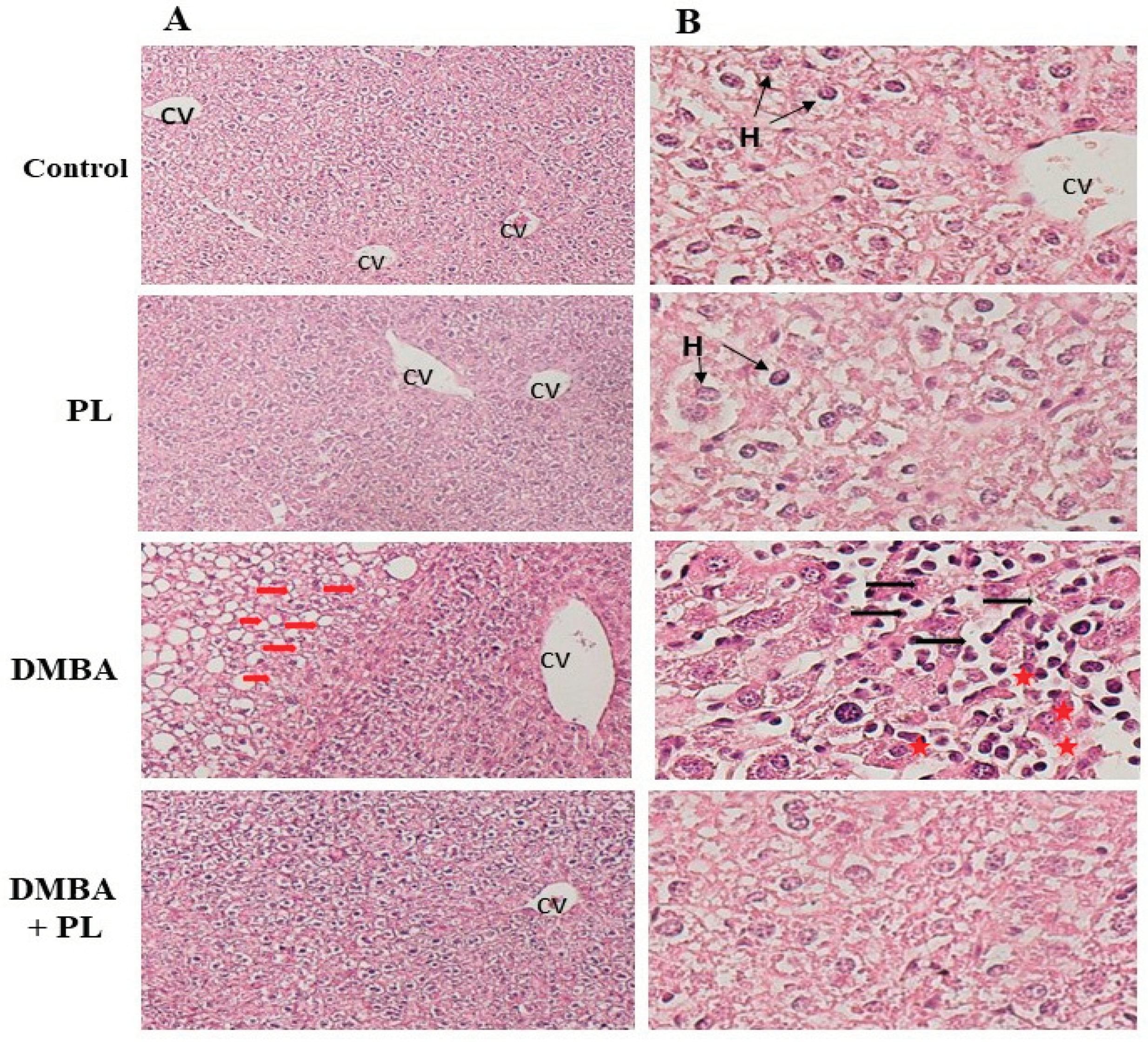
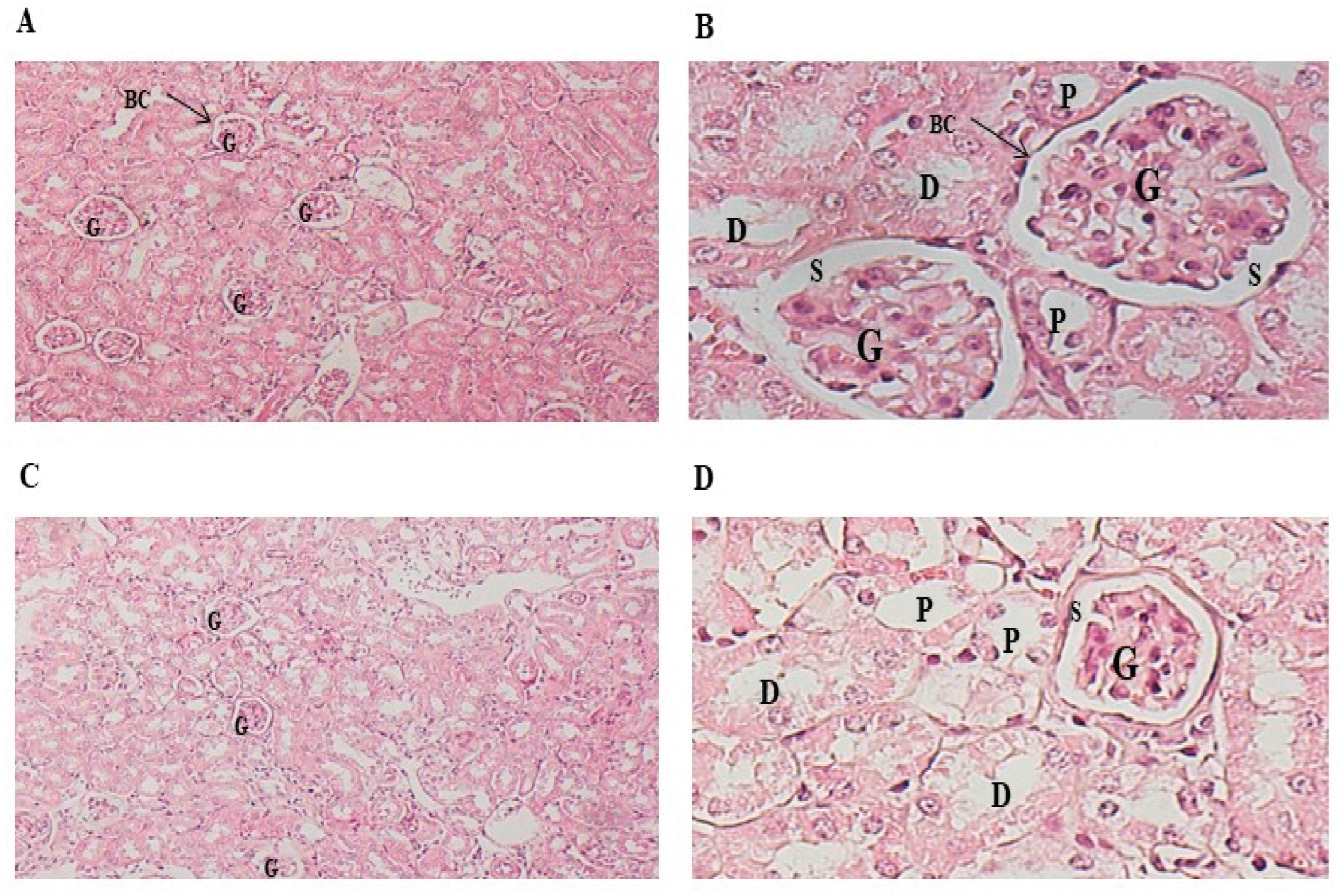
3.6. Histopathology Section
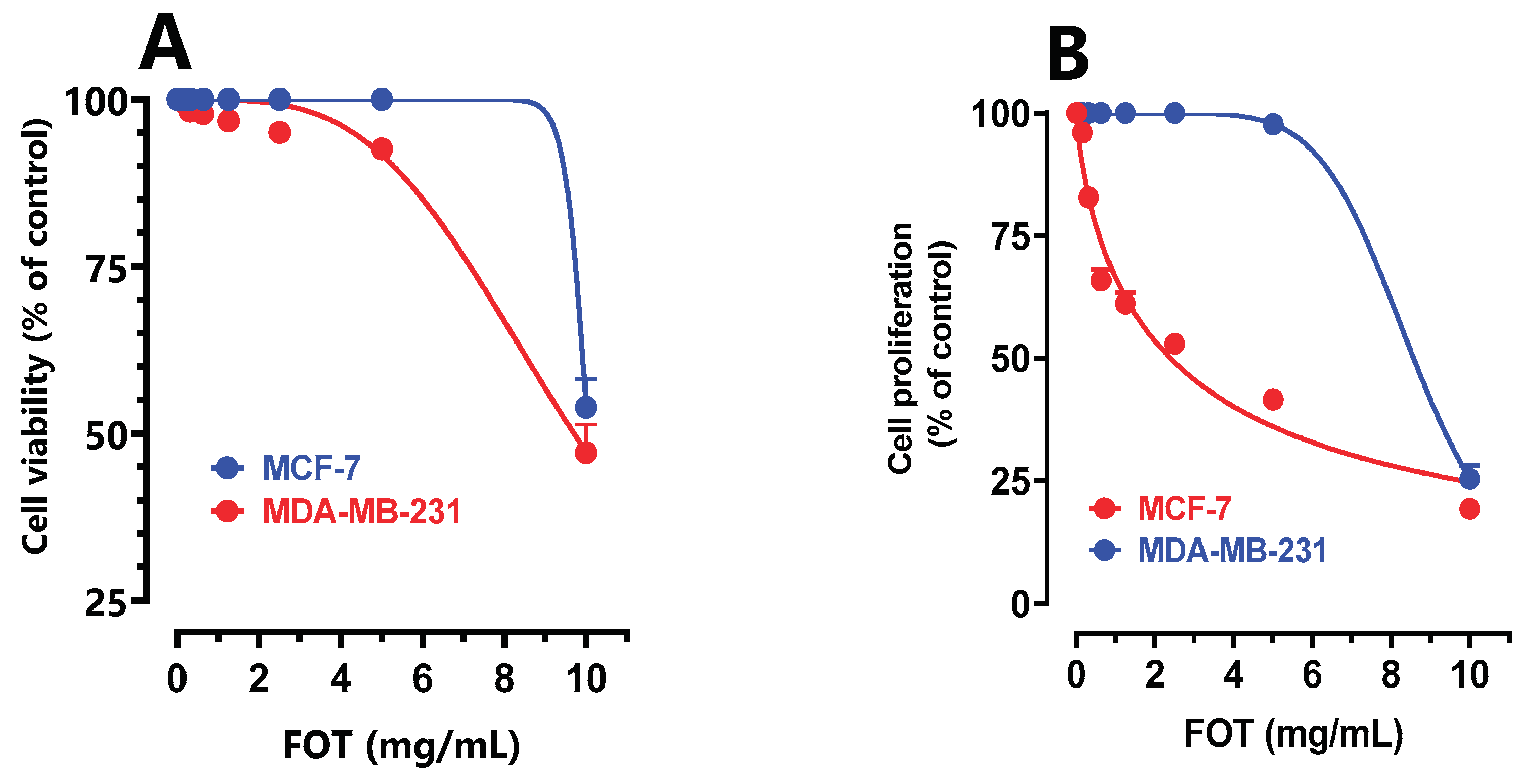
3.7. In Vitro Anticancer Effect
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Blumer, M.; Youngblood, W.W. Polycyclic Aromatic Hydrocarbons in Soils and Recent Sediments. Science 1975, 188, 53–55. [Google Scholar] [CrossRef] [PubMed]
- Ananda, H.; Kumar, K.S.S.; Sudhanva, M.S.; Rangappa, S.; Rangappa, K.S. A trisubstituted pyrazole derivative reduces DMBA-induced mammary tumor growth in rats by inhibiting estrogen receptor-α expression. Mol. Cell. Biochem. 2018, 449, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Sofi, M.S.; Sateesh, M.K.; Bashir, M.; Ganie, M.A.; Nabi, S. Chemopreventive and anti-breast cancer activity of compounds isolated from leaves of Abrus precatorius L. 3 Biotech 2018, 8, 371. [Google Scholar] [CrossRef] [PubMed]
- Omayma, A.; Zaineb, Abdelkafi-Koubaa., Ilhem Bettaieb-Dridi, Lamjed Toumi, Lamjed Marzouki, and Ouajdi Souilem, Pistacia lentiscus L. revealed potential anti-inflammatory, antioxidant and antiproliferative enhancement in vitro breast cancer cells and in vivo DMBA-C57BL/6 mammary cancer. PLoS ONE, 2024.
- Bagheri, S.; et al. Effects of Pistacia atlantica on oxidative stress markers and antioxidant enzymes expression in diabetic rats. Journal of the American College of Nutrition 2019, 38, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Abidi, O.; Abdelkafi-Koubaa, Z.; Fares-Zagrouba, N.; Elkahoui, S.; Toumi, L.; Marzouki, L.; Souilem, O. Hytochemical Analysis And Biological Activities Of Two Oil-Bearing Extracts From Fresh Pistacia Lentiscus. Bull. Chem. Soc. Ethiop. 2023, 37, 1487–1501. [Google Scholar] [CrossRef]
- Shamsi-Baghbanan, H.; Sharifiyan, A.; Esmaeili, S.; Minaei, B. Hepatoprotective Herbs, Avicenna Viewpoint. Iran. Red Crescent Med J. 2014, 16, e12313. [Google Scholar] [CrossRef]
- Nikolakopoulou, V.; Printezis, P.; Maniatis, V.; Kontizas, D.; Vosinakis, S.; Chatzigrigoriou, P.; Koutsabasis, P. Conveying Intangible Cultural Heritage in Museums with Interactive Storytelling and Projection Mapping: The Case of the Mastic Villages. Heritage 2022, 5, 1024–1049. [Google Scholar] [CrossRef]
- Kim, K.E.; Ko, K.-H.; Heo, R.W.; Yi, C.-O.; Shin, H.J.; Kim, J.Y.; Park, J.-H.; Nam, S.; Kim, H.; Roh, G.S. Artemisia annuaLeaf Extract Attenuates Hepatic Steatosis and Inflammation in High-Fat Diet-Fed Mice. J. Med. Food 2016, 19, 290–299. [Google Scholar] [CrossRef]
- Klibet, F.; et al. , Oxidative stress-related liver dysfunction by sodium arsenite: Alleviation by Pistacia lentiscus oil. Pharm. Biol. 2016, 54, 354–363. [Google Scholar] [CrossRef]
- Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Lampri, E.; Fitsiou, E.; Vasileiadis, S.; Vamvakias, M.; Bardouki, H.; Goussia, A.; Malamou-Mitsi, V.; Panayiotidis, M.I.; et al. Dietary mastic oil extracted from Pistacia lentiscus var. chia suppresses tumor growth in experimental colon cancer models. Sci. Rep. 2017, 7, 3782. [Google Scholar] [CrossRef]
- Belyagoubi-Benhammou, N.; Belyagoubi, L.; El Zerey-Belaskri, A.; Zitouni, A.; Ghembaza, N.; Benhassaini, H.; Atik-Bekkara, F.; Piras, A.; Falconieri, D.; Rosa, A. Fatty acid composition and antioxidant activity of Pistacia lentiscus L. fruit fatty oil from Algeria. J. Food Meas. Charact. 2018, 12, 1408–1412. [Google Scholar] [CrossRef]
- Abidi, O.; Abdelkafi-Koubaa, Z.; Fares-Zagrouba, N.; Elkahoui, S.; Toumi, L.; Marzouki, L.; Souilem, O. Phytochemical analysis and biological activities of two oil-bearing extracts from fresh Pistacia lentiscus. Bull. Chem. Soc. Ethiop. 2023, 37, 1487–1501. [Google Scholar] [CrossRef]
- Hawkins, P.; Morton, D.B.; Burman, O.; Dennison, N.; Honess, P.; Jennings, M.; Lane, S.; Middleton, V.; Roughan, J.V.; Wells, S.; et al. A guide to defining and implementing protocols for the welfare assessment of laboratory animals: Eleventh report of the BVAAWF/FRAME/RSPCA/UFAW Joint Working Group on Refinement. Lab. Anim. 2011, 45, 1–13. [Google Scholar] [CrossRef] [PubMed]
- AVMA American Veterinary Medical Association guidelines for the euthanasia of animals: 2020 edition. 2020. 2020.0.1, 1–121.
- Reitman, S.; Frankel, S. A Colorimetric Method for the Determination of Serum Glutamic Oxalacetic and Glutamic Pyruvic Transaminases. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, S.; Barr, J.K. A simplified method of quantitating protein using the biuret and phenol reagents. Anal. Biochem. 1978, 86, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Draper, H.H.; Hadley, M. Malondialdehyde determination as index of lipid Peroxidation. Methods Enzymol. 1990, 186, 421–431. [Google Scholar]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Kakinuma, K.; et al. A Determination of H2O2 Release by the Treatment of Human: Blood Polymorphonuclear Leukocytes with Myristate. The Journal of Biochemistry 1979, 86, 87–95. [Google Scholar]
- Rotruck, J.T.; Pope, A.L.; Ganther, H.E.; Swanson, A.B.; Hafeman, D.G.; Hoekstra, W.G. Selenium: Biochemical Role as a Component of Glutathione Peroxidase. Science 1973, 179, 588–590. [Google Scholar] [CrossRef]
- Kakkar, P.; Das, B.; Viswanathan, P.N. A modified spectrophotometric assay of superoxide dismutase. Indian Journal of Biochemistry & Biophysics 1984, 21, 130–132. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Vaya, J.; Mahmood, S. Flavonoid content in leaf extracts of the fig (Ficus carica L.), carob (Ceratonia siliqua L.) and pistachio (Pistacia lentiscus L.). Biofactors 2006, 28, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Yuan, G.; Xu, M.; Tan, M.; Dong, J.; Chen, X. Supplementation with Docosahexaenoic Acid and Vitamin E Improves Hepatic Triglyceride Accumulation Induced by High-Fat Diet in Mice. Eur. J. Lipid Sci. Technol. 2020, 123, 2000224. [Google Scholar] [CrossRef]
- Liao, C.-C.; Day, Y.-J.; Lee, H.-C.; Liou, J.-T.; Chou, A.-H.; Liu, F.-C. Baicalin Attenuates IL-17-Mediated Acetaminophen-Induced Liver Injury in a Mouse Model. PLoS ONE 2016, 11, e0166856. [Google Scholar] [CrossRef] [PubMed]
- Ostovan, M.; et al. The short-term effects of Pistacia lentiscus oil and sesame oil on liver and kidney pathology of rats and human cancer cell lines. Galen Medical Journal 2020, 9, e2001. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, H.B.; Santani, D.D.; Sagar, R. Chemopreventive potential of Apium leptophyllum (Pers.) against DMBA induced skin carcinogenesis model by modulatory influence on biochemical and antioxidant biomarkers in Swiss mice. Indian Journal of Pharmacology 2014, 46, 531. [Google Scholar] [PubMed]
- Balan, K.; Prince, J.; Han, Z.; Dimas, K.; Cladaras, M.; Wyche, J.; Sitaras, N.; Pantazis, P. Antiproliferative activity and induction of apoptosis in human colon cancer cells treated in vitro with constituents of a product derived from Pistacia lentiscus L. var. chia. Phytomedicine 2007, 14, 263–272. [Google Scholar] [CrossRef]
- Pachi, V.K.; Mikropoulou, E.V.; Gkiouvetidis, P.; Siafakas, K.; Argyropoulou, A.; Angelis, A.; Mitakou, S.; Halabalaki, M. Traditional uses, phytochemistry and pharmacology of Chios mastic gum (Pistacia lentiscus var. Chia, Anacardiaceae): A review. J. Ethnopharmacol. 2020, 254, 112485. [Google Scholar] [CrossRef]
| Lipid profile | Liver function | kidney function | ||||||||||
| TC (g/L) | TG (g/L) | HDL (g/L) | LDL (g/L) | ALT (UI/I) | AST (UI/I) | PAL (UI/I) | C-RP (µg/dL) | Crea (mg/L) | Urea (g/L) | |||
| Group control | 1 ± 0.2 | 1.9 ± 0.1 | 1.1 ± 0.2 | 0.9 ± 0.1 | 98 ± 52 | 196 ± 72 | 212 ± 38 | 0.9 ± 0.6 | 4.9 ± 0.9 | 0.6 ± 0.1 | ||
| Group P.L | 0.9 ± 0.03 | 1.8 ± 0.5 | 1 ± 0.03 | 0.8 ± 0.05 | 96 ± 62 | 186 ± 82 | 204± 50 | 0.82 ± 0.7 | 4.9 ± 0.5 | 0.6 ± 0.05 | ||
| Group DMBA | 1.5 ± 0.2a | 2.2 ± 0.9a | 1.5 ± 0.2a | 1.2 ± 0.09a | 130 ± 88 a | 230 ± 51 a | 249 ± 65 a | 1.5 ± 0.7 a | 6.2 ± 0.5a | 0.9 ± 0.09a | ||
| Group DMBA+ P.L | 1.2 ± 0.1b | 2 ± 0.8b | 1.2 ± 0.1b | 1 ± 0.1b | 158 ± 73 b | 213 ± 62b | 223 ± 51b | 1.1 ± 0.3 b | 5.9 ± 0.3b | 0.7 ± 0.1b | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




